Lacticaseibacillus casei Strain Shirota Modulates Macrophage-Intestinal Epithelial Cell Co-Culture Barrier Integrity, Bacterial Sensing and Inflammatory Cytokines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Culture and Preparation of Heat Killed Extracts
2.2. Cell Culture & Co-Culture
2.3. Modulatory Effect of Heat-Killed LcS Probiotic Bacteria on Epithelial Cell Cytokine Production
2.4. Detection of Endogenous IL-10 Activity and Its Effect on Modulation of Epithelial Cell Bacterial Sensing Molecules
2.5. Measurement of Trans-Epithelial Electrical Resistance (TEER)
2.6. Immunohistochemical (IHC) Localisation of Epithelial Cell Zona Occludin-1 (ZO-1)
2.7. Detection of Membrane TLR2 and TLR4 Protein by Flow Cytometry
2.8. Real Time qPCR Analysis
2.9. Quantification of Cytokine Secretion
2.10. Statistical Analysis
3. Results
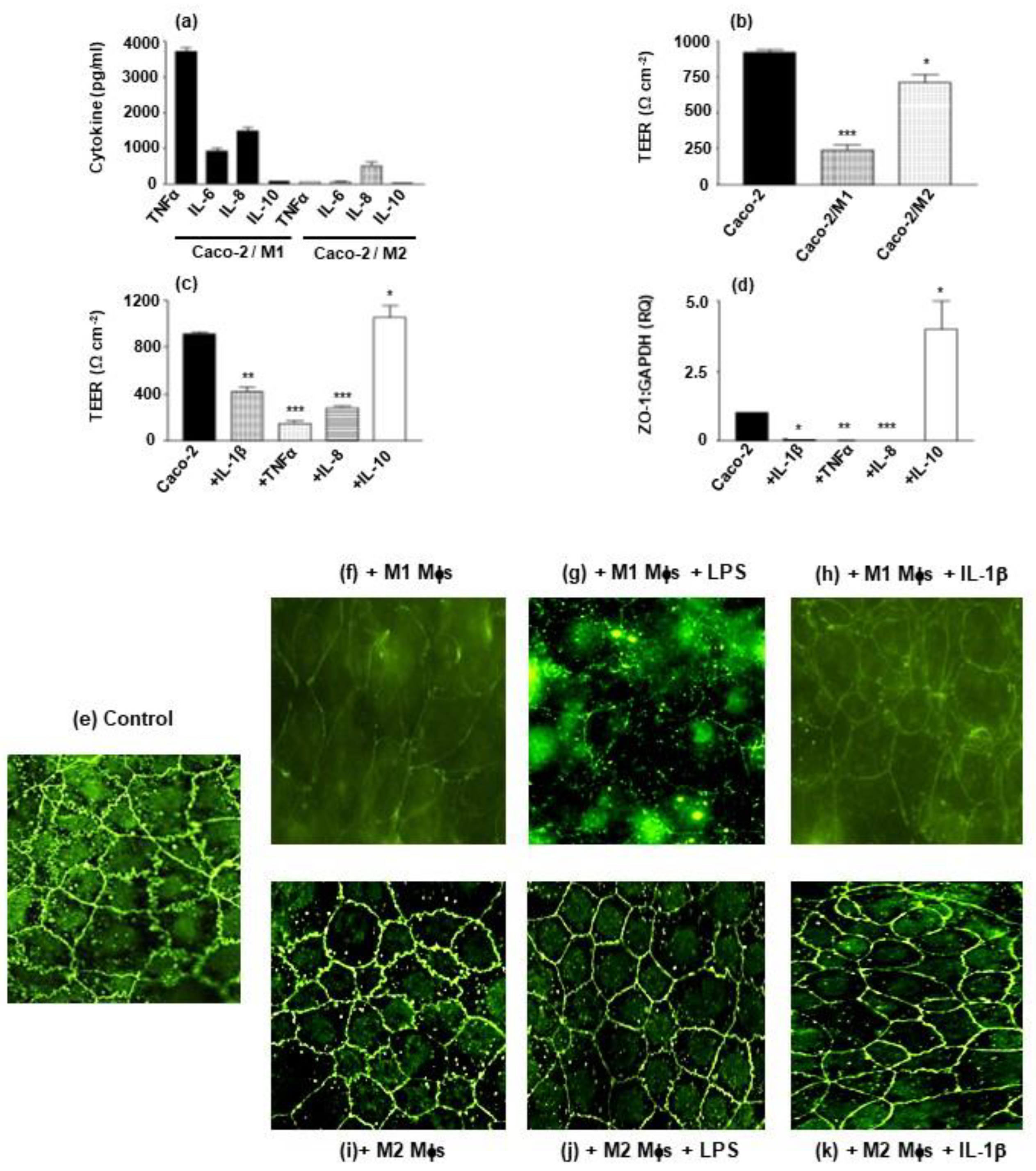
3.1. Modulation of IEC Cytokines by Heat-Killed LcS Is Dependent on Inflammatory Environment
3.2. Macrophage Subset and Inflammatory Cytokines Determine IEC Barrier Integrity
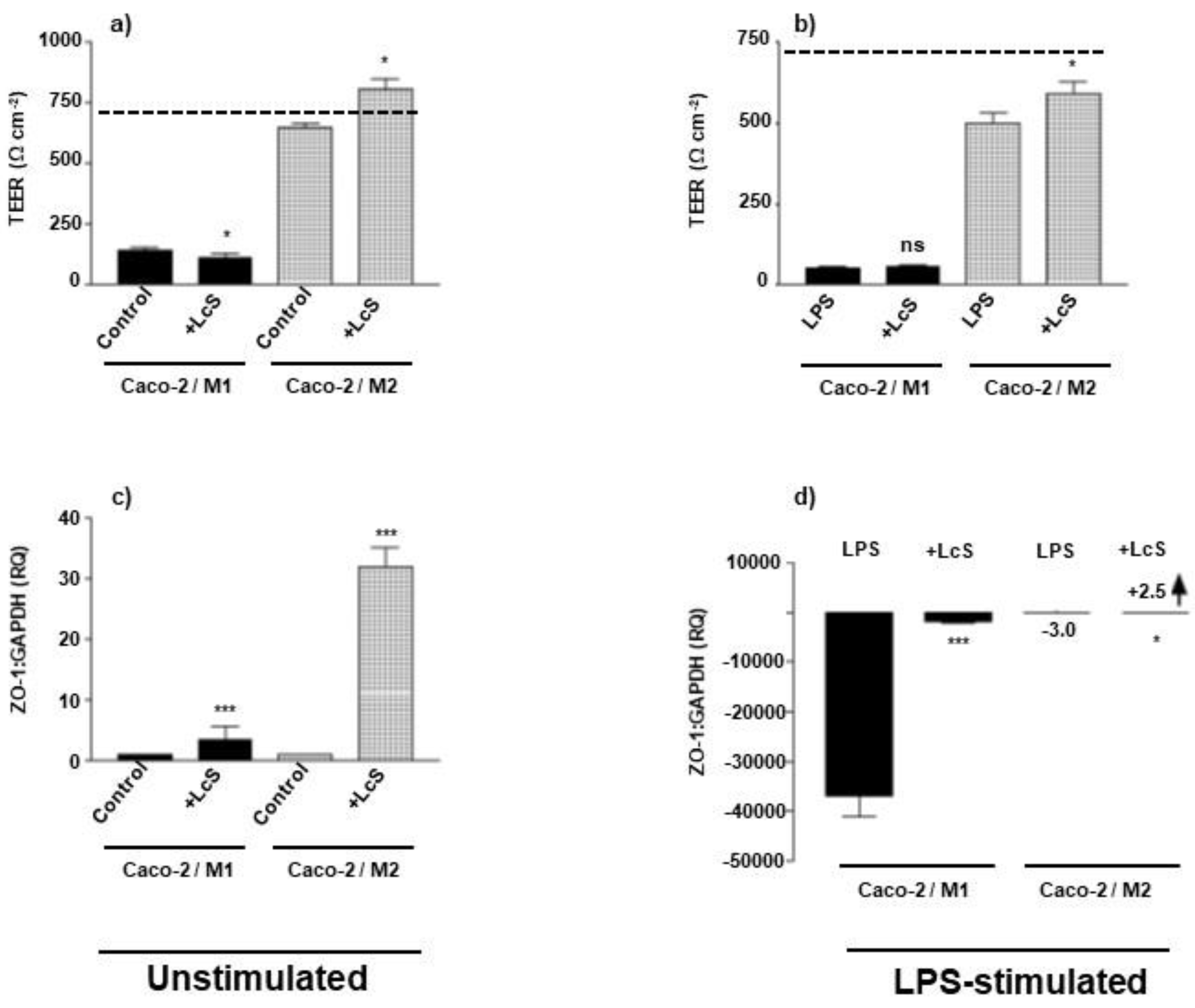
3.3. LcS Differentially Rescues Epithelial Barrier Integrity in Macrophage Subset-IEC Co-Cultures
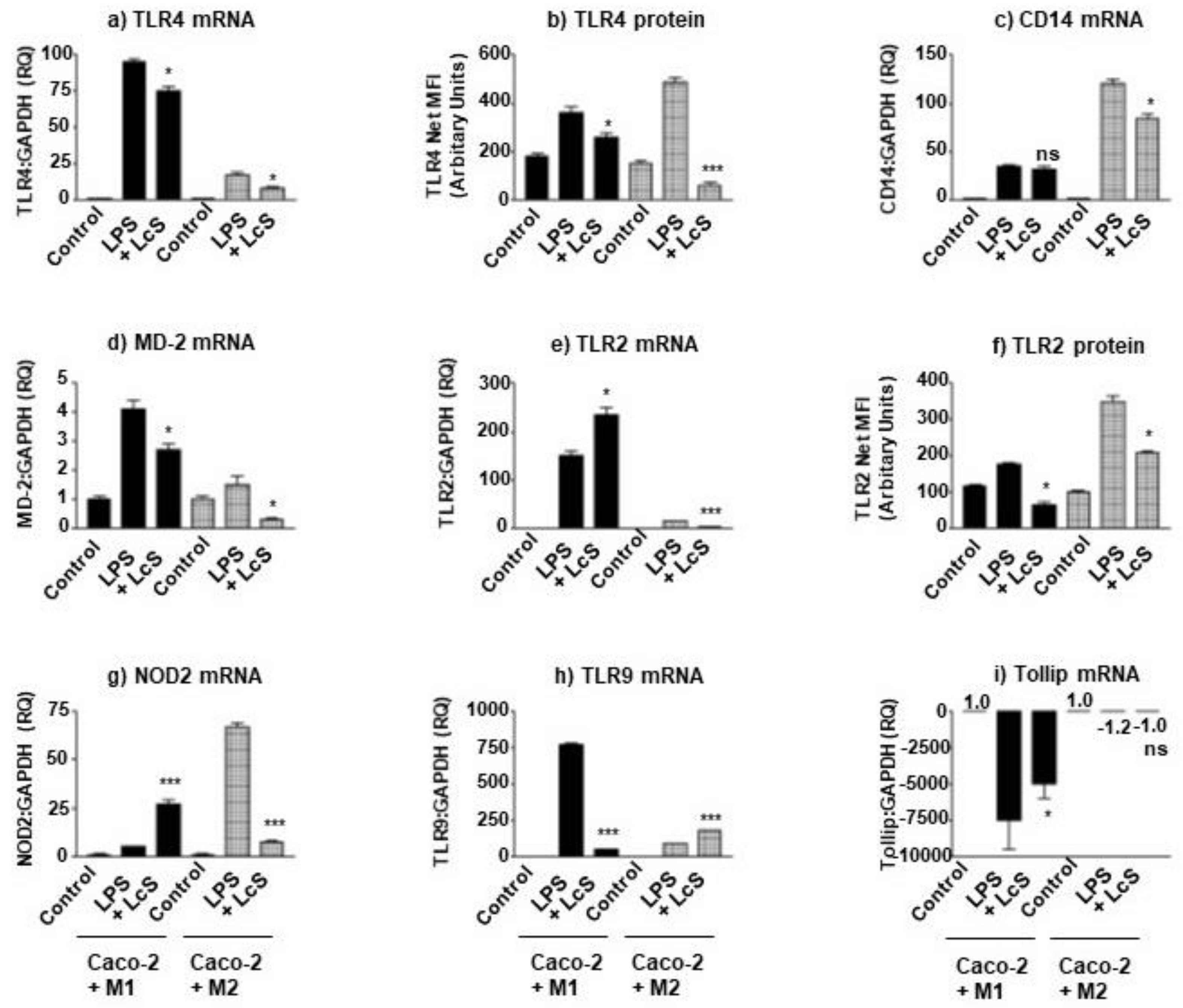
3.4. LcS Differentially Modulates IEC LPS-Sensing Molecules in Macrophage Subset Co-Culture
3.5. LcS Modulation of Co-Culture Pro- and Anti-Inflammatory Cytokines Is Dependent on Macrophage Subset and Stimulation Status
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Foey, A.D. Mucosal macrophages: Phenotype and functionality in homeostasis and pathology. In Handbook of Macrophages: Life Cycle, Functions and Diseases; Takahashi, R., Kai, H., Eds.; Nova Science Publishers Inc.: New York, NY, USA, 2012; Chapter 4; pp. 121–146. [Google Scholar]
- Foey, A.D. Macrophage polarisation: A collaboration of differentiation, activation and pre-programming? J. Clin. Cell. Immunol. 2015, 6, 293. [Google Scholar] [CrossRef]
- Smith, P.D.; Ochsenbauer-Jambor, C.; Smythies, L.E. Intestinal macrophages: Unique effector cells of the innate immune system. Immunol. Revs. 2005, 206, 149–159. [Google Scholar] [CrossRef]
- Smythies, L.E.; Sellers, M.; Clements, R.H.; Mosteller-Barnum, M.; Meng, G.; Benjamin, W.H.; Orenstein, J.M.; Smith, P.D. Human intestinal macrophages display profound inflammatory anergy despite avid phagocytic and bacteriocidal activity. J. Clin. Investig. 2005, 115, 66–75. [Google Scholar] [CrossRef] [Green Version]
- Platt, A.M.; Mowat, A.M. Mucosal macrophages and the regulation of immune responses in the intestine. Immunol. Lett. 2008, 119, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Hardy, H.; Harris, J.; Lyon, E.; Beal, J.; Foey, A.D. Probiotics, prebiotics and immunomodulation of gut mucosal defences: Homeostasis and immunopathology. Nutrients 2013, 5, 1869–1912. [Google Scholar] [CrossRef] [PubMed]
- Habil, N.; Al-Murrani, W.; Beal, J.; Foey, A.D. Probiotic bacterial strains differentially modulate macrophage cytokines in a strain-dependent and cell subset-specific manner. Benef. Microbes 2011, 2, 283–293. [Google Scholar] [CrossRef]
