B Vitamins and Their Roles in Gut Health
Abstract
1. Introduction
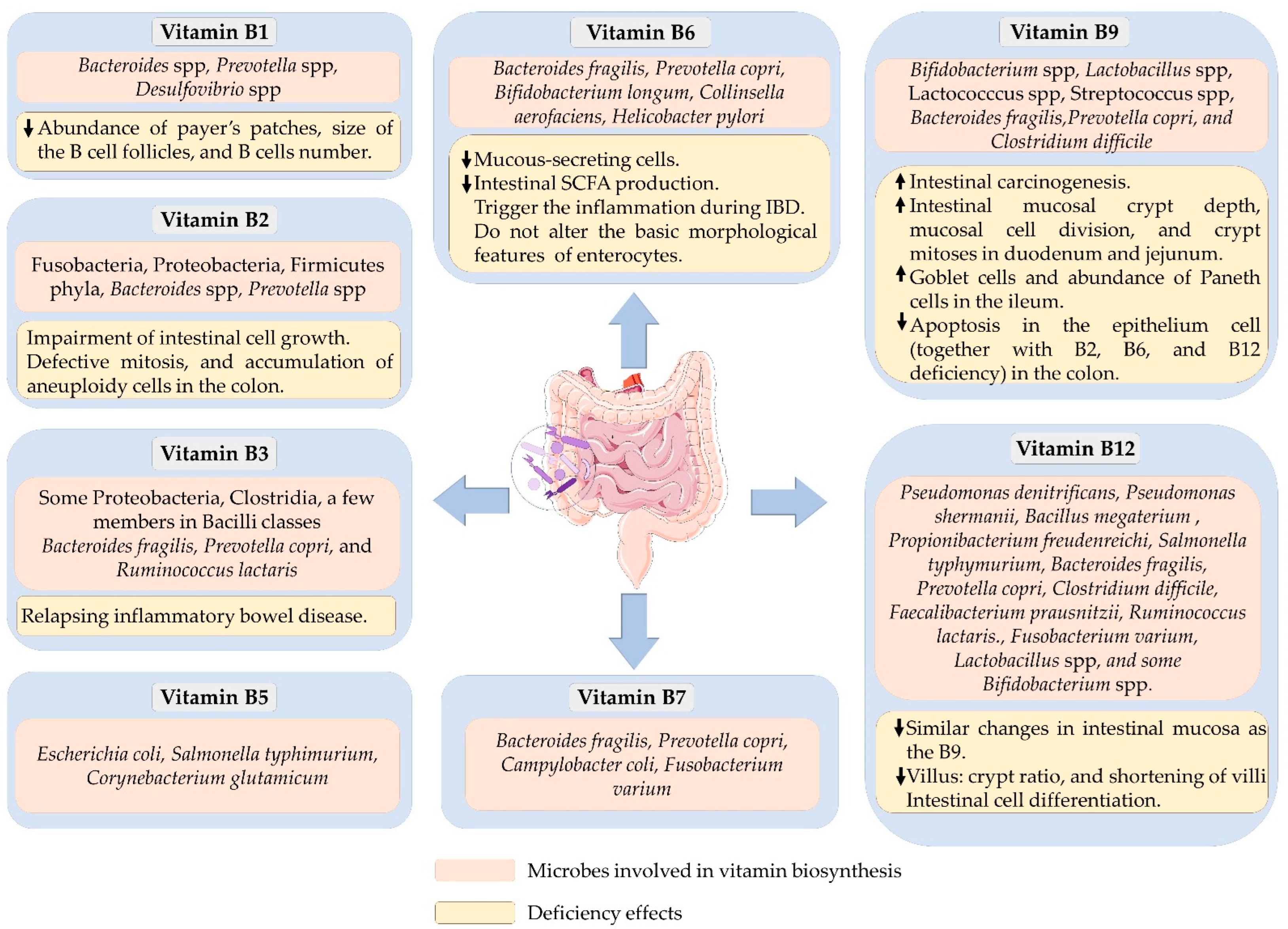
2. Vitamin B1/Thiamin
3. Vitamin B2/Riboflavin
4. Vitamin B3/Niacin
5. Vitamin B5/Pantothenic Acid
6. Vitamin B6/Pyridoxine
7. Vitamin B7/Biotin
8. Vitamin B9/Folate
9. Vitamin B12/Cobalamin
10. Factors Affecting Gut Microbial B-Vitamin Synthesis
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kennedy, D.O. B Vitamins and the Brain: Mechanisms, Dose and Efficacy—A Review. Nutrients 2016, 8, 68. [Google Scholar] [CrossRef]
- Depeint, F.; Bruce, W.R.; Shangari, N.; Mehta, R.; O’Brien, P.J. Mitochondrial function and toxicity: Role of the B vitamin family on mitochondrial energy metabolism. Chem. Biol. Interact. 2006, 163, 94–112. [Google Scholar] [CrossRef]
- Rahman, S.; Baumgartner, M. B Vitamins: Small molecules, big effects. J. Inherit. Metab. Dis. 2019, 42, 579–580. [Google Scholar] [CrossRef]
- Murphy, M.M.; Guéant, J.-L. B vitamins and one carbon metabolism micronutrients in health and disease. Biochimie 2020, 173, 1–2. [Google Scholar] [CrossRef]
- Peterson, C.T.; Rodionov, D.A.; Osterman, A.L.; Peterson, S.N. B Vitamins and Their Role in Immune Regulation and Cancer. Nutrients 2020, 12, 3380. [Google Scholar] [CrossRef]
- Martinez, D.L.; Tsuchiya, Y.; Gout, I. Coenzyme A biosynthetic machinery in mammalian cells. Biochem. Soc. Trans. 2014, 42, 1112–1117. [Google Scholar] [CrossRef]
- Spinneker, A.; Sola, R.; Lemmen, V.; Castillo, M.J.; Pietrzik, K.; González-Gross, M. Vitamin B6 status, deficiency and its consequences--an overview. Nutr. Hosp. 2007, 22, 7–24. [Google Scholar]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Marchesi, J.R. Prokaryotic and Eukaryotic Diversity of the Human Gut. Adv. Appl. Microbiol. 2010, 72, 43–62. [Google Scholar] [CrossRef]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef]
- Bäumler, A.J.; Sperandio, V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature 2016, 535, 85–93. [Google Scholar] [CrossRef]
- Den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.-J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef]
- Gensollen, T.; Iyer, S.S.; Kasper, D.L.; Blumberg, R.S. How colonization by microbiota in early life shapes the immune system. Science 2016, 352, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Dogra, S.K.; Doré, J.; Damak, S. Gut Microbiota Resilience: Definition, Link to Health and Strategies for Intervention. Front. Microbiol. 2020, 11, 572921. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, P.W.; Claesson, M. Gut microbiota: Changes throughout the lifespan from infancy to elderly. Int. Dairy J. 2010, 20, 281–291. [Google Scholar] [CrossRef]
- Biasucci, G.; Benenati, B.; Morelli, L.; Bessi, E.; Boehm, G. Cesarean Delivery May Affect the Early Biodiversity of Intestinal Bacteria. J. Nutr. 2008, 138, 1796S–1800S. [Google Scholar] [CrossRef]
- Neu, J.; Rushing, J. Cesarean Versus Vaginal Delivery: Long-term Infant Outcomes and the Hygiene Hypothesis. Clin. Perinatol. 2011, 38, 321–331. [Google Scholar] [CrossRef]
- Odamaki, T.; Kato, K.; Sugahara, H.; Hashikura, N.; Takahashi, S.; Xiao, J.-Z.; Abe, F.; Osawa, R. Age-Related changes in gut microbiota composition from newborn to centenarian: A cross-sectional study. BMC Microbiol. 2016, 16, 90. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.M.; Aronoff, D.M. The influence of non-steroidal anti-inflammatory drugs on the gut microbiome. Clin. Microbiol. Infect. 2016, 22, 178.e1–178.e9. [Google Scholar] [CrossRef]
- Jernberg, C.; Löfmark, S.; Edlund, C.; Jansson, J.K. Long-Term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology 2010, 156, 3216–3223. [Google Scholar] [CrossRef]
