Abstract
Background: Hippeastrum species have a wide range of biological properties. In Argentina, this genus comprises ten widely distributed species. Purpose: To evaluate the antiparasitic and anticholinesterase activities and chemical profiles of seven Argentinean Hippeastrum species and determine the synergism between the major isolated alkaloid—montanine—and benznidazole in anti-Trypanosoma cruzi activity. Methods: The antiparasitic activity was evaluated through antiproliferative and viability assays against T. cruzi epimastigotes. Synergism assays were performed using the Chou–Talalay method. AChE and BuChE inhibitory activities were also assessed. The alkaloid composition was obtained using GC-MS analysis. Results: All extracts showed strong growth inhibition of T. cruzi epimastigote proliferation. The extracts from H. aglaiae, H. aulicum, and H. hybrid stand out for their potent and total growth inhibition, which was comparable to benznidazole. The H. reticulatum extract showed strong Acetylcholinesterase (AChE) inhibitory activities, while five species showed moderate Butyrylcholinesterase (BuChE) inhibition. Fifteen alkaloids were identified by means of GC-MS. Regarding the synergism assessment, the highest synergistic effect was obtained from the combination of montanine and benznidazole. Conclusion: Hippeastrum species bulb extracts from Argentina were shown to be a good source of antiparasitic alkaloids and cholinesterase inhibitors. The synergism between montanine and benznidazole emerges as a potential combination for future studies to treat Chagas disease.
1. Introduction
Natural products have been a source of compounds useful in medicine, pharmaceuticals, and biology. Neglected Tropical Diseases (NTDs) are a diverse group of communicable diseases, and they are prevalent in tropical and subtropical conditions and affect more than 1 billion people [1]. WHO lists 17 NTDs, including Chagas disease (CD), which is caused by the protozoan parasite Trypanosoma cruzi. CD is mainly found in endemic areas of 21 countries in Latin America, and it was estimated that in the world, there are between 6 and 7 million infected people [1]. The drugs nifurtimox and benznidazole (Bzn) are effective against T. cruzi during the acute phase of the disease but are less useful in the chronic phase [2]. These drugs cause frequent adverse effects, such as allergic skin reactions, rashes, nerve damage (peripheral neuropathy), and diminished bone marrow function (neutropenia and thrombocytopenia). Limitations especially arise during prolonged treatment if the patient discontinues treatment [3,4]. Numerous synthetic and natural compounds were tested against T. cruzi for decades, but in most cases, their use is restricted by their high cytotoxicity and low efficacy in the chronic phase of CD [5,6]. Considering all of the disadvantages of current antichagasic chemotherapy, alternative drugs with safer, more tolerable, and effective profiles remain a critical need. Moreover, there is an increasing interest in drug combinations for Chagas disease, allowing a reduction in the dose of each constituent and a consequent reduction in adverse effects and treatment withdrawal [7]. In fact, the majority of cancer chemotherapies and several antimicrobial treatments are based on multidrug regimens [8]. On the other hand, 50 million people around the world live with dementia [9]. Alzheimer’s disease (AD) is the most common dementia type and may account for 60–70% of dementia cases [10,11].
Alkaloids are secondary metabolites of plants that are of great interest for drug development [12]. A particular characteristic of the Amaryllidaceae family plants is an exclusive, large, and still-expanding group of alkaloids that are characterized by a unique skeletal arrangement and a broad spectrum of biological activities [12,13,14]. In northeastern Argentina, indigenous communities use some species of the Amaryllidaceae family in traditional medicine, including Hippeastrum species [15]. The genus Hippeastrum, belonging to the Amaryllidoideae subfamily, is endemic to South America and is characterized by large bulbs and prominent and colorful flowers [16]. Hippeastrum species have a wide range of biological properties, including antimicrobial, cholinesterase inhibitory, cytotoxic, psychoactive, antiepileptic, and anti-inflammatory activities [16,17,18,19,20]. In Argentina, this genus comprises nine widely distributed and poorly studied species that vary in habitat, ranging from tropical to subtropical areas and from sea level to high altitudes [21,22].
In this work, an in vitro study of basic alkaloid-rich extracts (BAREs) from Hippeastrum spp. was performed to determine their antiparasitic activities, as well as to estimate their potential as cholinesterase inhibitors. Furthermore, the alkaloid profile of each species using GC-MS was obtained. Finally, the combination of the isolated montanine alkaloid, H. hybrid BARE, and Bzn was tested to determine their synergistic effect against T. cruzi.
2. Materials and Methods
2.1. Plant Material
Bulbs of the species Hippeastrum aglaiae, H. aulicum, H. glaucescens, H. hybrid, H. petiolatum, H. puniceum, and H. reticulatum were collected in Northeast Argentina during the flowering period between the years 2012 and 2015. The specimens were identified by MSc. German Roitman and then deposited in the herbarium of Universidad de Nacional de San Juan under specific herbarium codes as follows: IBT-Arg 20, IBT-Arg 21, IBT-Arg 22, IBT-Arg 23, IBT-Arg 24, IBT-Arg 25, and IBT-Arg 26.
2.2. Alkaloid Extraction and Isolation
Dry powdered bulb material (20–100 g) was macerated in H2SO4 2% for 4 h in an ultrasonic bath (3 × 1000 mL). Subsequently, samples were centrifuged at 4200× g (10 min), and the supernatant was transferred to another flask, where it was defatted with diethyl ether (Et2O) (3 × 500 mL). The aqueous solution was brought to pH 10–11 with 10% NaOH, and the alkaloids were extracted with CH2Cl2 (3 × 500 mL). The organic phase was dried with anhydrous sodium sulfate and then evaporated to obtain the basic alkaloid-rich extracts (BAREs) of each species. The average alkaloid yield of the extraction process was 0.55% from the dry plant material. Montanine was previously isolated from the bulbs of Hippeastrum argentinum [17]. The H. hybrid BARE was chromatographed on a Sephadex LH-20 column (length 50 cm, 3.5 cm i.d.; equilibrated to MeOH) and then eluted with MeOH. Twenty-one fractions were obtained and grouped according to their TLC profiles as follows: fractions 1 to 10 (F1), fractions 11 and 12 (F2), fractions 13 and 14 (F3), fractions 15 and 16 (F4), fractions 17 to 19 (F5), and fractions 20 and 21 (F6). As a result of the isolation process, six fractions were obtained (F1–F6).
2.3. GC-MS and UHPLC-MS/MS Analysis
The alkaloids were identified by comparing their GC-MS spectra and Kovats retention index (RI) values against authentic Amaryllidaceae alkaloids previously isolated and identified. Spectral data were processed with AMDIS 2.64 software. Alkaloids were identified by comparing their fragmentation patterns and RIs with those of the Amaryllidaceae alkaloids in our laboratory library, in which the isolated compounds were identified by NMR and other spectroscopic techniques (UV, CD, and MS), as well as the NIST database and literature data. The RI values were calibrated with an n-hydrocarbon calibration mixture (C9–C36). The results obtained were analyzed using AMDIS 2.64 software and the NIST database. Chromatograms indicating the identified alkaloids are shown in Supplementary Materials (Figure S1).
