Abstract
A post-COVID surge of Bordetella pertussis was observed globally. China has reported a high level of macrolide-resistant Bordetella pertussis (MRBP) in recent years; however, the epidemiology of MRBP in Hong Kong remains unknown. We retrieved archived B. pertussis isolates from respiratory samples collected at five regional public hospitals in Hong Kong between 2015 and 2024 and tested their minimum inhibitory concentration (MIC) for macrolides and other non-macrolide antibiotics using the Etest method. All isolates were also subjected to whole genome sequencing for genotypic resistance, Multi-locus Antigen Sequence Typing (MLST) and Multi-locus Variable Number of Tandem Repeat Analysis (MLVA) typing. Twenty-nine isolates of B. pertussis were included in the study. All isolates demonstrating phenotypic macrolide resistance harbored the A2047G mutation while showing low MIC to trimethoprim-sulfamethoxazole, doxycycline, levofloxacin, piperacillin-tazobactam and meropenem. In 2023 and 2024, 100% were MRBP and all belonged to the MT28 clone with the ptxP3 antigenic type. The MRBP isolates in Hong Kong were phylogenetically related to those from mainland China during the same period. There was no obvious correlation between macrolide resistance and clinical presentation, laboratory findings, management and outcome. Phylogenetic analysis suggests that MRBP isolates in Hong Kong and mainland China are closely related.
1. Introduction
Pertussis, also known as whooping cough, is a highly contagious bacterial infection that was a leading cause of childhood illness and death in the pre-vaccine era. It classically presents in young infants with the characteristic paroxysmal cough, which may be lead to feeding difficulties, respiratory failure and even death in severe cases [1]. In the 1940s, pertussis caused three times more infant deaths than measles, mumps, chicken pox, rubella, scarlet fever, diphtheria, poliomyelitis and bacterial meningitis combined [2]. Global vaccination campaigns started in the 1950s with whole-cell vaccines, and later switched to acellular vaccines in a routine schedule in 1990s [3]. Despite these efforts, it was estimated that pertussis accounted for around 160,000 deaths worldwide in the year 2014 alone [4].
The pertussis-containing vaccine was first introduced in Hong Kong in 1957 [5]. Under the current Hong Kong Childhood Immunization Program, children are given three primary doses of pertussis-containing vaccine at two, four and six months and three booster doses from 18 months onwards. Acellular pertussis toxoid-containing vaccines haven been used since 2006 [6]. The immunization coverage rate has been over 95% for many years. In response to rising infant pertussis cases in Hong Kong, one dose of pertussis-containing vaccine in the second or third trimester is now offered to pregnant women as part of routine antenatal care since 2020, in the form of dTap (diphtheria (reduced dose), tetanus and acellular pertussis (reduced dose)) [5]. In mainland China, pertussis-toxoid acellular vaccine is administered at three, four, five and 18 months [7]. Vaccine coverage is reported at 99% [8]. The maternal pertussis vaccination is currently not integrated into healthcare practices in mainland China. A study shows that only 36% of expectant mothers intended to receive the pertussis-containing vaccine in a province in mainland China [9].
Despite the implementation of the childhood vaccination program and maternal vaccination, a recent upsurge of pertussis has been reported globally after the relaxation of non-pharmacological interventions for coronavirus disease-2019 (COVID-19) [10,11,12]. Data from the World Health Organization (WHO) show that the global incidence of pertussis increased from 3.6 million in 2021 to 22.8 million in 2023, nearing the pre-pandemic level [13]. A similar increase has been observed in Hong Kong [1]. The rise in incidence of whooping cough is most notable in vaccinated individuals and older children [14,15]. It has been proposed that vaccine escape of B. pertussis is due to the antigenic shift from the ptxP1 to ptxP3 subtype and the acquisition of macrolide resistance [16].
Macrolide resistance in B. pertussis is most commonly caused by an A-to-G mutation at position 2047 of the Sanger Center sequence of the B. pertussis 23s rRNA gene [17]. The mutation affects the macrolide binding site in the 23S rRNA component of the 50S ribosomal subunit, preventing macrolide inhibition on peptide elongation. There are three copies of the 23S rRNA gene in B. pertussis, but it was expected that mutation in at least two copies would be sufficient to confer resistance [17]. Macrolide-resistant Bordetella pertussis (MRBP) was first reported in the United States in 1994 [18]. The prevalence of MRBP in North America, Europe, Southeast Asia and Australia remained low at 0–13% from 1967 to 2020 [18], and has only sporadically been reported since, including one isolate recently reported from France [19]. In contrast, since the first report of MRBP in Shandong in mainland China in 2011, its prevalence in the major cities of mainland China, including Beijing and Shanghai, has been observed to reach up to 90–100% after the COVID-19 pandemic [15,20,21]. Pertussis toxin promoter (ptxP) is one of the antigenic subtypes included in the Multi-locus Sequence Typing (MLST) scheme historically used to study shifts in B. pertussis populations. The variant ptxP3 is traditionally believed to enhance the production of toxins [22]. Previous studies showed that MRBP in China was mostly linked to the ptxP1 allele [21,23]. In 2007, mainland China introduced the acellular diphtheria–tetanus–pertussis (DTaP) vaccine which comprises strains with the ptxP1 antigen [24]. The ptxP3-MRBP antigen has emerged sporadically in mainland China since 2017 and was found to be prevalent in Shanghai in 2021. This means that the ptxP3 strain likely holds a competitive advantage over the ptxP1 strain under vaccine-driven selection pressure [25].
Hong Kong was experiencing a similar upsurge of pertussis in 2024 [26]. Although in close geographical proximity to mainland China, the prevalence and circulating antigenic genotype of MRBP in Hong Kong remained unknown. Azithromycin is currently the antibiotic recommended by local guidelines for pertussis treatment and post-exposure prophylaxis (PEP) [27], similar to the practice in the United States, United Kingdom and Australia where MRBP prevalence has remained low [28,29,30,31]. However, macrolides are ineffective against MRBP, permitting further transmission [28,30]. Trimethoprim-sulfamethoxazole is the alternative antibiotic choice, especially against MRBP. This retrospective study aims to investigate the epidemiology and genetic evolution of MRBP in Hong Kong.
2. Materials and Methods
2.1. Strain Recovery and Identification
All archived clinical B. pertussis isolates from respiratory samples collected during patient care at five regional public hospitals (Queen Elizabeth Hospital, United Christian Hospital, Tuen Mun Hospital, Kwong Wah Hospital and Pamela Youde Nethersole Eastern Hospital) in Hong Kong during 2015–2024 were retrospectively retrieved. Archived bacterial isolates stored at −70 °C in Microbank 2D (Pro-lab diagnostics; Richmond Hill, ON, Canada) were subcultured onto Regan–Lowe agar (Oxoid CM0119; Thermo Fisher, Basingstoke, UK) in an ambient moist chamber at 35 °C for up to 7 days. Bacterial speciation was reconfirmed by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS, IVD library version: DB-11758 MSP; Bruker, Bremen, Germany). Only one isolate will be retrieved for analysis for an individual pertussis case of close contact.
2.2. Antimicrobial Susceptibility Testing
The minimum inhibitory concentration (MIC) of three macrolides (erythromycin, azithromycin, clarithromycin), trimethoprim-sulfamethoxazole, doxycycline, levofloxacin, piperacillin-tazobactam and meropenem were determined after 72 h of incubation at 35 °C using MIC test strips (Liofilchem, Copenhagen, Denmark) and interpreted as previously described [32]. There is no standardized method for susceptibility testing of B. pertussis; however, the MICs of erythromycin against susceptible isolates were previously reported to range from 0.02 to 0.12 μg/mL [33].
2.3. Detection of A2047G Mutation
B. pertussis colonies were inoculated into 1.5 mL nuclease free water to create a suspension of 1.0 McFarland standard and heated to 100 °C. A total of 200 μL of the suspension was then added to 200 μL of MagNA Pure Bacterial Lysis Buffer (Roche, Basel, Switzerland) and MagNA Pure extraction was carried out according to the manufacturer’s instructions [34]. The extracted DNA was subjected to A2047G mutation detection by polymerase chain reaction-based sequencing of the 23S rRNA gene, as previously described [35].