- Habil, N.; Beal, J.; Foey, A.D. Lactobacillus casei strain Shirota selectively modulates macrophage subset cytokine production. Int. J. Probiotics Prebiotics 2012, 7, 1–12. [Google Scholar]
- Habil, N.; Abate Woldie, W.; Beal, J.; Foey, A.D. Probiotic bacterial species selectively modulate gut epithelial cell beta-defensin-2. Benef. Microbes 2014, 5, 483–495. [Google Scholar] [CrossRef]
- Haller, D.; Bode, C.; Hammes, W.P.; Pfeifer, A.M.A.; Schiffrin, E.J.; Blum, S. Non-pathogenic bacteria elicit a differential cytokine response by intestinal epithelial cell/leucocyte co-cultures. Gut 2000, 47, 79–87. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.M.; Kim, N.-H.; Lee, S.; Kim, Y.N.; Heo, J.-D.; Rho, J.-R.; Jeong, E.J. (10Z)-Debromohymenialdisine from marine sponge Stylissa sp. Regulates intestinal inflammatory responses in co-culture model of epithelial Caco-2 cells and THP-1 macrophage cells. Molecules 2019, 24, 3394. [Google Scholar] [CrossRef] [Green Version]
- Noel, G.; Baetz, N.W.; Staab, J.F.; Donowitz, M.; Kovbasnjuk, O.; Pasetti, M.F.; Zachos, N.C. A primary human macrophage-enteroid co-culture model to investigate mucosal gut physiology and host-pathogen interactions. Sci. Rep. 2017, 7, 45270. [Google Scholar] [CrossRef] [PubMed]
- Kampfer, A.A.M.; Urban, P.; Gioria, S.; Kanase, N.; Stone, V.; Kinsner-Ovaskainen, A. Development of an in vitro co-culture model to mimic the human intestine in healthy and diseased state. Toxicol. Vitr. 2017, 45, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, J.-L.; Finlay, B.B. Effect of cell polarisation and differentiation on entry of Listeria monocytogenes into the enterocyte-like Caco-2 cell line. Infect. Immun. 1996, 64, 1299–1308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nollevaux, G.; Deville, C.; El Moualij, B.; Zorzi, W.; Deloyer, P.; Schneider, Y.-J.; Peulen, O.; Dandrifosse, G. Development of a serum-free co-culture of human intestinal epithelium cell-lines (Caco-2/HT29-5M21). BMC Cell Biol. 2006, 7, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parlesak, A.; Haller, D.; Brinz, S.; Baeuerlein, A.; Bode, C. Modulation of cytokine release by differentiated Caco-2 cells in a compartmentalised co-culture model with mononuclear leukocytes and non-pathogenic bacteria. Scand. J. Immunol. 2004, 60, 477–485. [Google Scholar] [CrossRef]
- Lavelle, E.C.; Murphy, C.; O’Neill, L.A.J.; Creagh, E.M. The role of TLRs, NLRs and RLRs in mucosal innate immunity and homeostasis. Mucosal Immunol. 2010, 3, 17–28. [Google Scholar] [CrossRef] [Green Version]
- Melmed, G.; Thomas, L.S.; Lee, N.; Tesfay, S.Y.; Lukasek, K.; Michelsen, K.S.; Zhou, Y.; Hu, B.; ArditI, M.; Abreu, M.T. Human intestinal epithelial cells are broadly unresponsive to Toll-Like Receptor, 2-dependent bacterial ligands: Implications for host-microbial interactions in the gut. J. Immunol. 2003, 170, 1406–1415. [Google Scholar] [CrossRef] [Green Version]
- Young, S.L.; Simon, M.A.; Baird, M.A.; Tannock, G.W.; Bibiloni, R.; Spencely, K.; Lane, J.M.; Fitzharris, P.; Crane, J.; Town, I.; et al. Bifidobacterial species differentially affect expression of cell surface markers and cytokines of dendritic cells harvested from cord blood. Clin. Diagn. Lab. Immunol. 2004, 11, 686–690. [Google Scholar] [CrossRef] [Green Version]
