Electrospinning Microencapsulation of Lactobacillus fermentum K73 Using Gelatin as the Main Component of a Food-Grade Matrix
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Bacterial Strain and Culture Conditions
2.3. Preparation of Carrier Material
2.3.1. Mixture Experimental Design to Select the Ratio of Wall Materials
2.3.2. Mixture Characterization
2.4. Improvement in Probiotic Viability during the Electrospinning Process through Box–Behnken Design
2.4.1. Electrospinning Process
2.4.2. Cell Count
2.4.3. Scanning Electron Microscopy (SEM)
2.5. Survival Percentage of Encapsulated L. fermentum K73 during Simulated Gastrointestinal Conditions
2.6. Statistical Analysis
3. Results
3.1. Characterization of Polymeric Solutions of the Mixture Experimental Design
3.2. Formulation of Carrier Material
3.3. Cell Counting Process: Release of Probiotics from Electrospun Fibers
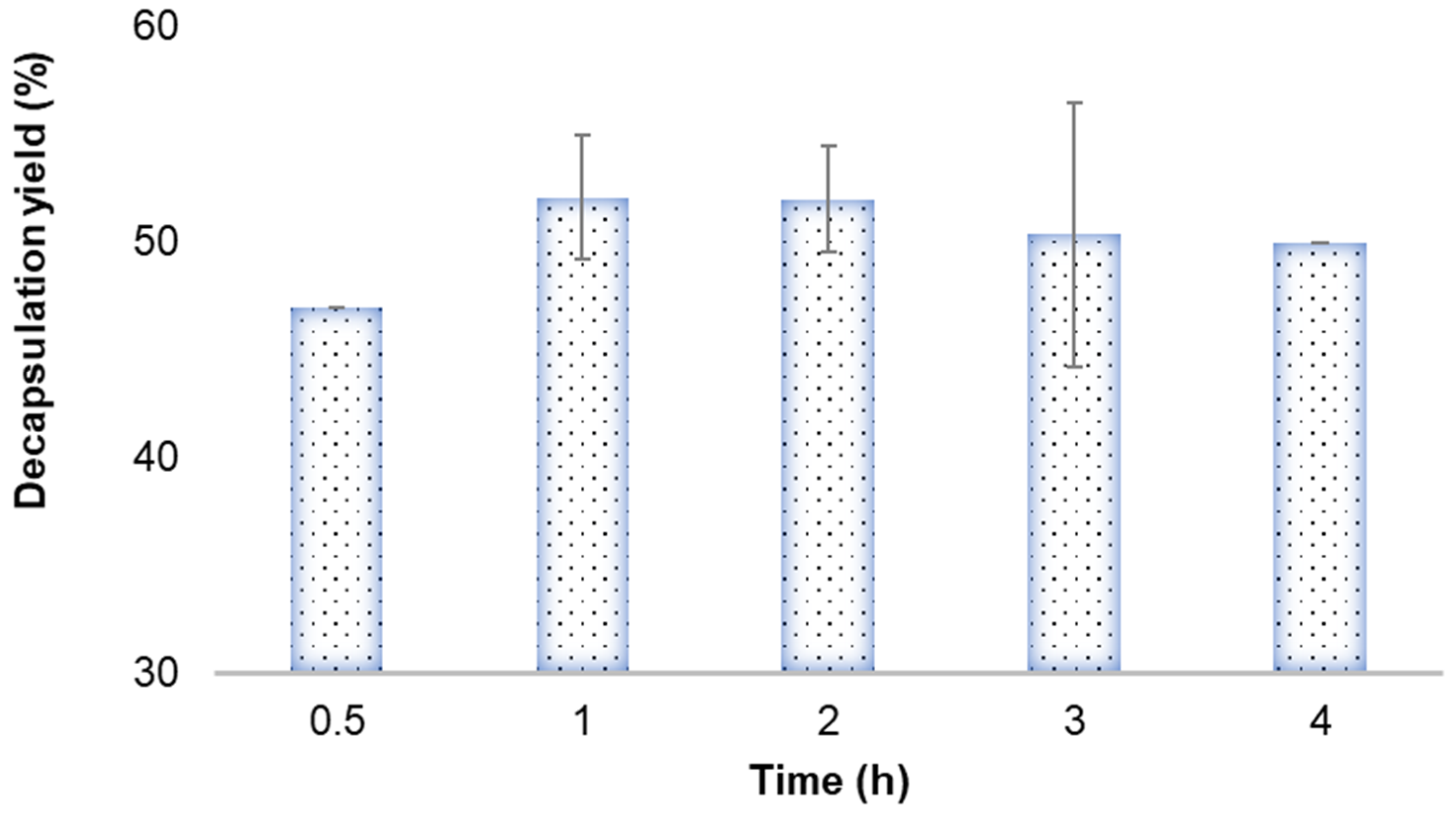
3.4. Improvement of Encapsulation Yield in the Electrospinning Process
3.5. Morphological Characteristics
3.6. Tolerance of Encapsulated and Non-Encapsulated Probiotic to Simulated Gastrointestinal Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Haffner, F.; Diab, R.; Pasc, A. Encapsulation of probiotics: Insights into academic and industrial approaches. AIMS Mater. Sci. 2016, 3, 114–136. [Google Scholar] [CrossRef]
- Siciliano, R.A.; Reale, A.; Mazzeo, M.F.; Morandi, S.; Silvetti, T.; Brasca, M. Paraprobiotics: A New Perspective for Functional Foods and Nutraceuticals. Nutrients 2021, 13, 1225. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Niro, C.M.; Bresolin, J.D.; Soares, V.F.; Ferreira, M.D.; Sivieri, K.; Azeredo, H.M.C. Dehydrated strawberries for probiotic delivery: Influence of dehydration and probiotic incorporation methods. LWT-Food Sci. Technol. 2021, 144, 111105. [Google Scholar] [CrossRef]
- de Simone, C. The Unregulated Probiotic Market. Clin. Gastroenterol. Hepatol. 2019, 17, 809–817. [Google Scholar] [CrossRef]
- Dobson, A.; Cotter, P.D.; Ross, R.P.; Hill, C. Bacteriocin production: A probiotic trait? Appl. Environ. Microbiol. 2012, 78, 1–6. [Google Scholar] [CrossRef]
- Hernández-González, J.C.; Martínez-Tapia, A.; Lazcano-Hernández, G.; García-Pérez, B.E.; Castrejón-Jiménez, N.S. Bacteriocins from lactic acid bacteria. A powerful alternative as antimicrobials, probiotics, and immunomodulators in veterinary medicine. Animals 2021, 11, 979. [Google Scholar] [CrossRef]
- Cueto, C.; Aragón, S. Evaluation of probiotic potential of lactic acid bacteria to reduce in vitro cholesterol. Sci. Agropecu. 2012, 3, 45–50. [Google Scholar] [CrossRef]
- Li, P.; Gu, Q.; Yang, L.; Yu, Y.; Wang, Y. Characterization of extracellular vitamin B12 producing Lactobacillus plantarum strains and assessment of the probiotic potentials. Food Chem. 2017, 234, 494–501. [Google Scholar] [CrossRef]
- Maldonado Galdeano, C.; Cazorla, S.I.; Lemme Dumit, J.M.; Vélez, E.; Perdigón, G. Beneficial effects of probiotic consumption on the immune system. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar] [CrossRef]
- Azad, M.A.K.; Sarker, M.; Wan, D. Immunomodulatory effects of probiotics on cytokine profiles. Biomed Res. Int. 2018, 2018, 8063647. [Google Scholar] [CrossRef]
