Mid-Term Results of Single-Stage Surgery for Patients with Chronic Osteomyelitis Using Antibiotic-Loaded Resorbable PerOssal® Beads
Abstract
1. Introduction
2. Methods
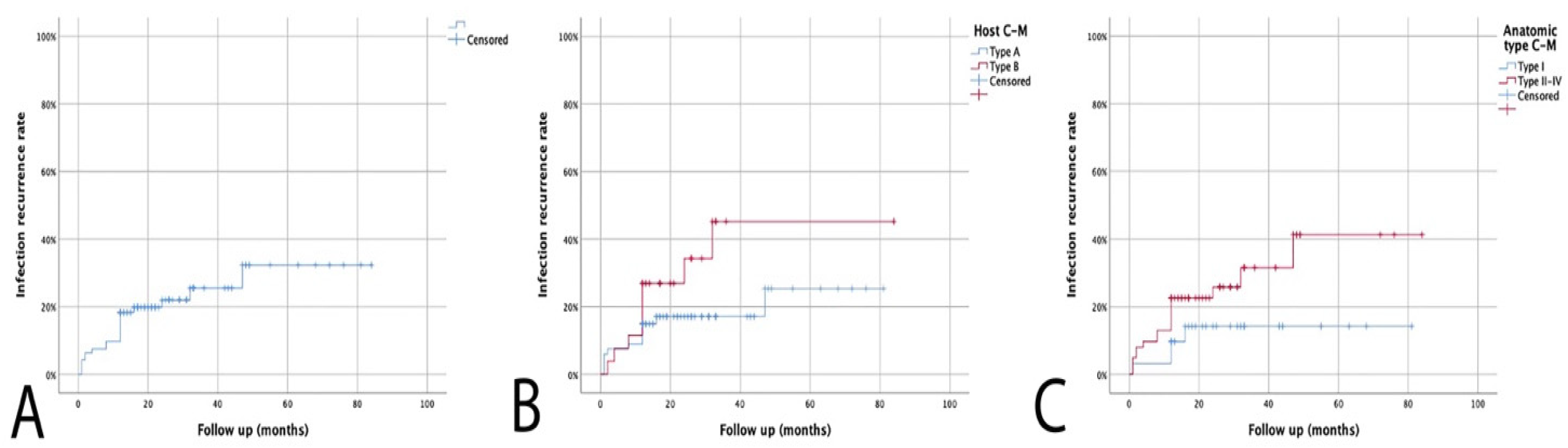
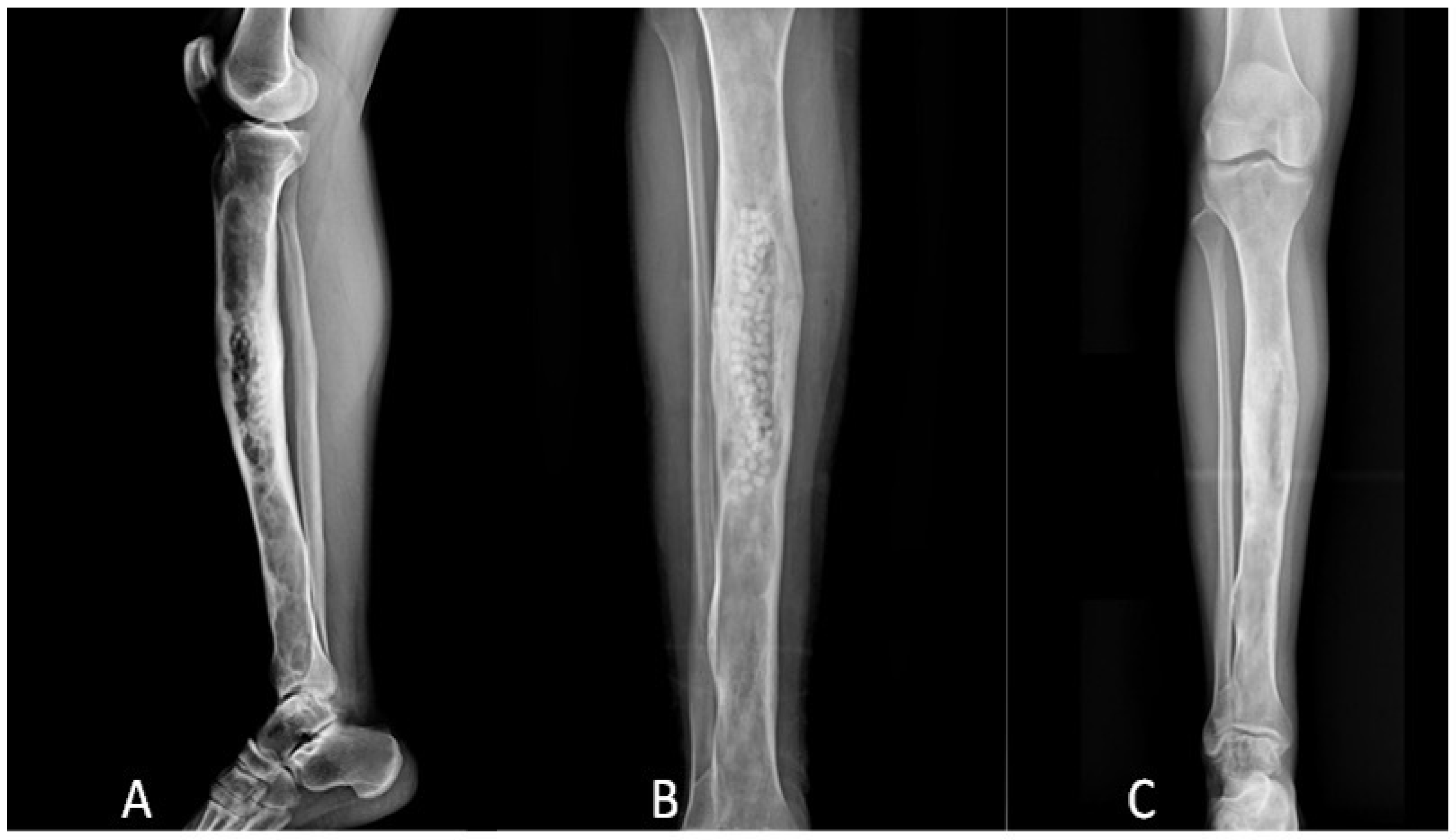
3. Results
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Salvana, J.; Rodner, C.; Browner, B.D.; Livingston, K.; Schreiber, J.; Pesanti, E. Chronic osteomyelitis: Results obtained by an integrated team approach to management. Conn. Med. 2005, 69, 195–202. [Google Scholar] [PubMed]
- Panteli, M.; Giannoudis, P.V. Chronic osteomyelitis: What the surgeon needs to know. EFORT Open Rev. 2016, 1, 128–135. [Google Scholar] [CrossRef]
- Maffulli, N.; Papalia, R.; Zampogna, B.; Torre, G.; Albo, E.; Denaro, V. The management of osteomyelitis in the adult. Surgeon 2016, 14, 345–360. [Google Scholar] [CrossRef] [PubMed]
- Ziran, B.H.; Rao, N.; Hall, R.A. A dedicated team approach enhances outcomes of osteomyelitis treatment. Clin. Orthop. Relat. Res. 2003, 414, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Sambri, A.; Fiore, M.; Tedeschi, S.; De Paolis, M. The Need for Multidisciplinarity in Modern Medicine: An Insight into Orthopaedic Infections. Microorganisms 2022, 10, 756. [Google Scholar] [CrossRef]
- Swiontkowski, M.F.; Hanel, D.P.; Vedder, N.B.; Schwappach, J.R. A comparison of short- and long-term intravenous antibiotic therapy in the postoperative management of adult osteomyelitis. J. Bone Jt. Surg. Br. 1999, 81, 1046–1050. [Google Scholar] [CrossRef]
- McNally, M.A.; Small, J.O.; Tofighi, H.G.; Mollan, R.A. Two-stage management of chronic osteomyelitis of the long bones. The Belfast technique. J. Bone Jt. Surg. Br. 1993, 75, 375–380. [Google Scholar] [CrossRef]
- Parsons, B.; Strauss, E. Surgical management of chronic osteomyelitis. Am. J. Surg. 2004, 188, 57–66. [Google Scholar] [CrossRef]
- Chan, J.K.K.; Ferguson, J.Y.; Scarborough, M.; McNally, M.A.; Ramsden, A.J. Management of Post-Traumatic Osteomyelitis in the Lower Limb: Current State of the Art. Indian J. Plast. Surg. 2019, 52, 62–72. [Google Scholar] [CrossRef]
- Wahl, P.; Guidi, M.; Benninger, E.; Rönn, K.; Gautier, E.; Buclin, T.; Magnin, J.L.; Livio, F. The levels of vancomycin in the blood and the wound after the local treatment of bone and soft-tissue infection with antibiotic-loaded calcium sulphate as carrier material. Bone Jt. J. 2017, 99-B, 1537–1544. [Google Scholar] [CrossRef]
- Kinik, H.; Karaduman, M. Cierny-Mader Type III chronic osteomyelitis: The results of patients treated with debridement, irrigation, vancomycin beads and systemic antibiotics. Int. Orthop. 2008, 32, 551–558. [Google Scholar] [CrossRef]