- Hilgers, A.R.; Conradi, R.A.; Burton, P.S. Caco-2 cell monolayers as a model for drug transport across the intestinal mucosa. Pharm. Res. 1990, 7, 902–910. [Google Scholar] [CrossRef]
- Belfield, L.A. Interactions between Porphyromonas gingivalis and macrophages in oral pathology. Ph.D. Thesis, University of Plymouth, Plymouth, UK, April 2013. [Google Scholar]
- Daigneault, M.; Preston, J.A.; Marriott, H.M.; Whyte, M.K.B.; Dockrell, D.H. The Identification of Markers of Macrophage Differentiation in PMA-Stimulated THP-1 Cells and Monocyte-Derived Macrophages. PLoS ONE 2010, 5, e8668. [Google Scholar] [CrossRef]
- Foey, A.D. Butyrate regulation of distinct macrophage subsets: Opposing effects on m1 and m2 macrophages. Int. J. Probiotics Prebiotics 2011, 6, 147–158. [Google Scholar]
- Habil, N. Probiotic Modulation of Mucosal Immune Responses in an In Vitro Co-Culture Model. Ph.D. Thesis, University of Plymouth, Plymouth, UK, February 2013. [Google Scholar]
- Kohro, T.; Tanaka, T.; Murakami, T.; Wada, Y.; Aburatani, H.; Hamakubo, T.; Kodama, T. A Comparison of Differences in the Gene Expression Profiles of Phorbol 12-myristate 13-acetate Differentiated THP-1 Cells and Human Monocyte-derived Macrophage. J. Atheroscler. Thromb. 2004, 11, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Schwende, H.; Fitzke, E.; Ambs, P.; Dieter, P. Differences in the state of differentiation of THP-1 cells induced by phorbol ester and 1,25-dihydroxyvitamin D3. J. Leukoc. Biol. 1996, 59, 555–561. [Google Scholar] [CrossRef]
- Tadesco, S.; De Majo, F.; Kim, J.; Trenti, A.; Trevisi, L.; Fadini, G.P. Convenience versus biological significance: Are PMA-differentiated THP-1 cells a reliable substitute for blood-derived macrophages when studying in vitro polarisation? Front. Pharmacol. 2018, 9, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, R.; Kim, J.Y. 1,25-Dihydroxyvitamin D3 Facilitates M2 Polarization and Upregulates TLR10 Expression on Human Microglial Cells. Neuroimmunomodulation 2016, 23, 75–80. [Google Scholar] [CrossRef]
- Vey, E.; Zhang, J.H.; Dayer, J.M. IFN-gamma and 1,25(OH)2D3 induce on THP-1 cells distinct patterns of cell surface antigen expression, cytokine production, and responsiveness to contact with activated T cells. J. Immunol. 1992, 149, 2040–2046. [Google Scholar]
- Vogel, D.Y.; Glim, J.E.; Stavenuiter, A.W.; Breur, M.; Heijnen, P.; Amor, S.; Dijkstra, C.D.; Beelen, R.H. Human mac-rophage polarisation in vitro: Maturation and activation methods compared. Immunobiology 2014, 219, 695–703. [Google Scholar] [CrossRef]
- Zarember, K.A.; Godowski, P.J. Tissue Expression of Human Toll-Like Receptors and Differential Regulation of Toll-Like Receptor mRNAs in Leukocytes in Response to Microbes, Their Products, and Cytokines. J. Immunol. 2002, 168, 554–561. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Zhou, M.; Guo, Y.; Song, Z.; Liu, B. 1, 25-Dihydroxyvitamin D3 Promotes High Glucose-Induced M1 Macrophage Switching to M2 via the VDR-PPAR Signaling Pathway. BioMed Res. International 2015, 2015, 157834. [Google Scholar]
