Spatial and Temporal Variability in Prevalence Rates of Members of the Borrelia burgdorferi Species Complex in Ixodes ricinus Ticks in Urban, Agricultural and Sylvatic Habitats in Slovakia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Tick Collections
2.2. DNA Isolation
2.3. PCR Amplification and Molecular Analysis
2.4. Identification of Borrelia Species in Samples with Mixed Infections
2.4.1. Sequencing
2.4.2. Reverse Line Blotting (RLB)
2.4.3. Cloning and Transformation of the Competent Cells
2.4.4. RFLP Analysis
2.5. Statistical Analysis
3. Results
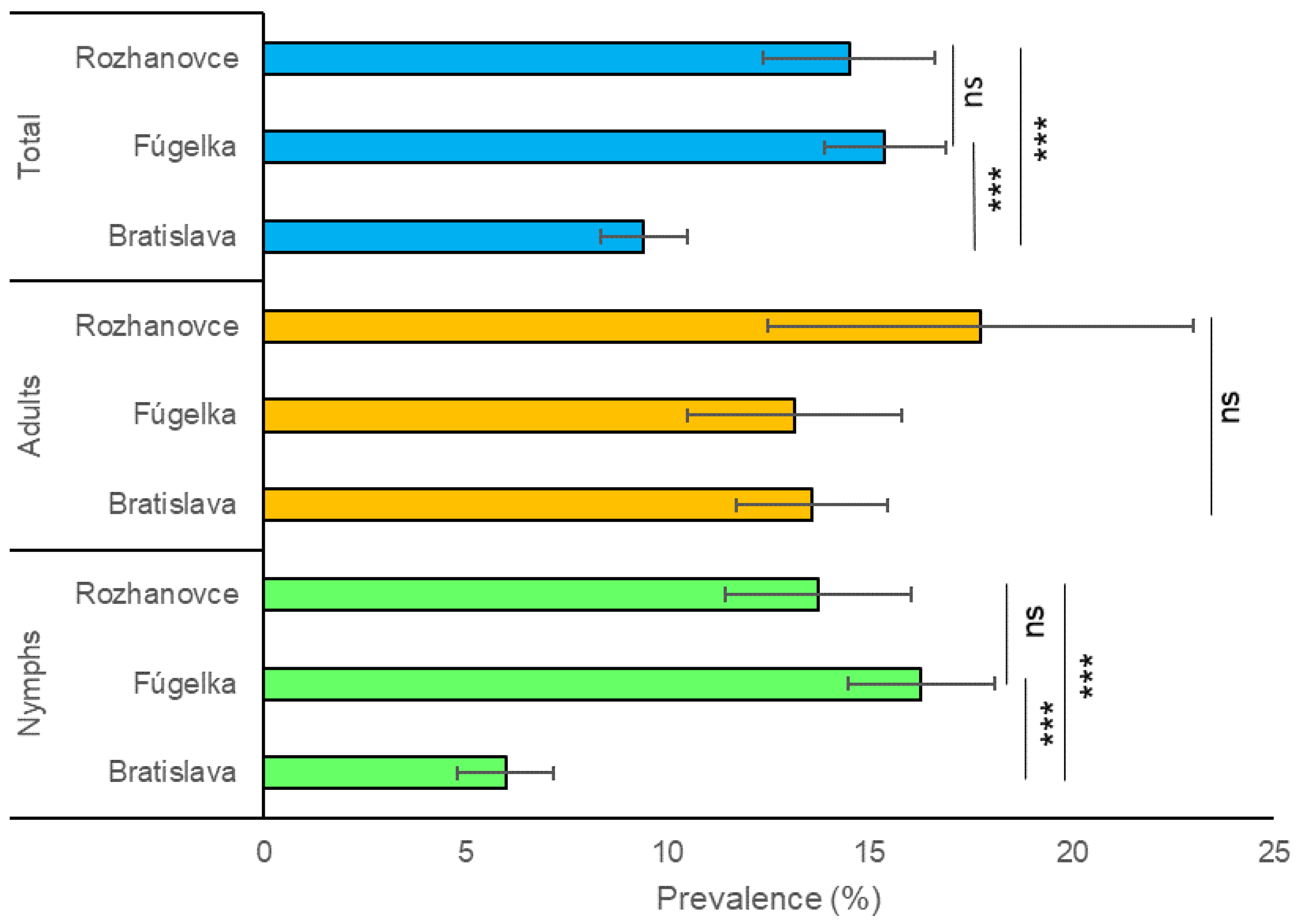
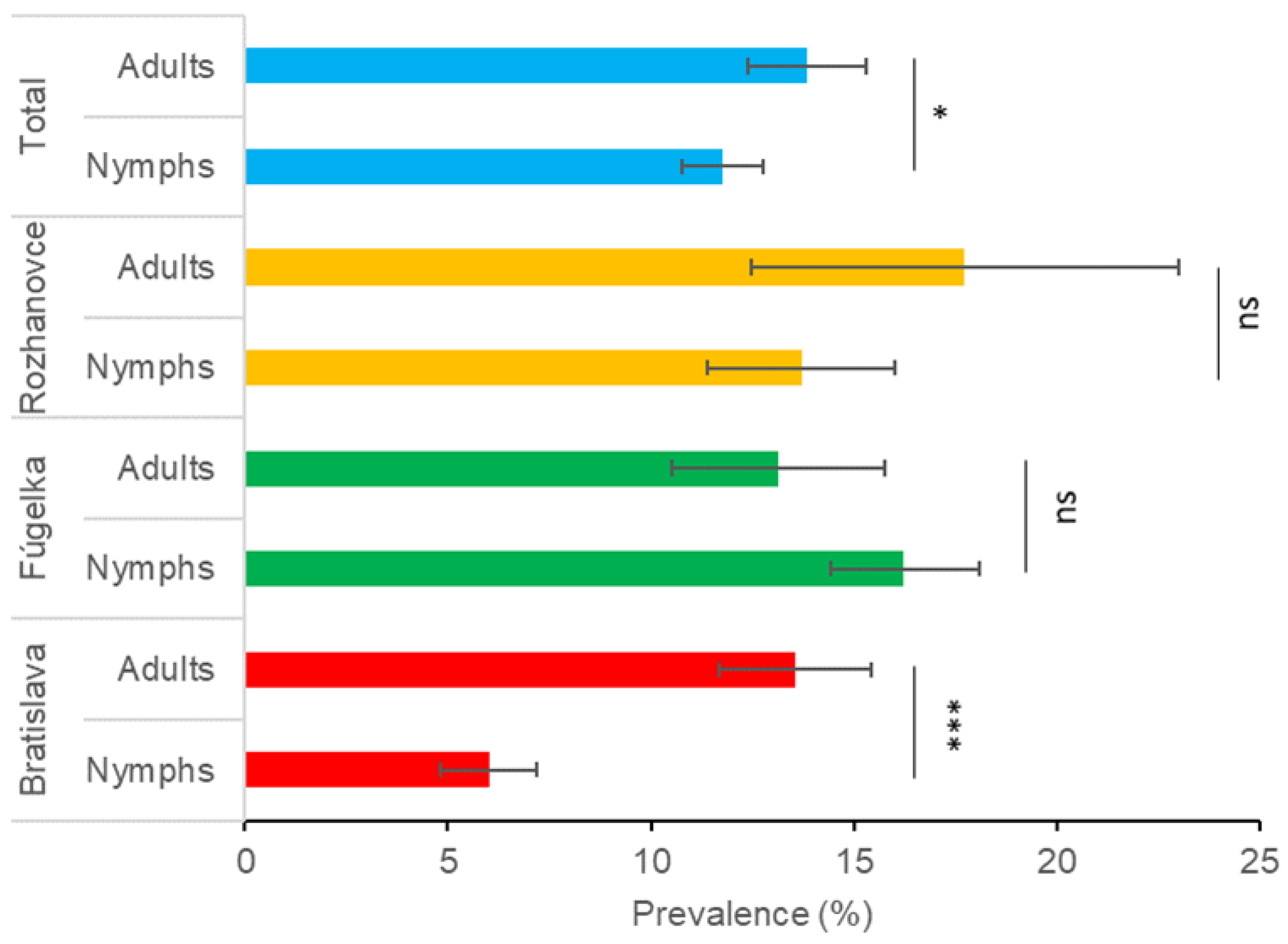
3.1. Prevalence of Borrelia burgdorferi s.l. in Questing Ixodes ricinus
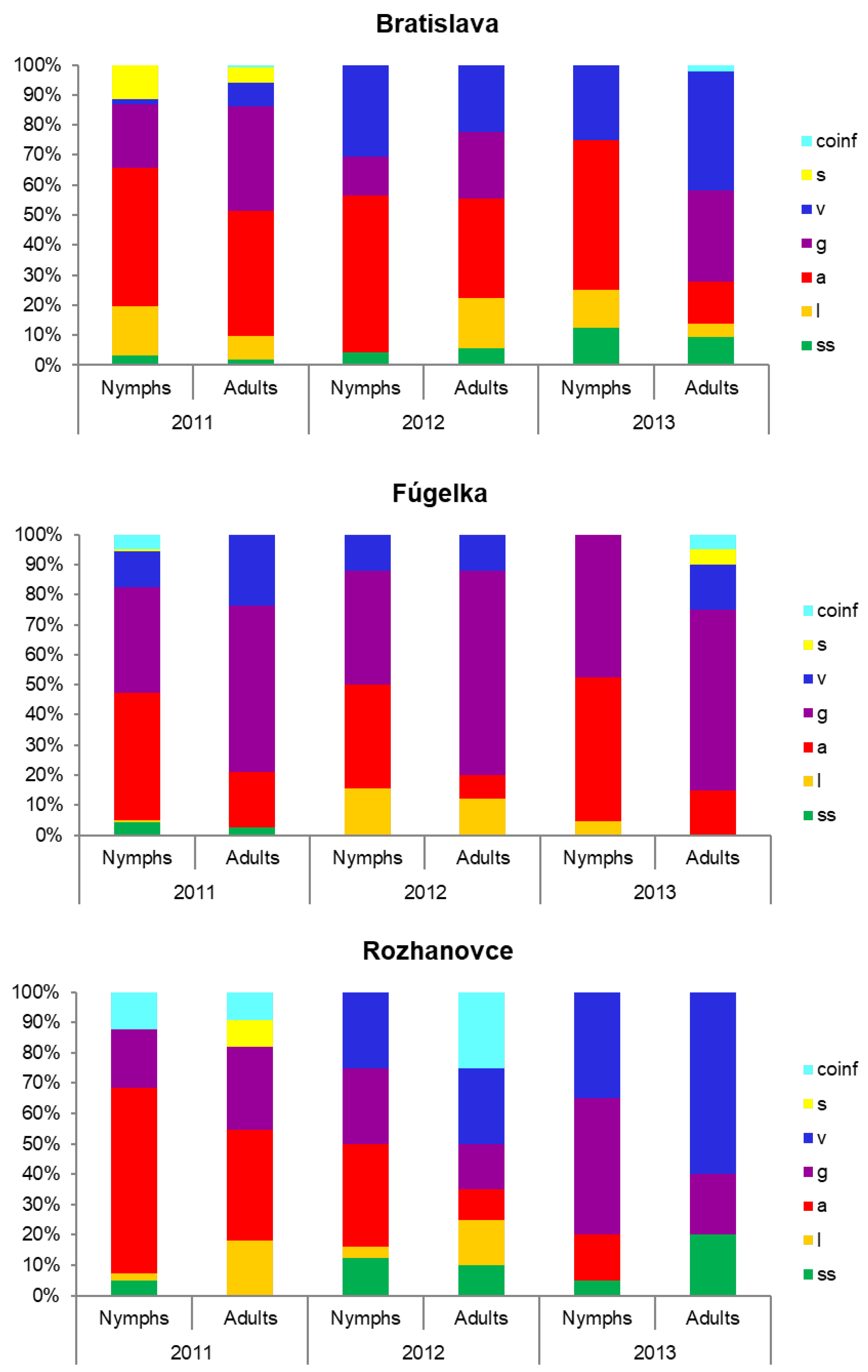
3.2. Occurrence of Borrelia burgdorferi s.l. Species
3.3. Variability in Infection Prevalence and Distribution of Borrelia burgdorferi s.l. Species in Bratislava
3.4. Borrelia burgdorferi s.l. Species in Mixed Infections
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rizzoli, A.; Hauffe, H.C.; Carpi, G.; Vourc’h, G.I.; Neteler, M.; Rosà, R. Lyme borreliosis in Europe. Euro Surveill. 2011, 16, 19906. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Updates on Borrelia burgdorferi sensu lato complex with respect to public health. Ticks Tick Borne Dis. 2011, 2, 123–128. [Google Scholar] [CrossRef]
- Steinbrink, A.; Brugger, K.; Margos, G.; Kraiczy, P.; Klimpel, S. The evolving story of Borrelia burgdorferi sensu lato transmission in Europe. Parasitol. Res. 2022, 121, 781–803. [Google Scholar] [CrossRef] [PubMed]
- Wodecka, B.; Kolomiiets, V. Genetic diversity of Borreliaceae species detected in natural populations of Ixodes ricinus ticks in Northern Poland. Life 2023, 13, 972. [Google Scholar] [CrossRef] [PubMed]
- Wodecka, B.; Michalik, J.; Grochowalska, R. Red foxes (Vulpes vulpes) are exposed to high diversity of Borrelia burgdorferi sensu lato species infecting fox-derived Ixodes ticks in west-central Poland. Pathogens 2022, 11, 696. [Google Scholar] [CrossRef] [PubMed]
- Majerová, K.; Hönig, V.; Houda, M.; Papežík, P.; Fonville, M.; Sprong, H.; Rudenko, N.; Golovchenko, M.; Černá Bolfíková, B.; Hulva, P.; et al. Hedgehogs, squirrels, and blackbirds as sentinel hosts for active surveillance of Borrelia miyamotoi and Borrelia burgdorferi complex in urban and rural environments. Microorganisms 2020, 8, 1908. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M. Sexual transmission of Lyme borreliosis? The question that calls for an answer. Trop. Med. Infect. Dis. 2021, 6, 87. [Google Scholar] [CrossRef]
- Stanek, G.; Reiter, M. The expanding Lyme Borrelia complex-clinical significance of genomic species? Clin. Microbiol. Infect. 2011, 17, 487–493. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Ortega, C.; Sánchez, N.; Desimone, L.; Sudre, B.; Suk, J.E.; Semenza, J.C. Correlation of Borrelia burgdorferi sensu lato prevalence in questing Ixodes ricinus ticks with specific abiotic traits in the western Palearctic. Appl. Environ. Microbiol. 2011, 77, 3838–3845. [Google Scholar] [CrossRef]
- Strnad, M.; Honig, V.; Růžek, D.; Grubhoffer, L.; Rego, R.O.M. Europe-wide meta-analysis of Borrelia burgdorferi sensu lato prevalence in questing Ixodes ricinus ticks. Appl. Environ. Microbiol. 2017, 83, e00609-17. [Google Scholar] [CrossRef]
- Kurtenbach, K.; De Michelis, S.; Etti, S.; Schäfer, S.M.; Sewell, H.S.; Brade, V.; Kraiczy, P. Host association of Borrelia burgdorferi sensu lato–the key role of host complement. Trends Microbiol. 2002, 10, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Kurtenbach, K.; Hanincová, K.; Tsao, J.I.; Margos, G.; Fish, D.; Ogden, N.H. Fundamental processes in the evolutionary ecology of Lyme borreliosis. Nat. Rev. Microbiol. 2006, 4, 660–669. [Google Scholar] [CrossRef]
- O’Keeffe, K.R.; Oppler, Z.J.; Brisson, D. Evolutionary ecology of Lyme Borrelia. Infect. Genet. Evol. 2020, 85, 104570. [Google Scholar] [CrossRef] [PubMed]
