Comparison of the Effects of Feeding Compound Probiotics and Antibiotics on Growth Performance, Gut Microbiota, and Small Intestine Morphology in Yellow-Feather Broilers
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Experimental Design and Sampling
2.3. Measurement of Growth Performance
2.4. HE Staining and Histomorphology Analysis
2.5. Extraction of Genomic DNA and 16S rRNA Sequencing
2.6. Processing of Sequencing Data and Functional Prediction
2.7. Quantification of Antibiotic Resistance Genes
2.8. Plasma Biochemical Analysis
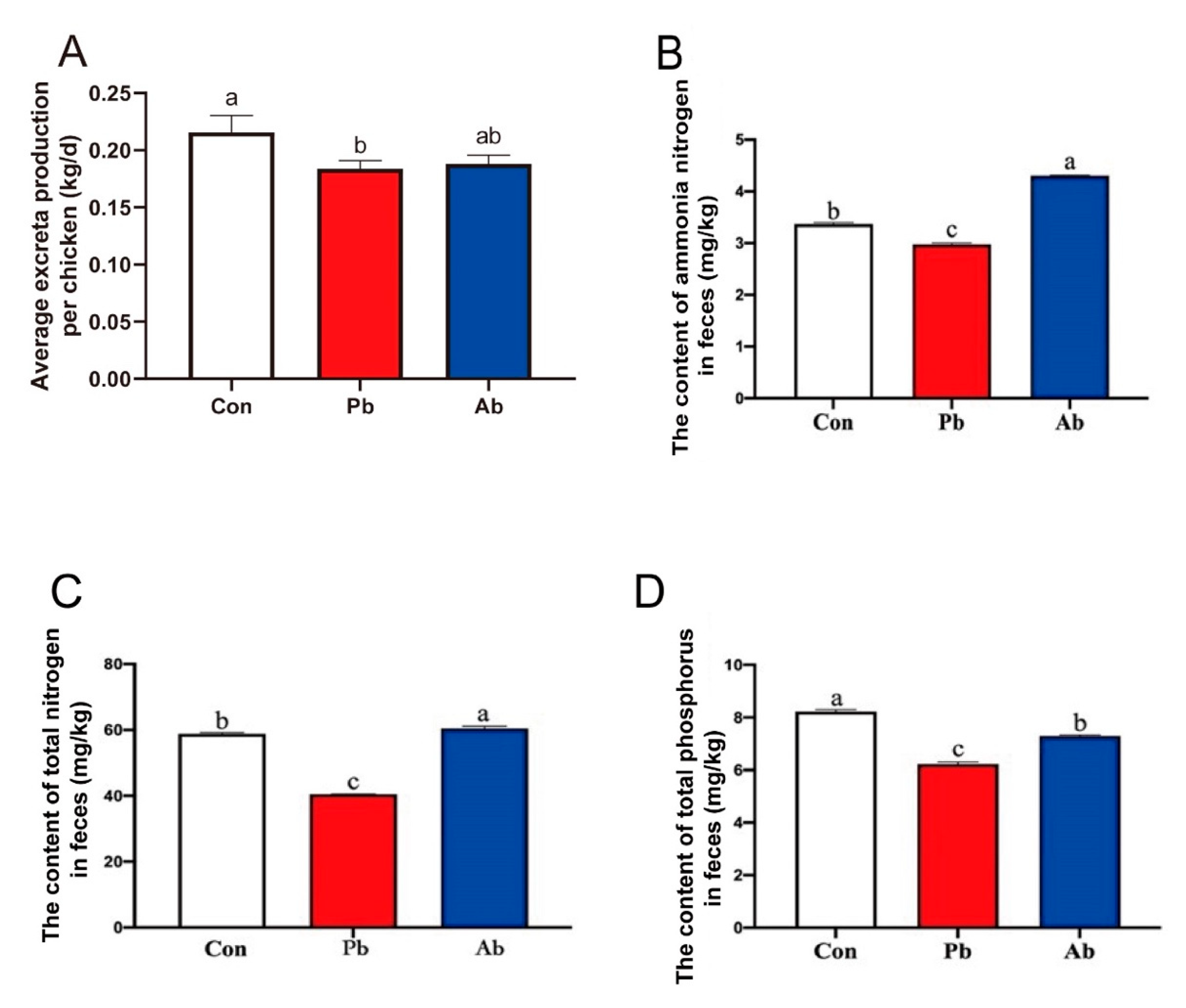
2.9. Determination of Total Phosphorus, Nitrogen, and Ammonia Nitrogen in Feces
2.10. Statistical Analysis
3. Result
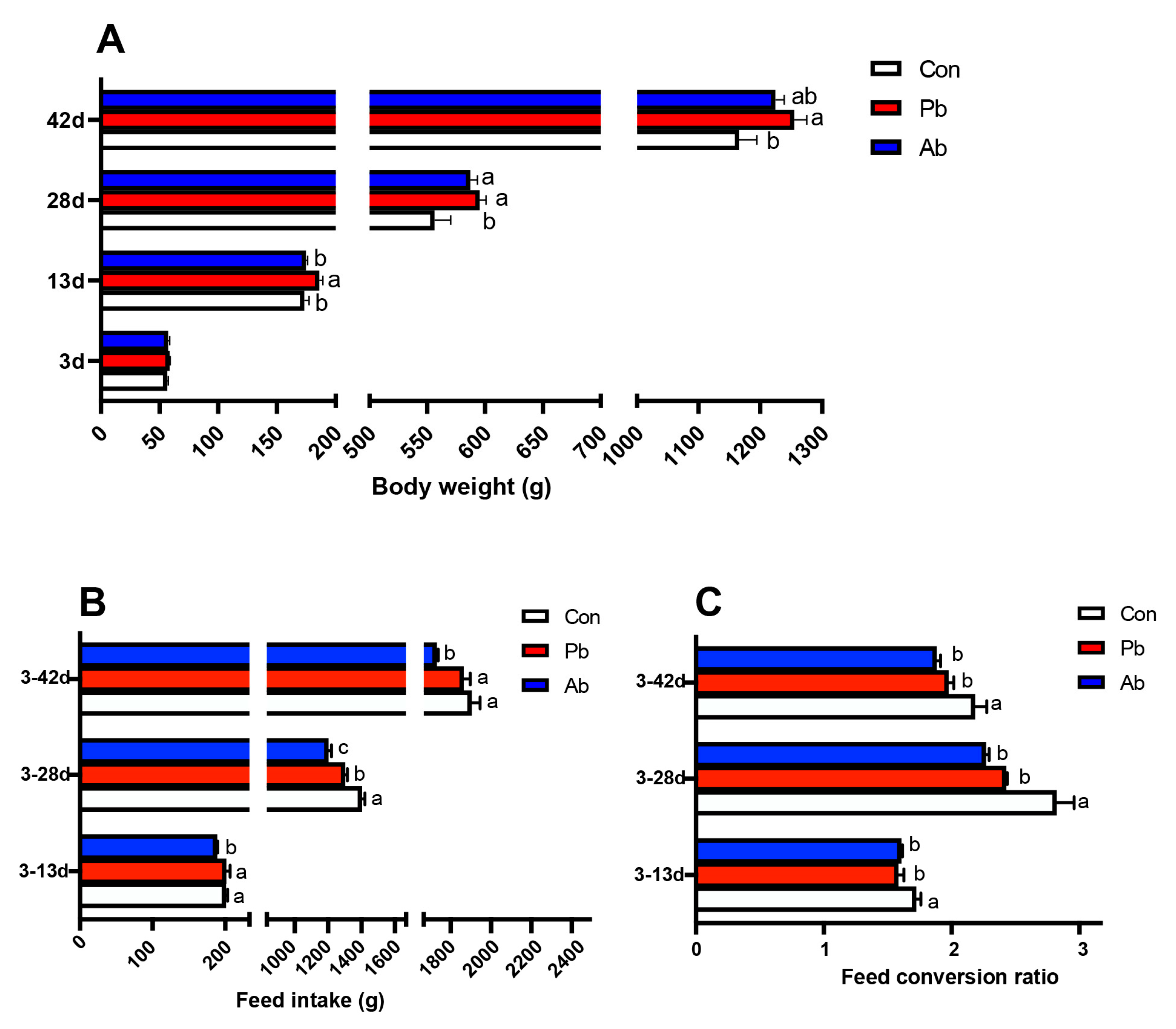
3.1. Body Weight, Food Intake, and Feed Conversion Ratio
3.2. Organ Indexes and Plasma Biochemical Indicators
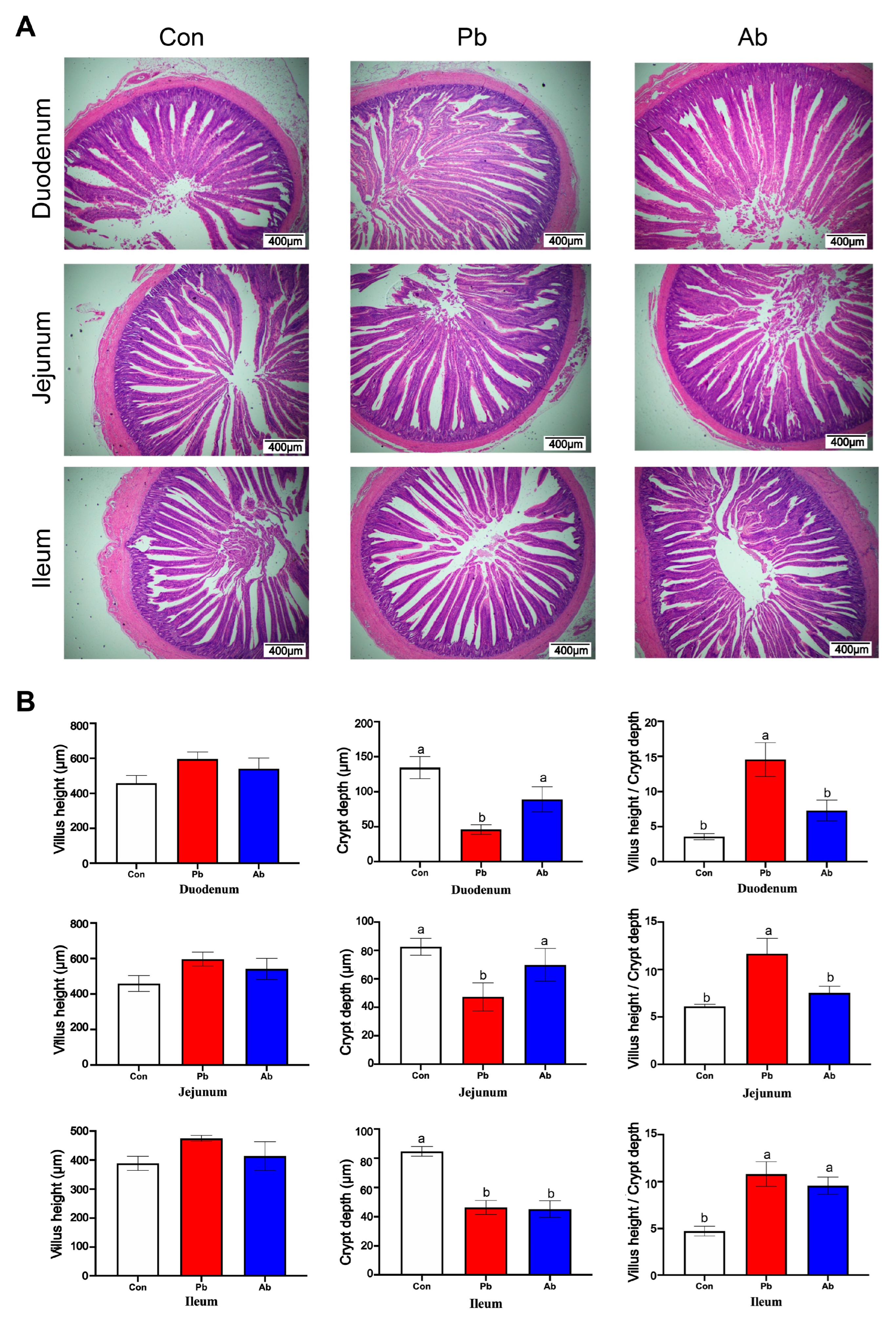
3.3. Morphology of the Small Intestines
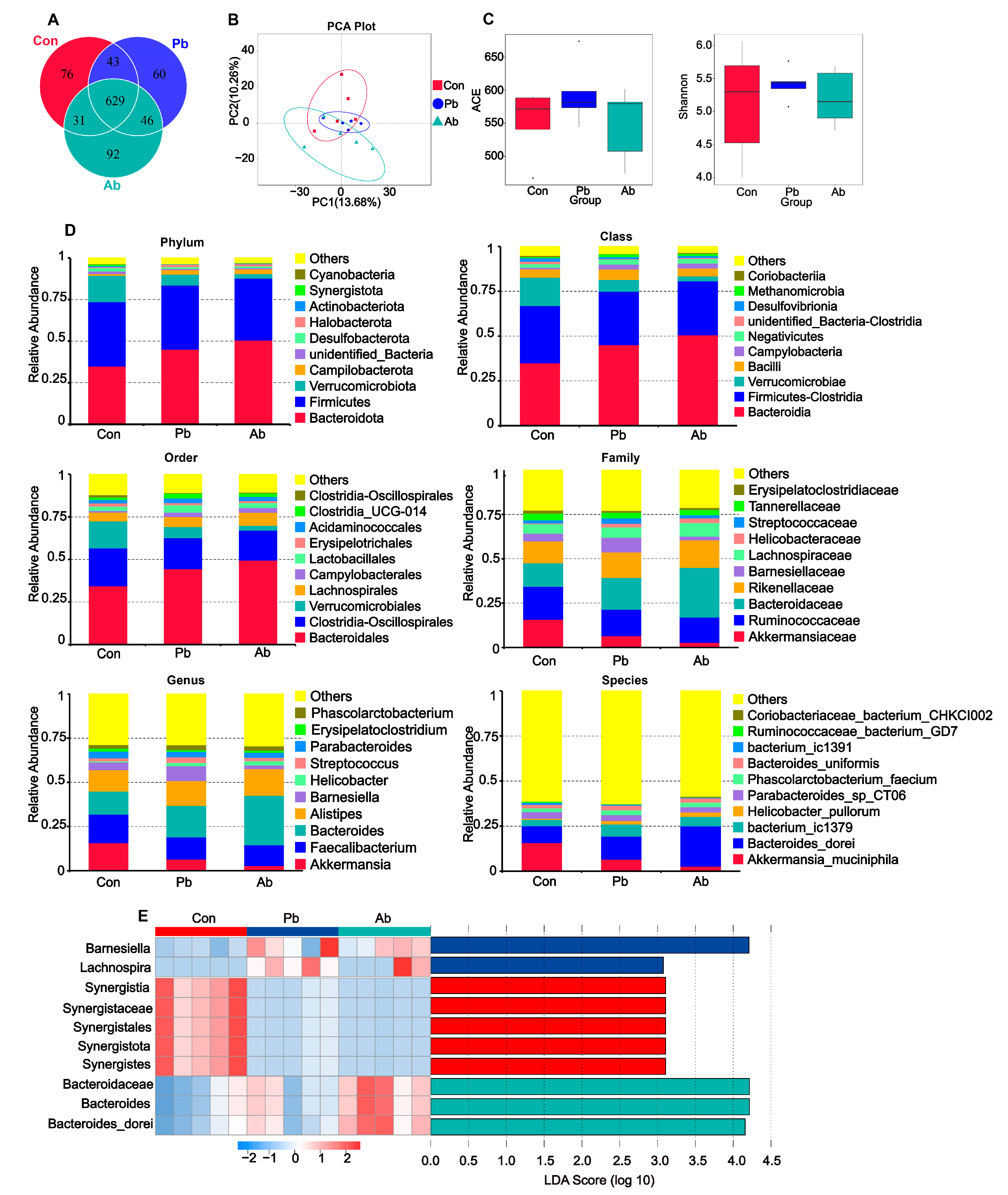
3.4. Bacterial Community Characterization in Cecal Contents
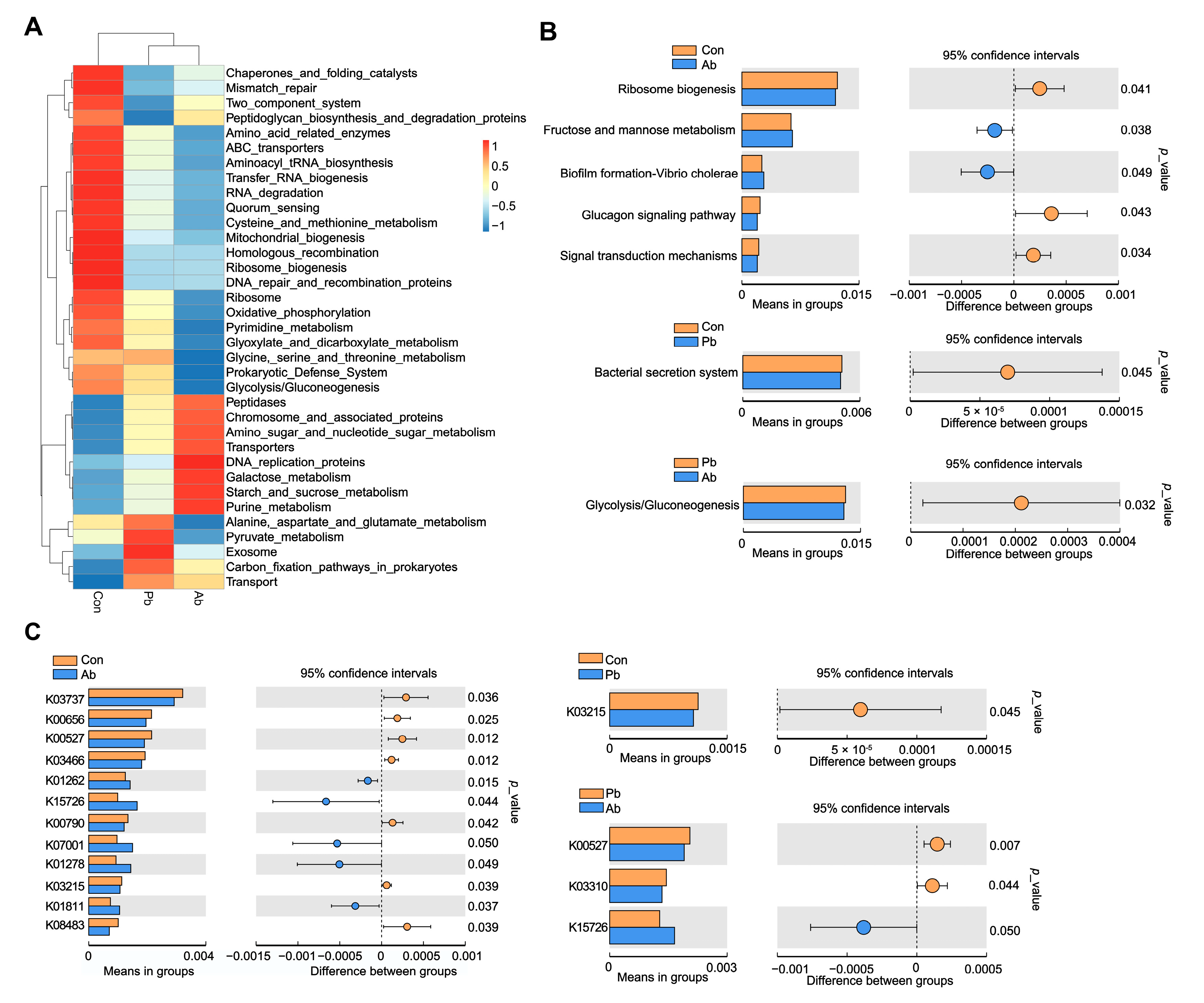
3.5. Functional Prediction of Microbiota
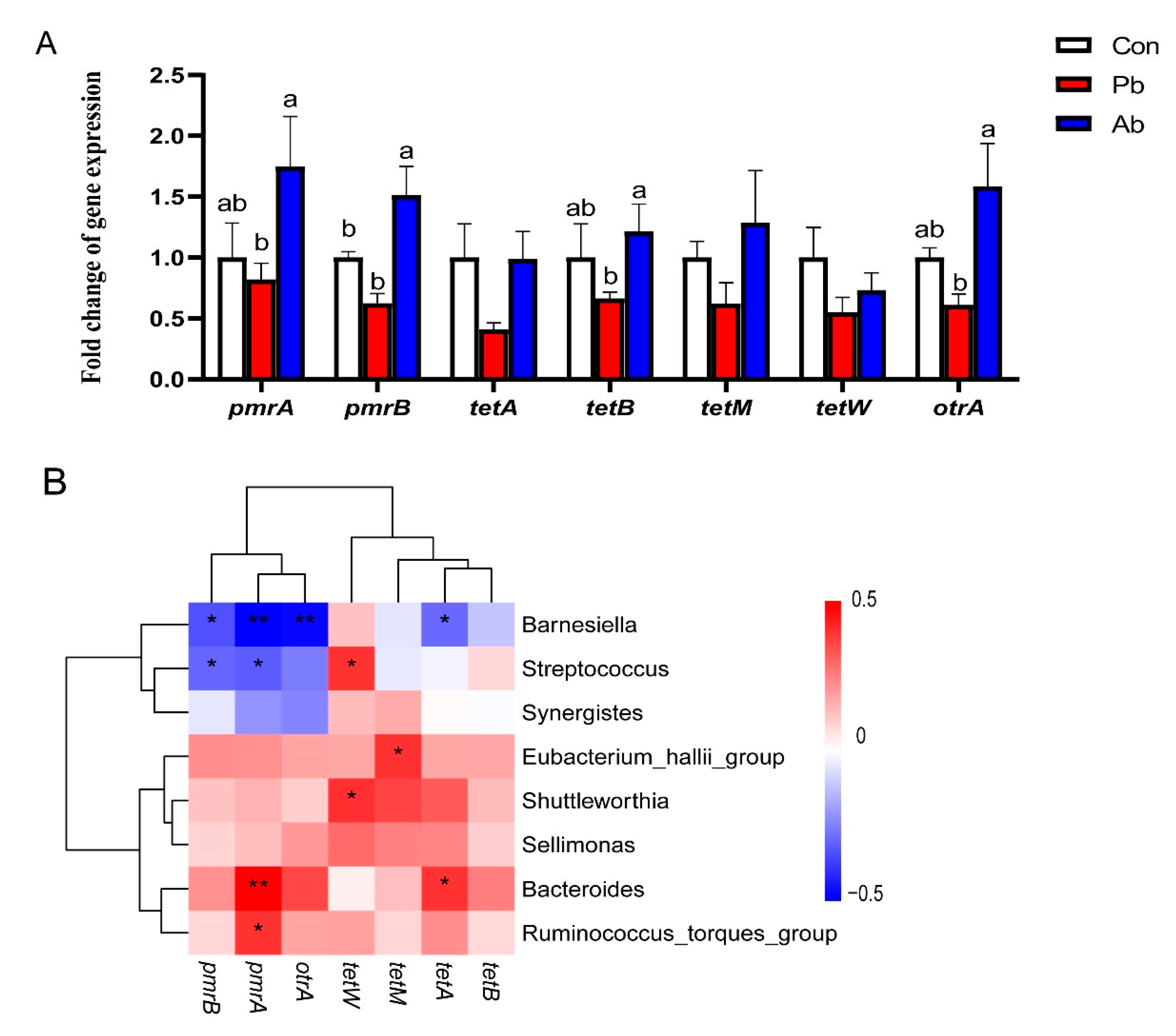
3.6. The Expression of Genes Related to Antibiotic Resistance
3.7. The Level of Total Nitrogen, Total Phosphorus, and Ammonia Nitrogen in Feces
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alagawany, M.; Abd El-Hack, M.E.; Arif, M.; Ashour, E.A. Individual and combined effects of crude protein, methionine, and probiotic levels on laying hen productive performance and nitrogen pollution in the manure. Environ. Sci. Pollut. Res. Int. 2016, 23, 22906–22913. [Google Scholar] [CrossRef] [PubMed]
- Popova, T. Effect of probiotics in poultry for improving meat quality. Curr. Opin. Food Sci. 2017, 14, 72–77. [Google Scholar] [CrossRef]