2.4. Whole Genome Sequencing
Genomic DNA from B. pertussis isolates was extracted using DNeasy Blood & Tissue Kit (Qiagen, Hilden, Germany), followed by library preparation using Illumina DNA Prep (Illumina, San Diego, CA, USA). Sequencing of the pooled library was performed using a MiSeq sequencer (Illumina) with paired-end 300 bp reads, as previously described [36]. Briefly, the low-quality bases in the paired-end reads were filtered and trimmed using Trimmomatic v0.39 [37]. Quality-trimmed reads were then de-novo assembled using SPAdes v3.15.3 and Ragout v2.3 for the reference-assisted scaffolding [38,39]. Single nucleotide polymorphism (SNP) analysis was performed using Snippy v4.60 (https://github.com/tseemann/snippy (accessed on 13 August 2024)). B. pertussis strain Tohama I (Genbank accession no. NC_002929.2) was selected as the reference genome. A maximum likelihood phylogeny was constructed from the SNP data using IQ-TREE v2.1.1 with a K2P with ascertainment bias correction. The whole-genome sequencing project has been deposited in GenBank (accession numbers SAMN48174304-32).
2.5. Multi-Locus Sequence Typing (MLST) and Virulence Gene Analysis
The whole-genome sequencing (WGS) data was used to determine the multi-locus sequence type of each isolate. Allele numbers and sequence types (STs) were assigned by querying the Bordetella database in the BIGSdb-Pasteur platform (https://bigsdb.pasteur.fr/bordetella/ (accessed on 13 August 2024)). The presence and sequence variants of key virulence genes (ptxP, ptxA, ptxC, prn, fim2, fim3, tcfA, fhaB and bscI) were also determined from the WGS data using the Bordetella database in the BIGSdb-Pasteur platform.
2.6. Multi-Locus Variable Number of Tandem Repeat Analysis (MLVA) Typing
MLVA typing was performed according to a protocol previously described [40].
2.7. Retrieval of Clinical Data
Demographic data of the patients, including age, gender, isolation source and date, symptoms and signs of presentation, investigation results, clinical progress and outcome were retrieved by reviewing electronic medical records in the Hospital Authority Clinical Management System.
2.8. Consumption of Macrolides
Data on consumption of macrolides were obtained from the Department of Health in Hong Kong. The department collected the annual wholesale supply data from all registered drug wholesalers in Hong Kong. This served as a good proxy of antimicrobial consumption for the whole territory of 7.3 million people. Consumption of macrolides and other antimicrobials from 2015 to 2024 were analyzed and presented as Defined Daily Dose (DDD) and DID (DDD per 1000 Inhabitants per Day) as the units adopted by the World Health Organization (WHO). Version 2024 of the DDD values was adopted for the calculations.
2.9. Statistical Analysis
Statistical analysis was performed using SPSS version 24 (SPSS, Chicago, IL, USA). Statistical significance for the clinical severity of diseases due to MRBP and macrolide-sensitive B. pertussis (MSBP), ptxP1 and ptxP3 subtype, was determined by the Mann–Whitney U test and Fisher’s Exact Test. The consumption of macrolides over the surveillance period was tested for any statistical change using linear regression.
3. Results
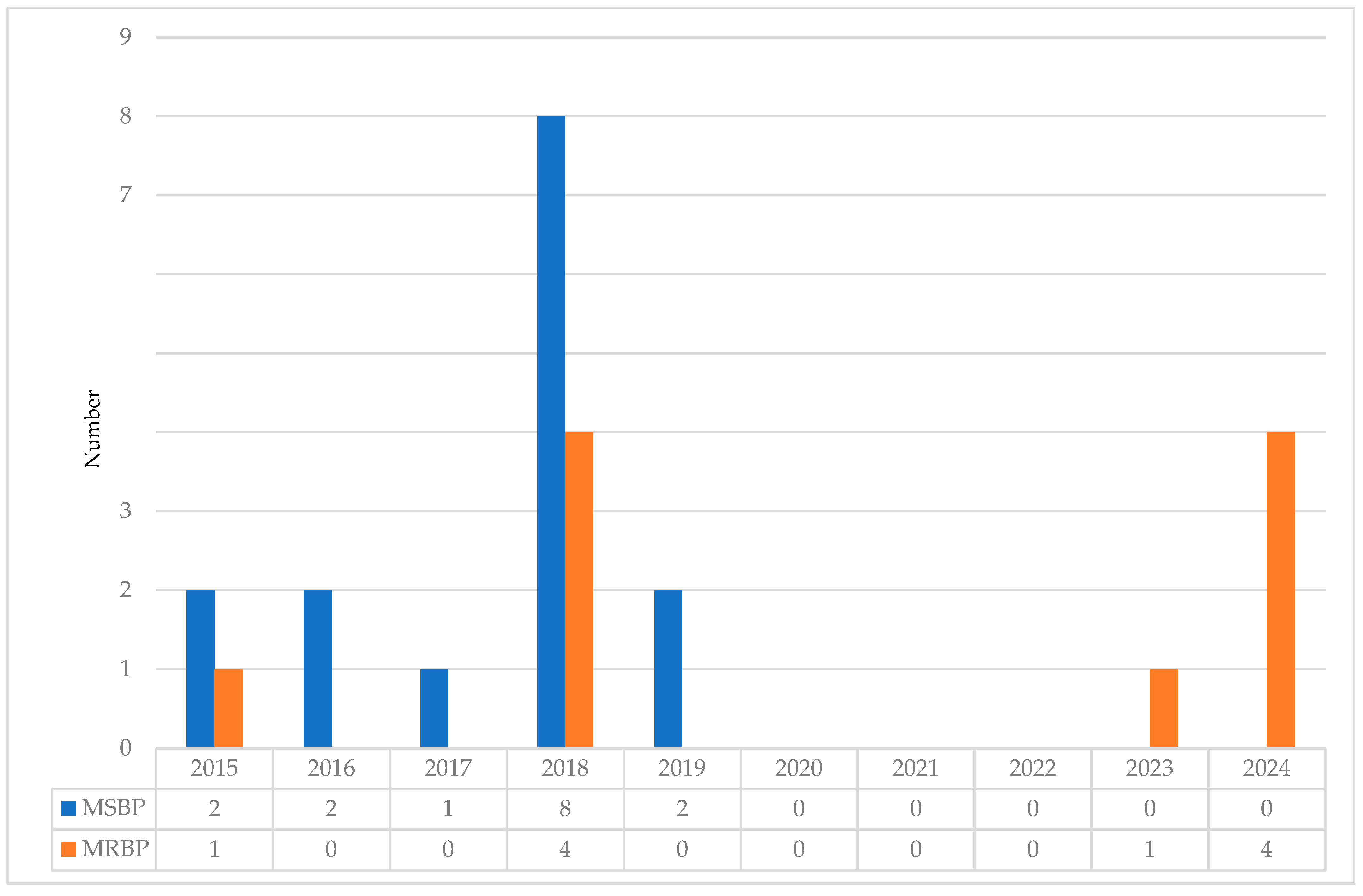
Twenty-nine clinical B. pertussis isolates were recovered between January 2015 and May 2024 and included in our study. Macrolide-resistance was identified in 34.5% (10/29). The first MRBP was detected in 2015, and its prevalence reached 33% (4/12) in 2018. In 2023 and 2024, after the end of COVID-19 pandemic was declared by the World Health Organization [41], MRBP became dominant and now accounts for all of the tested isolates (1/1 in 2023, 4/4 in 2024) (Figure 1).
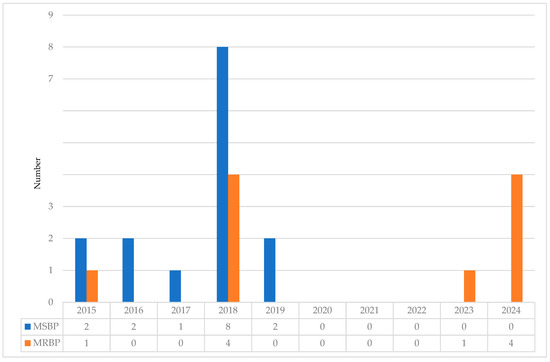
Figure 1.
Macrolide resistance of Bordetella pertussis in Hong Kong, 2015–2024. MSBP, macrolide-sensitive Bordetella pertussis; MRBP, macrolide-resistant Bordetella pertussis.