The UHPLC-MS/MS quantification analyses were performed on a Waters Acquity H-class with a Xevo TQ-S micro mass spectrometer detector, and the MassLynx software was used for data acquisition and treatment. An Acquity UPLC BEH C18, 130 Å, 1.7 µm, 2.1 mm × 100 mm column was used. The elution system was composed of formic acid 0.1% (mobile phase A), acetonitrile–formic acid 0.1% (mobile phase B), and MeOH (mobile phase C). The flow rate was 0.2 mL·min−1, while the temperature of the sample compartment and column were 22 °C and 40 °C, respectively. The gradient started with 95% A and 5% B (hold for 2 min); 85% A and 15% B (3 min); 80% A, 10% B, and 10% C (5 min) and hold for 7 min; 95% A and 5% B (1 min) and hold for 2 min, totaling 20 min of analysis for montanine quantification. The spectrometric parameters were ESI + and the daughter ion function for the [M + H]+ of montanine (m/z 302), with a voltage cone of 46.15 V, capillary energy of 2.00 kV, source temperature of 124 °C, and collision energy of 20 V. Standard solutions of montanine for the calibration curve (0.1, 1, 5, and 10 ppm), as well as the BARE solutions (84, 96, and 98 ppm for H. hybrid, H. aglaiae, and H. puniceum, respectively), were prepared by dissolving them in a mixture of methanol and water (50:50). The solutions were filtered through a 0.22 μm membrane filter, and the injection volume was 10 μL.
2.4. AChE and BuChE Inhibitory Activities
Cholinesterase inhibitory activities were determined according to Ellman et al. [23] with some modifications [16]. Acetylcholinesterase (AChE) from Electrophorus electricus (C3389), Butyrylcholinesterase (BuChE) from equine serum (C7512), acetylthiocoline iodide (ATCI, A5751), butyrylthiocholine iodide (BTCI, 20820), 5,5′-dithio-bis (2-nitrobenzoic acid) (DTNB) (D-8130), and galantamine hydrobromide (GAL) were purchased from Sigma-Aldrich (St. Louis, MO, USA. A volume of 50 μL of 0.25 U/mL AChE or BuChE in phosphate buffer (8 mM K2HPO4, 2.3 mM NaH2PO4, and 0.15 M NaCl, pH 7.6) and 50 μL of each BARE concentration dissolved in the same buffer were added to the wells. The plates were incubated for 30 min at room temperature before 100 μL of the substrate solution (0.1 M Na2HPO4, 0.5 M DTNB, and 0.6 mM ATCI or BTCI in Millipore water, pH 7.5) was added. The absorbance was read in a Thermo Scientific Multiskan FC microplate spectrophotometer at 405 nm after 5 min. The enzyme-inhibitory activity was calculated as a percentage compared to an assay using a buffer without any inhibitor. The enzyme-inhibitory data were analyzed with the software package Prism (Graph Pad Inc., San Diego, CA, USA). The BARE concentrations used to calculate the IC50 values were 1, 20, 40, 60, 80, and 100 μg/mL in both AChE and BuChE assays. The IC50 values are the means ± SD of three individual determinations, each performed in triplicate.
2.5. Trypanosoma cruzi
2.5.1. Culture
T. cruzi epimastigotes (Dm28c strain—DTU: TcI) were cultured at 28 °C in Diamond medium (0.1 M NaCl, 0.05 M K2HPO4, 0.625% (w/v) tryptose, 0.625% (w/v) tryptone, and 0.625% (w/v) yeast extract, pH 7.2), supplemented with 10% inactivated FBS (Gibco) and 12.5 µg/mL hemin and antibiotics 0.1% (penicillin 75 U/mL and streptomycin 75 µg/mL).
2.5.2. Growth Inhibition Assay
Parasites were incubated at 28 °C in sterile plastic tubes with 10 and 50 µg/mL of each BARE; 1, 2.5, 5, and 10 µg/mL of H. aglaiae and H. hybrid BAREs; 5 and 10 µg/mL of each fraction of H. hybrid BARE; and 0.1, 0.5, 1, 2.5, and 5 µg/mL of montanine. The initial concentration (InC) of parasites was 3 × 106/mL (or 2 × 106/mL) in a final volume adjusted to 1 mL. The negative controls were parasites without treatment, and the positive control was treated with Bzn (5 µg/mL). According to Spina et al. [24], aliquots were collected at 24, 48, and 72 h, and they were suspended in 2% p-formaldehyde in PBS (0.15 M NaCl, 0.02 M NaH2PO4, and 0.017 M NaOH; pH 7.2). Then, the number of parasites (n° p) was counted in a Neubauer chamber. The percentage of inhibition was calculated as:
% inhibition = 100 − {[(n° p treated − InC)/(n° p control − InC)] × 100}
2.5.3. Viability Assay
T. cruzi epimastigotes were incubated with 50 µg/mL of each BARE. Aliquots were taken at 24 and 48 h from each treated culture and were placed on slides for 3 min with 2% eosin in PBS (pH 7.2) and observed under a light microscope. The percentage of dead cells (stained) was determined for each treatment, which was carried out in triplicate. The percentage viability was calculated as:
% viability = [n° p alive/(n° p dead + n° p alive)] × 100
2.5.4. Combination Assay (Synergism)
Drug Treatment
The IC50 value was first determined for each drug alone against T. cruzi epimastigotes (Dm28c strain). H. hybrid BARE concentrations ranged from 1 to 10 μg/mL for the single-drug treatment. Montanine concentrations ranged from 0.1 to 5 μg/mL. Bzn concentrations ranged from 1 to 5 μg/mL for the single-drug treatment. Combination studies were performed by combining montanine–H. hybrid BARE, montanine–Bzn, and H. hybrid BARE–Bzn. T. cruzi epimastigotes were treated with the drugs alone and combined in a fixed ratio [25] at concentrations of 0.25 × IC50, 0.5 × IC50, IC50, 2 × IC50, and 4 × IC50 (Figure S2).
Analysis of Drug Interactions
To quantify drug interaction, the Combination Index (CI) and Dose Reduction Index (DRI) were assessed with the Chou and Talalay method [26] using the CompuSyn software (ComboSyn, Inc., New York, NY, USA). The mutually exclusive model was used, which is based on the assumption that drugs act through entirely different mechanisms [27]. CI was plotted on the y-axis as a function of the fraction affected (Fa) on the x-axis to assess drug synergism between drug combinations. Fa is a value between 0 and 1, where 0 means the drug had no effect on cell viability, and 1 means the drug produced a full effect on decreasing cell viability. The CI is a quantitative representation of pharmacological interactions. CI < 1 indicates synergism, CI = 1 indicates an additive interaction, and CI > 1 indicates antagonism. The DRI is a dimensionless measure of how much the dose of each drug in a synergistic combination may be reduced at a given fractional inhibition compared with the doses of each drug alone, where DRI > 1 indicates a favorable dose reduction, DRI < 1 indicates an unfavorable dose reduction, and finally, DRI = 1 indicates no dose reduction [26]. Experiments were conducted in triplicate.
2.6. Statistical Analysis
Student’s t-test was used to determine the statistical significance of the differences between treated and control groups. The effect of each treatment was analyzed by one-way analysis of variance (ANOVA).
3. Results
3.1. GC-MS and UHPLC-MS/MS Analyses
Based on the total ion current (TIC) in the GC-MS analysis, the most abundant alkaloids identified in the Hippeastrum BAREs were montanine (H. hybrid); lycorine (H. aglaiae, H. aulicum, and H. petiolatum); hippeastrine (H. puniceum); 8-O-demethylhomolycorine (H. reticulatum); and tazettine (H. glaucescens). Further information regarding the alkaloid profile obtained by the GC-MS analysis of each BARE is shown in Table 1. The alkaloid chemical structures are shown in Figure S3.

Table 1.
Main alkaloids identified in Argentinean Hippeastrum spp. samples by GC-MS analysis.
