Plant Growth-Promoting Bacterial Consortia as a Strategy to Alleviate Drought Stress in Spinacia oleracea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Phenotypic Characterization and Growth Conditions
2.3. Bioassays for PGP Traits
2.3.1. Biofilm Production and Swarming Motility
2.3.2. Phosphate Solubilization
2.3.3. Indole-Acetic Acid (IAA) Detection
2.3.4. Ammonia Production
2.3.5. Siderophores Production
2.3.6. Biosurfactants Production
2.3.7. Screening for Hydrolytic Enzymatic Activity
2.3.8. Catalase Assay
2.3.9. DPPH Assay
2.4. Germination Assay
2.5. Adhesion Assay
2.6. Microbial Compatibility In Vitro
2.7. Drought Stress Treatment
2.8. Fluorescence Emission Measurements and Photosynthetic Pigment Determination
2.9. Statistical Analysis
3. Results
3.1. In Vitro Characterization of Potential DT-PGPB
3.2. Characterization of PGP Traits under Drought Stress Conditions
3.3. Antioxidant and Scavenging Activity
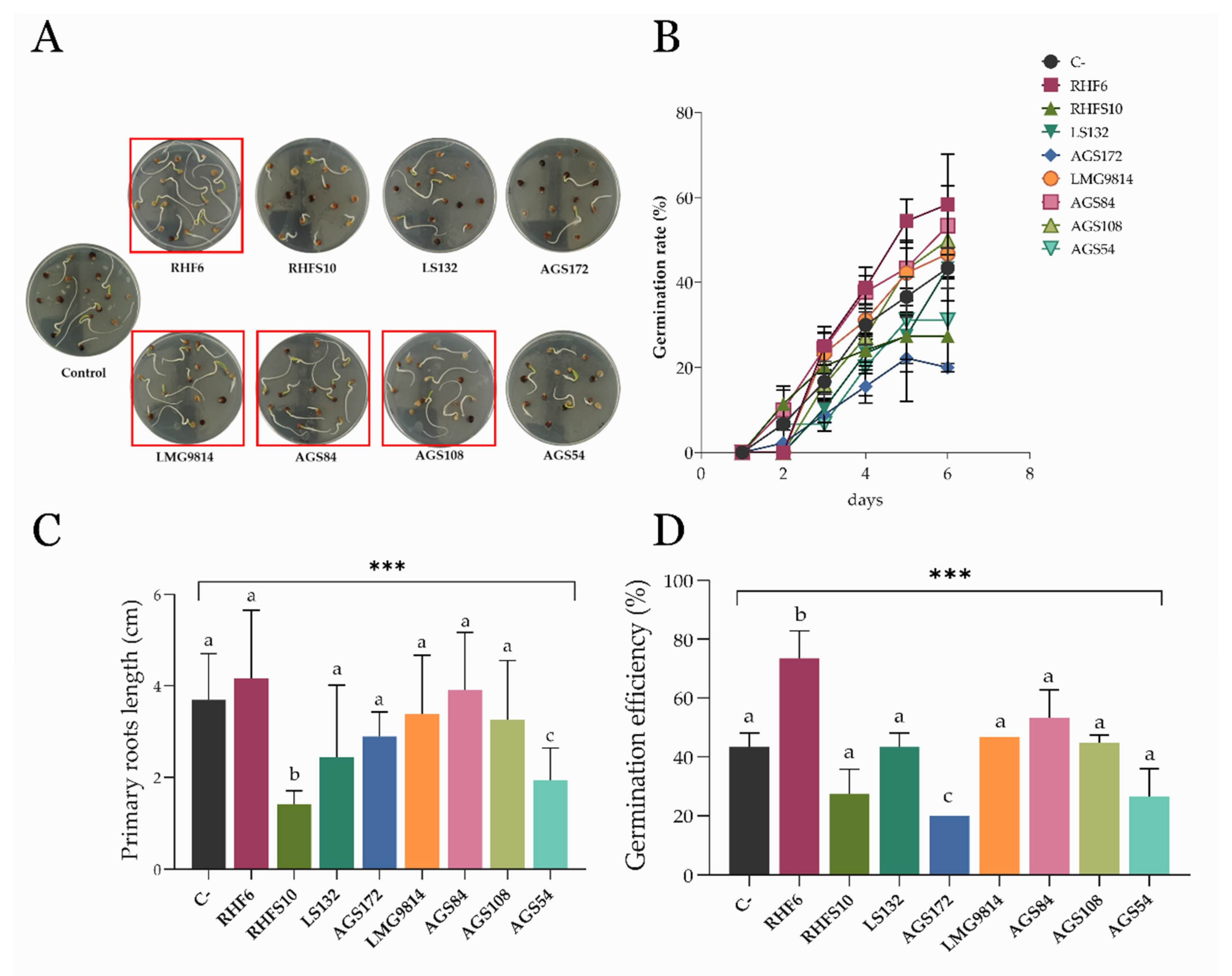
3.4. Effects of Seed-Biopriming on S. oleracea Germination In Vitro
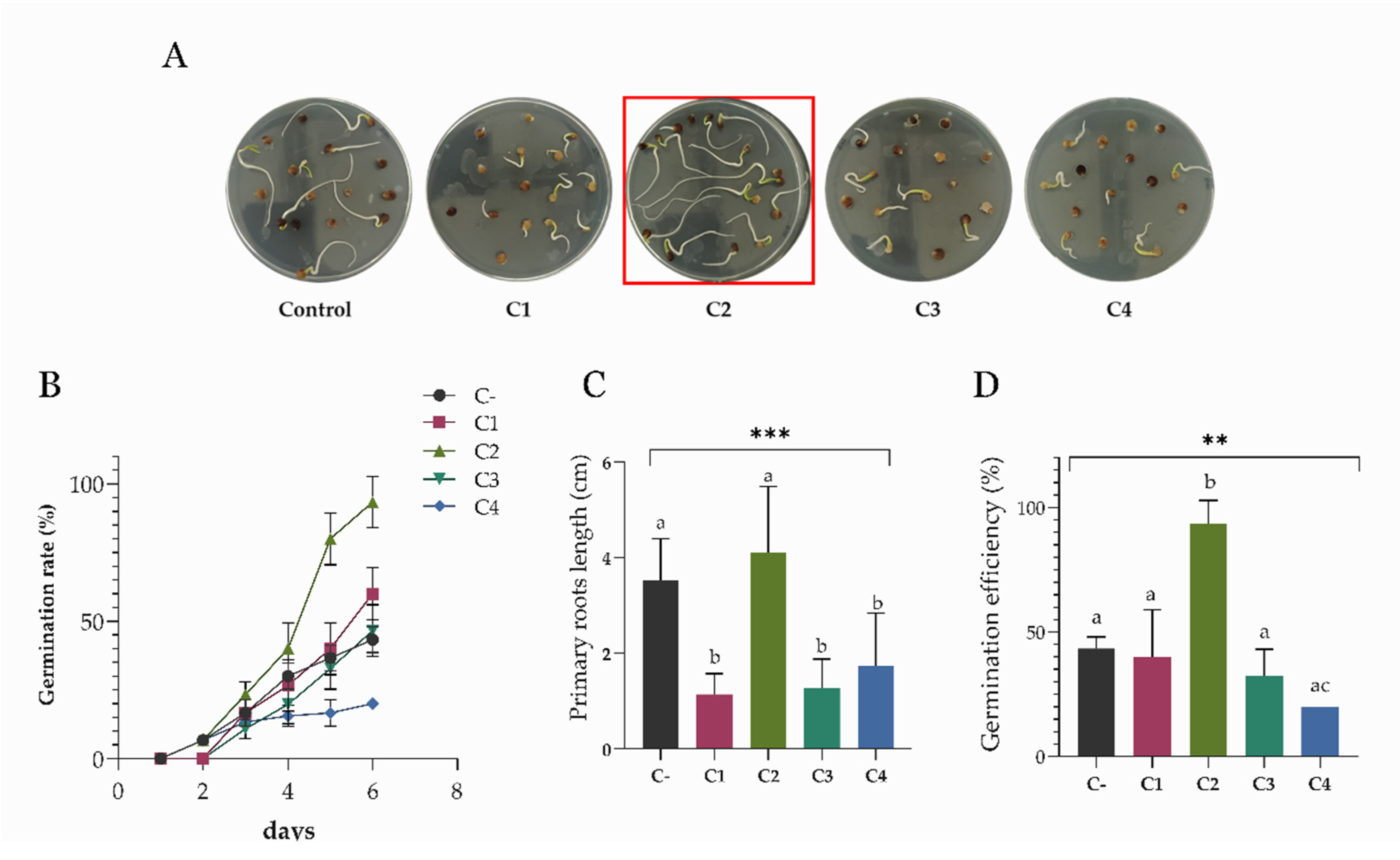
3.5. Effects of Bacterial Consortia on S. oleracea Germination In Vitro
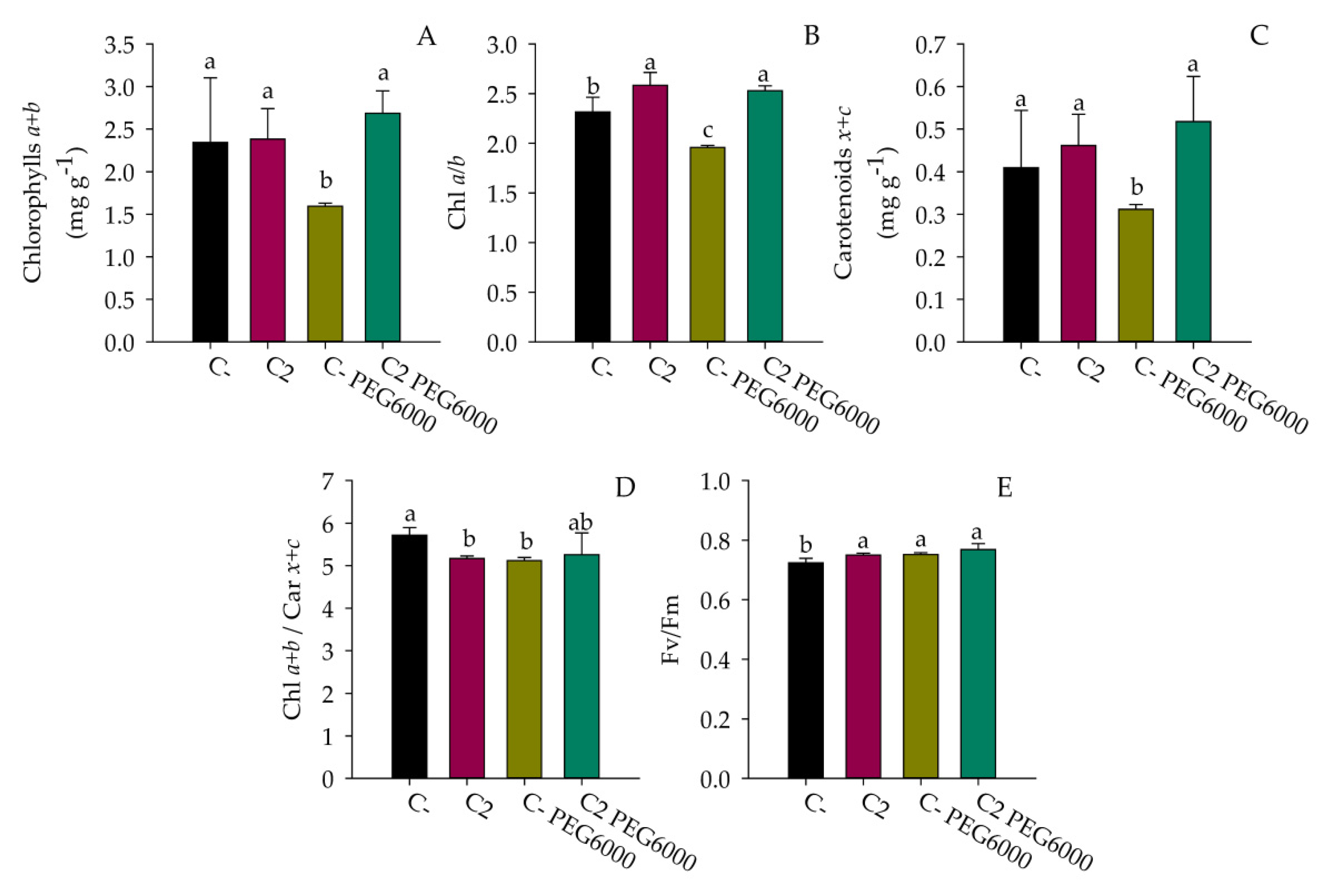
3.6. Drought Stress Treatment
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hanaka, A.; Ozimek, E.; Reszczyńska, E.; Jaroszuk-Ściseł, J.; Stolarz, M. Plant Tolerance to Drought Stress in the Presence of Supporting Bacteria and Fungi: An Efficient Strategy in Horticulture. Horticulturae 2021, 7, 390. [Google Scholar] [CrossRef]
- Seleiman, M.F.; Al-Suhaibani, N.; Ali, N.; Akmal, M.; Alotaibi, M.; Refay, Y.; Dindaroglu, T.; Abdul-Wajid, H.H.; Battaglia, M.L. Drought Stress Impacts on Plants and Different Approaches to Alleviate Its Adverse Effects. Plants 2021, 10, 259. [Google Scholar] [CrossRef] [PubMed]
- Sati, D.; Pande, V.; Pandey, S.C.; Samant, M. Recent Advances in PGPR and Molecular Mechanisms Involved in Drought Stress Tolerance. Preprints 2021, 2021050331. [Google Scholar] [CrossRef]