- Mandal, S.; Mandal, N.C. Current trends and future perspectives of probiotics on human health: An overview. In Recent Advances and Future Perspectives of Microbial Metabolites; Academic Press: Cambridge, MA, USA, 2023; pp. 81–122. [Google Scholar]
- Van Biervliet, S.; Declercq, D.; Somerset, S. Clinical effects of probiotics in cystic fibrosis patients: A systematic review. Clin. Nutr. ESPEN 2017, 18, 37–43. [Google Scholar] [CrossRef]
- Hernández-Gómez, J.G.; López-Bonilla, A.; Trejo-Tapia, G.; Ávila-Reyes, S.V.; Jiménez-Aparicio, A.R.; Hernández-Sánchez, H. In vitro bile salt hydrolase (BSH) activity screening of different probiotic microorganisms. Foods 2021, 10, 674. [Google Scholar] [CrossRef] [PubMed]
- Aragón-Rojas, S.; Hernández-Álvarez, A.J.; Mainville, I.; Arcand, Y.; Quintanilla-Carvajal, M.X. Effect of the carrier material, drying technology and dissolution media on the viability of Lactobacillus fermentum K73 during simulated gastrointestinal transit. Food Funct. 2020, 11, 2339–2348. [Google Scholar] [CrossRef]
- Feng, K.; Huang, R.-M.; Wu, R.-Q.; Wei, Y.-S.; Zong, M.-H.; Linhardt, R.J.; Wu, H. A novel route for double-layered encapsulation of probiotics with improved viability under adverse conditions. Food Chem. 2019, 310, 125977. [Google Scholar] [CrossRef] [PubMed]
- Fiocco, D.; Longo, A.; Arena, M.P.; Russo, P.; Spano, G.; Capozzi, V. How probiotics face food stress: They get by with a little help. Crit. Rev. Food Sci. Nutr. 2020, 60, 1552–1580. [Google Scholar] [CrossRef]
- Mohammad, S.; Gharibzahedi, T.; Smith, B. Legume proteins are smart carriers to encapsulate hydrophilic and hydrophobic bioactive compounds and probiotic bacteria: A review. Compr. Rev. Food Sci. Food Saf. 2021, 20, 1250–1279. [Google Scholar] [CrossRef]
- Ranadheera, C.S.; Evans, C.A.; Adams, M.C.; Baines, S.K. Effect of dairy probiotic combinations on in vitro gastrointestinal tolerance, intestinal epithelial cell adhesion and cytokine secretion. J. Funct. Foods 2014, 8, 18–25. [Google Scholar] [CrossRef]
- Ma, J.; Xu, C.; Yu, H.; Feng, Z.; Yu, W.; Gu, L.; Liu, Z.; Chen, L.; Jiang, Z.; Hou, J. Food Hydrocolloids Electro-encapsulation of probiotics in gum Arabic-pullulan blend nanofibres using electrospinning technology. Food Hydrocoll. 2021, 111, 106381. [Google Scholar] [CrossRef]
- Rodrigues, F.J.; Cedran, M.F.; Bicas, J.L.; Sato, H.H. Encapsulated probiotic cells: Relevant techniques, natural sources as encapsulating materials and food applications—A narrative review. Food Res. Int. 2020, 137, 109682. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, C.; García-Moreno, P.J.; Mendes, A.C.; Mateiu, R.V.; Chronakis, I.S. Use of Electrohydrodynamic Processing for Encapsulation of Sensitive Bioactive Compounds and Applications in Food. Annu. Rev. Food Sci. Technol. 2018, 9, 525–549. [Google Scholar] [CrossRef]
- Chávarri, M.; Marañon, I.; Villarán, M.C. Encapsulation Technology to Protect Probiotic Bacteria. In Probiotics; Intech: London, UK, 2012; pp. 501–540. [Google Scholar] [CrossRef]
