Antimicrobial Resistance and Biofilm Formation Capacity of Salmonella enterica Serovar Enteritidis Strains Isolated from Poultry and Humans in Poland
Abstract
1. Introduction
2. Results
2.1. Molecular Identification of Salmonella enterica ser. Enteritidis
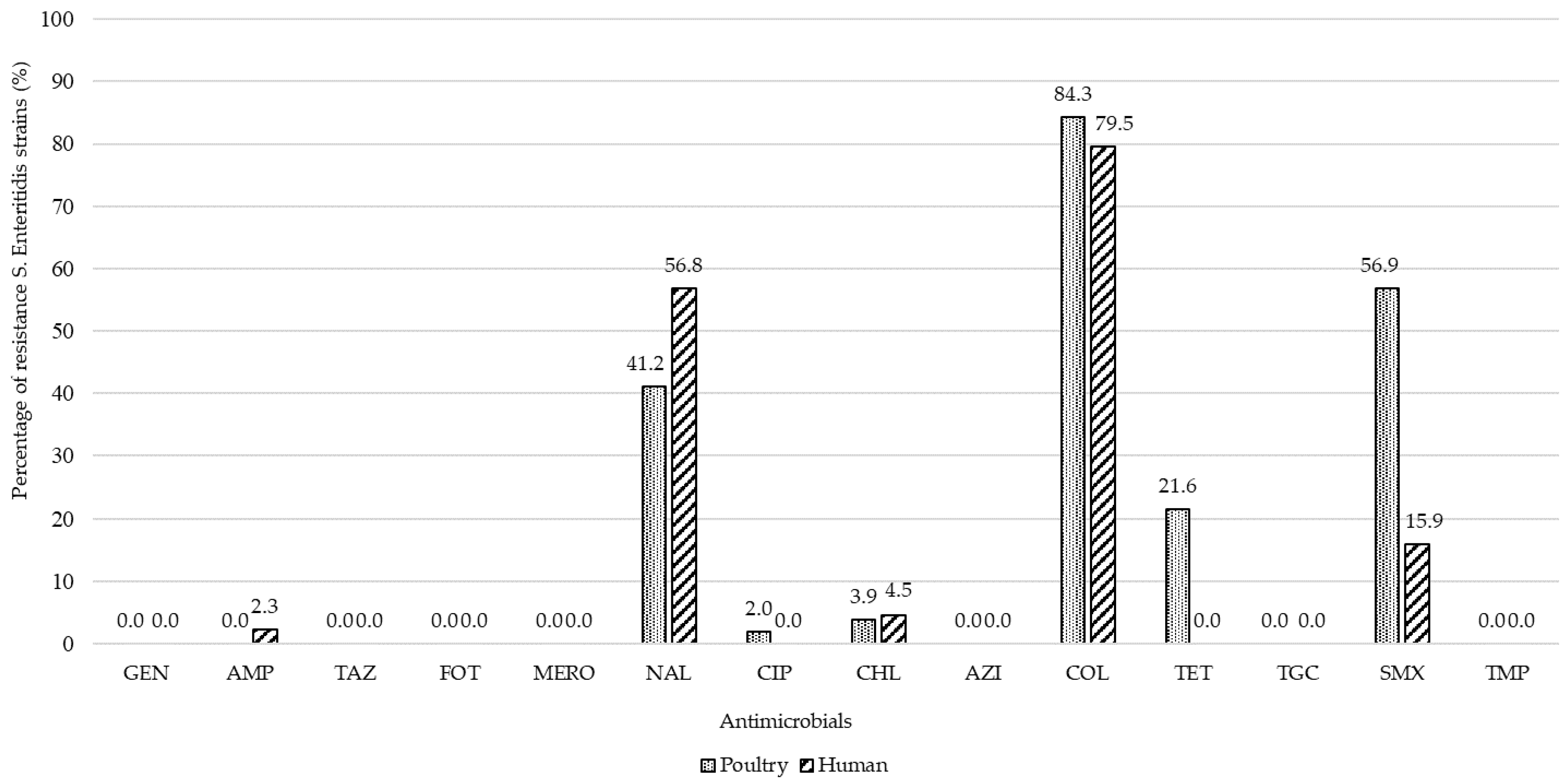
2.2. Antimicrobial Susceptibility and Resistance Gene Prevalence in Salmonella enterica ser. Enteritidis
2.3. Salmonella enterica ser. Enteritidis Biofilm Formation
2.3.1. Prevalence of Biofilm-Related Genes
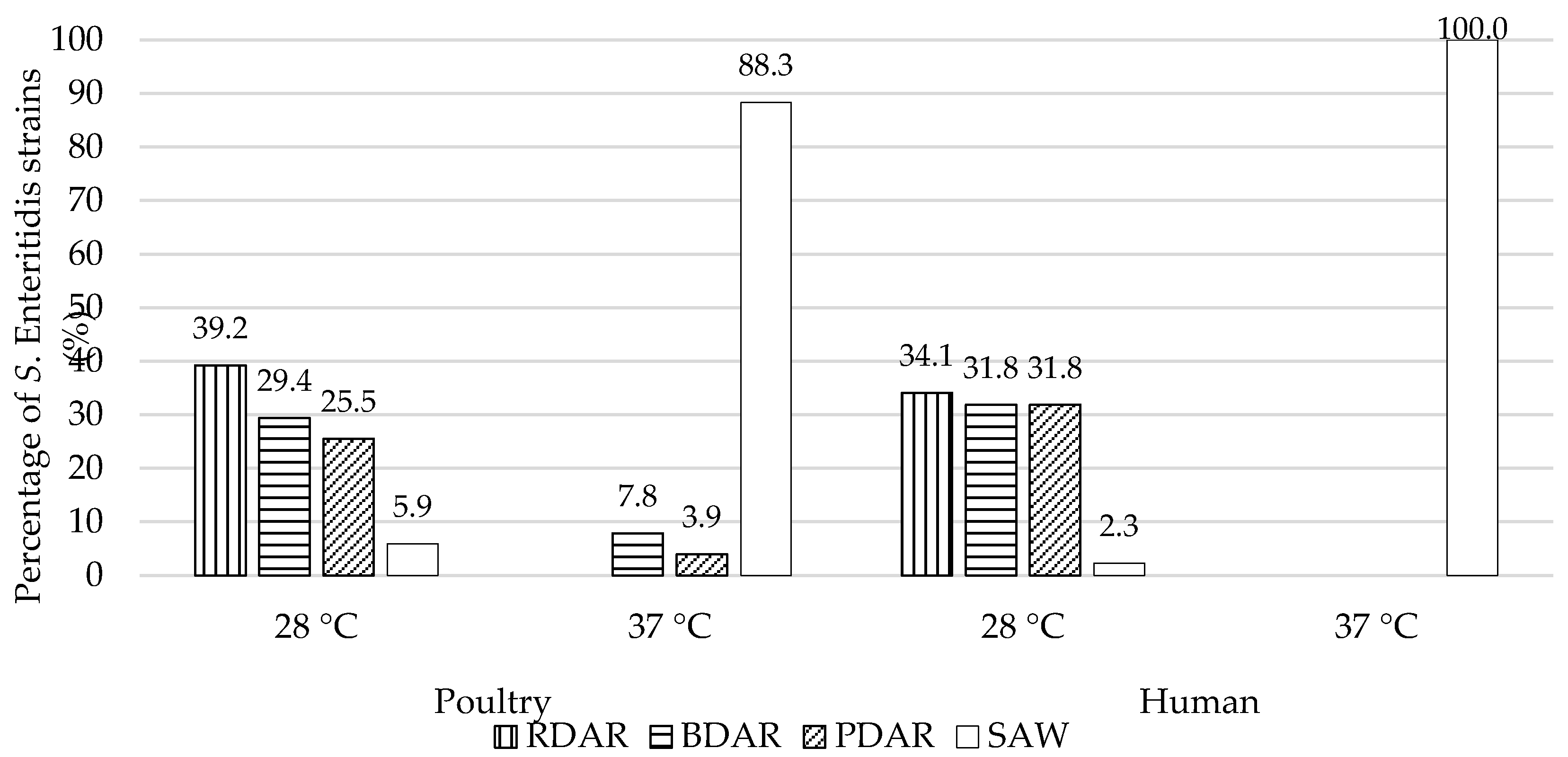
2.3.2. Assessment of Salmonella enterica ser. Enteritidis Morphotypes
2.3.3. Biofilm Formation Assay
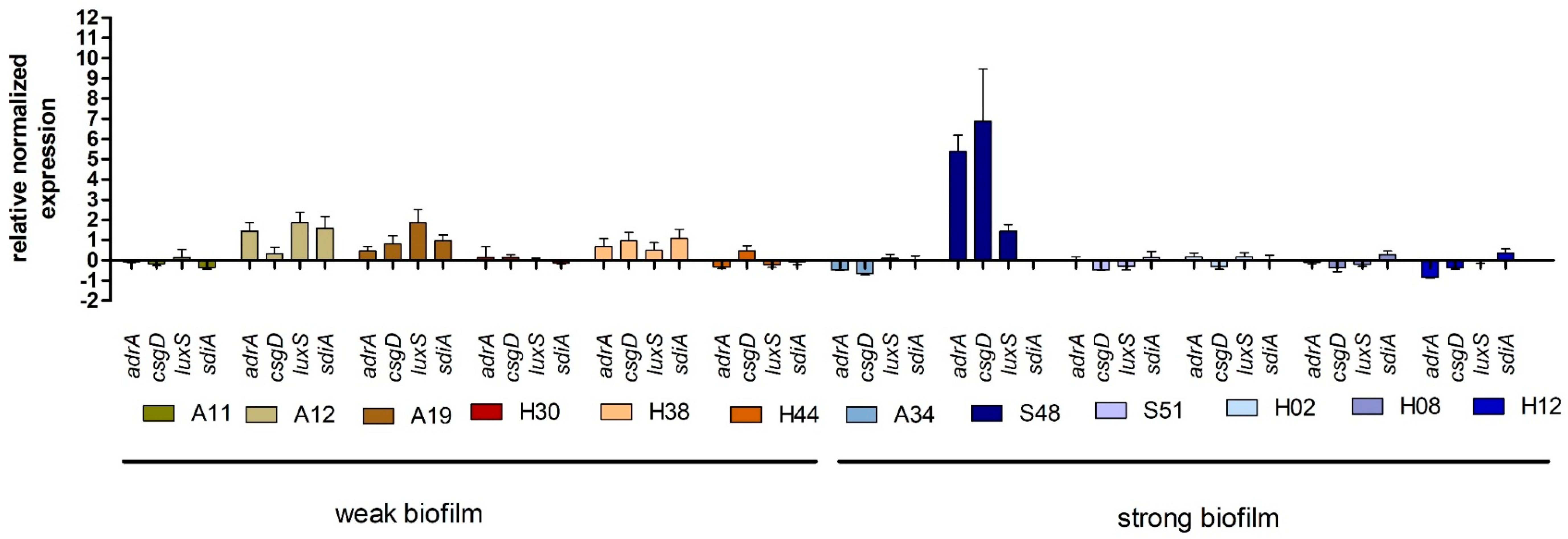
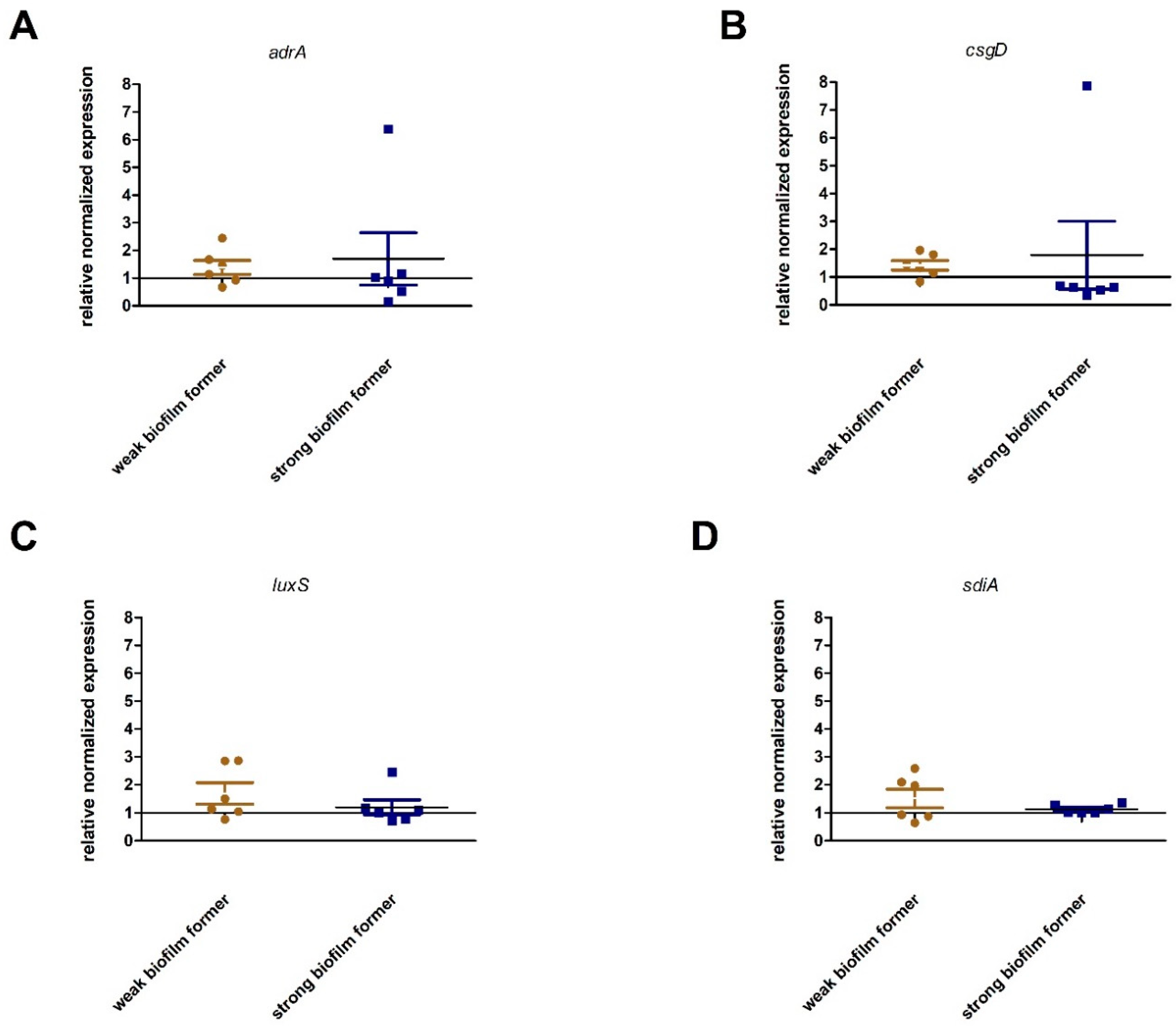