- Abbasi, T.; Abbasi, S.A. Biomass Energy and the Environmental Impacts Associated with Its Production and Utilization. Renew. Sustain. Energy Rev. 2010, 14, 919–937. [Google Scholar] [CrossRef]
- Ullah, A.; Nisar, M.; Ali, H.; Hazrat, A.; Hayat, K.; Keerio, A.A.; Ihsan, M.; Laiq, M.; Ullah, S.; Fahad, S.; et al. Drought Tolerance Improvement in Plants: An Endophytic Bacterial Approach. Appl. Microbiol. Biotechnol. 2019, 103, 7385–7397. [Google Scholar] [CrossRef]
- Khan, N.; Ali, S.; Shahid, M.A.; Mustafa, A.; Sayyed, R.Z.; Curá, J.A. Insights into the Interactions among Roots, Rhizosphere, and Rhizobacteria for Improving Plant Growth and Tolerance to Abiotic Stresses: A Review. Cells 2021, 10, 1551. [Google Scholar] [CrossRef] [PubMed]
- Arena, C.; Vitale, L.; De Santo, A.V. Paraheliotropism in Robinia pseudoacacia L.: An Efficient Strategy to Optimise Photosynthetic Performance under Natural Environmental Conditions. Plant Biol. 2008, 10, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Vitale, L.; Tommasi, P.; Arena, C.; Riondino, M.; Forte, A.; Verlotta, A.; Fierro, A.; Virzo De Santo, A.; Fuggi, A.; Magliulo, V. Growth and Gas Exchange Response to Water Shortage of a Maize Crop on Different Soil Types. Acta Physiol. Plant. 2009, 31, 331–341. [Google Scholar] [CrossRef]
- Rigano, M.M.; Arena, C.; Di Matteo, A.; Sellitto, S.; Frusciante, L.; Barone, A. Eco-Physiological Response to Water Stress of Drought-Tolerant and Drought-Sensitive Tomato Genotypes. Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2016, 150, 682–691. [Google Scholar] [CrossRef]
- Fang, Y.; Xiong, L. General Mechanisms of Drought Response and Their Application in Drought Resistance Improvement in Plants. Cell. Mol. Life Sci. 2015, 72, 673–689. [Google Scholar] [CrossRef]
- Ngumbi, E.; Kloepper, J. Bacterial-mediated drought tolerance: Current and future prospects. Appl. Soil Ecol. 2016, 105, 109–125. [Google Scholar] [CrossRef]
- Aslam, M.U.; Raza, M.A.S.; Saleem, M.F.; Waqas, M.; Iqbal, R.; Ahmad, S. Imran Haider Improving Strategic Growth Stage-Based Drought Tolerance in Quinoa by Rhizobacterial Inoculation. Commun. Soil Sci. Plant Anal. 2020, 51, 853–868. [Google Scholar] [CrossRef]