Twenty-five (25/29, 86.2%) of the cultured B. pertussis isolates were isolated from symptomatic cases, and the remaining four (13.8%) were isolated from close contacts who retrospectively reported mild upper respiratory tract infection symptoms (Table 1). The median age of our patients was three months, and 72.4% (21/29) were infants under six months old who would not have completed their primary immunization course against pertussis according to the local schedule. A majority (93.1%, 27/29) of patients had cough, but only 10.3% (3/29) exhibited the classical symptom of paroxysmal cough, while 13.8% (4/29) demonstrated wheezing. B. pertussis infection resulted in cyanosis in 31.0% (9/29), pneumonia in 17.2% (5/29) and respiratory failure in 13.8% (4/29) of patients, while 6.9% (2/29) of patients required mechanical ventilation and 10.3% (3/29) required supportive nasogastric feeding. All cases presenting with cyanosis, pneumonia, respiratory failure and requiring mechanical ventilation were infants under 6 months of age. The median white blood cell, lymphocyte and C reactive protein of the patients were 15.15 × 109/L, 10.40 × 109/L and 0.6 mg/L, respectively. One (3.5%) of the patients had recent use of a third-generation cephalosporin, while none had recently used a macrolide group antibiotic. All patients were treated with a macrolide group antibiotic, but one patient (case 25) was later switched to trimethoprim-sulfamethoxazole due to treatment failure and subsequent detection of phenotypic resistance. All patients survived and recovered after a median 4 days of hospital stay (Table 1).

Table 1.
Clinical Symptoms, Laboratory Findings, Clinical Outcome and Management of 29 Cases with Culture-proven Pertussis—by A2047G mutation.
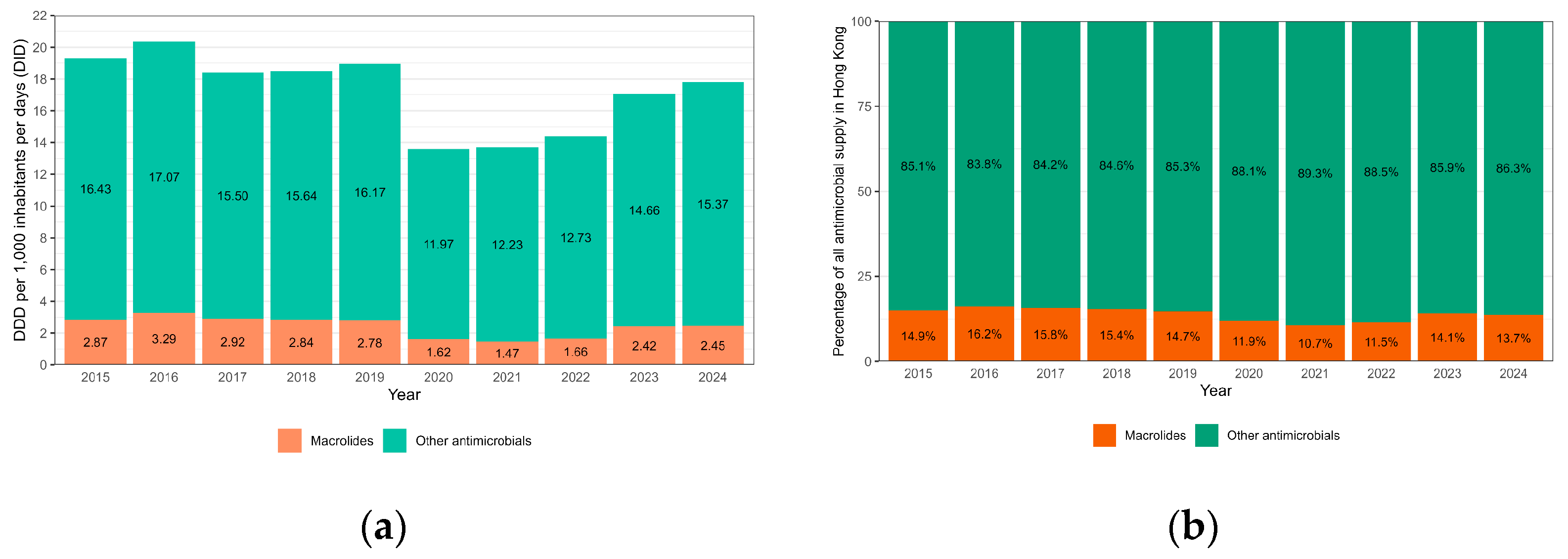
Clinically, there was no obvious correlation between macrolide resistance and clinical presentation, laboratory findings, disease severity, management and outcome in our cohort (Table 1). Travel history to mainland China within 30 days was noted in 40% (4/10) of the patients infected with MRBP (Table 1). The other clinical data have been summarized in Table 1. Figure 2a shows the supply of macrolides and other antimicrobials in Hong Kong over the study period. The proportion of macrolides among all antimicrobials fluctuated between 14.7% and 16.2% from 2015 to 2019 before it dropped slightly during the COVID-19 period from 2020 to 2022, ranging from 10.7% to 11.9% (Figure 2b). Overall, there was no statistically significant change in the consumption pattern of macrolides from 2015 to 2024.
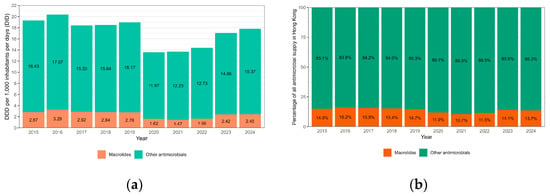
Figure 2.
(a) Wholesale supply of antimicrobials in Hong Kong, 2015–2024, measured in daily defined dose per 1000 inhabitants; (b) Wholesale supply of antimicrobials in Hong Kong, 2015–2024, measured in percentage. Linear regression: p = 0.06.
Macrolide resistance in B. pertussis is associated with the A2047G mutation in the 23S rRNA gene [35]. Although standardized breakpoints for detecting macrolide resistance in B. pertussis have not been established by the European Committee on Antimicrobial Susceptibility Testing (EUCAST) or the Clinical and Laboratory Standards Institute (CLSI), all B. pertussis isolates with the A2047G mutation in our study exhibited MICs of ≥256 μg/mL when tested with all three macrolides (azithromycin, erythromycin, clarithromycin) using the MIC test strip method. All isolates, irrespective of macrolide susceptibility, showed low MIC to all other tested antibiotics (Table 2). The MICs of individual isolates are summarized in Appendix A Table A1.

Table 2.
Phenotypic and Genotypic Test Results of 29 Bordetella pertussis Isolates Isolated from Hong Kong.
