Consumption of N2O by Flavobacterium azooxidireducens sp. nov. Isolated from Decomposing Leaf Litter of Phragmites australis (Cav.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Primariy Classification
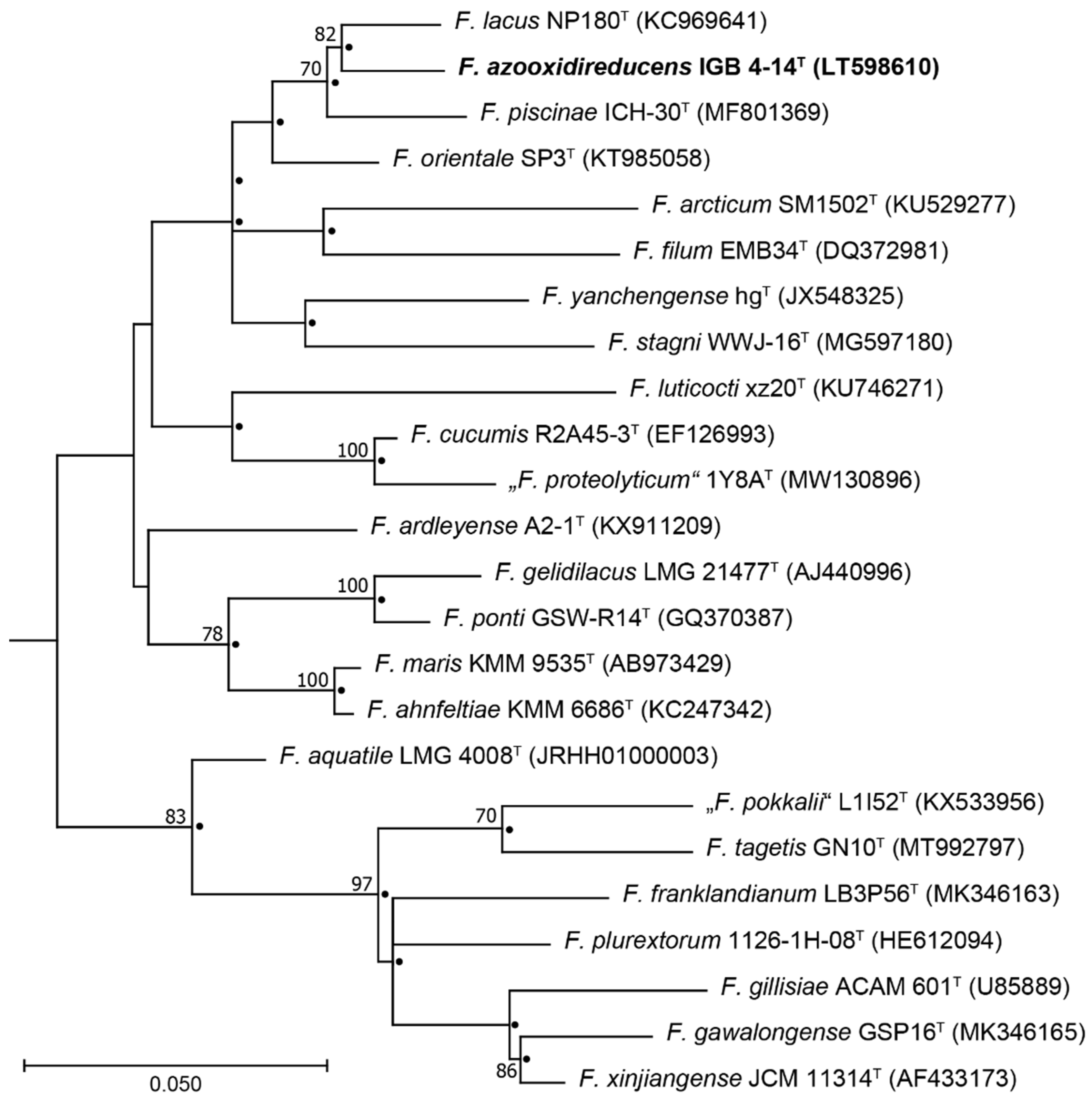
2.2. Phylogenetic Analysis
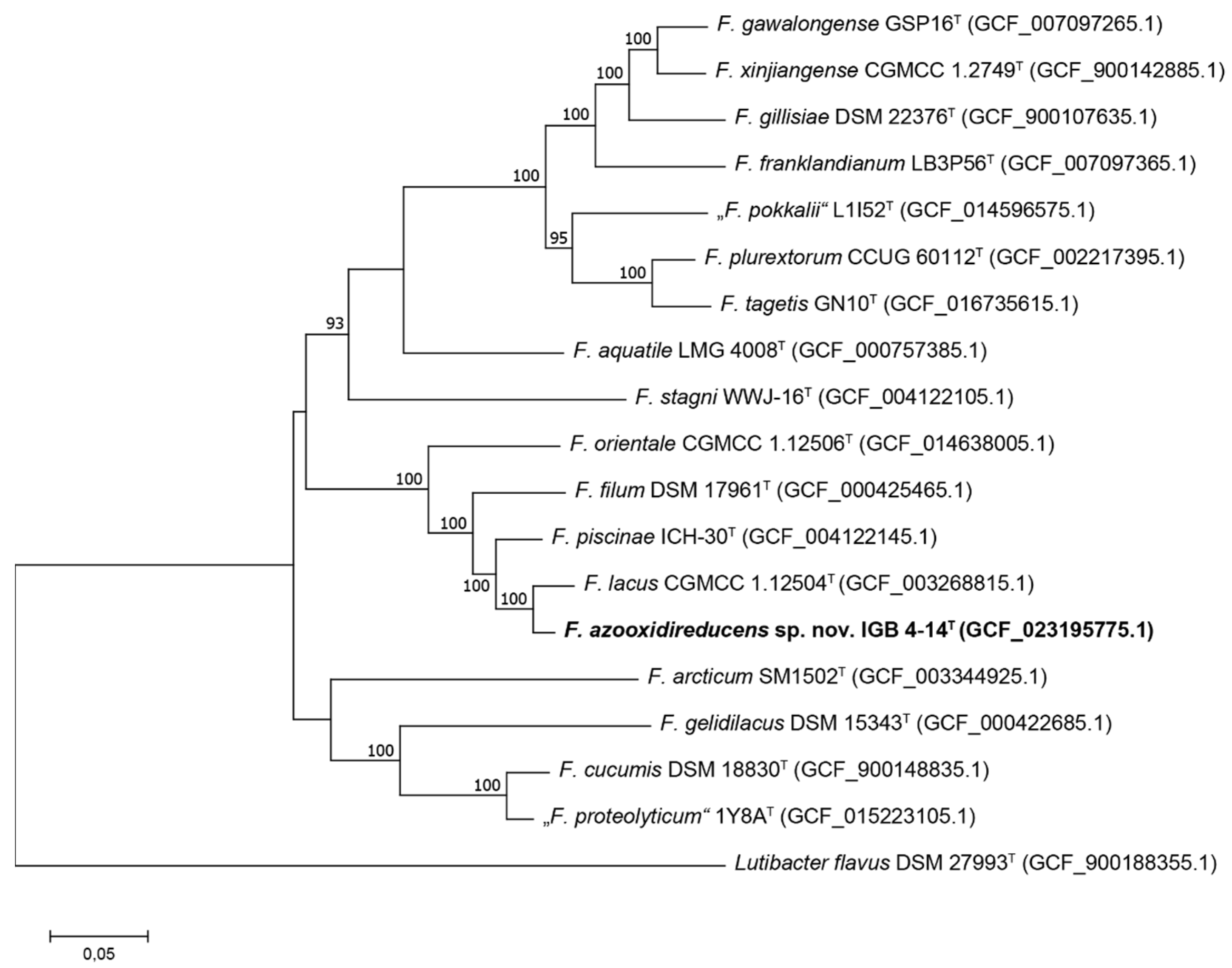
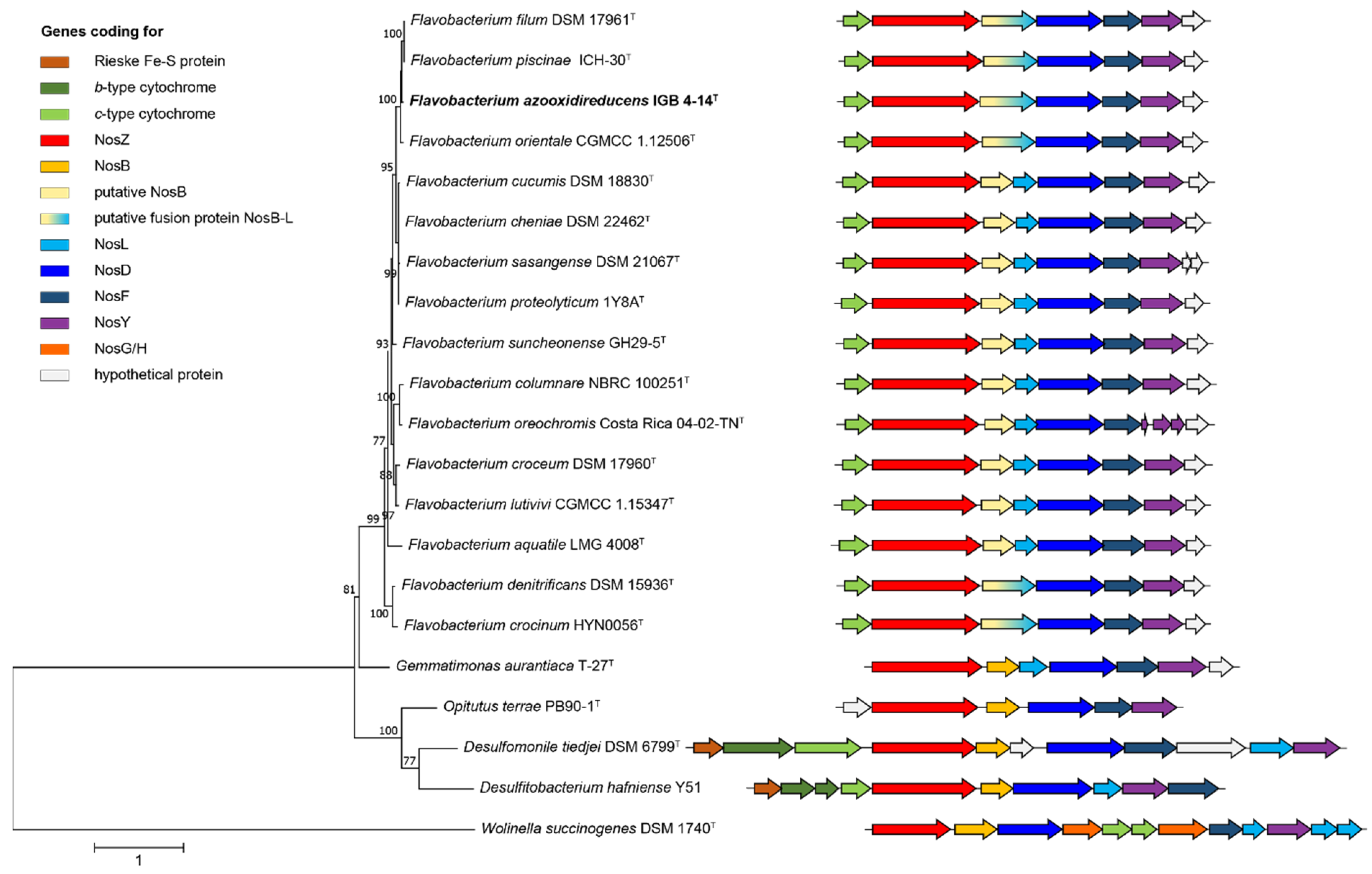
2.3. Genome Sequencing and Bioinformatic Analyses
2.4. Phenotypic Characterisation
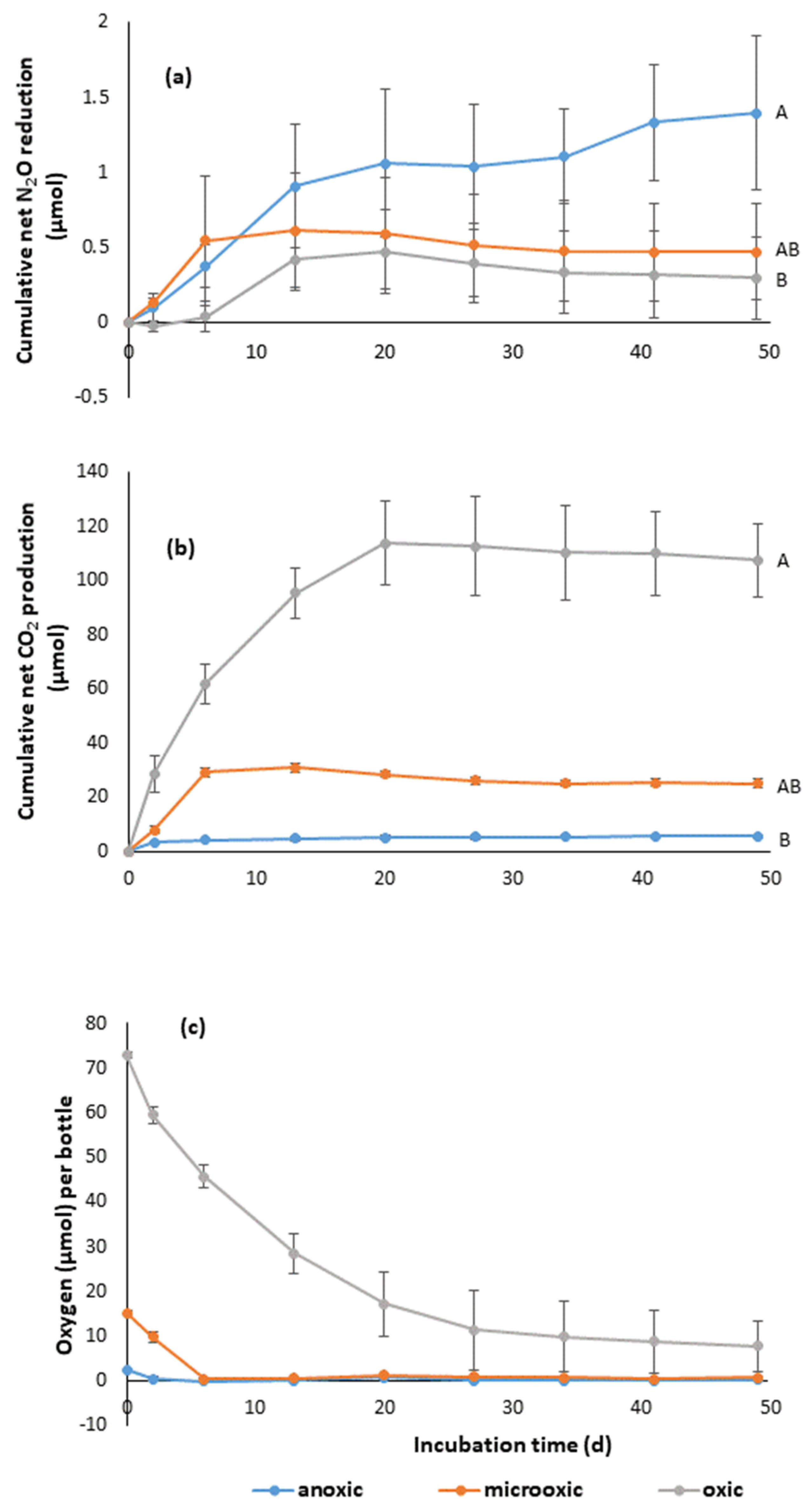
2.5. N2O Consumption
3. Results and Discussion
3.1. Phylogenetic Analysis
3.2. Phenotypic Analysis
3.3. Genome Inventory with Regard to the N-Oxide Reduction
3.4. Consumption of N2O
4. Protologue—Description of Flavobacterium azooxidireducens sp. nov.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ravinshakara, A.; Daniel, J.; Portmann, R. Nitrous oxide (N2O): The dominant ozone-depleting substance emitted in the 21st century. Science 2009, 326, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Hallin, S.; Philippot, L.; Löffler, F.E.; Sanford, R.A.; Jones, C.M. Genomics and ecology of novel N2O-reducing microorganisms. Trends Microbiol. 2018, 26, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Pauleta, S.R.; Carepo, M.S.P.; Moura, I. Source and reduction of nitrous oxide. Coord. Chem. Rev. 2019, 387, 436–449. [Google Scholar] [CrossRef]
- Sanford, R.A.; Wagner, D.D.; Wu, Q.; Chee-Sanford, J.C.; Thomas, S.H.; Cruz-Garcia, C.; Rodriguez, G.; Massol-Deya, A.; Krishnani, K.K.; Ritalahti, K.M.; et al. Unexpected nondenitrifier nitrous oxide reductase gene diversity and abundance in soils. Proc. Natl. Acad. Sci. USA 2012, 109, 19709–19714. [Google Scholar] [CrossRef]
- Orellana, L.H.; Rodriguez-R, L.M.; Higgins, S.; Chee-Sanford, J.C.; Sanford, R.A.; Ritalahti, K.M.; Löffler, F.E.; Konstantinidis, K.T. Detecting nitrous oxide reductase nosZ genes in soil metagenomes: Method development and implications for the nitrogen cycle. Mbio 2014, 5, e01193-14. [Google Scholar] [CrossRef]
- Simon, J. Mitigation of laughing gas emissions by nitrous oxide respiring microorganisms. In Enzymes for Solving Humankind’s Problems: Natural and Artificial Systems in Health, Agriculture, Environment and Energy; Moura, J.J.G., Moura, I., Maia, L.B., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 185–211. [Google Scholar] [CrossRef]
