Applications of Probiotic-Based Multi-Components to Human, Animal and Ecosystem Health: Concepts, Methodologies, and Action Mechanisms
Abstract
1. Introduction
2. Probiotic-Based Multi-Components
2.1. Probiotics and Synbiotics
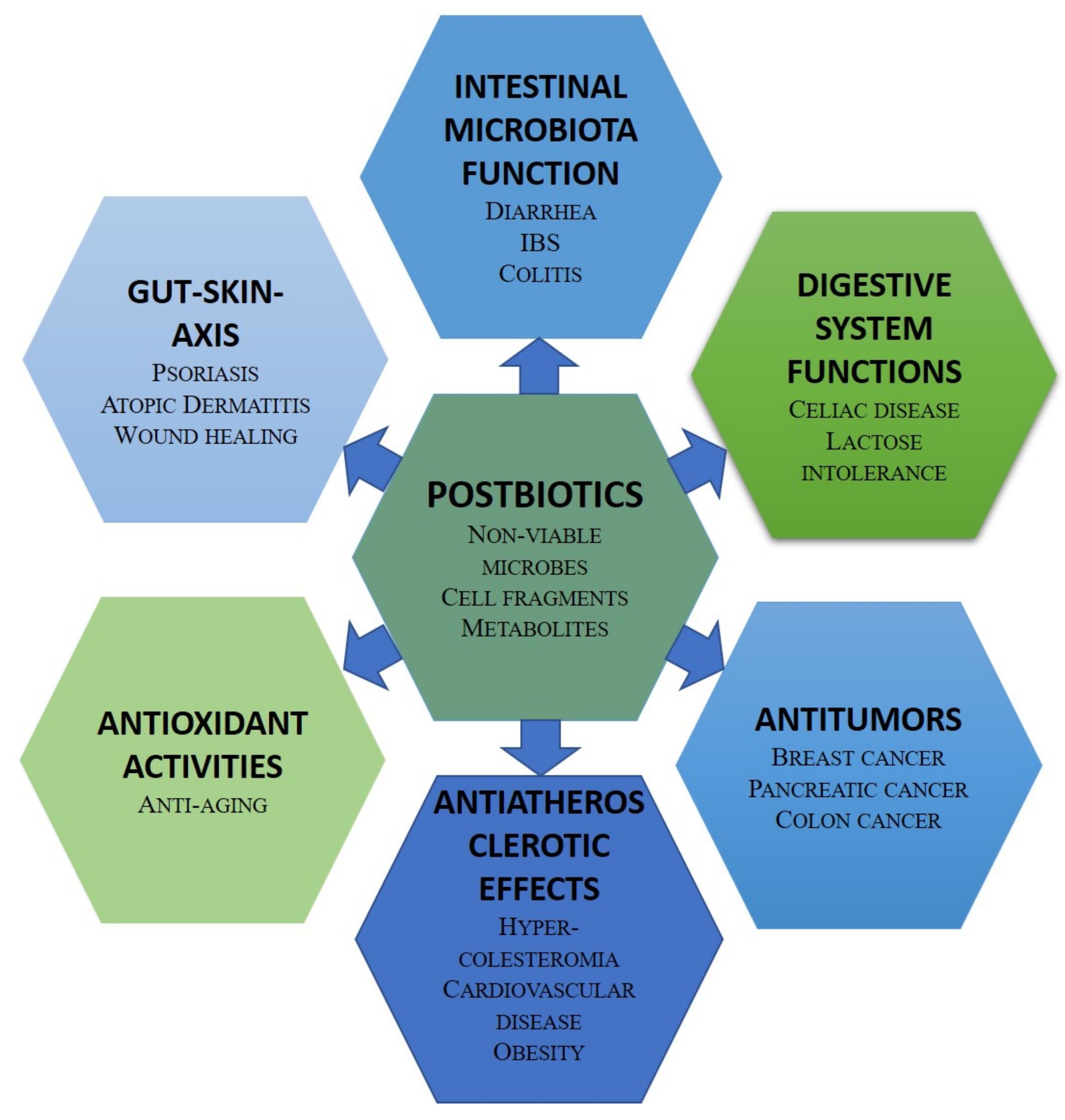
2.2. Postbiotics
3. Innovative Profiling and Monitoring Methodologies
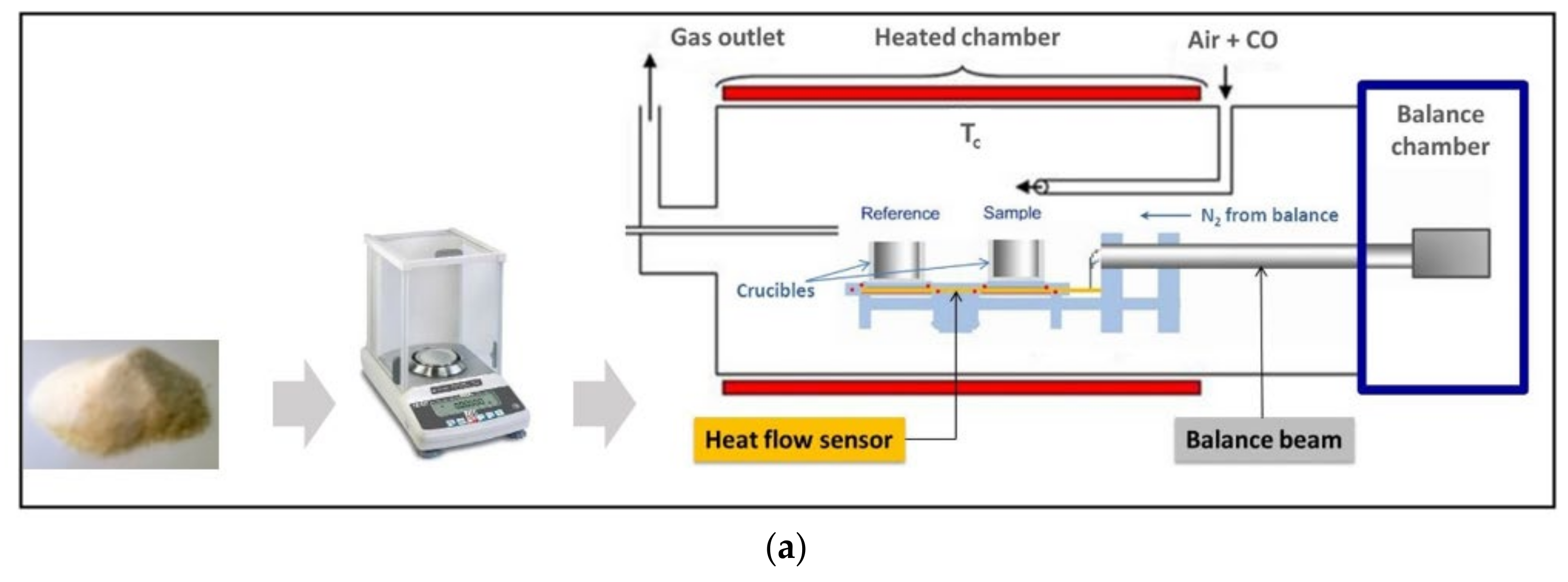
3.1. Thermophysical Profiling
3.1.1. Principle
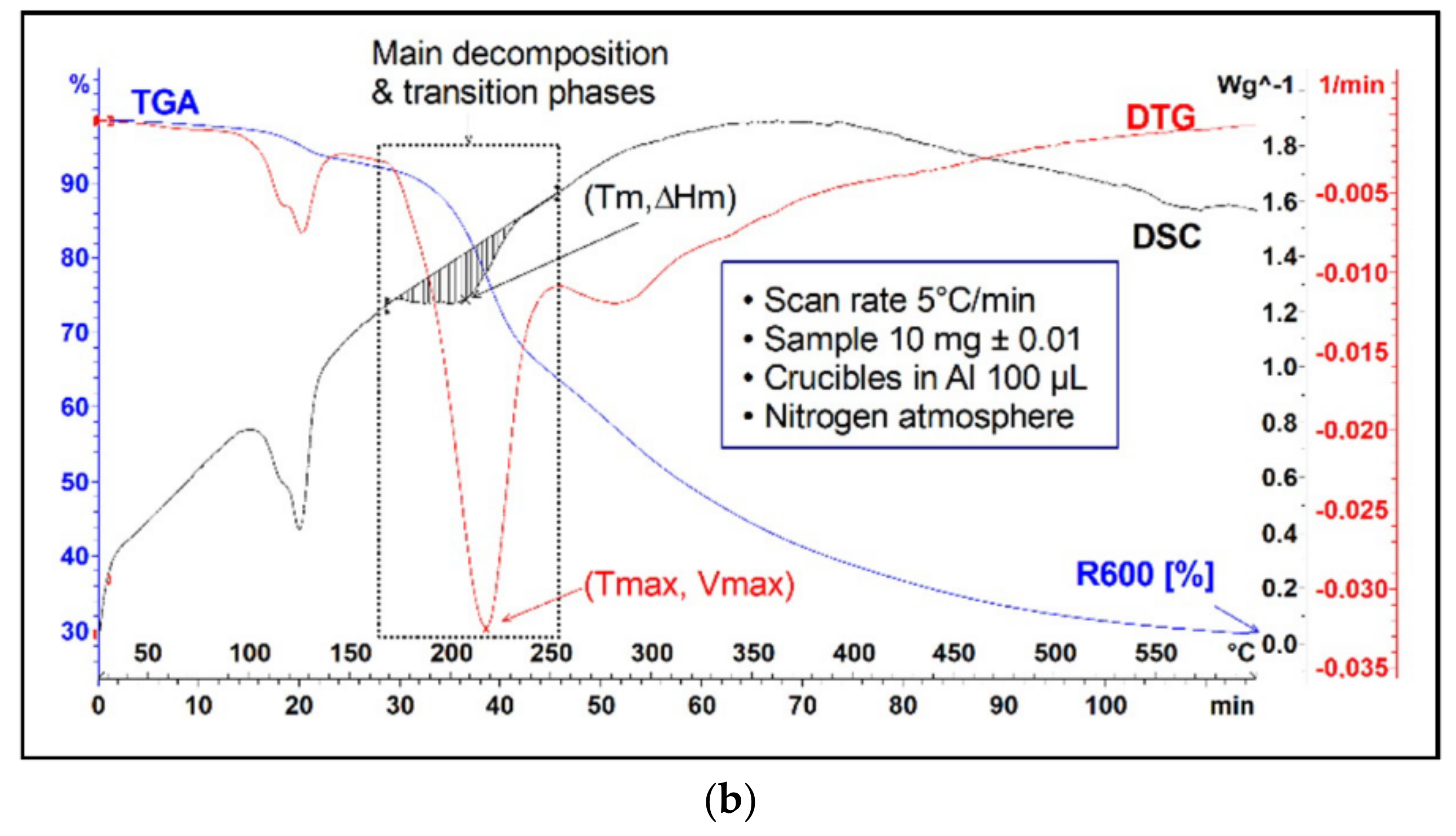
3.1.2. Practice of TGA-DSC Coupling Method
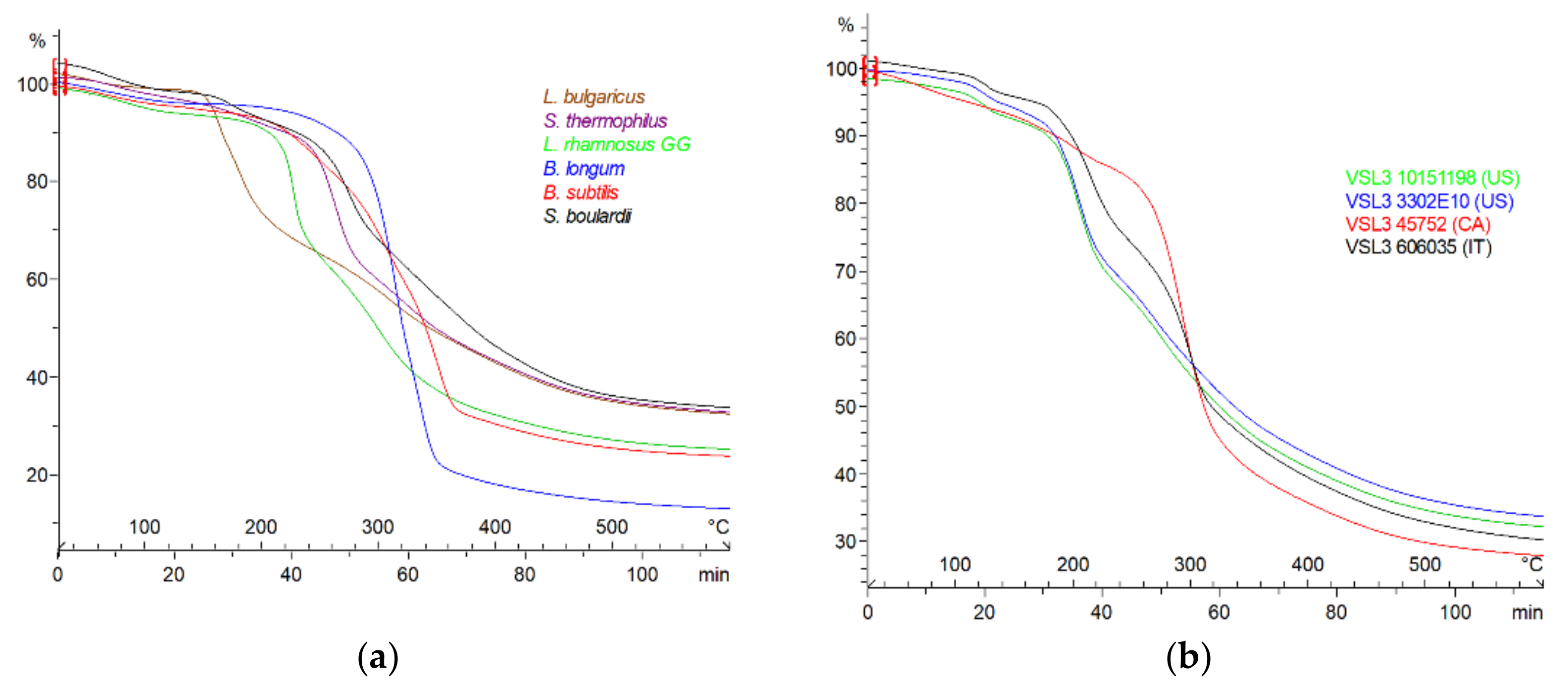
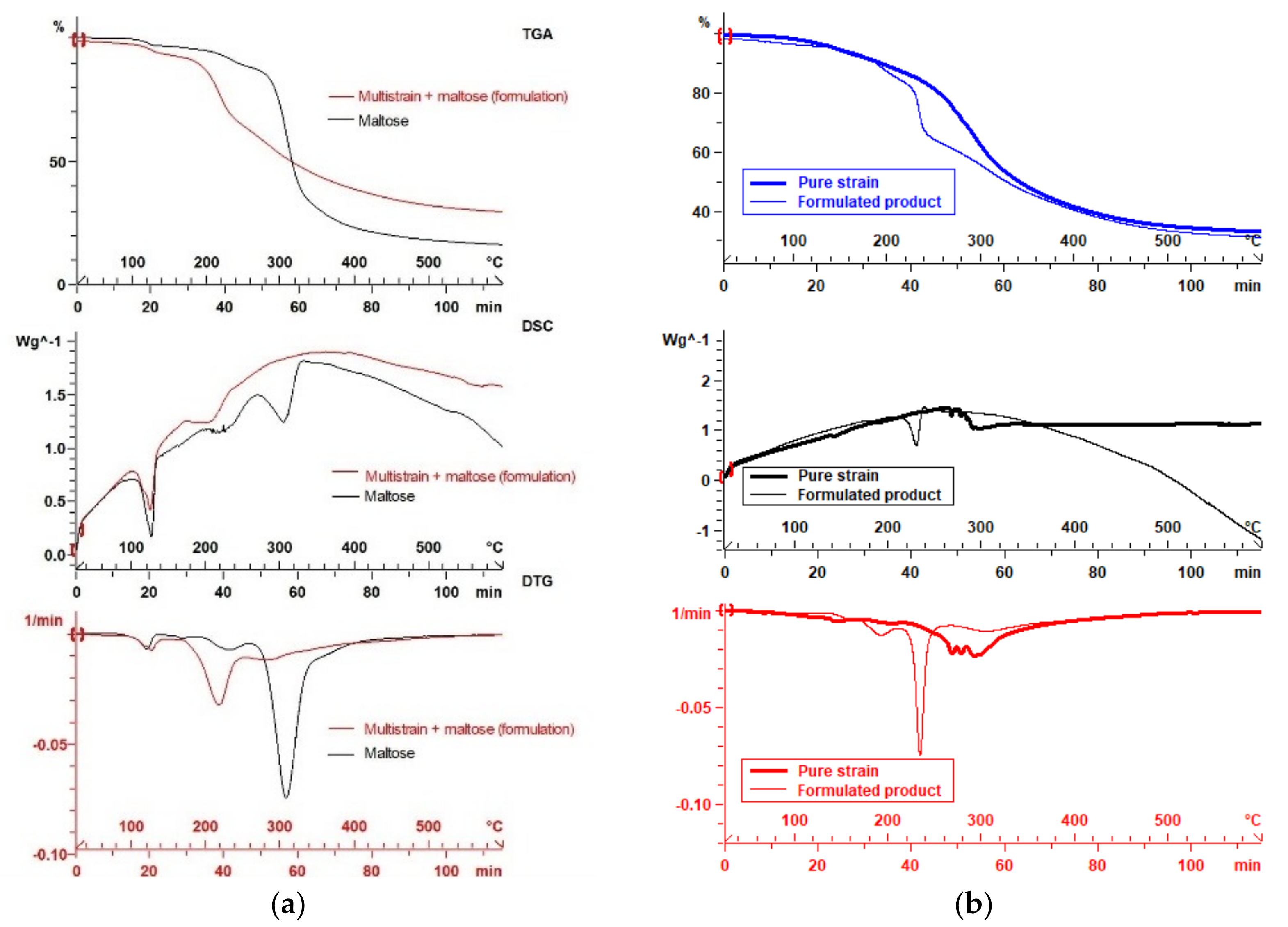
3.1.3. Examples of Mono- and Multi-Strain Thermophysical Profiling
3.1.4. Advantages of the TGA-DSC Approach
3.2. Advanced Testing for Genetic Composition (ATGC)
3.2.1. Origins of ATGC
3.2.2. Principles of ATGC
3.2.3. Probiotic Quality Control Application
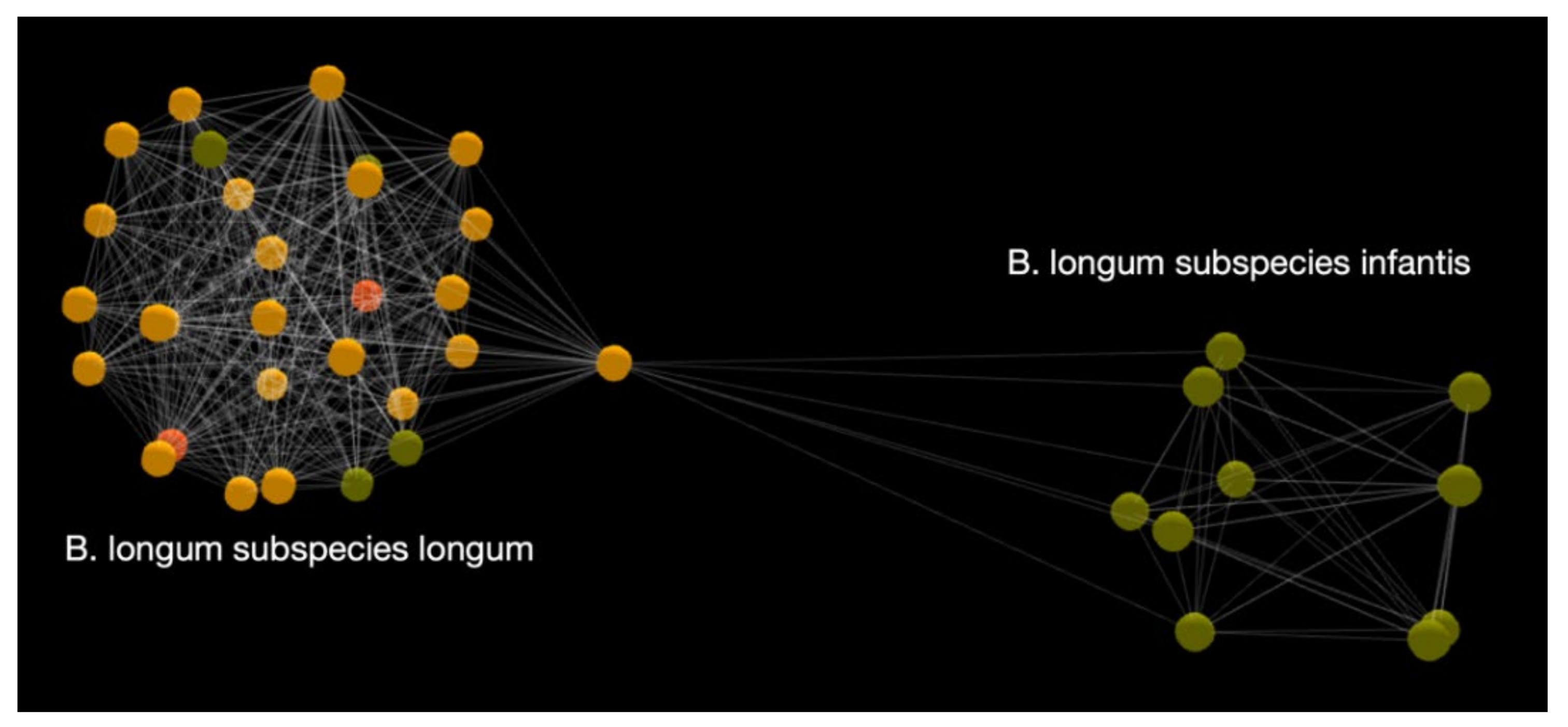
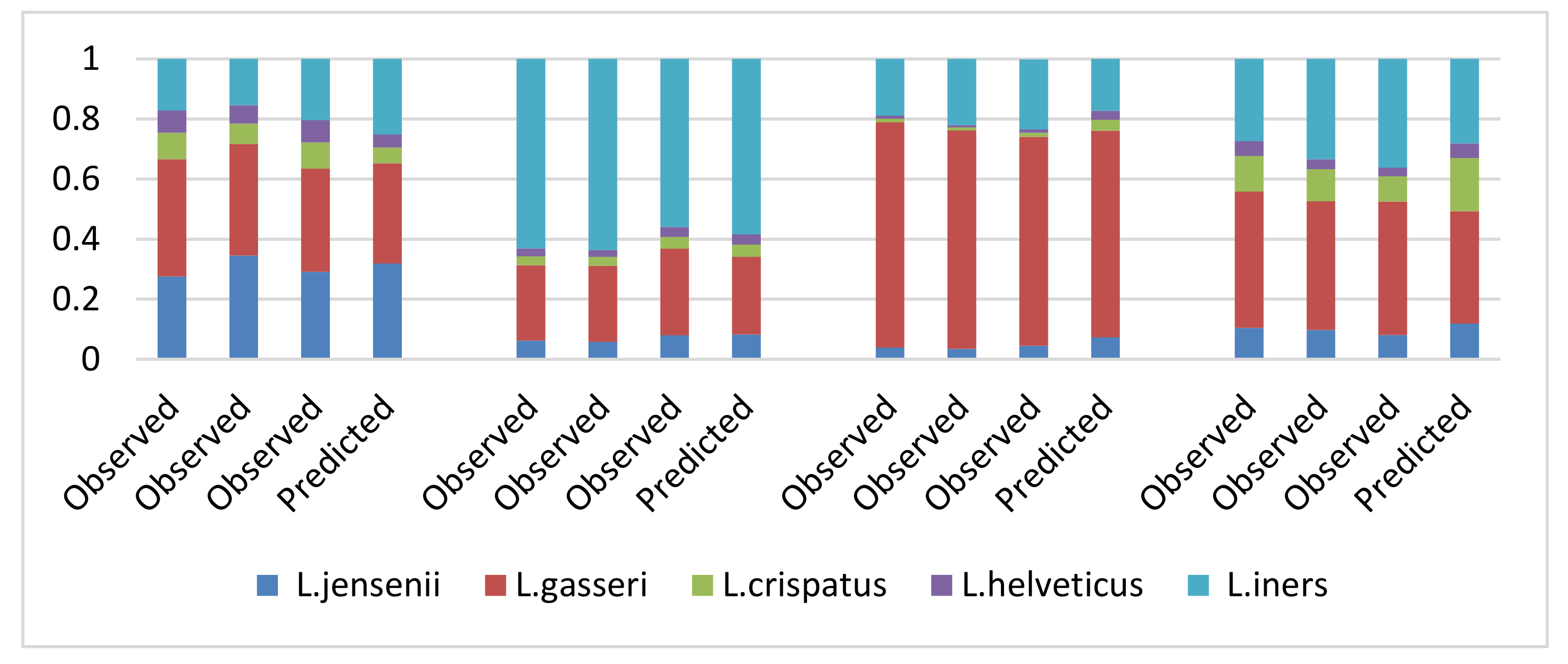
3.2.4. Human Microbiome Application
3.2.5. Soil Microbiome Application
3.2.6. Limitations, Challenges, and Future Developments
- Lactobacillus and Bifidobacterium
- Spore-forming (e.g., Bacillus)
- Gram-negative (e.g., Hafnia alvei, E. coli Nissle)
4. Applications and Action Mechanisms
4.1. Human Health
4.2. Animal Health
4.2.1. Poultry
4.2.2. Pigs and Piglets
4.2.3. Ruminants
4.2.4. Fish
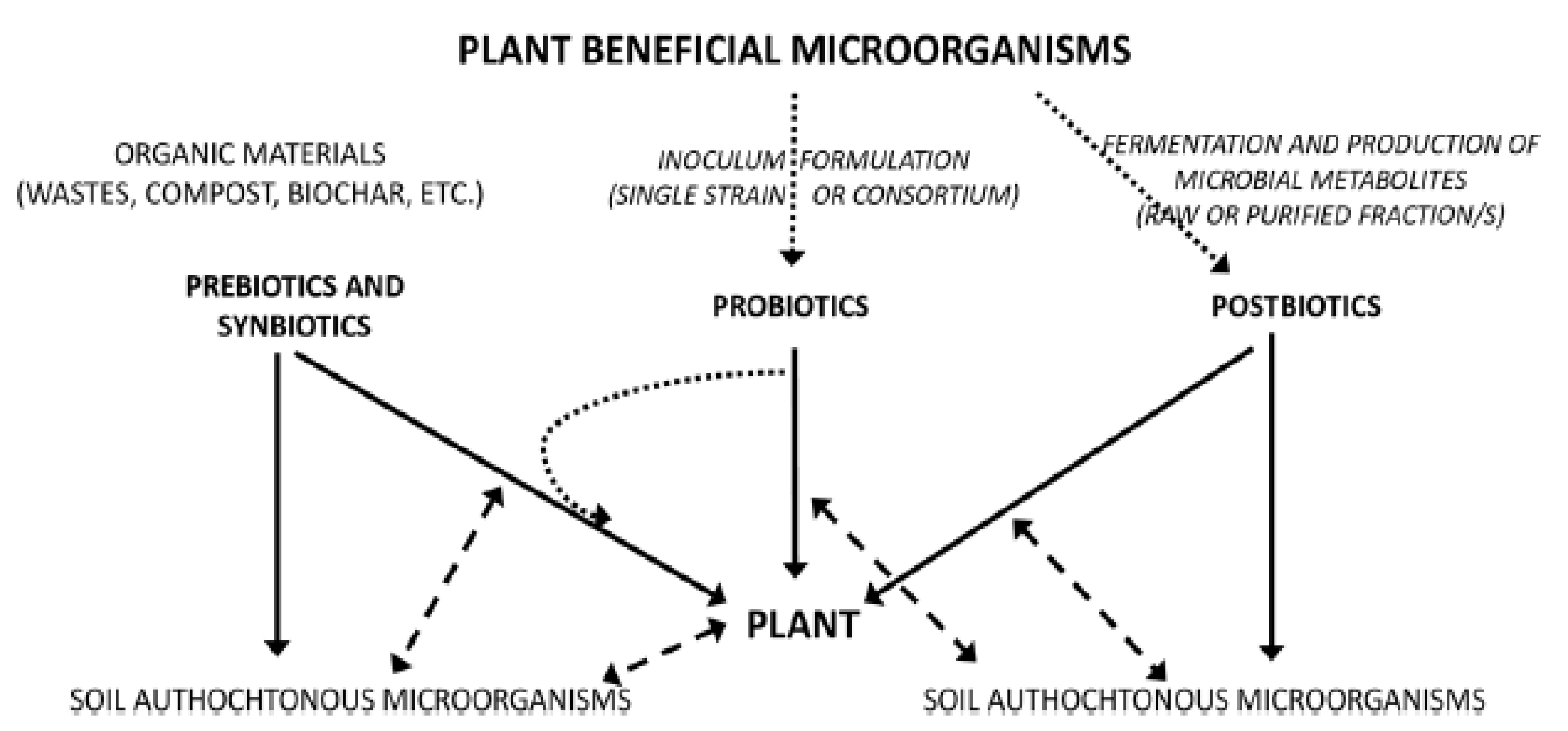
4.3. Plant and Soil Health
4.4. Environmental Health
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fuller, R. Probiotics in Man and Animals. J. Appl. Bacteriol. 1989, 66, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Ibrahim, S.; Hayek, S. Recent Application of Probiotics in Food and Agricultural Science. In Probiotics; Rigobelo, E., Ed.; InTech: London, UK, 2012; ISBN 978-953-51-0776-7. [Google Scholar]
- Zoghi, A.; Khosravi-Darani, K.; Sohrabvandi, S. Surface Binding of Toxins and Heavy Metals by Probiotics. Mini Rev. Med. Chem. 2014, 14, 84–98. [Google Scholar] [CrossRef] [PubMed]
- Ebenso, B.; Otu, A.; Alessandro, G.; Philipe, C.; Victor, A.; Razafindralambo, H. Nature-Based One Health Approaches to Urban Agriculture Can Deliver Food and Nutrition Security. Front. Nutr. 2022, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.-K.; Paik, H.-D. Prophylactic Effects of Probiotics on Respiratory Viruses Including COVID-19: A Review. Food Sci. Biotechnol. 2021, 30, 773–781. [Google Scholar] [CrossRef]
- Razafindralambo, H. Trends in Probiotic Applications; Studium Press LLC: Houston, TX, USA, 2018; ISBN 1-62699-110-3. [Google Scholar]
- Singh, K.; Rao, A. Probiotics: A Potential Immunomodulator in COVID-19 Infection Management. Nutr. Res. 2021, 87, 1–12. [Google Scholar] [CrossRef]
- Soleymani, S.; Ebrahimi, F.; Rezaeizadeh, H.; Rahimi, R. Probiotics and Cancer. In Nutraceuticals and Cancer Signaling: Clinical Aspects and Mode of Action; Jafari, S.M., Nabavi, S.M., Silva, A.S., Eds.; Food Bioactive Ingredients; Springer International Publishing: Cham, Switzerland, 2021; pp. 467–527. ISBN 978-3-030-74035-1. [Google Scholar]
- Goyal, P.; Belapurkar, P.; Kar, A. A Review on in Vitro and in Vivo Bioremediation Potential of Environmental and Probiotic Species of Bacillus and Other Probiotic Microorganisms for Two Heavy Metals, Cadmium and Nickel. Biosci. Biotechnol. Res. Asia 2019, 16, 01–13. [Google Scholar] [CrossRef]
- Helmy, Q.; Kardena, E.; Gustiani, S. Probiotics and Bioremediation. In Microorganisms; IntechOpen: London, UK, 2019. [Google Scholar]
- Yadav, M.K.; Kumari, I.; Singh, B.; Sharma, K.K.; Tiwari, S.K. Probiotics, Prebiotics and Synbiotics: Safe Options for next-Generation Therapeutics. Appl. Microbiol. Biotechnol. 2022, 106, 505–521. [Google Scholar] [CrossRef]
- Cavattoni, F. Probiotic Products: Technological and Regulatory Aspects. In Trends in Probiotic Applications; Hary Razafindralambo: Houxton, TX, USA, 2018; pp. 3–12. [Google Scholar]
- Cottet, C.; Ramírez Tapias, Y.A.; Delgado, J.F.; de la Osa, O.; Salvay, A.G.; Peltzer, M.A. Biobased Materials from Microbial Biomass and Its Derivatives. Materials 2020, 13, 1263. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S. Expert Consensus Document: The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Rabetafika, H.-N.; Razafindralambo, A.; Razafindralambo, H. Probiotics from Food and Non-Food Sources. Trends Probiotic Appl. 2018, 53, 33–49. [Google Scholar]
- Oelschlaeger, T.A. Mechanisms of Probiotic Actions—A Review. Int. J. Med. Microbiol. 2010, 300, 57–62. [Google Scholar] [CrossRef]
- Zambori, C.; Cumpănăşoiu, C.; Moţ, D.; Huţu, I.; Gurban, C.; Tîrziu, E. The Antimicrobial Role of Probiotics in the Oral Cavity in Humans and Dogs. Anim. Sci. Biotechnol. 2014, 47, 126–130. [Google Scholar]
- Muller, J.A.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Manufacture of Probiotic Bacteria. In Prebiotics and Probiotics Science and Technology; Springer: New York, NY, USA, 2009; Volume 1, pp. 725–759. [Google Scholar]
- Baral, K.C.; Bajracharya, R.; Lee, S.H.; Han, H.-K. Advancements in the Pharmaceutical Applications of Probiotics: Dosage Forms and Formulation Technology. Int. J. Nanomed. 2021, 16, 7535. [Google Scholar] [CrossRef]
- Huang, S.; Vignolles, M.-L.; Chen, X.D.; Le Loir, Y.; Jan, G.; Schuck, P.; Jeantet, R. Spray Drying of Probiotics and Other Food-Grade Bacteria: A Review. Trends Food Sci. Technol. 2017, 63, 1–17. [Google Scholar] [CrossRef]
- Su, Y.; Zheng, X.; Zhao, Q.; Fu, N.; Xiong, H.; Wu, W.D.; Chen, X.D. Spray Drying of Lactobacillus Rhamnosus GG with Calcium-Containing Protectant for Enhanced Viability. Powder Technol. 2019, 358, 87–94. [Google Scholar] [CrossRef]
- Charalampopoulos, D.; Rastall, R.A. Prebiotics and Probiotics Science and Technology; Springer: New York, NY, USA, 2009; Volume 1. [Google Scholar]
- Burgain, J.; Gaiani, C.; Linder, M.; Scher, J. Encapsulation of Probiotic Living Cells: From Laboratory Scale to Industrial Applications. J. Food Eng. 2011, 104, 467–483. [Google Scholar] [CrossRef]
- Centurion, F.; Basit, A.W.; Liu, J.; Gaisford, S.; Rahim, M.A.; Kalantar-Zadeh, K. Nanoencapsulation for Probiotic Delivery. ACS Nano 2021, 15, 18653–18660. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D. The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Vinderola, G.; Sanders, M.E.; Salminen, S. The Concept of Postbiotics. Foods 2022, 11, 1077. [Google Scholar] [CrossRef]
- Elko, N.; Foster, D.; Kleinheinz, G.; Raubenheimer, B.; Brander, S.; Kinzelman, J.; Kritzer, J.P.; Munroe, D.; Storlazzi, C.; Sutula, M.; et al. Human and Ecosystem Health in Coastal Systems. Shore Beach 2022, 90, 28. [Google Scholar]
- Vignieri, S.; Fahrenkamp-Uppenbrink, J. Ecosystem Earth. Science 2017, 356, 258–259. [Google Scholar] [CrossRef][Green Version]
- Razafindralambo, H. Advances in Physical Chemistry Tools for Probiotic Characterization. In Trends Probiotic Applications; Razafindralambo, H., Ed.; Studium Press LLC: Houston, TX, USA, 2018; pp. 50–81. [Google Scholar]
- Trush, E.A.; Poluektova, E.A.; Beniashvilli, A.G.; Shifrin, O.S.; Poluektov, Y.M.; Ivashkin, V.T. The Evolution of Human Probiotics: Challenges and Prospects. Probiotics Antimicrob. Proteins 2020, 12, 1291–1299. [Google Scholar] [CrossRef]
- Niki, E. Free Radicals in the 1900’s: From in Vitro to in Vivo. Free Radic. Res. 2000, 33, 693–704. [Google Scholar] [CrossRef]
- Barbosa, J.; Borges, S.; Teixeira, P. Influence of Sub-Lethal Stresses on the Survival of Lactic Acid Bacteria after Spray-Drying in Orange Juice. Food Microbiol. 2015, 52, 77–83. [Google Scholar] [CrossRef]