- Hofmeester, T.R.; Coipan, E.C.; van Wieren, S.E.; Prins, H.H.T.; Takken, W.; Sprong, H. Few vertebrate species dominate the Borrelia burgdorferi s.l. life cycle. Environ. Res. Lett. 2016, 11, 043001. [Google Scholar] [CrossRef]
- Heylen, D.; Lasters, R.; Adriaensen, F.; Fonville, M.; Sprong, H.; Matthysen, E. Ticks and tick-borne diseases in the city: Role of landscape connectivity and green space characteristics in a metropolitan area. Sci. Total Environ. 2019, 670, 941–949. [Google Scholar] [CrossRef]
- Gern, L. Borrelia burgdorferi sensu lato, the agent of Lyme borreliosis: Life in the wilds. Parasite 2008, 15, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Humair, P.F.; Gern, L. The wild hidden face of Lyme borreliosis in Europe. Microbes Infect. 2000, 2, 915–922. [Google Scholar] [CrossRef]
- Hanincová, K.; Schäfer, S.M.; Etti, S.; Sewell, H.S.; Tarageľová, V.; Žiak, D.; Labuda, M.; Kurtenbach, K. Association of Borrelia afzelii with rodents in Europe. Parasitology 2003, 126, 11–20. [Google Scholar] [CrossRef]
- Humair, P.F.; Gern, L. Relationship between Borrelia burgdorferi sensu lato species, red squirrels (Sciurus vulgaris) and Ixodes ricinus in enzootic areas in Switzerland. Acta Trop. 1998, 69, 213–227. [Google Scholar] [CrossRef]
- Hanincová, K.; Tarageľová, V.; Koči, J.; Schäfer, S.M.; Hails, R.; Ullmann, A.J.; Piesman, J.; Labuda, M.; Kurtenbach, K. Association of Borrelia garinii and B. valaisiana with songbirds in Slovakia. Appl. Environ. Microbiol. 2003, 69, 2825–2830. [Google Scholar] [CrossRef]
- Tarageľová, V.; Koči, J.; Hanincová, K.; Kurtenbach, K.; Derdáková, M.; Ogden, N.H.; Literák, I.; Kocianová, E.; Labuda, M. Blackbirds and song thrushes constitute a key reservoir of Borrelia garinii, the causative agent of borreliosis in central Europe. Appl. Environ. Microbiol. 2008, 74, 1289–1293. [Google Scholar] [CrossRef] [PubMed]
- Richter, D.; Matuschka, F.R. Perpetuation of the Lyme disease spirochete Borrelia lusitaniae by lizards. Appl. Environ. Microbiol. 2006, 72, 4627–4632. [Google Scholar] [CrossRef] [PubMed]
- Majláthová, V.; Majláth, I.; Víchová, B.; Derdáková, M.; Peťko, B. Borrelia lusitaniae and green lizards (Lacerta viridis), Karst Region, Slovakia. Emerg. Infect. Dis. 2006, 12, 1895–1901. [Google Scholar] [CrossRef] [PubMed]
- Richter, D.; Schlee, D.B.; Allgöwer, R.; Matuschka, F.-R. Relationships of a novel Lyme disease spirochete, Borrelia spielmanii sp. nov., with its hosts in Central Europe. Appl. Environ. Microbiol. 2004, 70, 6414–6419. [Google Scholar] [CrossRef]
- Durand, J.; Herrmann, C.; Genné, D.; Sarr, A.; Gern, L.; Voordouw, M.J. Multistrain infections with Lyme borreliosis pathogens in the tick vector. Appl. Environ. Microbiol. 2017, 83, e02552-16. [Google Scholar] [CrossRef]
- Rudenko, N.; Grubhoffer, L.; Golovchenko, M. Climate Impact on Lyme Borreliosis and Its Causative Agents. In Climate, Ticks and Disease; Nuttall, P., Ed.; University of Oxford: Oxford, UK, 2021; pp. 349–354. [Google Scholar]
- Hanincova, K.; Kurtenbach, K.; Diuk-Wasser, M.; Brei, B.; Fish, D. Epidemic spread of Lyme borreliosis, Northeastern United States. Emerg. Infect. Dis. 2006, 12, 604–611. [Google Scholar] [CrossRef]
- Černý, J.; Lynn, G.; Hrnková, J.; Golovchenko, M.; Rudenko, N.; Grubhoffer, L. Management options for Ixodes ricinus-associated pathogens: A review of prevention strategies. Int. J. Environ. Res. Public Health 2020, 17, 1830. [Google Scholar] [CrossRef]
- Kahl, O.; Gray, J.S. The biology of Ixodes ricinus with emphasis on its ecology. Ticks Tick Borne Dis. 2023, 14, 102114. [Google Scholar] [CrossRef]
- Rizzoli, A.; Silaghi, C.; Obiegala, A.; Rudolf, I.; Hubálek, Z.; Földvári, G.; Plantard, O.; Vayssier-Taussat, M.; Bonnet, S.; Špitalská, E.; et al. Ixodes ricinus and its transmitted pathogens in urban and peri-urban areas in Europe: New hazards and relevance for public health. Front. Publ. Health 2014, 2, 251. [Google Scholar] [CrossRef]
