Changes in the Proliferation of the Neural Progenitor Cells of Adult Mice Chronically Infected with Toxoplasma gondii
Abstract
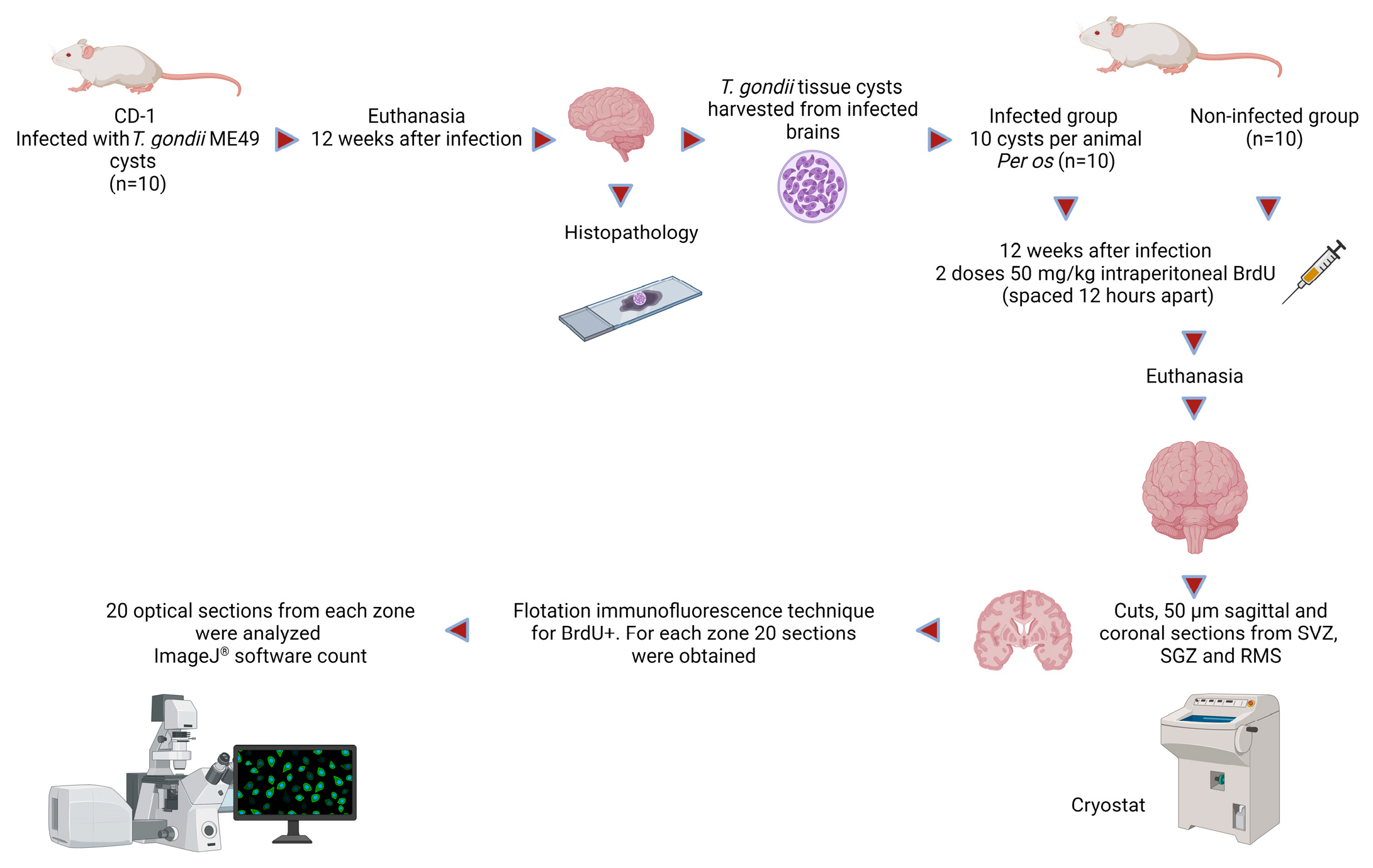
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Parasites
2.3. Purification of ME49 T. gondii Tissue Cysts and Mouse Infection
2.4. BrdU Administration and Preparation of Coronal and Sagittal Sections of the Subventricular Zone, Subgranular Dentate Gyrus Zone, and Rostral Migratory Stream
2.5. Histology Process
2.6. Flotation Immunofluorescence Technique for the Identification of NPC BrdU+
2.7. Scanning and Counting of BrdU+ Cells
2.8. Statistics
3. Results
BrdU-Positive Cells’ Evaluation by Confocal Scanning
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gaddi, P.J.; Yap, G.S. Cytokine regulation of immunopathology in toxoplasmosis. Immunol. Cell Biol. 2007, 85, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Attia, M.M.; El-Gameel, S.M.; Ismael, E. Evaluation of tumor necrosis factor-alpha (TNF-α); gamma interferon (IFN-γ) genes and oxidative stress in sheep: Immunological responses induced by Oestrus ovis (Diptera: Oestridae) infestation. J. Parasit. Dis. 2020, 44, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Castro-Elizalde, K.N.; Hernández-Contreras, P.; Ramírez-Flores, C.J.; González-Pozos, S.; Gómez de León, C.T.; Mondragón-Castelán, M.; Mondragón-Flores, R. Mycophenolic acid induces differentiation of Toxoplasma gondii RH strain tachyzoites into bradyzoites and formation of cyst-like structure in vitro. Parasitol. Res. 2018, 117, 547–563. [Google Scholar] [CrossRef] [PubMed]
- Sullivan Jr, W.J.; Smith, A.T.; Joyce, B.R. Understanding mechanisms and the role of differentiation in pathogenesis of Toxoplasma gondii: A review. Mem. Inst. Oswaldo Cruz 2009, 104, 155–161. [Google Scholar] [CrossRef]
- Delgado, I. Unraveling the Replication Process of Toxoplasma gondii through the MOB1 Protein; Doutor em Ciências Veterinárias na especialidade de de Ciências Biológicas e Biomédicas, Universidade de Lisboa: Lisboa, Portugal, 2022. [Google Scholar]
- Augusto, L.; Martynowicz, J.; Amin, P.H.; Carlson, K.R.; Wek, R.C.; Sullivan, W.J. TgIF2K-B is an eIF2α kinase in Toxoplasma gondii that responds to oxidative stress and optimizes pathogenicity. MBio 2021, 12, 1110–1128. [Google Scholar] [CrossRef]
- Ribeiro-Andrade, M.; Carvalho, J.D.C.S.; da Silva, R.A.; da Conceição Carvalho, M.; Porto, W.J.N.; Mota, R.A. Inter-and intra-genotype differences in induced cystogenesis of recombinant strains of Toxoplasma gondii isolated from chicken and pigs. Exp. Parasitol. 2019, 207, 107775. [Google Scholar] [CrossRef]