- Romanò, C.L.; Logoluso, N.; Meani, E.; Romanò, D.; De Vecchi, E.; Vassena, C.; Drago, L. A comparative study of the use of bioactive glass S53P4 and antibiotic-loaded calcium-based bone substitutes in the treatment of chronic osteomyelitis: A retrospective comparative study. Bone Jt. J. 2014, 96-B, 845–850. [Google Scholar] [CrossRef]
- Arshad, Z.; Lau, E.J.; Aslam, A.; Thahir, A.; Krkovic, M. Management of chronic osteomyelitis of the femur and tibia: A scoping review. EFORT Open Rev. 2021, 6, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Rand, B.C.; Penn-Barwell, J.G.; Wenke, J.C. Combined local and systemic antibiotic delivery improves eradication of wound contamination: An animal experimental model of contaminated fracture. Bone Jt. J. 2015, 97-B, 1423–1427. [Google Scholar] [CrossRef] [PubMed]
- McKee, M.D.; Li-Bland, E.A.; Wild, L.M.; Schemitsch, E.H. A prospective, randomized clinical trial comparing an antibiotic-impregnated bioabsorbable bone substitute with standard antibiotic-impregnated cement beads in the treatment of chronic osteomyelitis and infected nonunion. J. Orthop. Trauma 2010, 24, 483–490. [Google Scholar] [CrossRef]
- Ferguson, J.Y.; Dudareva, M.; Riley, N.D.; Stubbs, D.; Atkins, B.L.; McNally, M.A. The use of a biodegradable antibiotic-loaded calcium sulphate carrier containing tobramycin for the treatment of chronic osteomyelitis: A series of 195 cases. Bone Jt. J. 2014, 96-B, 829–836. [Google Scholar] [CrossRef]
- von Stechow, D.; Rauschmann, M.A. Effectiveness of combination use of antibiotic-loaded PerOssal with spinal surgery in patients with spondylodiscitis. Eur. Surg. Res. 2009, 43, 298–305. [Google Scholar] [CrossRef]
- Glaudemans, A.W.J.M.; Jutte, P.C.; Cataldo, M.A.; Cassar-Pullicino, V.; Gheysens, O.; Borens, O.; Trampuz, A.; Wörtler, K.; Petrosillo, N.; Winkler, H.; et al. Consensus document for the diagnosis of peripheral bone infection in adults: A joint paper by the EANM, EBJIS, and ESR (with ESCMID endorsement). Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 957–970. [Google Scholar] [CrossRef]
- Cierny, G.; DiPasquale, D. Treatment of chronic infection. J. Am. Acad. Orthop. Surg. 2006, 14, S105–S110. [Google Scholar] [CrossRef]
- Sambri, A.; Spinnato, P.; Tedeschi, S.; Zamparini, E.; Fiore, M.; Zucchini, R.; Giannini, C.; Caldari, E.; Crombé, A.; Viale, P.; et al. Bone and Joint Infections: The Role of Imaging in Tailoring Diagnosis to Improve Patients’ Care. J. Pers. Med. 2021, 11, 1317. [Google Scholar] [CrossRef] [PubMed]