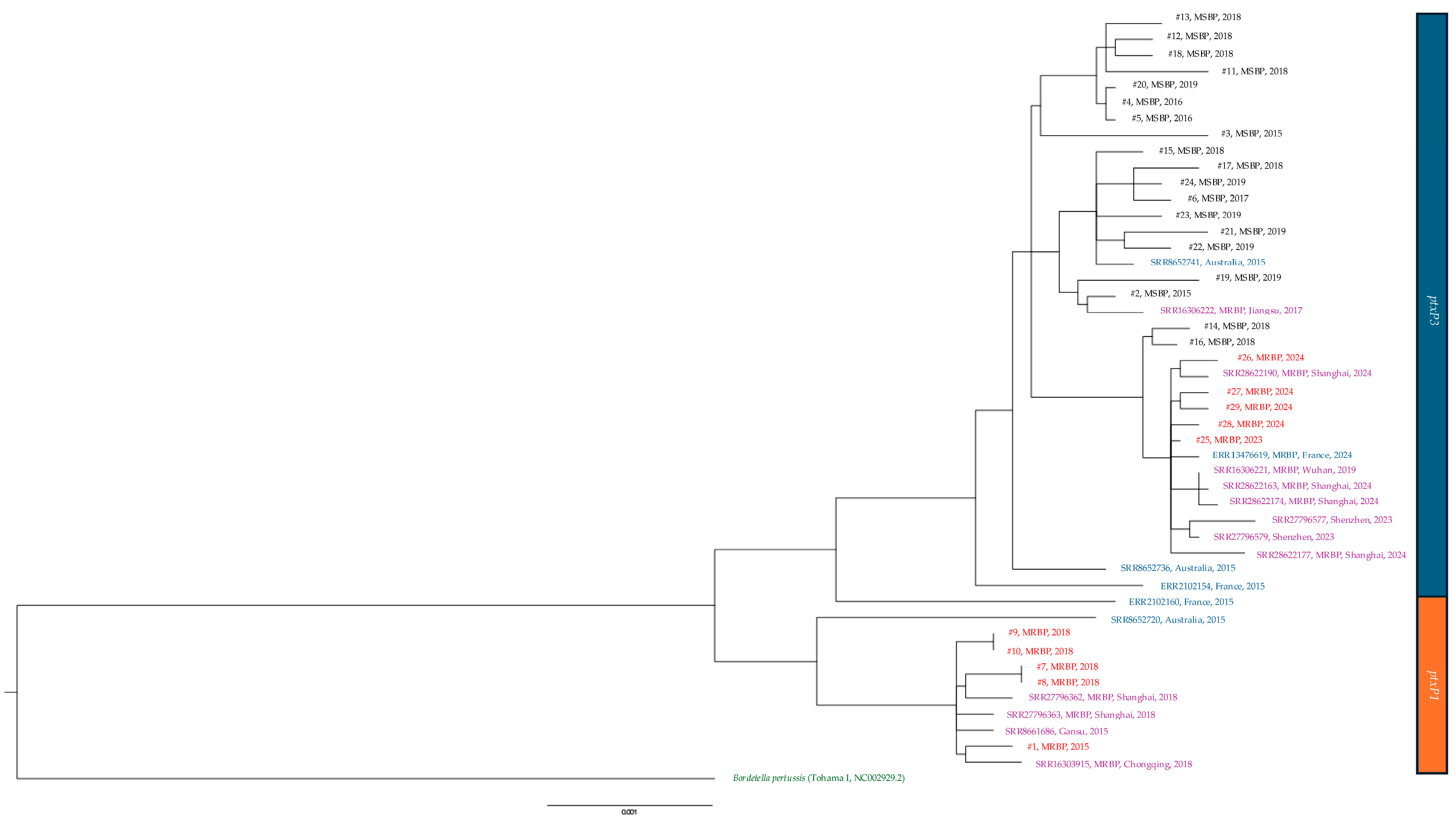
All the 29 isolates were subjected to WGS analysis. Based on the results obtained, before 2019, all MSBP harbored ptxP3/ptxA1/ptxC2/prn2/fim2-1/fim3-1/tcfA2/fhaB1/bscI2 and all MRBP harbored ptxP1/ptxA1/ptxC1/prn1/fim2-1/fim3-1/tcfA2/fhaB3/bscI1. After 2019, all MRBP harbored ptxP3/ptxA1/ptxC2/prn150/fim2-1/fim3-1/tcfA2/fhaB1/bscI2. The molecular typing results of the isolates are listed in Table 3, and all isolates after 2023 were MT28 (Table 3). No pertactin-deficient isolates were found in the current study. The phylogenetic relationship between our isolates, isolates from mainland China and worldwide are shown in Figure 3 and Figure 4.

Table 3.
Demographics, Genotypic, MLST and MLVA Results of 29 Bordetella pertussis Isolates Isolated from Hong Kong.

Figure 3.
Phylogenetic tree based on the complete genome of macrolide-resistant Bordetella pertussis isolates and other isolates of the same period. In red, isolates from study; in purple, isolates from mainland China; in blue, isolates from other countries; in green, reference strain. #, isolate; MRBP, macrolide-resistant Bordetella pertussis; ptxP1, pertussis toxin promoter 1; ptxP3, pertussis toxin promoter 3.
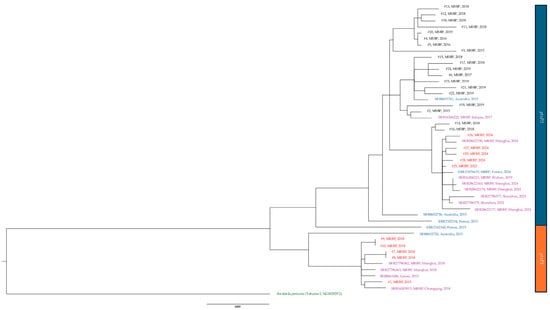
Figure 4.
Phylogenetic tree based on the complete genome of macrolide-sensitive and macrolide-resistant Bordetella pertussis isolates and other isolates of the same period. In red, MRBP from study; in black, MSBP from study; in purple, MRBP from mainland China; in blue, MRBP from other countries; in green, reference strain. #, isolate; MSBP, Macrolide-sensitive Bordetella pertussis; MRBP, macrolide-resistant Bordetella pertussis; ptxP1, pertussis toxin promoter 1; ptxP3, pertussis toxin promoter 3.
4. Discussion
This is the first report to describe the molecular characteristics and macrolide resistance of B. pertussis in Hong Kong. MRBP was detected as early as 2015 in Hong Kong and was demonstrated to have reached a prevalence of 100% after the COVID-19 pandemic in our cohort. The COVID-19 pandemic was also observed to be a critical timepoint for the transformation and epidemiology of MRBP in the major cities of mainland China, such as in Shanghai (36.4% in 2016 to 97.2% in 2022) [16], Tianjin (94.44% in 2012–2017) [42], Shenzhen (46.8% in 2015–2017) [43], Hunan (49% in 2017) [44] and Xi’an (79.3% in 2018–2020) [45].
Previous genotypic studies show that the rising macrolide resistance and shifting virulence genes of B. pertussis were largely influenced by the overuse of macrolides in mainland China [46]. In Hong Kong, the apparent rise in MRBP was observed despite a fairly stable consumption of macrolides during the study period. The slight drop in macrolide usage during the COVID-19 pandemic was probably due to reduced respiratory infections, for which macrolides were often prescribed especially in outpatient settings [47]. Instead, we observed that a higher proportion (40%, 4/6) of the MRBP cases in our study had a recent travel history to mainland China. Our latest MRBP isolates in 2023 and 2024 exhibited the same genotype and virulence-associated allelic profiles as the recent ptxP3/prn150 MR-MT28 strain circulating in mainland China [8,15]. Our genome analysis shows that the macrolide-resistant isolates in our cohort are closely associated with MRBP from mainland China during the same time period (Figure 3). The MRBP collected in 2024 in France (ERR13476619) was genotypically and phylogenetically related to isolates from China [19]. The above suggests that the MRBP seen in Hong Kong could have been introduced from mainland China.
Consistent with previous studies, we demonstrated no significant difference in clinical outcomes between MSBP and MRBP infections despite the use of ineffective antimicrobials in the latter group of patients [15]. Despite similar clinical outcomes, MRBP-infected patients can act as a persistent source of transmission due to use of ineffective antibiotics [15]. Macrolides were shown to be ineffective for eradication of MRBP in the nasopharynx in previous studies, with clearance of 51.2% and 74.2% on day 7 and 14 after treatment, respectively, with the longest carriage time of up to 81 days after initial detection [48]. The selection of MRBP and its prolonged shedding with the use of macrolides can facilitate its transmission in the community.
The clinical significance of the allelic divergence of B. pertussis remains controversial. Strains containing ptxP3 were believed to be more virulent as they produced more pertussis toxins than ptxP1 strains in some previous studies, but infections by ptxP3 strains were previously noted to be milder and were more likely to affect older, vaccinated individuals [14,22,49]. When stratifying the clinical outcomes according to pertussis promotor toxins, we demonstrated that our ptxP3-containing B. pertussis strains resulted in significantly higher hospitalization rates compared to ptxP1-containing strains, but other clinical outcomes were not significantly different (Appendix A Table A2). Our study also demonstrated a shift in the dominant virulence gene prn. All MSBP harbored the prn2 allele and all MRBP harbored the prn1 allele before 2019 (Table 3). After 2019, all B. pertussis isolates were macrolide-resistant and harbored prn150. The shift to prn150 was also observed in other studies as a result of divergence from the vaccine strain [24]. The clinical significance of prn150 has not been described.
4.1. Future Directions
Without effective antimicrobials to eradicate the nasopharyngeal carriage of MRBP, prolonged asymptomatic shedding can act as a continuous source leading to community outbreaks. This is an urgent call to review local treatment and post-exposure prophylaxis strategies by considering alternatives to macrolides to control pertussis. Both trimethoprim-sulfamethoxazole and doxycycline are shown to be effective in vitro for killing B. pertussis in previous studies [50]. Several fluoroquinolones, including levofloxacin, which was also tested in our study, showed good in vitro activity against B. pertussis and achieved concentrations in respiratory secretions that were well above their MICs [51]. However, trimethoprim-sulfamethoxazole is not licensed for infants younger than 6 weeks old [30]. Tetracyclines were believed to cause dental staining and were previously avoided in children younger than 8 years old. Recent guidelines suggest that newer generation tetracyclines, e.g., doxycycline, is not likely to cause visible teeth staining or enamel hypoplasia in children younger than 8 years when administered for short durations [52]. Although current guidelines do not routinely recommend the use of levofloxacin in children younger than 18 years, it can be used in specific conditions when there are no alternatives, or when the drug is known to be effective for the specific situation [53]. For children requiring inpatient treatment, beta-lactam group antibiotics such as piperacillin-tazobactam or meropenem may be considered. The effectiveness of beta-lactam group antibiotics was demonstrated in previous in vivo studies, where 91.5% and 95.7% clearance of MRBP in the nasopharynx was achieved on day 7 and day 14 after treatment, respectively [48]. Cefoperazone-sulbactam was also demonstrated to result in satisfactory clinical clearance of B. pertussis in the nasopharynx [54]. These drugs can be safely used in infants but require intravenous access and hospitalization.