The UHPLC-MS/MS quantification analysis showed a concentration of 14.32% montanine for the H. hybrid BARE, while the concentrations for H. puniceum and H. aglaiae BAREs were 1.28% and 4.98%, respectively. Finally, H. petiolatum showed the presence of montanine at a lower concentration (<1%). The linear correlation curve was r2 = 0.9202, and the linear equation was y = 113.638 x + 37,289.
3.2. Cholinesterase Inhibitory Activities
The BAREs were tested for in vitro AChE and BuChE inhibitory activities. The results, expressed as IC50 values, are summarized in Table 2. Galanthamine was used as the positive control. All BAREs of Hippeastrum species showed strong activity against AChE. The most active BARE against AChE was H. reticulatum, followed by H. petiolatum, H. puniceum, and H. aulicum (IC50 = 3.13 ± 0.53, 5.07 ± 0.75, 5 ± 0.64, and 6.33 ± 0.81 μg/mL, respectively). Likewise, all BAREs showed moderate to low inhibitory activity against BuChE (IC50 ≥ 50 μg/mL).

Table 2.
Cholinesterase inhibitory activities of Argentinean Hippeastrum spp. BAREs.
3.3. Anti-T. cruzi Activity
3.3.1. Growth Inhibition
Activity of Argentinean Hippeastrum spp. BAREs
The anti-T. cruzi activity of the BAREs in the epimastigote stage was evaluated at concentrations of 10 and 50 µg/mL. In Table 3, the results are shown. All Hippeastrum BAREs tested at 50 µg/mL presented 100% inhibition of proliferation at 24, 48, and 72 h, while at 10 µg/mL, the most active BAREs were H. aglaiae and H. hybrid, which inhibited 100% of the proliferation of epimastigotes at 72 h. Likewise, H. aulicum, H. glaucescens, H. petiolatum, H. puniceum, and H. reticulatum showed the potent inhibition of epimastigote proliferation at 48 h. Bzn presented strong proliferation inhibition, with a slight loss of efficacy throughout the assay.

Table 3.
In vitro activity of Hippeastrum spp. BAREs from Argentina on proliferation of T. cruzi epimastigotes (mean ± SD).
Antiproliferative T. cruzi Activity of H. aglaiae and H. hybrid BAREs
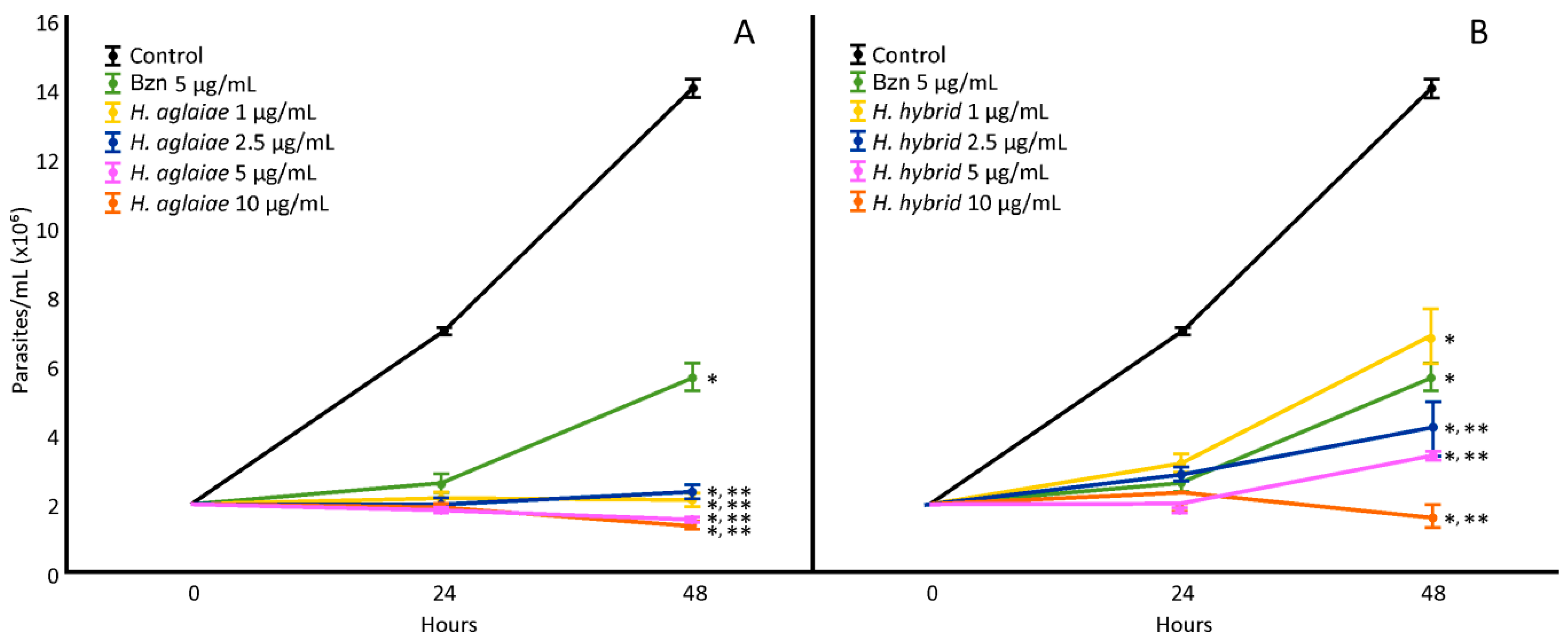
H. aglaiae and H. hybrid BAREs were evaluated (1, 2.5, 5, and 10 µg/mL) in order to determine the IC50 values (Figure 1). The H. aglaiae BARE showed 100% inhibition of the proliferation of T. cruzi at 48 h at all concentrations tested. Since all concentrations showed the potent inhibition of epimastigote proliferation, no dose-dependent relationship was observed. Regarding the H. hybrid BARE, all of the concentrations assayed (2.5, 5, and 10 µg/mL) reduced the number of parasites compared to the control. These results indicate an effect on proliferation greater than that of Bzn (positive control). Thus, the IC50 values for the H. aglaiae and H. hybrid BAREs were 0.0026 and 0.96 µg/mL, respectively, which are lower than that of Bzn (4.58 µg/mL).
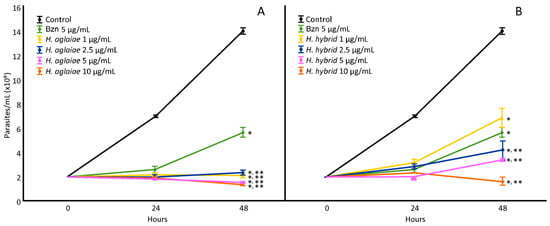
Figure 1.
Effect of the H. aglaiae BARE (A) and H. hybrid BARE (B) on T. cruzi epimastigote proliferation at concentrations of 1, 2.5, 5, and 10 µg/mL. *: Significant difference from negative control; **: significant difference from positive control. p < 0.05.