- Anu Bhushani, J.; Anandharamakrishnan, C. Electrospinning and electrospraying techniques: Potential food based applications. Trends Food Sci. Technol. 2014, 38, 21–33. [Google Scholar] [CrossRef]
- Duconseille, A.; Astruc, T.; Quintana, N.; Meersman, F.; Sante-Lhoutellier, V. Gelatin structure and composition linked to hard capsule dissolution: A review. Food Hydrocoll. 2015, 43, 360–376. [Google Scholar] [CrossRef]
- Duman, D.; Karadag, A. Inulin added electrospun composite nanofibres by electrospinning for the encapsulation of probiotics: Characterisation and assessment of viability during storage and simulated gastrointestinal digestion. J. Food Sci. Technol. 2020, 56, 927–935. [Google Scholar] [CrossRef]
- Emrerk, T.; Rungsardthong, V.; Vatanyoopaisarn, S. Processed flavors derived from combined bromelain hydrolyzed jellyfish protein hydrolysate, reducing sugars and arginine. Sci. Eng. Health Stud. 2021, 15, 21030002. [Google Scholar]
- Wu, W.; Li, B.; Hou, H.; Zhang, H.; Zhao, X. Identification of iron-chelating peptides from Pacific cod skin gelatin and the possible binding mode. J. Funct. Foods 2017, 35, 418–427. [Google Scholar] [CrossRef]
- De Prisco, A.; Mauriello, G. Probiotication of foods: A focus on microencapsulation tool. Trends Food Sci. Technol. 2016, 48, 27–39. [Google Scholar] [CrossRef]
- Razavi, S.; Janfaza, S.; Tasnim, N.; Gibson, D.L.; Hoorfar, M. Microencapsulating polymers for probiotics delivery systems: Preparation, characterization, and applications. Food Hydrocoll. 2021, 120, 106882. [Google Scholar] [CrossRef]
- Mendes, A.C.; Stephansen, K.; Chronakis, I.S. Electrospinning of food proteins and polysaccharides. Food Hydrocoll. 2017, 68, 53–68. [Google Scholar] [CrossRef]
- Ricaurte, L.; Quintanilla-Carvajal, M.X. Use of electrospinning technique to produce nanofibres for food industries: A perspective from regulations to characterisations. Trends Food Sci. Technol. 2019, 85, 92–106. [Google Scholar] [CrossRef]
- Ward, S.; Powles, N.T.; Page, M.I. Peptide biomarkers for identifying the species origin of gelatin using coupled UPLC-MS/MS. J. Food Compos. Anal. 2018, 73, 83–90. [Google Scholar] [CrossRef]
- Asadpour, S.; Kargozar, S.; Moradi, L.; Ai, A.; Nosrati, H.; Ai, J. Natural biomacromolecule based composite scaffolds from silk fi broin, gelatin and chitosan toward tissue engineering applications. Int. J. Biol. Macromol. 2019, 154, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Zhang, X.; Li, Y.; Que, F.; Kang, X.; Liu, Y.; Feng, F.; Zhang, H. Characterization of gelatin/zein nanofibers by hybrid electrospinning. Food Hydrocoll. 2018, 75, 72–80. [Google Scholar] [CrossRef]
- Yang, G.; Xiao, Z.; Long, H.; Ma, K.; Zhang, J.; Ren, X. Assessment of the characteristics and biocompatibility of gelatin sponge scaffolds prepared by various crosslinking methods. Sci. Rep. 2018, 8, 1616. [Google Scholar] [CrossRef] [PubMed]
- Abedinia, A.; Mohammadi, A.; Sharifi, M. Poultry gelatin: Characteristics, developments, challenges, and future outlooks as a sustainable alternative for mammalian gelatin. Trends Food Sci. Technol. 2020, 104, 14–26. [Google Scholar] [CrossRef]
