Actinobacteria as Effective Biocontrol Agents against Plant Pathogens, an Overview on Their Role in Eliciting Plant Defense
Abstract
1. Introduction
2. Actinobacteria as Successful Biocontrol Agents
| Strain | Host | Pathogen | Reference |
|---|---|---|---|
| Streptomyces halstedii AJ-7 | Red pepper | Phytophthora capsici | [52] |
| Streptomyces sp. CA2, AA2 | Tomato | Rhizoctonia solani | [22] |
| S. griseus | Tomato | Fusarium sp. | [53] |
| Streptomyces sp. S2,C | Sugar beet | Rhizoctonia solani | [54] |
| Streptomyces sp. MBCu-56 | Cucurbit | Colletotrichum orbiculare | [55] |
| S. aurantiogriseus VSMGT1014 | Rice | Rhizoctonia solani | [56] |
| Streptomyces sp. J-2 | Sugar beet | Sclerotium rolfsii | [57] |
| Streptomyces spp. | Sugar beet | Fusarium spp. | [58] |
| Actinoplanes campanulatus #2 Micromonospora chalcea #8 S. spiralis #17 | Cucumber | Pythium aphanidermatum | [41] |
| Streptomyces sp. strain g10 S. malaysiensis 8ZJF-21 | Banana | Fusarium oxysporum f.sp. cubense | [59] [60] |
| Streptomyces sp. S160 | Chickpea | Macrophomina phaseolina | [61] |
| Amycolatopsis sp. 521 | Apple | Colletotrichum gloeosporioides | [62] |
| S. albidoflavus | Tomato | Alternaria solani, A. alternata, Colletotrichum gloeosporioides, Fusarium oxysporum, Fusarium solani, Rhizoctonia solani, and Botrytis cinerea | [63] |
| Streptomyces sp. A1022 | Pepper, Cherry Tomato | Colletotrichum gloeosporioides | [64] |
| S. misionensis BH4-1,BH4-3 | Pistachio | Paecilomycesformosus | [65] |
| S. globisporus JK-1 | Rice | Magnaporthe oryzae | [66] |
| Streptomyces sp. MT7 | - | Wood-rotting fungi | [42] |
| S. mutabilis IA1 | Wheat | Fusarium culmorum | [67] |
| Micromonospora sp. ALFpr18c, ALFb5 | Tomato | Botrytis cinerea | [68] |
| S. globosus UAE1 | Date Palm | Thielaviopsis punctulata | [69] |
| Streptomyces spp. A20, 5.1, 7.1 | Rice | Burkholderia glumae | [70] |
| S. angustmyceticus NR8-2 | Cabbage | Colletotrichum sp. and Curvularia lunata | [51] |
| Streptomyces sp. HAAG3-15 | Cucumber | F. oxysporum f.sp. cucumerinum | [71] |
| Streptomyces spp. R7,F8 | Tomato | R. solani | [72] |
| S. laydicus M01 | Cucumber | A. alternata | [73] |
| S. fulvissimus Uts22 | Cucumber Wheat | Pythium aphanidermatum and Gaeumannomyces graminis var. tritici | [74] [75] |
| Streptomyces sp. TP199 | Potato | Pectobacterium carotovorum subsp. Carotovorum, and Pectobacterium atrosepticum | [76] |
| S. violaceusniger AC12AB | Potato | Streptomyces scabies | [77] |
| Streptomyces sp. AN090126 | Tomato Red Pepper Creeping bentgrass | Ralstonia solanacearum, Xanthomonas euvesicatoria, and Sclerotinia homoeocarpa | [78] |
3. The Potential of Actinobacteria to Induce Systemic Resistance in Plants
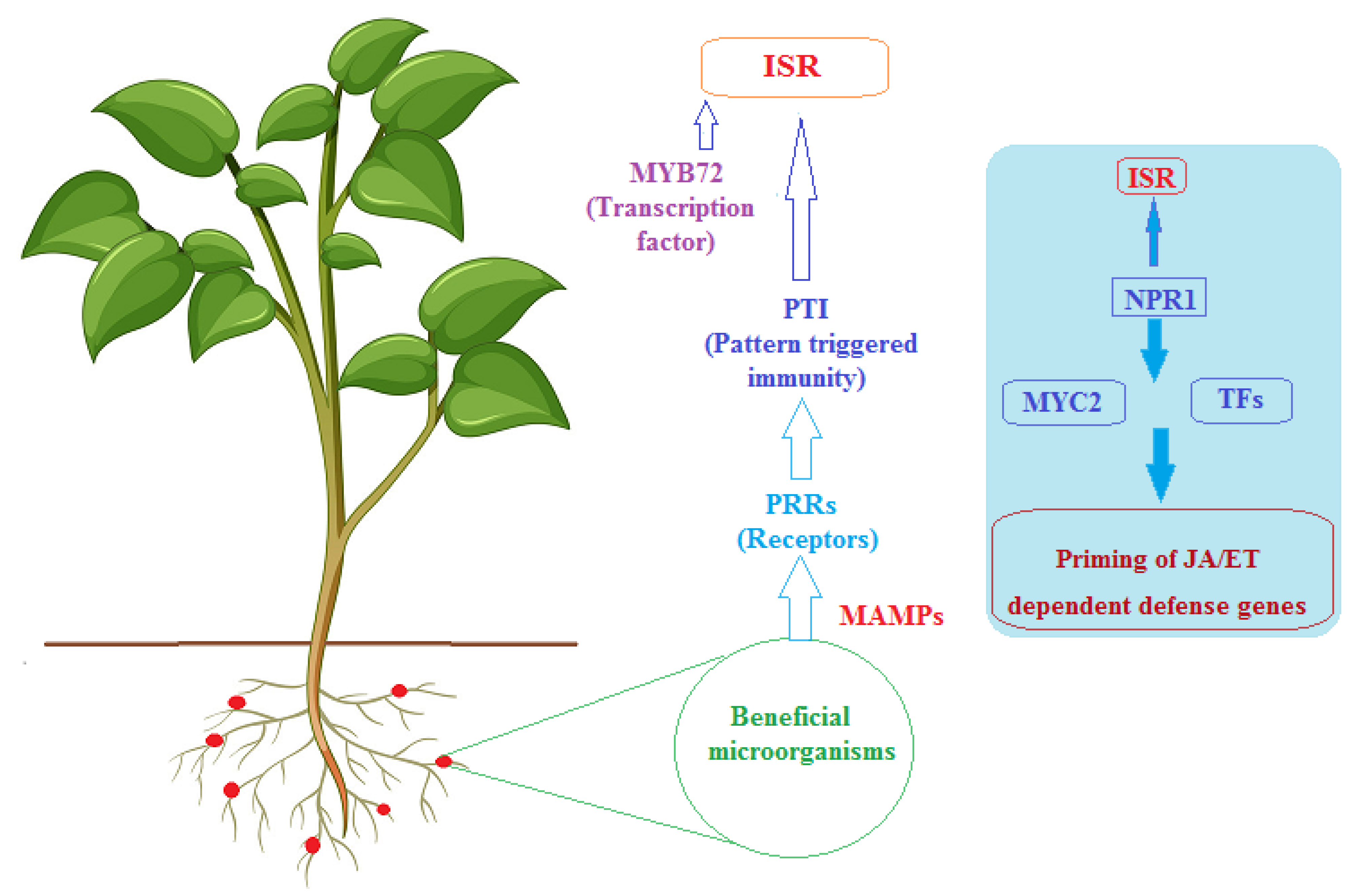
3.1. General Mechanisms of Induced Systemic Resistance (ISR)
3.2. Actinobacteria Priming Plant Defense
4. Enrichment of Actinobacteria during the Establishment of Suppressive Soils, Pathogen Attacks and Abiotic Stress: A Sign of Their Central Role in Plant Protection?