Anti-T. cruzi Activity of H. hybrid BARE Fractions
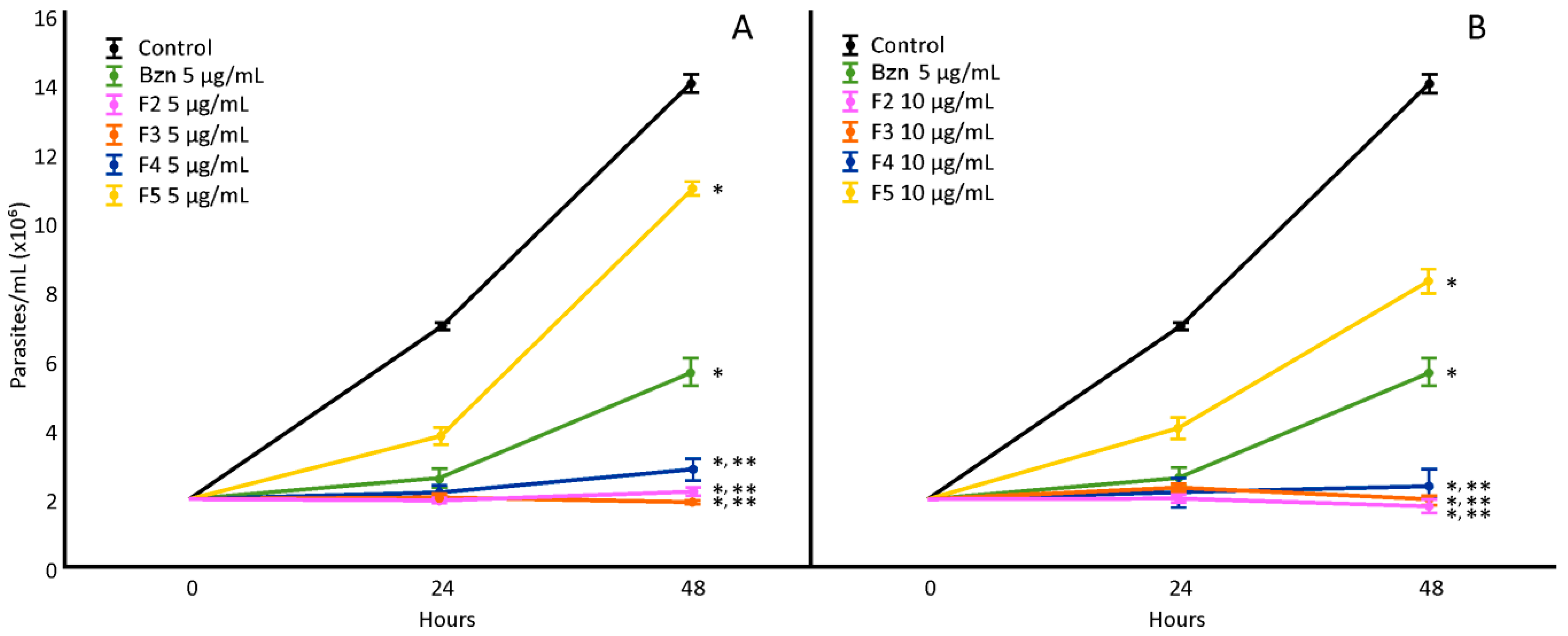
The H. hybrid BARE fractions (F1–F6) were tested at 5 and 10 µg/mL (Figure 2), showing a notable decrease in proliferation compared to the untreated control for fractions F2, F3, F4, and F5 (F1 and F6 did not show differences; data not shown). F3 was the most active at 5 µg/mL, and F2 was the most active at 10 µg/mL. Through UHPLC-MS/MS analysis, fraction F2 revealed mainly the presence of montanine (13.82%), while F3 showed almost double the amount of this alkaloid (24.76%) and also hippeastrine. F4 showed a mixture of 7-hydroxyclivonine, hippeastrine, and montanine (10.20%). Finally, in F5, the fragmentation patterns indicated the presence of 7-hydroxyclivonine and a low concentration of montanine (<1%).
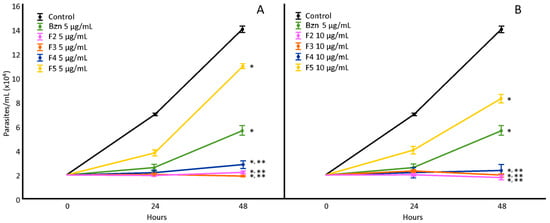
Figure 2.
Effect of H. hybrid BARE fractions on T. cruzi epimastigotes at concentrations of 5 µg/mL (A) and 10 µg/mL (B). *: Significant difference from negative control; **: significant difference from positive control. p < 0.05.
Antiproliferative Activity of Montanine
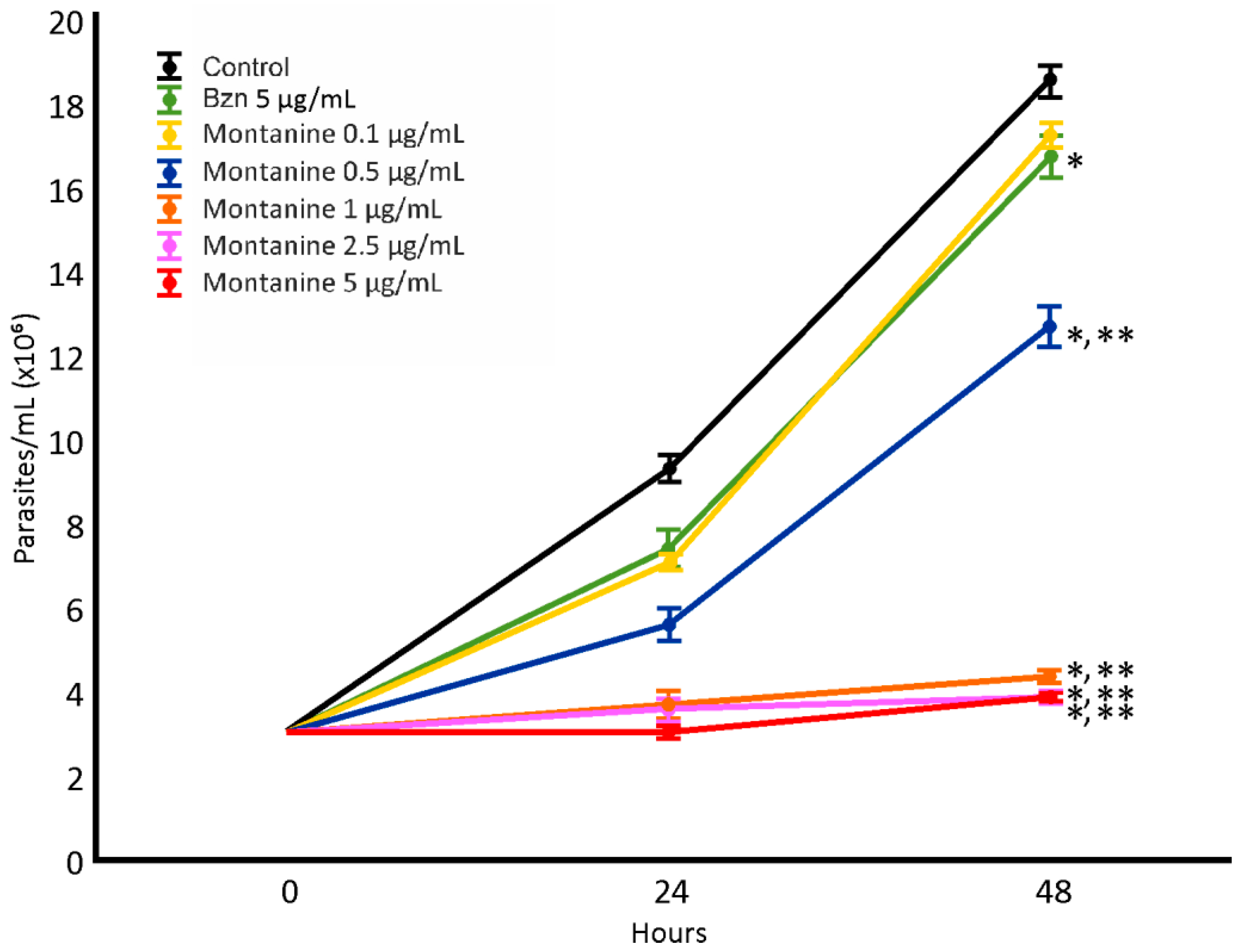
The epimastigote antiproliferative assay was performed with montanine at concentrations of 0.1, 0.5, 1, 2.5, and 5 µg/mL (Figure 3). Potent dose-dependent activity was observed, and all concentrations tested (except 0.1 µg/mL) were more active than Bzn. The IC50 value for montanine was 0.55 µg/mL.
