Occurrence of Leishmaniasis in Iberian Wolves in Northwestern Spain
Abstract
1. Introduction
2. Materials and Methods
2.1. Origin of the Animals
2.2. Sampling
2.3. PCR Analyses of the Samples
3. Results
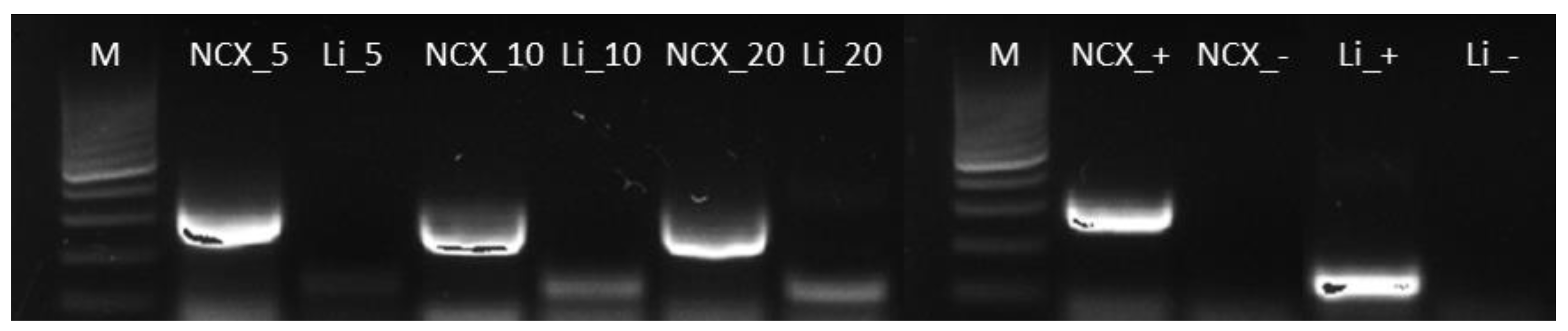
3.1. Set-Up of a PCR-Based Method for L. infantum DNA in Wolf Hair
3.2. Occurrence of Leishmaniasis in Living Wolves
3.3. Occurrence of Leishmaniasis in Dead Wolves
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reguera, R.M.; Morán, M.; Pérez-Pertejo, Y.; García-Estrada, C.; Balaña-Fouce, R. Current status on prevention and treatment of canine leishmaniasis. Vet. Parasitol. 2016, 227, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.R.; Michalick, M.S.M.; da Silva, M.E.; Dos Santos, C.C.P.; Frézard, F.J.G.; da Silva, S.M. Canine leishmaniasis: An overview of the current status and strategies for control. Biomed. Res. Int. 2018, 2018, 3296893. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, M.G. Leishmaniosis of companion animals in Europe: An update. Vet. Parasitol. 2015, 208, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Barriga, D.; Ruiz-Arrondo, I.; Peña, R.E.; Lucientes, J.; Delacour-Estrella, S. Phlebotomine sand flies (Diptera, Psychodidae) from Spain: An updated checklist and extended distributions. Zookeys 2022, 1106, 81–99. [Google Scholar] [CrossRef]
- Travi, B.L.; Cordeiro-da-Silva, A.; Dantas-Torres, F.; Miró, G. Canine visceral leishmaniasis: Diagnosis and management of the reservoir living among us. PLoS Negl. Trop. Dis. 2018, 12, e0006082. [Google Scholar] [CrossRef]
- Meléndez-Lazo, A.; Ordeix, L.; Planellas, M.; Pastor, J.; Solano-Gallego, L. Clinicopathological findings in sick dogs naturally infected with Leishmania infantum: Comparison of five different clinical classification systems. Res. Vet. Sci. 2018, 117, 18–27. [Google Scholar] [CrossRef]
- Morales-Yuste, M.; Martín-Sánchez, J.; Corpas-Lopez, V. Canine leishmaniasis: Update on epidemiology, diagnosis, treatment, and prevention. Vet. Sci. 2022, 9, 387. [Google Scholar] [CrossRef]
- Vaselek, S. Canine Leishmaniasis in Balkan—A review of occurrence and epidemiology. Acta Trop. 2021, 224, 106110. [Google Scholar] [CrossRef]
- Baldi, L.; Mizzoni, V.; Guarino, A. Canine leishmaniasis in Campania: New and old foci. Parassitologia 2004, 46, 217–220. [Google Scholar]
- Le Rutte, E.A.; van der Wilt, L.S.; Bulstra, C.A.; Nieboer, D.; Kontoroupis, P.; de Vlas, S.J.; Richardus, J.H. Incidence and geographical distribution of canine leishmaniosis in 2016–2017 in Spain and France. Vet. Parasitol. Reg. Stud. Rep. 2021, 25, 100613. [Google Scholar] [CrossRef]
- Vélez, R.; Ballart, C.; Domenech, E.; Abras, A.; Fernández-Arévalo, A.; Gómez, S.A.; Tebar, S.; Muñoz, C.; Cairó, J.; Gállego, M. Seroprevalence of canine Leishmania infantum infection in the Mediterranean region and identification of risk factors: The example of North-Eastern and Pyrenean areas of Spain. Prev. Vet. Med. 2019, 162, 67–75. [Google Scholar] [CrossRef]