- Heiman, M.L.; Greenway, F.L. A healthy gastrointestinal microbiome is dependent on dietary diversity. Mol. Metab. 2016, 5, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Uebanso, T.; Shimohata, T.; Mawatari, K.; Takahashi, A. Functional Roles of B-Vitamins in the Gut and Gut Microbiome. Mol. Nutr. Food Res. 2020, 64, 2000426. [Google Scholar] [CrossRef]
- Manzetti, S.; Zhang, J.; van der Spoel, D. Thiamin function, metabolism, uptake, and transport. Biochemistry 2014, 53, 821–835. [Google Scholar] [CrossRef]
- Sriram, K.; Manzanares, W.; Joseph, K. Thiamine in Nutrition Therapy. Nutr. Clin. Pract. 2012, 27, 41–50. [Google Scholar] [CrossRef]
- Said, H.M. Recent advances in transport of water-soluble vitamins in organs of the digestive system: A focus on the colon and the pancreas. Am. J. Physiol. Liver Physiol. 2013, 305, G601–G610. [Google Scholar] [CrossRef] [PubMed]
- Costliow, Z.A.; Degnan, P.H. Thiamine Acquisition Strategies Impact Metabolism and Competition in the Gut Microbe Bacteroides thetaiotaomicron. mSystems 2017, 2, e00116-17. [Google Scholar] [CrossRef]
- Said, H.M.; Ortiz, A.; Subramanian, V.S.; Neufeld, E.J.; Moyer, M.P.; Dudeja, P.K. Mechanism of thiamine uptake by human colonocytes: Studies with cultured colonic epithelial cell line NCM460. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G144–G150. [Google Scholar] [CrossRef] [PubMed]
- Nabokina, S.M.; Inoue, K.; Subramanian, V.S.; Valle, J.E.; Yuasa, H.; Said, H.M. Molecular Identification and Functional Characterization of the Human Colonic Thiamine Pyrophosphate Transporter. J. Biol. Chem. 2014, 289, 4405–4416. [Google Scholar] [CrossRef] [PubMed]
- Mathis, D.; Shoelson, S.E. Immunometabolism: An emerging frontier. Nat. Rev. Immunol. 2011, 11, 81–83. [Google Scholar] [CrossRef]
- Kunisawa, J.; Sugiura, Y.; Wake, T.; Nagatake, T.; Suzuki, H.; Nagasawa, R.; Shikata, S.; Honda, K.; Hashimoto, E.; Suzuki, Y.; et al. Mode of Bioenergetic Metabolism during B Cell Differentiation in the Intestine Determines the Distinct Requirement for Vitamin B 1. Cell Rep. 2015, 13, 122–131. [Google Scholar] [CrossRef]
- Pinto, J.T.; Zempleni, J. Riboflavin. Adv. Nutr. 2016, 7, 973–975. [Google Scholar] [CrossRef]
- Sundaram, U. Regulation of intestinal vitamin B(2) absorption. Focus on “Riboflavin uptake by human-derived colonic epithelial NCM460 cells”. Am. J. Physiol. Cell Physiol. 2000, 278, C268–C269. [Google Scholar] [CrossRef] [PubMed]
- Steinert, R.E.; Sadaghian Sadabad, M.; Harmsen, H.J.; Weber, P. The prebiotic concept and human health: A changing landscape with riboflavin as a novel prebiotic candidate? Eur. J. Clin. Nutr. 2016, 70, 1348–1353. [Google Scholar] [CrossRef] [PubMed]
- Magnusdottir, S.; Ravcheev, D.; De Crécy-Lagard, V.; Thiele, I. Systematic genome assessment of B-vitamin biosynthesis suggests co-operation among gut microbes. Front. Genet. 2015, 6, 148. [Google Scholar] [CrossRef]
- Thakur, K.; Tomar, S.K.; De, S. Lactic acid bacteria as a cell factory for riboflavin production. Microb. Biotechnol. 2016, 9, 441–451. [Google Scholar] [CrossRef]
- García-Angulo, V.A. Overlapping riboflavin supply pathways in bacteria. Crit. Rev. Microbiol. 2017, 43, 196–209. [Google Scholar] [CrossRef]
- Yates, C.; Evans, G.; Pearson, T.; Powers, H. Absence of luminal riboflavin disturbs early postnatal development of the gastrointestinal tract. Dig. Dis. Sci. 2003, 48, 1159–1164. [Google Scholar] [CrossRef]
- Yates, C.A.; Evans, G.S.; Powers, H.J. Riboflavin deficiency: Early effects on post-weaning development of the duodenum in rats. Br. J. Nutr. 2001, 86, 593–599. [Google Scholar] [CrossRef]