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, B.; Trivedi, P.; Singh, S.; Macdonald, C.; Verma, J. Emerging microbiome technologies for sustainable increase in farm productivity and environmental security. Microbiol. Aust. 2018, 39, 17–23. [Google Scholar] [CrossRef]
- Nega, A. Review on concepts in biological control of plant pathogens. J. Biol. Agric. Health 2014, 4, 33–54. [Google Scholar]
- Jamali, F.; Sharifi-Tehrani, A.; Okhovvat, M.; Zakeri, Z.; Saberi-Riseh, R. Biological control of chickpea Fusarium wilt by antagonistic bacteria under greenhouse condition. Commun. Agric. Appl. Biol. Sci. 2004, 69, 649–651. [Google Scholar] [PubMed]
- Moradi Pour, M.; Saberi-Riseh, R.; Mohammadinejad, R.; Hosseini, A. Investigating the formulation of alginate-gelatin encapsulated Pseudomonas fluorescens (VUPF5 and T17-4 strains) for controlling Fusarium solani on potato. Int. J. Biol. Macromol. 2019, 133, 603–613. [Google Scholar] [CrossRef]
- Fathi, F.; Saberi-Riseh, R.; Khodaygan, P. Survivability and controlled release of alginate-microencapsulated Pseudomonas fluorescens VUPF506 and their effects on biocontrol of Rhizoctonia solani on potato. Int. J. Biol. Macromol. 2021, 183, 627–634. [Google Scholar] [CrossRef]
- Saberi-Riseh, R.; Hajieghrari, B.; Rouhani, H.; Sharifi-Tehrani, A. Effects of inoculum density and substrate type on saprophytic survival of Phytophthora drechsleri, the causal agent of gummosis (crown and root rot) on pistachio in Rafsanjan, Iran. Commun. Agric. Appl. Biol. Sci. 2004, 69, 653–656. [Google Scholar]
- Saberi Riseh, R.; Skorik, Y.A.; Thakur, V.K.; Moradi Pour, M.; Tamanadar, E.; Noghabi, S.S. Encapsulation of plant biocontrol bacteria with alginate as a main polymer material. Int. J. Mol. Sci. 2021, 22, 11165. [Google Scholar] [CrossRef]
- Saberi Riseh, R.; Javan-Nikkhah, M.; Heidarian, R.; Hosseini, S.; Soleimani, P. Detection of fungal infectous agent of wheat grains in store-pits of Markazi province, Iran. Commun. Agric. Appl. Biol. Sci. 2004, 69, 541–544. [Google Scholar]
- Morales-Cedeño, L.R.; Orozco-Mosqueda, M.d.C.; Loeza-Lara, P.D.; Parra-Cota, F.I.; de los Santos-Villalobos, S.; Santoyo, G. Plant growth-promoting bacterial endophytes as biocontrol agents of pre- and post-harvest diseases: Fundamentals, methods of application and future perspectives. Microbiol. Res. 2021, 242, 126612. [Google Scholar] [CrossRef]
- Ludwig, W.; Euzéby, J.; Schumann, P.; Busse, H.-J.; Trujillo, M.; Kämpfer, P.; Whitman, W. Road Map of the Phylum Actinobacteria. In Bergey’s Manual of Systematic Bacteriology, 2nd ed.; Goodfellow, M., Kämpfer, P., Busse, H.-J., Trujillo, M.E., Suzuki, K., Ludwig, W., Whitman, W.B., Eds.; Springer Nature: Cham, Switzerland, 2012; pp. 1–28. [Google Scholar]
- Palaniyandi, S.A.; Yang, S.H.; Zhang, L.; Suh, J.-W. Effects of actinobacteria on plant disease suppression and growth promotion. Appl. Microbiol. Biotechnol. 2013, 97, 9621–9636. [Google Scholar] [CrossRef]
- Viaene, T.; Langendries, S.; Beirinckx, S.; Maes, M.; Goormachtig, S. Streptomyces as a plant’s best friend? FEMS Microbiol. Ecol. 2016, 92, fiw119. [Google Scholar] [CrossRef] [PubMed]
- Khamna, S.; Yokota, A.; Peberdy, J.; Lumyong, S. Indole3-acetic acid production by Streptomyces sp. isolated from some Thai medicinal plant rhizosphere soils. EurAsian J. Biosci. 2010, 4, 23–32. [Google Scholar] [CrossRef]
- Chukwuneme, C.F.; Babalola, O.O.; Kutu, F.R.; Ojuederie, O.B. Characterization of actinomycetes isolates for plant growth promoting traits and their effects on drought tolerance in maize. J. Plant Interact. 2020, 15, 93–105. [Google Scholar] [CrossRef]
- Yamaura, M.; Uchiumi, T.; Higashi, S.; Abe, M.; Kucho, K.-I. Identification by suppression subtractive hybridization of Frankia genes induced under nitrogen-fixing conditions. Appl. Environ. Microbiol. 2010, 76, 1692–1694. [Google Scholar] [CrossRef] [PubMed]
- Hamdali, H.; Hafidi, M.; Virolle, M.J.; Ouhdouch, Y. Rock phosphate-solubilizing Actinomycetes: Screening for plant growth-promoting activities. World J. Microbiol. Biotechnol. 2008, 24, 2565–2575. [Google Scholar] [CrossRef]
- Oliveira, C.A.; Alves, V.M.C.; Marriel, I.E.; Gomes, E.A.; Scotti, M.R.; Carneiro, N.P.; Guimarães, C.T.; Schaffert, R.E.; Sá, N.M.H. Phosphate solubilizing microorganisms isolated from rhizosphere of maize cultivated in an oxisol of the Brazilian Cerrado Biome. Soil Biol. Biochem. 2009, 41, 1782–1787. [Google Scholar] [CrossRef]
- Franco-Correa, M.; Quintana, A.; Duque, C.; Suarez, C.; Rodríguez, M.X.; Barea, J.-M. Evaluation of actinomycete strains for key traits related with plant growth promotion and mycorrhiza helping activities. Appl. Soil Ecol. 2010, 45, 209–217. [Google Scholar] [CrossRef]
- Boubekri, K.; Soumare, A.; Mardad, I.; Lyamlouli, K.; Hafidi, M.; Ouhdouch, Y.; Kouisni, L. The screening of potassium- and phosphate-solubilizing Actinobacteria and the assessment of their ability to promote wheat growth parameters. Microorganisms 2021, 9, 470. [Google Scholar] [CrossRef]
