Studies Using Mutant Strains of Azospirillum brasilense Reveal That Atmospheric Nitrogen Fixation and Auxin Production Are Light Dependent Processes
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacteria Growth
2.2. Plant Growth
2.3. Acetylene Reduction Assay (ARA)
2.4. Microbial 59Fe Uptake and Metabolic Transformation
2.5. Luciferase Chemiluminescence ATP Assay
2.6. Spectrophotometric Auxin Assay
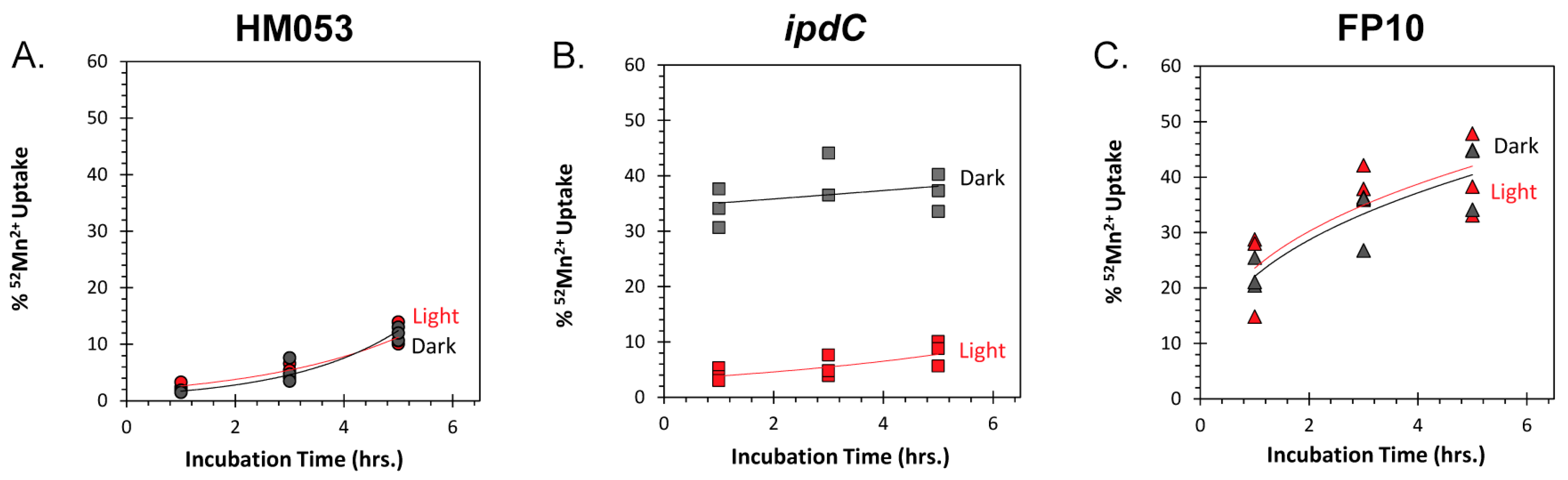
2.7. Bacteria 52Mn Uptake
2.8. Statistical Analysis
3. Results and Discussion
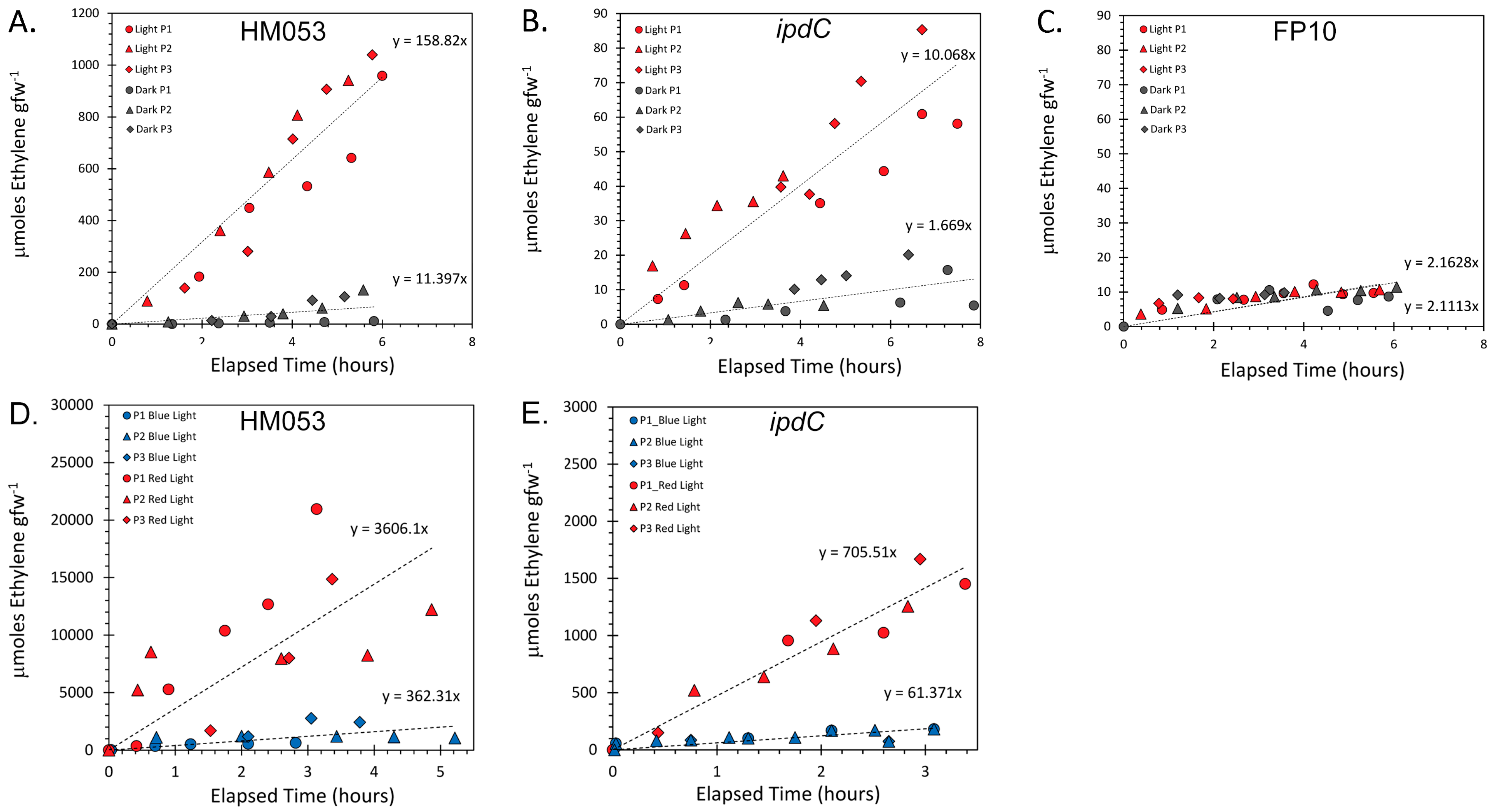
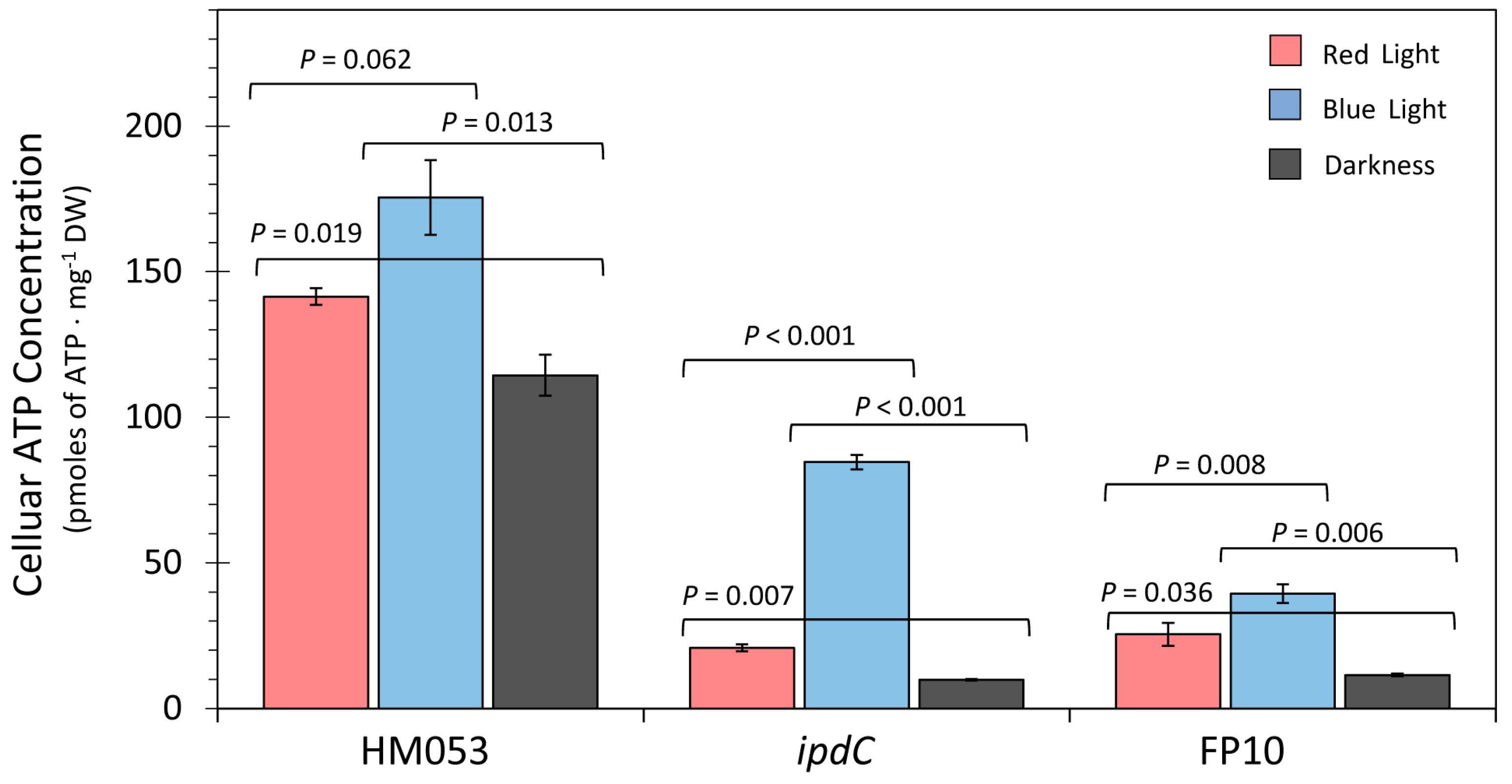
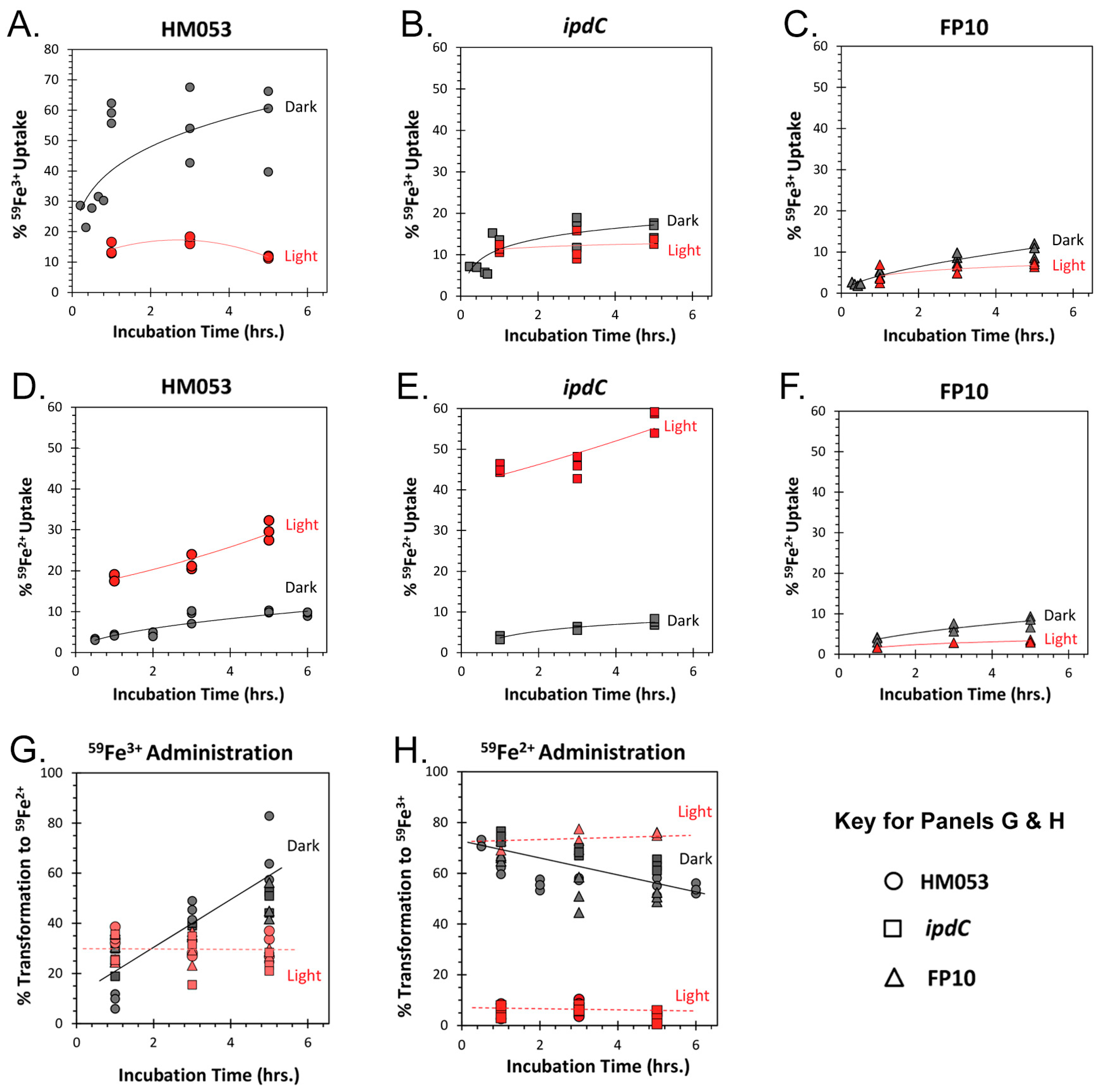
3.1. Light Dependencies of BNF and Its Supporting Processes in A. brasilense
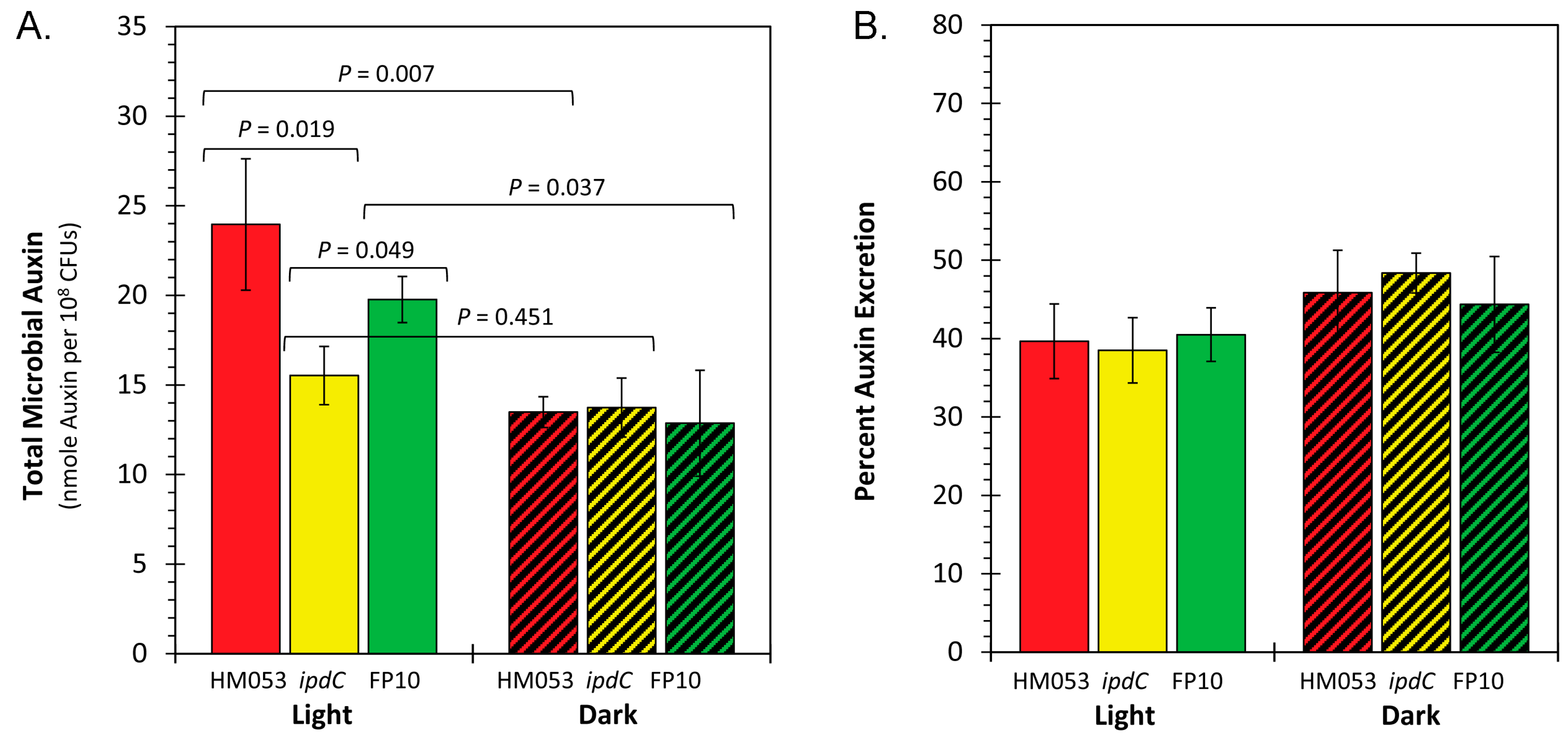
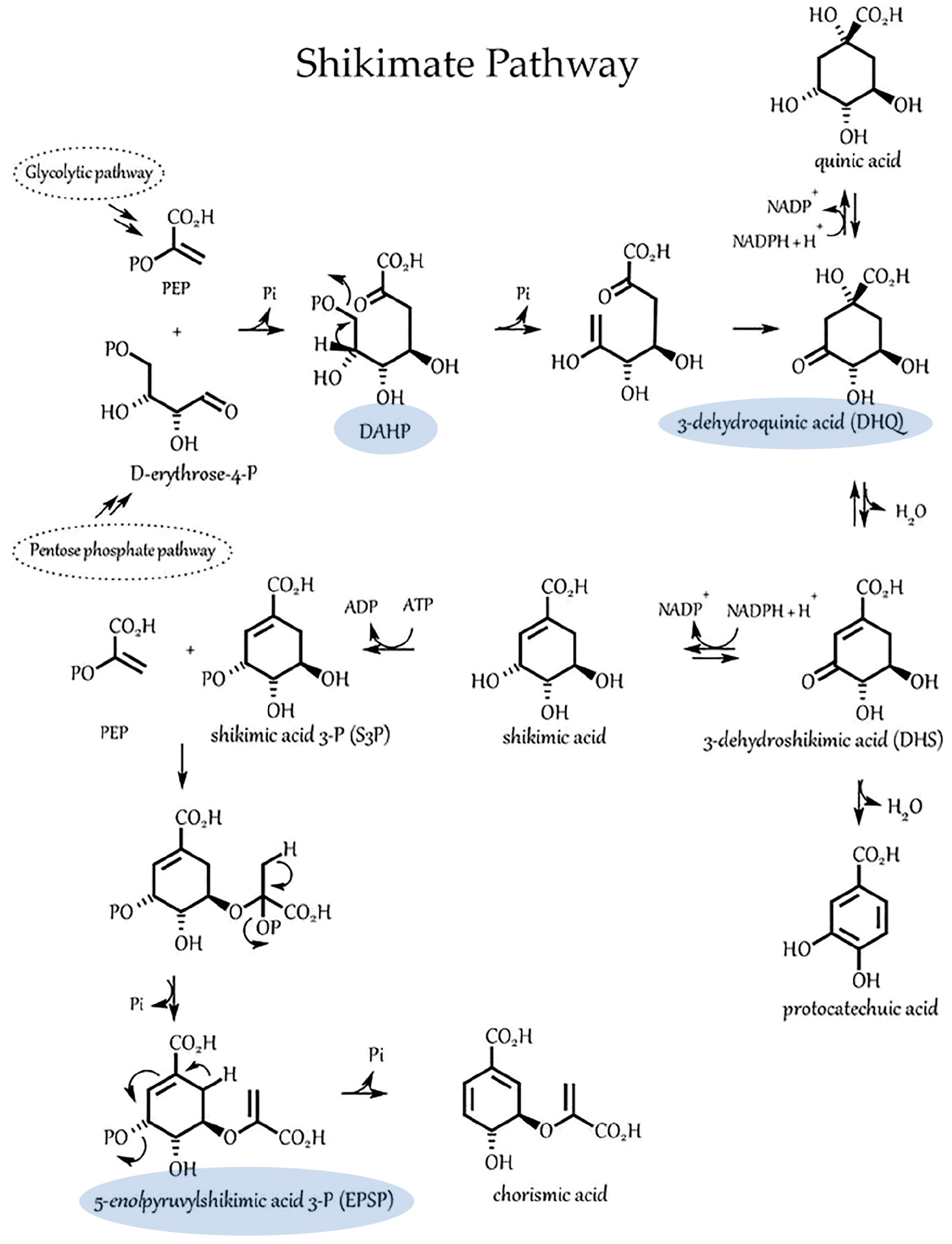
3.2. Light Dependencies of Auxin Biosynthesis and Its Supporting Processes in A. brasilense
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumar, S.; Kateriya, S.; Singh, V.S.; Tanwar, M.; Agarwal, S.; Singh, H.; Khurana, J.P.; Amla, D.V.; Tripathi, A.K. Bacteriophytochrome controls carotenoid-independent response to photodynamic stress in a non-photosynthetic rhizobacterium, Azospirillum brasilense Sp7. Sci. Rep. 2012, 2, 872. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Ha, J.H.; Kim, S.G.; Choi, H.K.; Kim, Z.H.; Han, Y.-J.; Kim, J.-I.; Oh, Y.; Fragoso, V.; Shin, K.; et al. Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots. Sci. Signal. 2016, 9, ra106. [Google Scholar] [CrossRef] [PubMed]
- van Gelderen, K.; Kang, C.; Pierik, R. Light Signaling, Root Development, and Plasticity. Plant Physiol. 2018, 176, 1049–1060. [Google Scholar] [CrossRef]
- Kakuszi, A.; Sárvári, É.; Solti, Á.; Czégény, G.; Hideg, É.; Hunyadi-Gulyás, É.; Bóka, K.; Böddi, B. Light piping driven photosynthesis in the soil: Low-light adapted active photosynthetic apparatus in the under-soil hypocotyl segments of bean (Phaseolus vulgaris). J. Photochem. Photobiol. B Biol. 2016, 161, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Waller, S.; Wilder, S.L.; Schueller, M.J.; Ferrieri, R.A. Plants use the suberin biopolymer to conduct light. Polymers 2022, 14, 5387. [Google Scholar] [CrossRef]