- Clavijo, V.; Florez, M.J.V. The gastrointestinal microbiome and its association with the control of pathogens in broiler chicken production: A review. Poult. Sci. 2018, 97, 1006–1021. [Google Scholar] [CrossRef] [PubMed]
- Ritzi, M.M.; Abdelrahman, W.; Mohnl, M.; Dalloul, R.A. Effects of probiotics and application methods on performance and response of broiler chickens to an Eimeria challenge. Poult. Sci. 2014, 93, 2772–2778. [Google Scholar] [CrossRef]
- Broadway, P.R.; Carroll, J.A.; Sanchez, N.C. Live Yeast and Yeast Cell Wall Supplements Enhance Immune Function and Performance in Food-Producing Livestock: A Review. Microorganisms 2015, 3, 417–427. [Google Scholar] [CrossRef]
- Zeng, X.; Li, Q.; Yang, C.; Yu, Y.; Fu, Z.; Wang, H.; Fan, X.; Yue, M.; Xu, Y. Effects of Clostridium butyricum- and Bacillus spp.-Based Potential Probiotics on the Growth Performance, Intestinal Morphology, Immune Responses, and Caecal Microbiota in Broilers. Antibiotics 2021, 10, 624. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, K.; Zhang, A.; Chang, W.; Liu, G. Effects of Lactobacillus acidophilus on the Growth Performance, Immune Response, and Intestinal Barrier Function of Broiler Chickens Challenged with Escherichia coli O78. Poult. Sci. 2021, 100, 101323. [Google Scholar] [CrossRef]
- Misra, A.K.; Kuila, R.K. Use of Bifidobacterium bifidum in the manufacture of bifidus milk and its antibacterial activity. Dairy Sci. Technol. 1992, 72, 213–220. [Google Scholar] [CrossRef][Green Version]
- Vol, N. Effect of administration of bifidobacteria and lactic acid bacteria to newborn calves and piglets. J. Dairy Sci. 1995, 78, 2838–2846. [Google Scholar]
- Ozawa, K.; Yabu-Uchi, K.; Yamanaka, K.; Yamashita, Y.; Oku, I. Effect of Streptococcus faecalis BIO-4R on intestinal flora of weanling piglets and calves. Appl. Environ. Microbiol. 1983, 45, 1513. [Google Scholar] [CrossRef]