- Kour, D.; Rana, K.L.; Yadav, A.N.; Sheikh, I.; Kumar, V.; Dhaliwal, H.S.; Saxena, A.K. Amelioration of drought stress in Foxtail millet (Setaria italica L.) by P-solubilizing drought-tolerant microbes with multifarious plant growth promoting attributes. Environ. Sustain. 2020, 3, 23–34. [Google Scholar] [CrossRef]
- Francesca, S.; Arena, C.; Hay Mele, B.; Schettini, C.; Ambrosino, P.; Barone, A.; Rigano, M.M. The Use of a Plant-Based Biostimulant Improves Plant Performances and Fruit Quality in Tomato Plants Grown at Elevated Temperatures. Agronomy 2020, 10, 363. [Google Scholar] [CrossRef]
- Rouphael, Y.; Micco, V.D.; Arena, C.; Raimondi, G.; Colla, G.; Pascale, S.D. Effect of Ecklonia Maxima Seaweed Extract on Yield, Mineral Composition, Gas Exchange, and Leaf Anatomy of Zucchini Squash Grown under Saline Conditions. J. Appl. Phycol. 2016, 1, 459–470. [Google Scholar] [CrossRef]
- Niu, X.; Song, L.; Xiao, Y.; Ge, W. Drought-Tolerant Plant Growth-Promoting Rhizobacteria Associated with Foxtail Millet in a Semi-Arid Agroecosystem and Their Potential in Alleviating Drought Stress. Front. Microbiol. 2018, 8, 2580. [Google Scholar] [CrossRef]
- Castaldi, S.; Petrillo, C.; Donadio, G.; Piaz, F.D.; Cimmino, A.; Masi, M.; Evidente, A.; Isticato, R. Plant Growth Promotion Function of Bacillus Sp. Strains Isolated from Salt-Pan Rhizosphere and Their Biocontrol Potential against Macrophomina Phaseolina. Int. J. Mol. Sci. 2021, 22, 3324. [Google Scholar] [CrossRef]
- Petrillo, C.; Castaldi, S.; Lanzilli, M.; Selci, M.; Cordone, A.; Giovannelli, D.; Isticato, R. Genomic and Physiological Characterization of Bacilli Isolated from Salt-Pans with Plant Growth Promoting Features. Front. Microbiol. 2021, 12, 715678. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Ali, S.; Babar, M.A. Crosstalk amongst phytohormones from planta and PGPR under biotic and abiotic stresses. Plant Growth Regul. 2020, 90, 189–203. [Google Scholar] [CrossRef]