Despite the high prevalence of MRBP, the latest Chinese National Guidelines for Treatment and Post-exposure Prophylaxis for B. pertussis in 2024 still recommend azithromycin as the first-line treatment for whooping cough, but recommend hospitals with adequate resources to perform resistance testing for B. pertussis [55]. Second-line agents include trimethoprim-sulfamethoxazole or levofloxacin for infants older than 2 months and adults, and piperacillin-tazobactam or cefoperazone-sulbactam for those younger than 2 months [55]. The local guidelines in Hong Kong give similar recommendations [27]. Our latest data suggest that there is an imminent need for Hong Kong to take the lead to update its guidelines to control MRBP.
4.2. Call to Action
In the context of globalization, a comprehensive surveillance system for MRBP is necessary [56]. This includes a high index of suspicion for whooping cough even in older and vaccinated individuals, and a low threshold for testing. Cultures should be routinely performed for polymerase chain reaction (PCR) positive cases, and the isolate should be subjected to antimicrobial resistance testing and epidemiological study using molecular methods. Hong Kong, an international financial hub with up to 34 million international visitors in 2023 [57], can potentially disseminate MRBP to other geographical regions if the transmission of MRBP is not adequately controlled. Prompt actions to update guidelines with the use of non-macrolide antibiotics may be required to help contain MRBP and prevent regional and global spread.
4.3. Limitations
Only 6.7% (29/431) of pertussis case reported to the Centre for Health Protection in Hong Kong from 2015 to June 2024 could be included in the current study. Several factors contributed to the low inclusion rate. Firstly, the molecular method is the main avenue for diagnosis of pertussis in Hong Kong, while B. pertussis culture is only performed on PCR-positive patients from respiratory specimens taken after the PCR result is available [58]. As macrolides are often started empirically for highly suspicious cases before PCR results are available in practice, a per-nasal culture swab taken after the PCR positive result and empirical treatment may have a lower yield. Due to the retrospective nature of our study, some archived isolates were no longer available at the time of our study.
Additionally, a majority of the isolates (24/29) from our study were from children, with 72.4% being under 6 months of age. Unfortunately, data on maternal immunization against pertussis was not available from the electronic medical record. However, as the maternal immunization program was implemented only after July 2020, it is likely that none of the mothers of the infant cases would have received the pertussis vaccine before 2020.
Finally, B. pertussis infections in adults are often mild and less symptomatic, so diagnosing pertussis is challenging in adults and could be underrepresented in our cohort [59]. Only 1/5 of our isolates from adult patients presented symptomatically, while the rest were isolated by contact tracing.
5. Conclusions
This is the first study to describe the epidemiology and prevalence of MRBP in Hong Kong. The first MRBP isolate was detected in 2015, and after the COVID-19 pandemic its prevalence has reached 100%. MRBP isolates in Hong Kong showed phylogenetic relatedness to isolates in mainland China around the same period. All MRBP showed low MIC to non-macrolide antimicrobials, which should be considered as first-line agents for effective treatment and eradication of MRBP to prevent regional and global spread.
Author Contributions
Conceptualization, T.-Y.H. and D.C.L.; methodology, H.K.-H.L. and G.K.-Y.C.; validation, G.K.-Y.C., H.T., S.C.-Y.W. and D.C.L.; formal analysis, T.-Y.H., G.K.-Y.C., H.T., S.C.-Y.W. and D.C.L.; data curation, S.K.-Y.C., L.-M.T., C.W.-S.T., K.-K.F., J.Y.-W.L., H.-L.N., T.H.-C.T., E.S.-K.M., V.W.-M.C. and D.C.L.; writing—original draft preparation, T.-Y.H.; writing—review and editing, S.K.-Y.C., L.-M.T., C.W.-S.T., K.-K.F., J.Y.-W.L., H.-L.N., T.H.-C.T., E.S.-K.M., S.C.-Y.W., V.W.-M.C. and D.C.L.; supervision, D.C.L.; project administration, D.C.L. All authors have read and agreed to the published version of the manuscript.
Funding
The article processing charge funded by the Department of Pathology, Queen Elizabeth Hospital.
Institutional Review Board Statement
Ethical approval was obtained from Research Ethics Committee of Kowloon Central/Kowloon East Cluster, Hospital Authority (HA) (Reference Number: PAED-2024-078), approval date on 1 November 2024.
Informed Consent Statement
Patient consent was waived as this is an observational retrospective study and no additional testing was performed on patients.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
Thanks must be given to technical staff in Queen Elizabeth Hospital and Hong Kong Children’s Hospital for performing laboratory tests, Public Health Laboratory Service Branch colleagues in retrieving culture isolates and clinicians for taking care of the patients involved. This article is an expanded version of a poster entitled “First report of Macrolide-resistant Bordetella pertussis in Hong Kong”, which was presented at Asian Congress of Pediatric Infectious Diseases (ACPID), Hong Kong, 13–15 November 2024. The feedback from the attendees was greatly appreciated.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| MRBP | Macrolide-resistant Bordetella pertussis |
| MIC | Minimum inhibitory concentration |
| MLST | Multi-locus antigen sequence typing |
| MLVA | Multi-locus variable number of tandem repeat analysis |
| dTap | Diphtheria (reduced dose), tetanus & acellular pertussis (reduced dose) |
| COVID-19 | Coronavirus disease-2019 |
| WHO | World Health Organization |
| PEP | Post-exposure prophylaxis |
| DDD | Defined daily dose |
| MSBP | Macrolide-sensitive B. pertussis |
| EUCAST | European Committee on Antimicrobial Susceptibility Testing |
| CLSI | Clinical and Laboratory Standards Institute |
| PCR | Polymerase chain reaction |
Appendix A

Table A1.
Patient Demographics, Phenotypic and Genotypic Testing Results of 29 Bordetella pertussis Isolates.
Table A1.
Patient Demographics, Phenotypic and Genotypic Testing Results of 29 Bordetella pertussis Isolates.