- Gandy, S.; Kilbride, E.; Biek, R.; Millins, C.; Gilbert, L. Experimental evidence for opposing effects of high deer density on tick-borne pathogen prevalence and hazard. Parasit. Vectors 2021, 14, 509. [Google Scholar] [CrossRef]
- Stanko, M.; Derdáková, M.; Špitalská, E.; Kazimírová, M. Ticks and their epidemiological role in Slovakia: From the past till present. Biologia 2022, 77, 575–1610. [Google Scholar] [CrossRef] [PubMed]
- Kmety, E.; Rehácek, J.; Výrosteková, V. Investigations of ticks for the presence of Borrelia in Czechoslovakia. Zentralbl. Bakteriol. Mikrobiol. Hyg. A 1987, 263, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Drgoňová, M.; Řeháček, J. Prevalence of Lyme borrelia in ticks in Bratislava, Slovak Republic. Cent. Eur. J. Public Health 1995, 3, 134–137. [Google Scholar]
- Gern, L.; Hu, C.M.; Kocianova, E.; Vyrostekova, V.; Rehacek, J. Genetic diversity of Borrelia burgdorferi sensu lato isolates obtained from Ixodes ricinus ticks collected in Slovakia. Eur. J. Epidemiol. 1999, 15, 665–669. [Google Scholar] [CrossRef]
- Štepánová-Tresová, G.; Peťko, B.; Štefančíková, A.; Nadzamová, D. Occurrence of Borrelia burgdorferi sensu stricto, Borrelia garinii and Borrelia afzelii in the Ixodes ricinus ticks from Eastern Slovakia. Eur. J. Epidemiol. 2000, 16, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Lenčáková, D.; Hizo-Teufel, C.; Petko, B.; Schulte-Spechtel, U.; Stanko, M.; Wilske, B.; Fingerle, V. Prevalence of Borrelia burgdorferi s.l. OspA types in Ixodes ricinus ticks from selected localities in Slovakia and Poland. Int. J. Med. Microbiol. 2006, 296 (Suppl. S40), 108–118. [Google Scholar] [CrossRef]
- Pangrácová, L.; Derdáková, M.; Pekárik, L.; Hviščová, I.; Víchová, B.; Stanko, M.; Hlavatá, H.; Peťko, B. Ixodes ricinus abundance and its infection with the tick-borne pathogens in urban and suburban areas of Eastern Slovakia. Parasit. Vectors 2013, 6, 238. [Google Scholar] [CrossRef] [PubMed]
- Rusňáková Tarageľová, V.; Mahríková, L.; Selyemová, D.; Václav, R.; Derdáková, M. Natural foci of Borrelia lusitaniae in a mountain region of Central Europe. Ticks Tick Borne Dis. 2016, 7, 350–356. [Google Scholar] [CrossRef]
- Chvostáč, M.; Špitalská, E.; Václav, R.; Vaculová, T.; Minichová, L.; Derdáková, M. Seasonal patterns in the prevalence and diversity of tick-borne Borrelia burgdorferi sensu lato, Anaplasma phagocytophilum and Rickettsia spp. in an urban temperate forest in south western Slovakia. Int. J. Environ. Res. Public Health 2018, 15, 994. [Google Scholar] [CrossRef]
- Vaculová, T.; Derdáková, M.; Špitalská, E.; Václav, R.; Chvostáč, M.; Rusňáková Tarageľová, V. Simultaneous occurrence of Borrelia miyamotoi, Borrelia burgdorferi sensu lato, Anaplasma phagocytophilum and Rickettsia helvetica in Ixodes ricinus ticks in urban foci in Bratislava, Slovakia. Acta Parasitol. 2019, 64, 19–30. [Google Scholar] [CrossRef]
- Mtierová, Z.; Derdáková, M.; Chvostáč, M.; Didyk, Y.M.; Mangová, B.; Rusňáková Tarageľová, V.; Selyemová, D.; Šujanová, A.; Václav, R. Local population structure and seasonal variability of Borrelia garinii genotypes in Ixodes ricinus ticks, Slovakia. Int. J. Environ. Res. Public Health. 2020, 17, 3607. [Google Scholar] [CrossRef]
- Subramanian, G.; Sekeyova, Z.; Raoult, D.; Mediannikov, O. Multiple tick-associated bacteria in Ixodes ricinus from Slovakia. Ticks Tick Borne Dis. 2012, 3, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Hamšíková, Z.; Coipan, C.; Mahríková, L.; Minichová, L.; Sprong, H.; Kazimírová, M. Borrelia miyamotoi and co-infection with Borrelia afzelii in Ixodes ricinus ticks and rodents from Slovakia. Microb. Ecol. 2017, 73, 1000–1008. [Google Scholar] [CrossRef] [PubMed]
- Rosà, R.; Andreo, V.; Tagliapietra, V.; Baráková, I.; Arnoldi, D.; Hauffe, H.C.; Manica, M.; Rosso, F.; Blaňarová, L.; Bona, M.; et al. Effect of climate and land use on the spatio-temporal variability of tick-borne bacteria in Europe. Int. J. Environ. Res. Public Health 2018, 15, 732. [Google Scholar] [CrossRef]