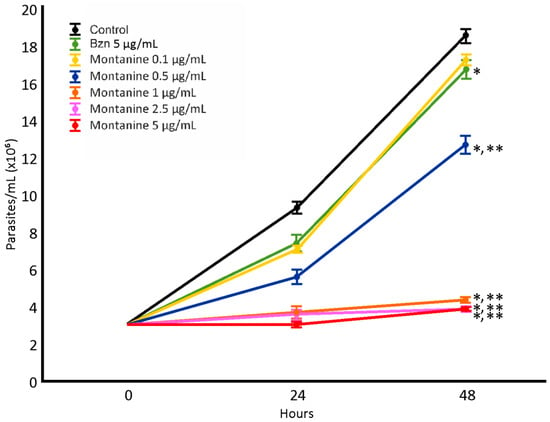
Figure 3.
Effect of montanine in T. cruzi epimastigotes. *: Significant difference from negative control; **: significant difference from positive control. p < 0.05.
3.3.2. Viability Assay
The viability percentages of epimastigote treated with Hippeastrum BAREs at 50 µg/mL and Bzn at 5 µg/mL for 24 and 48 h are shown in Table 4. All Hippeastrum BAREs produced a decrease in the viability of T. cruzi epimastigotes, with the H. reticulatum and H. hybrid BAREs being those with the greatest effect in the first 24 h (87.84 and 86.59%, respectively). Likewise, the H. glaucescens BARE decreased the viability of T. cruzi epimastigotes to a value of 72.88% at 48 h. Regarding Bzn, the viability of epimastigotes was not affected, presenting approximately 98% living cells.

Table 4.
In vitro activity of Hippeastrum spp. BAREs from Argentina on viability of T. cruzi epimastigotes (mean ± SD).
3.3.3. Synergistic Combinations
The growth inhibition assay was performed for each drug alone against T. cruzi epimastigotes, and the CompuSyn software was used for the generation of single-drug dose–effect curves (Figure S4A). The combinations of montanine + H. hybrid BARE, montanine + Bzn, and H. hybrid BARE + Bzn were evaluated against T. cruzi (Figures S4B and S5).
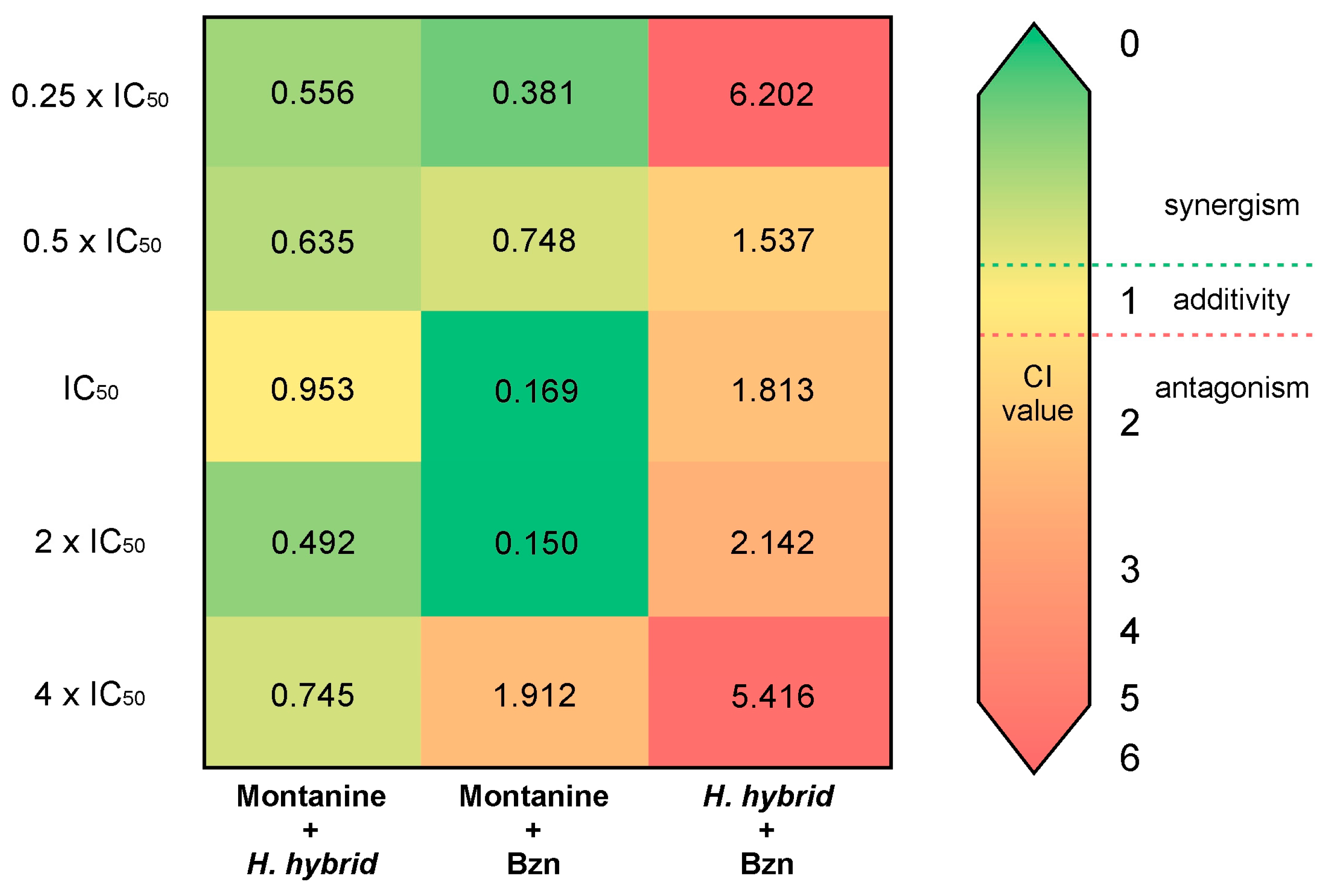
The results of the combinations are summarized in Table S1. The combination of montanine + Bzn shows the best synergistic effect, particularly in the mixtures of IC50 and 2 × IC50 (CI = 0.169 and 0.150, respectively). This interaction also demonstrated a potent effect on the proliferation of T. cruzi epimastigotes, with Fa values = 0.972 and 0.99, respectively. A combination of lower concentrations of montanine + Bzn also resulted in a synergistic effect (CI < 1) with a Fa of nearly 0.7. However, the combination of a higher concentration of montanine + Bzn (4 × IC50) resulted in an antagonistic effect (CI = 1.912). Likewise, all combinations of montanine + H. hybrid BARE showed synergistic interactions, with the combination of 2 × IC50 (montanine = 1.1 µg/mL + H. hybrid BARE = 1.24 µg/mL) being the one with the highest synergistic effect (CI = 0.492) and that of 4 × IC50 being the one with the highest inhibitory effect on the proliferation of T. cruzi (Fa = 0.972). However, H. hybrid BARE + Bzn showed antagonistic effects in all combinations (CI = 1.537–6.202) and low to moderate epimastigote antiproliferative effects (Fa = 0.05–0.708). The CI values of all combinations are shown in Figure 4. Based on the DRI values of the actual experimental data points, all synergistic drug combinations achieved favorable dose reduction indices (DRI > 1) for both drugs involved (Table S1). Montanine + Bzn reached the highest value of the DRI at a concentration of 2 × IC50, reducing the dose of montanine by 9.9 times and the dose of Bzn by 20.19 times to obtain 99% inhibition of the proliferation of T. cruzi. These interesting results were obtained by combining montanine + Bzn at their IC50 concentration values, achieving an inhibition value of 97% for both compounds and dose reductions of 9.4 and 15.8 times for montanine and Bzn, respectively. Finally, for all montanine + H. hybrid BARE combinations, favorable DRI values were obtained (1.63 to 5.57).
