The Effects of Perchlorates on the Permafrost Methanogens: Implication for Autotrophic Life on Mars
Abstract
:1. Introduction
2. Experimental Section
2.1. Archaeal Strains
| Methanogens | Substrates for Methanogenesis | Source of Isolation | References |
|---|---|---|---|
| Methanobacterium bryantii M.o.H.T | H2 + CO2 | Syntrophic association | [54] |
| Methanosarcina mazei S-6T | Acetate, methanol, methylamine, trimethylamine, H2 + CO2 | Anaerobic sewage digester | [55] |
| Methanobacterium arcticum M2T | H2+CO2, formate | Holocene permafrost | [22] |
| Methanobacterium veterum MK4T | H2 + CO2, H2 + methanol, H2 + methylamine | Plieocene permafrost | [21] |
| Methanosarcina sp. JL01 | Acetate, methanol, methylamine, trimethylamine | Holocene permafrost | [20] |
2.2. Media and Cultivation
2.3. Determination of Inhibitory Concentrations
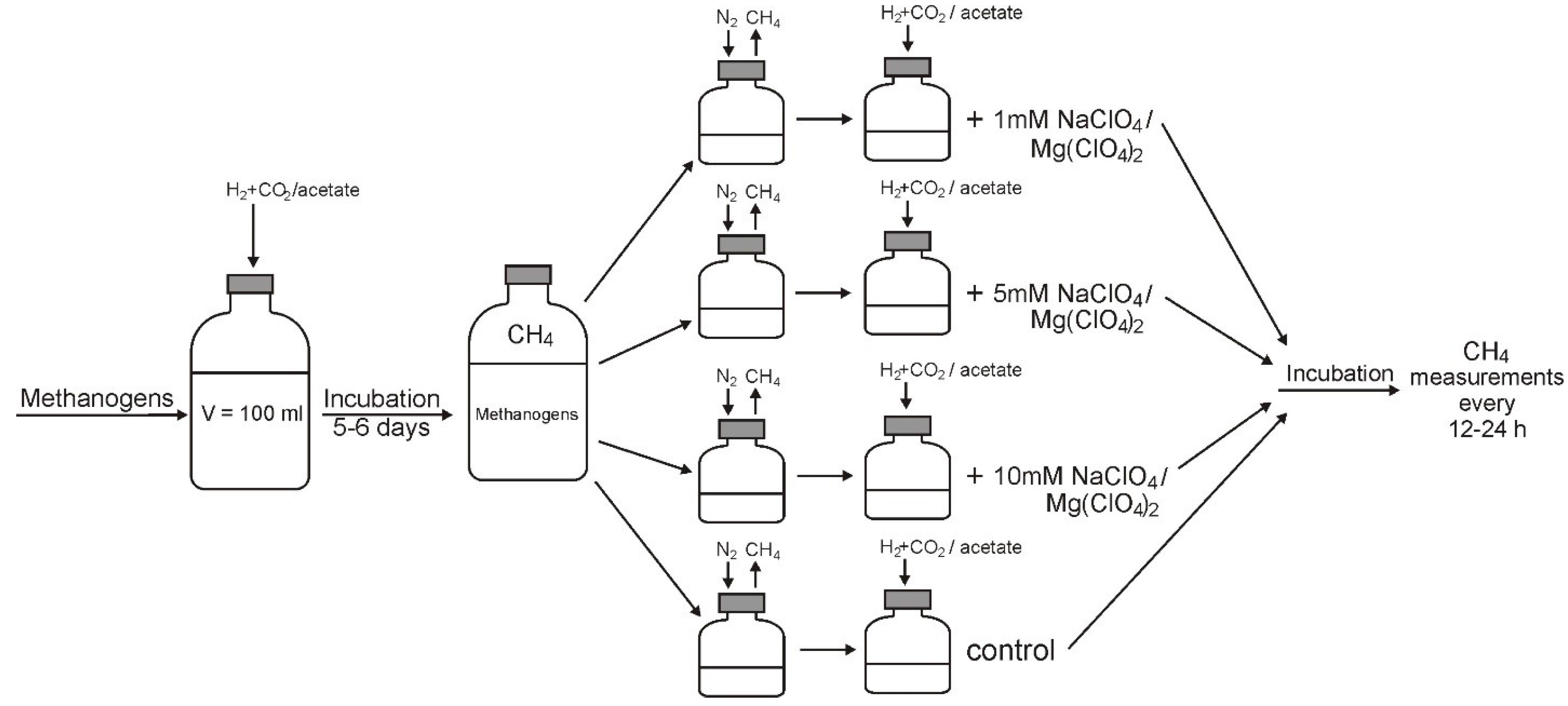
2.4. Microscopic Examination
2.5. Analytical Procedures
3. Results and Discussion
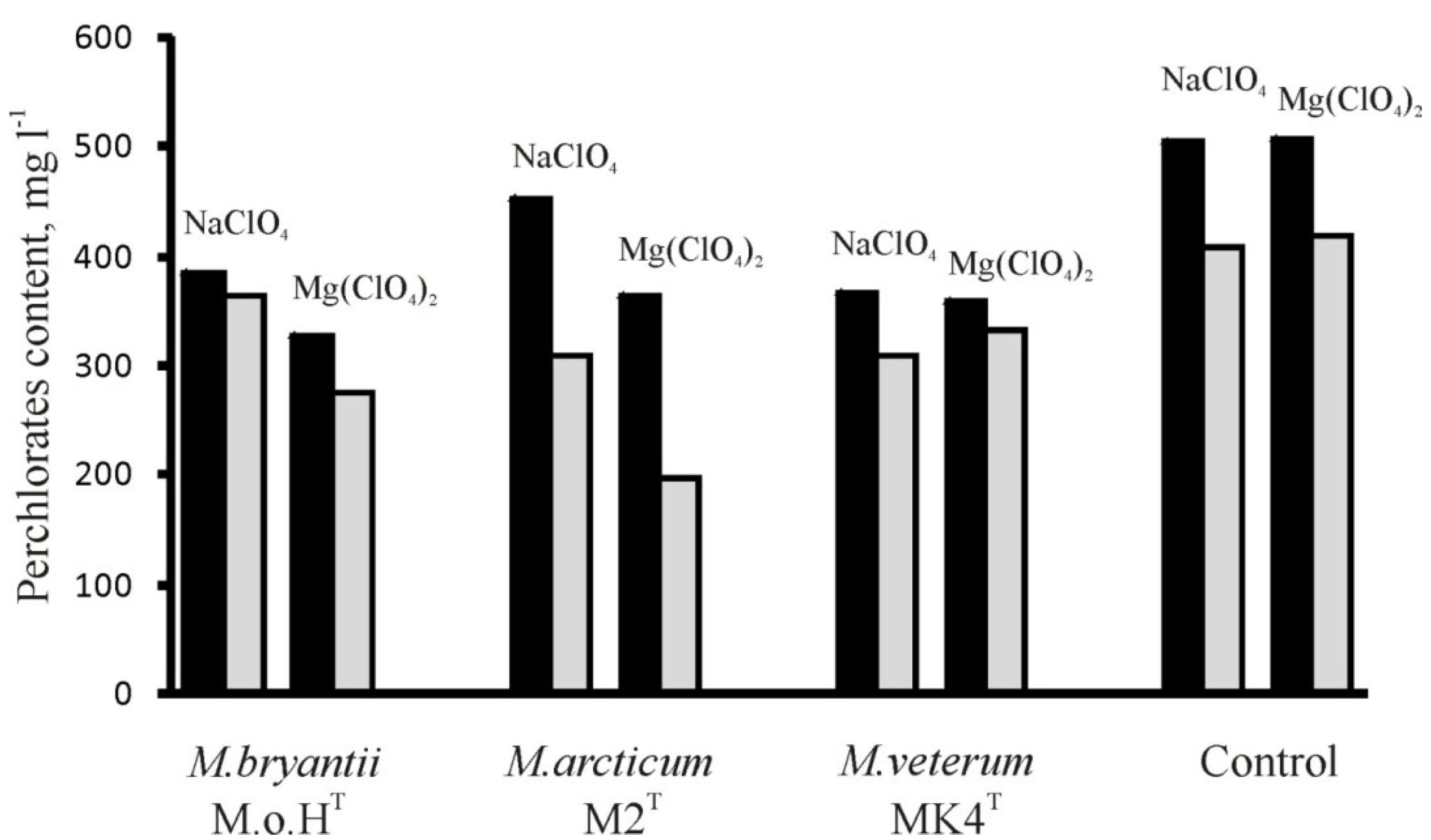
3.1. Determination of Perchlorates Inhibitory Concentrations
| Strains | Mg(ClO4)2, mM | NaClO4, mM | Mg(ClO4)2 + NaClO4, mM | ||||||
|---|---|---|---|---|---|---|---|---|---|
| IC20 | IC50 | IC80 | IC20 | IC50 | IC80 | IC20 | IC50 | IC80 | |
| M. bryantii M.o.HT | 3.5 | 6.2 | 9.0 | 2.8 | 6.0 | 9.8 | 2.2 | 4.0 | 8.1 |
| M. arcticum M2T | 9.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 |
| M. veterum MK4T | 2.6 | 6.6 | >10.0 | 4.1 | 8.4 | >10.0 | 2.5 | 5.6 | 9.0 |
| M. mazei S-6T | 5.0 | 9.2 | >10.0 | 7.8 | >10.0 | >10.0 | 4.8 | >10.0 | >10.0 |
| Methanosarcina sp. JL01 | 2.1 | 5.2 | >10.0 | 3.9 | 9.7 | >10.0 | 1.8 | 4.8 | >10.0 |
3.2. Methane Generation in Perchlorate-Supplemented Medium
