Evaluation of the Immunomodulatory Ability of Lactic Acid Bacteria Isolated from Feedlot Cattle Against Mastitis Using a Bovine Mammary Epithelial Cells In Vitro Assay
Abstract
1. Introduction
2. Results
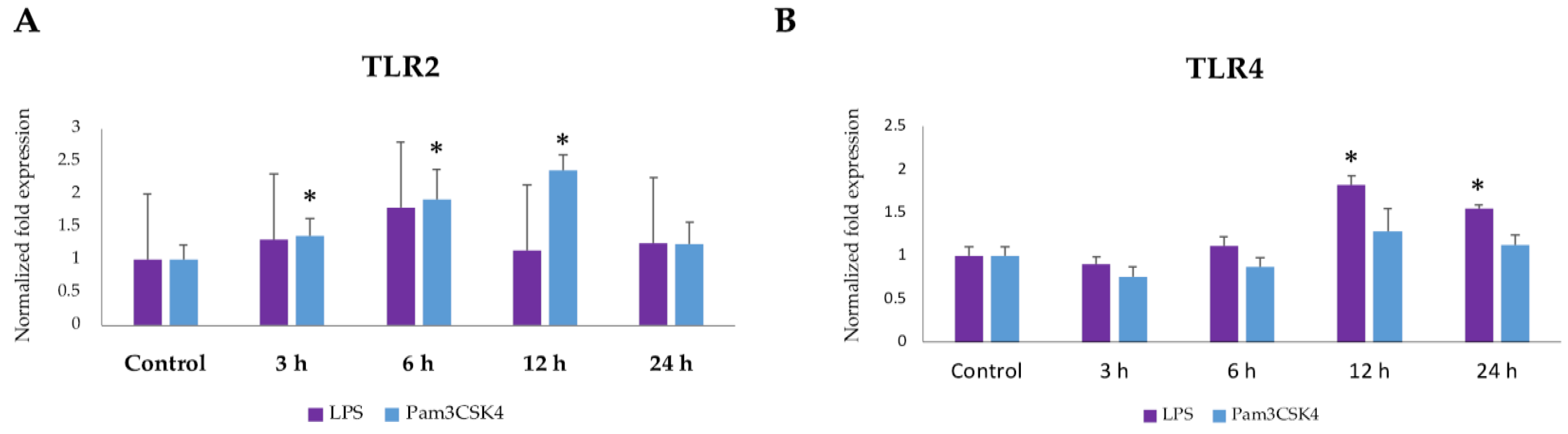
2.1. Expression Dynamics of TLR2 and TLR4 in BME Cells after Ligand Stimulation
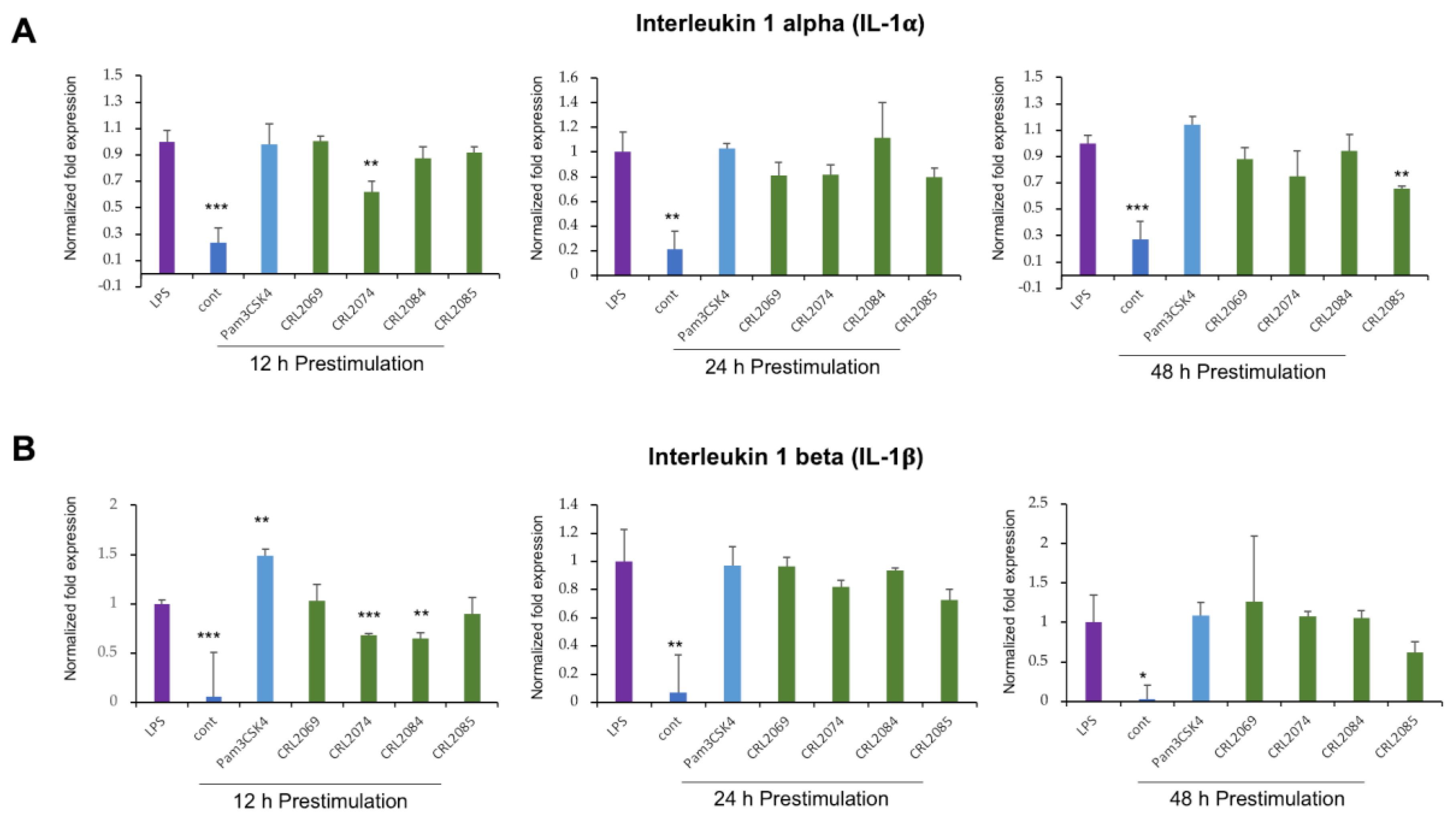
2.2. Modulation of LPS-Induced Cytokine Expression in BME Cells by Lactobacilli
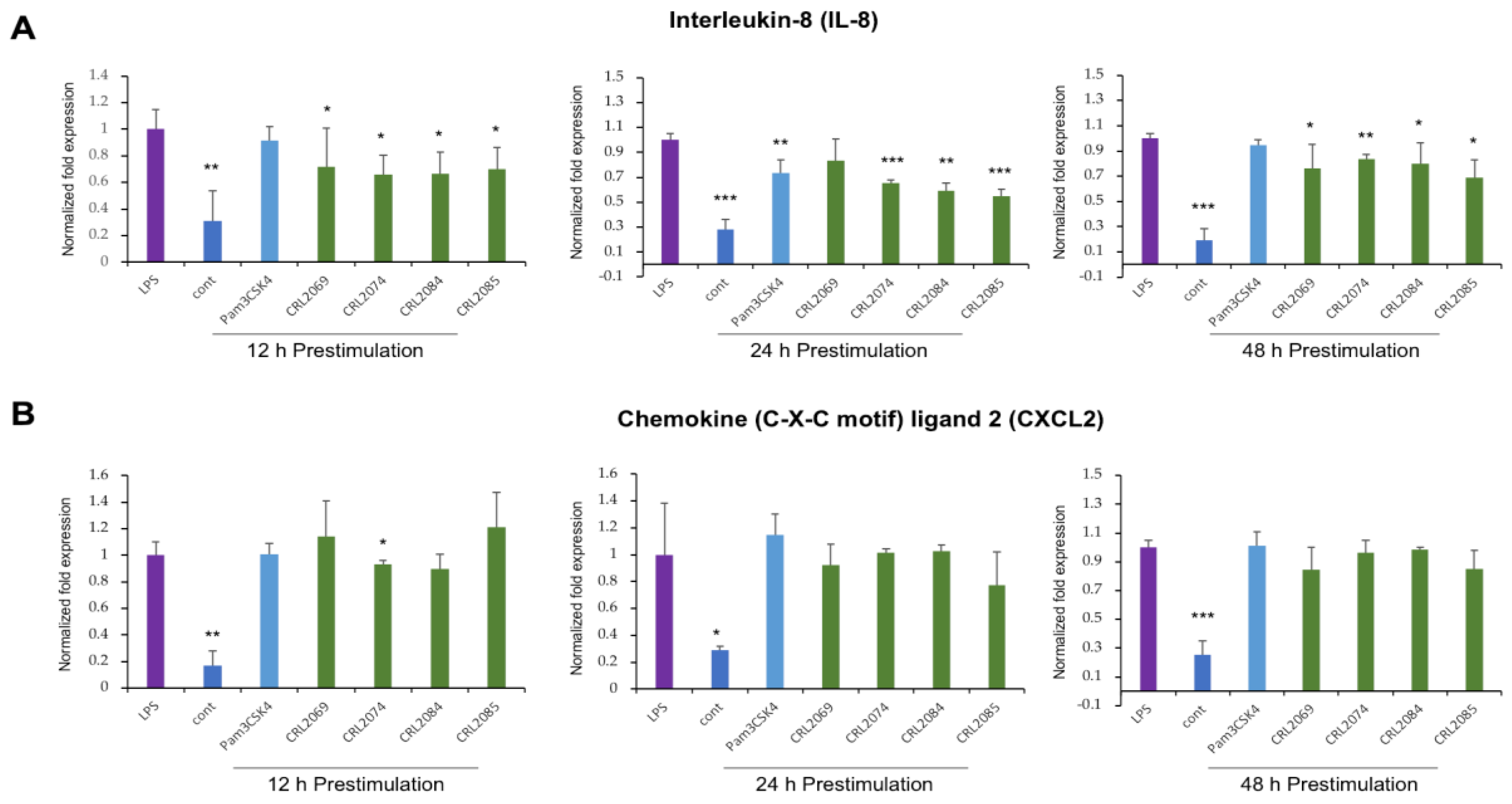
2.3. Modulation of LPS-Induced Nutrophil Chemoattractants in BME Cells by Lactobacilli
2.4. Modulation of LPS-Induced Monocyte Chemoattractants in BME Cells by Lactobacilli
2.5. Modulation of Negative Regulators of LPS-Induced Inflammation in BME Cells by Lactobacilli
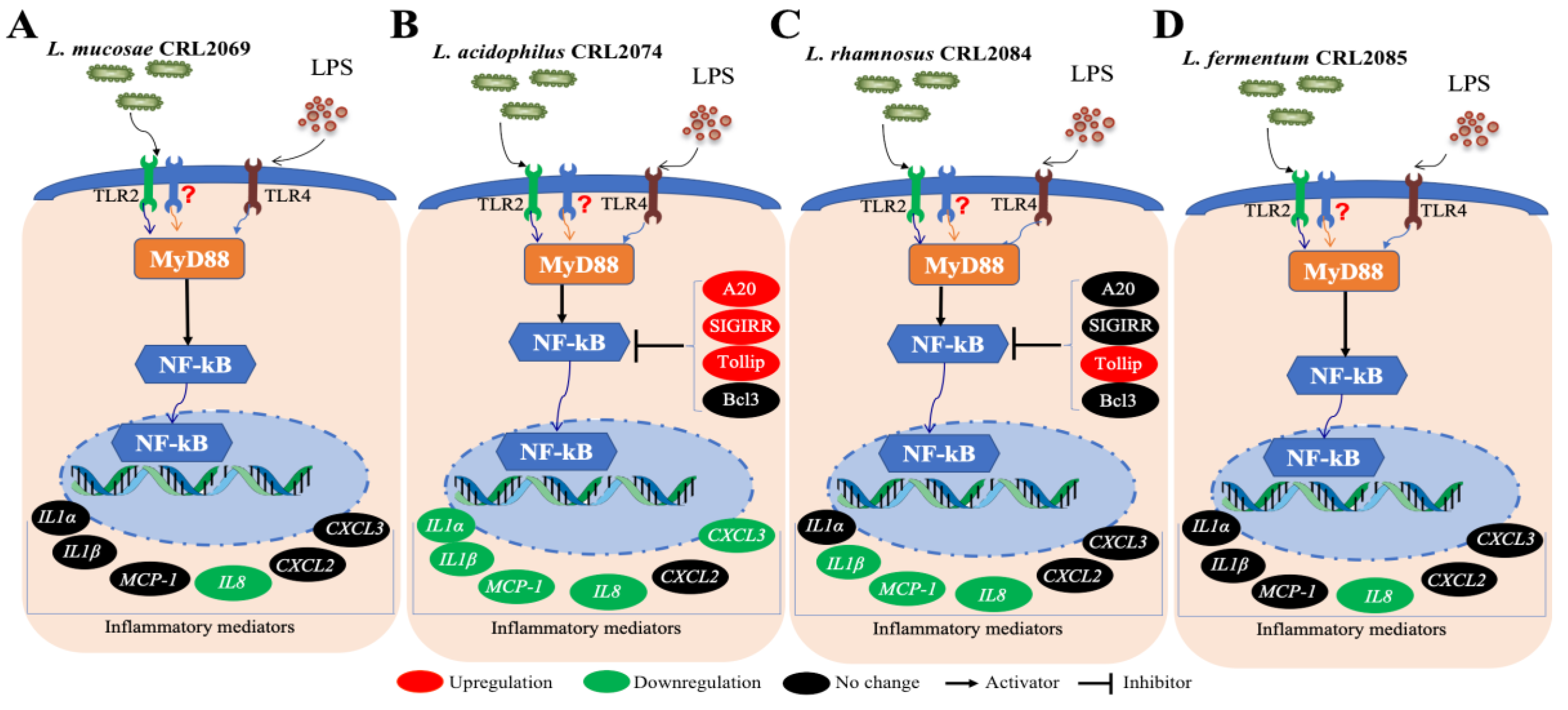
3. Discussion
4. Materials and Methods
4.1. Cell Line and Culture Condition
4.2. Growth and Maintenance of Microorganisms
4.3. Immunobiotic Evaluation Assay in BME Cells
4.4. Real-Time Quantitative PCR
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Halasa, T.; Huijps, K.; Østerås, O.; Hogeveen, H. Economic effects of bovine mastitis and mastitis management: A review. Vet. Q. 2007, 29, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.; Günther, J.; Talbot, R.; Petzl, W.; Zerbe, H.; Schuberth, H.J.; Seyfert, H.M.; Glass, E.J. Escherichia coli- and Staphylococcus aureus-induced mastitis differentially modulate transcriptional responses in neighbouring uninfected bovine mammary gland quarters. BMC Genom. 2013, 14, 36. [Google Scholar] [CrossRef] [PubMed]