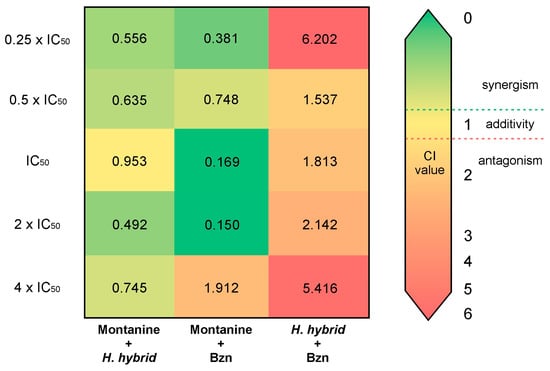
Figure 4.
Synergy heat map compiling CI values. The squares in the figure indicate antagonism (CI > 1.2, orange-red), additivity (0.9 < CI < 1.1, yellow), and synergism (CI < 0.9, green) among montanine, H. hybrid BARE, and Bzn against T. cruzi epimastigotes.
4. Discussion
The chemical profiles of the Hippeastrum species BAREs showed a total of fifteen alkaloids. This is the first report of the presence of 11,12-dehydroanhydrolycorine in H. aulicum, 8-O-demethylhomolycorine, hippeastrine, and montanine in H. puniceum, and 8-O-demethylhomolycorine in H. reticulatum, expanding the list of alkaloids previously reported for these species [28,29,30]. This work presents the first alkaloid profiles of H. aglaiae, which shows the presence of the alkaloids 8-O-demethylhomolycorine, lycorine, montanine, and norlycoramine, and of H. petiolatum, which shows the presence of montanine and lycorine. In the H. hybrid BARE, the alkaloids 2-hydroxyhomolycorine, 7-hydroxyclivonine, galanthamine, hippeastrine, lycorine, montanine, and pancracine were identified.
The alkaloids pancracine, montanine, lycorine, hamayne, 8-O-demethylhomolycorine, hippeastrine, and norlycoramine identified in the Hippeastrum BAREs have previously been tested against AChE and BuChE, reporting low or null inhibitory activities [11,16,31,32,33]. Herein, the H. reticulatum BARE exhibited the highest inhibitory activity against both AChE (IC50 = 3 ± 0.53) and BuChE (IC50 = 50 ± 1.20). The alkaloids lycorine, 11,12-dehydroanhydrolycorine, and 8-O-demetylhomolycorine and three unidentified narcissidine-type alkaloids were identified in this BARE, suggesting that the inhibition could be due to the combined effect of these compounds or other alkaloids present in low abundance.
Regarding anti-T. cruzi activity, the alkaloids identified in the BAREs, lycorine, hippeastrine, and montanine, have been reported for their inhibitory effect against the amastigote form [34], whereas pancracine has shown activity against trypomastigotes of T. cruzi [35]. Recently, de Souza and Barrias [36] proposed a new scheme of the life cycle of T. cruzi, describing epimastigote-type forms with infective and proliferative capacity, highlighting the importance of taking into account these stages (epimastigote-type forms), and establishing that it is a new target for treatment during the course of Chagas disease. This new approach supports the previous work developed by Kessler et al. [37], who reported differentiated epimastigotes with the ability to infect mammalian cells. Based on these advances in CD research, the Hippeastrum BAREs were assayed on T. cruzi epimastigotes.
In the literature, the IC50 values of Amaryllidaceae alkaloid extracts have been reported for their anti-T. cruzi activity when tested at ≥100 µg/mL [38,39]. The H. aglaiae and H. hybrid BAREs showed the highest effect against the proliferation of T. cruzi epimastigotes, with IC50 values lower than that of Bzn (IC50 = 0.0026 µg/mL, IC50 = 0.96 µg/mL, and IC50 = 4.58 µg/mL, respectively). In addition, the H. aglaiae and H. hybrid BAREs’ antiproliferative activity persisted even at 72 h. The H. aglaiae and H. hybrid BARE activities could be explained by the presence of lycorine and montanine, respectively. In order to prove the compound responsible for the H. hybrid BARE activity and its fractions, the assay of pure montanine was evaluated against T. cruzi, showing a dose-dependent response.
Combined therapies are used to treat various infectious diseases, including toxoplasmosis, malaria, tuberculosis, and AIDS [1]. Likewise, therapies for the treatment of CD aim to reduce the side effects produced by Bzn [7,40]. Some alkaloids, such as carbol, have been tested in combination with Bzn, reducing parasitemia in murine models [41]. Herein, the synergistic effect of the combination of montanine with Bzn stands out, managing to reduce the dose of Bzn by around 20 times. In addition, montanine is reported to have a low rate of cytotoxicity in HepG2 cells, with a value of TC50 = 13.9 µg/mL (46.10 µM) [34]. This is the first study showing that montanine and Bzn, as a combined strategy, have a powerful inhibitory effect against T. cruzi forms. These results position the montanine alkaloid as an enhancer of the effect of Bzn, reducing the doses used to achieve inhibition close to 100%. Also interesting is the synergistic effect produced by adding montanine to the H. hybrid BARE. However, the combination of the H. hybrid BARE and Bzn showed an antagonistic effect. These results indicate that the combination of several compounds reported as active does not always imply greater activity. In this case, the alkaloids present in the BARE reduced the effect of Bzn.
Possibly, the pool of alkaloids present in H. hybrid with the extra addition of montanine enhances the antiparasitic activity, leaving a window for future trials on the effect of the interaction between alkaloids of the Amaryllidaceae family against T. cruzi.
5. Conclusions
The combination of the montanine alkaloid and Bzn showed a potent synergistic effect, reducing the concentration of Bzn twenty-fold against T. cruzi, and could be considered for future research to reduce the side effects of Bzn to treat CD. To a lesser extent, montanine and the H. hybrid BARE presented a synergistic effect. Thus, further mechanistic studies, such as the determination of the structure–activity relationship, must be performed to identify the potential compounds responsible for these properties. Finally, Argentinean Hippeastrum species represent a promising candidate for the treatment of CD.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/microorganisms11010144/s1. Figure S1: GC-MS chromatograms of Hippeastrum BAREs a: H. aglaiae; b: H. aulicum; c: H. glaucescens; d: H. hybrid; e: H. petiolatum; f: H. puniceum; g: H. reticulatum. Figure S2: Representation of the design of the combination experiences; Figure S3: Alkaloids identified in Hippeastrum BAREs; Figure S4: The dose-effect curves of single drugs (A) and drug combos (B); Figure S5. Chou-Talalay method Fa-CI plot of montanine, H. hybrid BARE, and Bzn. Table S1: CI, DRI and Fa values of combinations of montanine, H. hybrid BARE and Bzn.