2.4. Expression of Genes Associated with Biofilm Production
2.5. Statistical Analysis
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains
- (1)
- Poultry S. enterica ser. Enteritidis strains (n = 51) isolated from the environment of chicken broilers, turkey broilers and laying hen farms. Isolates were obtained from two veterinary laboratories: AGRO-VET Laboratory in Wroclaw and SLW BIOLAB in Ostróda. These laboratories collected strains in the period 2009–2016. For our research, strains isolated from poultry are designated as “A” or “S”.
- (2)
- Human S. enterica ser. Enteritidis strains (n = 44) isolated from human fecal samples were collected in the period 2015–2017 by Dialab Laboratory in Wroclaw. Strains isolated from humans are designated as “H”.
4.2. Molecular Identification of Salmonella enterica ser. Enteritidis
4.3. Antimicrobial Susceptibility of Salmonella enterica ser. Enteritidis Strains
4.4. Antimicrobial Resistance Gene Detection
4.5. Salmonella enterica ser. Enteritidis Biofilm
4.5.1. Prevalence of Biofilm-Related Genes
4.5.2. Salmonella enterica ser. Enteritidis Morphotype on Congo Red Agar
4.5.3. Biofilm Formation Assay
4.5.4. Expression of Genes Associated with Biofilm Production
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. 2014 Antimicrobial Resistance Global Report on Surveillance. Available online: https://www.who.int/antimicrobial-resistance/publications/surveillancereport/en/ (accessed on 23 June 2020).