- Lee, E.-S.; Corfe, B.M.; Powers, H.J. Riboflavin depletion of intestinal cells in vitro leads to impaired energy generation and enhanced oxidative stress. Eur. J. Nutr. 2013, 52, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.A.; Rumsey, R.D.; Powers, H.J. An investigation into the reversibility of the morphological and cytokinetic changes seen in the small intestine of riboflavin deficient rats. Gut 1996, 39, 220–225. [Google Scholar] [CrossRef]
- Williams, E.A.; Rumsey, R.D.; Powers, H.J. Cytokinetic and structural responses of the rat small intestine to riboflavin depletion. Br. J. Nutr. 1996, 75, 315–324. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Williams, E.A.; Powers, H.J.; Rumsey, R.D.E. Morphological changes in the rat small intestine in response to riboflavin depletion. Br. J. Nutr. 1995, 73, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Nakano, E.; Mushtaq, S.; Heath, P.R.; Lee, E.-S.; Bury, J.P.; Riley, S.A.; Powers, H.J.; Corfe, B.M. Riboflavin Depletion Impairs Cell Proliferation in Adult Human Duodenum: Identification of Potential Effectors. Am. J. Dig. Dis. 2010, 56, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- Carrothers, J.M.; York, M.A.; Brooker, S.L.; Lackey, K.A.; Williams, J.E.; Shafii, B.; Price, W.J.; Settles, M.L.; McGuire, M.A.; McGuire, M.K. Fecal Microbial Community Structure Is Stable over Time and Related to Variation in Macronutrient and Micronutrient Intakes in Lactating Women. J. Nutr. 2015, 145, 2379–2388. [Google Scholar] [CrossRef]
- Qi, Y.; Lohman, J.; Bratlie, K.M.; Peroutka-Bigus, N.; Bellaire, B.; Wannemuehler, M.; Yoon, K.; Barrett, T.A.; Wang, Q. Vitamin C and B 3 as new biomaterials to alter intestinal stem cells. J. Biomed. Mater. Res. Part A 2019, 107, 1886–1897. [Google Scholar] [CrossRef]
- Gazzaniga, F.; Stebbins, R.; Chang, S.Z.; McPeek, M.A.; Brenner, C. Microbial NAD Metabolism: Lessons from Comparative Genomics. Microbiol. Mol. Biol. Rev. 2009, 73, 529–541. [Google Scholar] [CrossRef]
- Kurnasov, O.; Goral, V.; Colabroy, K.; Gerdes, S.; Anantha, S.; Osterman, A.; Begley, T.P. NAD Biosynthesis: Identification of the Tryptophan to Quinolinate Pathway in Bacteria. Chem. Biol. 2003, 10, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, Y.; Morishita, T.; Mutai, M. Comparative Studies on Synthesis of Water-soluble Vitamins among Human Species of Bifidobacteria. Agric. Biol. Chem. 1985, 49, 13–19. [Google Scholar]
- Rossolillo, P.; Marinoni, I.; Galli, E.; Colosimo, A.; Albertini, A.M. YrxA Is the Transcriptional Regulator That Represses De Novo NAD Biosynthesis in Bacillus subtilis. J. Bacteriol. 2005, 187, 7155–7160. [Google Scholar] [CrossRef]
- Rodionov, D.A.; Li, X.; Rodionova, I.A.; Yang, C.; Sorci, L.; Dervyn, E.; Martynowski, D.; Zhang, H.; Gelfand, M.; Osterman, A.L. Transcriptional regulation of NAD metabolism in bacteria: Genomic reconstruction of NiaR (YrxA) regulon. Nucleic Acids Res. 2008, 36, 2032–2046. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.S.; Subramanian, V.S.; Kapadia, R.; Kashyap, M.L.; Said, H.M. Mammalian colonocytes possess a carrier-mediated mechanism for uptake of vitamin B3 (niacin): Studies utilizing human and mouse colonic preparations. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G207–G213. [Google Scholar] [CrossRef] [PubMed]
- Fangmann, D.; Theismann, E.-M.; Türk, K.; Schulte, D.M.; Relling, I.; Hartmann, K.; Keppler, J.K.; Knipp, J.-R.; Rehman, A.; Heinsen, F.-A.; et al. Targeted Microbiome Intervention by Microencapsulated Delayed-Release Niacin Beneficially Affects Insulin Sensitivity in Humans. Diabetes Care 2017, 41, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kong, D.; Wang, Q.; Wu, W.; Tang, Y.; Bai, T.; Guo, L.; Wei, L.; Zhang, Q.; Yu, Y.; et al. Niacin ameliorates ulcerative colitis via prostaglandin D 2 -mediated D prostanoid receptor 1 activation. EMBO Mol. Med. 2017, 9, 571–588. [Google Scholar] [CrossRef]