- Talafantova, M.; Mandel, L. Protective activity of Streptococcus faecalis against pathogenic action of Escherichia coli O55 in gnotobiotic pigs. Folia Microbiol. 1985, 30, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Bilal, R.M.; Hassan, F.U.; Saeed, M.; Rafeeq, M.; Zahra, N.; Fraz, A.; Saeed, S.; Khan, M.A.; Mahgoub, H.A.M.; Farag, M.R.; et al. Role of Yeast and Yeast-Derived Products as Feed Additives in Broiler Nutrition. Anim. Biotechnol. 2023, 34, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Council, N.R. Nutrient Requirements of Poultry: 1994; National Academies Press: Washington, DC, USA, 1994. [Google Scholar]
- Qiu, K.; Qin, C.F.; Luo, M.; Zhang, X.; Sun, W.J.; Jiao, N.; de Li, F.; Yin, J.D. Protein Restriction with Amino Acid-Balanced Diets Shrinks Circulating Pool Size of Amino Acid by Decreasing Expression of Specific Transporters in the Small Intestine. PLoS ONE 2016, 11, e0162475. [Google Scholar] [CrossRef]
- Haas, B.J.; Gevers, D.; Earl, A.M.; Feldgarden, M.; Ward, D.V.; Giannoukos, G.; Ciulla, D.; Tabbaa, D.; Highlander, S.K.; Sodergren, E.; et al. Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res. 2011, 21, 494–504. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996. [Google Scholar] [CrossRef]
- Sjofjan, O.; Adli, D.N.; Harahap, R.P.; Jayanegara, A.; Utama, D.T.; Seruni, A.P. The effects of lactic acid bacteria and yeasts as probiotics on the growth performance, relative organ weight, blood parameters, and immune responses of broiler: A meta-analysis. F1000Research 2021, 10, 183. [Google Scholar] [CrossRef] [PubMed]