- Sinai, A.P.; Watts, E.A.; Dhara, A.; Murphy, R.D.; Gentry, M.S.; Patwardhan, A. Reexamining Chronic Toxoplasma gondii Infection: Surprising Activity for a “Dormant” Parasite. Curr. Clin. Microbiol. Rep. 2016, 3, 175–185. [Google Scholar] [CrossRef]
- Chaker, Z.; Codega, P.; Doetsch, F. A mosaic world: Puzzles revealed by adult neural stem cell heterogeneity. Wiley Interdiscip. Rev. Dev. 2016, 5, 640–658. [Google Scholar] [CrossRef]
- Shohayeb, B.; Diab, M.; Ahmed, M.; Ng, D.C.H. Factors that influence adult neurogenesis as potential therapy. Transl. Neurodegener. 2018, 7, 4. [Google Scholar] [CrossRef]
- Turnquist, C.; Harris, B.T.; Harris, C.C. Radiation-induced brain injury: Current concepts and therapeutic strategies targeting neuroinflammation. Neuro-Oncol. Adv. 2020, 2, vdaa057. [Google Scholar] [CrossRef]
- Hermes, G.; Ajioka, J.W.; Kelly, K.A.; Mui, E.; Roberts, F.; Kasza, K.; Mayr, T.; Kirisits, M.J.; Wollmann, R.; Ferguson, D.J.; et al. Neurological and behavioral abnormalities, ventricular dilatation, altered cellular functions, inflammation, and neuronal injury in brains of mice due to common, persistent, parasitic infection. J. Neuroinflamm. 2008, 5, 48. [Google Scholar] [CrossRef]
- Wang, T.; Sun, X.; Qin, W.; Zhang, X.; Wu, L.; Li, Y.; Cong, H. From inflammatory reactions to neurotransmitter changes: Implications for understanding the neurobehavioral changes in mice chronically infected with Toxoplasma gondii. Behav. Brain Res. 2019, 359, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J. Behavioral changes induced by latent toxoplasmosis could arise from CNS inflammation and neuropathogenesis. In Microorganisms and Mental Health; Springer: Berlin/Heidelberg, Germany, 2022; pp. 303–313. [Google Scholar]
- Bond, A.M.; Ming, G.-L.; Song, H. Adult Mammalian Neural Stem Cells and Neurogenesis: Five Decades Later. Cell Stem Cell 2015, 17, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Płatek, R.; Rogujski, P.; Mazuryk, J.; Wiśniewska, M.B.; Kaczmarek, L.; Czupryn, A. Impaired Generation of Transit-Amplifying Progenitors in the Adult Subventricular Zone of Cyclin D2 Knockout Mice. Cells 2022, 11, 135. [Google Scholar] [CrossRef]
- Abbott, L.C.; Nigussie, F. Adult neurogenesis in the mammalian dentate gyrus. Anat. Histol. Embryol. 2020, 49, 3–16. [Google Scholar] [CrossRef]
- Abdelhak, B.; Phuong, B.; Tran, D.R.; Richard, J.M. Chemokines regulate the migration of neural progenitors to sites of neuroinflammation. J. Neurosci 2006, 26, 3182–3191. [Google Scholar] [CrossRef]
- Bénédicte, M.; Garcia-Verdugo, J.M.; Yaschine, C.; Gonzalez-Perez, O.; Rowitch, D.; Alvarez-Buylla, A. Origin of Oligodendrocytes in the Subventricular Zone of the Adult Brain. J. Neurosci. 2006, 26, 7907. [Google Scholar] [CrossRef]