- Solans, M. Discaria trinervis—Frankia symbiosis promotion by saprophytic actinomycetes. J. Basic Microbiol. 2007, 47, 243–250. [Google Scholar] [CrossRef]
- Riedlinger, J.; Schrey, S.D.; Tarkka, M.T.; Hampp, R.; Kapur, M.; Fiedler, H.P. Auxofuran, a novel metabolite that stimulates the growth of fly agaric, is produced by the mycorrhiza helper bacterium Streptomyces strain AcH 505. Appl. Environ. Microbiol. 2006, 72, 3550–3557. [Google Scholar] [CrossRef]
- Goudjal, Y.; Toumatia, O.; Yekkour, A.; Sabaou, N.; Mathieu, F.; Zitouni, A. Biocontrol of Rhizoctonia solani damping-off and promotion of tomato plant growth by endophytic actinomycetes isolated from native plants of Algerian Sahara. Microbiol. Res. 2014, 169, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Newitt, J.T.; Prudence, S.M.M.; Hutchings, M.I.; Worsley, S.F. Biocontrol of cereal crop diseases using Streptomycetes. Pathogens 2019, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Van Wees, S.C.; Van der Ent, S.; Pieterse, C.M. Plant immune responses triggered by beneficial microbes. Curr. Opin. Plant Biol. 2008, 11, 443–448. [Google Scholar] [CrossRef] [PubMed]
- De Vleesschauwer, D.; Djavaheri, M.; Bakker, P.A.H.M.; Höfte, M. Pseudomonas fluorescens WCS374r-induced systemic resistance in rice against Magnaporthe oryzae is based on pseudobactin-mediated priming for a salicylic acid-repressible multifaceted defense response. Plant Physiol. 2008, 148, 1996–2012. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; van Wees, S.C.M.; van Pelt, J.A.; Knoester, M.; Laan, R.; Gerrits, H.; Weisbeek, P.J.; van Loon, L.C. A novel signaling pathway controlling induced systemic resistance in Arabidopsis. Plant Cell 1998, 10, 1571–1580. [Google Scholar] [CrossRef]
- Conrath, U.; Beckers, G.J.M.; Langenbach, C.J.G.; Jaskiewicz, M.R. Priming for enhanced defense. Annu. Rev. Phytopathol. 2015, 53, 97–119. [Google Scholar] [CrossRef]
- Mauch-Mani, B.; Baccelli, I.; Luna Diez, E.; Flors, V. Defense priming: An adaptive part of induced resistance. Annu. Rev. Plant Biol. 2017, 68, 485–512. [Google Scholar] [CrossRef]
- Conn, V.M.; Walker, A.R.; Franco, C.M. Endophytic actinobacteria induce defense pathways in Arabidopsis thaliana. Mol. Plant Microbe Interact. 2008, 21, 208–218. [Google Scholar] [CrossRef]
- Cheng, J.; Yang, S.H.; Palaniyandi, S.A.; Han, J.S.; Yoon, T.-M.; Kim, T.-J.; Suh, J.-W. Azalomycin F complex is an antifungal substance produced by Streptomyces malaysiensis MJM1968 isolated from agricultural soil. J. Korean Soc. Appl. Biol. Chem. 2010, 53, 545–552. [Google Scholar] [CrossRef]
- Meschke, H.; Walter, S.; Schrempf, H. Characterization and localization of prodiginines from Streptomyces lividans suppressing Verticillium dahliae in the absence or presence of Arabidopsis thaliana. Environ. Microbiol. 2012, 14, 940–952. [Google Scholar] [CrossRef]
- Rungin, S.; Indananda, C.; Suttiviriya, P.; Kruasuwan, W.; Jaemsaeng, R.; Thamchaipenet, A. Plant growth enhancing effects by a siderophore-producing endophytic streptomycete isolated from a Thai jasmine rice plant (Oryza sativa L. cv. KDML105). Antonie Leeuwenhoek 2012, 102, 463–472. [Google Scholar] [CrossRef]
- Aznar, A.; Dellagi, A. New insights into the role of siderophores as triggers of plant immunity: What can we learn from animals? J. Exp. Bot. 2015, 66, 3001–3010. [Google Scholar] [CrossRef]
- Sadeghi, A.; Koobaz, P.; Azimi, H.; Karimi, E.; Akbari, A. Plant growth promotion and suppression of Phytophthora drechsleri damping-off in cucumber by cellulase-producing Streptomyces. BioControl 2017, 62, 805–819. [Google Scholar] [CrossRef]
- El-Shatoury, S.; Elkraly, O.; El Kazzaz, W.; Dewedar, A. Antimicrobial activities of actinomycetes inhabiting Achillea fragrantissima (Family: Compositae). Egypt. J. Nat. Toxins 2009, 6, 1–15. [Google Scholar]
- Dimkpa, C.O.; Svatos, A.; Dabrowska, P.; Schmidt, A.; Boland, W.; Kothe, E. Involvement of siderophores in the reduction of metal-induced inhibition of auxin synthesis in Streptomyces spp. Chemosphere 2008, 74, 19–25. [Google Scholar] [CrossRef]
- Jog, R.; Nareshkumar, G.; Rajkumar, S. Enhancing Soil Health and Plant Growth Promotion by Actinomycetes. In Plant Growth Promoting Actinobacteria: A New Avenue for Enhancing the Productivity and Soil Fertility of Grain Legumes; Subramaniam, G., Arumugam, S., Rajendran, V., Eds.; Springer: Singapore, 2016; pp. 33–45. [Google Scholar]
- Lee, S.Y.; Tindwa, H.; Lee, Y.S.; Naing, K.W.; Hong, S.H.; Nam, Y.; Kim, K.Y. Biocontrol of anthracnose in pepper using chitinase, beta-1,3 glucanase, and 2-furancarboxaldehyde produced by Streptomyces cavourensis SY224. J. Microbiol. Biotechnol. 2012, 22, 1359–1366. [Google Scholar] [CrossRef]
- Joo, G.J. Purification and characterization of an extracellular chitinase from the antifungal biocontrol agent Streptomyces halstedii. Biotechnol. Lett. 2005, 27, 1483–1486. [Google Scholar] [CrossRef]
- Gherbawy, Y.; Elhariry, H.; Altalhi, A.; El-Deeb, B.; Khiralla, G. Molecular screening of Streptomyces isolates for antifungal activity and family 19 chitinase enzymes. J. Microbiol. 2012, 50, 459–468. [Google Scholar] [CrossRef]