| Isolate | Case/ Contact | Sex | Age | Year | Travel History to Mainland China * | A2047G Mutation | Minimum Inhibitory Concentration (μg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ERY | AZT | CLR | SXT | DOX | LEV | TZP | MEM | |||||||
| 1 | Case | F | 4 m | 2015 | Y | D | ≥256 | ≥256 | ≥256 | 0.38 | 2 | 0.25 | <0.016 | 0.094 |
| 2 | Case | F | 4 m | 2015 | Y | ND | 0.064 | 0.094 | 0.75 | 0.5 | 2 | 0.25 | <0.016 | 0.125 |
| 3 | Case | M | 2 m | 2015 | N | ND | 0.094 | 0.094 | 0.5 | 1 | 3 | 0.25 | <0.016 | 0.094 |
| 4 | Case | M | 1 m | 2016 | N | ND | 0.19 | 0.094 | 0.38 | 0.38 | 3 | 0.38 | <0.016 | 0.094 |
| 5 | Case | M | 1 m | 2016 | N | ND | 0.047 | 0.094 | 0.38 | 0.38 | 2 | 0.25 | <0.016 | 0.064 |
| 6 | Case | M | 1 m | 2017 | N | ND | 0.047 | 0.125 | 0.75 | 0.38 | 2 | 0.25 | <0.016 | 0.094 |
| 7 | Case | F | 2 m | 2018 | N | D | ≥256 | ≥256 | ≥256 | 0.5 | 2 | 0.25 | <0.016 | 0.125 |
| 8 | Contact | M | 32 y | 2018 | N | D | ≥256 | ≥256 | ≥256 | 0.032 | 1 | 0.25 | <0.016 | 0.032 |
| 9 | Contact | M | 53 y | 2018 | N | D | ≥256 | ≥256 | ≥256 | 0.064 | 2 | 0.25 | <0.016 | 0.094 |
| 10 | Contact | M | 14 y | 2018 | N | D | ≥256 | ≥256 | ≥256 | 0.38 | 2 | 0.25 | <0.016 | 0.094 |
| 11 | Case | F | 1 m | 2018 | N | ND | 0.094 | 0.094 | 0.75 | 0.75 | 2 | 0.25 | <0.016 | 0.064 |
| 12 | Case | F | 3 m | 2018 | N | ND | 0.19 | 0.094 | 0.75 | 0.5 | 2 | 0.25 | <0.016 | 0.064 |
| 13 | Case | M | 1 m | 2018 | N | ND | 0.125 | 0.094 | 0.5 | 0.094 | 2 | 0.25 | <0.016 | 0.125 |
| 14 | Case | F | 1 m | 2018 | N | ND | 0.064 | 0.064 | 0.75 | 0.5 | 2 | 0.38 | <0.016 | 0.047 |
| 15 | Case | M | 1 m | 2018 | N | ND | 0.19 | 0.064 | 0.75 | 0.25 | 2 | 0.25 | <0.016 | 0.008 |
| 16 | Case | M | 1 m | 2018 | Y | ND | 0.125 | 0.094 | 0.75 | 1.5 | 2 | 0.25 | <0.016 | 0.064 |
| 17 | Case | F | 3 m | 2018 | N | ND | 0.094 | 0.047 | 0.75 | 0.047 | 2 | 0.38 | <0.016 | 0.047 |
| 18 | Case | M | 35 y | 2018 | N | ND | 0.125 | 0.094 | 2 | 1 | 1.5 | 0.25 | <0.016 | 0.094 |
| 19 | Case | F | 2 m | 2019 | N | ND | 0.125 | 0.125 | 1.5 | 0.75 | 3 | 0.19 | <0.016 | 0.125 |
| 20 | Case | F | 5 m | 2019 | N | ND | 0.064 | 0.032 | 0.5 | 0.5 | 2 | 0.25 | <0.016 | 0.094 |
| 21 | Contact | F | 58 y | 2019 | N | ND | 0.094 | 0.032 | 0.5 | 0.38 | 3 | 0.38 | <0.016 | 0.094 |
| 22 | Case | F | 2 m | 2019 | N | ND | 0.094 | 0.032 | 0.5 | 0.25 | 2 | 0.38 | <0.016 | 0.094 |
| 23 | Case | F | 6 m | 2019 | N | ND | 0.064 | 0.047 | 0.75 | 0.047 | 1 | 0.19 | <0.016 | 0.064 |
| 24 | Case | M | 42 y | 2019 | N | ND | 0.094 | 0.064 | 0.75 | 1 | 4 | 0.25 | <0.016 | 0.094 |
| 25 | Case | M | 3 m | 2023 | Y | D | ≥256 | ≥256 | ≥256 | 0.5 | 3 | 0.25 | <0.016 | 0.064 |
| 26 | Case | M | 3 m | 2024 | Y | D | ≥256 | ≥256 | ≥256 | 1 | 3 | 0.25 | <0.016 | 0.094 |
| 27 | Case | M | 2 m | 2024 | N | D | ≥256 | ≥256 | ≥256 | 0.5 | 3 | 0.25 | <0.016 | 0.094 |
| 28 | Case | M | 9 y | 2024 | Y | D | ≥256 | ≥256 | ≥256 | 0.5 | 2 | 0.25 | <0.016 | 0.094 |
| 29 | Case | M | 4 m | 2024 | N | D | ≥256 | ≥256 | ≥256 | 1.5 | 2 | 0.38 | <0.016 | 0.094 |
M, male; F, female; m, month; y, year; Y, yes; N, no; D, detected; ND, not detected; ERY, erythromycin; AZT, azithromycin; CLR, clarithromycin; SXT, trimethoprim-sulfamethoxazole; DOX, doxycycline; LEV, levofloxacin; TZP, piperacillin-tazobactam; MEM, meropenem. * within 4 weeks.

Table A2.
Clinical Symptoms, Laboratory Findings, Clinical Outcome and Management of 29 Cases with Culture-proven Pertussis—by ptxP.
Table A2.
Clinical Symptoms, Laboratory Findings, Clinical Outcome and Management of 29 Cases with Culture-proven Pertussis—by ptxP.
| Characteristics | All Cases (n = 29) | ptxP1 (n = 5) | ptxP3 (n = 24) | p-Value | Odds Ratio a | |
|---|---|---|---|---|---|---|
| Estimate | 95% CI | |||||
| Clinical symptoms—n (%) | ||||||
| Paroxysmal cough | 3 (10.3%) | 1 (20%) | 2 (8.3%) | NS | NA | NA |
| Fever | 2 (6.90%) | 1 (20%) | 1 (4.2%) | NS | NA | NA |
| Cyanosis | 9 (31.0%) | 2 (40%) | 7 (29.2%) | NS | NA | NA |
| Wheezing | 4 (13.8%) | 1 (20%) | 3 (12.5%) | NS | NA | NA |
| Pneumonia | 5 (17.2%) | 0 (0%) | 5 (20.8%) | NS | NA | NA |
| Respiratory failure | 4 (13.8%) | 0 (0%) | 4 (16.7%) | NS | NA | NA |
| Laboratory findings—median (IQR) | ||||||
| White blood cell (×109/L) b | 15.15 (10.70–23.70) | 27.6 (16.40–38.80) | 13.75 (10.70–23.65) | NS | NA | NA |
| Lymphocyte (×109/L) b | 10.40 (6.40–18.10) | 19.6 (10.10–29.10) | 10.40 (6.40–13.70) | NS | NA | NA |
| C reactive protein (mg/L) b | 0.60 (0.25–1.00 | 0.91 (0.25–1.56) | 0.60 (0.20–1.00) | NS | NA | NA |
| Clinical outcome—n (%) | ||||||
| Survival | 29 (100%) | 5 (100%) | 24 (100%) | NA | NA | NA |
| Hospitalization | 22 (75.9%) | 2 (40%) | 21 (87.5%) | <0.05 | 0.10 | (0.01, 0.83) |
| Length of hospital stay, median (interquartile range, days) | 4.00 (3.00–6.00) | 0.00 (0.00–6.00) | 4.00 (3.00–5.50) | NS | NA | NA |
| Required oxygen therapy | 4 (13.8%) | 0 (0.0%) | 4 (16.7%) | NS | NA | NA |
| Required mechanical ventilation | 2 (6.90%) | 0 (0.0%) | 2 (8.3%) | NS | NA | NA |
| Required nasogastric feeding | 3 (10.3%) | 1 (20%) | 2 (8.3%) | NS | NA | NA |
| Antibiotic therapy c—n (%) | ||||||
| Macrolide | 28 (96.6%) | 4 (80%) | 24 (100%) | NS | NA | NA |
| Trimethoprim-sulfamethoxazole b | 1 (3.45%) | 0 (0.0%) | 1 (4.2%) | NS | NA | NA |
| Prior antibiotic usage—n (%) | ||||||
| Third-generation cephalosporin | 1 (3.45%) | 0 (0.0%) | 1 (4.2%) | NS | NA | NA |
| Macrolide | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | NA | NA | NA |
ptxP, pertussis toxin promoter; IQR, interquartile range; NA, not applicable; NS, not significant. a p-value was calculated with Mann–Whitney U test for parameters including white blood cells, lymphocytes, C reactive protein and length of stay. The p value was calculated with Fisher’s Exact Test for parameters including paroxysmal cough, fever, cyanosis, wheezing, pneumonia, respiratory failure, hospitalization, survival, oxygen therapy, mechanical ventilation and nasogastric feeding. b Result of this parameter is not available for all cases. c One patient did not receive treatment, while one patient received macrolide followed by trimethoprim-sulfamethoxazole due to treatment failure.
References
- Ho, F.W. Resurgence of Pertussis: A Global and Local Update; Centre for Health Protection (CHP), Department of Health: Hong Kong, 2025. Available online: https://www.chp.gov.hk/files/pdf/cdw_v21_6.pdf (accessed on 13 August 2025).