- Schukken, Y.H.; Günther, J.; Fitzpatrick, J.; Fontaine, M.C.; Goetze, L.; Holst, O.; Leigh, J.; Petzl, W.; Schuberth, H.-J.; Sipka, A.; et al. Host-response patterns of intramammary infections in dairy cows. Vet. Immunol. Immunopathol. 2011, 144, 270–289. [Google Scholar] [CrossRef] [PubMed]
- Burvenich, C.; Van Merrid, V.; Mehrzad, J.; ez-Fraile, A.; Duchateau, L. Severity of E. coli mastitis is mainly determined by cow factors. Vet. Res. 2003, 34, 521–564. [Google Scholar] [CrossRef]
- Mitterhuemer, S.; Petzl, W.; Krebs, S.; Mehne, D.; Klanner, A.; Wolf, E.; Zerbe, H.; Blum, H. Escherichia coli infection induces distinct local and systemic transcriptome responses in the mammary gland. BMC Genom. 2010, 11, 138. [Google Scholar] [CrossRef]
- Taponen, S.; Jantunen, A.; Pyoerala, E.; Pyoerala, S. Efficacy of targeted 5 day combined parenteral and intramammary treatment of clinical mastitis caused by penicillin-susceptible or penicillin-resistant Staphylococcus aureus. Acta Vet. Scand. 2003, 44, 53–62. [Google Scholar] [CrossRef]
- Bradley, A.J.; Breen, J.E.; Payne, B.; Williams, P.; Green, M.J. The use of a cephalonium containing dry cow therapy and an internal teat sealant, both alone and in combination. J. Dairy Sci. 2010, 93, 1566–1577. [Google Scholar] [CrossRef]