- Nematbakhsh, S.; Pei Pei, C.; Selamat, J.; Nordin, N.; Idris, L.H.; Abdull Razis, A.F. Molecular Regulation of Lipogenesis, Adipogenesis and Fat Deposition in Chicken. Genes 2021, 12, 414. [Google Scholar] [CrossRef]
- Liu, W.; Liu, Y.; Fang, S.; Yao, W.; Wang, X.; Bao, Y.; Shi, W. Salvia miltiorrhiza polysaccharides alleviates florfenicol-induced liver metabolic disorder in chicks by regulating drug and amino acid metabolic signaling pathways. Poult. Sci. 2022, 101, 101989. [Google Scholar] [CrossRef]
- El Aidy, S.; van den Bogert, B.; Kleerebezem, M. The small intestine microbiota, nutritional modulation and relevance for health. Curr. Opin. Biotechnol. 2015, 32, 14–20. [Google Scholar] [CrossRef]
- Dunsford, B.R.; Knabe, D.A.; Haensly, W.E. Effect of dietary soybean meal on the microscopic anatomy of the small intestine in the early-weaned pig. J. Anim. Sci. 1989, 67, 1855–1863. [Google Scholar] [CrossRef]
- Qiu, K.; Li, C.L.; Wang, J.; Qi, G.H.; Gao, J.; Zhang, H.J.; Wu, S.G. Effects of Dietary Supplementation With Bacillus subtilis, as an Alternative to Antibiotics, on Growth Performance, Serum Immunity, and Intestinal Health in Broiler Chickens. Front. Nutr. 2021, 8, 786878. [Google Scholar] [CrossRef] [PubMed]
- Sittipo, P.; Pham, H.Q.; Park, C.E.; Kang, G.U.; Zhi, Y.; Ji, H.J.; Jang, A.; Seo, H.S.; Shin, J.H.; Lee, Y.K. Irradiation-Induced Intestinal Damage Is Recovered by the Indigenous Gut Bacteria Lactobacillus acidophilus. Front. Cell. Infect. Microbiol. 2020, 10, 415. [Google Scholar] [CrossRef] [PubMed]
- Michaudel, C.; Sokol, H. The Gut Microbiota at the Service of Immunometabolism. Cell Metab. 2020, 32, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Tilocca, B.; Burbach, K.; Heyer, C.M.E.; Hoelzle, L.E.; Mosenthin, R.; Stefanski, V.; Camarinha-Silva, A.; Seifert, J. Dietary changes in nutritional studies shape the structural and functional composition of the pigs’ fecal microbiome-from days to weeks. Microbiome 2017, 5, 144. [Google Scholar] [CrossRef]