- Cierny, G.; Mader, J.T.; Penninck, J.J. A clinical staging system for adult osteomyelitis. Clin. Orthop. Relat. Res. 2003, 414, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Rauschmann, M.A.; Wichelhaus, T.A.; Stirnal, V.; Dingeldein, E.; Zichner, L.; Schnettler, R.; Alt, V. Nanocrystalline hydroxyapatite and calcium sulphate as biodegradable composite carrier material for local delivery of antibiotics in bone infections. Biomaterials 2005, 26, 2677–2684. [Google Scholar] [CrossRef]
- Visani, J.; Staals, E.L.; Donati, D. Treatment of chronic osteomyelitis with antibiotic-loaded bone void filler systems: An experience with hydroxyapatites calcium-sulfate biomaterials. Acta Orthop. Belg. 2018, 84, 25–29. [Google Scholar]
- Schierholz, J.M.; Morsczeck, C.; Brenner, N.; König, D.P.; Yücel, N.; Korenkov, M.; Neugebauer, E.; Rump, A.F.; Waalenkamp, G.; Beuth, J.; et al. Special aspects of implant-associated infection in orthopedic surgery. From the pathophysiology to custom-tailored prevention strategies. Orthopade 2004, 33, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.H.; Deakin, M.; Latham, J.M. Chronic osteomyelitis. The effect of the extent of surgical resection on infection-free survival. J. Bone Jt. Surg. Br. 2001, 83, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.; Colangeli, M.; Colangeli, S.; Di Bella, C.; Gozzi, E.; Donati, D. Adult osteomyelitis: Debridement versus debridement plus Osteoset T pellets. Acta Orthop. Belg. 2007, 73, 238–243. [Google Scholar] [PubMed]
- McNally, M.A.; Ferguson, J.Y.; Lau, A.C.; Diefenbeck, M.; Scarborough, M.; Ramsden, A.J.; Atkins, B.L. Single-stage treatment of chronic osteomyelitis with a new absorbable, gentamicin-loaded, calcium sulphate/hydroxyapatite biocomposite: A prospective series of 100 cases. Bone Jt. J. 2016, 98-B, 1289–1296. [Google Scholar] [CrossRef]
- McNally, M.A.; Ferguson, J.Y.; Scarborough, M.; Ramsden, A.; Stubbs, D.A.; Atkins, B.L. Mid- to long-term results of single-stage surgery for patients with chronic osteomyelitis using a bioabsorbable gentamicin-loaded ceramic carrier. Bone Jt. J. 2022, 104-B, 1095–1100. [Google Scholar] [CrossRef]
- Fleiter, N.; Walter, G.; Bösebeck, H.; Vogt, S.; Büchner, H.; Hirschberger, W.; Hoffmann, R. Clinical use and safety of a novel gentamicin-releasing resorbable bone graft substitute in the treatment of osteomyelitis/osteitis. Bone Jt. Res. 2014, 3, 223–229. [Google Scholar] [CrossRef]
- Xie, Z.; Liu, X.; Jia, W.; Zhang, C.; Huang, W.; Wang, J. Treatment of osteomyelitis and repair of bone defect by degradable bioactive borate glass releasing vancomycin. J. Control Release 2009, 139, 118–126. [Google Scholar] [CrossRef]
- Turner, T.M.; Urban, R.M.; Hall, D.J.; Chye, P.C.; Segreti, J.; Gitelis, S. Local and systemic levels of tobramycin delivered from calcium sulfate bone graft substitute pellets. Clin. Orthop. Relat. Res. 2005, 437, 97–104. [Google Scholar] [CrossRef]