- European Food Safety Authority and European Centre for Disease Prevention and Control (EFSA and ECDC). The European Union One Health 2018 zoonoses report. EFSA J. 2019, 17, e05926. [Google Scholar]
- European Food Safety Authority, and European Centre for Disease Prevention and Control. The European Union Summary Report on Antimicrobial Resistance in zoonotic and indicator bacteria from humans, animals and food in 2017/2018. EFSA J. 2020, 18, e06007. [Google Scholar]
- Elbaz, A.; Elsherbini, M.; Abdelkhalek, A.; AlAshmawy, M. Prevalence and molecular characterization of Salmonella serovars in milk and cheese in Mansoura city, Egypt. J. Adv. Veter. Anim. Res. 2017, 4, 45–51. [Google Scholar] [CrossRef]
- Foley, S.L.; Lynne, A.M.; Nayak, R. Salmonella challenges: Prevalence in swine and poultry and potential pathogenicity of such isolates. J. Anim. Sci. 2008, 86, E149–E162. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.R.; Griffin, P.M.; Cole, D.; Walsh, K.A.; Chai, S.J. Outbreak-associated Salmonella enterica Serotypes and Food Commodities, United States, 1998–2008. Emerg. Infect. Dis. 2013, 19, 1239–1244. [Google Scholar] [CrossRef] [PubMed]
- Ung, A.; Baidjoe, A.Y.; Van Cauteren, D.; Fawal, N.; Fabre, L.; Guerrisi, C.; Danis, K.; Morand, A.; Donguy, M.-P.; Lucas, E.; et al. Disentangling a complex nationwide Salmonella Dublin outbreak associated with raw-milk cheese consumption, France, 2015 to 2016. Eurosurveillance 2019, 24, 1700703. [Google Scholar] [CrossRef] [PubMed]
- Stein, R.A. Super-spreaders in infectious diseases. Int. J. Infect. Dis. 2011, 15, e510–e513. [Google Scholar] [CrossRef]
- Woolhouse, M.E.; Dye, C.; Etard, J.-F.; Smith, T.; Charlwood, J.D.; Garnett, G.P.; Hagan, P.; Hii, J.L.K.; Ndhlovu, P.D.; Quinnell, R.J.; et al. Heterogeneities in the transmission of infectious agents: Implications for the design of control programs. Proc. Natl. Acad. Sci. USA 1997, 94, 338–342. [Google Scholar] [CrossRef]
- Pribul, B.R.; Festivo, M.L.; De Souza, M.M.S.; Rodrigues, D.D.P. Characterization of quinolone resistance in Salmonella spp. isolates from food products and human samples in Brazil. Braz. J. Microbiol. 2016, 47, 196–201. [Google Scholar] [CrossRef][Green Version]
- Ahmed, A.M.; Younis, E.E.; Ishida, Y.; Shimamoto, T. Genetic basis of multidrug resistance in Salmonella enterica serovars Enteritidis and Typhimurium isolated from diarrheic calves in Egypt. Acta Trop. 2009, 111, 144–149. [Google Scholar] [CrossRef]
- Ahmed, A.; Ishida, Y.; Shimamoto, T. Molecular characterization of antimicrobial resistance in Salmonella isolated from animals in Japan. J. Appl. Microbiol. 2009, 106, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Martínez, J.L.; Baquero, F. Interactions among Strategies Associated with Bacterial Infection: Pathogenicity, Epidemicity, and Antibiotic Resistance. Clin. Microbiol. Rev. 2002, 15, 647–679. [Google Scholar] [CrossRef] [PubMed]
- Wasyl, D.; Hoszowski, A.; Zając, M. Prevalence and characterisation of quinolone resistance mechanisms in Salmonella spp. Vet. Microbiol. 2014, 171, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Galiè, S.; García-Gutiérrez, C.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Biofilms in the Food Industry: Health Aspects and Control Methods. Front. Microbiol. 2018, 9, 898. [Google Scholar] [CrossRef] [PubMed]
- Solano, C.; Garcia, B.; Valle, J.; Berasain, C.; Ghigo, J.-M.; Gamazo, C.; Lasa, I. Genetic analysis of Salmonella enteritidis biofilm formation: Critical role of cellulose. Mol. Microbiol. 2002, 43, 793–808. [Google Scholar] [CrossRef] [PubMed]
- Barnhart, M.M.; Chapman, M.R. Curli biogenesis and function. Annu. Rev. Microbiol. 2006, 60, 131–147. [Google Scholar] [CrossRef]
- Römling, U.; Rohde, M.; Olsen, A.; Normark, S.; Reinkoster, J. AgfD, the checkpoint of multicellular and aggregative behaviour in Salmonella typhimurium regulates at least two independent pathways. Mol. Microbiol. 2000, 36, 10–23. [Google Scholar] [CrossRef]
- Römling, U.; Bokranz, W.; Rabsch, W.; Zogaj, X.; Nimtz, M.; Tschäpe, H. Occurrence and regulation of the multicellular morphotype in Salmonella serovars important in human disease. Int. J. Med. Microbiol. 2003, 293, 273–285. [Google Scholar] [CrossRef]
- Schonewille, E.; Windhorst, D.; Bräuni, I. Biofilm building properties of Salmonella on the poultry farm. Int. Poult. Prod. 2012, 20, 13–15. [Google Scholar]
- Wang, H.; Ding, S.; Dong, Y.; Ye, K.; Xu, X.; Zhou, G. Biofilm Formation of Salmonella Serotypes in Simulated Meat Processing Environments and Its Relationship to Cell Characteristics. J. Food Prot. 2013, 76, 1784–1789. [Google Scholar] [CrossRef]
- Lamas, A.; Regal, P.; Vazquez, B.; Cepeda, A.; Miranda, J.M.; Franco, C.M. Salmonella and Campylobacter biofilm formation: A comparative assessment from farm to fork. J. Sci. Food Agric. 2018, 98, 4014–4032. [Google Scholar] [CrossRef] [PubMed]
- Wasyl, D.; Hoszowski, A. Antimicrobial resistance in Salmonella isolated from animals and feed in Poland. Bull. Vet. Inst. Pulawy 2004, 48, 233–240. [Google Scholar]
- Łukasz, M.; Maćkiw, E.; Ścieżyńska, H.; Pawłowska, K.; Popowska, M. Antimicrobial susceptibility of Salmonella strains isolated from retail meat products in Poland between 2008 and 2012. Food Control. 2014, 36, 199–204. [Google Scholar]
- Łukasz, M.; Maćkiw, E.; Ścieżyńska, H.; Popowska, M. Occurrence and antimicrobial resistance of Salmonella spp. isolated from food other than meat in Poland. Ann. Agric. Environ. Med. 2015, 22, 403–408. [Google Scholar] [CrossRef]
- Skarzyńska, M.; Hoszowski, A.; Zając, M.; Lalak, A.; Samcik, I.; Kwit, R.; Wasyl, D. Distribution of Salmonella serovars along the food chain in Poland, 2010–2015. J. Vet. Res. 2017, 61, 173–179. [Google Scholar] [CrossRef]
- Witkowska, D.; Kuncewicz, M.; Żebrowska, J.P.; Sobczak, J.; Sowińska, J. Prevalence of Salmonella spp. in broiler chicken flocks in northern Poland in 2014–2016. Ann. Agric Environ. Med. 2018, 25, 693–697. [Google Scholar] [CrossRef]
- WHO. Integrated Surveillance of Antimicrobial Resistance in Foodborne Bacteria. Application of a One Health Approach. Available online: https://www.who.int/foodsafety/publications/agisar_guidance2017/en/ (accessed on 23 June 2020).