- Nait-Oumesmar, B.; Decker, L.; Lachapelle, F.; Avellana-Adalid, V.; Bachelin, C.; Baron, A.; Evercooren, V. Progenitor cells of the adult mouse subventricular zone proliferate, migrate and differentiate into oligodendrocytes after demyelination. Eur. J. Neurosci. 1999, 11, 4357–4366. [Google Scholar] [CrossRef]
- Brousse, B.; Magalon, M.; Durbec, P.; Cayre, M. Region and dynamic specificities of adult neural stem cells and oligodendrocyte precursors in myelin regeneration in the mouse brain. Biol. Open 2015, 4, 980–992. [Google Scholar] [CrossRef]
- Xing, X.Y.; Philipp, T.; Röth, J.; Stratton, A.S.; Bernard, H.A.; Chuang, J.D.; Ellis, S.; Woei, N.; Kilpatrick, T.; Merson, T. Adult Neural Precursor Cells from the Subventricular Zone Contribute Significantly to Oligodendrocyte Regeneration and Remyelination. J. Neurosci. 2014, 34, 14128. [Google Scholar] [CrossRef]
- Zong, W.; Gouda, M.; Cai, E.; Wang, R.; Xu, W.; Wu, Y.; Lorenzo, J.M. The antioxidant phytochemical schisandrin a promotes neural cell proliferation and differentiation after ischemic brain injury. Molecules 2021, 26, 7466. [Google Scholar] [CrossRef]
- Gan, X.; Zhang, X.; Cheng, Z.; Chen, L.; Ding, X.; Du, J.; Cai, Y.; Luo, Q.; Shen, J.; Wang, Y.; et al. Toxoplasma gondii inhibits differentiation of C17.2 neural stem cells through Wnt/β-catenin signaling pathway. Biochem. Biophys. Res. Commun. 2016, 473, 187–193. [Google Scholar] [CrossRef]
- NOM-062-ZOO-1999; Especificaciones Técnicas Para la Producción, Cuidado y uso de los Animales de Laboratorio. NORMA Oficial Mexicana: Gobierno de México, Mexico. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwiC1tjvl56CAxWrJEQIHQWWCrUQFnoECBEQAQ&url=https%3A%2F%2Fwww.gob.mx%2Fcms%2Fuploads%2Fattachment%2Ffile%2F203498%2FNOM-062-ZOO-1999_220801.pdf&usg=AOvVaw0w8KmBjbQNtDTmfAUv92Bv&opi=89 (accessed on 25 October 2023).
- Watson, G.F.; Davis, P.H. Systematic review and meta-analysis of variation in Toxoplasma gondii cyst burden in the murine model. Exp. Parasitol. 2019, 196, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Tuttle, A.H.; Philip, V.M.; Chesler, E.J.; Mogil, J.S. Comparing phenotypic variation between inbred and outbred mice. Nat. Methods 2018, 15, 994–996. [Google Scholar] [CrossRef] [PubMed]
- Haroon, F.; Händel, U.; Angenstein, F.; Goldschmidt, J.; Kreutzmann, P.; Lison, H.; Fischer, K.D.; Scheich, H.; Wetzel, W.; Schlüter, D.; et al. Toxoplasma gondii Actively Inhibits Neuronal Function in Chronically Infected Mice. PLoS ONE 2012, 7, e35516. [Google Scholar] [CrossRef]