- Iaconelli, C.; Lemetais, G.; Kechaou, N.; Chain, F.; Bermúdez-Humarán, L.G.; Langella, P.; Gervais, P.; Beney, L. Drying Process Strongly Affects Probiotics Viability and Functionalities. J. Biotechnol. 2015, 214, 17–26. [Google Scholar] [CrossRef]
- Rajam, R.; Subramanian, P. Encapsulation of Probiotics: Past, Present and Future. Beni-Suef Univ. J. Basic Appl. Sci. 2022, 11, 46. [Google Scholar] [CrossRef]
- Grumet, L.; Tromp, Y.; Stiegelbauer, V. The Development of High-Quality Multispecies Probiotic Formulations: From Bench to Market. Nutrients 2020, 12, 2453. [Google Scholar] [CrossRef]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M. The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef]
- Cheng, C.; Yu, X.; Geng, F.; Wang, L.; Yang, J.; Huang, F.; Deng, Q. Review on the Regulation of Plant Polyphenols on the Stability of Polyunsaturated-Fatty-Acid-Enriched Emulsions: Partitioning Kinetic and Interfacial Engineering. J. Agric. Food Chem. 2022, 70, 3569–3584. [Google Scholar] [CrossRef]
- Bindels, L.B.; Delzenne, N.M.; Cani, P.D.; Walter, J. Towards a More Comprehensive Concept for Prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 303–310. [Google Scholar] [CrossRef]
- Malik, J.K.; Prakash, A.; Srivastava, A.K.; Gupta, R.C. Synbiotics in Animal Health and Production. In Nutraceuticals in Veterinary Medicine; Springer: Cham, Switzerland, 2019; pp. 287–301. [Google Scholar]
- Tabacchioni, S.; Passato, S.; Ambrosino, P.; Huang, L.; Caldara, M.; Cantale, C.; Hett, J.; Del Fiore, A.; Fiore, A.; Schlüter, A. Identification of Beneficial Microbial Consortia and Bioactive Compounds with Potential as Plant Biostimulants for a Sustainable Agriculture. Microorganisms 2021, 9, 426. [Google Scholar] [CrossRef] [PubMed]
- Tsilingiri, K.; Rescigno, M. Postbiotics: What Else? Benef. Microbes 2013, 4, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Żółkiewicz, J.; Marzec, A.; Ruszczyński, M.; Feleszko, W. Postbiotics—A Step beyond Pre-and Probiotics. Nutrients 2020, 12, 2189. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667. [Google Scholar] [CrossRef]
- Rad, A.H.; Hosseini, S.; Pourjafar, H. Postbiotics as Dynamic Biological Molecules for Antimicrobial Activity: A Mini-Review. Biointerface Res. Appl. Chem. 2022, 12, 6543–6556. [Google Scholar]
- Abbasi, A.; Sheykhsaran, E.; Kafil, H.S. Postbiotics: Science, Technology and Applications; Bentham Science Publishers: Sharjah, United Arab Emirates, 2021; ISBN 1-68108-839-8. [Google Scholar]
- Aghebati-Maleki, L.; Hasannezhad, P.; Abbasi, A.; Khani, N. Antibacterial, Antiviral, Antioxidant, and Anticancer Activities of Postbiotics: A Review of Mechanisms and Therapeutic Perspectives. Biointerface Res. Appl. Chem. 2021, 12, 2629–2645. [Google Scholar]
- Wegh, C.A.; Geerlings, S.Y.; Knol, J.; Roeselers, G.; Belzer, C. Postbiotics and Their Potential Applications in Early Life Nutrition and Beyond. Int. J. Mol. Sci. 2019, 20, 4673. [Google Scholar] [CrossRef]
- Asachi, M.; Nourafkan, E.; Hassanpour, A. A Review of Current Techniques for the Evaluation of Powder Mixing. Adv. Powder Technol. 2018, 29, 1525–1549. [Google Scholar] [CrossRef]
- Loskyll, J.; Maier, W.F.; Stoewe, K. Application of a Simultaneous TGA-DSC Thermal Analysis System for High-Throughput Screening of Catalytic Activity. ACS Comb. Sci. 2012, 14, 600–604. [Google Scholar] [CrossRef]
- Yu, L. Amorphous Pharmaceutical Solids: Preparation, Characterization and Stabilization. Adv. Drug Deliv. Rev. 2001, 48, 27–42. [Google Scholar] [CrossRef]
- Razafindralambo, H.; Razafindralambo, A.; Blecker, C. Thermophysical Fingerprinting of Probiotic-Based Products. Sci. Rep. 2019, 9, 1–8. [Google Scholar]
- Razafindralambo, H.; Correani, V.; Fiorucci, S.; Mattei, B. Variability in Probiotic Formulations Revealed by Proteomics and Physico-Chemistry Approach in Relation to the Gut Permeability. Probiotics Antimicrob. Proteins 2020, 12, 1193–1202. [Google Scholar] [CrossRef]
- Razafindralambo, H. Application of a Rapid Micro-Calorimetry Coupling Method in Quality and Integrity Control of Probiotic-Based Products. In New Visions in Science and Technology; Book Publisher International: West Bengal, India, 2021; Volume 2, pp. 6–11. ISBN 978-93-91882-98-3. [Google Scholar]
- Higgins, D.; Dworkin, J. Recent Progress in Bacillus Subtilis Sporulation. FEMS Microbiol. Rev. 2012, 36, 131–148. [Google Scholar] [CrossRef]
- Zhao, F.; Jin, H.; Shen, X.; Li, Q.; Liu, X.; Zhang, L.; Sun, Z.; Yu, J. Effect of the Administration of Probiotics on the Fecal Microbiota of Adult Individuals. Food Sci. Nutr. 2021, 9, 6471–6479. [Google Scholar] [CrossRef]
- Khrapko, K.; Hanekamp, J.S.; Thilly, W.G.; Belenkii, A.; Foret, F.; Karger, B.L. Constant Denaturant Capillary Electrophoresis (CDCE): A High Resolution Approach to Mutational Anaylsis. Nucleic Acids Res. 1994, 22, 364–369. [Google Scholar] [CrossRef]
- Khrapko, K.; Coller, H.A.; André, P.C.; Li, X.-C.; Hanekamp, J.S.; Thilly, W.G. Mitochondrial Mutational Spectra in Human Cells and Tissues. Proc. Natl. Acad. Sci. USA 1997, 94, 13798–13803. [Google Scholar] [CrossRef]
- Andre, P.; Kim, A.; Khrapko, K.; Thilly, W.G. Fidelity and Mutational Spectrum of Pfu DNA Polymerase on a Human Mitochondrial DNA Sequence. Genome Res. 1997, 7, 843–852. [Google Scholar] [CrossRef][Green Version]
- Sudo, H.; Li-Sucholeiki, X.-C.; Marcelino, L.A.; Gruhl, A.N.; Herrero-Jimenez, P.; Zarbl, H.; Willey, J.C.; Furth, E.E.; Morgenthaler, S.; Coller, H.A. Fetal–Juvenile Origins of Point Mutations in the Adult Human Tracheal–Bronchial Epithelium: Absence of Detectable Effects of Age, Gender or Smoking Status. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 2008, 646, 25–40. [Google Scholar] [CrossRef]
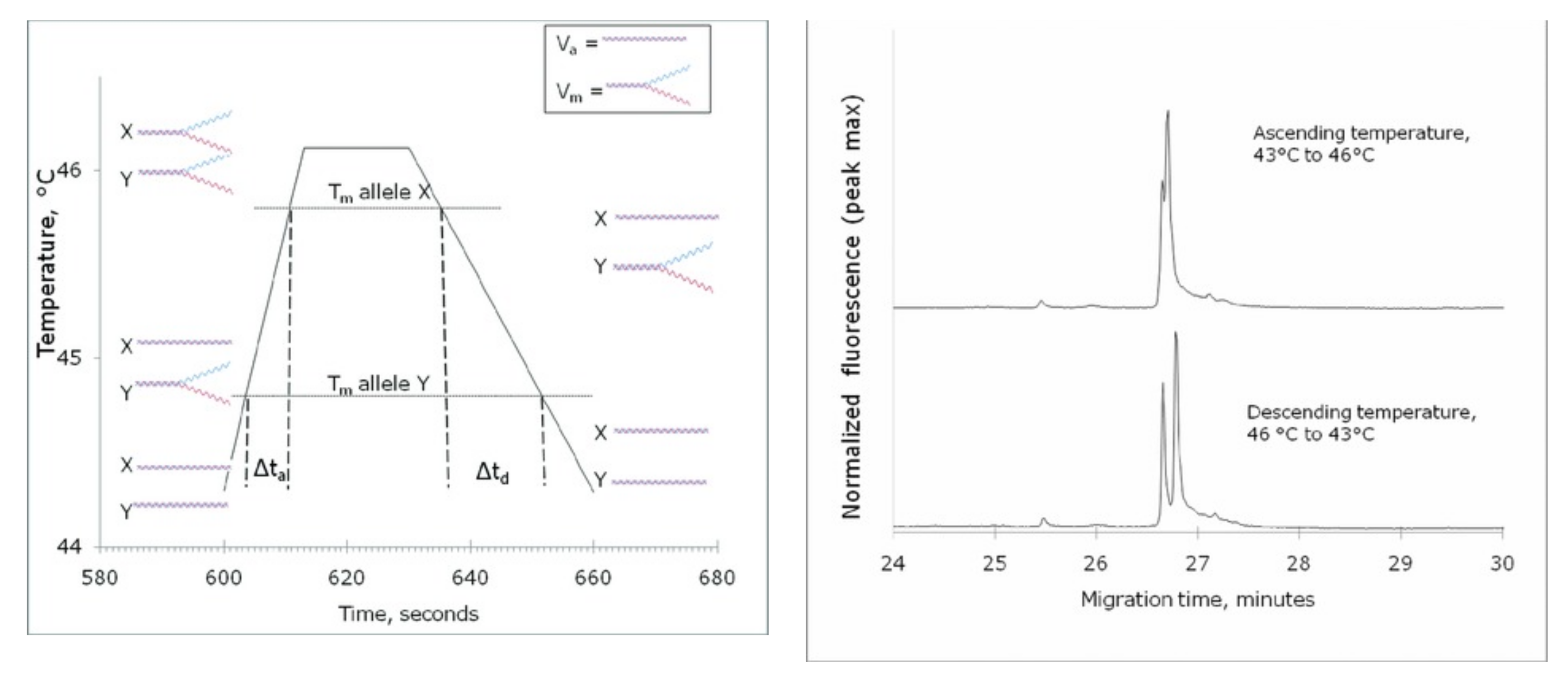
- Ekstrøm, P.O.; Warren, D.J.; Thilly, W.G. Separation Principles of Cycling Temperature Capillary Electrophoresis. Electrophoresis 2012, 33, 1162–1168. [Google Scholar] [CrossRef]
- Ekstrøm, P.O.; Khrapko, K.; Li-Sucholeiki, X.-C.; Hunter, I.W.; Thilly, W.G. Analysis of Mutational Spectra by Denaturing Capillary Electrophoresis. Nat. Protoc. 2008, 3, 1153–1166. [Google Scholar] [CrossRef]
- Ekstrøm, P.O.; Nakken, S.; Johansen, M.; Hovig, E. Automated Amplicon Design Suitable for Analysis of DNA Variants by Melting Techniques. BMC Res. Notes 2015, 8, 667. [Google Scholar] [CrossRef] [PubMed]
- Refinetti, P.; Morgenthaler, S.; Ekstrøm, P.O. Cycling Temperature Capillary Electrophoresis: A Quantitative, Fast and Inexpensive Method to Detect Mutations in Mixed Populations of Human Mitochondrial DNA. Mitochondrion 2016, 29, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Arstad, C.; Refinetti, P.; Kristensen, A.T.; Giercksky, K.-E.; Ekstrøm, P.O. Is Detection of Intraperitoneal Exfoliated Tumor Cells after Surgical Resection of Rectal Cancer a Prognostic Factor of Survival? BMC Cancer 2017, 17, 406. [Google Scholar] [CrossRef] [PubMed]
- Refinetti, P.; Arstad, C.; Thilly, W.G.; Morgenthaler, S.; Ekstrøm, P.O. Mapping Mitochondrial Heteroplasmy in a Leydig Tumor by Laser Capture Micro-Dissection and Cycling Temperature Capillary Electrophoresis. BMC Clin. Pathol. 2017, 17, 6. [Google Scholar] [CrossRef] [PubMed]
- Arstad, C.; Refinetti, P.; Warren, D.; Giercksky, K.-E.; Ekstrøm, P.O. Scanning the Mitochondrial Genome for Mutations by Cycling Temperature Capillary Electrophoresis. Mitochondrial DNA Part A 2018, 29, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Arstad, C.; Taskén, K.; Refinetti, P.; Axcrona, U.; Giercksky, K.-E.; Ekstrøm, P.O. Somatic Mitochondrial DNA Point Mutations Used as Biomarkers to Demonstrate Genomic Heterogeneity in Primary Prostate Cancer. Prostate Cancer 2020, 2020, 7673684. [Google Scholar] [CrossRef] [PubMed]