- Haug, I.J.; Draget, K.I.; Smidsrød, O. Physical and rheological properties of fish gelatin compared to mammalian gelatin. Food Hydrocoll. 2004, 18, 203–213. [Google Scholar] [CrossRef]
- Guo, L.; Colby, R.H.; Lusignan, C.P.; Whitesides, T.H. Kinetics of Triple Helix Formation in Semidilute Gelatin Solutions. Macromolecules 2003, 36, 9999–10008. [Google Scholar] [CrossRef]
- Ricaurte, L.; Hernández-Carrión, M.; Moyano-Molano, M.; Clavijo-Romero, A.; Quintanilla-Carvajal, M.X. Physical, thermal and thermodynamical study of high oleic palm oil nanoemulsions. Food Chem. 2018, 256, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Coelho, S.C.; Estevinho, B.N.; Rocha, F. Encapsulation in food industry with emerging electrohydrodynamic techniques: Electrospinning and electrospraying—A review. Food Chem. 2021, 339, 127850. [Google Scholar] [CrossRef]
- Gomez-Mascaraque, L.G.; Morfin, R.C.; Pérez-Masiá, R.; Sanchez, G.; Lopez-Rubio, A. Optimization of electrospraying conditions for the microencapsulation of probiotics and evaluation of their resistance during storage and in-vitro digestion. LWT-Food Sci. Technol. 2016, 69, 438–446. [Google Scholar] [CrossRef]
- Mehrali, F.; Ziyadi, H.; Hekmati, M.; Faridi, R.; Mahnaz, M. Kefiran/poly (vinyl alcohol)/poly (vinyl pyrrolidone) composite nanofibers: Fabrication, characterization and consideration of effective parameters in electrospinning. SN Appl. Sci. 2020, 2, 1–13. [Google Scholar] [CrossRef]
- Diep, E.; Schi, J.D. Encapsulating bacteria in alginate-based electrospun nanofiber. Biomater. Sci. 2021, 9, 4364–4373. [Google Scholar] [CrossRef]
- Mojaveri, S.J.; Hosseini, S.F.; Gharsallaoui, A. Viability improvement of Bifidobacterium animalis Bb12 by encapsulation in chitosan/poly(vinyl alcohol) hybrid electrospun fiber mats. Carbohydr. Polym. 2020, 241, 116278. [Google Scholar] [CrossRef]
- Zhong, J.; Mohan, S.D.; Bell, A.; Terry, A.; Mitchell, G.; Davis, F.J. Electrospinning of food grade nanofibres from whey protein. Int. J. Biol. Macromol. 2018, 113, 764–773. [Google Scholar] [CrossRef] [PubMed]
- Aragón-Rojas, S.; Quintanilla-Carvajal, M.X.; Hernández-Sánchez, H. Multifunctional Role of the Whey Culture Medium in the Spray-Drying Microencapsulation of Lactic Acid Bacteria. Food Technol. Biotechnol. 2018, 56, 381–397. [Google Scholar] [CrossRef] [PubMed]
- Ricaurte, L.; Tello-Camacho, E.; Quintanilla-Carvajal, M.X. Hydrolysed Gelatin-Derived, Solvent-Free, Electrospun Nanofibers for Edible Applications: Physical, Chemical and Thermal Behaviour. Food Biophys. 2019, 15, 133–142. [Google Scholar] [CrossRef]
- Wyma, A.; Martin-Alarcon, L.; Walsh, T.; Schmidt, T.A.; Gates, I.D.; Kallos, M.S. Non-Newtonian rheology in suspension cell cultures significantly impacts bioreactor shear stress quantification. Biotechnol. Bioeng. 2018, 115, 2101–2113. [Google Scholar] [CrossRef]
- Zaeim, D.; Sarabi-Jamab, M.; Ghorani, B.; Kadkhodaee, R.; Tromp, R.H. Electrospray assisted fabrication of hydrogel microcapsules by single- and double-stage procedures for encapsulation of probiotics. Food Bioprod. Process. 2017, 102, 250–259. [Google Scholar] [CrossRef]