- Torres, M.J.; Simon, J.; Rowley, G.; Bedmar, E.J.; Richardson, D.J.; Gates, A.J.; Delgado, M.J. Nitrous oxide metabolism in nitrate-reducing bacteria: Physiology and regulatory mechanisms. In Advances in Bacterial Electron Transport Systems and Their Regulation; Poole, R.K., Ed.; Advances in Microbial Physiology; Academic Press Ltd-Elsevier Science Ltd.: London, UK, 2016; Volume 68, pp. 353–432. [Google Scholar] [CrossRef]
- Yoon, S.; Nissen, S.; Park, D.; Sanford, R.A.; Löffler, F.E. Nitrous oxide reduction kinetics distinguish bacteria harboring clade I NosZ from those harboring clade II NosZ. Appl. Environ. Microbiol. 2016, 82, 3793–3800. [Google Scholar] [CrossRef]
- Hein, S.; Simon, J. Chapter Four—Bacterial nitrous oxide respiration: Electron transport chains and copper transfer reactions. Adv. Microb. Physiol. 2019, 75, 137–175. [Google Scholar] [CrossRef]
- Shan, J.; Sanford, R.A.; Chee-Sanford, J.; Ooi, S.K.; Löffler, F.E.; Konstantinidis, K.T.; Yang, W.H. Beyond denitrification: The role of microbial diversity in controlling nitrous oxide reduction and soil nitrous oxide emissions. Glob. Change Biol. 2021, 27, 2669–2683. [Google Scholar] [CrossRef]
- Suenaga, T.; Hori, T.; Riya, S.; Hosomi, M.; Smets, B.F.; Terada, A. Enrichment, isolation, and characterization of high-affinity N2O-reducing bacteria in a gas-permeable membrane reactor. Environ. Sci. Technol. 2019, 53, 12101–12112. [Google Scholar] [CrossRef]
- Suenaga, T.; Riya, S.; Hosomi, M.; Terada, A. Biokinetic Characterization and activities of N2O-reducing bacteria in response to various oxygen levels. Front. Microbiol. 2018, 9, 697. [Google Scholar] [CrossRef]
- Graf, D.R.H.; Jones, C.M.; Hallin, S. Intergenomic comparisons highlight modularity of the denitrification pathway and underpin the importance of community structure for N2O emissions. PLoS ONE 2014, 9, e114118. [Google Scholar] [CrossRef] [PubMed]
- Bernardet, J.-F. Flavobacteriaceae. In Bergey’s Manual of Systematics of Archaea and Bacteria; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Kolton, M.; Erlacher, A.; Berg, G.; Cytryn, E. The Flavobacterium genus in the plant holobiont: Ecological, physiological, and applicative insights. In Microbial Models: From Environmental to Industrial Sustainability; Castro-Sowinski, S., Ed.; Springer: Singapore, 2016; pp. 189–207. [Google Scholar] [CrossRef]
- Bernardet, J.-F.; Bowman, J.P. The genus Flavobacterium. In The Prokaryotes: Volume 7: Proteobacteria: Delta, Epsilon Subclass; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 481–531. [Google Scholar] [CrossRef]