Author Contributions
M.P., J.E.O., M.A.S. and G.E.F. conceptualized the study. M.P., R.M.S.Z., P.A.B. and J.E.O. performed all biological assays. J.E.O., J.B. and G.E.F. performed chemical studies and provided the extracts and purified alkaloids. G.R. identified the species and provided the bulb samples. M.P., J.E.O., G.E.F. and J.B. wrote the article. All authors agree to be accountable for all aspects of this work ensuring integrity and accuracy. All authors have read and agreed to the published version of the manuscript.
Funding
This work was partially supported by ANPCyT (PICT-2020-SERIEA-03883), CONICET PIP 112202101 00902CO, and CICITCA-UNSJ (Argentina), and BIFRENES RED 416RT0511 CYTED España.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
M.P., J.E.O. and R.M.S.Z. hold a fellowship from CONICET. G.E.F. is a researcher from CONICET. The authors thank Juan Veja-Moreno for the support and donation. M.P. thanks Paula Wetten for her assistance and hospitality.
Conflicts of Interest
The authors declare no conflict of interest.
References
- World Health Organization. Neglected Tropical Diseases. Available online: http://www.who.int/neglected_diseases/diseases/en/ (accessed on 25 July 2022).
- Vermelho, A.B.; Cardoso, V.; Mansoldo, F.R.P.; Supuran, C.T.; Cedrola, S.M.L.; Rodrigues, I.A.; Rodrigues, G.C. Chagas Disease: Drug Development and Parasite Targets. In Antiprotozoal Drug Development and Delivery; Vermelho, A.B., Supuran, C.T., Eds.; Springer: Cham, Germany, 2022; Volume 39. [Google Scholar] [CrossRef]
- Molina, I.; Gómez, I.; Prat, J.; Salvador, F.; Treviño, B.; Sulleiro, E.; Serre, N.; Pou, D.; Roure, S.; Cabezos, J.; et al. Randomized trial of posaconazole and benznidazole for chronic Chagas’ disease. N. Engl. J. Med. 2014, 370, 1899–1908. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.L.; Lima, F.D.C.; Cruz, J.V.; dos Reis Almeida, T.; da Silva, C.B.B. In silic study of alkaloids derived from Catharanthus roseus in the active site of Trypanosoma cruzi by molecular docking. Res. Soc. Dev. 2022, 11, e23711528114. [Google Scholar] [CrossRef]
- Bermudez, J.; Davies, C.; Simonazzi, A.; Real, J.P.; Palma, S. Current drug therapy and pharmaceutical challenges for Chagas disease. Acta Trop. 2016, 156, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Field, M.C.; Horn, D.; Fairlamb, A.H.; Ferguson, M.A.J.; Gray, D.W.; Read, K.D.; de Rycker, M.; Torrie, L.S.; Wyatt, P.G.; Wyllie, S.; et al. Anti-trypanosomatid drug discovery: An ongoing challenge and a continuing need. Nat. Rev. Microbiol. 2017, 15, 217–231. [Google Scholar] [CrossRef]
- Ribeiro, V.; Dias, N.; Paiva, T.; Hagström-Bex, L.; Nitz, N.; Pratesi, R.; Hecht, M. Current trends in the pharmacological management of Chagas disease. Int. J. Parasitol. Drugs Drug Resist. 2020, 12, 7–17. [Google Scholar] [CrossRef]
- Sbaraglini, M.L.; Bellera, C.L.; Braghini, J.Q.; Areco, Y.; Miranda, C.; Carrillo, C.; Kelly, J.; Buchholz, B.; Gelpi, R.; Talevi, A.; et al. Combined therapy with Benznidazole and repurposed drugs Clofazimine and Benidipine for chronic Chagas disease. Eur. J. Med. Chem. 2019, 184, 111778. [Google Scholar] [CrossRef]
- Browne, B.; Kupeli, N.; Moore, K.J.; Sampson, E.L.; Davies, N. Defining end of life in dementia: A systematic review. Palliat. Med. 2021, 35, 1733–1746. [Google Scholar] [CrossRef]
- Huang, L.K.; Chao, S.P.; Hu, C.J. Clinical trials of new drugs for Alzheimer disease. J. Biomed. Sci. 2020, 27, 1–13. [Google Scholar] [CrossRef]
- Al Shammari, L.; Hulcová, D.; Maříková, J.; Kučera, T.; Šafratová, M.; Nováková, L.; Schmidt, M.; Pulkrabkova, L.; Janousek, J.; Soukup, O.; et al. Amaryllidaceae alkaloids from Hippeastrum x hybridum CV. Ferrari, and preparation of vittatine derivatives as potential ligands for Alzheimer’s disease. S. Afr. J. Bot. 2020, 136, 137–146. [Google Scholar] [CrossRef]
- Rodrigues, T.; Reker, D.; Schneider, P.; Schneider, G. Counting on natural products for drug design. Nat. Chem. 2016, 8, 531–541. [Google Scholar] [CrossRef]
- Stratton, C.F.; Newman, D.J.; Tan, D.S. Cheminformatics comparison of approved drugs from natural product versus synthetic origins. Bioorganic Med. Chem. Lett. 2015, 25, 4802–4807. [Google Scholar] [CrossRef] [PubMed]
- Bastida, J.; Lavilla, R.; Viladomat, F. Chemical and biological aspects of Narcissus Alkaloids. In The Alkaloids: Chemistry and Physiology; Cordell, G.A., Ed.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 63, pp. 87–179. [Google Scholar]
- Martínez, G.J.; Barboza, G.E. Natural pharmacopoeia used in traditional Toba medicine for the treatment of parasitosis and skin disorders (Central Chaco, Argentina). J. Ethnopharmacol. 2010, 132, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, J.E.; Pigni, N.B.; Andujar, S.A.; Roitman, G.; Suvire, F.D.; Enriz, R.D.; Tapia, A.; Bastida, J.; Feresin, G.E. Alkaloids from Hippeastrum argentinum and their cholinesterase-inhibitory activities: An in vitro and in silico study. J. Nat. Prod. 2016, 79, 1241–1248. [Google Scholar] [CrossRef] [PubMed]
- da Silva, A.F.S.; de Andrade, J.P.; Bevilaqua, L.R.; de Souza, M.M.; Izquierdo, I.; Henriques, A.T.; Zuanazzi, J.Â.S. Anxiolytic-, antidepressant- and anticonvulsant-like effects of the alkaloid montanine isolated from Hippeastrum vittatum. Pharmacol. Biochem. Behav. 2006, 85, 148–154. [Google Scholar] [CrossRef]
- da Silva, A.F.S.; de Andrade, J.P.; Machado, K.R.B.; Rocha, A.B.; Apel, M.A.; Sobral, M.E.G.; Henriques, A.T.; Zuanazzi, J.A.S. Screening for cytotoxic activity of extracts and isolated alkaloids from bulbs of Hippeastrum vittatum. Phytomedicine 2008, 15, 882–885. [Google Scholar] [CrossRef]