- Svitálková, Z.; Haruštiaková, D.; Mahríková, L.; Berthová, L.; Slovák, M.; Kocianová, E.; Kazimírová, M. Anaplasma phagocytophilum prevalence in ticks and rodents in an urban and natural habitat in South-Western Slovakia. Parasit. Vectors 2015, 8, 276. [Google Scholar] [CrossRef] [PubMed]
- Kazimírová, M.; Hamšíková, Z.; Kocianová, E.; Marini, G.; Mojšová, M.; Mahríková, L.; Berthová, L.; Slovák, M.; Rosá, R. Relative density of host-seeking ticks in different habitat types of south-western Slovakia. Exp. Appl. Acarol. 2016, 69, 205–224. [Google Scholar] [CrossRef]
- Špitalská, E.; Stanko, M.; Mošanský, L.; Kraljik, J.; Miklisová, D.; Mahríková, L.; Bona, M.; Kazimírová, M. Sesonal analysis of Rickettsia species in ticks in an agricultural site of Slovakia. Exp. Appl. Acarol. 2016, 68, 315–324. [Google Scholar] [CrossRef]
- Stanko, M.; Mošanský, L.; Fričová, J.; Čisláková, L.; Schniererová, E. Long-Time Study of Natural Focus and Its Structure in Eastern Slovakia. In Stawonogi Roznorodnosc Form i Oddzialywan; Buczek, A., Blaszak, C., Eds.; Koliber: Lublin, Poland, 2005; pp. 113–119. [Google Scholar]
- Siuda, K. Kleszcze Polski (Acari: Ixodida). Część II. Systematyka i Rozmieszczenie. In Monografie Parazytologiczne; Polskie Towarzystwo Parazytologiczne: Warszawa, Poland, 1993; pp. 1–375. [Google Scholar]
- Filippova, N.A. Fauna of the USSR: Arachnida, Subfamily Ixodinae 4(4); Nauka Publisher: Leningrad, Russia, 1977. [Google Scholar]
- Mydlová, L.; Svitálková, Z.; Slovák, M.; Kazimírová, M. Variability in Prevalence and Distribution of Borrelia burgdorferi s.l. In Ixodes ricinus Ticks across Bratislava Forest Park and Adjacent Sites (SW Slovakia). In Zborník z Konferencie III. Labudove dni; Kocianová, E., Kazimírová, M., Lúčanová, V., Eds.; Virologický ústav SAV: Bratislava, Slovakia, 2013; pp. 26–29. [Google Scholar]
- Derdáková, M.; Beati, L.; Peťko, B.; Stanko, M.; Fish, D. Genetic variability within Borrelia burgdorferi sensu lato genospecies established by PCR-single-strand conformation polymorphism analysis of the RrfA-RrlB intergenic spacer in Ixodes ricinus ticks from the Czech Republic. Appl. Environ. Microbiol. 2003, 69, 509–516. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Borrelia carolinensis sp. nov., a new (14th) member of the Borrelia burgdorferi sensu lato complex from the southeastern region of the United States. J. Clin. Microbiol. 2009, 47, 134–141. [Google Scholar] [CrossRef]
- Bunikis, J.; Garpmo, U.; Tsao, J.; Berglund, J.; Fish, D.; Barbour, A.G. Sequence typing reveals extensive strain diversity of the Lyme borreliosis agents Borrelia burgdorferi in North America and Borrelia afzelii in Europe. Microbiology 2004, 150, 1741–1755. [Google Scholar] [CrossRef]
- Clark, K.; Hendricks, A.; Burge, D. Molecular identification and analysis of Borrelia burgdorferi sensu lato in lizards in the southeastern United States. Appl. Environ. Microbiol. 2005, 71, 2616–2625. [Google Scholar] [CrossRef] [PubMed]
- Alekseev, A.N.; Dubinina, H.V.; Van De Pol, I.; Schouls, L.M. Identification of Ehrlichia spp. and Borrelia burgdorferi in Ixodes ticks in the Baltic regions of Russia. J. Clin. Microbiol. 2001, 39, 2237–2242. [Google Scholar] [CrossRef]
- Blazejak, K.; Raulf, M.K.; Janecek, E.; Jordan, D.; Fingerle, V.; Strube, C. Shifts in Borrelia burgdorferi (s.l.) geno-species infections in Ixodes ricinus over a 10-year surveillance period in the city of Hanover (Germany) and Borrelia miyamotoi-specific Reverse Line Blot detection. Parasit. Vectors 2018, 11, 304. [Google Scholar] [CrossRef] [PubMed]
- Gern, L.; Douet, V.; López, Z.; Rais, O.; Cadenas, F.M. Diversity of Borrelia genospecies in Ixodes ricinus ticks in a Lyme borreliosis endemic area in Switzerland identified by using new probes for reverse line blotting. Ticks Tick Borne Dis. 2010, 1, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Springer, A.; Jordan, D.; Glass, A.; Kahl, O.; Fingerle, V.; Girl, P.; Chitimia-Dobler, L.; Strube, C. Borrelia infections in ageing ticks: Relationship with morphometric age ratio in field-collected Ixodes ricinus nymphs. Microorganisms 2022, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, M.; Okada, K.; Nakao, M.; Konishi, T.; Sato, Y. Phylogenetic analysis of Borrelia species based on flagellin gene sequences and its application for molecular typing of Lyme disease borreliae. Int. J. Syst. Bacteriol. 1996, 46, 898–905. [Google Scholar] [CrossRef] [PubMed]