- Horn, M.A.; Ihssen, J.; Matthies, C.; Schramm, A.; Acker, G.; Drake, H.L. Dechloromonas denitrificans sp. nov., Flavobacterium denitrificans sp. nov., Paenibacillus anaericanus sp. nov. and Paenibacillus terrae strain MH72, N2O-producing bacteria isolated from the gut of the earthworm Aporrectodea caliginosa. Int. J. Syst. Evol. Microbiol. 2005, 55, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Bernardet, J.-F.; Bowman, J.P. Flavobacterium. In Bergey’s Manual of Systematics of Archaea and Bacteria; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Yoon, H.; Song, M.J.; Kim, D.D.; Sabba, F.; Yoon, S. A serial biofiltration system for effective removal of low-concentration nitrous oxide in oxic gas streams: Mathematical modeling of reactor performance and experimental validation. Environ. Sci. Technol. 2019, 53, 2063–2074. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Song, M.J.; Yoon, S. Design and feasibility analysis of a self-sustaining biofiltration system for removal of low concentration N2O emitted from wastewater treatment plants. Environ. Sci. Technol. 2017, 51, 10736–10745. [Google Scholar] [CrossRef]
- Kim, D.D.; Park, D.; Yoon, H.; Yun, T.; Song, M.J.; Yoon, S. Quantification of nosZ genes and transcripts in activated sludge microbiomes with novel group-specific qPCR methods validated with metagenomic analyses. Water Res. 2020, 185, 116261. [Google Scholar] [CrossRef]
- Li, W.; O’Neill, K.R.; Haft, D.H.; DiCuccio, M.; Chetvernin, V.; Badretdin, A.; Coulouris, G.; Chitsaz, F.; Derbyshire, M.K.; Durkin, A.S.; et al. RefSeq: Expanding the Prokaryotic Genome Annotation Pipeline reach with protein family model curation. Nucleic Acids Res. 2021, 49, D1020–D1028. [Google Scholar] [CrossRef]
- Reuter, H.; Gensel, J.; Elvert, M.; Zak, D. Evidence for preferential protein depolymerization in wetland soils in response to external nitrogen availability provided by a novel FTIR routine. Biogeosciences 2020, 17, 499–514. [Google Scholar] [CrossRef]
- Rillig, M.C.; Hoffmann, M.; Lehmann, A.; Liang, Y.; Lück, M.; Augustin, J. Microplastic fibers affect dynamics and intensity of CO2 and N2O fluxes from soil differently. Microplastics Nanoplastics 2021, 1, 3. [Google Scholar] [CrossRef]
- Heylen, K.; Vanparys, B.; Wittebolle, L.; Verstraete, W.; Boon, N.; De Vos, P. Cultivation of denitrifying bacteria: Optimization of isolation conditions and diversity study. Appl. Environ. Microbiol. 2006, 72, 2637–2643. [Google Scholar] [CrossRef]
- Stanier, R.Y.; Palleroni, N.J.; Doudoroff, M. The aerobic pseudomonads: A taxonomic study. J. Gen. Microbiol. 1966, 43, 159–271. [Google Scholar] [CrossRef]