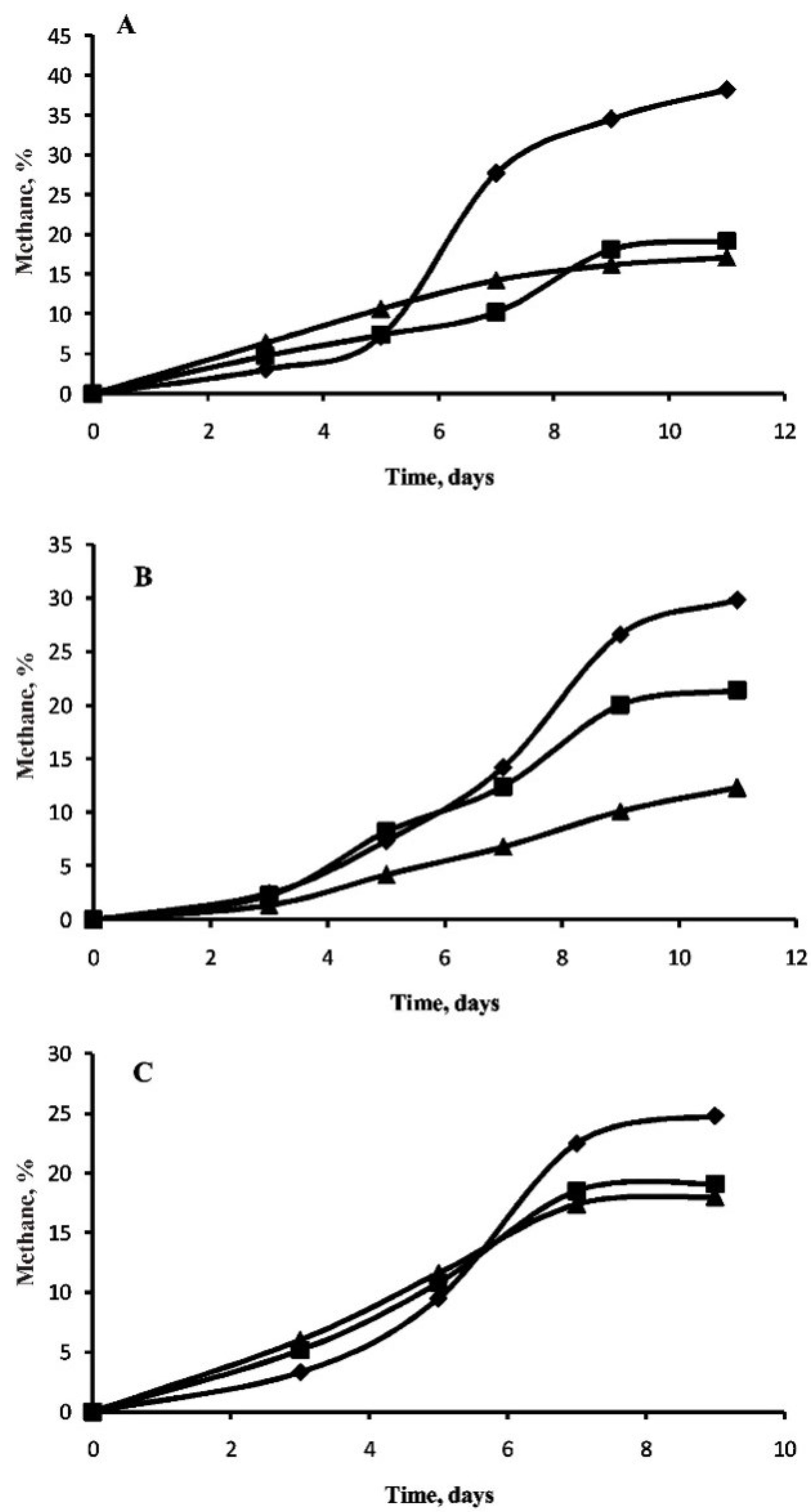
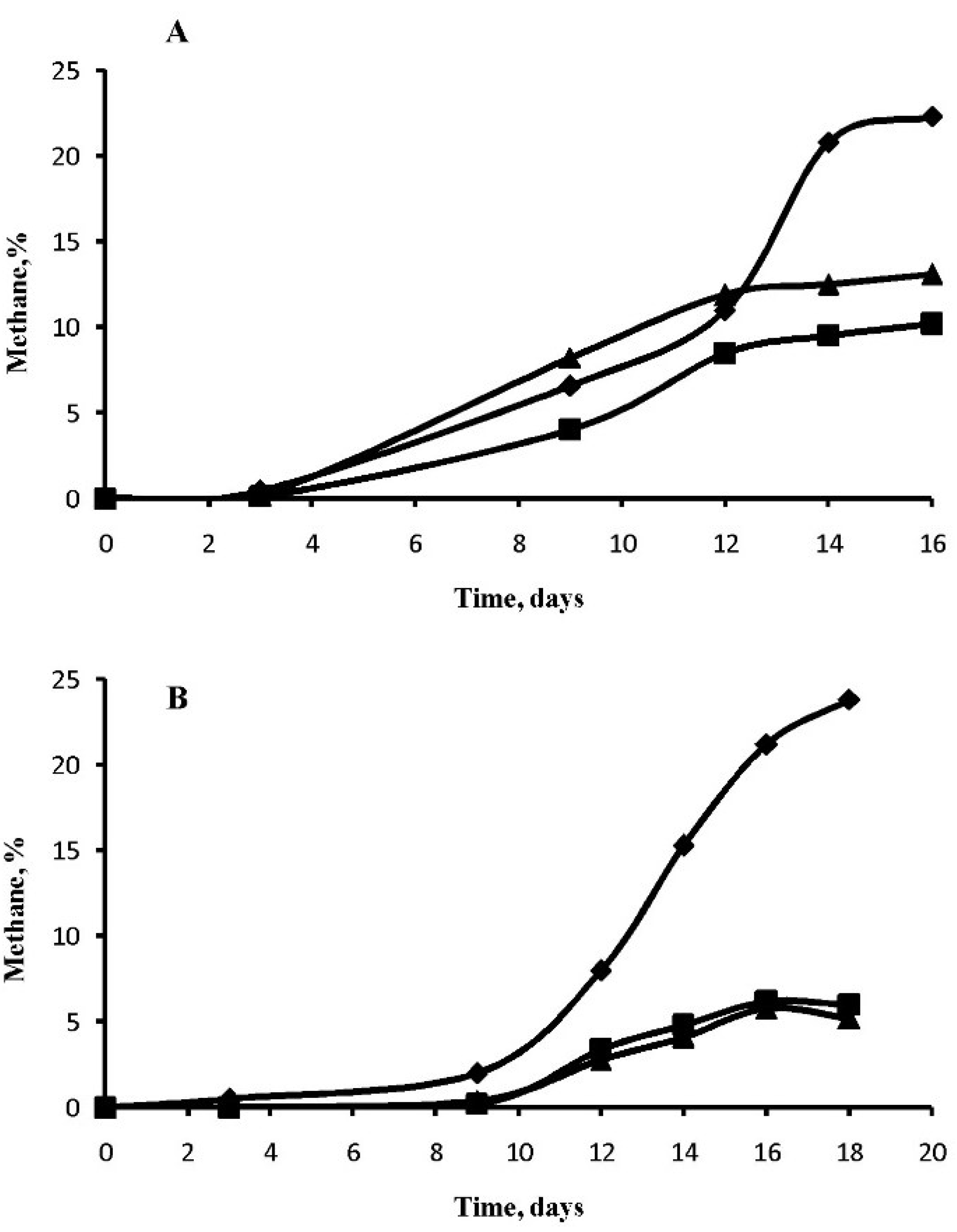
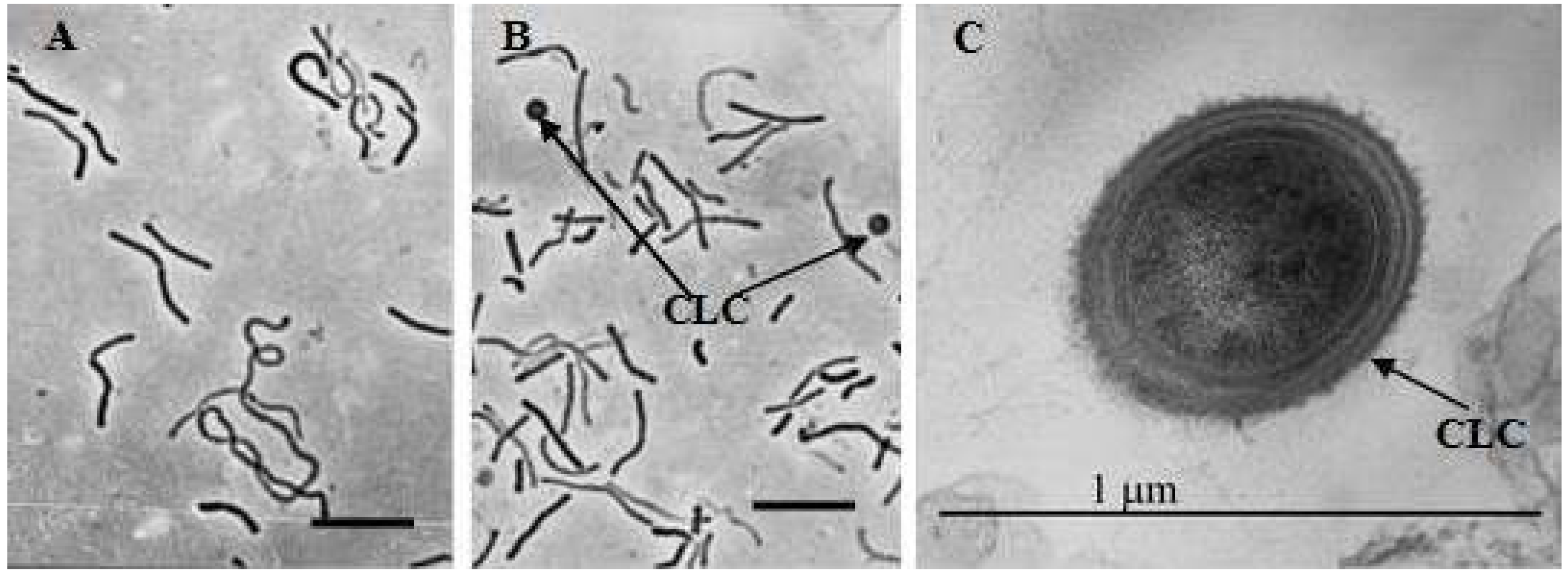
3.3. Effect of Perchlorate on Morphology of M. arcticum M2T Cells
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cameron, R.E.; Morelli, F.A. Viable microorganisms from ancient Ross Island and Taylor Valley drill core. Antarct. J. U. S. 1974, 9, 113–116. [Google Scholar]
- Gilichinsky, D. Permafrost as a microbial habitat. In Encyclopedia of Environmental Microbiology; Willey-Interscience: New York, NY, USA, 2002; pp. 932–956. [Google Scholar]
- Janssen, J.K.; Tas, N. The microbial ecology of permafrost. Nat. Rev. Microbiol. 2014, 12, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Gilichinsky, D.A.; Wilson, G.S.; Friedmann, E.I.; McKay, C.P.; Sletten, R.S.; Rivkina, E.M.; Vishnivetskaya, T.A.; Erokhina, L.G.; Ivanushkina, N.E.; Kochkina, G.A.; et al. Microbial populations in Antarctic permafrost: Biodiversity, state, age, and implication for astrobiology. Astrobiology 2007, 7, 275–311. [Google Scholar] [CrossRef] [PubMed]
- Steven, B.; Leveille, R.; Pollard, W.H.; Whyte, L.G. Microbial ecology and biodiversity in permafrost. Extremophiles 2006, 10, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Gilichinsky, D.A. Permafrost model of extraterrestrial habitat. In Astrobiology, 2nd ed.; Horneck, G., Baumstark-Khan, C., Eds.; Springer: Berlin, Germany, 2002; pp. 125–142. [Google Scholar]