- Parry, S.; Sebbag, M.; Feldmann, M.; Brennan, F.M. Contact with T cells modulates monocyte IL-10 production: Role of T cell membrane TNF-alpha. J. Immunol. 1997, 158, 3673–3681. [Google Scholar]
- Livak, J.K.; Schmittgen, T.D. Analysis of relative gene expression data using real time quantitative PCR and the, 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Fleming, S.D.; Campbell, P.A. Macrophages have cell surface IL-10 that regulates macrophage bacteriocidal activity. J. Immunol. 1996, 156, 1143–1150. [Google Scholar] [PubMed]
- Al-Shaghdali, K.; Durante, B.; Hayward, C.; Beal, J.; Foey, A. Macrophage subsets exhibit distinct E. coli-LPS tolerisable cytokines associated with the negative regulators, IRAK-M and Tollip. PLoS ONE 2019, 14, e0214681. [Google Scholar]
- Fiorentino, D.F.; Zlotnik, A.; Mosmann, T.R.; Howard, M.; O’Garra, A. IL-10 inhibits cytokine production by activated macrophages. J. Immunol. 1991, 147, 3815–3822. [Google Scholar] [PubMed]
- Lan, J.-G.; Cruickshank, S.M.; Singh, J.C.I.; Farrar, M.; Lodge, J.P.A.; Felsburg, P.J.; Carding, S.R. Different cytokine response of primary colonic epithelial cells to commensal bacteria. World J. Gastroenterol. 2005, 11, 3375–3384. [Google Scholar] [CrossRef]
- Strober, W.; Zhang, F.; Kitani, A.; Fuss, I.; Fichtner-Feigl, S. Pro-inflammatory cytokines underlying the inflammation of Crohn’s disease. Curr. Opin. Gastroenterol. 2010, 26, 301–307. [Google Scholar] [CrossRef]
- Hyun, J.; Romero, L.; Riveron, R.; Flores, C.; Kanagavelu, S.; Chung, K.D.; Alonso, A.; Sotolongo, J.; Ruiz, J.; Manukyan, A.; et al. Human intestinal epithelial cells express IL-10 through Toll-like receptor 4 (TLR4)-mediated epithelial-macrophage crosstalk. J. Innate Immun. 2015, 7, 87–101. [Google Scholar] [CrossRef]
- Tilg, H.; van Montfrans, C.; van den Ende, A.; Kaser, A.; van Deventer, S.J.H.; Schreiber, S.; Gregor, M.; Ludwiczek, O.; Rutgeerts, P.; Gasche, C.; et al. Treatment of Crohn’s disease with recombinant human interleukin 10 induces the proinflammatory cytokine interferon γ. Gut 2002, 50, 191–195. [Google Scholar] [CrossRef] [Green Version]
- Mowat, A.M.; Bain, C.C. Mucosal macrophages in intestinal homeostasis and inflammation. J. Innate Immun. 2011, 3, 550–564. [Google Scholar] [CrossRef]
- Das, P.; Rampal, R.; Udinia, S.; Kumar, T.; Pilli, S.; Wari, N.; Ahmed, I.K.; Kedia, S.; Gupta, S.D.; Kumar, D.; et al. Selective M1macrophage polarisation in granuloma-positive and granuloma-negative Crohn’s disease in comparison to intestinal tuberculosis. Intest. Res. 2018, 16, 426–435. [Google Scholar] [CrossRef] [Green Version]
- Ma, T.Y.; Iwamoto, G.K.; Hoa, N.T.; Akotia, V.; Pedrami, A.; Boivin, M.A.; Said, H.M. TNFα-induced increase in intestinal epithelial tight junction permeability requires NF-κB activation. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G367–G376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardari, D.; Dusio, G.F.; Luchini, P.; Sciarabba, M.; Solimene, U.; Rumio, C. Oral administration of Interleukin-10 and anti-IL-1 antibody ameliorates experimental intestinal inflammation. Gastroenterol. Res. 2013, 6, 124–133. [Google Scholar]