- Cohen-Bazire, G.; Sistrom, W.R.; Stanier, R.Y. Kinetic studies of pigment synthesis by non-sulphur purple bacteria. J. Cell. Comp. Physiol. 1957, 49, 25–68. [Google Scholar] [CrossRef] [PubMed]
- Kniemeyer, O.; Probian, C.; Rossello-Mora, R.; Harder, J. Anaerobic Mineralization of Quaternary Carbon Atoms: Isolation of Denitrifying Bacteria on Dimethylmalonate. Appl. Environ. Microbiol. 1999, 65, 3319–3324. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, K.; Becker, R.; Behrendt, U.; Kube, M.; Ulrich, A. A comparative analysis of ash leaf-colonizing bacterial communities identifies putative antagonists of Hymenoscyphus fraxineus. Front. Microbiol. 2020, 11, 966. [Google Scholar] [CrossRef] [PubMed]
- Behrendt, U.; Ulrich, A.; Schumann, P. Fluorescent pseudomonads associated with the phyllosphere of grasses; Pseudomonas trivialis sp. nov., Pseudomonas poae sp. nov. and Pseudomonas congelans sp. nov. Int. J. Syst. Evol. Microbiol. 2003, 53, 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.J. 16S/23S rRNA sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds.; John Wiley and Sons: Chichester, UK, 1991; pp. 115–175. [Google Scholar]
- Ulrich, A.; Wirth, S. Phylogenetic diversity and population densities of culturable cellulolytic soil bacteria across an agricultural encatchment. Microb. Ecol. 1999, 37, 238–247. [Google Scholar] [CrossRef]
- Chun, J.; Goodfellow, M. A phylogenetic analysis of the genus Nocardia with 16S rRNA gene sequences. Int. J. Syst. Bacteriol. 1995, 45, 240–245. [Google Scholar] [CrossRef]
- Yoon, S.H.; Ha, S.M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Behrendt, U.; Ulrich, A.; Schumann, P. Chryseobacterium gregarium sp. nov., isolated from decaying plant material. Int. J. Syst. Evol. Microbiol. 2008, 58, 1069–1074. [Google Scholar] [CrossRef]
- Tokajian, S.; Issa, N.; Salloum, T.; Ibrahim, J.; Farah, M. 16S-23S rRNA gene intergenic spacer region variability helps resolve closely related sphingomonads. Front. Microbiol. 2016, 7, 149. [Google Scholar] [CrossRef]
- Ulrich, K.; Kube, M.; Becker, R.; Schneck, V.; Ulrich, A. Genomic analysis of the endophytic Stenotrophomonas strain 169 reveals features related to plant-growth promotion and stress tolerance. Front. Microbiol. 2021, 12, 1542. [Google Scholar] [CrossRef] [PubMed]