- Postic, D.; Assous, M.V.; Grimont, P.A.; Baranton, G. 1994. Diversity of Borrelia burgdorferi sensu lato evidenced by restriction fragment length polymorphism of rrf (5S)-rrl (23S) intergenic spacer amplicons. Int. J. Syst. Bacteriol. 1994, 44, 743–752. [Google Scholar] [CrossRef]
- Wang, G.; van Dam, A.P.; Schwartz, I.; Dankert, J. 1999. Molecular typing of Borrelia burgdorferi sensu lato: Taxonomic, epidemiological, and clinical implications. Clin. Microbiol. Rev. 1999, 2, 633–653. [Google Scholar] [CrossRef]
- Hammer, O.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Jouda, F.; Perret, J.L.; Gern, L. Density of questing Ixodes ricinus nymphs and adults infected by Borrelia burgdorferi sensu lato in Switzerland: Spatio-temporal pattern at a regional scale. Vector Borne Zoonotic Dis. 2004, 4, 23–32. [Google Scholar] [CrossRef]
- Reye, A.L.; Hübschen, J.M.; Sausy, A.; Muller, C.P. Prevalence and seasonality of tick-borne pathogens in questing Ixodes ricinus ticks from Luxembourg. Appl. Environ. Microbiol. 2010, 76, 2923–2931. [Google Scholar] [CrossRef] [PubMed]
- Venclíková, K.; Betášová, L.; Sikutová, S.; Jedličková, P.; Hubálek, Z.; Rudolf, I. Human pathogenic borreliae in Ixodes ricinus ticks in natural and urban ecosystem (Czech Republic). Acta Parasitol. 2014, 59, 717–720. [Google Scholar] [CrossRef]
- Hönig, V.; Svec, P.; Halas, P.; Vavruskova, Z.; Tykalova, H.; Kilian, P.; Vetiskova, V.; Dornakova, V.; Sterbova, J.; Simonova, Z.; et al. Ticks and tick-borne pathogens in South Bohemia (Czech Republic)—Spatial variability in Ixodes ricinus abundance, Borrelia burgdorferi and tick-borne encephalitis virus prevalence. Ticks Tick Borne Dis. 2015, 6, 559–567. [Google Scholar] [CrossRef]
- Honig, V.; Carolan, H.E.; Vavruskova, Z.; Massire, C.; Mosel, M.R.; Crowder, C.D.; Rounds, M.A.; Ecker, D.J.; Ruzek, D.; Grubhoffer, L.; et al. Broad-range survey of vector-borne pathogens and tick host identification of Ixodes ricinus from Southern Czech Republic. FEMS Microbiol. Ecol. 2017, 93, fix129. [Google Scholar] [CrossRef]
- Raileanu, C.; Moutailler, S.; Pavel, I.; Porea, D.; Mihalca, A.D.; Savuta, G.; Vayssier-Taussat, M. Borrelia diversity and co-infection with other tick borne pathogens in ticks. Front. Cell. Infect. Microbiol. 2017, 7, 36. [Google Scholar] [CrossRef] [PubMed]
- Kubiak, K.; Dziekońska-Rynko, J.; Szymańska, H.; Kubiak, D.; Dmitryjuk, M.; Dzika, E. Questing Ixodes ricinus ticks (Acari, Ixodidae) as a vector of Borrelia burgdorferi sensu lato and Borrelia miyamotoi in an urban area of north-eastern Poland. Exp. Appl. Acarol. 2019, 78, 113–126. [Google Scholar] [CrossRef]
- Zubriková, D.; Wittmann, M.; Hönig, V.; Švec, P.; Víchová, B.; Essbauer, S.; Dobler, G.; Grubhoffer, L.; Pfister, K. Prevalence of tick-borne encephalitis virus and Borrelia burgdorferi sensu lato in Ixodes ricinus ticks in Lower Bavaria and Upper Palatinate, Germany. Ticks Tick Borne Dis. 2020, 11, 101375. [Google Scholar] [CrossRef] [PubMed]
- Hartemink, N.; van Vliet, A.J.H.; Gort, G.; Gassner, F.; Jacobs, F.; Fonville, M.; Takken, W.; Sprong, H. Seasonal patterns and spatial variation of Borrelia burgdorferi (sensu lato) infections in Ixodes ricinus in the Netherlands. Parasit. Vectors 2021, 14, 121. [Google Scholar] [CrossRef] [PubMed]
- Răileanu, C.; Silaghi, C.; Fingerle, V.; Margos, G.; Thiel, C.; Pfister, K.; Overzier, E. Borrelia burgdorferi sensu lato in questing and engorged ticks from different habitat types in southern Germany. Microorganisms 2021, 9, 1266. [Google Scholar] [CrossRef]