- Parker, B.J.; Wearsch, P.A.; Veloo, A.C.M.; Rodriguez-Palacios, A. The Genus Alistipes: Gut Bacteria With Emerging Implications to Inflammation, Cancer, and Mental Health. Front. Immunol. 2020, 11, 906. [Google Scholar] [CrossRef]
- Roth, N.; Kasbohrer, A.; Mayrhofer, S.; Zitz, U.; Hofacre, C.; Domig, K.J. The application of antibiotics in broiler production and the resulting antibiotic resistance in Escherichia coli: A global overview. Poult. Sci. 2019, 98, 1791–1804. [Google Scholar] [CrossRef]
- Diarra, M.S.; Malouin, F. Antibiotics in Canadian poultry productions and anticipated alternatives. Front. Microbiol. 2014, 5, 282. [Google Scholar] [CrossRef]
- Wexler, H.M. Bacteroides: The good, the bad, and the nitty-gritty. Clin. Microbiol. Rev. 2007, 20, 593–621. [Google Scholar] [CrossRef]
- Salyers, A.A.; Gupta, A.; Wang, Y. Human intestinal bacteria as reservoirs for antibiotic resistance genes. Trends Microbiol. 2004, 12, 412–416. [Google Scholar] [CrossRef]
- Gunn, J.S.; Miller, S.I. PhoP-PhoQ activates transcription of pmrAB, encoding a two-component regulatory system involved in Salmonella typhimurium antimicrobial peptide resistance. J. Bacteriol. 1996, 178, 6857–6864. [Google Scholar] [CrossRef]
- Chopra, I.; Roberts, M. Tetracycline antibiotics: Mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef] [PubMed]
- Presley, L.L.; Wei, B.; Braun, J.; Borneman, J. Bacteria associated with immunoregulatory cells in mice. Appl. Env. Microbiol. 2010, 76, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Ubeda, C.; Bucci, V.; Caballero, S.; Djukovic, A.; Toussaint, N.C.; Equinda, M.; Lipuma, L.; Ling, L.; Gobourne, A.; No, D.; et al. Intestinal microbiota containing Barnesiella species cures vancomycin-resistant Enterococcus faecium colonization. Infect. Immun. 