- El-Tarabily, K.A.; Nassar, A.H.; Hardy, G.E.; Sivasithamparam, K. Plant growth promotion and biological control of Pythium aphanidermatum, a pathogen of cucumber, by endophytic actinomycetes. J. Appl. Microbiol. 2009, 106, 13–26. [Google Scholar] [CrossRef]
- Nagpure, A.; Choudhary, B.; Kumar, S.; Gupta, R.K. Isolation and characterization of chitinolytic Streptomyces sp. MT7 and its antagonism towards wood-rotting fungi. Ann. Microbiol. 2014, 64, 531–541. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Pande, S.; Sharma, M.; Humayun, P.; Kiran, B.K.; Sandeep, D.; Vidya, M.S.; Deepthi, K.; Rupela, O. Evaluation of actinomycete isolates obtained from herbal vermicompost for the biological control of Fusarium wilt of chickpea. Crop Prot. 2011, 30, 1070–1078. [Google Scholar] [CrossRef]
- Kamil, F.H.; Saeed, E.E.; El-Tarabily, K.A.; AbuQamar, S.F. Biological control of mango dieback disease caused by Lasiodiplodia theobromae using Streptomycete and Non-streptomycete Actinobacteria in the United Arab Emirates. Front. Microbiol. 2018, 9, 829. [Google Scholar] [CrossRef]
- Zhou, Z.; Wu, X.; Li, J.; Zhang, Y.; Huang, Y.; Zhang, W.; Shi, Y.; Wang, J.; Chen, S. A novel quorum quencher, Rhodococcus pyridinivorans XN-36, is a powerful agent for the biocontrol of soft rot disease in various host plants. Biol. Control 2022, 169, 104889. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, S.J.; Oh, T.K.; Oh, J.W.; Koo, B.T.; Yum, D.Y.; Lee, J.K. AhlD, an N-acylhomoserine lactonase in Arthrobacter sp., and predicted homologues in other bacteria. Microbiology 2003, 149, 1541–1550. [Google Scholar] [CrossRef]
- Citron, C.A.; Barra, L.; Wink, J.; Dickschat, J.S. Volatiles from nineteen recently genome sequenced actinomycetes. Org. Biomol. Chem. 2015, 13, 2673–2683. [Google Scholar] [CrossRef]
- Cordovez, V.; Carrion, V.J.; Etalo, D.W.; Mumm, R.; Zhu, H.; van Wezel, G.P.; Raaijmakers, J.M. Diversity and functions of volatile organic compounds produced by Streptomyces from a disease-suppressive soil. Front. Microbiol. 2015, 6, 1081. [Google Scholar] [CrossRef]
- Schmidt, R.; Cordovez, V.; de Boer, W.; Raaijmakers, J.; Garbeva, P. Volatile affairs in microbial interactions. ISME J. 2015, 9, 2329–2335. [Google Scholar] [CrossRef]
- Wan, M.; Li, G.; Zhang, J.; Jiang, D.; Huang, H.-C. Effect of volatile substances of Streptomyces platensis F-1 on control of plant fungal diseases. Biol. Control 2008, 46, 552–559. [Google Scholar] [CrossRef]
- Wonglom, P.; Suwannarach, N.; Lumyong, S.; Ito, S.-i.; Matsui, K.; Sunpapao, A. Streptomyces angustmyceticus NR8-2 as a potential microorganism for the biological control of leaf spots of Brassica rapa subsp. pekinensis caused by Colletotrichum sp. and Curvularia lunata. Biol. Control 2019, 138, 104046. [Google Scholar] [CrossRef]
- Joo, G.-J. Production of an anti-fungal substance for biological control of Phytophthora capsici causing phytophthora blight in red-peppers by Streptomyces halstedii. Biotechnol. Lett. 2005, 27, 201–205. [Google Scholar] [CrossRef]
- Anitha, A.; Rabeeth, M. Control of Fusarium wilt of tomato by bioformulation of Streptomyces griseus in green house condition. Afr. J. Basic Appl. Sci. 2009, 1, 9–14. [Google Scholar]
- Sadeghi, A.; Hessan, A.R.; Askari, H.; Aghighi, S.; Shahidi Bonjar, G.H. Biological control potential of two Streptomyces isolates on Rhizoctonia solani, the causal agent of damping-off of sugar beet. Pak. J. Biol. Sci. 2006, 9, 904–910. [Google Scholar] [CrossRef][Green Version]
- Shimizu, M.; Yazawa, S.; Ushijima, Y. A promising strain of endophytic Streptomyces sp. for biological control of cucumber anthracnose. J. Gen. Plant Pathol. 2009, 75, 27–36. [Google Scholar] [CrossRef]
- Harikrishnan, H.; Shanmugaiah, V.; Balasubramanian, N.; Sharma, M.P.; Kotchoni, S.O. Antagonistic potential of native strain Streptomyces aurantiogriseus VSMGT1014 against sheath blight of rice disease. World J. Microbiol. Biotechnol. 2014, 30, 3149–3161. [Google Scholar] [CrossRef]
- Errakhi, R.; Bouteau, F.; Lebrihi, A.; Barakate, M. Evidences of biological control capacities of Streptomyces spp. against Sclerotium rolfsii responsible for damping-off disease in sugar beet (Beta vulgaris L.). World J. Microbiol. Biotechnol. 2007, 23, 1503–1509. [Google Scholar] [CrossRef]
- Aallam, Y.; Dhiba, D.; El Rasafi, T.; Lemriss, S.; Haddioui, A.; Tarkka, M.; Hamdali, H. Growth promotion and protection against root rot of sugar beet (Beta vulgaris L.) by two rock phosphate and potassium solubilizing Streptomyces spp. under greenhouse conditions. Plant Soil 2022, 472, 407–420. [Google Scholar] [CrossRef]
- Getha, K.; Vikineswary, S.; Wong, W.H.; Seki, T.; Ward, A.; Goodfellow, M. Evaluation of Streptomyces sp. strain g10 for suppression of Fusarium wilt and rhizosphere colonization in pot-grown banana plantlets. J. Ind. Microbiol. Biotech. 2005, 32, 24–32. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Z.; Wang, Y.; Zhang, J.; Wan, S.; Huang, Y.; Yun, T.; Xie, J.; Wang, W. Biocontrol potential of endophytic Streptomyces malaysiensis 8ZJF-21 from medicinal plant against banana Fusarium wilt caused by Fusarium oxysporum f.sp. cubense tropical race 4. Front. Plant Sci. 2022, 13, 874819. [Google Scholar] [CrossRef]