- Zhang, G.; Ma, X.; Niu, F.; Dong, M.; Feng, H.; An, L.; Cheng, G. Diversity and distribution of alkaliphilic psychrotolerant bacteria in the Qinghai-Tibet Plateau permafrost region. Extremophiles 2007, 11, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Reid, I.; Sparks, W.; Lubow, S.; McGrath, M.; Livio, M.; Valenti, J.; Sowers, K.; Shukla, H.; MacAuley, S.; Miller, T. Terrestrial models for extraterrestrial life: Methanogens and halophiles at Martian temperatures. Int. J. Astrobiol. 2006, 5, 89–97. [Google Scholar] [CrossRef]
- Demidov, N.E.; Gilichinsky, D.A. Terrestrial permafrost models and analogues of Martian habitats and inhabitans. In Permafrost Soil, 6th ed.; Margesin, R., Ed.; Springer Berlin Heidelberg: Berlin, Germany, 2009. [Google Scholar]
- Rummel, J.D.; Beaty, D.W.; Jones, M.A.; Bakermans, C.; Barlow, N.G.; Boston, P.J.; Chevrier, V.F.; Clark, B.C.; de Vera, J.P.; Gough, R.V.; et al. A new analysis of Mars “Special Regions”: Findings of the second MEPAG Special Regions Science Analysis Group (SR-SAG2). Astrobiology 2014, 14, 887–968. [Google Scholar] [CrossRef] [PubMed]
- Boynton, W.V.; Feldman, W.C.; Mitrofanov, I.; Evans, L.G.; Reedy, R.C.; Squyres, S.W.; Starr, R.; Trombka, J.I.; D’Uston, C.; Arnold, J.R.; et al. Distribution of hydrogen in the near surface of Mars: Evidence for subsurface ice deposits. Science 2002, 297, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Demidov, N.; Boynton, W.; Gilichinsky, D.; Zuber, M.; Kozyrev, A.; Litvak, M.; Mitrofanov, I.; Sanin, A.; Saunders, R.; Smith, D. Water distribution in Martian permafrost regions from joint analysis of HEND (Mars Odyssey) and MOLA (Mars Global Surveyor) data. Astron. Lett. 2008, 34, 713–723. [Google Scholar] [CrossRef]
- Cull, S.; Arvidson, R.E.; Catalano, J.G.; Ming, D.W.; Morris, R.V.; Mellon, M.T.; Lemmon, M. Concentrated perchlorate at the Mars Phoenix landing site: Evidence for thin film liquid water on Mars. Geophys. Res. Lett. 2010, 37, L22203. [Google Scholar] [CrossRef]