2013, 81, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Gong, L.; Huang, K.; Li, F.; Tong, D.; Zhang, H. Effect of Heat-Inactivated Compound Probiotics on Growth Performance, Plasma Biochemical Indices, and Cecal Microbiome in Yellow-Feathered Broilers. Front. Microbiol. 2020, 11, 585623. [Google Scholar] [CrossRef]
- Kaur, U.J.; Preet, S.; Rishi, P. Augmented antibiotic resistance associated with cadmium induced alterations in Salmonella enterica serovar Typhi. Sci. Rep. 2018, 8, 12818. [Google Scholar] [CrossRef]
- Parsons, C.M. Determination of digestible and available amino acids in meat meal using conventional and caecectomized cockerels or chick growth assays. Br. J. Nutr. 1986, 56, 227–240. [Google Scholar] [CrossRef]
- Fen, W.; Rajput, L.; Rashid, I.; Xu, X.; Li, Y. Effects of Probiotic (Bacillus subtilis) on Laying Performance, Blood Biochemical Properties and Intestinal Microflora of Shaoxing Duck. Int. J. Poult. Sci. 2011, 10, 583–589. [Google Scholar]
- Abd El-Hack, M.E.; Mahgoub, S.A.; Alagawany, M.; Ashour, E.A. Improving productive performance and mitigating harmful emissions from laying hen excreta via feeding on graded levels of corn DDGS with or without Bacillus subtilis probiotic. J. Anim. Physiol. Anim. Nutr. 2017, 101, 904–913. [Google Scholar] [CrossRef]
| Ages (Day) | ||
|---|---|---|
| Items | 1–21 | 22–42 |
| Ingredients (%) | ||
| Corn | 56.90 | 59.66 |
| Soybean meal | 33.00 | 32.20 |
| Fish meal | 3.00 | 0 |
| Limestone | 1.20 | 1.15 |
| Dicalcium phosphate | 1.45 | 1.65 |
| Soybean oil | 3.00 | 3.90 |
| L-methionine | 0.15 | 0.14 |
| Sodium chloride | 0.30 | 0.30 |
| Vitamin-mineral premix * | 1 | 1 |
| Nutritional composition (%) | ||