- Ansari, F.A.; Jabeen, M.; Ahmad, I. Pseudomonas azotoformans FAP5, a novel biofilm-forming PGPR strain, alleviates drought stress in wheat plant. Int. J. Environ. Sci. Technol. 2021, 18, 3855–38706. [Google Scholar] [CrossRef]
- Wang, C.-J.; Yang, W.; Wang, C.; Gu, C.; Niu, D.-D.; Liu, H.-X.; Wang, Y.-P.; Guo, J.-H. Induction of Drought Tolerance in Cucumber Plants by a Consortium of Three Plant Growth-Promoting Rhizobacterium Strains. PLoS ONE 2012, 7, e52565. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Bano, A.; Rahman, M.A.; Guo, J.; Kang, Z.; Babar, M.A. Comparative physiological and metabolic analysis reveals a complex mechanism involved in drought tolerance in chickpea (Cicer arietinum L.) induced by PGPR and PGRs. Sci. Rep. 2019, 9, 2097. [Google Scholar] [CrossRef]
- Meenakshi, K.; Govindasamy, V.; Ajit, V.; Choudhary, D.K. Mitigation of drought stress in wheat crop by drought tolerant endophytic bacterial isolates. Vegetos 2019, 32, 486–493. [Google Scholar] [CrossRef]
- Bianchi, A.; Masseroni, D.; Facchi, A. Modelling Water Requirements of Greenhouse Spinach for Irrigation Management Purposes. Hydrol. Res. 2016, 48, 776–788. [Google Scholar] [CrossRef]
- Giglio, R.; Fani, R.; Isticato, R.; De Felice, M.; Ricca, E.; Baccigalupi, L. Organization and Evolution of the CotG and CotH Genes of Bacillus Subtilis. J. Bacteriol. 2011, 193, 6664–6673. [Google Scholar] [CrossRef]
- Cangiano, G.; Sirec, T.; Panarella, C.; Isticato, R.; Baccigalupi, L.; De Felice, M.; Ricca, E. The Sps Gene Products Affect the Germination, Hydrophobicity, and Protein Adsorption of Bacillus Subtilis Spores. Appl. Environ. Microbiol. 2014, 80, 7293–7302. [Google Scholar] [CrossRef]
- Petrillo, C.; Castaldi, S.; Lanzilli, M.; Saggese, A.; Donadio, G.; Baccigalupi, L.; Ricca, E.; Isticato, R. The Temperature of Growth and Sporulation Modulates the Efficiency of Spore-Display in Bacillus Subtilis. Microb. Cell Fact. 2020, 19, 185. [Google Scholar] [CrossRef]
- Ahmad, M.A.; Javed, R.; Adeel, M.; Rizwan, M.; Yang, Y. PEG 6000-Stimulated Drought Stress Improves the Attributes of In Vitro Growth, Steviol Glycosides Production, and Antioxidant Activities in Stevia rebaudiana Bertoni. Plants 2020, 9, 1552. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, G.A. Microtiter Dish Biofilm Formation Assay. J. Vis. Exp. 2011, 2437. [Google Scholar] [CrossRef]