- Fischer, E.; Martínez, G.M.; Elliott, H.M.; Rennó, N.O. Experimental evidence for the formation of liquid saline water on Mars. Geophys. Res. Lett. 2014, 41, 4456–4462. [Google Scholar] [CrossRef] [PubMed]
- Mumma, M.; Novak, R.; DiSanti, M.; Bonev, B. A sensitive search for methane on Mars. Bull. Am. Astron. Soc. 2003, 35, 937. [Google Scholar]
- Formisano, V.; Atreya, S.; Encrenaz, T.R.; Ignatiev, N.; Giuranna, M. Detection of methane in the atmosphere of Mars. Science 2004, 306, 1758–1761. [Google Scholar] [CrossRef] [PubMed]
- Krasnopolsky, V.A.; Maillard, J.P.; Owen, T.C. Detection of methane in the Martian atmosphere: Evidence for life? Icarus 2004, 172, 537–547. [Google Scholar] [CrossRef]
- Mumma, M.J.; Villanueva, G.L.; Novak, R.E.; Hewagama, T.; Bonev, B.P.; Disanti, M.A.; Mandell, A.M.; Smith, M.D. Strong release of methane on Mars in northern summer 2003. Science 2009, 323, 1041–1045. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, D.R.; Lovelace, J.E. Life Detection by Atmospheric Analysis. Icarus 1967, 7, 149–159. [Google Scholar] [CrossRef]
- Rivkina, E.; Shcherbakova, V.; Laurinavichius, K.; Petrovskaya, L.; Krivushin, K.; Kraev, G.; Pecheritsina, S.; Gilichinsky, D. Biogeochemistry of methane and methanogenic archaea in permafrost. FEMS Microbiol. Ecol. 2007, 61, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Krivushin, K.V.; Shcherbakova, V.A.; Petrovskaya, L.E.; Rivkina, E.M. Methanobacterium veterum sp. nov., from ancient Siberian permafrost. IJSEM 2010, 60, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Shcherbakova, V.; Rivkina, E.; Pecheritsyna, S.; Laurinavichius, K.; Suzina, N.; Gilichinsky, D. Methanobacterium