- Harb, A.; Habib, I.; Mezal, E.H.; Kareem, H.S.; Laird, T.; A O’Dea, M.; Abraham, S. Occurrence, antimicrobial resistance and whole-genome sequencing analysis of Salmonella isolates from chicken carcasses imported into Iraq from four different countries. Int. J. Food Microbiol. 2018, 284, 84–90. [Google Scholar] [CrossRef]
- Beshiru, A.; Igbinosa, I.H.; Igbinosa, E.O. Prevalence of Antimicrobial Resistance and Virulence Gene Elements of Salmonella Serovars from Ready-to-Eat (RTE) Shrimps. Front. Microbiol. 2019, 10, 1613. [Google Scholar] [CrossRef]
- De Oliveira, F.A.; Pasqualotto, A.P.; Da Silva, W.P.; Tondo, E.C. Characterization of Salmonella Enteritidis isolated from human samples. Food Res. Int. 2012, 45, 1000–1003. [Google Scholar] [CrossRef]
- Zhu, Y.; Lai, H.; Zou, L.; Yin, S.; Wang, C.; Han, X.; Xia, X.; Hu, K.; He, L.; Zhou, K.; et al. Antimicrobial resistance and resistance genes in Salmonella strains isolated from broiler chickens along the slaughtering process in China. Int. J. Food Microbiol. 2017, 259, 43–51. [Google Scholar] [CrossRef]
- Chuanchuen, R.; Padungtod, P. Antimicrobial resistance genes in Salmonella enterica isolates from poultry and swine in Thailand. J. Vet. Med. Sci. 2009, 71, 1349–1355. [Google Scholar] [CrossRef] [PubMed]
- Türkyılmaz, S.; Hazimoglu, S.; Bozdogan, B. Antimicrobial susceptibility and resistance genes in Salmonella enterica serovar enteritidis isolated from Turkeys. Isr. J. Vet. Med. 2009, 64, 72–77. [Google Scholar]
- Michael, G.B.; Schwarz, S. Microbial resistance in zoonotic nontyphoidal Salmonella: An alarming trend? Clin. Microbiol. Infect. 2016, 22, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Adesiji, Y.O.; Deekshit, V.K.; Karunasagar, I. Antimicrobial-resistant genes associated with Salmonella spp. isolated from human, poultry, and seafood sources. Food Sci. Nutr. 2014, 2, 436–442. [Google Scholar]
- Chen, S.; Zhao, S.; White, D.G.; Schroeder, C.M.; Lu, R.; Yang, H.; McDermott, P.F.; Ayers, S.; Meng, J. Characterization of Multiple-Antimicrobial-Resistant Salmonella Serovars Isolated from Retail Meats. Appl. Environ. Microbiol. 2004, 70, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Duffy, G.; Cloak, O.M.; O’Sullivan, M.G.; Guillet, A.; Sheridan, J.; Blair, I.; McDowell, D. The incidence and antibiotic resistance profiles of Salmonella spp. on Irish retail meat products. Food Microbiol. 1999, 16, 623–631. [Google Scholar] [CrossRef]
- Thong, K.L.; Modarressi, S. Antimicrobial resistant genes associated with Salmonella from retail meats and street foods. Food Res. Int. 2011, 44, 2641–2646. [Google Scholar] [CrossRef]
- FDA. Available online: https://www.fda.gov/animal-veterinary/national-antimicrobial-resistance-monitoring-system/2015-narms-integrated-report (accessed on 11 January 2017).
- Yaqoob, E.; Hussain, I.; Rahman, I.U. Molecular characterization by using random amplified polymorphic DNA (RAPD) analysis of Salmonella enteritidis isolates recovered from avian and human sources. Pak. Vet. J. 2007, 27, 102–104. [Google Scholar]
- Campioni, F.; Bergamini, A.M.M.; Falcão, J.P. Genetic diversity, virulence genes and antimicrobial resistance of Salmonella enteritidis isolated from food and humans over a 24-year period in Brazil. Food Microbiol. 2012, 32, 254–264. [Google Scholar] [CrossRef]
- Kim, J.H.; Cho, J.K.; Kim, K.S. Prevalence, and characterization of plasmid-mediated quinolone resistance genes in Salmonella isolated from poultry in Korea. Avian Pathol. 2013, 42, 221–229. [Google Scholar] [CrossRef]
- Figueiredo, R.; Card, R.M.; Nunez, J.; Pomba, C.; Mendonça, N.; Anjum, M.F.; Da Silva, G.J. Detection of an mcr-1-encoding plasmid mediating colistin resistance in Salmonella enterica from retail meat in Portugal: Table 1. J. Antimicrob. Chemother. 2016, 71, 2338–2340. [Google Scholar] [CrossRef]
- CLSI. Subcommittee on Antimicrobial Susceptibility Testing. In M100 Performance Standards for Antimicrobial Susceptibility Testing, 29th ed.; CLSI: Wayne, PA, USA, 2019. [Google Scholar]
- EUCAST. The European Committee on Antimicrobial Susceptibility Testing. In Breakpoint Tables for Interpretation of MICs and Zone Diameters; Version 9.0; EUCAST: Basel, Switzerland, 2019. [Google Scholar]
- Quesada, A.; Ugarte-Ruiz, M.; Iglesias, M.R.; Porrero, M.C.; Martínez, R.; Florez-Cuadrado, D.; Campos, M.J.; García, M.; Píriz, S.; Sáez, J.L.; et al. Detection of plasmid mediated colistin resistance (MCR-1) in Escherichia coli and Salmonella enterica isolated from poultry and swine in Spain. Res. Vet. Sci. 2016, 105, 134–135. [Google Scholar] [CrossRef] [PubMed]
- Carattoli, A.; Villa, L.; Feudi, C.; Curcio, L.; Orsini, S.; Luppi, A.; Pezzotti, G.; Magistrali, C.F. Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Eurosurveillance 2017, 22, 30589. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Graells, C.; Keersmaecker, S.C.; Vanneste, K.; Pochet, B.; Vermeersch, K.; Roosens, N.; Dierick, K.; Botteldoorn, N. Detection of Plasmid-Mediated Colistin Resistance, mcr-1 and mcr-2 Genes, in Salmonella spp. Isolated from Food at Retail in Belgium from 2012 to 2015. Foodborne Pathog. Dis. 2018, 15, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Litrup, E.; Kiil, K.; Hammerum, A.M.; Roer, L.; Nielsen, E.M.; Torpdahl, M. Plasmid-borne colistin resistance gene mcr-3 in Salmonella isolates from human infections, Denmark, 2009–2017. Eurosurveillance 2017, 22, 30587. [Google Scholar] [CrossRef] [PubMed]
- KORLD. Available online: http://korld.nil.gov.pl/pdf/strona_23-06-2020-Stanowisko_Zespolu_Roboczego.pdf (accessed on 23 June 2020).