- Gálvez, R.; Montoya, A.; Cruz, I.; Fernández, C.; Martín, O.; Checa, R.; Chicharro, C.; Migueláñez, S.; Marino, V.; Miró, G. Latest trends in Leishmania infantum infection in dogs in Spain, Part I: Mapped seroprevalence and sand fly distributions. Parasit. Vectors 2020, 13, 204. [Google Scholar] [CrossRef]
- Baneth, G.; Koutinas, A.F.; Solano-Gallego, L.; Bourdeau, P.; Ferrer, L. Canine leishmaniosis—New concepts and insights on an expanding zoonosis: Part one. Trends Parasitol. 2008, 24, 324–330. [Google Scholar] [CrossRef]
- Shaw, S.E.; Langton, D.A.; Hillman, T.J. Canine leishmaniosis in the United Kingdom: A zoonotic disease waiting for a vector? Vet. Parasitol. 2009, 163, 281–285. [Google Scholar] [CrossRef]
- Maia, C.; Cardoso, L. Spread of Leishmania infantum in Europe with dog travelling. Vet Parasitol. 2015, 213, 2–11. [Google Scholar] [CrossRef]
- Rioux, J.A.; Albaret, J.L.; Houin, R.; Dedet, J.P.; Lanotte, G. Ecologie des leishmanioses dans le sud de la France. 2. Les réservoirs selvatiques. Infestation spontanée du Renard (Vulpes vulpes L.). Ann. Parasitol. Hum. Comp. 1968, 43, 421–428. [Google Scholar] [CrossRef]
- Sgroi, G.; Iatta, R.; Veneziano, V.; Bezerra-Santos, M.A.; Lesiczka, P.; Hrazdilová, K.; Annoscia, G.; D’Alessio, N.; Golovchenko, M.; Rudenko, N.; et al. Molecular survey on tick-borne pathogens and Leishmania infantum in red foxes (Vulpes vulpes) from southern Italy. Ticks Tick Borne Dis. 2021, 12, 101669. [Google Scholar] [CrossRef]
- Mitková, B.; Hrazdilová, K.; D’Amico, G.; Duscher, G.G.; Suchentrunk, F.; Forejtek, P.; Gherman, C.M.; Matei, I.A.; Ionică, A.M.; Daskalaki, A.A.; et al. Eurasian golden jackal as host of canine vector-borne protists. Parasit. Vectors 2017, 10, 183. [Google Scholar] [CrossRef]
- Lima, C.M.; Santarém, N.; Neves, N.C.; Sarmento, P.; Carrapato, C.; de Sousa, R.; Cardoso, L.; Cordeiro-da-Silva, A. Serological and molecular survey of Leishmania infantum in a population of Iberian Lynxes (Lynx pardinus). Microorganisms 2022, 10, 2447. [Google Scholar] [CrossRef]
- Del Río, L.; Chitimia, L.; Cubas, A.; Victoriano, I.; De la Rúa, P.; Gerrikagoitia, X.; Barral, M.; Muñoz-García, C.I.; Goyena, E.; García-Martínez, D.; et al. Evidence for widespread Leishmania infantum infection among wild carnivores in L. infantum periendemic northern Spain. Prev. Vet. Med. 2014, 113, 430–435. [Google Scholar] [CrossRef]
- Alcover, M.M.; Ribas, A.; Guillén, M.C.; Berenguer, D.; Tomás-Pérez, M.; Riera, C.; Fisa, R. Wild mammals as potential silent reservoirs of Leishmania infantum in a Mediterranean area. Prev. Vet. Med. 2020, 175, 104874. [Google Scholar] [CrossRef] [PubMed]
- Ortega-García, M.V.; Salguero, F.J.; Rodríguez-Bertos, A.; Moreno, I.; García, N.; García-Seco, T.; Luz Torre, G.; Domínguez, L.; Domínguez, M. A pathological study of Leishmania infantum natural infection in European rabbits (Oryctolagus cuniculus) and Iberian hares (Lepus granatensis). Transbound. Emerg. Dis. 2019, 66, 2474–2481. [Google Scholar] [CrossRef] [PubMed]