- López-Rubio, A.; Sanchez, E.; Wilkanowicz, S.; Sanz, Y.; Lagaron, J.M. Electrospinning as a useful technique for the encapsulation of living bifidobacteria in food hydrocolloids. Food Hydrocoll. 2012, 28, 159–167. [Google Scholar] [CrossRef]
- Librán, C.M.; Castro, S.; Lagaron, J.M. Encapsulation by electrospray coating atomization of probiotic strains. Innov. Food Sci. Emerg. Technol. 2017, 39, 216–222. [Google Scholar] [CrossRef]
- Mendes, A.C.; Chronakis, I.S. Electrohydrodynamic encapsulation of probiotics: A review. Food Hydrocoll. 2021, 117, 106688. [Google Scholar] [CrossRef]
- Yu, H.; Liu, W.; Li, D.; Liu, C.; Feng, Z.; Jiang, B. Targeting delivery system for Lactobacillus plantarum based on functionalized electrospun nanofibers. Polymers 2020, 12, 1565. [Google Scholar] [CrossRef] [PubMed]
- Nuryana, I.; Andriani, A.; Lisdiyanti, P. Analysis of organic acids produced by lactic acid bacteria. IOP Conf. Ser. Earth Environ. Sci. 2019, 251, 012054. [Google Scholar] [CrossRef]
- Škrlec, K.; Zupančič, Š.; Prpar Mihevc, S.; Kocbek, P.; Kristl, J.; Berlec, A. Development of electrospun nanofibers that enable high loading and long-term viability of probiotics. Eur. J. Pharm. Biopharm. 2019, 136, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Hoch, E.; Schuh, C.; Hirth, T.; Tovar, G.E.M.; Borchers, K. Stiff gelatin hydrogels can be photo-chemically synthesized from low viscous gelatin solutions using molecularly functionalized gelatin with a high degree of methacrylation. J. Mater. Sci. Mater. Med. 2012, 23, 2607–2617. [Google Scholar] [CrossRef]
- Pang, Z.; Deeth, H.; Prakash, S.; Bansal, N. Development of rheological and sensory properties of combinations of milk proteins and gelling polysaccharides as potential gelatin replacements in the manufacture of stirred acid milk gels and yogurt. J. Food Eng. 2016, 169, 27–37. [Google Scholar] [CrossRef]
- Liu, H.; Cui, S.W.; Chen, M.; Li, Y.; Liang, R.; Xu, F.; Zhong, F. Protective approaches and mechanisms of microencapsulation to the survival of probiotic bacteria during processing, storage and gastrointestinal digestion: A review. Crit. Rev. Food Sci. Nutr. 2019, 59, 2863–2878. [Google Scholar] [CrossRef] [PubMed]
- Solikhin, A.; Mustopa, A.Z.; Putranto, W.S. Partial purification, characterization, and application of extracellular aspartic protease form Lactobacillus casei WSP in producing the bioactive peptides with antibacterial and antioxidant activity. Ann. Bogor. 2018, 22, 47–56. [Google Scholar] [CrossRef]
- Cheng, F.; Chen, H.; Lei, N.; Zhang, M.; Wan, H.; Shu, G. Effect of prebiotics, inorganic salts and Amino Acids for cell envelope proteinase production from Lactobacillus plantarum Lp69. Acta Sci. Pol. Technol. Aliment. 2019, 18, 269–278. [Google Scholar] [CrossRef]
- Shu, G.; Zhang, B.; Chen, S.; Wan, H.; Chen, H.E. Effect of amino acids added to culture medium on the growth and survival of Lactobacillus bulgaricus LB6 during freeze-drying. Food Technol. 2017, 41, 106–117. [Google Scholar]
- Burgain, J.; Gaiani, C.; Francius, G.; Revol-Junelles, A.M.; Cailliez-Grimal, C.; Lebeer, S.; Tytgat, H.L.P.; Vanderleyden, J.; Scher, J. In vitro interactions between probiotic bacteria and milk proteins probed by atomic force microscopy. Colloids Surf. B 2012, 104, 153–162. [Google Scholar] [CrossRef]