| Crude protein | 20.98 | 18.83 |
| Total lysine | 1.17 | 1.00 |
| Total methionine and cysteine | 0.80 | 0.72 |
| Calcium | 1.08 | 0.88 |
| Nonphytate phosphorus | 0.45 | 0.40 |
| Total phosphorus | 0.68 | 0.66 |
| Metabolic energy (Mcal/kg) | 2.96 | 3.00 |
| Gene | Primer Sequences (5′-3′) |
|---|---|
| pmrA | F: AGTTTTCCTCATTCGCGACCA |
| R: TACCAGGCTGCGGATGATATTCT | |
| pmrB | F: GGATGGCCTGATGTGACGCTGTC |
| R: GCGCGGCTTTGGCTATATGCTG | |
| tetA | F: TTGGCATTCTGCATTCACTC |
| R: GTATAGCTTGCCGGAAGTCG | |
| tetB | F: AGTGCGCTTTGGATGCTGTA |
| R: AGCCCCAGTAGCTCCTGTGA | |
| tetM | F: ACAGAAAGCTTATTATATAAC |
| R: TGGCGTGTCTATGATGTTCAC | |
| tetW | F: GAGAGCCTGCTATATGCCAGC |
| R: GGGCGTATCCACAATGTTAAC | |
| otrA | F: GGCATYCTGGCCCACGT |
| R: CCCGGGGTGTCGTASAGG |
| Items, % | Con | Pb | Ab | |
|---|---|---|---|---|
| Liver | D21 | 2.59 ± 0.12 | 2.64 ± 0.10 | 2.57 ± 0.14 |
| D42 | 2.79 ± 0.35 b | 1.95 ± 0.05 a | 1.91 ± 0.06 a | |
| Breast muscle | D21 | 4.58 ± 0.11 | 4.89 ± 0.07 | 4.90 ± 0.13 |
| D42 | 8.37 ± 0.21 | 9.49 ± 0.56 | 9.21 ± 0.35 | |
| Abdominal fat | D21 | 1.17 ± 0.09 ab | 0.96 ± 0.03 a | 1.33 ± 0.86 b |
| D42 | 2.56 ± 0.10 b | 1.54 ± 0.13 a | 1.80 ± 0.12 a | |
| Bursa of Fabricius | D21 | 0.24 ± 0.03 | 0.23 ± 0.03 | 0.26 ± 0.02 |
| D42 | 0.24 ± 0.03 | 0.23 ± 0.02 | 0.21 ± 0.01 |
| Items | Con | Pb | Ab | |
|---|---|---|---|---|
| ALT | D21 | 1.67 ± 0.88 | 1.67 ± 0.67 | 3.17 ± 1.01 |
| D42 | 3.38 ± 0.84 | 2.75 ± 0.37 | 2.14 ± 0.14 | |
| AST | D21 | 205.67 ± 6.15 | 233.67 ± 10.67 | 225.50 ± 9.31 |
| D42 | 223.88 ± 16.42 | 228.25 ± 8.53 | 218.00 ± 6.82 | |
| GLU | D21 | 13.19 ± 0.38 | 12.37 ± 0.27 | 13.26 ± 0.36 |
| D42 | 12.20 ± 0.25 a | 12.03 ± 0.24 a | 15.34 ± 1.52 b | |
| TG | D21 | 0.47 ± 0.03 a | 0.60 ± 0.02 b | 0.57 ± 0.06 ab |