- Refinetti, P.; Morgenthaler, S.; Thilly, W.G.; Arstad, C.; Ekstrøm, P.O. Tracing of Human Tumor Cell Lineages by Mitochondrial Mutations. Front. Oncol. 2020, 10, 523860. [Google Scholar] [CrossRef]
- Karlen, Y.; McNair, A.; Perseguers, S.; Mazza, C.; Mermod, N. Statistical Significance of Quantitative PCR. BMC Bioinform. 2007, 8, 131. [Google Scholar] [CrossRef]
- Ruijter, J.M.; Ramakers, C.; Hoogaars, W.M.H.; Karlen, Y.; Bakker, O.; Van den Hoff, M.J.B.; Moorman, A. Amplification Efficiency: Linking Baseline and Bias in the Analysis of Quantitative PCR Data. Nucleic Acids Res. 2009, 37, e45. [Google Scholar] [CrossRef]
- Jackson, S.A.; Schoeni, J.L.; Vegge, C.; Pane, M.; Stahl, B.; Bradley, M.; Goldman, V.S.; Burguière, P.; Atwater, J.B.; Sanders, M.E. Improving End-User Trust in the Quality of Commercial Probiotic Products. Front. Microbiol. 2019, 10, 739. [Google Scholar] [CrossRef]
- Chiron, C.; Tompkins, T.A.; Burguière, P. Flow Cytometry: A Versatile Technology for Specific Quantification and Viability Assessment of Micro-Organisms in Multistrain Probiotic Products. J. Appl. Microbiol. 2018, 124, 572–584. [Google Scholar] [CrossRef]
- Petrova, M.I.; Lievens, E.; Malik, S.; Imholz, N.; Lebeer, S. Lactobacillus Species as Biomarkers and Agents That Can Promote Various Aspects of Vaginal Health. Front. Physiol. 2015, 6, 81. [Google Scholar] [CrossRef]
- Sela, D.A.; Mills, D.A. Nursing Our Microbiota: Molecular Linkages between Bifidobacteria and Milk Oligosaccharides. Trends Microbiol. 2010, 18, 298–307. [Google Scholar] [CrossRef]
- Hashem, A.; Tabassum, B.; Abd_Allah, E.F. Bacillus Subtilis: A Plant-Growth Promoting Rhizobacterium That Also Impacts Biotic Stress. Saudi J. Biol. Sci. 2019, 26, 1291–1297. [Google Scholar] [CrossRef]
- Ghahremani, Z.; Escudero, N.; Beltrán-Anadón, D.; Saus, E.; Cunquero, M.; Andilla, J.; Loza-Alvarez, P.; Gabaldón, T.; Sorribas, F.J. Bacillus Firmus Strain I-1582, a Nematode Antagonist by Itself and through the Plant. Front. Plant Sci. 2020, 11, 796. [Google Scholar] [CrossRef]
- Roh, J.-Y.; Choi, J.-Y.; Li, M.-S.; Jin, B.-R.; Je, Y.-H. Bacillus Thuringiensis as a Specific, Safe, and Effective Tool for Insect Pest Control. J. Microbiol. Biotechnol. 2007, 17, 547–559. [Google Scholar]
- Basak, B.B.; Biswas, D.R. Influence of Potassium Solubilizing Microorganism (Bacillus Mucilaginosus) and Waste Mica on Potassium Uptake Dynamics by Sudan Grass (Sorghum Vulgare Pers.) Grown under Two Alfisols. Plant Soil 2009, 317, 235–255. [Google Scholar] [CrossRef]
- Puri, A.; Padda, K.P.; Chanway, C.P. Seedling Growth Promotion and Nitrogen Fixation by a Bacterial Endophyte Paenibacillus Polymyxa P2b-2R and Its GFP Derivative in Corn in a Long-Term Trial. Symbiosis 2016, 69, 123–129. [Google Scholar] [CrossRef]
- Azcón, R.; del Carmen Perálvarez, M.; Roldán, A.; Barea, J.-M. Arbuscular Mycorrhizal Fungi, Bacillus Cereus, and Candida Parapsilosis from a Multicontaminated Soil Alleviate Metal Toxicity in Plants. Microb. Ecol. 2010, 59, 668–677. [Google Scholar] [CrossRef]
- Shen, Z.; Wang, B.; Lv, N.; Sun, Y.; Jiang, X.; Li, R.; Ruan, Y.; Shen, Q. Effect of the Combination of Bio-Organic Fertiliser with Bacillus Amyloliquefaciens NJN-6 on the Control of Banana Fusarium Wilt Disease, Crop Production and Banana Rhizosphere Culturable Microflora. Biocontrol Sci. Technol. 2015, 25, 716–731. [Google Scholar] [CrossRef]
- Masood, S.; Zhao, X.Q.; Shen, R.F. Bacillus Pumilus Promotes the Growth and Nitrogen Uptake of Tomato Plants under Nitrogen Fertilization. Sci. Hortic. 2020, 272, 109581. [Google Scholar] [CrossRef]
- Kang, S.-M.; Radhakrishnan, R.; You, Y.-H.; Joo, G.-J.; Lee, I.-J.; Lee, K.-E.; Kim, J.-H. Phosphate Solubilizing Bacillus Megaterium Mj1212 Regulates Endogenous Plant Carbohydrates and Amino Acids Contents to Promote Mustard Plant Growth. Indian J. Microbiol. 2014, 54, 427–433. [Google Scholar] [CrossRef]
- Kerry, R.G.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.-S.; Das, G. Benefaction of Probiotics for Human Health: A Review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef]
- Rao, S.C.; Athalye-Jape, G.K.; Deshpande, G.C.; Simmer, K.N.; Patole, S.K. Probiotic Supplementation and Late-Onset Sepsis in Preterm Infants: A Meta-Analysis. Pediatrics 2016, 137, e20153684. [Google Scholar] [CrossRef]
- Shehata, A.A.; Yalçın, S.; Latorre, J.D.; Basiouni, S.; Attia, Y.A.; Abd El-Wahab, A.; Visscher, C.; El-Seedi, H.R.; Huber, C.; Hafez, H.M. Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry. Microorganisms 2022, 10, 395. [Google Scholar] [CrossRef]
- Illiano, P.; Brambilla, R.; Parolini, C. The Mutual Interplay of Gut Microbiota, Diet and Human Disease. FEBS J. 2020, 287, 833–855. [Google Scholar] [CrossRef] [PubMed]
- Bottari, B.; Quartieri, A.; Prandi, B.; Raimondi, S.; Leonardi, A.; Rossi, M.; Ulrici, A.; Gatti, M.; Sforza, S.; Nocetti, M. Characterization of the Peptide Fraction from Digested Parmigiano Reggiano Cheese and Its Effect on Growth of Lactobacilli and Bifidobacteria. Int. J. Food Microbiol. 2017, 255, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Polk, D.B. Probiotics and Immune Health. Curr. Opin. Gastroenterol. 2011, 27, 496. [Google Scholar] [CrossRef] [PubMed]
- Breton, J.; Galmiche, M.; Déchelotte, P. Dysbiotic Gut Bacteria in Obesity: An Overview of the Metabolic Mechanisms and Therapeutic Perspectives of next-Generation Probiotics. Microorganisms 2022, 10, 452. [Google Scholar] [CrossRef]
- Senok, A.C.; Ismaeel, A.Y.; Botta, G.A. Probiotics: Facts and Myths. Clin. Microbiol. Infect. 2005, 11, 958–966. [Google Scholar] [CrossRef]
- Mizock, B.A. Probiotics. Dis. -A-Mon. DM 2015, 61, 259–290. [Google Scholar] [CrossRef]
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, Prebiotics and Synbiotics-a Review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef]
- Suez, J.; Zmora, N.; Segal, E.; Elinav, E. The Pros, Cons, and Many Unknowns of Probiotics. Nat. Med. 2019, 25, 716–729. [Google Scholar] [CrossRef]
- Da Cruz, M.F.; Magno, M.B.; Jural, L.A.; Pimentel, T.C.; Masterson, D.; Esmerino, E.A.; Ramos, G.L.P.A.; Gomila, J.V.; Silva, M.C.; Cruz, A.G. Probiotics and Dairy Products in Dentistry: A Bibliometric and Critical Review of Randomized Clinical Trials. Food Res. Int. 2022, 157, 111228. [Google Scholar] [CrossRef]
- Ganji-Arjenaki, M.; Rafieian-Kopaei, M. Probiotics Are a Good Choice in Remission of Inflammatory Bowel Diseases: A Meta Analysis and Systematic Review. J. Cell. Physiol. 2018, 233, 2091–2103. [Google Scholar] [CrossRef]
- Plaza-Diaz, J.; Ruiz-Ojeda, F.J.; Gil-Campos, M.; Gil, A. Mechanisms of Action of Probiotics. Adv. Nutr. 2019, 10, S49–S66. [Google Scholar] [CrossRef]
- Islam, S.U. Clinical Uses of Probiotics. Medicine 2016, 95, e2658. [Google Scholar] [CrossRef]
- Liu, Y.; Tran, D.Q.; Rhoads, J.M. Probiotics in Disease Prevention and Treatment. J. Clin. Pharmacol. 2018, 58, S164–S179. [Google Scholar] [CrossRef]
- Tillisch, K. The Effects of Gut Microbiota on CNS Function in Humans. Gut Microbes 2014, 5, 404–410. [Google Scholar] [CrossRef]
- Kahouli, I.; Malhotra, M.; Alaoui-Jamali, M.; Prakash, S. In-Vitro Characterization of the Anti-Cancer Activity of the Probiotic Bacterium Lactobacillus Fermentum NCIMB 5221 and Potential against Colorectal Cancer. J. Cancer Sci. 2015, 7, 224–235. [Google Scholar]
- Hilton, E.; Kolakowski, P.; Singer, C.; Smith, M. Efficacy of Lactobacillus GG as a Diarrheal Preventive in Travelers. J. Travel Med. 1997, 4, 41–43. [Google Scholar] [CrossRef]
- McFarland, L.V. Meta-Analysis of Probiotics for the Prevention of Traveler’s Diarrhea. Travel Med. Infect. Dis. 2007, 5, 97–105. [Google Scholar] [CrossRef]