- Sun, Q.; Yoda, K.; Suzuki, H. Internal axial light conduction in the stems and roots of herbaceous plants. J. Exp. Bot. 2005, 56, 191–203. [Google Scholar] [CrossRef]
- Okon, Y. Azospirillum/Plant Associations; CRC Press: Boca Raton, FL, USA, 1993; 192p. [Google Scholar]
- Okon, Y.; Vanderleyden, J. Root-associated Azospirillum species can stimulate plants. ASM News 1997, 63, 366–370. [Google Scholar]
- Steenhoudt, O.; Vanderleyden, J. Azospirillum, a free-living nitrogen-fixing bacterium closely associated with grasses: Genetic, biochemical, and ecological aspects. FEMS Microbiol. Rev. 2000, 24, 487–506. [Google Scholar] [CrossRef]
- Josiane Fukami1, J.; Paula Cerezini, P.; Hungria, M. Azospirillum: Benefits that go far beyond biological nitrogen fixation. AMB Express 2018, 8, 73–85. [Google Scholar] [CrossRef]
- Cassán, F.; Coniglio, A.; López, G.; Molina, R.; Nievas, S.; Le Noir de Carlan, C.; Donadio, F.; Torres, D.; Rosas, S.; Pedrosa, F.O.; et al. Everything you must know about Azospirillum and its impact on agriculture and beyond. Biol. Fertil. Soils 2020, 56, 461–479. [Google Scholar] [CrossRef]
- Aloo, B.N.; Tripathi, V.; Makumba, B.A.; Mbega, E.R. Plant growth-promoting rhizobacterial biofertilizers for crop production: The past, present, and future. Front. Plant Sci. 2022, 13, 1002448. [Google Scholar] [CrossRef]
- Cruz-Hernández, M.A.; Mendoza-Herrera, A.; Bocanegra-García, V.; Rivera, G. Azospirillum spp. from plant growth-promoting bacteria to their use in bioremediation. Microorganisms 2022, 10, 1057–1068. [Google Scholar] [CrossRef]
- James, E.; Baldani, J. The role of biological nitrogen fixation by non-legumes in the sustainable production of food and biofuels. Plant Soil 2012, 356, 1–3. [Google Scholar] [CrossRef]