- Gasca, C.A.; Moreira, N.C.; de Almeida, F.C.; Gomes, J.V.D.; Castillo, W.O.; Fagg, C.W.; Magalhãesa, P.O.; Fonseca-Bazzoa, Y.; Sakamoto-Hojo, E.; de Medeiros, Y.K.; et al. Acetylcholinesterase inhibitory activity, anti-inflammatory, and neuroprotective potential of Hippeastrum psittacinum (Ker Gawl.) herb (Amaryllidaceae). Food Chem. Toxicol. 2020, 145, 111703. [Google Scholar] [CrossRef]
- Santana, O.; Reina, M.; Anaya, A.L.; Hernández, F.; Izquierdo, M.E.; González-Coloma, A. 3-O-Acetyl-narcissidine, a bioactive alkaloid from Hippeastrum puniceum Lam. (Amaryllidaceae). Z. Naturforsch. C Biosci. 2008, 63, 639–643. [Google Scholar] [CrossRef]
- Sultana, J.; Sutlana, N.; Siddique, M.N.A.; Islam, A.K.M.A.; Hossain, M.M.; Hossain, T. In vitro bulb production in Hippeastrum (Hippeastrum hybridum). J. Cent. Eur. Agric. 2010, 11, 469–474. [Google Scholar]
- Zuloaga, F.O.; Morrone, O.; Belgrano, M.J. Catálogo de las plantas vasculares del Cono Sur (Argentina, sur de Brasil, Chile, Paraguay, y Uruguay) Ptherydophyta, Gymnospernae, Monocotyledoneae. Syst. Bot. 2009, 34, 610–611. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Spina, R.M.; Lozano, E.; Barrera, P.A.; Agüero, M.B.; Tapia, A.; Feresin, G.E.; Sosa, M.Á. Antiproliferative effect and ultrastructural alterations induced by 5-O-methylembelin on Trypanosoma cruzi. Phytomedicine 2018, 46, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Duarte, D.; Vale, N. New trends for antimalarial drugs: Synergism between antineoplastics and antimalarials on breast cancer cells. Biomolecules 2020, 10, 1623. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C. Drug combination studies and their synergy quantification using the chou-talalay method. Cancer Res. 2010, 70, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Roell, K.R.; Reif, D.M.; Motsinger-Reif, A.A. An introduction to terminology and methodology of chemical synergy—Perspectives from across disciplines. Front. Pharmacol. 2017, 8, 158. [Google Scholar] [CrossRef]
- Bessa, C.D.; Andrade, J.P.D.; Oliveira, R.S.D.; Domingos, E.; Santos, H.; Romão, W.; Bastida, J.; Borges, W.S. Identification of alkaloids from Hippeastrum aulicum (Ker Gawl.) Herb. (Amaryllidaceae) using CGC-MS and ambient ionization mass spectrometry (PS-MS and LS-MS). J. Braz. Chem. Soc. 2017, 28, 819–830. [Google Scholar] [CrossRef]
- Soprani, L.C.; Andrade, J.P.D.; Santos, V.D.D.; Alves-Araújo, A.; Bastida, J.; Silva, C.A.G.; Silveira, D.; de Souza, W.; Jamal, C.M. Chemical evaluation and anticholinesterase activity of Hippeastrum puniceum (Lam.) Kuntz bulbs (Amaryllidaceae). Braz. J. Pharm. Sci. 2021, 57, e19154. [Google Scholar] [CrossRef]
- Tallini, L.R.; Osorio, E.H.; Santos, V.D.D.; Borges, W.D.S.; Kaiser, M.; Viladomat, F.; Zuanazzi, J.A.S.; Bastida, J. Hippeastrum reticulatum (Amaryllidaceae): Alkaloid profiling, biological activities and molecular docking. Molecules 2017, 22, 2191. [Google Scholar] [CrossRef]
- Karakoyun, Ç.; Bozkurt, B.; Çoban, G.; Masi, M.; Cimmino, A.; Evidente, A.; Somer, N.U. A comprehensive study on Narcissus tazetta subsp. tazetta L.: Chemo-profiling, isolation, anticholinesterase activity and molecular docking of Amaryllidaceae alkaloids. S. Afr. J. Bot. 2020, 130, 148–154. [Google Scholar] [CrossRef]
- López, S.; Bastida, J.; Viladomat, F.; Codina, C. Acetylcholinesterase inhibitory activity of some Amaryllidaceae alkaloids and Narcissus extracts. Life Sci. 2002, 71, 2521–2529. [Google Scholar] [CrossRef]
- Šafratová, M.; Hošťálková, A.; Hulcová, D.; Breiterová, K.; Hrabcová, V.; Machado, M.; Fontinha, D.; Prudêncio, M.; Kuneš, J.; Chlebek, J.; et al. Alkaloids from Narcissus poeticus cv. Pink Parasol of various structural types and their biological activity. Arch. Pharm. Res. 2018, 41, 208–218. [Google Scholar] [CrossRef]
- Martinez-Peinado, N.; Cortes-Serra, N.; Torras-Claveria, L.; Pinazo, M.J.; Gascon, J.; Bastida, J.; Alonso-Padilla, J. Amaryllidaceae alkaloids with anti-Trypanosoma cruzi activity. Parasit. Vectors 2020, 13, 1–10. [Google Scholar] [CrossRef]
- Labraña, J.; Machocho, A.K.O.; Kricsfalusy, V.; Brun, R.; Codina, C.; Viladomat, F.; Bastida, J. Alkaloids from Narcissus angustifolius subsp. transcarpathicus (Amaryllidaceae). Phytochemistry 2002, 60, 847–852. [Google Scholar] [CrossRef] [PubMed]
- de Souza, W.; Barrias, E.S. May the epimastigote form of Trypanosoma cruzi be infective? Acta Trop. 2020, 212, 105688. [Google Scholar] [CrossRef]
- Kessler, R.L.; Contreras, V.T.; Marliére, N.P.; Aparecida Guarneri, A.; Villamizar Silva, L.H.; Mazzarotto, G.A.C.A.; Batista, M.; Soccol, V.T.; Krieger, M.A.; Probst, C.M. Recently differentiated epimastigotes from Trypanosoma cruzi are infective to the mammalian host. Mol. Microbiol. 2017, 104, 712–736. [Google Scholar] [CrossRef] [PubMed]
- Castañeda, J.S.; Suta-Velásquez, M.; Mateus, J.; Pardo-Rodriguez, D.; Puerta, C.J.; Cuéllar, A.; Robles, J.; Cuervo, C. Preliminary chemical characterization of ethanolic extracts from Colombian plants with promising anti-Trypanosoma cruzi activity. Exp. Parasitol. 2021, 223, 108079. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Peinado, N.; Ortiz, J.E.; Cortes-Serra, N.; Pinazo, M.J.; Gascon, J.; Tapia, A.; Roitman, G.; Bastida, J.; Feresin, G.E.; Alonso-Padilla, J. Anti-Trypanosoma cruzi activity of alkaloids isolated from Habranthus brachyandrus (Amaryllidaceae) from Argentina. Phytomedicine 2022, 101, 154126. [Google Scholar] [CrossRef] [PubMed]
- Puente, V.; Demaria, A.; Frank, F.M.; Batlle, A.; Lombardo, M.E. Anti-parasitic effect of vitamin C alone and in combination with benznidazole against Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2018, 12, e0006764. [Google Scholar] [CrossRef]
- Santos, S.S.; de Araujo, R.V.; Giarolla, J.; El Seoud, O.; Ferreira, E.I. Searching for drugs for Chagas disease, leishmaniasis and schistosomiasis: A review. Int. J. Antimicrob. Agents 2020, 55, 105906. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).