- Parks, D.H.; Chuvochina, M.; Waite, D.W.; Rinke, C.; Skarshewski, A.; Chaumeil, P.-A.; Hugenholtz, P. A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat. Biotechnol. 2018, 36, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Behrendt, U.; Wende, S.; Kolb, S.; Ulrich, A. Genome-based phylogeny of the genera Proteus and Cosenzaea and description of Proteus terrae subsp. terrae subsp. nov. and Proteus terrae subsp. cibarius subsp. nov. Int. J. Syst. Evol. Microbiol. 2019, 71, 004651. [Google Scholar] [CrossRef]
- Meier-Kolthoff, J.P.; Göker, M. TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat. Commun. 2019, 10, 2182. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.H.; Ha, S.M.; Lim, J.; Kwon, S.; Chun, J. A large-scale evaluation of algorithms to calculate average nucleotide identity. Antonie Van Leeuwenhoek 2017, 110, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Park, S.; Chun, J. Introducing EzAAI: A pipeline for high throughput calculations of prokaryotic average amino acid identity. J. Microbiol. 2021, 59, 476–480. [Google Scholar] [CrossRef]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res. 2013, 42, D206–D214. [Google Scholar] [CrossRef]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST Server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Kawashima, M.; Furumichi, M.; Tanabe, M. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res. 2015, 44, D457–D462. [Google Scholar] [CrossRef]
- Behrendt, U.; Ulrich, A.; Schumann, P.; Erler, W.; Burghardt, J.; Seyfarth, W. A taxonomic study of bacteria isolated from grasses: A proposed new species Pseudomonas graminis sp. nov. Int. J. Syst. Bacteriol. 1999, 49, 297–308. [Google Scholar] [CrossRef]
- Bernardet, J.F.; Nakagawa, Y.; Holmes, B. Proposed minimal standards for describing new taxa of the family Flavobacteriaceae and emended description of the family. Int. J. Syst. Evol. Microbiol. 2002, 52, 1049–1070. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.T. Single derivatization method for routine analysis of bacterial whole-cell fatty acid methyl esters, including hydroxy acids. J. Clin. Microbiol. 1982, 16, 584–586. [Google Scholar] [CrossRef] [PubMed]
- Kuykendall, L.D.; Roy, M.A.; O’Neill, J.J.; Devine, T.E. Fatty acids, antibiotic resistance, and deoxyribonucleic acid homology groups of Bradyrhizobium japonicum. Int. J. Syst. Evol. Microbiol. 1988, 38, 358–361. [Google Scholar] [CrossRef]
- Arai, H.; Mizutani, M.; Igarashi, Y. Transcriptional regulation of the nos genes for nitrous oxide reductase in Pseudomonas aeruginosa. Microbiology 2003, 149, 29–36. [Google Scholar] [CrossRef]