- Galdeano, C.M.; Perdigon, G. The probiotic bacterium Lactobacillus casei induces activation of the gut mucosal immune system through innate immunity. Clin. Vaccine Immunol. 2006, 13, 219–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiu, Y.-H.; Tsai, J.-J.; Lin, S.-L.; Lin, M.-Y. Lactobacillus casei MYL01 modulates the proinflammatory state induced by ethanol in an in vitro model. J. Dairy Sci. 2014, 97, 2009–2016. [Google Scholar] [CrossRef] [Green Version]
- Castillo, N.A.; Perdigon, G.; De Moreno de LeBlanc, A. Oral administration of a probiotic Lactobacillus modulates cytokine production and TLR expression improving the immune response against Salmonella enterica serovar Typhimurium infection in mice. BMC Microbiol. 2011, 11, 177. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.; Bhatia, R.; Singh, A.; Singh, P.; Kaur, R.; Khare, P.; Purama, R.K.; Boparai, R.K.; Rishi, P.; Ambalam, P.; et al. Probiotic attributes and prevention of LPS-induced pro-inflammatory stress in RAW264.7 macrophages and human intestinal epithelial cell line (Caco-2) by newly isolated Weissella cibaria strains. Food Funct. 2018, 9, 1254. [Google Scholar] [CrossRef]
- Almerighi, C.; Sinistro, A.; Cavazza, A.; Ciaprini, C.; Rocchi, G.; Bergamini, A. 1α,25-Dihydroxyvitamin D3 inhibits CD40L-induced pro-inflammatory and immunomodulatory activity in Human Monocytes. Cytokine 2009, 45, 190–197. [Google Scholar] [CrossRef]
- Auwerx, J. The human leukemia cell line, THP-1: A multifacetted model for the study of monocyte-macrophage differentiation. Experientia 1991, 47, 22–31. [Google Scholar] [CrossRef]
- Baqui, A.A.; Meiller, T.F.; Turng, B.-F.; Kelley, J.I.; Falkler, W.A. Functional Changes En Thp-1 Human Monocytic cells after Stimulation with Lipopolysaccharide of oral Microorganisms and Granulocyte Macrophage colony Stimulating Factor. Immunopharmacol. Immunotoxicol. 1998, 20, 493–518. [Google Scholar] [CrossRef]
- Baqui, A.A.; Meiller, T.F.; Falkler, W.A. Enhanced interleukin-8 production in THP-1 human monocytic cells by lipopol-ysaccharide from oral microorganisms and granulocyte-macrophage colony-stimulating factor. Oral Microbiol. Immunol. 1999, 14, 275–280. [Google Scholar] [CrossRef]
- Chanput, W.; Mes, J.; Vreeburg, R.A.M.; Savelkoul, H.F.J.; Wichers, H.J. Transcription profiles of LPS-stimulated THP-1 monocytes and macrophages: A tool to study inflammation modulating effects of food-derived compounds. Food Funct. 2010, 1, 254–261. [Google Scholar] [CrossRef]
- Chanput, W.; Mes, J.J.; Savelkoul, H.F.J.; Wichers, H.J. Characterization of polarized THP-1 macrophages and polarizing ability of LPS and food compounds. Food Funct. 2012, 4, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Chanput, W.; Mes, J.J.; Wichers, H.J. THP-1 cell line: An in vitro cell model for immune modulation approach. Int. Immunopharmacol. 2014, 23, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Kuhn, D.C.; Gaydos, L.J.; Demers, L.M. Induction of nitric oxide and nitric oxide synthase mRNA by silica and lipopolysaccharide in PMA-primed THP-1 cells. APMIS 1996, 104, 176–182. [Google Scholar] [CrossRef]