- McKee, M.D.; Wild, L.M.; Schemitsch, E.H.; Waddell, J.P. The use of an antibiotic-impregnated, osteoconductive, bioabsorbable bone substitute in the treatment of infected long bone defects: Early results of a prospective trial. J. Orthop. Trauma 2002, 16, 622–627. [Google Scholar] [CrossRef]
- Kanakaris, N.; Gudipati, S.; Tosounidis, T.; Harwood, P.; Britten, S.; Giannoudis, P.V. The treatment of intramedullary osteomyelitis of the femur and tibia using the Reamer-Irrigator-Aspirator system and antibiotic cement rods. Bone Jt. J. 2014, 96-B, 783–788. [Google Scholar] [CrossRef]
- Pincher, B.; Fenton, C.; Jeyapalan, R.; Barlow, G.; Sharma, H.K. A systematic review of the single-stage treatment of chronic osteomyelitis. J. Orthop. Surg. Res. 2019, 14, 393. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.J.; Florance, H.; McKinnon, J.L.; Laycock, P.A.; Aiken, S.S. Elution profiles of tobramycin and vancomycin from high-purity calcium sulphate beads incubated in a range of simulated body fluids. J. Biomater. Appl. 2016, 31, 357–365. [Google Scholar] [CrossRef]
- Monzón, M.; García-Alvarez, F.; Laclériga, A.; Gracia, E.; Leiva, J.; Oteiza, C.; Amorena, B. A simple infection model using pre-colonized implants to reproduce rat chronic Staphylococcus aureus osteomyelitis and study antibiotic treatment. J. Orthop. Res. 2001, 19, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Beuerlein, M.J.; McKee, M.D. Calcium sulfates: What is the evidence? J. Orthop. Trauma 2010, 24 (Suppl. S1), S46–S51. [Google Scholar] [CrossRef]
- Keller, J.; Brink, S.; Busse, B.; Schilling, A.F.; Schinke, T.; Amling, M.; Lange, T. Divergent resorbability and effects on osteoclast formation of commonly used bone substitutes in a human in vitro-assay. PLoS ONE 2012, 7, e46757. [Google Scholar] [CrossRef]
- Badie, A.A.; Arafa, M.S. One-stage surgery for adult chronic osteomyelitis: Concomitant use of antibiotic-loaded calcium sulphate and bone marrow aspirate. Int. Orthop. 2019, 43, 1061–1070. [Google Scholar] [CrossRef]
- Inzana, J.A.; Schwarz, E.M.; Kates, S.L.; Awad, H.A. Biomaterials approaches to treating implant-associated osteomyelitis. Biomaterials 2016, 81, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Rousset, M.; Walle, M.; Cambou, L.; Mansour, M.; Samba, A.; Pereira, B.; Ghanem, I.; Canavese, F. Chronic infection and infected non-union of the long bones in paediatric patients: Preliminary results of bone versus beta-tricalcium phosphate grafting after induced membrane formation. Int. Orthop. 2018, 42, 385–393. [Google Scholar] [CrossRef]