- Su, L.-H.; Chiu, C.-H.; Chu, C.; Ou, J.T. Antimicrobial Resistance in Nontyphoid Salmonella Serotypes: A Global Challenge. Clin. Infect. Dis. 2004, 39, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Van Doren, J.M.; Kleinmeier, D.; Hammack, T.S.; Westerman, A. Prevalence, serotype diversity, and antimicrobial resistance of Salmonella in imported shipments of spice offered for entry to the United States, FY2007–FY2009. Food Microbiol. 2013, 34, 239–251. [Google Scholar] [CrossRef]
- EUR-Lex. Available online: https://eurlex.europa.eu/legalcontent/EN/TXT/PDF/?uri=CELEX:52001DC0333&from=EN (accessed on 20 June 2001).
- Ćwiek, K.; Bugla-Płoskońska, G.; Wieliczko, A. Salmonella biofilm development: Structure and significance. Adv. Hyg. Exp. Med. 2019, 73, 937–943. [Google Scholar] [CrossRef]
- Wang, H.; Ye, K.; Wei, X.; Cao, J.-X.; Xu, X.; Zhou, G. Occurrence, antimicrobial resistance and biofilm formation of Salmonella isolates from a chicken slaughter plant in China. Food Control. 2013, 33, 378–384. [Google Scholar] [CrossRef]
- Halatsi, K.; Oikonomou, I.; Lambiri, M.; Mandilara, G.; Vatopoulos, A.C.; Kyriacou, A. PCR detection of Salmonella spp. using primers targeting the quorum sensing gene sdiA. FEMS Microbiol. Lett. 2006, 259, 201–207. [Google Scholar] [CrossRef]
- Bhowmick, P.P.; Devegowda, D.; Ruwandeepika, H.D.; Fuchs, T.M.; Srikumar, S.; Karunasagar, I.; Karunasagar, I. gcpA (stm1987) is critical for cellulose production and biofilm formation on polystyrene surface by Salmonella enterica serovar Weltevreden in both high and low nutrient medium. Microb. Pathog. 2011, 50, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Solomon, E.B.; Niemira, B.A.; Sapers, G.M.; Annous, B.A. Biofilm Formation, Cellulose Production, and Curli Biosynthesis by Salmonella Originating from Produce, Animal, and Clinical Sources. J. Food Prot. 2005, 68, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Lamas, A.; Fernandez-No, I.C.; Miranda, J.M.; Vazquez, B.; Cepeda, A.; Franco, C.M. Biofilm Formation and Morphotypes of Salmonella enterica subsp. arizonae Differs from Those of Other Salmonella enterica Subspecies in Isolates from Poultry Houses. J. Food Prot. 2016, 79, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Stepanović, S.; Ćirković, I.; Ranin, L.; Švabic-Vlahović, M. Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett. Appl. Microbiol. 2004, 38, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Rawool, D.B.; Doijad, S.; Poharkar, K.; Mohan, V.; Barbuddhe, S.; Kolhe, R.; Kurkure, N.V.; Kumar, A.; Malik, S.; et al. Biofilm formation and genetic diversity of Salmonella isolates recovered from clinical, food, poultry and environmental sources. Infect. Genet. Evol. 2015, 36, 424–433. [Google Scholar] [CrossRef]
- Paz-Méndez, A.M.; Lamas, A.; Vazquez, B.; Miranda, J.M.; Cepeda, A.; Franco, C.M. Effect of Food Residues in Biofilm Formation on Stainless Steel and Polystyrene Surfaces by Salmonella enterica Strains Isolated from Poultry Houses. Foods 2017, 6, 106. [Google Scholar] [CrossRef]
- Donlan, R.M. Biofilms: Microbial Life on Surfaces. Emerg. Infect. Dis. 2002, 8, 881–890. [Google Scholar] [CrossRef]
- Wang, H.; Dong, Y.; Wang, G.; Xu, X.; Zhou, G. Effect of growth media on gene expression levels in Salmonella typhimurium biofilm formed on stainless steel surface. Food Control. 2016, 59, 546–552. [Google Scholar] [CrossRef]
- Silva, P.L.A.P.A.; Goulart, L.R.; Reis, T.F.M.; Mendonça, E.P.; De Melo, R.T.; Penha, V.A.S.; Peres, P.A.B.M.; Hoepers, P.G.; Beletti, M.; Fonseca, B.B. Biofilm Formation in Different Salmonella Serotypes Isolated from Poultry. Curr. Microbiol. 2018, 76, 124–129. [Google Scholar] [CrossRef]
- Lamas, A.; Miranda, J.M.; Vázquez, B.I.; Cepeda, A.; Franco, C.M. Biofilm formation, phenotypic production of cellulose and gene expression in Salmonella enterica decrease under anaerobic conditions. Int. J. Food Microbiol. 2016, 238, 63–67. [Google Scholar] [CrossRef]