- Richardson, A.; Barea, J.; Mcneill, A.; Prigent-Combaret, C.; Richardson, A.E.; Barea, J.M.; McNeill, A.M.; Prigent-Combaret, C. Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant Soil 2009, 321, 305–339. [Google Scholar] [CrossRef]
- Pankievicz, V.C.S.; do Amaral, F.P.; Santos, K.F.D.N.; Agtuca, B.; Xu, Y.; Schueller, M.J.; Arisi, A.C.M.; Steffens, M.B.R.; de Souza, E.M.; Pedrosa, F.O.; et al. Robust biological nitrogen fixation in a model grass–bacterial association. Plant J. 2015, 81, 907–919. [Google Scholar] [CrossRef]
- Okon, Y.; Labandera-Gonzalez, C.A. Agronomic applications of Azospirillum: An evaluation of 20 years worldwide field inoculation. Soil Biol. Biochem. 1994, 26, 1591–1601. [Google Scholar] [CrossRef]
- Pedraza, R.O.; Bellone, C.H.; Carrizo de Bellone, S.; Boa Sorte, P.M.F.; Teixeira, K. Azospirillum inoculation and nitrogen fertilization effect on grain yield and on the diversity of endophytic bacteria in the phyllosphere of rice rainfed crop. Eur. J. Soil Biol. 2009, 45, 36–43. [Google Scholar] [CrossRef]
- Housh, A.B.; Powell, G.; Scott, S.; Anstaett, A.; Gerheart, A.; Benoit, M.; Waller, S.; Powell, A.; Guthrie, J.M.; Higgins, B.; et al. Functional mutants of Azospirillum brasilense elicit beneficial physiological and metabolic responses in Zea mays contributing to increased host iron assimilation. ISME J. 2021, 15, 1505–1522. [Google Scholar] [CrossRef]
- Dobbelaere, S.; Croonenborghs, A.; Thys, A.; Ptacek, D.; Vanderleyden, J.; Dutto, P.; Labandera-Gonzalez, C.; Caballero-Mellado, J.; Aguirre, J.F.; Kapulnik, Y.; et al. Responses of agronomically important crops to inoculation with Azospirillum. Funct. Plant Biol. 2001, 28, 871–879. [Google Scholar] [CrossRef]
- Suhameena, B.; Devi, S.; Gowri, R.; Kumar, S.D. Utilization of Azospirillum as a biofertilizer—An overview. Int. J. Pharm. Sci. Rev. Res. 2020, 62, 141–145. [Google Scholar]
- Giraud, E.; Lavergne, J.; Verméglio, A. Chapter 9—Characterization of Bacteriophytochromes from Photosynthetic Bacteria: Histidine Kinase Signaling Triggered by Light and Redox Sensing. Methods Enzymol. 2010, 471, 135–159. [Google Scholar] [PubMed]