- Clohisy, D.R.; Bar-Shavit, Z.; Chappel, J.C.; Teitelbaum, S. 1,25-Dihydroxyvitamin D3 modulates bone marrow macrophage precursor proliferation and differentiation. Up-regulation of the mannose receptor. J. Biol. Chem. 1987, 262, 15922–15929. [Google Scholar] [CrossRef]
- Dickie, L.J.; Church, L.D.; Coulthard, L.R.; Mathews, R.J.; Emery, P.; McDermott, M.F. Vitamin D3 down-regulates intracellular Toll-like receptor 9 expression and Toll-like receptor 9-induced IL-6 production in human monocytes. Rheumatology 2010, 49, 1466–1471. [Google Scholar] [CrossRef] [Green Version]
- Fleit, H.B.; Kobasiuk, C.D. The human monocyte-like cell line THP-1 expresses Fc gamma RI and Fc gamma RII. J. Leukoc. Biol. 1991, 49, 556–565. [Google Scholar] [CrossRef]
- Foey, A.D.; Brennan, F.M. Conventional protein kinase C and atypical protein kinase Czeta differentially regulate mac-rophage production of tumour necrosis factor-alpha and interleukin-10. Immunol. 2004, 112, 44–53. [Google Scholar] [CrossRef]
- Foey, A.D.; Crean, S. Macrophage Subset Sensitivity to Endotoxin Tolerisation by Porphyromonas gingivalis. PLoS ONE 2013, 8, e67955. [Google Scholar] [CrossRef] [Green Version]
- Foey, A.D.; Habil, N.; Al-Shaghdali, K.; Crean, S. Porphyromonas gingivalis-stimulated macrophage subsets exhibit differ-ential induction and responsiveness to interleukin-10. Arch Oral Biol. 2017, 73, 282–288. [Google Scholar] [CrossRef] [Green Version]
- Gordon, S.; Taylor, P.R. Monocyte and macrophage heterogeneity. Nat. Rev. Immunol. 2005, 5, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Gynther, P.; Toropainen, S.; Matilainen, J.M.; Seuter, S.; Carlberg, C.; Vaisanen, S. Mechanism of 1α,25-dihydroxyvitamin D(3)-dependent repression of interleukin-12B. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Res. 2011, 1813, 810–818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoe, E.; Nathanielsz, J.; Toh, Z.Q.; Spry, L.; Marimla, R.; Balloch, A.; Mulholland, K.; Licciardi, P.V. Anti-Inflammatory Effects of Vitamin D on Human Immune Cells in the Context of Bacterial Infection. Nutrients 2016, 8, 806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahmoud, L.; Al-Enezi, F.; Al-Saif, M.; Warsy, A.; Khabar, K.S.A.; Hitti, E.G. Sustained stabilization of Interleukin-8 mRNA in human macrophages. RNA Biol. 2014, 11, 124–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mantovani, A.; Sozzani, S.; Locati, M.; Allavena, P.; Sica, A. Macrophage polarization: Tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002, 23, 549–555. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000Prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef] [Green Version]
- Matilainen, J.M.; Husso, T.; Toropainen, S.; Seuter, S.; Turunen, M.P.; Gynther, P.; Yla-Herttuala, S.; Carlberg, C.; Vaisanen, S. Primary effect of 1α,25(OH)₂D₃ on IL-10 expression in monocytes is short-term down-regulation. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Res. 2010, 1803, 1276–1286. [Google Scholar] [CrossRef]