arcticum sp. nov., a methanogenic archaeon from Holocene Arctic permafrost. IJSEM 2011, 61, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Costa, K.C.; Leigh, J.A. Metabolic versatility in methanogens. Curr. Opin. Biotechnol. 2014, 29, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Ferry, J.G. The chemical biology of methanogenesis. Planet. Space Sci. 2010, 58, 1775–1783. [Google Scholar] [CrossRef]
- Wagner, D.; Spieck, E.; Bock, E.; Pfeiffer, E.-M. Microbial Life in Terrestrial Permafrost: Methanogenesis and Nitrification in Gelisols as Potentials for Exobiological Process. In Astrobiology, 2nd ed.; Horneck, G., Baumstark-Khan, C., Eds.; Springer Berlin Heidelberg: Berlin, Germany, 2002; pp. 143–159. [Google Scholar]
- Levin, G.V.; Straat, P.A. Methane and life on Mars. In Proceedings of the SPIE Optical Engineering + Applications, International Society for Optics and Photonics, San Diego, CA, USA, 2 August 2009.
- Kral, T.A.; Brink, K.M.; Miller, S.L.; McKay, C.P. Hydrogen consumption by methanogens on the early Earth. Orig. Life Evol. B 1998, 28, 311–319. [Google Scholar] [CrossRef]
- Kral, T.A.; Bekkum, C.R.; McKay, C.P. Growth of methanogens on a Mars soil simulant. Orig. Life Evol. B 2004, 34, 615–626. [Google Scholar] [CrossRef]
- Ormond, D.R.; Kral, T.A. Washing methanogenic cells with the liquid fraction from a Mars soil simulant and water mixture. J. Microbiol. Methods 2006, 67, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Gavin, P.; Kral, T.; Goodhart, T.; Chevrier, V. Methanogen Growth in Perchlorate-Supplemented Media and Implications for Life in Phoenix-Type Soils. LPI Contrib. 2009, 1502, 19. [Google Scholar]