- Schoebitz, M.; Ceballos, C.; Ciamp, L. Effect of Immobilized Phosphate Solubilizing Bacteria on Wheat Growth and Phosphate Uptake. J. Soil Sci. Plant Nutr. 2013, 13, 1–10. [Google Scholar] [CrossRef]
- Damodaran, T.; Rb, R.; Jha, S.; Pandey, B.K.; Sah, V.; Mishra, V.; Sharma, D. Rhizosphere and Endophytic Bacteria for Induction of Salt Tolerance in Gladiolus Grown in Sodic Soils. J. Plant Interact. 2014, 9, 577–584. [Google Scholar] [CrossRef]
- Gordon, S.A.; Weber, R.P. Colorimetric estimation of indoleacetic acid. Plant Physiol. 1951, 26, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Miranda, S.; Cabirol, N.; George-Téllez, R.; Zamudio-Rivera, L.S.; Fernández, F.J. O-CAS, a Fast and Universal Method for Siderophore Detection. J. Microbiol. Methods 2007, 70, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Sarwar, A.; Brader, G.; Corretto, E.; Aleti, G.; Abaidullah, M.; Sessitsch, A.; Hafeez, F.Y. Qualitative Analysis of Biosurfactants from Bacillus Species Exhibiting Antifungal Activity. PLoS ONE 2018, 13, e0198107. [Google Scholar] [CrossRef]
- Morris, L.S.; Evans, J.; Marchesi, J.R. A Robust Plate Assay for Detection of Extracellular Microbial Protease Activity in Metagenomic Screens and Pure Cultures. J. Microbiol. Methods 2012, 91, 144–146. [Google Scholar] [CrossRef]
- Alariya, S.S.; Sethi, S.; Gupta, S.; Gupta, B.L. Amylase Activity of a Starch Degrading Bacteria Isolated from Soil. Arch. Appl. Sci. Res. 2013, 5, 15–24. [Google Scholar]
- Meddeb-Mouelhi, F.; Moisan, J.K.; Beauregard, M. A Comparison of Plate Assay Methods for Detecting Extracellular Cellulase and Xylanase Activity. Enzyme Microb. Technol. 2014, 66, 16–19. [Google Scholar] [CrossRef]
- Hankin, L.; Anagnostakis, S.L. Solid Media Containing Carboxymethylcellulose to Detect CX Cellulose Activity of Micro-Organisms. J. Gen. Microbiol. 1977, 98, 109–115. [Google Scholar] [CrossRef]