- Wojtowicz, J.M.; Kee, N. BrdU assay for neurogenesis in rodents. Nat. Protoc. 2006, 1, 1399–1405. [Google Scholar] [CrossRef] [PubMed]
- Nowakowski, R.S.; Lewin, S.B.; Miller, M.W. Bromodeoxyuridine immunohistochemical determination of the lengths of the cell cycle and the DNA-synthetic phase for an anatomically defined population. J. Neurocytol. 1989, 18, 311–318. [Google Scholar] [CrossRef]
- Kee, N.; Sivalingam, S.; Boonstra, R.; Wojtowicz, J.M. The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J. Neurosci. Methods 2002, 11, 97–105. [Google Scholar] [CrossRef]
- Berenreiterová, M.; Flegr, J.; Kuběna, A.A.; Němec, P. The Distribution of Toxoplasma gondii Cysts in the Brain of a Mouse with Latent Toxoplasmosis: Implications for the Behavioral Manipulation Hypothesis. PLoS ONE 2011, 6, e28925. [Google Scholar] [CrossRef]
- Grishagin, I.V. Automatic cell counting with ImageJ. Anal. Biochem. 2015, 473, 63–75. [Google Scholar] [CrossRef]
- Basler, L.; Gerdes, S.; Wolfer, D.P.; Slomianka, L. Sampling the Mouse Hippocampal Dentate Gyrus. Front. Neuroanat. 2017, 11, 123. [Google Scholar] [CrossRef] [PubMed]
- Melzer, T.C.; Cranston, H.J.; Weiss, L.M.; Halonen, S.K. Host Cell Preference of Toxoplasma gondii Cysts in Murine Brain: A Confocal Study. J. Neuroparasitol. 2010, 1, N100505. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Dubey, J.P. Isolation and genotyping of Toxoplasma gondii strains. In Toxoplasma gondii: Methods and Protocols; Humana: New York, NY, USA, 2020; pp. 49–80. [Google Scholar] [CrossRef]
- Wang, Q.; Sibley, L.D. Assays for monitoring Toxoplasma gondii infectivity in the laboratory mouse. In Toxoplasma gondii: Methods and Protocols; Humana: New York, NY, USA, 2020; pp. 99–116. [Google Scholar] [CrossRef]
- Wang, J.L.; Li, T.T.; Elsheikha, H.M.; Liang, Q.L.; Zhang, Z.W.; Wang, M.; Zhu, X.Q. The protein phosphatase 2A holoenzyme is a key regulator of starch metabolism and bradyzoite differentiation in Toxoplasma gondii. Nat. Commun. 2022, 13, 7560. [Google Scholar] [CrossRef] [PubMed]
- Afonso, C.; Paixão, V.B.; Costa, R.M. Chronic Toxoplasma Infection Modifies the Structure and the Risk of Host Behavior. PLoS ONE 2012, 7, e32489. [Google Scholar] [CrossRef] [PubMed]
- Pittman, K.J.; Aliota, M.T.; Knoll, L.J. Dual transcriptional profiling of mice and Toxoplasma gondii during acute and chronic infection. BMC Genom. 2014, 15, 806. [Google Scholar] [CrossRef] [PubMed]