- Yadav, A.K.; Yandigeri, M.S.; Vardhan, S.; Sivakumar, G.; Rangeshwaran, R.; Tripathi, C.P.M. Streptomyces sp. S160: A potential antagonist against chickpea charcoal root rot caused by Macrophomina phaseolina (Tassi) Goid. Ann. Microbiol. 2014, 64, 1113–1122. [Google Scholar] [CrossRef]
- Sadeghian, M.; Shahidi Bonjar, G.H.; Sharifi Sirchi, G.R. Post harvest biological control of apple bitter rot by soil-borne Actinomycetes and molecular identification of the active antagonist. Postharvest Biol. Technol. 2016, 112, 46–54. [Google Scholar] [CrossRef]
- Haggag, W.M.; Singer, S.M.; Aly, M.D.E.H. Application of broad-spectrum of marine Streptomyces albidoflavus as biofungicide and plant growth promoting of tomato diseases. Res. J. Pharm. Biol. Chem. Sci. 2014, 5, 142–148. [Google Scholar]
- Kim, H.; Lee, E.; Park, S.; Lee, H.-S.; Chung, N. Biological control of anthracnose (Colletotrichum gloeosporioides) in pepper and cherry tomato by Streptomyces sp. A1022. J. Agric. Sci. 2014, 6, 54. [Google Scholar] [CrossRef]
- Torabi, A.; Shahidi Bonjar, G.H.; Abdolshahi, R.; Pournamdari, M.; Saadoun, I.; Barka, E.A. Biological control of Paecilomyces formosus, the causal agent of dieback and canker diseases of pistachio by two strains of Streptomyces misionensis. Biol. Control 2019, 137, 104029. [Google Scholar] [CrossRef]
- Li, Q.; Jiang, Y.; Ning, P.; Zheng, L.; Huang, J.; Li, G.; Jiang, D.; Hsiang, T. Suppression of Magnaporthe oryzae by culture filtrates of Streptomyces globisporus JK-1. Biol. Control 2011, 58, 139–148. [Google Scholar] [CrossRef]
- Toumatia, O.; Compant, S.; Yekkour, A.; Yacine, G.; Sabaou, N.; Mathieu, F.; Sessitsch, A.; Zitouni, A. Biocontrol and plant growth promoting properties of Streptomyces mutabilis strain IA1 isolated from a Saharan soil on wheat seedlings and visualization of its niches of colonization. S. Afr. J. Bot. 2016, 105, 234–239. [Google Scholar] [CrossRef]
- Martínez-Hidalgo, P.; García, J.M.; Pozo, M.J. Induced systemic resistance against Botrytis cinerea by Micromonospora strains isolated from root nodules. Front. Microbiol. 2015, 6, 922. [Google Scholar] [CrossRef]
- Saeed, E.E.; Sham, A.; Salmin, Z.; Abdelmowla, Y.; Iratni, R.; El-Tarabily, K.; AbuQamar, S. Streptomyces globosus UAE1, a potential effective biocontrol agent for black scorch disease in date palm plantations. Front. Microbiol. 2017, 8, 1455. [Google Scholar] [CrossRef]
- Suárez-Moreno, Z.R.; Vinchira-Villarraga, D.M.; Vergara-Morales, D.I.; Castellanos, L.; Ramos, F.A.; Guarnaccia, C.; Degrassi, G.; Venturi, V.; Moreno-Sarmiento, N. Plant-growth promotion and biocontrol properties of three Streptomyces spp. isolates to control bacterial rice pathogens. Front. Microbiol. 2019, 10, 290. [Google Scholar] [CrossRef]
- Cao, P.; Li, C.; Wang, H.; Yu, Z.; Xu, X.; Wang, X.; Zhao, J.; Xiang, W. Community structures and antifungal activity of root-associated endophytic Actinobacteria in healthy and diseased cucumber plants and Streptomyces sp. HAAG3-15 as a promising biocontrol agent. Microorganisms 2020, 8, 236. [Google Scholar] [CrossRef]
- Ebrahimi-Zarandi, M.; Bonjar, G.H.; Riseh, R.S.; El-Shetehy, M.; Saadoun, I.; Barka, E.A. Exploring two Streptomyces species to control Rhizoctonia solani in tomato. Agronomy 2021, 11, 1384. [Google Scholar] [CrossRef]
- Wang, M.; Xue, J.; Ma, J.; Feng, X.; Ying, H.; Xu, H. Streptomyces lydicus M01 regulates soil microbial community and alleviates foliar disease caused by Alternaria alternata on cucumbers. Front. Microbiol. 2020, 11, 942. [Google Scholar] [CrossRef] [PubMed]
- Saberi Riseh, R.; Moradi Pour, M.; Ait Barka, E. A novel route for double-layered encapsulation of Streptomyces fulvissimus Uts22 by alginate and arabic gum for controlling of Pythium aphanidermatum in cucumber. Agronomy 2022, 12, 655. [Google Scholar] [CrossRef]
- Saberi-Riseh, R.; Moradi-Pour, M. A novel encapsulation of Streptomyces fulvissimus Uts22 by spray drying and its biocontrol efficiency against Gaeumannomyces graminis, the causal agent of take-all disease in wheat. Pest Manag. Sci. 2021, 77, 4357–4364. [Google Scholar] [CrossRef] [PubMed]
- Padilla-Gálvez, N.; Luengo-Uribe, P.; Mancilla, S.; Maurin, A.; Torres, C.; Ruiz, P.; France, A.; Bravo, I.; Urrutia, H. Antagonistic activity of endophytic actinobacteria from native potatoes (Solanum tuberosum subsp. tuberosum L.) against Pectobacterium carotovorum subsp. carotovorum and Pectobacterium atrosepticum. BMC Microbiol. 2021, 21, 335. [Google Scholar] [CrossRef]
- Sarwar, A.; Latif, Z.; Zhang, S.; Hao, J.; Bechthold, A. A potential biocontrol agent Streptomyces violaceusniger AC12AB for managing potato common scab. Front. Microbiol. 2019, 10, 202. [Google Scholar] [CrossRef]
- Le, K.D.; Yu, N.H.; Park, A.R.; Park, D.J.; Kim, C.J.; Kim, J.C. Streptomyces sp. AN090126 as a biocontrol agent against bacterial and fungal plant diseases. Microorganisms 2022, 10, 791. [Google Scholar] [CrossRef]