- Papadimitriou, J.M.; Ashman, R.B. Macrophages: Current Views on Their Differentiation, Structure, and Function. Ultrastruct. Pathol. 1989, 13, 343–372. [Google Scholar] [CrossRef]
- Prieto, J.; Eklund, A.; Patarroyo, M. Regulated Expression of Integrins and Other Adhesion Molecules during Differentiation of Monocytes into Macrophages. Cell. Immunol. 1994, 156, 191–211. [Google Scholar] [CrossRef]
- Starr, T.; Bauler, T.; Malik-Kale, P.; Steele-Mortimer, O. The phorbol 12-myristate-13-acetate differentiation protocol is critical to the interaction of THP-1 macrophages with Salmonella Typhimurium. PLoS ONE 2018, 13, e0193601. [Google Scholar] [CrossRef]
- Traore, K.; Trush, M.A.; George, M., Jr.; Spannhake, E.W.; Anderson, W.; Asseffa, A. Signal transduction of phorbol 12-myristate 13-acetate (PMA)-induced growth inhibition of human monocytic leukemia THP-1 cells is reactive oxygen dependent. Leuk. Res. 2005, 29, 863–879. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, S.; Kobayashi, Y.; Goto, Y.; Okumura, H.; Nakae, S.; Konno, T.; Tada, K. Induction of maturation in cultured human monocytic leukemia cells by a phorbol diester. Cancer Res. 1982, 42, 1530–1536. [Google Scholar]
- Tsuchiya, S.; Yamabe, M.; Yamaguchi, Y.; Kobayashi, Y.; Konno, T.; Tada, K. Establishment and characterization of a human acute monocytic leukemia cell line (THP-1). Int. J. Cancer 1980, 26, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Jung, J.H.; Kim, J.Y. 1,25-Dihydroxyvitamin D3 up-regulates TLR10 while down-regulating TLR2, 4, and 5 in human monocyte THP-1. J. Steroid Biochem. Mol. Biol. 2014, 141, 1–6. [Google Scholar] [CrossRef]

| Target Gene | Forward Primer 5′- | Size (bp) | Reverse Primer 3′- | Size (bp) | Product Size (bp) |
|---|---|---|---|---|---|
| GAPDH | CTGCTCCTCCTGTTCGACAGT | 21 | CCGTTGACTCCGACCTTCAC | 23 | 100 |
| IL-6 | TGGCTGCAGGACATGACAAC | 20 | TGAGGTGCCCATGCTACATTT | 20 | 100 |
| IL-8 | TCAGAGACAGCAGAGCACACAA | 22 | GGCCAGCTTGGAAGTCATGT | 20 | 100 |
| IL-10 | AGGAGGTGATGCCCCAAGCTGA | 22 | TCGATGACAGCGCCGTAGCCT | 21 | 110 |
| TNFα | ACATCCAACCTTCCCAAACG | 20 | GCCCCCAATTCTCTTTTTGAG | 22 | 151 |
| ZO-1 | GCAATGGAGGAAACAGCTATATGG | 24 | TGAGGATTATCTCGTCCACCAGAT | 24 | 104 |
| TLR2 | GGCATGTGCTGTGCTCTGTT | 20 | GGAGCCAGGCCCACATC | 17 | 100 |
| TLR4 | AGCCCTTCACCCCGATTC | 18 | TAGAAATTCAGCTCCATGCATTG | 23 | 100 |
| TLR9 | GGACCTCTGGTACTGCTTCCA | 21 | AAGCTCGTTGTACACCCAGTCT | 22 | 151 |
| CD14 | ACCCTAGCGCTCCGAGATG | 19 | AGCTTGGCTGGCAGTCCTTT | 20 | 100 |
| MD-2 | TGCACATTTTCTACATTCCAAGGA | 24 | ATAACTTCTTTGCGCTTTGGAAGA | 24 | 100 |
| NOD2 | CAGAATTTCAAACGGCCTCACTA | 23 | ATGAAATGGAACTGCCTCTTGTG | 23 | 102 |
| Tollip | TCTCATGCCGTTCTGGAAAAT | 21 | TCACATCACAAAATGCCATGAA | 22 | 110 |
| Readout/Treatment | Unstimulated | TNFα | TNFα + LcS | p Values |
|---|---|---|---|---|
| IL-8 mRNA IL-8 Protein | 1.0 ± 0 15 ± 1 | 50 ± 1 25 ± 1 | 2700 ± 100 30 ± 2 | <0.01 ** ns |
| IL-6 mRNA IL-6 Protein | 1.0 ± 0 <7 ND | 3 ± 1 <7 ND | 4 ± 1 <7 ND | ns ns |
| TNFα mRNA TNFα Protein | 1.0 ± 0 13 ± 1 | 500 ± 10 75 ± 3 | 2300 ± 200 60 ± 5 | <0.001 *** <0.05 * |
| IL-10 mRNA IL-10 Protein | 1.0 ± 0 20 ± 1 | 100 ± 1 175 ± 20 | 2100 ± 1 190 ± 10 | <0.01 ** ns |