- Cooper-Kuhn, C.M.; Georg-Kuhn, H. Is it all DNA repair?: Methodological considerations for detecting neurogenesis in the adult brain. Brain Res. Dev. 2002, 134, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Gionchiglia, N.; Granato, A.; Merighi, A.; Lossi, L. Association of Caspase 3 Activation and H2AX γ phosphorylation in the aging brain: Studies on untreated and irradiated mice. Biomedicines 2021, 9, 1166. [Google Scholar] [CrossRef]
- Martínez-Cerdeño, V.; Noctor, S.C. Neural Progenitor Cell Terminology. Front. Neuroanat. 2018, 12, 104. [Google Scholar] [CrossRef]
- Liang, H.; Zhao, H.; Gleichman, A.; Machnicki, M.; Telang, S.; Tang, S.; Carmichael, S.T. Region-specific and activity-dependent regulation of SVZ neurogenesis and recovery after stroke. Proc. Natl. Acad. Sci. USA 2019, 116, 13621–13630. [Google Scholar] [CrossRef]
- Kuipers, S.D.; Bramham, C.R.; Cameron, H.A.; Fitzsimons, C.P.; Korosi, A.; Lucassen, P.J. Modulation of Adult Hippocampal Neurogenesis by Early-Life Environmental Challenges Triggering Immune Activation. Neural Plast. 2014, 2014, 194396. [Google Scholar] [CrossRef]
- Sharma, A.; Valadi, N.; Miller, A.H.; Pearce, B.D. Neonatal Viral Infection Decreases Neuronal Progenitors and Impairs Adult Neurogenesis in the Hippocampus. Neurobiol. Dis. 2002, 11, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Rosa-Fernandes, L.; Cugola, F.R.; Russo, F.B.; Kawahara, R.; Freire, C.C.; Leite, P.E.C.; Palmisano, G. Zika Virus Impairs Neurogenesis and Synaptogenesis Pathways in Human Neural Stem Cells and Neurons. Front. Cell. Neurosci. 2019, 13, 64. [Google Scholar] [CrossRef] [PubMed]
- Putatunda, R.; Ho, W.Z.; Hu, W. HIV-1 and Compromised Adult Neurogenesis: Emerging Evidence for a New Paradigm of HAND Persistence. AIDS Rev. 2019, 21, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Miranda, A.S.; Brant, F.; Campos, A.C.; Vieira, L.B.; Rocha, N.P.; Cisalpino, D.; Binda, N.S.; Rodrigues, D.H.; Ransohoff, R.M.; Machado, F.S.; et al. Evidence for the contribution of adult neurogenesis and hippocampal cell death in experimental cerebral malaria cognitive outcome. Neuroscience 2015, 284, 920–933. [Google Scholar] [CrossRef] [PubMed]
- Jalland, C.M.; Scheffler, K.; Benestad, S.L.; Moldal, T.; Ersdal, C.; Gunnes, G.; Suganthan, R.; Bjørås, M.; Tranulis, M.A. Neil3 induced neurogenesis protects against prion disease during the clinical phase. Sci. Rep. 2016, 6, 37844. [Google Scholar] [CrossRef]
- Sawada, N.; Kotani, T.; Konno, T.; Setiawan, J.; Nishigaito, Y.; Saito, Y.; Matozaki, T. Regulation by commensal bacteria of neurogenesis in the subventricular zone of adult mouse brain. Biochem. Biophys. Res. Commun. 2018, 498, 824–829. [Google Scholar] [CrossRef]