- Sabaratnam, S.; Traquair, J.A. Formulation of a Streptomyces biocontrol agent for the suppression of Rhizoctonia damping-off in tomato transplants. Biol. Control 2002, 23, 245–253. [Google Scholar] [CrossRef]
- Copping, L.G.; Duke, S.O. Natural products that have been used commercially as crop protection agents. Pest Manag. Sci. 2007, 63, 524–554. [Google Scholar] [CrossRef]
- Saxena, S.; Pandey, A.K. Microbial metabolites as eco-friendly agrochemicals for the next millennium. Appl. Microbiol. Biotechnol. 2001, 55, 395–403. [Google Scholar] [CrossRef]
- Kabaluk, J.T.; Svircev, A.M.; Goettel, M.S.; Woo, S.G. The Use and Regulation of Microbial Pesticides in Representative Jurisdiction Worldwide; IOBC Global: Hong Kong, China, 2010; p. 99. [Google Scholar]
- Aggarwal, N.; Thind, S.K.; Sharma, S. Role of Secondary Metabolites of Actinomycetes in Crop Protection. In Plant Growth Promoting Actinobacteria: A New Avenue for Enhancing the Productivity and Soil Fertility of Grain Legumes; Subramaniam, G., Arumugam, S., Rajendran, V., Eds.; Springer: Singapore, 2016; pp. 99–121. [Google Scholar]
- Bailey, K.L.; Falk, S.P. Turning research on microbial bioherbicides into commercial products a Phoma Story. Pest Technol. 2011, 5, 73–79. [Google Scholar]
- Raymaekers, K.; Ponet, L.; Holtappels, D.; Berckmans, B.; Cammue, B.P.A. Screening for novel biocontrol agents applicable in plant disease management—A review. Biol. Control 2020, 144, 104240. [Google Scholar] [CrossRef]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.-P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33. [Google Scholar] [CrossRef]
- Kloepper, J.W.; Ryu, C.M.; Zhang, S. Induced systemic resistance and promotion of plant growth by Bacillus spp. Phytopathology 2004, 94, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- De Vleesschauwer, D.; Höfte, M. Rhizobacteria-Induced Systemic Resistance. In Advances in Botanical Research; Academic Press: Cambridge, MA, USA, 2009; Volume 51, pp. 223–281. [Google Scholar]
- Ryu, C.M.; Farag, M.A.; Hu, C.H.; Reddy, M.S.; Kloepper, J.W.; Paré, P.W. Bacterial volatiles induce systemic resistance in Arabidopsis. Plant Physiol. 2004, 134, 1017–1026. [Google Scholar] [CrossRef]
- Meziane, H.; van der Sluis, I.; van Loon, L.C.; Höfte, M.; Bakker, P.A.H.M. Determinants of Pseudomonas putida WCS358 involved in inducing systemic resistance in plants. Mol. Plant Pathol. 2005, 6, 177–185. [Google Scholar] [CrossRef]
- van Loon, L.C.; Bakker, P.A.H.M.; van der Heijdt, W.H.W.; Wendehenne, D.; Pugin, A. Early responses of tobacco suspension cells to rhizobacterial elicitors of induced systemic resistance. Mol. Plant Microbe Interact. 2008, 21, 1609–1621. [Google Scholar] [CrossRef]
- Verhagen, B.W.; Trotel-Aziz, P.; Couderchet, M.; Höfte, M.; Aziz, A. Pseudomonas spp. induced systemic resistance to Botrytis cinerea is associated with induction and priming of defence responses in grapevine. J. Exp. Bot. 2010, 61, 249–260. [Google Scholar] [CrossRef]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Tjamos, S.E.; Flemetakis, E.; Paplomatas, E.J.; Katinakis, P. Induction of resistance to Verticillium dahliae in Arabidopsis thaliana by the biocontrol agent K-165 and pathogenesis-related proteins gene expression. Mol. Plant Microbe Interact. 2005, 18, 555–561. [Google Scholar] [CrossRef]
- van de Mortel, J.E.; de Vos, R.C.; Dekkers, E.; Pineda, A.; Guillod, L.; Bouwmeester, K.; van Loon, J.J.; Dicke, M.; Raaijmakers, J.M. Metabolic and transcriptomic changes induced in Arabidopsis by the rhizobacterium Pseudomonas fluorescens SS101. Plant Physiol. 2012, 160, 2173–2188. [Google Scholar] [CrossRef]
- Audenaert, K.; Pattery, T.; Cornelis, P.; Höfte, M. Induction of systemic resistance to Botrytis cinerea in tomato by Pseudomonas aeruginosa 7NSK2: Role of salicylic acid, pyochelin, and pyocyanin. Mol. Plant Microbe Interact. 2002, 15, 1147–1156. [Google Scholar] [CrossRef]
- Zhao, S.; Du, C.M.; Tian, C.Y. Suppression of Fusarium oxysporum and induced resistance of plants involved in the biocontrol of cucumber Fusarium wilt by Streptomyces bikiniensis HD-087. World J. Microbiol. Biotechnol. 2012, 28, 2919–2927. [Google Scholar] [CrossRef]
- Baz, M.; Tran, D.; Kettani-Halabi, M.; Samri, S.E.; Jamjari, A.; Biligui, B.; Meimoun, P.; El-Maarouf-Bouteau, H.; Garmier, M.; Saindrenan, P.; et al. Calcium- and ROS-mediated defence responses in BY2 tobacco cells by nonpathogenic Streptomyces sp. J. Appl. Microbiol. 2012, 112, 782–792. [Google Scholar] [CrossRef]
- Patil, H.J.; Srivastava, A.K.; Singh, D.P.; Chaudhari, B.L.; Arora, D.K. Actinomycetes mediated biochemical responses in tomato (Solanum lycopersicum) enhances bioprotection against Rhizoctonia solani. Crop Prot. 2011, 30, 1269–1273. [Google Scholar] [CrossRef]
- Singh, S.; Gupta, R.; Gaur, R.; Srivastava, A. Antagonistic actinomycetes mediated resistance in Solanum lycopersicon Mill. against Rhizoctonia solani Kühn. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2015, 87, 789–798. [Google Scholar] [CrossRef]