- Carithers, R.P.; Yoch, D.C.; Arnon, D.I. Two Forms of Nitrogenase from the Photosynthetic Bacterium Rhodospirillum rubrum. J. Bacteriol. 1979, 137, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Molina, R.; López, G.; Rodríquez, B.; Rosas, S.; Mora, V.; Cassán, F. Evaluation of growth and motility in non-photosynthetic Azospirillum brasilense exposed to red, blue, and white light. Arch. Microbiol. 2020, 202, 1193–1201. [Google Scholar] [CrossRef]
- Machado, H.B.; Funayama, S.; Rigo, L.U.; Pedrosa, F.O. Excretion of ammonium by Azospirillum brasilense mutants resistant to ethylenediamine. Can. J. Microbiol. 1991, 37, 549–553. [Google Scholar] [CrossRef]
- Santos, A.R.S.; Etto, R.M.; Furmam, R.W.; Freitas, D.L.; de Santos, K.F.; d’Eça, N.; de Souza, E.M.; Pedrosa, F.O.; Ayub, R.A.; Steffens, M.B.R.; et al. Labeled Azospirillum brasilense wild type and excretion-ammonium strains in association with barley roots. Plant Physiol. Biochem. 2017, 118, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Näsvall, J.; Knöppel, A.; Andersson, D.I. Duplication-Insertion Recombineering: A fast and scar-free method for efficient transfer of multiple mutations in bacteria. Nucleic Acids Res. 2017, 45, e3. [Google Scholar] [CrossRef]
- Pedrosa, F.O.; Yates, M.G. Regulation of nitrogen fixation (nif) genes of Azospirillum brasilense by nifA and ntr (gln) type gene products. FEMS Microbiol. Lett. 1984, 23, 95–101. [Google Scholar] [CrossRef]
- Lamb, A.B.; Jacques, A.O. The slow hydrolysis of ferric chloride in dilute solution. II. The change in hydrogen-ion concentration. J. Am. Chem. Soc. 1938, 60, 967–981. [Google Scholar] [CrossRef]
- Mempin, R.; Tran, H.; Chen, C.; Gong, H.; Ho, K.K.; Lu, S. Release of extracellular ATP by bacteria during growth. BMC Microbiol. 2013, 13, 301–314. [Google Scholar] [CrossRef]
- Yang, N.-C.; Ho, W.-M.; Chen, Y.-H.; Hu, M.-L. A convenient one-step extraction of cellular ATP using boiling water for the Luciferin-Luciferase assay of ATP. Anal. Biochem. 2003, 306, 323–327. [Google Scholar] [CrossRef]