- Azami-Conesa, I.; Martínez-Díaz, R.A.; González, F.; Gómez-Muñoz, M.T. First detection of Leishmania infantum in common urban bats Pipistrellus pipistrellus in Europe. Res. Vet. Sci. 2020, 132, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Navea-Pérez, H.M.; Díaz-Sáez, V.; Corpas-López, V.; Merino-Espinosa, G.; Morillas-Márquez, F.; Martín-Sánchez, J. Leishmania infantum in wild rodents: Reservoirs or just irrelevant incidental hosts? Parasitol. Res. 2015, 114, 2363–2370. [Google Scholar] [CrossRef]
- Ortuño, M.; Latrofa, M.S.; Iborra, M.A.; Pérez-Cutillas, P.; Bernal, L.J.; Risueño, J.; Muñoz, C.; Bernal, A.; Sánchez-López, P.F.; Segovia, M.; et al. Genetic diversity and phylogenetic rela-tionships between Leishmania infantum from dogs, humans and wildlife in south-east Spain. Zoonoses Public Health 2019, 66, 961–973. [Google Scholar] [CrossRef]
- Beck, A.; Beck, R.; Kusak, J.; Gudan, A.; Martinković, F.; Artuković, B.; Hohšteter, M.; Huber, Đ.; Marinculić, A.; Grabarević, Ž. A case of visceral leishmaniosis in a gray wolf (Canis lupus) from Croatia. J. Wildl. Dis. 2008, 44, 451–456. [Google Scholar] [CrossRef]
- Sobrino, R.; Ferroglio, E.; Oleaga, A.; Romano, A.; Millán, J.; Revilla, M.; Arnal, M.C.; Trisciuoglio, A.; Gortázar, C. Characterization of widespread canine leishmaniasis among wild carnivores from Spain. Vet. Parasitol. 2008, 155, 198–203. [Google Scholar] [CrossRef]
- Oleaga, A.; Zanet, S.; Espí, A.; Pegoraro de Macedo, M.R.; Gortázar, C.; Ferroglio, E. Leishmania in wolves in northern Spain: A spreading zoonosis evidenced by wildlife sanitary surveillance. Vet. Parasitol. 2018, 255, 26–31. [Google Scholar] [CrossRef]
- Risueño, J.; Ortuño, M.; Pérez-Cutillas, P.; Goyena, E.; Maia, C.; Cortes, S.; Campino, L.; Bernal, L.J.; Muñoz, C.; Arcenillas, I.; et al. Epidemiological and genetic studies suggest a common Leishmania infantum transmission cycle in wildlife, dogs and humans associated to vector abundance in Southeast Spain. Vet. Parasitol. 2018, 259, 61–67. [Google Scholar] [CrossRef]
- Piscopo, N.; Gentile, L.; Scioli, E.; Eguren, V.G.; Carvajal Urueña, A.M.; García, T.Y.; Alberti, J.P.; Esposito, L. Management models applied to the human-wolf conflict in agro-forestry-pastoral territories of two Italian protected areas and one Spanish game area. Animals 2021, 11, 1141. [Google Scholar] [CrossRef]
- Sáenz de Buruaga, M.; Canales, F.; Campos, M.A.; Noriega, A.; Muñoz, F.J.; Navamuel, N. Censo Regional de Lobo (Canis lupus) en Castilla y León; Consejería de Fomento y Medio Ambiente de la Junta de Castilla y León y Ministerio de Agricultura, Alimentación y Medio Ambiente, (TRAGSATEC); Consultora de Recursos Naturales, S.L.: Gasteiz, Spain, 2015. [Google Scholar]
- Fernández-Gil, A.; Barrientos, L.M.; Nuño Nuño, Á.; Naves Cienfuegos, J.; Ordiz, A.; Quevedo, M.; Revilla, M. Estimaciones de Tamaño de Grupo: Implicaciones para la Conservación y Gestión de las Poblaciones Ibéricas de Lobos III Congreso Ibérico del Lobo; Asociación Galega de Custodia do Territorio: Lugo, Spain, 2012. [Google Scholar]
- Centro del Lobo Ibérico de CyL—Félix Rodríguez de la Fuente. Available online: https://centrodellobo.es/ (accessed on 22 February 2021).