- Kurth, F.; Mailänder, S.; Bönn, M.; Feldhahn, L.; Herrmann, S.; Große, I.; Buscot, F.; Schrey, S.D.; Tarkka, M.T. Streptomyces-induced resistance against oak powdery mildew involves host plant responses in defense, photosynthesis, and secondary metabolism pathways. Mol. Plant Microbe Interact. 2014, 27, 891–900. [Google Scholar] [CrossRef]
- Singh, S.P.; Gaur, R. Endophytic Streptomyces spp. underscore induction of defense regulatory genes and confers resistance against Sclerotium rolfsii in chickpea. Biol. Control 2017, 104, 44–56. [Google Scholar] [CrossRef]
- Vatsa-Portugal, P.; Aziz, A.; Rondeau, M.; Villaume, S.; Morjani, H.; Clément, C.; Ait Barka, E. How Streptomyces anulatus primes grapevine defenses to cope with gray mold: A study of the early responses of cell suspensions. Front. Plant Sci. 2017, 8, 1043. [Google Scholar] [CrossRef]
- Awla, H.K.; Kadir, J.; Othman, R.; Rashid, T.S.; Hamid, S.; Wong, M.-Y. Plant growth-promoting abilities and biocontrol efficacy of Streptomyces sp. UPMRS4 against Pyricularia oryzae. Biol. Control 2017, 112, 55–63. [Google Scholar] [CrossRef]
- Zhang, Q.; Yong, D.; Zhang, Y.; Shi, X.; Li, B.; Li, G.; Liang, W.; Wang, C. Streptomyces rochei A-1 induces resistance and defense-related responses against Botryosphaeria dothidea in apple fruit during storage. Postharvest Biol. Technol. 2016, 115, 30–37. [Google Scholar] [CrossRef]
- Vilasinee, S.; Toanuna, C.; McGovern, R.; Nalumpang, S. Expression of pathogenesis-related (PR) genes in tomato against Fusarium wilt by challenge inoculation with Streptomyces NSP3. Int. J. Agric. Technol. 2019, 15, 157–170. [Google Scholar]
- Abbasi, S.; Safaie, N.; Sadeghi, A.; Shamsbakhsh, M. Streptomyces strains induce resistance to Fusarium oxysporum f.sp. lycopersici race 3 in tomato through different molecular mechanisms. Front. Microbiol. 2019, 10, 1505. [Google Scholar] [CrossRef]
- Mun, B.-G.; Lee, W.-H.; Kang, S.-M.; Lee, S.-U.; Lee, S.-M.; Lee, D.Y.; Shahid, M.; Yun, B.-W.; Lee, I.-J. Streptomyces sp. LH 4 promotes plant growth and resistance against Sclerotinia sclerotiorum in cucumber via modulation of enzymatic and defense pathways. Plant Soil 2020, 448, 87–103. [Google Scholar] [CrossRef]
- Saikia, K.; Bora, L.C. Exploring actinomycetes and endophytes of rice ecosystem for induction of disease resistance against bacterial blight of rice. Eur. J. Plant Pathol. 2021, 159, 67–79. [Google Scholar] [CrossRef]
- Lee, S.-M.; Kong, H.G.; Song, G.C.; Ryu, C.-M. Disruption of Firmicutes and Actinobacteria abundance in tomato rhizosphere causes the incidence of bacterial wilt disease. ISME J. 2021, 15, 330–347. [Google Scholar] [CrossRef]
- Abo-Zaid, G.A.; Matar, S.M.; Abdelkhalek, A. Induction of plant resistance against tobacco mosaic virus using the biocontrol agent Streptomyces cellulosae isolate Actino 48. Agronomy 2020, 10, 1620. [Google Scholar] [CrossRef]
- Vergnes, S.; Gayrard, D.; Veyssière, M.; Toulotte, J.; Martinez, Y.; Dumont, V.; Bouchez, O.; Rey, T.; Dumas, B. Phyllosphere colonization by a soil Streptomyces sp. promotes plant defense responses against fungal infection. Mol. Plant Microbe Interact. 2020, 33, 223–234. [Google Scholar] [CrossRef]
- Cha, J.-Y.; Han, S.; Hong, H.-J.; Cho, H.; Kim, D.; Kwon, Y.; Kwon, S.-K.; Crüsemann, M.; Bok Lee, Y.; Kim, J.F.; et al. Microbial and biochemical basis of a Fusarium wilt-suppressive soil. ISME J. 2016, 10, 119–129. [Google Scholar] [CrossRef]
- Schlatter, D.; Kinkel, L.; Thomashow, L.; Weller, D.; Paulitz, T. Disease suppressive soils: New insights from the soil microbiome. Phytopathology 2017, 107, 1284–1297. [Google Scholar] [CrossRef]
- Yuan, J.; Zhao, J.; Wen, T.; Zhao, M.; Li, R.; Goossens, P.; Huang, Q.; Bai, Y.; Vivanco, J.M.; Kowalchuk, G.A.; et al. Root exudates drive the soil-borne legacy of aboveground pathogen infection. Microbiome 2018, 6, 156. [Google Scholar] [CrossRef]
- Zhalnina, K.; Louie, K.B.; Hao, Z.; Mansoori, N.; da Rocha, U.N.; Shi, S.; Cho, H.; Karaoz, U.; Loqué, D.; Bowen, B.P.; et al. Dynamic root exudate chemistry and microbial substrate preferences drive patterns in rhizosphere microbial community assembly. Nat. Microbiol. 2018, 3, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Li, R.; Ren, Y.; Liu, C.; Zhao, Q.; Wu, H.; Jousset, A.; Shen, Q. Distinct roles for soil fungal and bacterial communities associated with the suppression of vanilla Fusarium wilt disease. Soil Biol. Biochem. 2017, 107, 198–207. [Google Scholar] [CrossRef]
- Zheng, Y.; Han, X.; Zhao, D.; Wei, K.; Yuan, Y.; Li, Y.; Liu, M.; Zhang, C.-S. Exploring biocontrol agents from microbial keystone taxa associated to suppressive soil: A new attempt for a biocontrol strategy. Front. Plant Sci. 2021, 12, 655673. [Google Scholar] [CrossRef]
- Tan, H.; Zhou, S.; Deng, Z.; He, M.; Cao, L. Ribosomal-sequence-directed selection for endophytic streptomycete strains antagonistic to Ralstonia solanacearum to control tomato bacterial wilt. Biol. Control 2011, 59, 245–254. [Google Scholar] [CrossRef]
- Gao, M.; Xiong, C.; Gao, C.; Tsui, C.K.M.; Wang, M.M.; Zhou, X.; Zhang, A.M.; Cai, L. Disease-induced changes in plant microbiome assembly and functional adaptation. Microbiome 2021, 9, 187. [Google Scholar] [CrossRef]