- Gordon, J.E.; Hood, R.I. Whooping cough and its epidemiological anomalies. Am. J. Med. Sci. 1951, 222, 333–361. [Google Scholar] [CrossRef] [PubMed]
- Domenech de Cellès, M.; Rohani, P. Pertussis vaccines, epidemiology and evolution. Nat. Rev. Microbiol. 2024, 22, 722–735. [Google Scholar] [CrossRef]
- Yeung, K.H.T.; Duclos, P.; Nelson, E.A.S.; Hutubessy, R.C.W. An update of the global burden of pertussis in children younger than 5 years: A modelling study. Lancet Infect. Dis. 2017, 17, 974–980. [Google Scholar] [CrossRef]
- Centre for Health Protection of the Department of Health of the Hong Kong Special Administrative Region. Consensus Recommendations on Pertussis Vaccination for Pregnant Women in Hong Kong. Available online: https://www.chp.gov.hk/files/pdf/recommendations_on_pertussis_vaccination_for_pregnant_women_in_hk_formatted.pdf (accessed on 11 August 2025).
- Centre for Health Protection of the Department of Health of the Hong Kong Special Administrative Region. Recommendations on Updated Childhood Immunisation Programme Containing Inactivated Poliovirus and Acellular Pertussis Vaccines. Available online: https://www.chp.gov.hk/files/pdf/sas6_recommendation_on_updated_childhood_immunisation_programme_(dec2006).pdf (accessed on 14 August 2025).
- National Health Commission of the People’s Republic of China. Childhood Immunization Schedule for National Immunization Program Vaccines—China (Version 2021). China CDC Wkly. 2021, 3, 1101–1108. [Google Scholar] [CrossRef]
- Cai, J.; Chen, M.; Liu, Q.; Luo, J.; Yuan, L.; Chen, Y.; Chen, M.; Zeng, M. Domination of an emerging erythromycin-resistant ptxP3 Bordetella pertussis clone in Shanghai, China. Int. J. Antimicrob. Agents 2023, 62, 106835. [Google Scholar] [CrossRef]
- Jiang, F.; Ye, X.; Wang, Y.; Tang, N.; Feng, J.; Gao, Y.; Bao, M. Factors associated with pregnant women’s willingness to receive maternal pertussis vaccination in Guizhou Province, China: An exploratory cross-sectional study. Hum. Vaccines Immunother. 2024, 20, 2331870. [Google Scholar] [CrossRef]
- Guo, M.; Hu, Y.; Meng, Q.; Shi, W.; Yao, K. Resurgence and atypical patterns of pertussis in China. J. Infect. 2024, 88, 106140. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention; Center for Surveillance, Epidemiology and Laboratory Services; National Notifiable Diseases Surveillance System. Pertussis (Week 49): Weekly Cases of Notifiable Diseases, United States, U.S. Territories, and Non-U.S. Residents Week Ending December 7, 2024. Available online: https://stacks.cdc.gov/view/cdc/174748 (accessed on 8 April 2025).
- UK Health Security Agency. Confirmed Cases of Whooping Cough in England by Month. Available online: https://www.gov.uk/government/publications/pertussis-epidemiology-in-england-2024/confirmed-cases-of-pertussis-in-england-by-month (accessed on 26 November 2024).
- World Health Organization. Pertussis Reported Cases and Incidence. Available online: https://immunizationdata.who.int/global/wiise-detail-page/pertussis-reported-cases-and-incidence?CODE=Global&YEAR= (accessed on 13 August 2025).
- Fu, P.; Wang, C.; Tian, H.; Kang, Z.; Zeng, M. Bordetella pertussis Infection in Infants and Young Children in Shanghai, China, 2016–2017: Clinical Features, Genotype Variations of Antigenic Genes and Macrolides Resistance. Pediatr. Infect. Dis. J. 2019, 38, 370–376. [Google Scholar] [CrossRef]
- Fu, P.; Yan, G.; Li, Y.; Xie, L.; Ke, Y.; Qiu, S.; Wu, S.; Shi, X.; Qin, J.; Zhou, J.; et al. Pertussis upsurge, age shift and vaccine escape post-COVID-19 caused by ptxP3 macrolide-resistant Bordetella pertussis MT28 clone in China. Clin. Microbiol. Infect. 2024, 30, 1439–1446. [Google Scholar] [CrossRef]
- Fu, P.; Zhou, J.; Yang, C.; Nijiati, Y.; Zhou, L.; Yan, G.; Lu, G.; Zhai, X.; Wang, C. Molecular Evolution and Increasing Macrolide Resistance of Bordetella pertussis, Shanghai, China, 2016–2022. Emerg. Infect. Dis. 2023, 30, 29–38. [Google Scholar] [CrossRef]
- Bartkus, J.M.; Juni, B.A.; Ehresmann, K.; Miller, C.A.; Sanden, G.N.; Cassiday, P.K.; Saubolle, M.; Lee, B.; Long, J.; Harrison, A.R.; et al. Identification of a Mutation Associated with Erythromycin Resistance in Bordetella pertussis: Implications for Surveillance of Antimicrobial Resistance. J. Clin. Microbiol. 2003, 41, 1167–1172. [Google Scholar] [CrossRef]
- Ivaska, L.; Barkoff, A.M.; Mertsola, J.; He, Q. Macrolide Resistance in Bordetella pertussis: Current Situation and Future Challenges. Antibiotics 2022, 11, 1570. [Google Scholar] [CrossRef]
- Rodrigues, C.; Bouchez, V.; Soares, A.; Trombert-Paolantoni, S.; Aït El Belghiti, F.; Cohen, J.F.; Armatys, N.; Landier, A.; Blanchot, T.; Hervo, M.; et al. Resurgence of Bordetella pertussis, including one macrolide-resistant isolate, France, 2024. Eurosurveillance 2024, 29, 2400459. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, M.; Wang, L.; Xin, T.; He, Q. High-resolution melting analysis for the detection of two erythromycin-resistant Bordetella pertussis strains carried by healthy schoolchildren in China. Clin. Microbiol. Infect. 2013, 19, E260–E262. [Google Scholar] [CrossRef]
- Yang, Y.; Yao, K.; Ma, X.; Shi, W.; Yuan, L.; Yang, Y. Variation in Bordetella pertussis Susceptibility to Erythromycin and Virulence-Related Genotype Changes in China (1970–2014). PLoS ONE 2015, 10, e0138941. [Google Scholar] [CrossRef] [PubMed]
- Mooi, F.R.; van Loo, I.H.; van Gent, M.; He, Q.; Bart, M.J.; Heuvelman, K.J.; de Greeff, S.C.; Diavatopoulos, D.; Teunis, P.; Nagelkerke, N.; et al. Bordetella pertussis strains with increased toxin production associated with pertussis resurgence. Emerg. Infect. Dis. 2009, 15, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Deng, J.; Ma, X.; Zhou, K.; Meng, Q.; Yuan, L.; Shi, W.; Wang, Q.; Li, Y.; Yao, K. High Prevalence of Macrolide-Resistant Bordetella pertussis and ptxP1 Genotype, Mainland China, 2014–2016. Emerg. Infect. Dis. 2019, 25, 2205–2214. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Li, Y.; Wang, H.; Wang, Y.; Gao, Y.; Xu, J.; Wang, F.; Peng, T.; Zhang, M.; Shao, Z. Emergence of Erythromycin-Resistant and Pertactin- and Filamentous Hemagglutinin-Deficient Bordetella pertussis Strains—Beijing, China, 2022–2023. China CDC Wkly. 2024, 6, 437–441. [Google Scholar] [CrossRef]
- Wu, X.; Du, Q.; Li, D.; Yuan, L.; Meng, Q.; Fu, Z.; Xu, H.; Yao, K.; Zhao, R. A Cross-Sectional Study Revealing the Emergence of Erythromycin-Resistant Bordetella pertussis Carrying ptxP3 Alleles in China. Front. Microbiol. 2022, 13, 901617. [Google Scholar] [CrossRef]
- Centre for Health Protection of the Department of Health of the Hong Kong Special Administrative Region. Update on the Regional and Local Situations of Pertussis. Available online: https://www.chp.gov.hk/files/pdf/cdw_v20_5.pdf (accessed on 10 April 2025).
- Hospital Authority. HA Central Committee on Infectious Disaes and Emergency Respsonse. Fact Sheet on Perrtussis (Whooping cough). Internal Document. 2019. Available online: https://ha.home/ho/ps/FactSheetonPertussis.pdf (accessed on 16 August 2025).
- Tiwari, T.; Murphy, T.V.; Moran, J. Recommended Antimicrobial Agents for the Treatment and Postexposure Prophylaxis of Pertussis: 2005 CDC Guidelines. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5414a1.htm (accessed on 19 March 2025).