- Huang, S.; Vignolles, M.-L.; Chen, X.D.; Le Loir, Y.; Jan, G.; Schuck, P.; Jeantet, R. Spray drying of probiotics and other food-grade bacteria: A review. Trends Food Sci. Technol. 2017, 63, 1–17. [Google Scholar] [CrossRef]
- Jain, S.; Valvi, P.U.; Swarnakar, N.K.; Thanki, K. Gelatin coated hybrid lipid nanoparticles for oral delivery of Amphotericin B. Mol. Pharm. 2012, 9, 2542–2553. [Google Scholar] [CrossRef] [PubMed]
- Gunzburg, W.H.; Myo Myint Aung1, P.T.; Ng, S.; Read, E.; Tan, W.J.; Brandtner, L.; Dangerfield, J.; Salmons, B. Efficient Protection of Probiotics for Delivery to the Gastric 2 Tract by Cellulose Sulphate Encapsulation. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Pang, Z.; Deeth, H.; Sopade, P.; Sharma, R.; Bansal, N. Rheology, texture and microstructure of gelatin gels with and without milk proteins. Food Hydrocoll. 2014, 35, 484–493. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Jia, L.; Qin, X.H. The effect of different surfactants on the electrospinning poly(vinyl alcohol) (PVA) nanofibers. J. Therm. Anal. Calorim. 2013, 112, 595–605. [Google Scholar] [CrossRef]
- Robert, C.; Couëdelo, L.; Vaysse, C.; Michalski, M.-C. Vegetable lecithins: A review of their compositional diversity, impact on lipid metabolism and potential in cardiometabolic disease prevention. Biochimie 2020, 169, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Aro, H.; Järvenpää, E.; Mäkinen, J.; Lauraeus, M.; Huopalahti, R.; Hietaniemi, V. The utilization of oat polar lipids produced by supercritical fluid technologies in the encapsulation of probiotics. LWT-Food Sci. Technol. 2013, 53, 540–546. [Google Scholar] [CrossRef]
- Zhuang, X.; Gaudino, N.; Clark, S.; Acevedo, N.C. Novel lecithin-based oleogels and oleogel emulsions delay lipid oxidation and extend probiotic bacteria survival. LWT-Food Sci. Technol. 2021, 136, 110353. [Google Scholar] [CrossRef]
- Donthidi, A.R.; Tester, R.F.; Aidoo, K.E. Effect of lecithin and starch on alginate-encapsulated probiotic bacteria. J. Microencapsul. 2010, 27, 67–77. [Google Scholar] [CrossRef]
- Pham, L.Q.; Uspenskaya, M.V.; Olekhnovich, R.O.; Bernal, R.A.O. A review on electrospun pvc nanofibers: Fabrication, properties, and application. Fibers 2021, 9, 12. [Google Scholar] [CrossRef]
- Tapia-Hernández, J.A.; Torres-Chávez, P.I.; Ramírez-Wong, B.; Rascón-Chu, A.; Plascencia-Jatomea, M.; Barreras-Urbina, C.G.; Rangel-Vázquez, N.A.; Rodríguez-Félix, F. Micro- and Nanoparticles by Electrospray: Advances and Applications in Foods. J. Agric. Food Chem. 2015, 63, 4699–4707. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Salam, M.H.; El-Shibiny, S. Preparation and properties of milk proteins-based encapsulated probiotics: A review. Dairy Sci. Technol. 2015, 95, 393–412. [Google Scholar] [CrossRef]
- Rasouli, M.; Pirsalami, S.; Zebarjad, S.M. Study on the formation and structural evolution of bead-on-string in electrospun polysulfone mats. Polym. Int. 2020, 69, 822–832. [Google Scholar] [CrossRef]
- Ubbink, J.; Schär-Zammaretti, P. The cell wall of lactic acid bacteria: Surface constituents and macromolecular conformations. Biophys. J. 2003, 85, 4076–4092. [Google Scholar]