- Bradley, A.J.; Breen, J.E.; Payne, B.; Green, M.J. A comparison of broad-spectrum and narrow-spectrum dry cow therapy used alone and in combination with a teat sealant. J. Dairy Sci. 2011, 94, 692–704. [Google Scholar] [CrossRef]
- Dalton, J.C. Antibiotic residue prevention in milk and dairy beef. West. Dairy News 2006, 6, 79. [Google Scholar]
- Saini, V.; McClure, J.T.; Léger, D.; Keefe, G.C.; Scholl, D.T.; Morck, D.W.; Barkema, H.W. Antimicrobial resistance profiles of common mastitis pathogens on Canadian dairy farms. J. Dairy Sci. 2012, 95, 4319–4332. [Google Scholar] [CrossRef]
- Cao, L.T.; Wu, J.Q.; Xie, F.; Hu, S.H.; Mo, Y. Efficacy of Nisin in treatment of clinical mastitis in lactating dairy cows. J. Dairy Sci. 2007, 90, 3980–3985. [Google Scholar] [CrossRef] [PubMed]
- FAO. WHO 2002 Guidelines for the Evaluation of Probiotics in Food; Food and Agriculture Organization of the United Nations and World Health Organization Working Group: Geneva, Switzerland, 2002. [Google Scholar]
- Villena, J.; Aso, H.; Rutten, V.P.M.G.; Takahashi, H.; van Eden, W.; Kitazawa, H. Immunobiotics for the Bovine Host: Their Interaction with Intestinal Epithelial Cells and Their Effect on Antiviral Immunity. Front. Immunol. 2018, 9, 326. [Google Scholar] [CrossRef]
- Uyeno, Y.; Shigemori, S.; Shimosato, T. Effect of Probiotics/Prebiotics on Cattle Health and Productivity. Microbes Environ. 2015, 30, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Chaimanee, V.; Sakulsingharoj, C.; Deejing, S.; Seetakoses, P.; Niamsup, P. Screening and characterization of bacteriocin-producing bacteria capable of inhibiting the growth of bovine mastitis. Maejo Int. J. Sci. Technol. 2009, 3, 43–52. [Google Scholar]