- Chastain, B.K.; Kral, T.A. Zero-valent iron on Mars: An alternative energy source for methanogens. Icarus 2010, 208, 198–201. [Google Scholar] [CrossRef]
- Sinha, N.; Kral, T. Growth of Methanogens on Different Mars Regolith Analogues and Stable Carbon Isotope Fractionation during Methanogenesis. In Proceedings of the Lunar and Planetary Science Conference, Woodlands, TX, USA, 16–20 March 2015.
- Kendrick, M.G.; Kral, T.A. Survival of methanogens during desiccation: Implications for life on Mars. Astrobiology 2006, 6, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Altheide, T.; Kral, T. Low-pressure desiccation effects on methane production by methanogens. In Proceedings of the Lunar and Planetary Science Conference, League City, TX, USA, 10–14 March 2008.
- Kral, T.A.; Altheide, T.S.; Lueders, A.E.; Schuerger, A.C. Low pressure and desiccation effects on methanogens: Implications for life on Mars. Planet. Space Sci. 2011, 59, 264–270. [Google Scholar] [CrossRef]
- Schirmack, J.; Alawi, M.; Wagner, D. Influence of Martian regolith analogs on the activity and growth of methanogenic archaea, with special regard to long-term desiccation. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Morozova, D.; Möhlmann, D.; Wagner, D. Survival of methanogenic archaea from Siberian permafrost under simulated Martian thermal conditions. Orig. Life Evol. Biosph. 2007, 37, 189–200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sinha, N.; Kral, T. Methanogen sensitivity to ultraviolet radiation: Implications for life on Mars. In Proceedings of the 76th Annual Meeting of the Meteoritical Society, Edmonton, AB, Canada, 29 July–7 August 2013.