- Geisweid, K.; Weber, K.; Sauter-Louis, C.; Hartmann, K. Evaluation of a conjunctival swab polymerase chain reaction for the detection of Leishmania infantum in dogs in a non-endemic area. Vet. J. 2013, 198, 187–192. [Google Scholar] [CrossRef]
- Azami-Conesa, I.; Gómez-Muñoz, M.T.; Martínez-Díaz, R.A. A systematic review (1990–2021) of wild animals infected with zoonotic Leishmania. Microorganisms 2021, 9, 1101. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control and European Food Safety Authority. Mosquito Maps; ECDC: Stockholm, Sweden, 2021; Available online: https://ecdc.europa.eu/en/disease-vectors/surveillance-and-disease-data/phlebotomine-maps (accessed on 22 March 2023).
- Arce, A.; Estirado, A.; Ordobás, M.; Sevilla, S.; García, N.; Moratilla, L.; de la Fuente, S.; Martínez, A.M.; Pérez, A.M.; Aránguez, E.; et al. Re-emergence of leishmaniasis in Spain: Community outbreak in Madrid, Spain, 2009 to 2012. Euro Surveill. 2013, 18, 20546. [Google Scholar] [CrossRef]
- Mohebali, M.; Hajjaran, H.; Hamzavi, Y.; Mobedi, I.; Arshi, S.; Zarei, Z.; Akhoundi, B.; Naeini, K.M.; Avizeh, R.; Fakhar, M. Epidemiological aspects of canine visceral leishmaniosis in the Islamic Republic of Iran. Vet. Parasitol. 2005, 129, 243–251. [Google Scholar] [CrossRef]
- Sastre, N.; Francino, O.; Ramírez, O.; Enseñat, C.; Sánchez, A.; Altet, L. Detection of Leishmania infantum in captive wolves from Southwestern Europe. Vet. Parasitol. 2008, 158, 117–120. [Google Scholar] [CrossRef]
- Souza, T.D.; Turchetti, A.P.; Fujiwara, R.T.; Paixão, T.A.; Santos, R.L. Visceral leishmaniasis in zoo and wildlife. Vet. Parasitol. 2014, 200, 233–241. [Google Scholar] [CrossRef]
- Oleaga, A.; Vicente, J.; Ferroglio, E.; Pegoraro de Macedo, M.R.; Casais, R.; del Cerro, A.; Espí, A.; García, E.J.; Gortázar, C. Concomitance and interactions of pathogens in the Iberian wolf (Canis lupus). Res. Vet. Sci. 2015, 101, 22–27. [Google Scholar] [CrossRef]
- Battisti, E.; Zanet, S.; Khalili, S.; Trisciuoglio, A.; Hertel, B.; Ferroglio, E. Molecular survey on vector-borne pathogens in alpine wild carnivorans. Front. Vet. Sci. 2020, 7, 1. [Google Scholar] [CrossRef]
- Mohebali, M.; Moradi-Asl, E.; Rassi, Y. Geographic distribution and spatial analysis of Leishmania infantum infection in domestic and wild animal reservoir hosts of zoonotic visceral leishmaniasis in Iran: A systematic review. J. Vector Borne Dis. 2018, 55, 173–183. [Google Scholar]

| Name (Id.) | Sample Source | Date | Gender | Years | Mouth | R. Ear | L. Ear | Hair |
|---|---|---|---|---|---|---|---|---|
| La Albañeza | Live capture | 8 November 2017 | female | 5 | - | - | - | n.d. * |
| Lobeznos | Live capture | 17 March 2018 | male | 7.5 | - | + | n.d. | n.d. |
| Brevanas | Live capture | 23 October 2018 | female | 0.5 | + | - | - | n.d. |
| Cabañas | Live capture | 23 October 2018 | female | 0.5 | - | - | - | n.d. |
| Pereruela | Live capture | 4 December 2018 | female | 1.5 | + | - | - | n.d. |
| Mahide | Live capture | 10 December 2018 | male | 0.5 | + | - | - | n.d. |
| Brasa | Iberian Wolf Center | 21 March 2018 | female | 6 | + | + | n.d. | + |
| El Casal | Live capture | 18 October 2019 | male | 0.5 | + | - | - | n.d. |
| Dakota | Iberian Wolf Center | 23 March 2021 | female | 9 | + | + | + | - |
| Clarita | Iberian Wolf Center | 7 May 2021 | female | 10 | - | - | - | n.d. |
| Sauron | Iberian Wolf Center | 7 May 2021 | male | 10 | - | - | + | n.d. |
| Jara | Iberian Wolf Center | 7 May 2021 | female | 8 | - | - | - | n.d. |