- Espeche, M.C.; Otero, M.C.; Sesma, F.; Nader-Macias, M.E.F. Screening of surface properties and antagonistic substances production by lactic acid bacteria isolated from the mammary gland of healthy and mastitic cows. Vet. Microbiol. 2009, 135, 346–357. [Google Scholar] [CrossRef]
- Crispie, F.; Alonso-Gomez, M.; O’Loughlin, C.; Klostermann, K.; Flynn, J.; Arkins, S.; Meaney, W.; Ross, R.P.; Hill, C. Intramammary infusion of a live culture for treatment of bovine mastitis: Effect of live lactococci on the mammary immune response. J. Dairy Res. 2008, 75, 374–384. [Google Scholar] [CrossRef]
- Beecher, C.; Daly, M.; Berry, D.P.; Klostermann, K.; Flynn, J.; Meaney, W.; Hill, C.; McCarthy, T.V.; Ross, R.P.; Giblin, L. Administration of a live culture of Lactococcus lactis DPC 3147 into the bovine mammary gland stimulates the local host immune response, particularly IL-1β and IL-8 gene expression. J. Dairy Res. 2009, 76, 340–348. [Google Scholar] [CrossRef]
- Camperio, C.; Armas, F.; Biasibetti, E.; Frassanito, P.; Giovannelli, C.; Spuria, L.; D’Agostino, C.; Tait, S.; Capucchio, M.T.; Marianelli, C. A mouse mastitis model to study the effects of the intramammary infusion of a food-grade Lactococcus lactis strain. PLoS ONE 2017, 12, 30184218. [Google Scholar] [CrossRef]