- Santoru, M.L.; Piras, C.; Murgia, F.; Spada, M.; Tronci, L.; Leoni, V.P.; Serreli, G.; Deiana, M.; Atzori, L. Modulatory Effect of Nicotinic Acid on the Metabolism of Caco-2 Cells Exposed to IL-1β and LPS. Metabolites 2020, 10, 204. [Google Scholar] [CrossRef]
- Filippi, J.; Al-Jaouni, R.; Wiroth, J.B.; Hébuterne, X.; Schneider, S.M. Nutritional deficiencies in patients with Crohn’s disease in remission. Inflamm. Bowel Dis. 2006, 12, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Ragaller, V.; Lebzien, P.; Südekum, K.H.; Hüther, L.; Flachowsky, G. Pantothenic acid in ruminant nutrition: A review. J. Anim. Physiol. Anim. Nutr. 2011, 95, 6–16. [Google Scholar] [CrossRef]
- Hayflick, S.J. Defective pantothenate metabolism and neurodegeneration. Biochem. Soc. Trans. 2014, 42, 1063–1068. [Google Scholar] [CrossRef]
- Leonardi, R.; Jackowski, S. Biosynthesis of Pantothenic Acid and Coenzyme A. EcoSal Plus 2007, 2, 2. [Google Scholar] [CrossRef]
- Begley, T.P.; Kinsland, C.; Strauss, E. The biosynthesis of coenzyme a in bacteria. Vitam. Horm. 2001, 61, 157–171. [Google Scholar] [CrossRef]
- Tahiliani, A.G.; Beinlich, C.J. Pantothenic Acid in Health and Disease. Vitam. Horm. 1991, 46, 165–228. [Google Scholar] [CrossRef]
- Frederick, C.; Neidhardt, J.L.I.; Magasanik, K.B.; Brooks, L.; Moselio, S.; Edwin, U.H. Escherichia coli and Salmonella typhimurium. Cell. Mol. Biol. 1988, 63, 463–464. [Google Scholar]
- Epelbaum, S.; LaRossa, R.A.; VanDyk, T.K.; Elkayam, T.; Chipman, D.M.; Barak, Z. Branched-Chain amino acid biosynthesis in Salmonella typhimurium: A quantitative analysis. J. Bacteriol. 1998, 180, 4056–4067. [Google Scholar] [CrossRef]
- Yao, C.; Chou, J.; Wang, T.; Zhao, H.; Zhang, B. Pantothenic Acid, Vitamin C, and Biotin Play Important Roles in the Growth of Lactobacillus helveticus. Front. Microbiol. 2018, 9, 1194. [Google Scholar] [CrossRef]
- Parra, M.; Stahl, S.; Hellmann, H. Vitamin B6 and Its Role in Cell Metabolism and Physiology. Cells 2018, 7, 84. [Google Scholar] [CrossRef]
- Mooney, S.; Leuendorf, J.-E.; Hendrickson, C.; Hellmann, H. Vitamin B6: A Long Known Compound of Surprising Complexity. Molecules 2009, 14, 329–351. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, S.R.; Singh, S.K.; Roy, S.; Sengupta, D.N. An insight into the sequential, structural and phylogenetic properties of banana 1-aminocyclopropane-1-carboxylate synthase 1 and study of its interaction with pyridoxal-5’-phosphate and aminoethoxyvinylglycine. J. Biosci. 2010, 35, 281–294. [Google Scholar] [CrossRef]
- Plecko, B.; Stöckler, S. Vitamin B6 dependent seizures. Can. J. Neurol. Sci. 2009, 36, S73–S77. [Google Scholar] [PubMed]
- Geng, M.-Y.; Saito, H.; Katsuki, H. Effects of vitamin B6 and its related compounds on survival of cultured brain neurons. Neurosci. Res. 1995, 24, 61–65. [Google Scholar] [CrossRef]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Mayengbam, S.; Chleilat, F.; Reimer, R.A. Dietary Vitamin B6 Deficiency Impairs Gut Microbiota and Host and Microbial Metabolites in Rats. Biomedicines 2020, 8, 469. [Google Scholar] [CrossRef] [PubMed]
- Vitellio, P.; Celano, G.; Bonfrate, L.; Gobbetti, M.; Portincasa, P.; De Angelis, M. Effects of Bifidobacterium longum and Lactobacillus rhamnosus on Gut Microbiota in Patients with Lactose Intolerance and Persisting Functional Gastrointestinal Symptoms: A Randomised, Double-Blind, Cross-Over Study. Nutrients 2019, 11, 886. [Google Scholar] [CrossRef] [PubMed]