- Ehmann, A. The van Urk-Salkowski reagent—A sensitive and specific chromogenic reagent for silica gel thin-layer chromatographic detection and identification of indole derivatives. J. Chromatogr. 1977, A132, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, A.-L.; Hong, Y.-K.; Shin, J.-H.; Joo, S.-H. A highly efficient auxin-producing bacterial strain and its effect on plant growth. J. Genet. Eng. Biotechnol. 2021, 19, 179–188. [Google Scholar] [CrossRef]
- Graves, S.A.; Hernandez, R.; Fonslet, J.; England, C.G.; Valdovinos, H.F.; Ellison, P.A.; Barnhart, T.E.; Elema, D.R.; Theuer, C.P.; Cai, W.; et al. Novel preparation methods of Mn-52 for immunoPET imaging. Bioconjugate Chem. 2015, 26, 2118–2124. [Google Scholar] [CrossRef] [PubMed]
- Inomura, K.; Deutsch, C.; Wilson, S.T.; Masuda, T.; Lawrenz, E.; Bučinská, L.; Sobotka, R.; Gauglitz, J.M.; Saito, M.A.; Prášil, O.; et al. Quantifying oxygen management and temperature and light dependencies of nitrogen fixation by Crocosphaera watsonii. MSphere 2019, 4, e00531-19. [Google Scholar] [CrossRef] [PubMed]
- Fixen, K.R.; Zheng, Y.; Harris, D.F.; Shaw, S.; Yang, Z.Y.; Dean, D.R.; Seefeldt, L.C.; Harwood, C.S. Light-driven carbon dioxide reduction to methane by nitrogenase in a photosynthetic bacterium. Proc. Natl. Acad. Sci. USA 2016, 113, 10163–10167. [Google Scholar] [CrossRef]
- Zhang, Y.; Burris, R.H.; Ludden, P.W.; Roberts, G.P. Regulation of nitrogen fixation in Azospirillum brasilense. FEMS Microbiol. Lett. 1997, 152, 195–204. [Google Scholar] [CrossRef]
- Vekshin, N.L. Light-dependent ATP synthesis in mitochondria. Biochem. Int. 1991, 25, 603–611. [Google Scholar]
- Fu, H.A.; Hartmann, A.; Lowery, R.G.; Fitzmaurice, W.P.; Roberts, G.P.; Burris, R.H. Posttranslational regulatory system for nitrogenase activity in Azospirillum spp. J. Bacteriol. 1989, 171, 4679–4685. [Google Scholar] [CrossRef]
- Klassen, G.; de Souza, E.M.; Yates, M.G.; Rigo, L.U.; Inaba, J.; Pedrosa, F.O. Control of nitrogenase reactivation by the GlnZ protein in Azospirillum brasilense. J. Bacteriol. 2001, 183, 6710–6713. [Google Scholar] [CrossRef]