- Beers, R.F.; Sizer, I.W. A Spectrophotometric Method for Measuring the Breakdown of Hydrogen Peroxide by Catalase. J. Biol. Chem. 1952, 195, 133–140. [Google Scholar] [CrossRef]
- Mazzoli, A.; Donadio, G.; Lanzilli, M.; Saggese, A.; Guarino, A.M.; Rivetti, M.; Crescenzo, R.; Ricca, E.; Ferrandino, I.; Iossa, S.; et al. Bacillus megaterium SF185 Spores Exert Protective Effects against Oxidative Stress in Vivo and in Vitro. Sci. Rep. 2019, 9, 12082. [Google Scholar] [CrossRef]
- Wang, C.; Wu, B.; Jiang, K. Allelopathic Effects of Canada Goldenrod Leaf Extracts on the Seed Germination and Seeding Growth of Lettuce Reinforced under Salt Stress. Ecotoxicology 2019, 28, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, I.; Paul, C.; Al-Dourobi, A.; Sandoz, F.; Deschamps, P.; Junier, T.; Junier, P.; Bindschedler, S. Comparison of the Plant Growth-Promotion Performance of a Consortium of Bacilli Inoculated as Endospores or as Vegetative Cells. FEMS Microbiol. Ecol. 2019, 95, fiz147. [Google Scholar] [CrossRef] [PubMed]
- Tabacchioni, S.; Passato, S.; Ambrosino, P.; Huang, L.; Caldara, M.; Cantale, C.; Hett, J.; Del Fiore, A.; Fiore, A.; Schlüter, A.; et al. Identification of Beneficial Microbial Consortia and Bioactive Compounds with Potential as Plant Biostimulants for a Sustainable Agriculture. Microorganisms 2021, 9, 426. [Google Scholar] [CrossRef] [PubMed]
- Muchate, N.S.; Rajurkar, N.S.; Suprasanna, P.; Nikam, T.D. NaCl Induced Salt Adaptive Changes and Enhanced Accumulation of 20-Hydroxyecdysone in the in Vitro Shoot Cultures of Spinacia oleracea (L.). Sci. Rep. 2019, 9, 12522. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- Amaya-Gómez, C.V.; Porcel, M.; Mesa-Garriga, L.; Gómez-Álvarez, M.I. A Framework for the Selection of Plant Growth-Promoting Rhizobacteria Based on Bacterial Competence Mechanisms. Appl. Environ. Microbiol. 2020, 86, e00760–20. [Google Scholar] [CrossRef]
- Plunkett, M.H.; Knutson, C.M.; Barney, B.M. Key Factors Affecting Ammonium Production by an Azotobacter Vinelandii Strain Deregulated for Biological Nitrogen Fixation. Microb. Cell Factories 2020, 19, 107. [Google Scholar] [CrossRef]