- Naylor, D.; DeGraaf, S.; Purdom, E.; Coleman-Derr, D. Drought and host selection influence bacterial community dynamics in the grass root microbiome. ISME J. 2017, 11, 2691–2704. [Google Scholar] [CrossRef]
- Hartman, K.; Tringe, S.G. Interactions between plants and soil shaping the root microbiome under abiotic stress. Biochem. J. 2019, 476, 2705–2724. [Google Scholar] [CrossRef]
- Xu, L.; Naylor, D.; Dong, Z.; Simmons, T.; Pierroz, G.; Hixson, K.K.; Kim, Y.-M.; Zink, E.M.; Engbrecht, K.M.; Wang, Y.; et al. Drought delays development of the sorghum root microbiome and enriches for monoderm bacteria. Proc. Natl. Acad. Sci. USA 2018, 115, E4284–E4293. [Google Scholar] [CrossRef]
- Fitzpatrick, C.; Copeland, J.; Wang, P.; Guttman, D.; Kotanen, P.; Johnson, M. Assembly and ecological function of the root microbiome across angiosperm plant species. Proc. Natl. Acad. Sci. USA 2018, 115, 201717617. [Google Scholar] [CrossRef]
- Yoolong, S.; Kruasuwan, W.; Thanh Phạm, H.T.; Jaemsaeng, R.; Jantasuriyarat, C.; Thamchaipenet, A. Modulation of salt tolerance in Thai jasmine rice (Oryza sativa L. cv. KDML105) by Streptomyces venezuelae ATCC 10712 expressing ACC deaminase. Sci. Rep. 2019, 9, 1275. [Google Scholar] [CrossRef]
- Gebauer, L.; Breitkreuz, C.; Heintz-Buschart, A.; Reitz, T.; Buscot, F.; Tarkka, M.; Bouffaud, M.-L. Water deficit history selects plant beneficial soil bacteria differently under conventional and organic farming. Front. Microbiol. 2022, 13, 824437. [Google Scholar] [CrossRef]
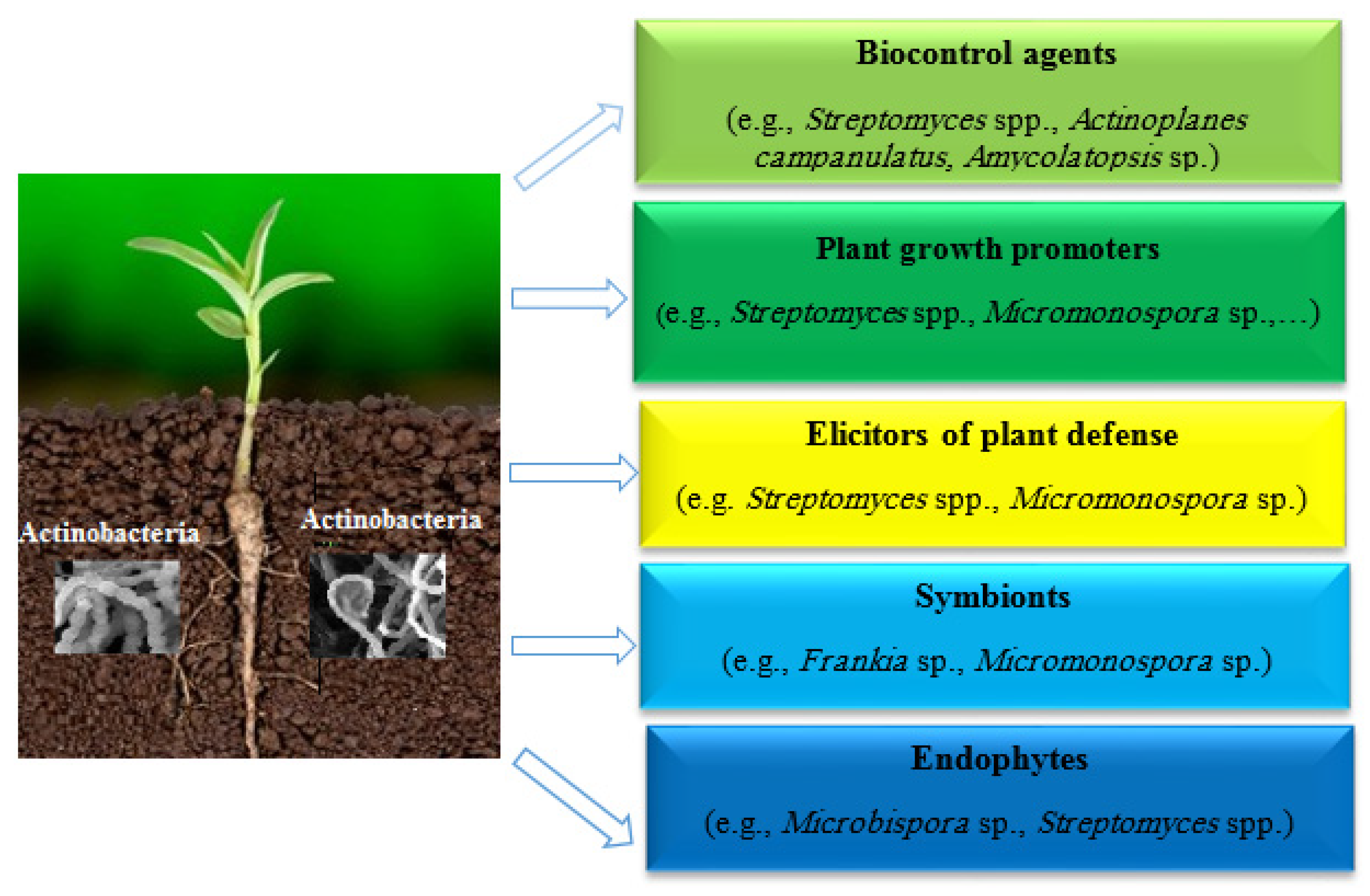
| Product Name | Organism | Targeted Pathogen/Disease |
|---|---|---|
| Mycostop, Verdera Oy, Finland | S. griseoviridis K61 | Damping off caused by Alternaria and R. solani and Fusarium, Phytophthora, and Pythium wilt and root diseases |
| Actinovate, Novozymes BioAg Inc., USA | S. lydicus WYEC 108 | Soilborne pathogens, viz. Pythium, Fusarium, Phytophthora, Rhizoctonia, and Verticillium; foliar diseases such as powdery and downy mildew, Botrytis, Alternaria, Postia, Geotrichum, and Sclerotinia |
| Mykocide KIBC Co., Ltd. South Korea | S. colombiensis | Powdery mildews, grey mold, and brown patch |
| Safegrow KIBC Co., Ltd. South Korea | S. kasugaensis | Sheath blight and large patch |
| Bactophil | S. albus | Seed germination diseases |
| Blasticidin-S BLA-S | S. griseochromogenes | Pyricularia oryzae |
| Kasugamycin Kasumin, Kasurab | S. kasugaensis | Leaf spot in sugar beet and celery (Cercospora spp.), scab in pears and apples (Venturia spp.), and soybean root rot (Phytophthora sojae) |
| Streptomycin Agrimycin, Paushak, Cuprimicin 17, AAstrepto 17, AS-50, Dustret, Cuprimic 100 and 500 | S. griseus | Bacterial rots, canker, and other bacterial diseases; Xanthomonas oryzae, Xanthomonas citri, and Pseudomonas tabaci of pome fruit, stone fruit, citrus, olives, vegetables, potatoes, tobacco, cotton, and ornamentals |
| Phytomycin Mycoshield, Cuprimic 100 and 500, Mycoject | S. rimosus | Fire blight (Erwinia amylovora) and diseases caused by Pseudomonas sp., Xanthomonas sp. and mycoplasma-like organisms |
| Validamycin Validacin, Valimun, Dantotsupadanvalida, Mycin Hustler, Valida, Sheathmar | S. hygroscopicus | R. solani and other Rhizoctonia in rice, potatoes, vegetables, strawberries, tobacco, ginger, cotton, sugar beet, etc. |
| Polyoxorim Endorse, PolyoxinZ, Stopit, Polyoxin AL and Z, Polybelin | S. cacaoi var. asoensis | Plant-pathogenic fungi, Sphaerotheca spp. and other powdery mildews; Botrytis cinerea, Sclerotinia sclerotiorum, Corynespora melonis, Cochliobolus miyabeanus, Alternaria alternata and other species in vines, apples, pears, vegetables, and ornamentals; rice sheath blight (R. solani), apple, pear canker, and Helminthosporium in rice |
| Natamycin Delvolan | S. natalensis and S. chattanoogensis | Basal rots on daffodils and ornamentals caused by Fusarium oxysporum |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ebrahimi-Zarandi, M.; Saberi Riseh, R.; Tarkka, M.T. Actinobacteria as Effective Biocontrol Agents against Plant Pathogens, an Overview on Their Role in Eliciting Plant Defense. Microorganisms 2022, 10, 1739. https://doi.org/10.3390/microorganisms10091739
Ebrahimi-Zarandi M, Saberi Riseh R, Tarkka MT. Actinobacteria as Effective Biocontrol Agents against Plant Pathogens, an Overview on Their Role in Eliciting Plant Defense. Microorganisms. 2022; 10(9):1739. https://doi.org/10.3390/microorganisms10091739
Chicago/Turabian StyleEbrahimi-Zarandi, Marzieh, Roohallah Saberi Riseh, and Mika T. Tarkka. 2022. "Actinobacteria as Effective Biocontrol Agents against Plant Pathogens, an Overview on Their Role in Eliciting Plant Defense" Microorganisms 10, no. 9: 1739. https://doi.org/10.3390/microorganisms10091739
APA StyleEbrahimi-Zarandi, M., Saberi Riseh, R., & Tarkka, M. T. (2022). Actinobacteria as Effective Biocontrol Agents against Plant Pathogens, an Overview on Their Role in Eliciting Plant Defense. Microorganisms, 10(9), 1739. https://doi.org/10.3390/microorganisms10091739