- Huergo, L.F.; Chubatsu, L.S.; de Souza, E.M.; Pedrosa, F.O.; Steffens, M.B.R.; Merrick, M. Interactions between PII proteins and the nitrogenase regulatory enzymes DraT and DraG in Azospirillum brasilense. FEBS Lett. 2006, 580, 5232–5236. [Google Scholar] [CrossRef]
- Wenke, B.B.; Spatzal, T.; Rees, D.C. Site-specific oxidation state assignments of the iron atoms in the [4Fe:4S]2+/1+/0 states of the nitrogenase Fe-protein. Angewandte Chemie. Int. Ed. 2019, 58, 3894–3897. [Google Scholar] [CrossRef]
- Bacchawat, A.K.; Ghosh, S. Iron transport in Azospirillum brasilense: Role of the siderophore spirilobactin. J. Gen. Microbiol. 1987, 133, 1759–1765. [Google Scholar]
- Mori, E.; Fulchieri, M.; Indorato, C.; Fani, R.; Bazzicalupo, M. Cloning, nucleotide sequencing, and expression of Azospirillum brasilense ion gene: Involvement in iron uptake. J. Bacteriol. 1996, 178, 3440–3446. [Google Scholar] [CrossRef]
- Kamnev, A.A.; Renou-Gonnord, M.-F.; Antonyuk, L.P.; Colina, M.; Chernyshev, A.V.; Frolov, I.; Ignatov, V.V. Spectroscopic characterization of the uptake of essential and xenobiotic metal cations in cells of the soil bacterium Azospirillum brasilense. Biochem. Mol. Biol. Int. 1997, 41, 123–130. [Google Scholar] [CrossRef]
- Barton, L.L.; Johnson, G.V.; Bishop, Y.M. The metabolism of iron by nitrogen-fixing rhizospheric bacteria. In Iron Nutrition in Plants and Rhizospheric Microorganisms; Barton, L.L., Abadia, J., Eds.; Springer: Dordrecht, The Netherlands, 2006; Chapter 9; pp. 199–214. [Google Scholar]
- Kamnev, A.A.; Tugarova, A.V. Bioanalytical applications of Mössbauer spectroscopy. Russ. Chem. Rev. 2021, 90, 1415–1453. [Google Scholar] [CrossRef]
- Kamnev, A.A.; Anna, V.; Tugarova, A.V.; Krisztina Kovács, K.; Biró, B.; Homonnay, Z.; Kuzmann, E. Mössbauer spectroscopic study of 57Fe metabolic transformations in the rhizobacterium Azospirillum brasilense Sp245. Hyperfine Interact. 2014, 226, 415–419. [Google Scholar] [CrossRef]