- Knoll, S.; Springer, A.; Hauck, D.; Schunack, B.; Pachnicke, S.; Fingerle, V.; Strube, C. Distribution of Borrelia burgdorferi s.l. and Borrelia miyamotoi in Ixodes tick populations in Northern Germany, co-infections with Rickettsiales and assessment of potential influencing factors. Med. Vet. Entomol. 2021, 35, 595–606. [Google Scholar] [CrossRef]
- Król, N.; Obiegala, A.; Imholt, C.; Arz, C.; Schmidt, E.; Jeske, K.; Ulrich, R.G.; Rentería-Solís, Z.; Jacob, J.; Pfeffer, M. Diversity of Borrelia burgdorferi sensu lato in ticks and small mammals from different habitats. Parasit. Vectors 2022, 15, 195. [Google Scholar] [CrossRef]
- Rauter, C.; Hartung, T. Prevalence of Borrelia burgdorferi sensu lato genospecies in Ixodes ricinus ticks in Europe: A metaanalysis. Appl. Environ. Microbiol. 2005, 71, 7203–7216. [Google Scholar] [CrossRef] [PubMed]
- Trávniček, M.; Guryčová, D.; Halánová, M.; Kožuch, O.; Nadzamová, D.; Miško, J. Presence of antibodies to some zoonoses in mouflons (Ovis musimon Pall.) and fallow deer (Dama dama L.) in eastern Slovakia. Vet. Med. 1999, 44, 215–219. [Google Scholar]
- Štefančíková, A.; Derdáková, M.; Lenčáková, D.; Ivanová, R.; Stanko, M.; Čisláková, L.; Peťko, B. Serological and molecular detection of Borrelia burgdorferi sensu lato and Anaplasmataceae in rodents. Folia Microbiol. 2008, 53, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Nadzamová, D.; Petko, B.; Stefancíková, A.; Cisláková, L.; Stĕpánová, G.; Seba, M. Potential foci of Lyme borreliosis in towns. Cent. Eur. J. Public Health 2000, 8, 176–178. [Google Scholar] [PubMed]
- Peťko, B.; Štefančíková, A.; Nadzamová, D.; Stanko, M.; Siuda, K.; Karbowiak, G.; Winiarczyjk, S. 2001. Epizootiological Aspects of Lyme Borreliosis in the City Agglomerations of the Carpathian Regions of Slovakia and Poland and Their Peripheral Parts. In Stawonogi Pasożyty i Nosiciele; Buczek, A., Błaszak, C., Eds.; Wyd. KGM: Lublin, Poland, 2001; pp. 157–164. [Google Scholar]
- Derdakova, M.; Halanova, M.; Stanko, M.; Stefancikova, A.; Cislakova, L.; Pet’ko, B. Molecular evidence for Anaplasma phagocytophilum and Borrelia burgdorferi sensu lato in Ixodes ricinus ticks from eastern Slovakia. Ann. Agric. Environ. Med. 2003, 10, 269–271. [Google Scholar]
- Smetanová, K.; Schwarzová, K.; Kocianová, E. Detection of Anaplasma phagocytophilum, Coxiella burnetti, Rickettsia spp., and Borrelia burgdorferi s.l. in ticks and wild living animals in western and middle Slovakia. Ann. N. Y. Acad. Sci. 2006, 1078, 312–315. [Google Scholar] [CrossRef]
- Smetanová, K.; Burri, C.; Pérez, D.; Gern, L.; Kocianová, E. Detection and identification of Borrelia burgdorferi sensu lato genospecies in ticks from three different regions in Slovakia. Wien. Klin. Wochenschr. 2007, 119, 534–537. [Google Scholar] [CrossRef]
- Danielová, V.; Daniel, M.; Schwarzová, L.; Materna, J.; Rudenko, N.; Golovchenko, M.; Holubová, J.; Grubhoffer, L.; Kilián, P. Integration of a tick-borne encephalitis virus and Borrelia burgdorferi sensu lato into mountain ecosystems, following a shift in the altitudinal limit of distribution of their vector, Ixodes ricinus (Krkonose mountains, Czech Republic). Vector Borne Zoonotic Dis. 2010, 10, 223–230. [Google Scholar] [CrossRef]
- Picken, M.M.; Cheng, Y.; Strle, F.; Picken, R.N. Patient isolates of Borrelia burgdorferi sensu lato with genomic and phenotypic similarities of strain 25015. J. Infect. Dis. 1996, 174, 1112–1115. [Google Scholar] [CrossRef]
- Strle, F.; Picken, R.N.; Cheng, Y.; Cimperman, J.; Maraspin, V.; Lotric-Furlan, S.; Ruzic-Sabljic, E.; Picken, M.M. Clinical findings for patients with Lyme borreliosis caused by Borrelia burgdorferi sensu lato with genomic and phenotypic similarities of strain 25015. Clin. Infect. Dis. 1997, 25, 273–280. [Google Scholar] [PubMed]