- Kundu, P.; Lee, H.U.; Garcia-Perez, I.; Tay, E.X.Y.; Kim, H.; Faylon, L.E.; Pettersson, S. Neurogenesis and prolongevity signaling in young germ-free mice transplanted with the gut microbiota of old mice. Sci. Transl. Med. 2019, 11, eaau4760. [Google Scholar] [CrossRef]
- Liebetanz, D.; Gerber, J.; Schiffner, C.; Schütze, S.; Klinker, F.; Jarry, H.; Nau, R.; Tauber, S.C. Pre-infection physical exercise decreases mortality and stimulates neurogenesis in bacterial meningitis. J. Neuroinflamm. 2012, 9, 168. [Google Scholar] [CrossRef]
- Guzzetta, K.E.; Cryan, J.F.; O’Leary, O.F. Microbiota-gut-brain axis regulation of adult hippocampal neurogenesis. Brain Plast. 2022, 8, 97–119. [Google Scholar] [CrossRef]
- Zhang, X.; Su, R.; Cheng, Z.; Zhu, W.; Li, Y.; Wang, Y.; Du, J.; Cai, Y.; Luo, Q.; Shen, J.; et al. A mechanistic study of Toxoplasma gondii ROP18 inhibiting differentiation of C17.2 neural stem cells. Parasites Vectors 2017, 10, 585. [Google Scholar] [CrossRef]
- Zhou, J.; Gan, X.; Wang, Y.; Zhang, X.; Ding, X.; Chen, L.; Du, J.; Luo, Q.; Wang, T.; Shen, J.; et al. Toxoplasma gondii prevalent in China induce weaker apoptosis of neural stem cells C17.2 via endoplasmic reticulum stress (ERS) signaling pathways. Parasites Vectors 2015, 8, 73. [Google Scholar] [CrossRef]
- Arrizabalaga, G. A Fine Balance between Life and Death: Modulation of BCL-2 Family Members by Toxoplasma Gondii. Front. Microbiol. 2011, 2, 39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hu, W. NFκB signaling regulates embryonic and adult neurogenesis. Front. Biol. 2012, 7, 277–291. [Google Scholar] [CrossRef]
- Zhang, R.; Xue, Y.Y.; Lu, S.D.; Wang, Y.; Zhang, L.M.; Huang, Y.L.; Signore, A.P.; Chen, J.; Sun, F.Y. Bcl-2 enhances neurogenesis and inhibits apoptosis of newborn neurons in adult rat brain following a transient middle cerebral artery occlusion. Neurobiol. Dis. 2006, 24, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Rumman, M.; Pandey, S.; Singh, B.; Gupta, M.; Mahdi, A.A. Genistein suppresses microglial activation and inhibits apoptosis in different brain regions of hypoxia-exposed mice model of amnesia. Metab. Brain Dis. 2022, 37, 2521–2532. [Google Scholar] [CrossRef]
- Picard-Riera, N.; Decker, L.; Delarasse, C.; Goude, K.; Nait-Oumesmar, B.; Liblau, R.; Pham-Dinh, D.; Evercooren, A. Experimental autoimmune encephalomyelitis mobilizes neural progenitors from the subventricular zone to undergo oligodendrogenesis in adult mice. Proc. Natl. Acad. Sci. USA 2002, 99, 13211–13216. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Huang, J.; Young, K. Neural Stem and Progenitor Cells in Nervous System Function and Therapy. Stem Cells Int. 2016, 2016, 1890568. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Castro, B.E.; Reyes-García, J.G.; Valenzuela-Vargas, M.T.; Martínez-Gómez, F. Histopathology of murine toxoplasmosis under treatment with dialyzable leukocyte extract. Mem. Inst. Oswaldo Cruz 2017, 112, 741–747. [Google Scholar] [CrossRef]