- Bouskill, N.J.; Wood, T.E.; Baran, R.; Ye, Z.; Bowen, B.P.; Lim, H.; Zhou, J.; Nostrand, J.D.V.; Nico, P.; Northen, T.R.; et al. Belowground Response to Drought in a Tropical Forest Soil. I. Changes in Microbial Functional Potential and Metabolism. Front. Microbiol. 2016, 7, 525. [Google Scholar] [CrossRef]
- Rezayian, M.; Niknam, V.; Ebrahimzadeh, H. Effects of Drought Stress on the Seedling Growth, Development, and Metabolic Activity in Different Cultivars of Canola. Soil Sci. Plant Nutr. 2018, 64, 360–369. [Google Scholar] [CrossRef]
- Baez-Rogelio, A.; Morales-García, Y.E.; Quintero-Hernández, V.; Muñoz-Rojas, J. Next Generation of Microbial Inoculants for Agriculture and Bioremediation. Microb. Biotechnol. 2017, 10, 19–21. [Google Scholar] [CrossRef]
- Mayak, S.; Tirosh, T.; Glick, B.R. Plant Growth-Promoting Bacteria That Confer Resistance to Water Stress in Tomatoes and Peppers. Plant Sci. 2004, 166, 525–530. [Google Scholar] [CrossRef]
- Arshad, M.; Shaharoona, B.; Mahmood, T. Inoculation with Pseudomonas Spp. Containing ACC-Deaminase Partially Eliminates the Effects of Drought Stress on Growth, Yield, and Ripening of Pea (Pisum sativum L.) *1 *1 Project Supported by the Higher Education Commission, Islamabad, Pakistan (No. PIN 041 211534 A-031). Pedosphere 2008, 18, 611–620. [Google Scholar] [CrossRef]
- Bashan, Y. Inoculants of Plant Growth-Promoting Bacteria for Use in Agriculture. Biotechnol. Adv. 1998, 16, 729–770. [Google Scholar] [CrossRef]
- Abdelaal, K.; Alkahtani, M.; Attia, K.; Hafez, Y.; Király, L.; Künstler, A. The Role of Plant Growth-Promoting Bacteria in Alleviating the Adverse Effects of Drought on Plants. Biology 2021, 10, 520. [Google Scholar] [CrossRef]


| Strain | Species | Source | Citation |
|---|---|---|---|
| RHF6 | B. amyloliquefaciens | Sand (Spain) | [18] |
| RHFS10 | B. vallismortis | Rhizosphere (Spain) | [17] |
| LS132 | A. chroococcum | Rhizosphere (Italy) | Agriges collection |
| AGS172 | B. subtilis | Unknown | Agriges collection |
| LMG9814 | B. amyloliquefaciens | Soil | Agriges collection |