- Ligaarden, S.C.; Farup, P. Low intake of vitamin B6 is associated with irritable bowel syndrome symptoms. Nutr. Res. 2011, 31, 356–361. [Google Scholar] [CrossRef]
- Yin, L.; Li, J.; Wang, H.; Yi, Z.; Wang, L.; Zhang, S.; Li, X.; Wang, Q.; Li, J.; Yang, H.; et al. Effects of vitamin B6 on the growth performance, intestinal morphology, and gene expression in weaned piglets that are fed a low-protein diet1. J. Anim. Sci. 2020, 98, skaa022. [Google Scholar] [CrossRef]
- Javanmardi, S.; Tavabe, K.R.; Rosentrater, K.A.; Solgi, M.; Bahadori, R. Effects of different levels of vitamin B6 in tank water on the Nile tilapia (Oreochromis niloticus): Growth performance, blood biochemical parameters, intestine and liver histology, and intestinal enzyme activity. Fish Physiol. Biochem. 2020, 46, 1909–1920. [Google Scholar] [CrossRef]
- Matyaszczyk, M.; Karczmarewicz, E.; Czarnowska, E.; Reynolds, R.D.; Lorenc, R.S. Vitamin B-6 deficiency alters rat enterocyte calcium homeostasis but not duodenal transport. J. Nutr. 1993, 123, 204–215. [Google Scholar] [PubMed]
- Tadi, K.G.B.P. Biotin; StatPearls: Tampa, FL, USA, 2021. [Google Scholar]
- Zempleni, J.; Wijeratne, S.S.; Hassan, Y.I. Biotin. Biofactors 2009, 35, 36–46. [Google Scholar] [CrossRef]
- Zempleni, J. Uptake, Localization, and Noncarboxylase Roles of Biotin. Annu. Rev. Nutr. 2005, 25, 175–196. [Google Scholar] [CrossRef]
- Kothapalli, N.; Camporeale, G.; Kueh, A.; Chew, Y.C.; Oommen, A.M.; Griffin, J.B.; Zempleni, J. Biological functions of biotinylated histones. J. Nutr. Biochem. 2005, 16, 446–448. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Agrawal, S.; Agrawal, A.; Said, H.M. Biotin deficiency enhances the inflammatory response of human dendritic cells. Am. J. Physiol. Physiol. 2016, 311, C386–C391. [Google Scholar] [CrossRef] [PubMed]
- Elahi, A.; Sabui, S.; Narasappa, N.N.; Agrawal, S.; Lambrecht, N.W.; Agrawal, A.; Said, H.M. Biotin Deficiency Induces Th1- and Th17-Mediated Proinflammatory Responses in Human CD4+ T Lymphocytes via Activation of the mTOR Signaling Pathway. J. Immunol. 2018, 200, 2563–2570. [Google Scholar] [CrossRef]
- Bi, H.; Zhu, L.; Jia, J.; Cronan, J.E. A Biotin Biosynthesis Gene Restricted to Helicobacter. Sci. Rep. 2016, 6, 21162. [Google Scholar] [CrossRef]
- Entcheva, P.; Phillips, D.A.; Streit, W.R. Functional Analysis of Sinorhizobium meliloti Genes Involved in Biotin Synthesis and Transport. Appl. Environ. Microbiol. 2002, 68, 2843–2848. [Google Scholar] [CrossRef]
- Hayashi, A.; Mikami, Y.; Miyamoto, K.; Kamada, N.; Sato, T.; Mizuno, S.; Naganuma, M.; Teratani, T.; Aoki, R.; Fukuda, S.; et al. Intestinal Dysbiosis and Biotin Deprivation Induce Alopecia through Overgrowth of Lactobacillus murinus in Mice. Cell Rep. 2017, 20, 1513–1524. [Google Scholar] [CrossRef]
- Iyer, R.; Tomar, S. Folate: A Functional Food Constituent. J. Food Sci. 2009, 74, R114–R122. [Google Scholar] [CrossRef]
- Da Silva, A.V.A.; Oliveira, S.B.D.C.; Di Rienzi, S.C.; Brown-Steinke, K.; Dehan, L.M.; Rood, J.K.; Carreira, V.S.; Le, H.; Maier, E.A.; Betz, K.J.; et al. Murine Methyl Donor Deficiency Impairs Early Growth in Association with Dysmorphic Small Intestinal Crypts and Reduced Gut Microbial Community Diversity. Curr. Dev. Nutr. 2019, 3, nzy070. [Google Scholar] [CrossRef]
- Stover, P.J. Physiology of folate and vitamin B12 in health and disease. Nutr. Rev. 2004, 62, S3–S12, discussion S13. [Google Scholar] [CrossRef]
- Gazzali, A.M.; Lobry, M.; Colombeau, L.; Acherar, S.; Azaïs, H.; Mordon, S.; Arnoux, P.; Baros, F.; Vanderesse, R.; Frochot, C. Stability of folic acid under several parameters. Eur. J. Pharm. Sci. 2016, 93, 419–430. [Google Scholar] [CrossRef]