- Sun, D. The potential of endogenous neurogenesis for brain repair and regeneration following traumatic brain injury. Neural Regen. Res. 2014, 9, 688–692. [Google Scholar] [CrossRef]
- Amor, S.; Woodroofe, M.N. Innate and adaptive immune responses in neurodegeneration and repair. Immunology 2014, 141, 287–291. [Google Scholar] [CrossRef]
- Li, Y.; Severance, E.G.; Viscidi, R.P.; Yolken, R.H.; Xiao, J. Persistent Toxoplasma Infection of the Brain Induced Neurodegeneration Associated with Activation of Complement and Microglia. Infect. Immun. 2019, 87, 10–1128. [Google Scholar] [CrossRef]
- Baron, R.; Nemirovsky, A.; Harpaz, I.; Cohen, H.; Owens, T.; Monsonego, A. IFN-γ enhances neurogenesis in wild-type mice and in a mouse model of Alzheimer’s disease. FASEB J. 2008, 22, 2843–2852. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.; Goldshmit, Y.; Turnley, A.M. Interferon-γ but not TNFα promotes neuronal differentiation and neurite outgrowth of murine adult neural stem cells. Exp. Neurol. 2004, 187, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Araki, T.; Ikegaya, Y.; Koyama, R. The effects of microglia-and astrocyte-derived factors on neurogenesis in health and disease. Eur. J. Neurosci. 2021, 54, 5880–5901. [Google Scholar] [CrossRef] [PubMed]
- Lang, D.; Schott, B.H.; Ham, M.; Morton, L.; Kulikovskaja, L.; Herrera-Molina, R.; Dunay, I.R. Chronic Toxoplasma infection is associated with distinct alterations in the synaptic protein composition. J. Neuroinflamm. 2018, 15, 216. [Google Scholar] [CrossRef]
- Hwang, Y.S.; Shin, J.H.; Yang, J.P.; Jung, B.K.; Lee, S.H.; Shin, E.H. Characteristics of Infection Immunity Regulated by Toxoplasma gondii to Maintain Chronic Infection in the Brain. Front. Immunol. 2018, 9, 158. [Google Scholar] [CrossRef]
- Watts, E.; Zhao, Y.; Dhara, A.; Eller, B.; Patwardhan, A.; Sinai, A.P. Novel Approaches Reveal that Toxoplasma gondii Bradyzoites within Tissue Cysts Are Dynamic and Replicating Entities In Vivo. mBio 2015, 6, 1128. [Google Scholar] [CrossRef]
- Webster, J.P.; Kaushik, M.; Bristow, G.C.; McConkey, G.A. Toxoplasma gondii infection, from predation to schizophrenia: Can animal behaviour help us understand human behaviour? J. Exp. Biol. 2013, 216, 99–112. [Google Scholar] [CrossRef]
- Parlog, A.; Harsan, L.A.; Zagrebelsky, M.; Weller, M.; Elverfeldt, D.; Mawrin, C.; Korte, M.; Dunay, I.R. Chronic murine toxoplasmosis is defined by subtle changes in neuronal connectivity. Dis. Model. Mech. 2014, 7, 459–469. [Google Scholar] [CrossRef]