- Sasaki, G.; Watanabe, Y.; Yasui, Y.; Matsui, K.; Kawano, H.; Miyamoto, W. Induced membrane technique using an equal portion of autologous cancellous bone and β-tricalcium phosphate provided a successful outcome for osteomyelitis in large part of the femoral diaphysis—Case report. Trauma Case Rep. 2021, 36, 100545. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Wang, G.; Zhang, Y.; Luo, W.; Liu, S.; Liu, Y.; Zhou, Y. The effect of calcium sulfate/calcium phosphate composite for the treatment of chronic osteomyelitis compared with calcium sulfate. Ann. Palliat. Med. 2020, 9, 1821–1833. [Google Scholar] [CrossRef] [PubMed]
- Urban, R.M.; Turner, T.M.; Hall, D.J.; Inoue, N.; Gitelis, S. Increased bone formation using calcium sulfate-calcium phosphate composite graft. Clin. Orthop. Relat. Res. 2007, 459, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Lazzarini, L.; Mader, J.T.; Calhoun, J.H. Osteomyelitis in long bones. J. Bone Jt. Surg. Am. 2004, 86, 2305–2318. [Google Scholar] [CrossRef]
- Bose, D.; Kugan, R.; Stubbs, D.; McNally, M. Management of infected nonunion of the long bones by a multidisciplinary team. Bone Jt. J. 2015, 97-B, 814–817. [Google Scholar] [CrossRef]
- Sambri, A.; Pignatti, M.; Tedeschi, S.; Lozano Miralles, M.E.; Giannini, C.; Fiore, M.; Filippini, M.; Cipriani, R.; Viale, P.; De Paolis, M. Combined Orthoplastic Approach in Fracture-Related Infections of the Distal Tibia. Microorganisms 2022, 10, 1640. [Google Scholar] [CrossRef] [PubMed]
- Gosain, A.; Chang, N.; Mathes, S.; Hunt, T.K.; Vasconez, L. A study of the relationship between blood flow and bacterial inoculation in musculocutaneous and fasciocutaneous flaps. Plast. Reconstr. Surg. 1990, 86, 1152–1162; discussion 1163. [Google Scholar] [CrossRef]
- Richards, R.R.; McKee, M.D.; Paitich, C.B.; Anderson, G.I.; Bertoia, J.T. A comparison of the effects of skin coverage and muscle flap coverage on the early strength of union at the site of osteotomy after devascularization of a segment of canine tibia. J. Bone Jt. Surg. Am. 1991, 73, 1323–1330. [Google Scholar] [CrossRef]
- Zweifel-Schlatter, M.; Haug, M.; Schaefer, D.J.; Wolfinger, E.; Ochsner, P.; Pierer, G. Free fasciocutaneous flaps in the treatment of chronic osteomyelitis of the tibia: A retrospective study. J. Reconstr. Microsurg. 2006, 22, 41–47. [Google Scholar] [CrossRef]