- Sazawal, S.; Hiremath, G.; Dhingra, U.; Malik, P.; Deb, S.; Black, R.E. Efficacy of Probiotics in Prevention of Acute Diarrhoea: A Meta-Analysis of Masked, Randomised, Placebo-Controlled Trials. Lancet Infect. Dis. 2006, 6, 374–382. [Google Scholar] [CrossRef]
- Cremonini, F.; Di Caro, S.; Nista, E.C.; Bartolozzi, F.; Capelli, G.; Gasbarrini, G.; Gasbarrini, A. Meta-Analysis: The Effect of Probiotic Administration on Antibiotic-Associated Diarrhoea. Aliment. Pharmacol. Ther. 2002, 16, 1461–1467. [Google Scholar] [CrossRef]
- Naidoo, K.; Gordon, M.; Fagbemi, A.O.; Thomas, A.G.; Akobeng, A.K. Probiotics for Maintenance of Remission in Ulcerative Colitis. Cochrane Database Syst. Rev. 2011, CD007443. [Google Scholar] [CrossRef]
- AlFaleh, K.; Anabrees, J. Probiotics for Prevention of Necrotizing Enterocolitis in Preterm Infants. Evid.-Based Child Health A Cochrane Rev. J. 2014, 9, 584–671. [Google Scholar] [CrossRef]
- Autran, C.A.; Schoterman, M.H.; Jantscher-Krenn, E.; Kamerling, J.P.; Bode, L. Sialylated Galacto-Oligosaccharides and 2′-Fucosyllactose Reduce Necrotising Enterocolitis in Neonatal Rats. Br. J. Nutr. 2016, 116, 294–299. [Google Scholar] [CrossRef]
- Vetvicka, V.; Oliveira, C. β(1-3)(1-6)-D-Glucans Modulate Immune Status in Pigs: Potential Importance for Efficiency of Commercial Farming. Ann. Transl. Med. 2014, 2, 16. [Google Scholar]
- Jacob, J.P.; Pescatore, A.J. Barley β-Glucan in Poultry Diets. Ann. Transl. Med. 2014, 2, 20. [Google Scholar]
- Alok, A.; Singh, I.D.; Singh, S.; Kishore, M.; Jha, P.C.; Iqubal, M.A. Probiotics: A New Era of Biotherapy. Adv. Biomed. Res. 2017, 6, 31. [Google Scholar] [CrossRef]
- Fiocco, D.; Longo, A.; Arena, M.P.; Russo, P.; Spano, G.; Capozzi, V. How Probiotics Face Food Stress: They Get by with a Little Help. Crit. Rev. Food Sci. Nutr. 2020, 60, 1552–1580. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, T.C.; da Costa, W.K.A.; Barão, C.E.; Rosset, M.; Magnani, M. Vegan Probiotic Products: A Modern Tendency or the Newest Challenge in Functional Foods. Food Res. Int. 2021, 140, 110033. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K. How Some Probiotic Scientists Are Working to Address COVID-19. 2020. Available online: https://isappscience.org/how-some-probiotic-and-prebiotic-scientists-are-working-to-address-covid-19/ (accessed on 29 June 2022).
- Bottari, B.; Castellone, V.; Neviani, E. Probiotics and COVID-19. Int. J. Food Sci. Nutr. 2021, 72, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
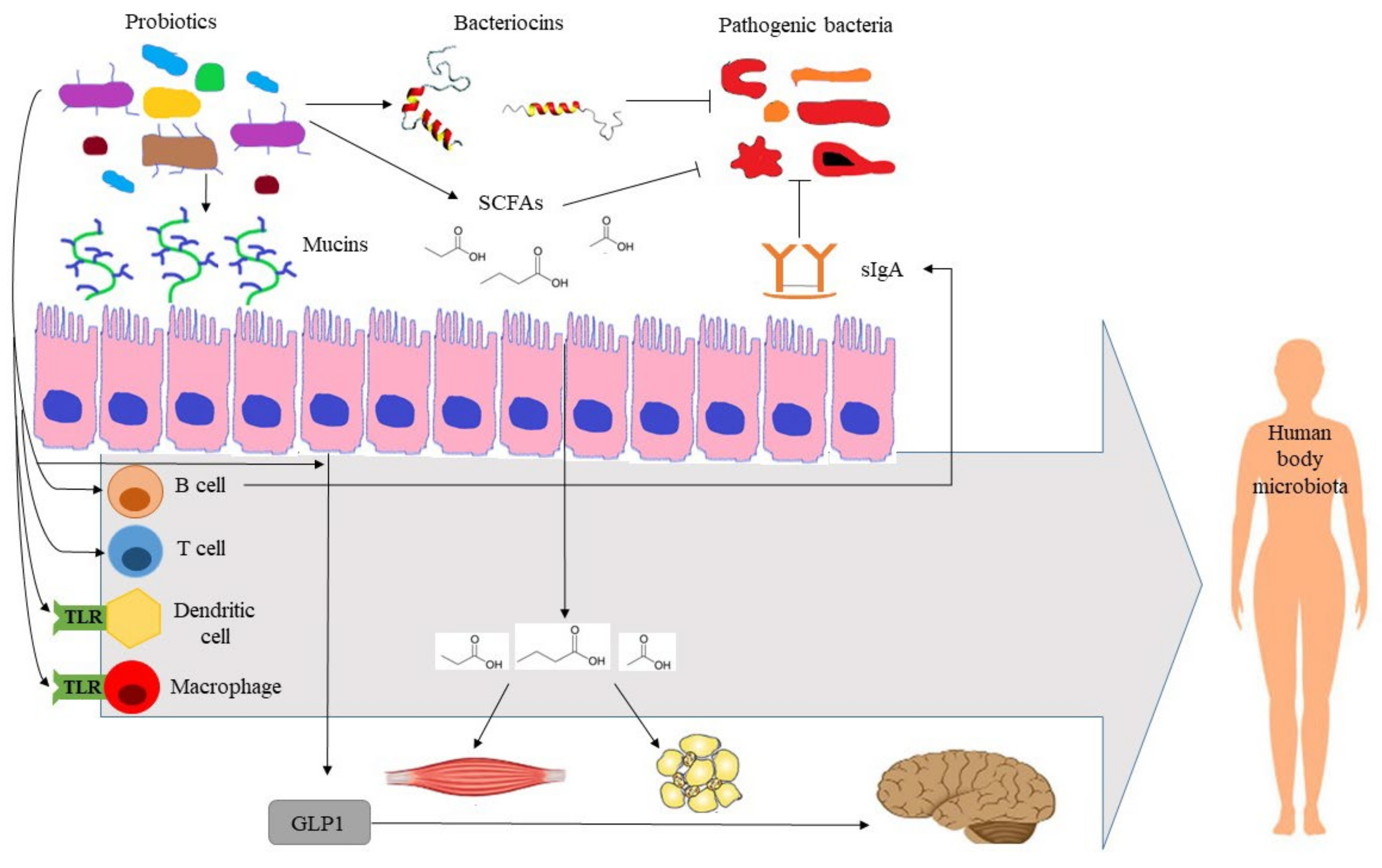
- Van Zyl, W.F.; Deane, S.M.; Dicks, L.M.T. Molecular Insights into Probiotic Mechanisms of Action Employed against Intestinal Pathogenic Bacteria. Gut Microbes 2020, 12, 1831339. [Google Scholar] [CrossRef]
- Burgueño, J.F.; Abreu, M.T. Epithelial Toll-like Receptors and Their Role in Gut Homeostasis and Disease. Nat Rev Gastroenterol Hepatol 2020, 17, 263–278. [Google Scholar] [CrossRef]
- Mirzaei, R.; Dehkhodaie, E.; Bouzari, B.; Rahimi, M.; Gholestani, A.; Hosseini-Fard, S.R.; Keyvani, H.; Teimoori, A.; Karampoor, S. Dual Role of Microbiota-Derived Short-Chain Fatty Acids on Host and Pathogen. Biomed. Pharmacother. 2022, 145, 112352. [Google Scholar] [CrossRef]
- Blaak, E.E.; Canfora, E.E.; Theis, S.; Frost, G.; Groen, A.K.; Mithieux, G.; Nauta, A.; Scott, K.; Stahl, B.; van Harsselaar, J.; et al. Short Chain Fatty Acids in Human Gut and Metabolic Health. Benef. Microbes 2020, 11, 411–455. [Google Scholar] [CrossRef]
- Lai, Y.; Dhingra, R.; Zhang, Z.; Ball, L.M.; Zylka, M.J.; Lu, K. Toward Elucidating the Human Gut Microbiota–Brain Axis: Molecules, Biochemistry, and Implications for Health and Diseases. Biochemistry 2021. [Google Scholar] [CrossRef]
- Fabbrizzi, A.; Amedei, A.; Lavorini, F.; Renda, T.; Fontana, G. The Lung Microbiome: Clinical and Therapeutic Implications. Intern. Emerg. Med. 2019, 14, 1241–1250. [Google Scholar] [CrossRef]
- Gutiérrez-Castrellón, P.; Gandara-Martí, T.; Abreu, Y.; Abreu, A.T.; Nieto-Rufino, C.D.; López-Orduña, E.; Jiménez-Escobar, I.; Jiménez-Gutiérrez, C.; López-Velazquez, G.; Espadaler-Mazo, J. Probiotic Improves Symptomatic and Viral Clearance in Covid19 Outpatients: A Randomized, Quadruple-Blinded, Placebo-Controlled Trial. Gut Microbes 2022, 14, 2018899. [Google Scholar] [CrossRef]
- Mercader-Barceló, J.; Truyols-Vives, J.; Río, C.; López-Safont, N.; Sala-Llinàs, E.; Chaplin, A. Insights into the Role of Bioactive Food Ingredients and the Microbiome in Idiopathic Pulmonary Fibrosis. Int. J. Mol. Sci. 2020, 21, 6051. [Google Scholar] [CrossRef]
- Sinha, S.; Lin, G.; Ferenczi, K. The Skin Microbiome and the Gut-Skin Axis. Clin. Dermatol. 2021, 39, 829–839. [Google Scholar] [CrossRef]
- Khan, I.; Ullah, N.; Zha, L.; Bai, Y.; Khan, A.; Zhao, T.; Che, T.; Zhang, C. Alteration of Gut Microbiota in Inflammatory Bowel Disease (IBD): Cause or Consequence? IBD Treatment Targeting the Gut Microbiome. Pathogens 2019, 8, 126. [Google Scholar] [CrossRef]
- Li, H.-Y.; Zhou, D.-D.; Gan, R.-Y.; Huang, S.-Y.; Zhao, C.-N.; Shang, A.; Xu, X.-Y.; Li, H.-B. Effects and Mechanisms of Probiotics, Prebiotics, Synbiotics, and Postbiotics on Metabolic Diseases Targeting Gut Microbiota: A Narrative Review. Nutrients 2021, 13, 3211. [Google Scholar] [CrossRef]
- Sergeev, I.N.; Aljutaily, T.; Walton, G.; Huarte, E. Effects of Synbiotic Supplement on Human Gut Microbiota, Body Composition and Weight Loss in Obesity. Nutrients 2020, 12, 222. [Google Scholar] [CrossRef]
- Kvakova, M.; Bertkova, I.; Stofilova, J.; Savidge, T.C. Co-Encapsulated Synbiotics and Immobilized Probiotics in Human Health and Gut Microbiota Modulation. Foods 2021, 10, 1297. [Google Scholar] [CrossRef]
- Krumbeck, J.A.; Walter, J.; Hutkins, R.W. Synbiotics for Improved Human Health: Recent Developments, Challenges, and Opportunities. Annu. Rev. Food Sci. Technol. 2018, 9, 451–479. [Google Scholar] [CrossRef]
- Ahmadi Badi, S.; Tarashi, S.; Fateh, A.; Rohani, P.; Masotti, A.; Siadat, S.D. From the Role of Microbiota in Gut-Lung Axis to SARS-CoV-2 Pathogenesis. Mediat. Inflamm. 2021, 2021, 6611222. [Google Scholar] [CrossRef]
- Antunes, A.E.; Vinderola, G.; Xavier-Santos, D.; Sivieri, K. Potential Contribution of Beneficial Microbes to Face the COVID-19 Pandemic. Food Res. Int. 2020, 136, 109577. [Google Scholar] [CrossRef]
- Eslamparast, T.; Zamani, F.; Hekmatdoost, A.; Sharafkhah, M.; Eghtesad, S.; Malekzadeh, R.; Poustchi, H. Effects of Synbiotic Supplementation on Insulin Resistance in Subjects with the Metabolic Syndrome: A Randomised, Double-Blind, Placebo-Controlled Pilot Study. Br. J. Nutr. 2014, 112, 438–445. [Google Scholar] [CrossRef]
- Rogha, M.; Esfahani, M.Z.; Zargarzadeh, A.H. The Efficacy of a Synbiotic Containing Bacillus Coagulans in Treatment of Irritable Bowel Syndrome: A Randomized Placebo-Controlled Trial. Gastroenterol. Hepatol. Bed Bench 2014, 7, 156. [Google Scholar]
- Dinleyici, E.C.; Dalgic, N.; Guven, S.; Ozen, M.; Kara, A.; Arica, V.; Metin-Timur, O.; Sancar, M.; Kurugol, Z.; Tanir, G. The Effect of a Multispecies Synbiotic Mixture on the Duration of Diarrhea and Length of Hospital Stay in Children with Acute Diarrhea in Turkey: Single Blinded Randomized Study. Eur. J. Pediatrics 2013, 172, 459–464. [Google Scholar] [CrossRef]
- Worthley, D.L.; Le Leu, R.K.; Whitehall, V.L.; Conlon, M.; Christophersen, C.; Belobrajdic, D.; Mallitt, K.-A.; Hu, Y.; Irahara, N.; Ogino, S. A Human, Double-Blind, Placebo-Controlled, Crossover Trial of Prebiotic, Probiotic, and Synbiotic Supplementation: Effects on Luminal, Inflammatory, Epigenetic, and Epithelial Biomarkers of Colorectal Cancer. Am. J. Clin. Nutr. 2009, 90, 578–586. [Google Scholar] [CrossRef]