- De Freitas, C.G.; Santana, A.P.; Da Silva, P.H.C.; Gonçalves, V.S.P.; Barros, M.D.A.F.; Torres, F.A.G.; Murata, L.S.; Perecmanis, S. PCR multiplex for detection of Salmonella Enteritidis, Typhi and Typhimurium and occurrence in poultry meat. Int. J. Food Microbiol. 2010, 139, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, L.; Wang, J.; Yassin, A.K.; Butaye, P.; Kelly, P.; Gong, J.; Guo, W.; Li, J.; Li, M.; et al. Molecular detection of colistin resistance genes (mcr-1, mcr-2 and mcr-3) in nasal/oropharyngeal and anal/cloacal swabs from pigs and poultry. Sci. Rep. 2018, 8, 1–9. [Google Scholar]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]


| Antimicrobial Agent | Antimicrobial Dilution Range (µg/mL) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.015 | 0.03 | 0.06 | 0.12 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | 16 | 32 | 64 | 128 | 256 | 512 | 1024 | |
| Poultry (n = 51) | |||||||||||||||||
| Gentamicin | x | x | x | x | x | 45 | 3 | 1 | 2 | x | x | x | x | x | |||
| Ampicillin | x | x | x | x | x | x | 11 | 35 | 4 | 1 | x | x | x | x | |||
| Tazobactam | x | x | x | x | x | 49 | 2 | x | x | x | x | x | x | x | |||
| Cefotaxime | x | x | x | x | 49 | 2 | x | x | x | x | x | x | x | x | |||
| Meropenem | x | 51 | x | x | x | x | x | x | |||||||||
| Nalidixic acid | x | x | x | x | x | x | x | x | 24 | 1 | 5 | 1 | 4 | (16 *) | x | x | x |
| Ciprofloxacin | 2 | 18 | 1 | 2 | 12 | 15 | 1 | x | x | x | x | x | x | x | |||
| Chloramphenicol | x | x | x | x | x | x | x | x | x | 49 | 1 | 1 | x | x | x | ||
| Azithromycin | x | x | x | x | x | x | x | 16 | 35 | x | x | x | x | ||||
| Colistin | x | x | x | x | x | x | 6 | 2 | 29 | 14 | x | x | x | x | x | x | |
| Tetracycline | x | x | x | x | x | x | x | 34 | 4 | 2 | 4 | 5 | 1(1 *) | x | x | x | x |
| Tigecycline | x | x | x | x | 36 | 15 | x | x | x | x | x | x | x | ||||
| Sulfamethoxazole | x | x | x | x | x | x | x | x | x | 11 | 8 | 2 | 1 | 1 | (28 *) | ||
| Trimethoprim | x | x | x | x | 12 | 36 | 12 | 1 | x | x | x | x | x | ||||
| Human (n = 44) | |||||||||||||||||
| Gentamicin | x | x | x | x | x | 39 | 5 | x | x | x | x | x | |||||
| Ampicillin | x | x | x | x | x | x | 5 | 35 | 3 | 1 | x | x | x | x | |||
| Tazobactam | x | x | x | x | x | 44 | x | x | x | x | x | x | x | ||||
| Cefotaxime | x | x | x | x | 44 | x | x | x | x | x | x | x | x | ||||
| Meropenem | x | 44 | x | x | x | x | x | x | |||||||||
| Nalidixic acid | x | x | x | x | x | x | x | x | 18 | 1 | 2 | 15 | (8 *) | x | x | x | |
| Ciprofloxacin | 1 | 18 | 2 | 1 | 13 | 9 | x | x | x | x | x | x | x | ||||
| Chloramphenicol | x | x | x | x | x | x | x | x | x | 42 | 2 | x | x | x | |||
| Azithromycin | x | x | x | x | x | x | x | 7 | 37 | x | x | x | x | ||||
| Colistin | x | x | x | x | x | x | 9 | 13 | 22 | x | x | x | x | x | x | ||
| Tetracycline | x | x | x | x | x | x | x | 37 | 6 | 1 | x | x | x | x | |||
| Tigecycline | x | x | x | x | 27 | 17 | x | x | x | x | x | x | x | ||||
| Sulfamethoxazole | x | x | x | x | x | x | x | x | x | 14 | 22 | 1 | (7 *) | ||||
| Trimethoprim | x | x | x | x | 12 | 30 | 2 | x | x | x | x | x | |||||
| No. | Multidrug Resistance Profile | S. enterica ser. Enteritidis Strains |
|---|---|---|
| 1 | NAL CHL COL TET SMX | A09 |
| 2 | NAL CIP COL SMX | A37 |
| 3 | NAL CHL COL SMX | A40 |
| 4 | NAL COL TET SMX | S47, S55, S56 |
| 5 | NAL COL SMX | A12, A41, A42, S60, H24, H34 |
| 6 | COL TET SMX | A22, A23, A25, A31 |
| 7 | CHL COL SMX | H33 |
| 8 | AMP NAL COL | H10 |
| Intensity at 28 °C | TSB | M9 | ||||||
| Poultry | Human | Poultry | Human | |||||
| n | % | n | % | n | % | n | % | |
| 0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| 1 | 4 | 7.8 | 6 | 13.6 | 12 | 23.5 | 25 | 56.8 |
| 2 | 34 | 66.7 | 30 | 68.2 | 31 | 60.8 | 18 | 40.9 |
| 3 | 13 | 25.5 | 8 | 18.2 | 8 | 15.7 | 1 | 2.3 |
| Intensity at 37 °C | TSB | M9 | ||||||
| Poultry | Human | Poultry | Human | |||||
| n | % | n | % | n | % | n | % | |
| 0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| 1 | 44 | 86.3 | 39 | 88.6 | 41 | 80.4 | 44 | 100.0 |