- Borrelli, J.; Prickett, W.D.; Ricci, W.M. Treatment of nonunions and osseous defects with bone graft and calcium sulfate. Clin. Orthop. Relat. Res. 2003, 411, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.M.; Wilkins, R.M.; Gitelis, S.; Hartjen, C.; Watson, J.T.; Kim, P.T. The use of a surgical grade calcium sulfate as a bone graft substitute: Results of a multicenter trial. Clin. Orthop. Relat. Res. 2001, 382, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.; Alk, D.; Sandbank, J.; Farber, R.; Halperin, N. Inflammatory reactions associated with a calcium sulfate bone substitute. Ann. Transplant. 1999, 4, 91–97. [Google Scholar] [PubMed]
| Characteristic | n (%) |
|---|---|
| Age, yeas (median, range) | 40 years (4 to 73). |
| Sex | |
| Male | 68 (73.1%) |
| Female | 25 (26.9%) |
| Site | |
| Femur | 24 (25.8%) |
| Tibia | 52 (55.9%) |
| Humerus | 6 (6.5%) |
| Radius | 4 (4.3%) |
| Other | 7 (7.l5%) |
| CM anatomic type | |
| I | 31 (33.3%) |
| II | 13 (14.0%) |
| III | 21 (22.6%) |
| IV | 28 (30.1%) |
| Local antibiotics Vancomycin Rifampicin Gentamycin Vancomycin + Rifampicin Rifampicin + Gentamycin | 56 (60.2%) 10 (10.8%) 2 (2.1%) 24 (25.8%) 1 (1.1%) |
| Plastic surgery | |
| None | 86 (92.5%) |
| Local flap | 2 (2.1%) |
| Free flap | 5 (5.4%) |
| Pathogen | |
| MRSA | 18 (19.4%) |
| MSSA | 21 (22.6%) |
| CoNS | 10 (10.8%) |
| Enterobacteriaceae | 7 (7.5%) |
| Polimicrobial | 5 (5.3%) |
| Negative | 32 (34.4%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sambri, A.; Cevolani, L.; Passarino, V.; Bortoli, M.; Parisi, S.C.; Fiore, M.; Campanacci, L.; Staals, E.; Donati, D.M.; De Paolis, M. Mid-Term Results of Single-Stage Surgery for Patients with Chronic Osteomyelitis Using Antibiotic-Loaded Resorbable PerOssal® Beads. Microorganisms 2023, 11, 1623. https://doi.org/10.3390/microorganisms11071623
Sambri A, Cevolani L, Passarino V, Bortoli M, Parisi SC, Fiore M, Campanacci L, Staals E, Donati DM, De Paolis M. Mid-Term Results of Single-Stage Surgery for Patients with Chronic Osteomyelitis Using Antibiotic-Loaded Resorbable PerOssal® Beads. Microorganisms. 2023; 11(7):1623. https://doi.org/10.3390/microorganisms11071623
Chicago/Turabian StyleSambri, Andrea, Luca Cevolani, Valentina Passarino, Marta Bortoli, Stefania Claudia Parisi, Michele Fiore, Laura Campanacci, Eric Staals, Davide Maria Donati, and Massimiliano De Paolis. 2023. "Mid-Term Results of Single-Stage Surgery for Patients with Chronic Osteomyelitis Using Antibiotic-Loaded Resorbable PerOssal® Beads" Microorganisms 11, no. 7: 1623. https://doi.org/10.3390/microorganisms11071623
APA StyleSambri, A., Cevolani, L., Passarino, V., Bortoli, M., Parisi, S. C., Fiore, M., Campanacci, L., Staals, E., Donati, D. M., & De Paolis, M. (2023). Mid-Term Results of Single-Stage Surgery for Patients with Chronic Osteomyelitis Using Antibiotic-Loaded Resorbable PerOssal® Beads. Microorganisms, 11(7), 1623. https://doi.org/10.3390/microorganisms11071623