| AGS84 | Bacillus. sp. | Grape leaves | Agriges collection |
| AGS108 | B. amyloliquefaciens | Unknown | Agriges collection |
| AGS54 | P. fluorescens | Soil | Agriges collection |
| PGPR Traits | Hydrolytic Activities (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Strain | PVK | IAA (µg/mL) | Ammonia Production (mg/L) | Siderophores (%) | Biosurfactants | Protease | Amylase | Xylanase | CMC |
| RHF6 | + | 4.2 ± 0.10 | 1.3 ± 0.02 | 3.5 | ++ | 0 | 0 | 0 | 0 |
| RHFS10 | + | 18 ± 0.03 | 0.0 | 25 | +++ | 0 | 50 | 75 | 0 |
| LS132 | − | 2.4 ± 0.03 | 1.8 ± 0.01 | 0 | + | 0 | 0 | 0 | 0 |
| AGS172 | + | 5.2 ± 0.02 | 1.7 ± 0.01 | 5.2 | ++ | 100 | 50 | 75 | 0 |
| LMG9814 | − | 3.5 ± 0.09 | 0.8 ± 0.09 | 3.2 | ++ | 100 | 66.7 | 0 | 0 |
| AGS84 | + | 4.4 ± 0.03 | 1.1 ± 0.02 | 4.6 | ++ | 0 | 100 | 75 | 0 |
| AGS108 | + | 2.3 ± 0.05 | 0.6 ± 0.01 | 3.5 | + | 100 | 75 | 75 | 0 |
| AGS54 | − | 2.1 ± 0.03 | 1.4 ± 0.04 | 20 | ++ | 0 | 0 | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrillo, C.; Vitale, E.; Ambrosino, P.; Arena, C.; Isticato, R. Plant Growth-Promoting Bacterial Consortia as a Strategy to Alleviate Drought Stress in Spinacia oleracea. Microorganisms 2022, 10, 1798. https://doi.org/10.3390/microorganisms10091798
Petrillo C, Vitale E, Ambrosino P, Arena C, Isticato R. Plant Growth-Promoting Bacterial Consortia as a Strategy to Alleviate Drought Stress in Spinacia oleracea. Microorganisms. 2022; 10(9):1798. https://doi.org/10.3390/microorganisms10091798
Chicago/Turabian StylePetrillo, Claudia, Ermenegilda Vitale, Patrizia Ambrosino, Carmen Arena, and Rachele Isticato. 2022. "Plant Growth-Promoting Bacterial Consortia as a Strategy to Alleviate Drought Stress in Spinacia oleracea" Microorganisms 10, no. 9: 1798. https://doi.org/10.3390/microorganisms10091798
APA StylePetrillo, C., Vitale, E., Ambrosino, P., Arena, C., & Isticato, R. (2022). Plant Growth-Promoting Bacterial Consortia as a Strategy to Alleviate Drought Stress in Spinacia oleracea. Microorganisms, 10(9), 1798. https://doi.org/10.3390/microorganisms10091798