| Oscura | Iberian Wolf Center | 7 May 2021 | female | 10 | n.d. | - | - | n.d. |
| Atila | Iberian Wolf Center | 10 November 2021 | male | 9 | - | - | - | n.d. |
| Llagu | Iberian Wolf Center | 10 November 2021 | male | 2 | - | - | - | n.d. |
| Robledo | Iberian Wolf Center | 10 November 2021 | male | 6 | - | - | - | n.d. |
| Sanabria | Iberian Wolf Center | 2 February 2022 | female | 3 | n.d. | n.d. | n.d. | + |
| Tera | Iberian Wolf Center | 2 February 2022 | female | 3 | n.d. | n.d. | n.d. | - |
| Felix | Iberian Wolf Center | 3 February 2022 | male | 1 | n.d. | n.d. | n.d. | + |
| Niebla | Iberian Wolf Center | 3 February 2022 | female | 1 | n.d. | n.d. | n.d. | + |
| Luna | Iberian Wolf Center | 3 February 2022 | female | 1 | n.d. | n.d. | n.d. | + |
| Sampling Place | Date | Cause of Death | Gender | Years | Mouth | R. Ear | L. Ear | Hair |
|---|---|---|---|---|---|---|---|---|
| Puebla de Sanabria 2 | 10 November 17 | Motor vehicle | male | 5 | - | - | - | n.d. * |
| Palacios de Sanabria | 10 January 18 | Motor vehicle | male | 3 | n.d. | + | + | n.d. |
| Puebla de Sanabria 3 | 1 March 18 | Motor vehicle | male | 2.5 | - | + | + | n.d. |
| Puebla de Sanabria | 2 March 18 | Motor vehicle | male | 2.5 | - | n.d. | n.d. | n.d. |
| Cañizo | 1 October 18 | Motor vehicle | female | 7.5 | + | - | - | n.d. |
| Vega del Castillo | 1 February 19 | Fighting | male | 5 | + | n.d. | n.d. | n.d. |
| San Cebrián | 16 April 20 | Motor vehicle | female | 4 | - | n.d. | n.d. | - |
| Ayoo | 18 December 20 | Legal hunting | male | 4 | - | n.d. | n.d. | + |
| Perilla de Castro | 30 January 21 | Legal hunting | male | 5 | - | - | - | n.d. |
| Cerezal | 23 February 21 | Motor vehicle | male | 4 | - | - | - | + |
| Santovenia | 12 March 21 | Motor vehicle | male | 5 | - | n.d. | n.d. | n.d. |
| Burganes | 12 March 21 | Legal hunting | male | 4 | - | n.d. | n.d. | n.d. |
| Torres del Carrizal 2 | 6 July 21 | Motor vehicle | male | 4 | - | n.d. | n.d. | n.d. |
| Torres del Carrizal | 5 September 21 | Motor vehicle | male | 1 | - | - | - | n.d. |
| Cerezal de Aliste | 1 March 22 | Motor vehicle | male | 4 | - | n.d. | n.d. | n.d. |
| Puebla de Sanabria | 1 March 22 | Motor vehicle | male | 3 | - | n.d. | n.d. | n.d. |
| Villanueva | 8 March 22 | Motor vehicle | male | 2 | - | n.d. | n.d. | n.d. |
| Gallegos del Río | 8 March 22 | Motor vehicle | male | 3 | - | n.d. | n.d. | n.d. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merino Goyenechea, J.; Castilla Gómez de Agüero, V.; Palacios Alberti, J.; Balaña Fouce, R.; Martínez Valladares, M. Occurrence of Leishmaniasis in Iberian Wolves in Northwestern Spain. Microorganisms 2023, 11, 1179. https://doi.org/10.3390/microorganisms11051179
Merino Goyenechea J, Castilla Gómez de Agüero V, Palacios Alberti J, Balaña Fouce R, Martínez Valladares M. Occurrence of Leishmaniasis in Iberian Wolves in Northwestern Spain. Microorganisms. 2023; 11(5):1179. https://doi.org/10.3390/microorganisms11051179
Chicago/Turabian StyleMerino Goyenechea, Javier, Verónica Castilla Gómez de Agüero, Jesús Palacios Alberti, Rafael Balaña Fouce, and María Martínez Valladares. 2023. "Occurrence of Leishmaniasis in Iberian Wolves in Northwestern Spain" Microorganisms 11, no. 5: 1179. https://doi.org/10.3390/microorganisms11051179
APA StyleMerino Goyenechea, J., Castilla Gómez de Agüero, V., Palacios Alberti, J., Balaña Fouce, R., & Martínez Valladares, M. (2023). Occurrence of Leishmaniasis in Iberian Wolves in Northwestern Spain. Microorganisms, 11(5), 1179. https://doi.org/10.3390/microorganisms11051179