- Courtemanche, C.; Elson-Schwab, I.; Mashiyama, S.T.; Kerry, N.; Ames, B.N. Folate Deficiency Inhibits the Proliferation of Primary Human CD8+T Lymphocytes In Vitro. J. Immunol. 2004, 173, 3186–3192. [Google Scholar] [CrossRef]
- Troen, A.M.; Mitchell, B.; Sorensen, B.; Wener, M.H.; Johnston, A.; Wood, B.; Selhub, J.; McTiernan, A.; Yasui, Y.; Oral, E.; et al. Unmetabolized Folic Acid in Plasma Is Associated with Reduced Natural Killer Cell Cytotoxicity among Postmenopausal Women. J. Nutr. 2006, 136, 189–194. [Google Scholar] [CrossRef]
- Bermingham, A.; Derrick, J.-P. The folic acid biosynthesis pathway in bacteria: Evaluation of potential for antibacterial drug discovery. BioEssays 2002, 24, 637–648. [Google Scholar] [CrossRef]
- Hanson, A.D.; Gregory, J.F., III. Synthesis and turnover of folates in plants. Curr. Opin. Plant Biol. 2002, 5, 244–249. [Google Scholar] [CrossRef]
- White, R.H. Analysis and characterization of the folates in the nonmethanogenic archaebacteria. J. Bacteriol. 1988, 170, 4608–4612. [Google Scholar] [CrossRef]
- Levin, I.; Giladi, M.; Altman-Price, N.; Ortenberg, R.; Mevarech, M. An alternative pathway for reduced folate biosynthesis in bacteria and halophilic archaea. Mol. Microbiol. 2004, 54, 1307–1318. [Google Scholar] [CrossRef] [PubMed]
- de Crécy-Lagard, V.; El Yacoubi, B.; de la Garza, R.D.; Noiriel, A.; Hanson, A.D. Comparative genomics of bacterial and plant folate synthesis and salvage: Predictions and validations. BMC Genom. 2007, 8, 245. [Google Scholar] [CrossRef]
- Crittenden, R.G.; Martinez, N.R.; Playne, M.J. Synthesis and utilisation of folate by yoghurt starter cultures and probiotic bacteria. Int. J. Food Microbiol. 2003, 80, 217–222. [Google Scholar] [CrossRef]
- Rossi, M.; Amaretti, A.; Raimondi, S. Folate Production by Probiotic Bacteria. Nutrients 2011, 3, 118–134. [Google Scholar] [CrossRef]
- Ventura, M.; Canchaya, C.; Tauch, A.; Chandra, G.; Fitzgerald, G.F.; Chater, K.F.; van Sinderen, D. Genomics of Actinobacteria: Tracing the Evolutionary History of an Ancient Phylum. Microbiol. Mol. Biol. Rev. 2007, 71, 495–548. [Google Scholar] [CrossRef] [PubMed]
- Ventura, M.; Turroni, F.; Zomer, A.; Foroni, E.; Giubellini, V.; Bottacini, F.; Canchaya, C.; Claesson, M.; He, F.; Mantzourani, M.; et al. The Bifidobacterium dentium Bd1 Genome Sequence Reflects Its Genetic Adaptation to the Human Oral Cavity. PLoS Genet. 2009, 5, e1000785. [Google Scholar] [CrossRef]
- Makarova, K.S.; Koonin, E.V. Evolutionary Genomics of Lactic Acid Bacteria. J. Bacteriol. 2007, 189, 1199–1208. [Google Scholar] [CrossRef]
- Klipstein, F.A.; Lipton, S.D.; Schenk, E.A. Folate deficiency of the intestinal mucosa. Am. J. Clin. Nutr. 1973, 26, 728–737. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.; Wagner, C.; Schenker, S. Malabsorption of Thiamin in Folate-deficient Rats. J. Nutr. 1974, 104, 1024–1032. [Google Scholar] [CrossRef]
- Kim, Y.; Shirwadkar, S.; Choi, S.; Puchyr, M.; Wang, Y.; Mason, J.B. Effects of dietary folate on DNA strand breaks within mutation-prone exons of the p53 gene in rat colon. Gastroenterology 2000, 119, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Pufulete, M.; Al-Ghnaniem, R.; Leather, A.J.; Appleby, P.; Gout, S.; Terry, C.; Emery, P.W.; Sanders, T.A. Folate status, genomic DNA hypomethylation, and risk of colorectal adenoma and cancer: A case control study. Gastroenterology 2003, 124, 1240–1248. [Google Scholar] [CrossRef]
- Liu, Z.; Choi, S.-W.; Crott, J.W.; Keyes, M.K.; Jang, H.; Smith, D.E.; Kim, M.; Laird, P.W.; Bronson, R.; Mason, J.B. Mild Depletion of Dietary Folate Combined with Other B Vitamins Alters Multiple Components of the Wnt Pathway in Mouse Colon. J. Nutr. 2007, 137, 2701–2708. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Watanabe, F. Vitamin B12 Sources and Bioavailability. Exp. Biol. Med. 2007, 232, 1266–1274. [Google Scholar] [CrossRef]