- Dehghani, H.; Heidari, F.; Mozaffari-Khosravi, H.; Nouri-Majelan, N.; Dehghani, A. Synbiotic Supplementations for Azotemia in Patients with Chronic Kidney Disease: A Randomized Controlled Trial. Iran. J. Kidney Dis. 2016, 10, 351–357. [Google Scholar]
- Javadi, L.; Khoshbaten, M.; Safaiyan, A.; Ghavami, M.; Abbasi, M.M.; Gargari, B.P. Pro-and Prebiotic Effects on Oxidative Stress and Inflammatory Markers in Non-Alcoholic Fatty Liver Disease. Asia Pac. J. Clin. Nutr. 2018, 27, 1031–1039. [Google Scholar]
- Ahanchian, H.; Jafari, S.A.; Ansari, E.; Ganji, T.; Kiani, M.A.; Khalesi, M.; Momen, T.; Kianifar, H. A Multi-Strain Synbiotic May Reduce Viral Respiratory Infections in Asthmatic Children: A Randomized Controlled Trial. Electron. Physician 2016, 8, 2833. [Google Scholar] [CrossRef]
- Wu, K.-G.; Li, T.-H.; Peng, H.-J. Lactobacillus Salivarius plus Fructo-Oligosaccharide Is Superior to Fructo-Oligosaccharide Alone for Treating Children with Moderate to Severe Atopic Dermatitis: A Double-Blind, Randomized, Clinical Trial of Efficacy and Safety. Br. J. Dermatol. 2012, 166, 129–136. [Google Scholar] [CrossRef]
- Haghighat, N.; Mohammadshahi, M.; Shayanpour, S.; Haghighizadeh, M.H.; Rahmdel, S.; Rajaei, M. The Effect of Synbiotic and Probiotic Supplementation on Mental Health Parameters in Patients Undergoing Hemodialysis: A Double-Blind, Randomized, Placebo-Controlled Trial. Indian J. Nephrol. 2021, 31, 149. [Google Scholar]
- Rodriguez-Herrera, A.; Tims, S.; Polman, J.; Porcel Rubio, R.; Muñoz Hoyos, A.; Agosti, M.; Lista, G.; Corvaglia, L.T.; Knol, J.; Roeselers, G. Early-Life Fecal Microbiome and Metabolome Dynamics in Response to an Intervention with Infant Formula Containing Specific Prebiotics and Postbiotics. Am. J. Physiol.-Gastrointest. Liver Physiol. 2022, 322, G571–G582. [Google Scholar] [CrossRef]
- Singh, A.; D’Amico, D.; Andreux, P.A.; Fouassier, A.M.; Blanco-Bose, W.; Evans, M.; Aebischer, P.; Auwerx, J.; Rinsch, C. Urolithin A Improves Muscle Strength, Exercise Performance, and Biomarkers of Mitochondrial Health in a Randomized Trial in Middle-Aged Adults. Cell Rep. Med. 2022, 3, 100633. [Google Scholar] [CrossRef] [PubMed]
- Ricci, A.; Allende, A.; Bolton, D.; Chemaly, M.; Davies, R.; Girones, R.; Herman, L.; Koutsoumanis, K.; Lindqvist, R.; Nørrung, B.; et al. Scientific Opinion on the Update of the List of QPS-recommended Biological Agents Intentionally Added to Food or Feed as Notified to EFSA. EFSA J. 2017, 15, e04664. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.A.; Burkholder, K.M. Application of Prebiotics and Probiotics in Poultry Production. Poult. Sci. 2003, 82, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Chen, Y.; Li, X.; Yang, W.; Wen, C.; Kang, Y.; Wang, A.; Zhou, Y. Effects of Synbiotic Supplementation on Growth Performance, Carcass Characteristics, Meat Quality and Muscular Antioxidant Capacity and Mineral Contents in Broilers: Effects of Synbiotic Supplementation. J. Sci. Food Agric. 2017, 97, 3699–3705. [Google Scholar] [CrossRef]
- Gaggìa, F.; Mattarelli, P.; Biavati, B. Probiotics and Prebiotics in Animal Feeding for Safe Food Production. Int. J. Food Microbiol. 2010, 141, S15–S28. [Google Scholar] [CrossRef]
- Stanczuk, J.; Zdunczyk, Z.; Juskiewicz, J.; Jankowski, J. Indices of Response of Young Turkeys to Diets Containing Mannan- Oligosaccharide or Inulin Raciono, Turin Č Io Monooligosacharid Ų Ir Inulino, Poveikis. Veterinarija Ir Zootechnika 2005, 31, 98–101. [Google Scholar]
- Kawashima, T.; Hayashi, K.; Kosaka, A.; Kawashima, M.; Igarashi, T.; Tsutsui, H.; Tsuji, N.M.; Nishimura, I.; Hayashi, T.; Obata, A. Lactobacillus Plantarum Strain YU from Fermented Foods Activates Th1 and Protective Immune Responses. Int. Immunopharmacol. 2011, 11, 2017–2024. [Google Scholar] [CrossRef]
- Guevarra, R.B.; Lee, J.H.; Lee, S.H.; Seok, M.J.; Kim, D.W.; Kang, B.N.; Johnson, T.J.; Isaacson, R.E.; Kim, H.B. Piglet Gut Microbial Shifts Early in Life: Causes and Effects. J. Anim. Sci. Biotechnol. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Zangeronimo, M.G.; Cantarelli, V.d.S.; Fialho, E.T.; Amaral, N.d.O.; Silveira, H.; Pereira, L.d.M.; Pereira, L.J. Herbal Extracts and Symbiotic Mixture Replacing Antibiotics in Piglets at the Initial Phase. Rev. Bras. De Zootec. 2011, 40, 1045–1051. [Google Scholar] [CrossRef]
- Casey, P.G.; Gardiner, G.E.; Casey, G.; Bradshaw, B.; Lawlor, P.G.; Lynch, P.B.; Leonard, F.C.; Stanton, C.; Ross, R.P.; Fitzgerald, G.F.; et al. A Five-Strain Probiotic Combination Reduces Pathogen Shedding and Alleviates Disease Signs in Pigs Challenged with Salmonella Enterica Serovar Typhimurium. Appl. Environ. Microbiol. 2007, 73, 1858–1863. [Google Scholar] [CrossRef]
- Duarte, M.E.; Tyus, J.; Kim, S.W. Synbiotic Effects of Enzyme and Probiotics on Intestinal Health and Growth of Newly Weaned Pigs Challenged with Enterotoxigenic F18+ Escherichia coli. Front. Vet. Sci. 2020, 7, 573. [Google Scholar] [CrossRef]
- Cadogan, D.J.; Choct, M. Pattern of Non-Starch Polysaccharide Digestion along the Gut of the Pig: Contribution to Available Energy. Anim. Nutr. 2015, 1, 160–165. [Google Scholar] [CrossRef]
- Passos, A.A.; Park, I.; Ferket, P.; von Heimendahl, E.; Kim, S.W. Effect of Dietary Supplementation of Xylanase on Apparent Ileal Digestibility of Nutrients, Viscosity of Digesta, and Intestinal Morphology of Growing Pigs Fed Corn and Soybean Meal Based Diet. Anim. Nutr. 2015, 1, 19–23. [Google Scholar] [CrossRef]
- Lee, Y.K. Selection and Maintenance of Probiotic Microorganisms. In Handbook of Probiotics and Prebiotics, 2nd ed.; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2008; pp. 177–187. ISBN 978-0-470-13544-0. [Google Scholar]
- Markowiak, P.; Śliżewska, K. The Role of Probiotics, Prebiotics and Synbiotics in Animal Nutrition. Gut Pathog. 2018, 10, 21. [Google Scholar] [CrossRef]
- Weimer, P.J. Redundancy, Resilience, and Host Specificity of the Ruminal Microbiota: Implications for Engineering Improved Ruminal Fermentations. Front. Microbiol. 2015, 6, 296. [Google Scholar] [CrossRef]
- Melara, E.G.; Avellaneda, M.C.; Valdivié, M.; García-Hernández, Y.; Aroche, R.; Martínez, Y. Probiotics: Symbiotic Relationship with the Animal Host. Animals 2022, 12, 719. [Google Scholar] [CrossRef]
- Stover, M.G.; Watson, R.R.; Collier, R.J. Pre- and Probiotic Supplementation in Ruminant Livestock Production. In Probiotics, Prebiotics, and Synbiotics: Bioactive Foods in Health Promotion; Academic Press: Cambridge, MA, USA, 2016; pp. 25–36. ISBN 978-0-12-802189-7. [Google Scholar]
- Reuben, R.C.; Elghandour, M.M.M.Y.; Alqaisi, O.; Cone, J.W.; Márquez, O.; Salem, A.Z.M. Influence of Microbial Probiotics on Ruminant Health and Nutrition: Sources, Mode of Action and Implications. J. Sci. Food Agric. 2022, 102, 1319–1340. [Google Scholar] [CrossRef]
- Stein, D.R.; Allen, D.T.; Perry, E.B.; Bruner, J.C.; Gates, K.W.; Rehberger, T.G.; Mertz, K.; Jones, D.; Spicer, L.J. Effects of Feeding Propionibacteria to Dairy Cows on Milk Yield, Milk Components, and Reproduction. J. Dairy Sci. 2006, 89, 111–125. [Google Scholar] [CrossRef]
- Poppy, G.D.; Rabiee, A.R.; Lean, I.J.; Sanchez, W.K.; Dorton, K.L.; Morley, P.S. A Meta-Analysis of the Effects of Feeding Yeast Culture Produced by Anaerobic Fermentation of Saccharomyces Cerevisiae on Milk Production of Lactating Dairy Cows. J. Dairy Sci. 2012, 95, 6027–6041. [Google Scholar] [CrossRef]