- Alenkina, I.V.; Oshtrakh, M.I.; Tugarova, A.V.; Biró, B.; Semionkin, V.A.; Kamnev, A.A. Study of the rhizobacterium Azospirillum brasilense Sp245 using Mössbauer spectroscopy with a high velocity resolution: Implication for the analysis of ferritin-like iron cores. J. Mol. Struct. 2014, 1073, 181–186. [Google Scholar] [CrossRef]
- Kamnev, A.A.; Tugarova, A.V.; Kovács, K.; Kuzmann, E.; Homonnay, Z.; Kulikov, L.A.; Perfiliev, Y.D. Mössbauer spectroscopic study of iron and cobalt metabolic transformations in cells of the bacterium Azospirillum brasilense Sp7. Bull. Russ. Acad. Sci. Phys. 2015, 79, 1036–1040. [Google Scholar] [CrossRef]
- Kovács, K.; Kamnev, A.A.; Pechoušek, J.; Tugarova, A.V.; Kuzmann, E.; Machala, L.; Zbořil, R.; Homonnay, Z.; Lázár, K. Evidence for ferritin as dominant iron-bearing species in the rhizobacterium Azospirillum brasilense Sp7 provided by low-temperature/in-field Mössbauer spectroscopy. Anal. Bioanal. Chem. 2016, 408, 1565–1571. [Google Scholar] [CrossRef]
- Kamnev, A.A.; Tugarova, A.V.; Shchelochkov, A.G.; Kovács, K.; Kuzmann, E. Diffuse reflectance infrared Fourier transform (DRIFT) and Mössbauer spectroscopic study of Azospirillum brasilense Sp7: Evidence for intracellular iron(II) oxidation in bacterial biomass upon lyophilization. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 229, 117970. [Google Scholar] [CrossRef]
- Woodward, A.W.; Bartel, B. Auxin: Regulation, action, and interaction. Ann. Bot. 2005, 95, 707–735. [Google Scholar] [CrossRef] [PubMed]
- McSteen, P. Auxin and monocot development. Cold Spring Harb. Perspect. Biol. 2010, 2, a001479. [Google Scholar] [CrossRef] [PubMed]
- Agtuca, B.; Rieger, E.; Hilger, K.; Song, L.; Robert, C.A.M.; Erb, M.; Karve, A.; Ferrieri, R.A. Carbon-11 reveals opposing roles of auxin and salicylate in regulating leaf physiology, leaf metabolism and resource allocation patterns that impact root growth in Zea mays. J. Plant Growth Regul. 2014, 33, 328–339. [Google Scholar] [CrossRef]