- Communicable Diseases Network Australia. Pertussis CDNA National Guidelines for Public Health Units. Available online: https://www.health.gov.au/sites/default/files/2024-10/pertussis-whooping-cough-cdna-national-guidelines-for-public-health-units.pdf (accessed on 18 April 2025).
- UK Health Security Agency. Guidance on the Management of Cases of Pertussis in England During the Re-Emergence of Pertussis in 2024 Update: August 2024. Available online: https://assets.publishing.service.gov.uk/media/66c4a642808b8c0aa08fa7e7/UKHSA-guidance-on-the-management-of-cases-of-pertussis-during-high-activity-august-2024.pdf (accessed on 10 April 2025).
- Treatment of Pertussis. U.S. Centerse for Disease Control and Prevention. Available online: https://www.cdc.gov/pertussis/hcp/clinical-care/index.html (accessed on 11 August 2025).
- European Centre for Disease Prevention and Control. Laboratory Diagnosis and Molecular Surveillance of Bordetella pertussis. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/bordetella-pertussis-laboratory-diagnosis-molecular-surveillance.pdf (accessed on 10 April 2025).
- Hill Bertha, C.; Baker Carolyn, N.; Tenover Fred, C. A Simplified Method for Testing Bordetella pertussis for Resistance to Erythromycin and Other Antimicrobial Agents. J. Clin. Microbiol. 2000, 38, 1151–1155. [Google Scholar] [CrossRef]
- Seven Treatments for Bacterial Sample Materials with MagNA Pure Bacteria Lysis Buffer. Available online: https://lifescience.roche.com/global/en/article-listing/article/seven-treatments-for-bacterial-sample-materials-with-magna-pure-.html (accessed on 10 April 2025).
- Wang, Z.; Cui, Z.; Li, Y.; Hou, T.; Liu, X.; Xi, Y.; Liu, Y.; Li, H.; He, Q. High prevalence of erythromycin-resistant Bordetella pertussis in Xi’an, China. Clin. Microbiol. Infect. 2014, 20, O825–O830. [Google Scholar] [CrossRef]
- Tse, H.; Tsang, A.K.L.; Chu, Y.-W.; Tsang, D.N.C. Draft Genome Sequences of 19 Clinical Isolates of Candida auris from Hong Kong. Microbiol. Resour. Announc. 2021, 10. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef]
- Kolmogorov, M.; Raney, B.; Paten, B.; Pham, S. Ragout-a reference-assisted assembly tool for bacterial genomes. Bioinformatics 2014, 30, i302–i309. [Google Scholar] [CrossRef]
- Schouls, L.M.; van der Heide, H.G.; Vauterin, L.; Vauterin, P.; Mooi, F.R. Multiple-locus variable-number tandem repeat analysis of Dutch Bordetella pertussis strains reveals rapid genetic changes with clonal expansion during the late 1990s. J. Bacteriol. 2004, 186, 5496–5505. [Google Scholar] [CrossRef] [PubMed]
- WHO Director-General’s Opening Remarks at the Media Briefing. 2023. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing---5-may-2023 (accessed on 14 August 2025).
- Guo, L.; Zhang, W.; Su, X.; Huang, H.; Liu, Y. Analysis on drug resistance of Bordetella pertussis isolated in Tianjin. Dis. Surveill. 2018, 33, 585–589. [Google Scholar] [CrossRef]
- Zhang, J.S.; Wang, H.M.; Yao, K.H.; Liu, Y.; Lei, Y.L.; Deng, J.K.; Yang, Y.H. Clinical characteristics, molecular epidemiology and antimicrobial susceptibility of pertussis among children in southern China. World J. Pediatr. 2020, 16, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Zou, J.; Yao, K.; Li, L.; Zhong, L. Analysis of antibiotic sensitivity and resistance genes of Bordetella pertussis in Chinese children. Medicine 2021, 100, e24090. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, D.; Wang, X.; Wei, X.; Li, H. Macrolide susceptibility and molecular characteristics of Bordetella pertussis. J. Int. Med. Res. 2022, 50, 3000605221078782. [Google Scholar] [CrossRef]
- Li, J.; Liu, L.; Zhang, H.; Guo, J.; Wei, X.; Xue, M.; Ma, X. Severe problem of macrolides resistance to common pathogens in China. Front. Cell. Infect. Microbiol. 2023, 13, 1181633. [Google Scholar] [CrossRef] [PubMed]
- Ma, E.S.; Hsu, E.; Chow, V.; Chow, T.; Kung, K.H.; Au, A.; Chen, H. Rebound of Antibiotic Use and Respiratory Infections After Resumption of Normalcy From COVID-19 in Hong Kong. Infect. Drug Resist. 2025, 18, 1325–1337. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.M.; Hua, C.Z.; Fang, C.; Liu, J.J.; Xie, Y.P.; Lin, L.N.; Wang, G.L. Effect of Macrolides and β-lactams on Clearance of Bordetella pertussis in the Nasopharynx in Children With Whooping Cough. Pediatr. Infect. Dis. J. 2021, 40, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.F.; Dalby, T.; Dragsted, D.M.; Mooi, F.; Lambertsen, L. Temporal trends in Bordetella pertussis populations, Denmark, 1949–2010. Emerg. Infect. Dis. 2012, 18, 767–774. [Google Scholar] [CrossRef]
- Shi, W.; Meng, Q.; Hu, Y.; Yao, K. Modifying antibiotic treatment strategies in the face of pertussis surge associated to erythromycin resistance in China. J. Infect. 2024, 88, 106174. [Google Scholar] [CrossRef]
- Gordon, K.A.; Fusco, J.; Biedenbach, D.J.; Pfaller, M.A.; Jones, R.N. Antimicrobial susceptibility testing of clinical isolates of Bordetella pertussis from northern California: Report from the SENTRY Antimicrobial Surveillance Program. Antimicrob. Agents Chemother. 2001, 45, 3599–3600. [Google Scholar] [CrossRef]
- Committee on Infectious Diseases; American Academy of Pediatrics. Tetracyclines. In Red Book: 2021–2024 Report of the Committee on Infectious Diseases; Kimberlin, D.W., Barnett, E.D., Lynfield, R., Sawyer, M.H., Eds.; American Academy of Pediatrics: Itasca, IL, USA, 2021; p. 866. [Google Scholar]
- Committee on Infectious Diseases; American Academy of Pediatrics. Fluoroquinolones. In Red Book: 2021–2024 Report of the Committee on Infectious Diseases; Kimberlin, D.W., Barnett, E.D., Lynfield, R., Sawyer, M.H., Eds.; American Academy of Pediatrics: Itasca, IL, USA, 2021; pp. 864–866. [Google Scholar]
- Koide, K.; Yao, S.; Chiang, C.S.; Thuy, P.T.B.; Nga, D.T.T.; Huong, D.T.; Dien, T.M.; Vichit, O.; Vutthikol, Y.; Sovannara, S.; et al. Genotyping and macrolide-resistant mutation of Bordetella pertussis in East and South-East Asia. J. Glob. Antimicrob. Resist. 2022, 31, 263–269. [Google Scholar] [CrossRef]
- Mei, Z.; Zhujun, S.; Wenhong, Z.; Jun, X. Guidelines for diagnosis and management and prevention of pertussis of China (2024 edition). Chin. Med. Assoc. 2024. [Google Scholar] [CrossRef]
- Feng, Y.; Chiu, C.-H.; Heininger, U.; Hozbor, D.F.; Tan, T.Q.; von König, C.-H.W. Emerging macrolide resistance in Bordetella pertussis in mainland China: Findings and warning from the global pertussis initiative. Lancet Reg. Health–West. Pac. 2021, 8, 100098. [Google Scholar] [CrossRef]
- Hong Kong Tourism Board. Monthly Report—Visitor Arrival Statistics: Jan 2024. Available online: https://www.discoverhongkong.com/content/dam/dhk/intl/corporate/newsroom/tourisum-statistics/2024/tourism_stat_01_2024.pdf (accessed on 26 November 2024).
- Centre for Health Protection of the Department of Health of the Hong Kong Special Administrative Region. Number of Notifiable Infectious Diseases by Month. Available online: https://www.chp.gov.hk/en/statistics/data/10/26/43/7060.html (accessed on 18 April 2025).
- Jenkinson, D. Pertussis (whooping cough) is common in teens and adults. BMJ 2019, 365, l1623. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).