- Scott, J.M. Bioavailability of vitamin B12. Eur. J. Clin. Nutr. 1997, 51 (Suppl. 1), S49–S53. [Google Scholar]
- Coates, P.M. Encyclopedia of Dietary Supplements; Dietary Supplements; Marcel Dekker: New York, NY, USA, 2005. [Google Scholar]
- Marriott, B.P.; Birt, D.F.; Stalling, V.A.; Yates, A.A. Present Knowledge in Nutrition, 11th ed.; Academic Press: Cambridge, MA, USA, 2020; p. 678. [Google Scholar]
- Thiamin, R. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academy Press: Washington, DC, USA, 1998. [Google Scholar]
- Ross, A.C.; Caballero, B.; Cousins, R.J.; Tucker, K.L. (Eds.) Modern Nutrition in Health and Disease, 11th ed.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Allen, L.H. Vitamin B-12. Adv. Nutr. 2012, 3, 54–55. [Google Scholar] [CrossRef]
- Degnan, P.H.; Barry, N.A.; Mok, K.C.; Taga, M.E.; Goodman, A.L. Human Gut Microbes Use Multiple Transporters to Distinguish Vitamin B12 Analogs and Compete in the Gut. Cell Host Microbe 2014, 15, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.J.; Alexeev, E.E.; Farb, L.; Vickery, T.W.; Zheng, L.; Eric, L.C.; Kitzenberg, D.A.; Battista, K.D.; Kominsky, D.J.; Robertson, C.E.; et al. Oral vitamin B12 supplement is delivered to the distal gut, altering the corrinoid profile and selectively depleting Bacteroides in C57BL/6 mice. Gut Microbes 2019, 10, 654–662. [Google Scholar] [CrossRef]
- Masuda, M.; Ide, M.; Utsumi, H.; Niiro, T.; Shimamura, Y.; Murata, M. Production Potency of Folate, Vitamin B12, and Thiamine by Lactic Acid Bacteria Isolated from Japanese Pickles. Biosci. Biotechnol. Biochem. 2012, 76, 2061–2067. [Google Scholar] [CrossRef]
- Gu, Q.; Zhang, C.; Song, D.; Li, P.; Zhu, X. Enhancing vitamin B12 content in soy-yogurt by Lactobacillus reuteri. Int. J. Food Microbiol. 2015, 206, 56–59. [Google Scholar] [CrossRef]
- Piwowarek, K.; Lipińska, E.; Hać-Szymańczuk, E.; Kieliszek, M.; Ścibisz, I. Propionibacterium spp.—source of propionic acid, vitamin B12, and other metabolites important for the industry. Appl. Microbiol. Biotechnol. 2018, 102, 515–538. [Google Scholar] [CrossRef]
- Lee, J.-H.; O’Sullivan, J.D. Genomic Insights into Bifidobacteria. Microbiol. Mol. Biol. Rev. 2010, 74, 378–416. [Google Scholar] [CrossRef]
- Song, H.; Yoo, Y.; Hwang, J.; Na, Y.-C.; Kim, H.S. Faecalibacterium prausnitzii subspecies–level dysbiosis in the human gut microbiome underlying atopic dermatitis. J. Allergy Clin. Immunol. 2015, 137, 852–860. [Google Scholar] [CrossRef]
- Kleerebezem, M.; Hugenholtz, J. Metabolic pathway engineering in lactic acid bacteria. Curr. Opin. Biotechnol. 2003, 14, 232–237. [Google Scholar] [CrossRef]
- Moore, S.; Warren, M.J. The anaerobic biosynthesis of vitamin B12. Biochem. Soc. Trans. 2012, 40, 581–586. [Google Scholar] [CrossRef]
- Benight, N.M.; Stoll, B.; Chacko, S.; da Silva, V.R.; Marini, J.C.; Gregory, J.F.; Stabler, S.P.; Burrin, D.G. B-Vitamin deficiency is protective against DSS-induced colitis in mice. Am. J. Physiol. Liver Physiol. 2011, 301, G249–G259. [Google Scholar] [CrossRef]
- Bressenot, A.; Pooya, S.; Bossenmeyer-Pourie, C.; Gauchotte, G.; Germain, A.; Chevaux, J.-B.; Coste, F.; Vignaud, J.-M.; Guéant, J.-L.; Peyrin-Biroulet, L. Methyl donor deficiency affects small-intestinal differentiation and barrier function in rats. Br. J. Nutr. 2013, 109, 667–677. [Google Scholar] [CrossRef]
- Berg, N.O.; Dahlqvist, A.; Lindberg, T.; Lindstrand, K.; Nordén, Å. Morphology, Dipeptidases and Disaccharidases of Small Intestinal Mucosa in Vitamin B12 and Folic Acid Deficiency. Scand. J. Haematol. 2009, 9, 167–173. [Google Scholar] [CrossRef]