- Martín, M.J.; Lara-Villoslada, F.; Ruiz, M.A.; Morales, M.E. Microencapsulation of bacteria: A review of different technologies and their impact on the probiotic effects. Innov. Food Sci. Emerg. Technol. 2015, 27, 15–25. [Google Scholar] [CrossRef]
- Hu, B.; Tian, F.; Wang, G.; Zhang, Q.; Zhao, J.; Zhang, H.; Chen, W. Enhancement of bile resistance in Lactobacillus plantarum strains by soy lecithin. Lett. Appl. Microbiol. 2015, 61, 13–19. [Google Scholar] [CrossRef]
- Bollom, M.A.; Clark, S.; Acevedo, N.C. Edible lecithin, stearic acid, and whey protein bigels enhance survival of probiotics during in vitro digestion. Food Biosci. 2021, 39, 100813. [Google Scholar] [CrossRef]





| Mixture | Ratio | Surface Tension (mN m−1) | Viscosity (mPa s−1) | Conductivity (mS cm−1) | Ph | |
|---|---|---|---|---|---|---|
| Gelatin | Culture | |||||
| 1 | 0.20 | 0.80 | 42.9 ± 0.03 a | 4.80 ± 0.28 d | 4.88 ± 0.25 a | 4.10 ± 0.06 a |
| 2 | 0.35 | 0.65 | 39.3 ± 0.025 b | 7.90 ± 0.70 d | 4.10 ± 0.17 b | 4.20 ± 0.00 a |
| 3 | 0.40 | 0.60 | 40.8 ± 1.76 ab | 19.55 ± 0.49 c | 3.76 ± 0.08 b | 4.40 ± 0.11 a |
| 4 | 0.50 | 0.50 | 42.0 ± 0.62 ab | 23.90 ± 0.56 c | 1.95 ± 0.03 c | 4.40 ± 0.05 a |
| 5 | 0.60 | 0.40 | 42.6 ± 0.98 a | 33.95 ± 2.19 b | 1.73 ± 0.04 c | 4.40 ± 0.04 a |
| 6 | 0.65 | 0.35 | 42.7 ± 1.08 a | 34.10 ± 1.6 b | 1.58 ± 0.00 c | 4.50 ± 0.19 a |
| 7 | 0.80 | 0.20 | 42.5 ± 0.82 a | 48.58 ± 1.38 a | 1.92 ± 0.07 c | 4.90 ± 0.02 a |
| Correlation | Pearson Correlation Coefficient | p-Value |
|---|---|---|
| A/B and pH | 0.937 | 0.002 |
| A/B and viscosity | 0.915 | 0.004 |
| B/A and conductivity | 0.916 | 0.004 |
| B/A and pH | −0.764 | 0.045 |
| B/A and viscosity | −0.858 | 0.013 |
| B/A and bacterial cycles change under bile salts | −0.830 | 0.021 |
| Conductivity and bacterial cycles change under bile salts | −0.953 | 0.001 |
| Run | Factor wt% (Wall Material) | Response Variable: Bacterial Cycles Change log (CFU/mL) | |||
|---|---|---|---|---|---|
| Gelatin [A] | Culture [B] | After Mixture | Gastric pH | Bile Salt | |
| 1 | 0.40 | 0.60 | 0.04 | 0.09 | −1.87 |
| 2 | 0.65 | 0.35 | 0.19 | −0.16 | −1.25 |
| 3 | 0.20 | 0.80 | −0.07 | −3.18 | −2.48 |
| 4 | 0.60 | 0.40 | 0.10 | −0.56 | −1.11 |
| 5 | 0.50 | 0.50 | 0.05 | 0.01 | −1.22 |
| 6 | 0.80 | 0.20 | −0.05 | −2.05 | −1.39 |
| 7 | 0.20 | 0.80 | −0.06 | −3.20 | −1.98 |
| 8 | 0.80 | 0.20 | −0.08 | −3.15 | −1.70 |
| 9 | 0.35 | 0.65 | 0.15 | −1.59 | −1.59 |
| 10 | 0.80 | 0.20 | −0.08 | −2.01 | −1.55 |
| p-value | 0.0008 | 0.0005 | 0.0080 | ||
| Source | Change in Bacterial Cycles | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| After Mixture | Gastric pH | Bile Salts | |||||||||||||
| SS * | Df ** | Mean Square | F-Value | p-Value | SS * | Df ** | Mean Square | F-Value | p-Value | SS * | Df ** | Mean Square | F-Value | p-Value | |
| Model | 0.09 | 4 | 0.02 | 34.5 | 0.0008 | 14.6 | 2 | 7.29 | 27.6 | 0.0005 | 1.39 | 2 | 0.69 | 10.4 | 0.0080 |
| Linear mixture | 6.19 × 10−4 | 1 | 6.19 × 10−4 | 0.95 | 0.3740 | 0.280 | 1 | 0.280 | 1.05 | 0.3399 | 0.59 | 1 | 0.59 | 8.89 | 0.0204 |