| D42 | 0.54 ± 0.03 | 0.45 ± 0.03 | 0.49 ± 0.05 | |
| CHOL | D21 | 3.40 ± 0.15 | 3.70 ± 0.14 | 3.70 ± 0.18 |
| D42 | 3.70 ± 0.10 | 3.69 ± 0.08 | 3.85 ± 0.09 | |
| HDL | D21 | 2.60 ± 0.12 | 2.82 ± 0.08 | 2.92 ± 0.12 |
| D42 | 2.29 ± 0.08 | 2.45 ± 0.07 | 2.47 ± 0.09 | |
| LDL | D21 | 0.53 ± 0.06 | 0.71 ± 0.60 | 0.66 ± 0.09 |
| D42 | 0.91 ± 0.12 | 0.74 ± 0.04 | 0.80 ± 0.08 | |
| ALB | D21 | 12.87 ± 0.27 a | 13.88 ± 0.41 b | 14.17 ± 0.08 b |
| D42 | 13.44 ± 0.47 | 13.79 ± 0.21 | 13.77 ± 0.24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, Y.; Wu, X.; Hu, D.; Wang, C.; Chen, Q.; Ni, Y. Comparison of the Effects of Feeding Compound Probiotics and Antibiotics on Growth Performance, Gut Microbiota, and Small Intestine Morphology in Yellow-Feather Broilers. Microorganisms 2023, 11, 2308. https://doi.org/10.3390/microorganisms11092308
Feng Y, Wu X, Hu D, Wang C, Chen Q, Ni Y. Comparison of the Effects of Feeding Compound Probiotics and Antibiotics on Growth Performance, Gut Microbiota, and Small Intestine Morphology in Yellow-Feather Broilers. Microorganisms. 2023; 11(9):2308. https://doi.org/10.3390/microorganisms11092308
Chicago/Turabian StyleFeng, Yuyan, Xiaoting Wu, Dan Hu, Canyang Wang, Qu Chen, and Yingdong Ni. 2023. "Comparison of the Effects of Feeding Compound Probiotics and Antibiotics on Growth Performance, Gut Microbiota, and Small Intestine Morphology in Yellow-Feather Broilers" Microorganisms 11, no. 9: 2308. https://doi.org/10.3390/microorganisms11092308
APA StyleFeng, Y., Wu, X., Hu, D., Wang, C., Chen, Q., & Ni, Y. (2023). Comparison of the Effects of Feeding Compound Probiotics and Antibiotics on Growth Performance, Gut Microbiota, and Small Intestine Morphology in Yellow-Feather Broilers. Microorganisms, 11(9), 2308. https://doi.org/10.3390/microorganisms11092308