- Hecht, M.; Kounaves, S.; Quinn, R.; West, S.; Young, S.; Ming, D.; Catling, D.; Clark, B.; Boynton, W.; Hoffman, J. Detection of perchlorate and the soluble chemistry of Martian soil at the Phoenix lander site. Science 2009, 325, 64–67. [Google Scholar] [PubMed]
- Zhang, Y.; Cremer, P.S. Interactions between macromolecules and ions: The Hofmeister series. Curr. Opin. Chem. Biol. 2006, 10, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Cray, J.A.; Russell, J.T.; Timson, D.J.; Singhal, R.S.; Hallsworth, J.E. A universal measure of chaotropicity and kosmotropicity. Environ. Microbiol. 2013, 15, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Cray, J.A.; Stevenson, A.; Ball, P.; Bankar, S.B.; Eleutherio, E.C.; Ezeji, T.C.; Singhal, R.S.; Thevelein, J.M.; Timson, D.J. Hallsworth, J.E. Chaotropicity: A key factor in product tolerance of biofuel-producing microorganisms. Curr. Opin. Biotechnol. 2015, 33, 228–259. [Google Scholar] [CrossRef] [PubMed]
- Rangel, D.E.; Alder-Rangel, A.; Dadachova, E.; Finlay, R.D.; Kupiec, M.; Dijksterhuis, J.; Gilberto, U.L.; Braga, G.U.L.; Luis, M.; Corrochano, L.M.; et al. Fungal stress biology: A preface to the Fungal Stress Responses special edition. Curr. Genet. 2015, 61, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Hallsworth, J.E.; Heim, S.; Timmis, K.N. Chaotropic solutes cause water stress in Pseudomonas putida. Environ. Microbiol. 2003, 5, 1270–1280. [Google Scholar] [CrossRef] [PubMed]
- Duda, V.I.; Danilevich, V.N.; Suzina, N.E.; Shorokhova, A.P.; Dmitriev, V.V.; Mokhova, O.N. Akimov, V.N. Changes in the fine structure of microbial cells induced by chaotropic salts. Mikrobiologiya 2004, 73, 341–349. [Google Scholar]
- Hallsworth, J.E.; Yakimov, M.M.; Golyshin, P.N.; Gillion, J.L.; D’Auria, G.; de Lima Alves, F.; la Cono, V.; McKew, B.A.; Hayes, S.L.; Harris, G.; et al. Limits of life in MgCl2-containing environments: Chaotropicity defines the window. Environ. Microbiol. 2007, 9, 801–813. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.P.; Hallsworth, J.E. Limits of life in hostile environments: No barriers to biosphere function. Environ. Microbiol. 2009, 11, 3292–3308. [Google Scholar] [CrossRef] [PubMed]
- Bhaganna, P.; Volkers, R.J.; Bell, A.N.; Kluge, K.; Timson, D.J.; McGrath, J.W.; Ruijssenaars, H.J.; Hallsworth, J.E. Hydrophobic substances induce water stress in microbial cells. Microbiol. Biotechnol. 2010, 3, 701–716. [Google Scholar] [CrossRef] [PubMed]
- Coates, J.D.; Michaelidou, U.; Bruce, R.A.; Connor, S.M.; Crespi, J.N.; Achenbach, L.A. Ubiquity and diversity of dissimilatory (per) chlorate-reducing bacteria. Appl. Environ. Microbiol. 1999, 65, 5234–5241. [Google Scholar] [PubMed]
- Coates, J.D.; Achenbach, L.A. Microbial perchlorate reduction: Rocket-fuelled metabolism. Nat. Rev. Microbiol. 2004, 2, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Liebensteiner, M.G.; Pinkse, M.W.; Schaap, P.J.; Stams, A.J.; Lomans, B.P. Archaeal (per) chlorate reduction at high temperature: An interplay of biotic and abiotic reactions. Science 2013, 340, 85–87. [Google Scholar] [CrossRef] [PubMed]
- Oren, A.; Bardavid, R.E.; Mana, L. Perchlorate and halophilic prokaryotes: Implications for possible halophilic life on Mars. Extremophiles 2014, 18, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Espinosa, R.M.; Richardson, D.J.; Bonete, M.J. Characterization of chlorate reduction in the haloarchaeon Haloferax mediterranei. BBA-Gen. Subj. 2015, 1850, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Balch, W.E.; Fox, G.; Magrum, L.; Woese, C.; Wolfe, R. Methanogens: Reevaluation of a unique biological group. Microbiol. Rev. 1979, 43, 260. [Google Scholar] [PubMed]
- Mah, R.A.; Kuhn, D.A. Transfer of the type species of the genus Methanococcus to the genus Methanosarcina, naming it Methanosarcina mazei (Barker 1936) comb. nov. et emend and conservation of the genus Methanococcus (approved lists 1980) with Methanococcus vannielii (approved lists 1980) as the type species request for an opinion. IJSB 1984, 34, 263–265. [Google Scholar]
- Hungate, R. A roll tube method for cultivation of strict anaerobes. Methods Microbiol. 1969, 3B, 117–132. [Google Scholar]
- Reynolds, E.S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 1963, 17, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Shcherbakova, V.A.; Obraztsova, A.Y.; Laurinavichius, K.S.; Kotel’nikova, S.V.; Akimenko, V.K.; Navoa, M.K.; Kruz, M. Physiological properties of thermophilic methanosarcina isolated from active sludge of anaerobic digesters. Microbiology 1991, 60, 466–471. [Google Scholar]