| 2 | 7 | 13.7 | 5 | 11.4 | 10 | 19.6 | 0 | 0.0 |
| 3 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| Intensity at 6 °C | TSB | M9 | ||||||
| Poultry | Human | Poultry | Human | |||||
| n | % | n | % | n | % | n | % | |
| 0 | 9 | 17.7 | 11 | 25.0 | 20 | 39.2 | 17 | 38.6 |
| 1 | 40 | 78.4 | 29 | 65.9 | 31 | 60.8 | 26 | 59.1 |
| 2 | 2 | 3.9 | 4 | 9.1 | 0 | 0.0 | 1 | 2.7 |
| 3 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| Gene | Sequence | PCR Product Size (bp) | Annealing Temperature (°C) | References |
|---|---|---|---|---|
| aadB | F: 5′-GAGCGAAATCTGCCGCTCTGG-3′ R: 5′-CTGTTACAACGGACTGGCCGC-3′ | 319 | 61 | [34] |
| aacC | F: 5′-GGCGCGATCAACGAATTTATCCGA-3′ R: 5′-CCATTCGATGCCGAAGGAAACGAT-3′ | 488 | 58 | |
| blaTEM | F: 5′-CATTTCCGTGTCGCCCTTAT-3′ R: 5′-TCCATAGTTGCCTGACTCCC-3′ | 793 | 55 | |
| blaPSE | F: 5′-AATGGCAATCAGCGCTTCCC-3′ R: 5′-GGGGCTTGATGCTCACTACA-3′ | 586 | 59 | |
| blaOXA | F: 5′-ACCAGATTCAACTTTCAA-3′ R: 5′-TCTTGGCTTTTATGCTTG-3′ | 590 | 55 | |
| tetA | F: 5′-GCTACATCCTGCTTGCCTTC-3′ R: 5′-CATAGATCGCCGTGAAGAGG-3′ | 210 | 55 | |
| tetB | F: 5′-TTGGTTAGGGGCAAGTTTTG-3′ R: 5′-GTAATGGGCCAATAACACCG-3′ | 659 | 53 | |
| tetC | F: 5′-CTTGAGAGCCTTCAACCCAG-3′ R: 5′-ATGGTCGTCATCTACCTGCC-3′ | 417 | 56 | |
| tetG | F: 5′-GCTCGGTGGTATCTCTGCTC-3′ R: 5′-AGCAACAGAATCGGGAACAC-3′ | 468 | 58 | |
| cat1 | F: 5′-CCTATAACCAGACCGTTCAG-3′ R: 5′-TCACAGACGGCATGATGAAC-3′ | 491 | 56 | |
| cat2 | F: 5′-CCGGATTGACCTGAATACCT-3′ R: 5′-TCACATACTGCATGATGAAC-3′ | 456 | 56 | |
| cat3 | F: 5′-CCCACAATTCACCGTATTCC-3′ R: 5′-GAACCTGTACTGAGAGCGGC-3′ | 310 | 58 | |
| floR | F: 5′-AACCCGCCCTCTGGATCAAGTCAA-3′ R: 5′-CAAATCACGGGCCACGCTGTATC-3′ | 548 | 60 | |
| mcr-1 | F: 5′-CGGTCAGTCCGTTTGTTC-3′ R: 5′-CTTGGTCGGTCTGTAGGG-3′ | 309 | 59 | [69] |
| mcr-2 | F: 5′-TGTTGCTTGTGCCGATTGGA-3′ R: 5′-AGATGGTATTGTTGGTTGCTG-3′ | 567 | 59 | |
| qnrA | F: 5′-ATTTCTCACGCCAGGATTTG-3′ R: 5′-GATCGGCAAAGGTTAGGTCA-3′ | 516 | 56 | [10] |
| qnrB | F: 5′-GATCGTGAAAGCCAGAAAGG-3′ R: 5′-ACGATGCCTGGTAGTTGTCC-3′ | 469 | 56 | |
| qnrC | F: 5′-GGGTTGTACATTTATTGAATCG-3′ R: 5′-CACCTACCCATTTATTTTCA-3′ | 307 | 54 | |
| qnrD | F: 5′-CGAGATCAATTTACGGGGAATA-3′ R: 5′-AACAAGCTGAAGCGCCTG-3′ | 582 | 52 | |
| qnrS | F: 5′-ACGACATTCGTCAACTGCAA-3′ R: 5′-TAAATTGGCACCCTGTAGGC-3′ | 417 | 56 |
| Gene/Region | Sequence |
|---|---|
| csgD | F: 5′-TCCTGGTCTTCAGTAGCGTAA-3′ R: 5′-TATGATGGA AGCGGATAAGAA-3′ |
| adrA | F: 5′-GAAGCTCGTCGCTGGAAGTC-3′ R: 5′-TTCCGCTTAATTTAATGGCCG-3′ |
| luxS | F: 5′-ATGCCATTATTAGATAGCTT-3′ R: 5′-GAGATGGTCGCGCATAAAGCCAGC-3′ |
| sdiA | F: 5′-AATATCGCTTCGTACCAC-3′ R: 5′-GTAGGTAAACGAGGAGCAG-3′ |
| 16S rRNA reference gene | F: 5′-AGGCCTTCGGGTTGTAAAGT-3′ R: 5′-GTTAGCCGGTGCTTCTTCTG-3′ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ćwiek, K.; Korzekwa, K.; Tabiś, A.; Bania, J.; Bugla-Płoskońska, G.; Wieliczko, A. Antimicrobial Resistance and Biofilm Formation Capacity of Salmonella enterica Serovar Enteritidis Strains Isolated from Poultry and Humans in Poland. Pathogens 2020, 9, 643. https://doi.org/10.3390/pathogens9080643
Ćwiek K, Korzekwa K, Tabiś A, Bania J, Bugla-Płoskońska G, Wieliczko A. Antimicrobial Resistance and Biofilm Formation Capacity of Salmonella enterica Serovar Enteritidis Strains Isolated from Poultry and Humans in Poland. Pathogens. 2020; 9(8):643. https://doi.org/10.3390/pathogens9080643
Chicago/Turabian StyleĆwiek, Katarzyna, Kamila Korzekwa, Aleksandra Tabiś, Jacek Bania, Gabriela Bugla-Płoskońska, and Alina Wieliczko. 2020. "Antimicrobial Resistance and Biofilm Formation Capacity of Salmonella enterica Serovar Enteritidis Strains Isolated from Poultry and Humans in Poland" Pathogens 9, no. 8: 643. https://doi.org/10.3390/pathogens9080643
APA StyleĆwiek, K., Korzekwa, K., Tabiś, A., Bania, J., Bugla-Płoskońska, G., & Wieliczko, A. (2020). Antimicrobial Resistance and Biofilm Formation Capacity of Salmonella enterica Serovar Enteritidis Strains Isolated from Poultry and Humans in Poland. Pathogens, 9(8), 643. https://doi.org/10.3390/pathogens9080643