- Estrada-Angulo, A.; Zapata-Ramírez, O.; Castro-Pérez, B.I.; Urías-Estrada, J.D.; Gaxiola-Camacho, S.; Angulo-Montoya, C.; Ríos-Rincón, F.G.; Barreras, A.; Zinn, R.A.; Leyva-Morales, J.B.; et al. The Effects of Single or Combined Supplementation of Probiotics and Prebiotics on Growth Performance, Dietary Energetics, Carcass Traits, and Visceral Mass in Lambs Finished under Subtropical Climate Conditions. Biology 2021, 10, 1137. [Google Scholar] [CrossRef]
- Uyeno, Y.; Shigemori, S.; Shimosato, T. Effect of Probiotics/Prebiotics on Cattle Health and Productivity. Microbes Environ. 2015, 30, 126–132. [Google Scholar] [CrossRef]
- Xu, H.; Huang, W.; Hou, Q.; Kwok, L.; Sun, Z.; Ma, H.; Zhao, F.; Lee, Y.-K.; Zhang, H. The Effects of Probiotics Administration on the Milk Production, Milk Components and Fecal Bacteria Microbiota of Dairy Cows. Sci. Bull. 2017, 62, 767–774. [Google Scholar] [CrossRef]
- Stella, A.V.; Paratte, R.; Valnegri, L.; Cigalino, G.; Soncini, G.; Chevaux, E.; Dell’Orto, V.; Savoini, G. Effect of Administration of Live Saccharomyces Cerevisiae on Milk Production, Milk Composition, Blood Metabolites, and Faecal Flora in Early Lactating Dairy Goats. Small Rumin. Res. 2007, 67, 7–13. [Google Scholar] [CrossRef]
- Callaway, T.R.; Edrington, T.S.; Anderson, R.C.; Nisbet, D.J. Efficiency of the Use of Pre- and Probiotics for Dairy Cows. In Proceedings of the 3rd International Livestock Farming Systems Symposium, Aberdeen, Scotland, 12 September 1996; pp. 33–54. [Google Scholar]
- Fleige, S.; Preißinger, W.; Meyer, H.H.D.; Pfaffl, M.W. Effect of Lactulose on Growth Performance and Intestinal Morphology of Pre-Ruminant Calves Using a Milk Replacer Containing Enterococcus Faecium. Animal 2007, 1, 367–373. [Google Scholar] [CrossRef]
- Morrison, S.J.; Dawson, S.; Carson, A.F. The Effects of Mannan Oligosaccharide and Streptococcus Faecium Addition to Milk Replacer on Calf Health and Performance. Livest. Sci. 2010, 131, 292–296. [Google Scholar] [CrossRef]
- Jonova, S.; Ilgaza, A.; Zolovs, M. The Impact of Inulin and a Novel Synbiotic (Yeast Saccharomyces Cerevisiae Strain 1026 and Inulin) on the Development and Functional State of the Gastrointestinal Canal of Calves. Vet. Med. Int. 2021, 2021, 1–9. [Google Scholar] [CrossRef]
- Guowei, S.; He, Y.; Wan, H.; Hui, Y.; Li, H. Effects of Prebiotics on Antioxidant Activity of Goat Milk Fermented by Lactobacillus Plantarum L60. Acta Univ. Cibiniensis. Ser. E Food Technol. 2017, 21, 11–18. [Google Scholar] [CrossRef][Green Version]
- Han, G.; Gao, X.; Duan, J.; Zhang, H.; Zheng, Y.; He, J.; Huo, N.; Pei, C.; Li, H.; Gu, S. Effects of Yeasts on Rumen Bacterial Flora, Abnormal Metabolites, and Blood Gas in Sheep with Induced Subacute Ruminal Acidosis. Anim. Feed Sci. Technol. 2021, 280, 115042. [Google Scholar] [CrossRef]
- Lettat, A.; Nozière, P.; Silberberg, M.; Morgavi, D.P.; Berger, C.; Martin, C. Rumen Microbial and Fermentation Characteristics Are Affected Differently by Bacterial Probiotic Supplementation during Induced Lactic and Subacute Acidosis in Sheep. BMC Microbiol. 2012, 12, 142. [Google Scholar] [CrossRef] [PubMed]
- Zapata, O.; Cervantes, A.; Barreras, A.; Monge-Navarro, F.; González-Vizcarra, V.M.; Estrada-Angulo, A.; Urías-Estrada, J.D.; Corona, L.; Zinn, R.A.; Martínez-Alvarez, I.G.; et al. Effects of Single or Combined Supplementation of Probiotics and Prebiotics on Ruminal Fermentation, Ruminal Bacteria and Total Tract Digestion in Lambs. Small Rumin. Res. 2021, 204, 106538. [Google Scholar] [CrossRef]
- Rohani, M.F.; Islam, S.M.; Hossain, M.K.; Ferdous, Z.; Siddik, M.A.B.; Nuruzzaman, M.; Padeniya, U.; Brown, C.; Shahjahan, M. Probiotics, Prebiotics and Synbiotics Improved the Functionality of Aquafeed: Upgrading Growth, Reproduction, Immunity and Disease Resistance in Fish. Fish Shellfish Immunol. 2022, 120, 569–589. [Google Scholar] [CrossRef] [PubMed]
- Tanwar, J.; Das, S.; Fatima, Z.; Hameed, S. Multidrug Resistance: An Emerging Crisis. Interdiscip. Perspect. Infect. Dis. 2014, 2014, 541340. [Google Scholar] [CrossRef] [PubMed]
- Irianto, A.; Austin, B. Probiotics in Aquaculture. J. Fish Dis. 2002, 25, 633–642. [Google Scholar] [CrossRef]
- Prasad, L.; Baghel, D.; Kumar, V.; Laxmi, P.; Dharmendra Singh, B.; Vikas, K. Role and Prospects of Probiotics Use in Aquaculture. Aquacult 2003, 4, 247–251. [Google Scholar]
- Verschuere, L.; Rombaut, G.; Sorgeloos, P.; Verstraete, W. Probiotic Bacteria as Biological Control Agents in Aquaculture. Microbiol. Mol. Biol. Rev. 2000, 64, 655. [Google Scholar] [CrossRef]
- Dawood, M.A.; Koshio, S. Recent Advances in the Role of Probiotics and Prebiotics in Carp Aquaculture: A Review. Aquaculture 2016, 454, 243–251. [Google Scholar] [CrossRef]
- Hoseinifar, S.H.; Yousefi, S.; Van Doan, H.; Ashouri, G.; Gioacchini, G.; Maradonna, F.; Carnevali, O. Oxidative Stress and Antioxidant Defense in Fish: The Implications of Probiotic, Prebiotic, and Synbiotics. Rev. Fish. Sci. Aquac. 2020, 29, 198–217. [Google Scholar] [CrossRef]
- Mohammadi, G.; Hafezieh, M.; Karimi, A.; Azra, M.N.; Van Doan, H.; Tapingkae, W.; Abdelrahman, H.A.; Dawood, M.A.O. The Synergistic Effects of Plant Polysaccharide and Pediococcus Acidilactici as a Synbiotic Additive on Growth, Antioxidant Status, Immune Response, and Resistance of Nile Tilapia (Oreochromis Niloticus) against Aeromonas Hydrophila. Fish Shellfish. Immunol. 2021, 120, 304–313. [Google Scholar] [CrossRef]
- El-Haroun, E.R.; Goda, A.M.A.S.; Kabir Chowdhury, M.A. Effect of Dietary Probiotic Biogen® Supplementation as a Growth Promoter on Growth Performance and Feed Utilization of Nile Tilapia Oreochromis Niloticus (L.). Aquac. Res. 2006, 37, 1473–1480. [Google Scholar] [CrossRef]
- Michael, E.T.; Amos, S.O.; Hussaini, L.T. A Review on Probiotics Application in Aquaculture. Fish. Aquac. J. 2014, 5, 1. [Google Scholar] [CrossRef]
- Batista, K.S.; de Albuquerque, J.G.; de Vasconcelos, M.H.A.; Bezerra, M.L.R.; da Silva Barbalho, M.B.; Oliveira, R.P.; de Souza Aquino, J. Probiotics and Prebiotics: Potential Prevention and Therapeutic Target for Nutritional Management of COVID-19? Nutr. Res. Rev. 2021, 1–42. [Google Scholar] [CrossRef]
- McKenzie, V.J.; Kueneman, J.G.; Harris, R.N. Probiotics as a Tool for Disease Mitigation in Wildlife: Insights from Food Production and Medicine. Ann. N. Y. Acad. Sci. 2018, 1429, 18–30. [Google Scholar] [CrossRef]
- Ray, P.; Lakshmanan, V.; Labbé, J.L.; Craven, K.D. Microbe to Microbiome: A Paradigm Shift in the Application of Microorganisms for Sustainable Agriculture. Front. Microbiol. 2020, 11, 622926. [Google Scholar] [CrossRef]
- Haque, M.M.; Mosharaf, M.K.; Khatun, M.; Haque, M.A.; Biswas, M.S.; Islam, M.S.; Islam, M.M.; Shozib, H.B.; Miah, M.M.U.; Molla, A.H. Biofilm Producing Rhizobacteria with Multiple Plant Growth-Promoting Traits Promote Growth of Tomato under Water-Deficit Stress. Front. Microbiol. 2020, 11, 542053. [Google Scholar] [CrossRef]
- Park, K.H.; Oh, S.-Y.; Yoo, S.; Fong, J.J.; Kim, C.S.; Jo, J.W.; Lim, Y.W. Influence of Season and Soil Properties on Fungal Communities of Neighboring Climax Forests (Carpinus cordata and Fraxinus rhynchophylla). Front. Microbiol. 2020, 11, 2643. [Google Scholar] [CrossRef]
- Vassilev, N.; Eichler-Löbermann, B.; Flor-Peregrin, E.; Martos, V.; Reyes, A.; Vassileva, M. Production of a Potential Liquid Plant Bio-Stimulant by Immobilized Piriformospora Indica in Repeated-Batch Fermentation Process. AMB Express 2017, 7, 1–7. [Google Scholar] [CrossRef]
- Rahman, M.; Sabir, A.A.; Mukta, J.A.; Khan, M.; Alam, M.; Mohi-Ud-Din, M.; Miah, M.; Rahman, M.; Islam, M.T. Plant Probiotic Bacteria Bacillus and Paraburkholderia Improve Growth, Yield and Content of Antioxidants in Strawberry Fruit. Sci. Rep. 2018, 8, 1–11. [Google Scholar]