| Readout/Treatment | Unstimulated | IL-1β | IL-1β + LcS | p Values |
| IL-8 mRNA IL-8 Protein | 1.0 ± 0 15 ± 1 | 1000 ± 10 620 ± 20 | 2700 ± 100 780 ± 10 | <0.01 ** <0.05 * |
| IL-6 mRNA IL-6 Protein | 1.0 ± 0 <7 ND | 3 ± 1 35 ± 2 | 4 ± 1 75 ± 4 | <0.05 * <0.01 ** |
| TNFα mRNA TNFα Protein | 1.0 ± 0 15 ± 1 | 220 ± 10 55 ± 15 | 1100 ± 300 50 ± 3 | <0.001 *** <0.05 * |
| IL-10 mRNA IL-10 Protein | 1.0 ± 0 30 ± 1 | 10 ± 1 270 ± 10 | 2 ± 1 120 ± 20 | <0.05 * <0.001 *** |
| Treatment | TNFα mRNA | TNFα Protein |
|---|---|---|
| Unstimulated +anti-IL-10 | 1.0 ± 0 6.1 ± 0.7 p < 0.001 *** | 13 ± 1 315 ± 5 p < 0.001 *** |
| TNFα +anti-IL-10 | 4.0 ± 0.5 11.2 ± 0.4 p < 0.001 *** | 80 ± 10 710 ± 20 p < 0.001 *** |
| IL-1β +anti-IL-10 | 5.0 ± 2.0 0.8 ± 0.1 p < 0.001 *** | 330 ± 20 250 ± 15 p < 0.01 ** |
| Readout/Treatment | Control | +anti-IL-10 | p Values |
|---|---|---|---|
| TLR4 mRNA—Unstim | 1.0 ± 0 | 50 ± 1 | <0.001 *** |
| +TNFα | 5 ± 0.4 | 20 ± 0.5 | <0.05 * |
| +IL-1β | 20 ± 1 | 100 ± 1 | <0.05 * |
| TLR2 mRNA—Unstim | 1.0 ± 0 | 100 ± 5 | <0.001 *** |
| +TNFα | 5 ± 1 | 500 ± 10 | <0.001 *** |
| +IL-1β | 5 ± 1 | 900 ± 15 | <0.001 *** |
| CD14 mRNA—Unstim | 1.0 ± 0 | 70 ± 2 | <0.001 *** |
| +TNFα | 5 ± 1 | 120 ± 3 | <0.001 *** |
| +IL-1β | 70 ± 2 | 70 ± 1 | ns |
| MD2 mRNA—Unstim | 1.0 ± 0 | 3 ± 1 | <0.05 * |
| +TNFα | 0.8 ± 0.2 | 3 ± 1 | <0.05 * |
| +IL-1β | 4 ± 1.5 | 2 ± 1 | <0.05 * |
| NOD2 mRNA—Unstim | 1.0 ± 0 | 50 ± 6 | <0.001 *** |
| +TNFα | 15 ± 1 | 150 ± 5 | <0.01 ** |
| +IL-1β | 70 ± 2 | 500 ± 10 | <0.001 *** |
| TLR9 mRNA—Unstim | 1.0 ± 0 | 90 ± 2 | <0.001 *** |
| +TNFα | 30 ± 2 | 110 ± 10 | <0.001 *** |
| +IL-1β | 3 ± 1 | 30 ± 4 | <0.001 *** |
| Tollip mRNA—Unstim | 1.0 ± 0 | 0.001 ± 0.0002 | <0.001 *** |
| +TNFα | 0.05 ± 0.002 | 0.10 ± 0.04 | <0.05 * |
| +IL-1β | 0.01 ± 0.001 | 0.10 ± 0.03 | <0.05 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Foey, A.; Habil, N.; Strachan, A.; Beal, J. Lacticaseibacillus casei Strain Shirota Modulates Macrophage-Intestinal Epithelial Cell Co-Culture Barrier Integrity, Bacterial Sensing and Inflammatory Cytokines. Microorganisms 2022, 10, 2087. https://doi.org/10.3390/microorganisms10102087
Foey A, Habil N, Strachan A, Beal J. Lacticaseibacillus casei Strain Shirota Modulates Macrophage-Intestinal Epithelial Cell Co-Culture Barrier Integrity, Bacterial Sensing and Inflammatory Cytokines. Microorganisms. 2022; 10(10):2087. https://doi.org/10.3390/microorganisms10102087
Chicago/Turabian StyleFoey, Andrew, Neama Habil, Alex Strachan, and Jane Beal. 2022. "Lacticaseibacillus casei Strain Shirota Modulates Macrophage-Intestinal Epithelial Cell Co-Culture Barrier Integrity, Bacterial Sensing and Inflammatory Cytokines" Microorganisms 10, no. 10: 2087. https://doi.org/10.3390/microorganisms10102087
APA StyleFoey, A., Habil, N., Strachan, A., & Beal, J. (2022). Lacticaseibacillus casei Strain Shirota Modulates Macrophage-Intestinal Epithelial Cell Co-Culture Barrier Integrity, Bacterial Sensing and Inflammatory Cytokines. Microorganisms, 10(10), 2087. https://doi.org/10.3390/microorganisms10102087