- Zaman, M.; Kleineidam, K.; Bakken, L.; Berendt, J.; Bracken, C.; Butterbach-Bahl, K.; Cai, Z.; Chang, S.X.; Clough, T.; Dawar, K.; et al. Automated Laboratory and Field Techniques to Determine Greenhouse Gas Emissions. In Measuring Emission of Agricultural Greenhouse Gases and Developing Mitigation Options Using Nuclear and Related Techniques: Applications of Nuclear Techniques for GHGs; Zaman, M., Heng, L., Müller, C., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 109–139. [Google Scholar] [CrossRef]
- Meier-Kolthoff, J.P.; Auch, A.F.; Klenk, H.-P.; Göker, M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform. 2013, 14, 60. [Google Scholar] [CrossRef]
- Hördt, A.; López, M.G.; Meier-Kolthoff, J.P.; Schleuning, M.; Weinhold, L.-M.; Tindall, B.J.; Gronow, S.; Kyrpides, N.C.; Woyke, T.; Göker, M. Analysis of 1,000+ type-strain genomes substantially improves taxonomic classification of Alphaproteobacteria. Front. Microbiol. 2020, 11, 468. [Google Scholar] [CrossRef]
- Konstantinidis, K.T.; Tiedje, J.M. Towards a genome-based taxonomy for prokaryotes. J. Bacteriol. 2005, 187, 6258–6264. [Google Scholar] [CrossRef]
- Palmer, M.; Steenkamp, E.T.; Blom, J.; Hedlund, B.P.; Venter, S.N. All ANIs are not created equal: Implications for prokaryotic species boundaries and integration of ANIs into polyphasic taxonomy. Int. J. Syst. Evol. Microbiol. 2020, 70, 2937–2948. [Google Scholar] [CrossRef]
- Chen, W.-M.; Guo, Y.-P.; Kwon, S.-W.; Sheu, S.-Y. Flavobacterium piscinae sp. nov., isolated from a fish pond. Int. J. Syst. Evol. Microbiol. 2019, 69, 1775–1782. [Google Scholar] [CrossRef]
- Li, A.; Liu, H.; Sun, B.; Zhou, Y.; Xin, Y. Flavobacterium lacus sp. nov., isolated from a high-altitude lake, and emended description of Flavobacterium filum. Int. J. Syst. Evol. Microbiol. 2014, 64, 933–939. [Google Scholar] [CrossRef]
- van Spanning, R.J.M. Structure, function, regulation and evolution of the nitrite and nitrous oxide reductases: Denitrification enzymes with a Beta-propeller fold. In Nitrogen Cycling in Bacteria Molecular Analysis; Moir, J.W.B., Ed.; Caister Academic Press: Norfolk, UK, 2011; pp. 135–161. [Google Scholar]
- Hein, S.; Witt, S.; Simon, J. Clade II nitrous oxide respiration of Wolinella succinogenes depends on the NosG,-C1,-C2, -H electron transport module, NosB and a Rieske/cytochrome bc complex. Environ. Microbiol. 2017, 19, 4913–4925. [Google Scholar] [CrossRef] [PubMed]