- Niemeyer, J.C.; Lolata, G.B.; de Carvalho, G.M.; Da Silva, E.M.; Sousa, J.P.; Nogueira, M.A. Microbial Indicators of Soil Health as Tools for Ecological Risk Assessment of a Metal Contaminated Site in Brazil. Appl. Soil Ecol. 2012, 59, 96–105. [Google Scholar] [CrossRef]
- Vassilev, N.; Vassileva, M.; Martos, V.; Garcia del Moral, L.F.; Kowalska, J.; Tylkowski, B.; Malusá, E. Formulation of Microbial Inoculants by Encapsulation in Natural Polysaccharides: Focus on Beneficial Properties of Carrier Additives and Derivatives. Front. Plant Sci. 2020, 11, 270. [Google Scholar]
- González-Salazar, C.; Stephens, C.R.; Marquet, P.A. Comparing the Relative Contributions of Biotic and Abiotic Factors as Mediators of Species’ Distributions. Ecol. Model. 2013, 248, 57–70. [Google Scholar] [CrossRef]
- Dwivedi, S.; Sahrawat, K.; Puppala, N.; Ortiz, R. Plant Prebiotics and Human Health: Biotechnology to Breed Prebiotic-Rich Nutritious Food Crops. Electron. J. Biotechnol. 2014, 17, 238–245. [Google Scholar] [CrossRef]
- Moradipour, M.; Saberi-Riseh, R.; Mohammadinejad, R.; Hosseini, A. Nano-Encapsulation of Plant Growth-Promoting Rhizobacteria and Their Metabolites Using Alginate-Silica Nanoparticles and Carbon Nanotube Improves Ucb1 Pistachio Micropropagation. J. Microbiol. Biotechnol. 2019, 29, 1096–1103. [Google Scholar] [CrossRef]
- Harman, G.; Khadka, R.; Doni, F.; Uphoff, N. Benefits to Plant Health and Productivity from Enhancing Plant Microbial Symbionts. Front. Plant Sci. 2021, 11, 610065. [Google Scholar] [CrossRef]
- Angelim, A.L.; Costa, S.P.; Farias, B.C.S.; Aquino, L.F.; Melo, V.M.M. An Innovative Bioremediation Strategy Using a Bacterial Consortium Entrapped in Chitosan Beads. J. Environ. Manag. 2013, 127, 10–17. [Google Scholar] [CrossRef]
- Yadav, K.K.; Gupta, N.; Kumar, V.; Singh, J.K. Bioremediation of Heavy Metals from Contaminated Sites Using Potential Species: A Review. Indian J. Environ. Prot. 2017, 37, 65. [Google Scholar]
- Baez-Rogelio, A.; Morales-García, Y.E.; Quintero-Hernández, V.; Muñoz-Rojas, J. Next Generation of Microbial Inoculants for Agriculture and Bioremediation. Microb. Biotechnol. 2017, 10, 19–21. [Google Scholar] [CrossRef]
- Farhadian, M.; Vachelard, C.; Duchez, D.; Larroche, C. In Situ Bioremediation of Monoaromatic Pollutants in Groundwater: A Review. Bioresour. Technol. 2008, 99, 5296–5308. [Google Scholar] [CrossRef]
- Cao, W.; Zhang, H.; Wang, Y.; Pan, J. Bioremediation of Polluted Surface Water by Using Biofilms on Filamentous Bamboo. Ecol. Eng. 2012, 42, 146–149. [Google Scholar] [CrossRef]
- Voskoboinikov, G.M.; Matishov, G.G.; Metelkova, L.O.; Zhakovskaya, Z.A.; Lopushanskaya, E.M. Participation of the Green Algae Ulvaria Obscura in Bioremediation of Sea Water from Oil Products. In Proceedings of the Doklady Biological Sciences; Pleiades Publishing: New York, NY, USA, 2018; Volume 481, pp. 139–141. [Google Scholar]
- Fragoso ados Santos, H.; Duarte, G.A.S.; Rachid, C.T.d.C.; Chaloub, R.M.; Calderon, E.N.; Marangoni, L.F.d.B.; Bianchini, A.; Nudi, A.H.; Do Carmo, F.L.; van Elsas, J.D. Impact of Oil Spills on Coral Reefs Can Be Reduced by Bioremediation Using Probiotic Microbiota. Sci. Rep. 2015, 5, 1–11. [Google Scholar] [CrossRef]
- Sarlak, Z.; Khosravi-Darani, K.; Rouhi, M.; Garavand, F.; Mohammadi, R.; Sobhiyeh, M.R. Bioremediation of Organophosphorus Pesticides in Contaminated Foodstuffs Using Probiotics. Food Control 2021, 126, 108006. [Google Scholar] [CrossRef]
- Nybom, S.M.K.; Dziga, D.; Heikkilä, J.E.; Kull, T.P.J.; Salminen, S.J.; Meriluoto, J.A.O. Characterization of Microcystin-LR Removal Process in the Presence of Probiotic Bacteria. Toxicon 2012, 59, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Mirza Alizadeh, A.; Hosseini, H.; Mohseni, M.; Eskandari, S.; Sohrabvandi, S.; Hosseini, M.-J.; Tajabadi-Ebrahimi, M.; Mohammadi-Kamrood, M.; Nahavandi, S. Analytic and Chemometric Assessments of the Native Probiotic Bacteria and Inulin Effects on Bioremediation of Lead Salts. J. Sci. Food Agric. 2021, 101, 5142–5153. [Google Scholar] [CrossRef] [PubMed]
- Niderkorn, V.; Boudra, H.; Morgavi, D.P. Binding of Fusarium Mycotoxins by Fermentative Bacteria In Vitro. J. Appl. Microbiol. 2006, 101, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Sampaio Baptista, A.; Horii, J.; Antonia Calori-Domingues, M.; Micotti da Glória, E.; Mastrodi Salgado, J.; Roberto Vizioli, M. The Capacity of Manno-Oligosaccharides, Thermolysed Yeast and Active Yeast to Attenuate Aflatoxicosis. World J. Microbiol. Biotechnol. 2004, 20, 475–481. [Google Scholar] [CrossRef]
- Shetty, P.H.; Hald, B.; Jespersen, L. Surface Binding of Aflatoxin B1 by Saccharomyces Cerevisiae Strains with Potential Decontaminating Abilities in Indigenous Fermented Foods. Int. J. Food Microbiol. 2007, 113, 41–46. [Google Scholar] [CrossRef]



| Synbiotics | ||
|---|---|---|
| Probiotics | Prebiotics | Products |
| Lacticaseibacillus paracasei YIT 9029 (strain Shirota: LcS) Bifidobacterium breve YIT 12272 (BbrY) | GOS | Super Synbiotics LBG-P (Yakult Honsha Co., Ltd., Tokyo, Japan) |
| Streptococcus thermophilus Lacticaseibacillus rhamnosus Lactobacillus acidophilus B. infantis B. lactis | FOS + Ascorbic acid | Probiotical (Laboratoires Phacobel Belgium, Soheit-Tinlot, Belgium) |
| B. breve | Short chain scGOS Long chain lcFOS | Danone Nutricia Research, Utrecht, The Netherlands |
| Bacillus coagulans MTCC 5856 | Cranberry fiber | Lactocran (Sabinsa Corporation, Piscataway, NJ, USA) |
| B. lactis, B. breve, B. infantis, B. longum | XOS | PrebioMed™ XOS (designs for health, Suffield, CT, USA) |
| Mix of Bifidobacteria and Lactobacilli | Whole fruit Indian Pomegranate (Punica granatum) (>40% Polyphenolic + Phenolic Bioactives) | DS-01 (Seed Health, Los Angeles, CA, USA) |
| L. acidophilus Lacticaseibacillus casei, L. rhamnosus Lactiplantibacillus plantarum, S. thermophilus B. longum | Oat Bran (10% β-Glucan fiber) Organic Red Beetroot Inulin from Organic Chicory Root | Beta Glucan Synbiotic (BioImmersion Inc, Bellevue, WA, USA) |
| B. animalis, Enterococcus faecium, Limosilactobacillus reuteri Ligilactobacillus salivarius Pediococcus acidilactici | Inulin | PoultryStar® (ME BIOMIN GmbH, Niederösterreich, Austria) |
| Enterococcus faecium | FOS | Biomin® IMBO (ME BIOMIN GmbH, Niederösterreich, Austria) |
| L. acidophilus L. casei L. salivarius L. plantarum, L. rhamnosus Levilactobacillus brevis B. bifidum B. lactis S. thermophilus | Inulin | Synbiotic poultry (Vetafarm, Wagga Wagga, Australia) |
| Thermophysical Quantities | Meaning | Source |
|---|---|---|
| Tmax [°C] | Maximum temperature of main decomposition (first derivative or DTG main peak) | TGA |
| Vmax [Kmin−1] | Maximum rate of main decomposition (first derivative or DTG main peak) | TGA |
| Tm [°C] | Temperature of phase transition of main decomposition | DSC |
| ΔHm [J/g] | Enthalpy of phase transition of main decomposition (area of the curve) | DSC |
| R600 [%] | Black carbon and mineral compounds at the end of the temperature scan (600 °C) | TGA |
| Probiotic Formulations | Tmax | Vmax | Tm | ΔHm | R600 |
|---|---|---|---|---|---|
| [°C] | 100 × [min−1] | [°C] | [J/g] | [%] | |
| Mono-strains | |||||
| B. subtilis | 350.7 ± 0.7 | 3.93 ± 0.11 | 352.9 ± 0.9 | −23.0 ± 3.0 | 24.0 ± 1.1 |
| B. longum | 314.3 ± 0.1 | 7.04 ± 0.07 | 310.9 ± 0.4 | −5.1 ± 1.7 | 12.8 ± 0.3 |
| S. boulardii | 275.4 ± 0.3 | 2.60 ± 0.03 | 279.5 ± 0.4 | 33.3 ± 2.0 | 33.4 ± 0.3 |
| S. thermophilus | 264.6 ± 0.1 | 4.01 ± 0.03 | 273.2 ± 1.0 | 12.4 ± 1.2 | 32.7 ± 0.2 |