- Herrmann, K.M.; Weaver, L.M. The shikimate pathway. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 473–503. [Google Scholar] [CrossRef]
- Bender, S.L.; Mehdi, S.; Knowles, J.R. Dehydroquinate synthase: The role of divalent metal cations and of nicotinamide adenine dinucleotide in catalysis. Biochemistry 1989, 28, 7555–7560. [Google Scholar] [CrossRef]
- Schneider, D.A.; Gourse, R.L. Relationship between growth rate and ATP concentration in Eschericha coli. J. Biol. Chem. 2004, 279, 8262–8268. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Housh, A.B.; Noel, R.; Powell, A.; Waller, S.; Wilder, S.L.; Sopko, S.; Benoit, M.; Powell, G.; Schueller, M.J.; Ferrieri, R.A. Studies Using Mutant Strains of Azospirillum brasilense Reveal That Atmospheric Nitrogen Fixation and Auxin Production Are Light Dependent Processes. Microorganisms 2023, 11, 1727. https://doi.org/10.3390/microorganisms11071727
Housh AB, Noel R, Powell A, Waller S, Wilder SL, Sopko S, Benoit M, Powell G, Schueller MJ, Ferrieri RA. Studies Using Mutant Strains of Azospirillum brasilense Reveal That Atmospheric Nitrogen Fixation and Auxin Production Are Light Dependent Processes. Microorganisms. 2023; 11(7):1727. https://doi.org/10.3390/microorganisms11071727
Chicago/Turabian StyleHoush, Alexandra Bauer, Randi Noel, Avery Powell, Spenser Waller, Stacy L. Wilder, Stephanie Sopko, Mary Benoit, Garren Powell, Michael J. Schueller, and Richard A. Ferrieri. 2023. "Studies Using Mutant Strains of Azospirillum brasilense Reveal That Atmospheric Nitrogen Fixation and Auxin Production Are Light Dependent Processes" Microorganisms 11, no. 7: 1727. https://doi.org/10.3390/microorganisms11071727
APA StyleHoush, A. B., Noel, R., Powell, A., Waller, S., Wilder, S. L., Sopko, S., Benoit, M., Powell, G., Schueller, M. J., & Ferrieri, R. A. (2023). Studies Using Mutant Strains of Azospirillum brasilense Reveal That Atmospheric Nitrogen Fixation and Auxin Production Are Light Dependent Processes. Microorganisms, 11(7), 1727. https://doi.org/10.3390/microorganisms11071727