- Gilichinsky, D.; Rivkina, E.; Shcherbakova, V.; Laurinavichuis, K.; Tiedje, J. Supercooled Water Brines within Permafrost-An Unknown Ecological Niche for Microorganisms: A Model for Astrobiology. Astrobiology 2003, 3, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Deming, J.W. Psychrophiles and polar regions. Curr. Opin. Microbiol. 2002, 5, 301–309. [Google Scholar] [CrossRef]
- Marchant, D.R.; Head, J.W. Antarctic dry valleys: Microclimate zonation, variable geomorphic processes, and implications for assessing climate change on Mars. Icarus 2007, 192, 187–222. [Google Scholar] [CrossRef]
- Yakimov, M.M.; la Cono, V.; Spada, G.L.; Bortoluzzi, G.; Messina, E.; Smedile, F.; Arcadi, E.; Borghini, M.; Ferrer, M.; Schmitt-Kopplin, P.; et al. Microbial community of the deep-sea brine Lake Kryos seawater–brine interface is active below the chaotropicity limit of life as revealed by recovery of mRNA. Environ. Microbiol. 2015, 17, 364–382. [Google Scholar] [CrossRef] [PubMed]
- Chin, J.P.; Megaw, J.; Magill, C.L.; Nowotarski, K.; Williams, J.P.; Bhaganna, P.; Linton, M.; Patterson, M.F.; Underwood, G.J.C.; Mswaka, A.; et al. Solutes determine the temperature windows for microbial survival and growth. Proc. Natl. Acad. Sci. USA 2010, 107, 7835–7840. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, A.; Cray, J.A.; Williams, J.P.; Santos, R.; Sahay, R.; Neuenkirchen, N.; McClure, C.D.; Grant, I.R.; Houghton, J.D.R.; Quinn, J.P.; et al. Is there a common water-activity limit for the three domains of life? ISME 2015, 9, 1333–1351. [Google Scholar] [CrossRef] [PubMed]
- Shcherbakova, V.; Rivkina, E.; Laurinavichuis, K.; Pecheritsina, S.; Gilchinsky, D. Physiological characteristics of bacteria isolated from water brines within permafrost. Int. J. Astrobiol. 2004, 3, 37–43. [Google Scholar] [CrossRef]
- Zehnder, A.; Brock, T. Methane formation and methane oxidation by methanogenic bacteria. J. Bacteriol. 1979, 137, 420–432. [Google Scholar] [PubMed]
- Boetius, A.; Ravenschlag, K.; Schubert, C.J.; Rickert, D.; Widdel, F.; Gieseke, A.; Amann, R.; Jёrgensen, B.B.; Witte, U.; Pfannkuche, O. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 2000, 407, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Ettwig, K.F.; Shima, S.; van de Pas-Schoonen, V.; Katinka, T.; Kahnt, J.R.; Medema, M.H.; Op Den Camp, H.J.; Jetten, M.S.; Strous, M. Denitrifying bacteria anaerobically oxidize methane in the absence of Archaea. Environ. Microbiol. 2008, 10, 3164–3173. [Google Scholar] [CrossRef] [PubMed]
- Beal, E.J.; House, C.H.; Orphan, V.J. Manganese-and iron-dependent marine methane oxidation. Science 2009, 325, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Karaevskaya, E.; Demchenko, L.; Demidov, N.; Rivkina, E.; Bulat, S.; Gilichinsky, D. Archaeal diversity in permafrost deposits of Bunger Hills Oasis and King George Island (Antarctica) according to the 16S rRNA gene sequencing. Microbiology 2014, 83, 398–406. [Google Scholar] [CrossRef]
- Krivushin, K.; Kondrashov, F.; Shmakova, L.; Tutukina, M.; Petrovskaya, L.; Rivkina, E. Two metagenomes from Late Pleistocene northeast Siberian permafrost. Genome Announc. 2015, 3, e01380-14. [Google Scholar] [CrossRef] [PubMed]
- Rivkina, E.; Laurinavichius, K.; McGrath, J.; Tiedje, J.; Shcherbakova, V.; Gilichinsky, D. Microbial life in permafrost. Adv. Space Res. 2004, 33, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Rivkina, E.; Laurinavichus, K.; Gilichinsky, D.; Shcherbakova, V. Methane generation in permafrost sediments. Dokl. Biol. Sci. 2002, 383, 179–181. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shcherbakova, V.; Oshurkova, V.; Yoshimura, Y. The Effects of Perchlorates on the Permafrost Methanogens: Implication for Autotrophic Life on Mars. Microorganisms 2015, 3, 518-534. https://doi.org/10.3390/microorganisms3030518
Shcherbakova V, Oshurkova V, Yoshimura Y. The Effects of Perchlorates on the Permafrost Methanogens: Implication for Autotrophic Life on Mars. Microorganisms. 2015; 3(3):518-534. https://doi.org/10.3390/microorganisms3030518
Chicago/Turabian StyleShcherbakova, Viktoria, Viktoria Oshurkova, and Yoshitaka Yoshimura. 2015. "The Effects of Perchlorates on the Permafrost Methanogens: Implication for Autotrophic Life on Mars" Microorganisms 3, no. 3: 518-534. https://doi.org/10.3390/microorganisms3030518
APA StyleShcherbakova, V., Oshurkova, V., & Yoshimura, Y. (2015). The Effects of Perchlorates on the Permafrost Methanogens: Implication for Autotrophic Life on Mars. Microorganisms, 3(3), 518-534. https://doi.org/10.3390/microorganisms3030518