- Desloover, J.; Roobroeck, D.; Heylen, K.; Puig, S.; Boeckx, P.; Verstraete, W.; Boon, N. Pathway of nitrous oxide consumption in isolated Pseudomonas stutzeri strains under anoxic and oxic conditions. Environ. Microbiol. 2014, 16, 3143–3152. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Cheng, D.Y.; Tan, P.; An, Q.; Guo, J.S. Characterization of an aerobic denitrifier Pseudomonas stutzeri strain XL-2 to achieve efficient nitrate removal. Bioresour. Technol. 2018, 250, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Suenaga, T.; Qi, C.; Riya, S.; Hosomi, M.; Terada, A. Temperature and oxygen level determine N2O respiration activities of heterotrophic N2O-reducing bacteria: Biokinetic study. Biotechnol. Bioeng. 2021, 118, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Kim, H.; Yoon, S.; Kivisaar, M. Nitrous oxide reduction by an obligate aerobic bacterium, Gemmatimonas aurantiaca strain T-27. Appl. Environ. Microbiol. 2017, 83, e00502–e00517. [Google Scholar] [CrossRef]
- Chee-Sanford, J.; Tian, D.; Sanford, R. Consumption of N2O and other N-cycle intermediates by Gemmatimonas aurantiaca strain T-27. Microbiology 2019, 165, 1345–1354. [Google Scholar] [CrossRef]
- Zhang, H.; Sekiguchi, Y.; Hanada, S.; Hugenholtz, P.; Kim, H.; Kamagata, Y.; Nakamura, K. Gemmatimonas aurantiaca gen. nov., sp. nov., a Gram-negative, aerobic, polyphosphate-accumulating micro-organism, the first cultured representative of the new bacterial phylum Gemmatimonadetes phyl. nov. Int. J. Syst. Evol. Microbiol. 2003, 53, 1155–1163. [Google Scholar] [CrossRef]
- Conthe, M.; Wittorf, L.; Kuenen, J.G.; Kleerebezem, R.; van Loosdrecht, M.C.M.; Hallin, S. Life on N2O: Deciphering the ecophysiology of N2O respiring bacterial communities in a continuous culture. ISME J. 2018, 12, 1142–1153. [Google Scholar] [CrossRef]
- Sullivan, M.J.; Gates, A.J.; Appia-Ayme, C.; Rowley, G.; Richardson, D.J. Copper control of bacterial nitrous oxide emission and its impact on vitamin B12-dependent metabolism. Proc. Natl. Acad. Sci. USA 2013, 110, 19926–19931. [Google Scholar] [CrossRef]
- Ferla, M.P.; Patrick, W.M. Bacterial methionine biosynthesis. Microbiology 2014, 160, 1571–1584. [Google Scholar] [CrossRef]
| Strain IGB 4-14T vs. Species | 16S rRNA Gene Similarity | dDDH Values (%) | ANI (%) 2 | AAI (%) |
|---|---|---|---|---|
| F. lacus CGMCC 1.12504T (NP180T) 1 | 97.29 | 32.2 | 86.68 | 89.78 |
| F. piscinae ICH-30T | 96.92 | 27.8 | 84.01 | 87.92 |
| F. orientale CGMCC 1.12506T (SP3T) 1 | 96.80 | 24.9 | 80.31 | 81.02 |
| F. filum DSM 17961T (EMB34T) 1 | 94.57 | 24.1 | 80.52 | 83.20 |