- Lurz, E.; Horne, R.G.; Määttänen, P.; Wu, R.Y.; Botts, S.R.; Li, B.; Rossi, L.; Johnson-Henry, K.C.; Pierro, A.; Surette, M.G.; et al. Vitamin B12 Deficiency Alters the Gut Microbiota in a Murine Model of Colitis. Front. Nutr. 2020, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Degnan, P.H.; Taga, M.E.; Goodman, A.L. Vitamin B 12 as a Modulator of Gut Microbial Ecology. Cell Metab. 2014, 20, 769–778. [Google Scholar] [CrossRef]
- Guggenheim, K.; Halevy, S.; Hartmann, I.; Zamir, R. The Effect of Antibiotics on the Metabolism of Certain B Vitamins. J. Nutr. 1953, 50, 245–253. [Google Scholar] [CrossRef]
- Tan, C.H.; Blaisdell, S.J.; Hansen, H.J. Mouse transcobalamin II metabolism: The effects of antibiotics on the clearance of vitamin B12 from the serum transcobalamin II-vitamin B12 complex and the reappearance of the free serum transcobalamin II in the mouse. Biochim. Biophys. Acta (BBA)-Gen. Subj. 1973, 320, 469–477. [Google Scholar] [CrossRef]
- Di Meo, S.; Venditti, P. Evolution of the Knowledge of Free Radicals and Other Oxidants. Oxidative Med. Cell. Longev. 2020, 2020, 9829176. [Google Scholar] [CrossRef]
- Haurani, F.I. The Effects of Free Radicals on Cobalamin and Iron. Free Radic. Res. Commun. 1989, 7, 241–243. [Google Scholar] [CrossRef]
- Rocha, E.R.; Selby, T.; Coleman, J.P.; Smith, C.J. Oxidative stress response in an anaerobe, Bacteroides fragilis: A role for catalase in protection against hydrogen peroxide. J. Bacteriol. 1996, 178, 6895–6903. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.B.; Tolonen, A.; Xavier, R.J. Human genetic variation and the gut microbiome in disease. Nat. Rev. Genet. 2017, 18, 690–699. [Google Scholar] [CrossRef]
- Flint, H.J.; Duncan, S.; Louis, P. The impact of nutrition on intestinal bacterial communities. Curr. Opin. Microbiol. 2017, 38, 59–65. [Google Scholar] [CrossRef]
- Bonder, M.J.; Kurilshikov, A.; Tigchelaar, E.F.; Mujagic, Z.; Imhann, F.; Vila, A.V.; Deelen, P.; Vatanen, T.; Schirmer, M.; Smeekens, S.P.; et al. The effect of host genetics on the gut microbiome. Nat. Genet. 2016, 48, 1407–1412. [Google Scholar] [CrossRef]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Links between diet, gut microbiota composition and gut metabolism. Proc. Nutr. Soc. 2014, 74, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Zoetendal, E.G.; de Vos, W.M. Effect of diet on the intestinal microbiota and its activity. Curr. Opin. Gastroenterol. 2014, 30, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for enriching next-generation health-promoting gut bacteria through prebiotics and other dietary components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Plamada, D.; Vodnar, D.C. Polyphenols—Gut Microbiota Interrelationship: A Transition to a New Generation of Prebiotics. Nutrients 2021, 14, 137. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hossain, K.S.; Amarasena, S.; Mayengbam, S. B Vitamins and Their Roles in Gut Health. Microorganisms 2022, 10, 1168. https://doi.org/10.3390/microorganisms10061168
Hossain KS, Amarasena S, Mayengbam S. B Vitamins and Their Roles in Gut Health. Microorganisms. 2022; 10(6):1168. https://doi.org/10.3390/microorganisms10061168
Chicago/Turabian StyleHossain, Khandkar Shaharina, Sathya Amarasena, and Shyamchand Mayengbam. 2022. "B Vitamins and Their Roles in Gut Health" Microorganisms 10, no. 6: 1168. https://doi.org/10.3390/microorganisms10061168
APA StyleHossain, K. S., Amarasena, S., & Mayengbam, S. (2022). B Vitamins and Their Roles in Gut Health. Microorganisms, 10(6), 1168. https://doi.org/10.3390/microorganisms10061168