- Hansford, K.M.; Wheeler, B.W.; Tschirren, B.; Medlock, J.M. Questing Ixodes ricinus ticks and Borrelia spp. in urban green space across Europe: A review. Zoonoses Public Health 2022, 69, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Hornok, S.; Meli, M.L.; Gönczi, E.; Halász, E.; Takács, N.; Farkas, R.; Hofmann-Lehmann, R. Occurrence of ticks and prevalence of Anaplasma phagocytophilum and Borrelia burgdorferi s.l. in three types of urban biotopes: Forests, parks and cemeteries. Ticks Tick Borne Dis. 2014, 5, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Richtrová, E.; Míchalová, P.; Lukavská, A.; Navrátil, J.; Kybicová, K. Borrelia burgdorferi sensu lato infection in Ixodes ricinus ticks in urban green areas in Prague. Ticks Tick Borne Dis. 2022, 13, 102053. [Google Scholar] [CrossRef] [PubMed]
- Glass, A.; Springer, A.; Raulf, M.K.; Fingerle, V.; Strube, C. 15-year Borrelia prevalence and species distribution monitoring in Ixodes ricinus/inopinatus populations in the city of Hanover, Germany. Ticks Tick Borne Dis. 2023, 14, 102074. [Google Scholar] [CrossRef]
- Rollins, R.E.; Yeyin, Z.; Wyczanska, M.; Alig, N.; Hepner, S.; Fingerle, V.; Margos, G.; Becker, N.S. Spatial variability in prevalence and genospecies distributions of Borrelia burgdorferi sensu lato from ixodid ticks collected in southern Germany. Ticks Tick Borne Dis. 2021, 12, 101589. [Google Scholar] [CrossRef]
- Kowalec, M.; Szewczyk, T.; Welc-Faleçiak, R.; Siński, E.; Karbowiak, G.; Bajer, A. Ticks and the city—Are there any differences between city parks and natural forests in terms of tick abundance and prevalence of spirochaetes? Parasit. Vectors 2017, 10, 573. [Google Scholar] [CrossRef]
- Gray, J.S.; Kahl, O.; Janetzki, C.; Stein, J. Studies on the ecology of Lyme disease in a deer forest in County Galway, Ireland. J. Med. Entomol. 1992, 29, 915–920. [Google Scholar] [CrossRef]
- Zeman, P.; Daniel, M. Mosaic pattern of Borrelia infection in a continuous population of the tick Ixodes ricinus (Acari: Ixodidae). Exp. Appl. Acarol. 1999, 23, 327–335. [Google Scholar] [CrossRef]
| 2011 | 2012 | 2013 | Total | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Site | % (pos/ex) | 95% CI | % (pos/ex) | 95% CI | % (pos/ex) | 95% CI | χ2 | p | % (pos/ex) | 95% CI | |
| Bratislava | Nymphs | 6.9 (61/887) | 5.4–8.7 | 11.6 (23/199) | 7.1–16.0 | 1.8 (8/448) | 0.6–3.0 | 26.227 | <0.0001 | 6.0 (92/1534) | 4.8–7.2 |
| (urban/suburban habitat) | Females | 18.6 (69/370) | 14.7–22.6 | 14.5 (9/62) | 5.7–23.3 | 17.7 (28/158) | 11.8–23.7 | 0.624 | 0.732 | 18.0 (106/590) | 14.9–21.1 |
| Males | 9.9 (44/443) | 7.1–12.7 | 13.2 (9/68) | 5.2–21.3 | 8.29 (15/181) | 4.3–12.3 | 1.381 | 0.501 | 9.8 (68/692) | 7.6–12.0 | |
| Adults total | 13.9 (113/813) | 11.5–16.3 | 13.8 (18/130) | 7.9–19.8 | 12.3 (43/339) | 9.1–16.2 | 0.310 | 0.856 | 13.6 (174/1282) | 11.7–15.4 | |
| Total | 10.2 (174/1700) | 8.8–11.7 | 12.5 (41/329) | 8.9–16.0 | 6.5 (51/787) | 4.8–8.2 | 12.829 | 0.002 | 9.4 (266/2816) | 8.3–10.5 | |
| Fúgelka | Nymphs | 17.0 (184/1084) | 14.7–19.2 | 19.5 (58/298) | 15.0–24.0 | 8.9 (21/237) | 5.2–12.5 | 12.190 | 0.002 | 16.2 (263/1619) | 14.4–18.0 |
| (natural habitat) | Females | 17.0 (26/153) | 11.0–22.9 | 25.0 (15/60) | 14.0–36.0 | 16.4 (12/73) | 7.9–24.9 | 2.114 | 0.347 | 18.5 (53/286) | 14.0–23.0 |
| Males | 7.2 (12/167) | 3.3–11.1 | 12.0 (10/83) | 5.0–19.0 | 8.3 (8/96) | 2.8–13.9 | 1.674 | 0.433 | 8.7 (30/346) | 5.7–11.6 | |
| Adults total | 11.9 (38/320) | 8.3–15.4 | 17.5 (25/143) | 11.3–23.7 | 11.8 (20/169) | 7.0–16.7 | 3.065 | 0.216 | 13.1 (83/632) | 10.5–15.8 | |
| Total | 15.8 (222/1404) | 13.9–17.7 | 18.8 (83/441) | 15.2–22.5 | 10.1 (41/406) | 7.2–13.0 | 12.921 | 0.002 | 15.4 (346/2251) | 13.9–16.9 | |
| Rozhanovce | Nymphs | 15.4 (41/266) | 11.1–19.7 | 12.9 (56/433) | 9.8–16.1 | 13.0 (20/154) | 7.7–18.3 | 0.941 | 0.625 | 13.7 (117/853) | 11.4–16.0 |
| (agricultural habitat) | Females | 21.2 (7/33) | 7.3–35.2 | 27.3 (9/33) | 12.1–42.5 | 8.8 (3/34) | 0.0–18.4 | 0.119 F | 19.0 (19/100) | 11.3–26.7 | |
| Males | 13.3 (4/30) | 1.2–25.5 | 25.6 (11/43) | 12.5–38.6 | 6.7 (2/30) | 0.0–15.6 | 0.101 F | 16.5 (17/103) | 9.3–23.7 | ||
| Adults total | 17.5 (11/63) | 8.1–26.8 | 26.3 (20/76) | 16.4–36.2 | 7.8 (5/64) | 1.2–14.4 | 8.158 | 0.017 | 17.7 (36/203) | 12.5–23.0 | |