| Fatty Acid 1/Strain | IGB 4-14T | NBRC 109715T |
|---|---|---|
| Saturated | ||
| iso-C13:0 | 1.32 | Tr |
| anteiso-C13:0 | Tr | ND |
| C15:0 | Tr | 2.4 |
| iso-C15:0 | 44.18 | 36.11 |
| anteiso-C15:0 | 2.36 | 1.1 |
| iso-C15:0 3-OH | 8.57 | 6.2 |
| iso-C17:0 3-OH | 5.30 | 8.49 |
| Unsaturated | ||
| iso-C15:1 G | 17.65 | 21.58 |
| C15:1 w6c | Tr | 2.61 |
| iso-C17:1 w9c | 5.63 | 9.88 |
| anteiso-C17:1 w9c | Tr | ND |
| C17:1 w6c | ND | Tr |
| C17:1 w8c | ND | Tr |
| Summed features 2 | ||
| Feature 1 | 1.52 | ND |
| Feature 3 | 1.92 | 1.71 |
| Feature 4 | Tr | 1.68 |
| Characteristic | IGB 4-14T | NBRC 109715T |
|---|---|---|
| Flexirubin-type pigments | + | - |
| absorption of Congo red | + | - |
| Oxidation (GEN III) of: | ||
| N-Acetyl-β-D-Mannosamine | + | - |
| D-Galactose | + | - |
| 3-Methyl Glucose | + | - |
| D-Fructose-6-PO4 | + | - |
| L-Arginine | + | - |
| L-Histidine | + | - |
| Pectin | - | + |
| D-Galacturonic Acid | + | - |
| L-Galactonic Acid Lactone | + | - |
| D-Glucuronic Acid | + | - |
| Glucuronamide | + | - |
| Mucic Acid | - | + |
| p-Hydroxy-Phenylacetic Acid | + | - |
| L-Lactic Acid | + | - |
| D-Malic Acid | - | + |
| L-Malic Acid | - | + |
| Propionic Acid | + | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behrendt, U.; Spanner, T.; Augustin, J.; Zak, D.H.; Horn, M.A.; Kolb, S.; Ulrich, A. Consumption of N2O by Flavobacterium azooxidireducens sp. nov. Isolated from Decomposing Leaf Litter of Phragmites australis (Cav.). Microorganisms 2022, 10, 2304. https://doi.org/10.3390/microorganisms10112304
Behrendt U, Spanner T, Augustin J, Zak DH, Horn MA, Kolb S, Ulrich A. Consumption of N2O by Flavobacterium azooxidireducens sp. nov. Isolated from Decomposing Leaf Litter of Phragmites australis (Cav.). Microorganisms. 2022; 10(11):2304. https://doi.org/10.3390/microorganisms10112304
Chicago/Turabian StyleBehrendt, Undine, Tobias Spanner, Jürgen Augustin, Dominik H. Zak, Marcus A. Horn, Steffen Kolb, and Andreas Ulrich. 2022. "Consumption of N2O by Flavobacterium azooxidireducens sp. nov. Isolated from Decomposing Leaf Litter of Phragmites australis (Cav.)" Microorganisms 10, no. 11: 2304. https://doi.org/10.3390/microorganisms10112304
APA StyleBehrendt, U., Spanner, T., Augustin, J., Zak, D. H., Horn, M. A., Kolb, S., & Ulrich, A. (2022). Consumption of N2O by Flavobacterium azooxidireducens sp. nov. Isolated from Decomposing Leaf Litter of Phragmites australis (Cav.). Microorganisms, 10(11), 2304. https://doi.org/10.3390/microorganisms10112304