- Zhou, Y.; Shao, A.; Yao, Y.; Tu, S.; Deng, Y.; Zhang, J. Dual roles of astrocytes in plasticity and reconstruction after traumatic brain injury. Cell Commun. Signal. 2020, 18, 62. [Google Scholar] [CrossRef]





| Brain Section | NPC Number in Control Group (Non-Infected Animals)/mm3 | NPC Number in T. gondii-Infected Animals/mm3 | Statistical Significance (p < 0.05) |
|---|---|---|---|
| Sagittal SGZ | 2.12 ± 0.71 | 10.00 ± 2.19 * | 0.0014 |
| Coronal SGZ | 2.12 ± 0.29 | 11.14 ± 1.14 * | 0.0001 |
| Sagittal SVZ | 2.57 ± 0.97 | 17.50 ± 3.93 * | 0.0018 |
| Coronal SVZ | 4.02 ± 0.61 | 11.49 ± 0.99 * | 0.0001 |
| Sagittal RMS | 6.00 ± 1.06 | 14.82 ± 2.01 * | 0.0030 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anaya-Martínez, V.; Anacleto-Santos, J.; Mondragón-Flores, R.; Zepeda-Rodríguez, A.; Casarrubias-Tabarez, B.; de Jesús López-Pérez, T.; de Alba-Alvarado, M.C.; Martínez-Ortiz-de-Montellano, C.; Carrasco-Ramírez, E.; Rivera-Fernández, N. Changes in the Proliferation of the Neural Progenitor Cells of Adult Mice Chronically Infected with Toxoplasma gondii. Microorganisms 2023, 11, 2671. https://doi.org/10.3390/microorganisms11112671
Anaya-Martínez V, Anacleto-Santos J, Mondragón-Flores R, Zepeda-Rodríguez A, Casarrubias-Tabarez B, de Jesús López-Pérez T, de Alba-Alvarado MC, Martínez-Ortiz-de-Montellano C, Carrasco-Ramírez E, Rivera-Fernández N. Changes in the Proliferation of the Neural Progenitor Cells of Adult Mice Chronically Infected with Toxoplasma gondii. Microorganisms. 2023; 11(11):2671. https://doi.org/10.3390/microorganisms11112671
Chicago/Turabian StyleAnaya-Martínez, Verónica, Jhony Anacleto-Santos, Ricardo Mondragón-Flores, Armando Zepeda-Rodríguez, Brenda Casarrubias-Tabarez, Teresa de Jesús López-Pérez, Mariana Citlalli de Alba-Alvarado, Cintli Martínez-Ortiz-de-Montellano, Elba Carrasco-Ramírez, and Norma Rivera-Fernández. 2023. "Changes in the Proliferation of the Neural Progenitor Cells of Adult Mice Chronically Infected with Toxoplasma gondii" Microorganisms 11, no. 11: 2671. https://doi.org/10.3390/microorganisms11112671
APA StyleAnaya-Martínez, V., Anacleto-Santos, J., Mondragón-Flores, R., Zepeda-Rodríguez, A., Casarrubias-Tabarez, B., de Jesús López-Pérez, T., de Alba-Alvarado, M. C., Martínez-Ortiz-de-Montellano, C., Carrasco-Ramírez, E., & Rivera-Fernández, N. (2023). Changes in the Proliferation of the Neural Progenitor Cells of Adult Mice Chronically Infected with Toxoplasma gondii. Microorganisms, 11(11), 2671. https://doi.org/10.3390/microorganisms11112671