| Total | 15.8 (52/329) | 11.9–19.7 | 14.9 (76/509) | 11.8–18.0 | 11.5 (25/218) | 7.2–15.7 | 2.146 | 0.342 | 14.5 (153/1056) | 12.4–16.6 |
| Species | ss | a | g | v | l | s | coinf | T | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | ||
| Bratislava | |||||||||||||||
| 2011 | 0.1 | 0–0.3 | 4.5 | 3.5–5.4 | 3.1 | 2.3–3.9 | 0.6 | 0.2–0.9 | 1.1 | 0.6–1.6 | 0.8 | 0.3–1.2 | 0.06 | 0–0.2 | 1700 |
| 2012 | 0.6 | 0–1.45 | 5.5 | 3.0–7.9 | 2.1 | 0.6–3.7 | 3.3 | 1.4–5.39 | 0.9 | 0–1.9 | 0 | 0 | 329 | ||
| 2013 | 0.6 | 0–1.2 | 1.3 | 0.5–2.0 | 1.6 | 0.8–2.5 | 2.4 | 1.3–3.5 | 0.4 | 0–0.8 | 0.1 | 0–0.4 | 787 | ||
| Total | 0.3 | 0.1–0.5 | 3.7 | 3.0–4.4 | 2.6 | 2.0–3.2 | 1.4 | 1.0–1.9 | 0.9 | 0.5–1.2 | 0.5 | 0.2–0.7 | 0.1 | 0–0.2 | 2816 |
| Fúgelka | |||||||||||||||
| 2011 | 0.6 | 0.2–1.1 | 6.5 | 4.8–7.3 | 6.1 | 4.9–7.4 | 2.2 | 1.4–3.0 | 0.1 | 0–0.2 | 0.1 | 0–0.2 | 0.6 | 0.2–1.1 | 1404 |
| 2012 | 0 | 5.0 | 3.0–7.0 | 8.8 | 6.2–11.5 | 2.3 | 0.9–3.7 | 2.7 | 1.2–4.2 | 0 | 0 | 441 | |||
| 2013 | 0 | 3.2 | 1.5–4.9 | 5.4 | 3.2–7.6 | 0.7 | 0–1.6 | 0.2 | 0–0.7 | 0.2 | 0–0.7 | 0.2 | 0–0.7 | 406 | |
| Total | 0.4 | 0.1–0.7 | 5.3 | 4.4–6.3 | 6.5 | 5.5–7.5 | 1.9 | 1.4–2.5 | 0.6 | 0.3–0.9 | 0.1 | 0–0.2 | 0.4 | 0.2–0.7 | 2251 |
| Rozhanovce | |||||||||||||||
| 2011 | 0.6 | 0–1.4 | 8.8 | 5.7–11.9 | 3.3 | 1.4–5.3 | 0.9 | 0–1.9 | 0.3 | 0–0.9 | 1.8 | 0.4–3.3 | 329 | ||
| 2012 | 1.8 | 0.6–2.9 | 4.1 | 2.4–5.8 | 3.3 | 1.8–4.9 | 3.7 | 2.1–5.4 | 1.0 | 0.1–1.8 | 0 | 1.0 | 0.1–1.8 | 509 | |
| 2013 | 0.9 | 0–2.2 | 1.4 | 0–2.9 | 4.6 | 1.8–7.4 | 4.6 | 1.8–7.4 | 0 | 0 | 0 | 218 | |||
| Total | 1.2 | 0.6–1.9 | 5.2 | 3.7–6.3 | 3.6 | 2.5–4.7 | 2.7 | 1.8–3.7 | 0.8 | 0.2–1.3 | 0.1 | 0–0.3 | 1.4 | 0.4–1.6 | 1056 |
| TOTAL | 0.5 | 0.3–0.7 | 4.5 | 4.0–5.0 | 4.2 | 3.7–4.7 | 1.8 | 1.5–2.2 | 0.8 | 0.5–1.0 | 0.3 | 0.1–0.4 | 0.4 | 0.2–0.5 | 6123 |
| Transect | Nymphs | Adults | Total | %N | |||
|---|---|---|---|---|---|---|---|
| % (pos/ex) | 95% CI | % (pos/ex) | 95% CI | % (pos/ex) | 95% CI | ||
| A | 2.6 (10/381) | 1.0–4.2 | 6.8 (6/88) | 1.5–12.1 | 3.4 (16/469) | 1.8–5.0 | 81.2 |
| B | 13.9 (25/180) | 8.8–18.9 | 8.6 (25/291) | 5.4–11.8 | 10.6 (50/471) | 7.8–13.4 | 38.2 |
| C | 14.3 (2/14) | 0.0–32.6 | 21.0 (41/195) | 15.3–26.7 | 20.6 (43/209) | 15.1–26.0 | 6.7 |
| D | 25.0 (19/76) | 15.3–34.7 | 25.4 (44/173) | 18.9–31.9 | 25.3 (63/249) | 19.9–30.7 | 30.5 |
| E | 5.1 (12/233) | 2.3–8.0 | 12.7 (8/63) | 4.5–20.9 | 6.8 (20/296) | 3.9–9.6 | 78.7 |
| χ2 | 58.554 | 33.947 | 101.798 | ||||
| p | <0.0001 | <0.0001 | <0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazimírová, M.; Mahríková, L.; Hamšíková, Z.; Stanko, M.; Golovchenko, M.; Rudenko, N. Spatial and Temporal Variability in Prevalence Rates of Members of the Borrelia burgdorferi Species Complex in Ixodes ricinus Ticks in Urban, Agricultural and Sylvatic Habitats in Slovakia. Microorganisms 2023, 11, 1666. https://doi.org/10.3390/microorganisms11071666
Kazimírová M, Mahríková L, Hamšíková Z, Stanko M, Golovchenko M, Rudenko N. Spatial and Temporal Variability in Prevalence Rates of Members of the Borrelia burgdorferi Species Complex in Ixodes ricinus Ticks in Urban, Agricultural and Sylvatic Habitats in Slovakia. Microorganisms. 2023; 11(7):1666. https://doi.org/10.3390/microorganisms11071666
Chicago/Turabian StyleKazimírová, Mária, Lenka Mahríková, Zuzana Hamšíková, Michal Stanko, Maryna Golovchenko, and Natalie Rudenko. 2023. "Spatial and Temporal Variability in Prevalence Rates of Members of the Borrelia burgdorferi Species Complex in Ixodes ricinus Ticks in Urban, Agricultural and Sylvatic Habitats in Slovakia" Microorganisms 11, no. 7: 1666. https://doi.org/10.3390/microorganisms11071666
APA StyleKazimírová, M., Mahríková, L., Hamšíková, Z., Stanko, M., Golovchenko, M., & Rudenko, N. (2023). Spatial and Temporal Variability in Prevalence Rates of Members of the Borrelia burgdorferi Species Complex in Ixodes ricinus Ticks in Urban, Agricultural and Sylvatic Habitats in Slovakia. Microorganisms, 11(7), 1666. https://doi.org/10.3390/microorganisms11071666