| AB | 9.07 × 10−3 | 1 | 9.07 × 10−3 | 13.9 | 0.0135 | 14.3 | 1 | 14.3 | 54.1 | 0.0002 | 0.80 | 1 | 0.80 | 11.90 | 0.0107 |
| AB(A-B) | 2.25 × 10−3 | 1 | 2.25 × 10−3 | 3.46 | 0.1220 | ||||||||||
| AB(A-B)2 | 0.02 | 1 | 0.023 | 34.7 | 0.0020 | ||||||||||
| Residual | 3.25 × 10−3 | 5 | 6.50 × 10−4 | 1.85 | 7 | 0.260 | 0.47 | 7 | 0.07 | ||||||
| Lack of fit | 2.60 × 10−3 | 2 | 1.30 × 10−3 | 6.00 | 0.0894 | 1.01 | 4 | 0.250 | 0.91 | 0.5543 | 0.29 | 4 | 0.07 | 1.28 | 0.4376 |
| Pure error | 6.50 × 10−4 | 3 | 2.17 × 10−4 | 0.84 | 3 | 0.280 | 0.17 | 3 | 0.06 | ||||||
| Corr. Total | 0.09 | 9 | 16.43 | 9 | 1.86 | 9 | |||||||||
| R2 | 0.96 | 0.88 | 0.74 | ||||||||||||
| Equation | N (log CFU/mL) = −0.045358 ∗ A − 0.040809 ∗ B + 1.73313 × 10−3 ∗ A ∗ B + 1.39291 × 10−6 ∗ B ∗ A ∗ (A − B) + 2.54879 × 10−7 ∗ A∗ B ∗ (A − B)2 | N (log CFU/mL Log) = −0.070443 ∗ A − 0.084910 ∗ B + 3.09535 × 10−3 ∗ A ∗ B | N (log CFU/mL) = 0.024460 ∗ A − 0.036966 ∗ B + 7.29884 × 10−4 ∗ A ∗ B | ||||||||||||
| Factors | Response Variable Encapsulation Yield and Survival (%) | |||
|---|---|---|---|---|
| Run | Bacteria Culture (g/L) | Soy Lecithin (%wt/v) | Collector Distance (cm) | |
| 1 | 150 | 5.0 | 9.0 | 65 |
| 2 | 200 | 2.5 | 9.0 | 67 |
| 3 | 150 | 2.5 | 8.0 | 66 |
| 4 | 100 | 2.5 | 7.0 | 73 |
| 5 | 200 | 2.5 | 7.0 | 87 |
| 6 | 150 | 2.5 | 8.0 | 69 |
| 7 | 100 | 5.0 | 8.0 | 65 |
| 8 | 150 | 2.5 | 8.0 | 70 |
| 9 | 100 | 0.0 | 8.0 | 75 |
| 10 | 100 | 2.5 | 9.0 | 71 |
| 11 | 150 | 2.5 | 8.0 | 68 |
| 12 | 150 | 5.0 | 7.0 | 63 |
| 13 | 200 | 5.0 | 8.0 | 63 |
| 14 | 150 | 0.0 | 9.0 | 86 |
| 15 | 150 | 2.5 | 8.0 | 61 |
| Sum of Squares | DF * | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|
| Model | 752.71 | 9 | 83.63 | 4.98 | 0.0459 |
| [A]-Bacteria culture | 76.80 | 1 | 76.80 | 4.57 | 0.0855 |
| [B]-Soy lecithin | 450.67 | 1 | 450.67 | 26.83 | 0.0035 |
| [C]-Collector distance | 76.80 | 1 | 76.80 | 4.57 | 0.0855 |
| AB | 78.11 | 1 | 78.11 | 4.65 | 0.0835 |
| AC | 72.25 | 1 | 72.25 | 4.30 | 0.0928 |
| BC | 24.11 | 1 | 24.11 | 1.44 | 0.2846 |
| A2 | 17.14 | 1 | 17.14 | 1.02 | 0.3587 |
| B2 | 70.08 | 1 | 70.08 | 4.17 | 0.0965 |
| C2 | 89.05 | 1 | 89.05 | 5.30 | 0.0695 |
| Residual | 83.97 | 5 | 16.79 | ||
| Lack of Fit | 37.50 | 1 | 37.50 | 3.23 | 0.1468 |
| Pure Error | 46.47 | 4 | 11.62 | ||
| R2 | 0.899 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano-Delgado, A.; Quintanilla-Carvajal, M.X. Electrospinning Microencapsulation of Lactobacillus fermentum K73 Using Gelatin as the Main Component of a Food-Grade Matrix. Microorganisms 2023, 11, 2682. https://doi.org/10.3390/microorganisms11112682
Serrano-Delgado A, Quintanilla-Carvajal MX. Electrospinning Microencapsulation of Lactobacillus fermentum K73 Using Gelatin as the Main Component of a Food-Grade Matrix. Microorganisms. 2023; 11(11):2682. https://doi.org/10.3390/microorganisms11112682
Chicago/Turabian StyleSerrano-Delgado, Arjana, and María Ximena Quintanilla-Carvajal. 2023. "Electrospinning Microencapsulation of Lactobacillus fermentum K73 Using Gelatin as the Main Component of a Food-Grade Matrix" Microorganisms 11, no. 11: 2682. https://doi.org/10.3390/microorganisms11112682