| L. rhamnosus GG | 228.2 ± 0.5 | 6.44 ± 0.06 | 228.1 ± 1.0 | 26.1 ± 4.0 | 24.5 ± 0.3 |
| L. bulgaricus | 165.2 ± 0.0 | 3.21 ± 0.04 | 165.6 ± 0.0 | −97.9 ± 1.3 | 32.2 ± 0.1 |
| Multi-strains VSL#3 | |||||
| Lot 606035 | 301.2 ± 0.2 | 2.97 ± 0.02 | 305.8 ± 1.7 | 31.8 ± 4.5 | 30.1 ± 0.0 |
| Lot 45752 | 294.1 ± 0.9 | 4.24 ± 0.03 | 290.0 ± 3.0 | 70.0 ± 8.3 | 28.4 ± 0.6 |
| Lot 10151198 | 206.9 ± 2.1 | 3.00 ± 0.01 | 196.4 ± 4.5 | −33.5 ± 2.8 | 32.9 ± 0.9 |
| Lot 3302E10 | 204.3 ± 3.0 | 3.13 ± 0.11 | 193.7 ± 4.6 | −41.2 ± 11.5 | 33.4 ± 0.2 |
| Species | Known Function | Reference |
|---|---|---|
| Bacillus subtilis | Solubilize soil (phosphorus), enhance nitrogen fixation, and produce siderophores that promote its growth and suppresses the growth of pathogens | [75] |
| Cytobacillus firmus | Nematode antagonist | [76] |
| Bacillus thuringiensis | Known pesticide activities | [77] |
| Bacillus mucilaginosus | Solubilize potassium from minerals in soil so that plants, such as food crops, are able to use it | [78] |
| Paenibacillus polymyxa | Fix nitrogen, making it available to plants | [79] |
| Bacillus cereus | Regenerate contaminated soils and promote mycorrhizae growth | [80] |
| Bacillus amyloliquefaciens | Prevent a series of bacterial pathogens in crops | [81] |
| Bacillus pumilus | Promote plant growth, fix nitrogen, and prevent the germination of several fungal pathogens on crop roots | [82] |
| Bacillus megaterium | Solubilizes phosphates, and promotes plant growth through signaling | [83] |
| Health Outcomes | Probiotic Strains | Prebiotics | Reference | |
|---|---|---|---|---|
| Gut Intestinal tract | Treatment of overweight and metabolic syndrome | L. casei PXN 37, L. rhamnosus PXN 54, S. thermophilus PXN 66, B. breve PXN 25, L. acidophilus PXN 35, B. longum PXN 30, L. bulgaricus PXN 39 | FOS | [133] |
| Treatment of IBS | Bacillus coagulans | FOS | [134] | |
| Acute diarrhea | L. acidophilus, L. rhamnosus, B. bifidum, B. longum, Enterococcus faecium | FOS | [135] | |
| Colorectal cancer | B. lactis | Resistant starch | [136] | |
| Kidney | Treatment of chronic kidney disease | L. casei, L. acidophilus, L. bulgaricus, L. rhamnosus, B. breve, B. longum, S. thermophilus | FOS | [137] |
| Liver | Treatment of non-alcoholic fatty liver disease Prevention of infections after liver transplant | B. longum L. acidophilus | Inulin HP | [138] |
| Lung | Reduction of viral respiratory infections in asthmatic children | L. casei, L. rhamnosus, S. thermophilus, B. breve, L. acidophilus, B. infantis, L. bulgaricus | FOS | [139] |
| Skin/derm | Treatment of atopic dermatitis | L. salivarius PM-A0006 | FOS | [140] |
| Brain | Improvement in mental health in hemodialysis patients | L. acidophilus strain T16, B. bifidum strain BIA-6, B. lactis strain BIA-7, B. longum strain BIA-8 | Equal mix of FOS, GOS and inulin | [141] |
| Ruminant Host | Pre-/Pro-/Synbiotics | Mode of Administration/Dose | Effect | Reference |
|---|---|---|---|---|
| Dairy cows | L. casei and L. plantarum | Combination of both in the feed (50 g/day) | Increases the milk production and the contents of milk immunoglobulin G, lactoferrin, lysozyme and lactoperoxidase | [165,166] |
| Propionibacterium spp. and S. cerevisiae | Oral administration, mixed in feed | Improves the feed conversion rate, milk production and dry matter intake | [162] | |
| S. cerevisiae | Oral administration, mixed in feed (0.2 g/day) | Improves the feed conversion rate, milk production and dry matter intake | [167] | |
| Fructo-oligosacchrides (FOSs) and Mannan-oligosaccharides (MOSs) | Oral administration | Provides specific bacteria with a competitive advantage in the gut. | [168] | |
| Enterococcus faecium + lactulose | Oral administration 109 colony forming units (probiotics) + 1–3% dry matter (prebiotics) | Decreases the ileal villus height, the depth of the crypts in the cecum, and the surface area of lymph follicles from Peyer’s patches | [169] | |
| Streptococcus faecium + Mannan-oligosaccharide (MOS) | Oral administration (0.6 kg/day) | Improves fecal consistency and reduced the fecal score of calves | [170] | |
| S. cerevisiae strain 1026 + Inulin | Oral administration (probiotic 5 g + prebiotic 6 g) | Impacts positively the development of morphological structures of digestive systems | [171] | |
| Goats | L. reuteri, L. alimentarius, Enterococcus faecium and B. bifidum | Oral administration, resuspended in milk (1 mL/two feeds per day) | Improves the microbial environment and intestinal health, in addition to the acid profile of milk, with an increase in unsaturated fatty acids, mainly linoleic, linolenic and conjugated linoleic acids, and a decrease in the atherogenic index | [159] |
| Inulin, fructo-oligosaccharide, galacto-oligosaccharide and xylo-oligosaccharide | 0.4 to 0.6% in milk | Antioxidant activity and promotes the development of functional goat milk | [172] | |
| Sheep | S. cerevisiae and two strains of rumen-derived Diutina rugosa | Oral administration (100 mL) | Stabilizes the ruminal pH, improves the richness of rumen microflora, relieves acidosis and inflammation, and prevents subacute ruminal acidosis | [173,174] |
| Propionibacterium P63, L. plantarum and L. rhamnosus | Intraruminal cannula (2 g/day) | Stabilizes the pH of the rumen and prevents acidosis | [173,174] | |
| Mannan-oligosaccharide and b-glucans | Intraruminal cannula | Additive effects on digestion and fermentation | [175] | |
| Cattle | Enterococcus faecium strain 26 and Clostridium butyricum strain Miyari | Oral administration | Reduces the ruminal pH and the concentration of lactic acid in the ruminal fluid, thus preventing acidosis | [159] |
| Cellooligosaccharide (CE), Mannan-oligosaccharides (MOSs) and fructo-oligosaccharides (FOSs) such as Galactosyl-lactose in combination with spray-dried bovine serum | Oral administration of supplemented milk replacer | Reduces the incidence and severity of enteric disease and modulate the intestinal bacterial community in calves | [165] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kouhounde, S.; Adéoti, K.; Mounir, M.; Giusti, A.; Refinetti, P.; Otu, A.; Effa, E.; Ebenso, B.; Adetimirin, V.O.; Barceló, J.M.; et al. Applications of Probiotic-Based Multi-Components to Human, Animal and Ecosystem Health: Concepts, Methodologies, and Action Mechanisms. Microorganisms 2022, 10, 1700. https://doi.org/10.3390/microorganisms10091700
Kouhounde S, Adéoti K, Mounir M, Giusti A, Refinetti P, Otu A, Effa E, Ebenso B, Adetimirin VO, Barceló JM, et al. Applications of Probiotic-Based Multi-Components to Human, Animal and Ecosystem Health: Concepts, Methodologies, and Action Mechanisms. Microorganisms. 2022; 10(9):1700. https://doi.org/10.3390/microorganisms10091700
Chicago/Turabian StyleKouhounde, Sonagnon, Kifouli Adéoti, Majid Mounir, Alessandro Giusti, Paulo Refinetti, Akaninyene Otu, Emmanuel Effa, Bassey Ebenso, Victor O. Adetimirin, Josep Mercader Barceló, and et al. 2022. "Applications of Probiotic-Based Multi-Components to Human, Animal and Ecosystem Health: Concepts, Methodologies, and Action Mechanisms" Microorganisms 10, no. 9: 1700. https://doi.org/10.3390/microorganisms10091700
APA StyleKouhounde, S., Adéoti, K., Mounir, M., Giusti, A., Refinetti, P., Otu, A., Effa, E., Ebenso, B., Adetimirin, V. O., Barceló, J. M., Thiare, O., Rabetafika, H. N., & Razafindralambo, H. L. (2022). Applications of Probiotic-Based Multi-Components to Human, Animal and Ecosystem Health: Concepts, Methodologies, and Action Mechanisms. Microorganisms, 10(9), 1700. https://doi.org/10.3390/microorganisms10091700