- Armas, F.; Camperio, C.; Marianelli, C. In Vitro Assessment of the Probiotic Potential of Lactococcus lactis LMG 7930 against Ruminant Mastitis-Causing Pathogens. PLoS ONE. 2017, 12, e0169543. [Google Scholar] [CrossRef]
- Klostermann, K.; Crispie, F.; Flynn, J.; Ross, R.P.; Hill, C.; Meaney, W.J. Intramammary infusion of a live culture of Lactococcus lactis for treatment of bovine mastitis: Comparison of antibiotic treatment in field trials. J. Dairy Res. 2008, 75, 365–373. [Google Scholar] [CrossRef]
- Pellegrino, M.; Berardo, N.; Giraudo, J.; Nader-Macias, M.E.F.; Bogni, C. Bovine mastitis prevention: Humoral and cellular response of dairy cows inoculated with lactic acid bacteria at the dry-off period. Benef Microbes 2017, 8, 589–596. [Google Scholar] [CrossRef]
- Diepers, A.; Krömker, V.; Zinke, C.; Wente, N.; Pan, L.; Paulsen, K.; Paduch, J.-H. In vitro ability of lactic acid bacteria to inhibit mastitis-causing pathogens. Sustain. Chem. Pharm. 2017, 5, 84–92. [Google Scholar] [CrossRef]
- Rose, T.M.; Aso, H.; Yonekura, S.; Komatsu, T.; Hagino, A.; Ozutsumi, K.; Obara, Y. In Vitro differentiation of a cloned bovine mammary epithelial cell. J. Dairy Res. 2002, 69, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.A.; Takagi, M.; Fukuyama, K.; Komatsu, R.; Albarracin, L.; Nochi, T.; Suda, Y.; Ikeda-Ohtsubo, W.; Rutten, V.; Eden, W.; et al. Transcriptome Analysis of The Inflammatory Responses of Bovine Mammary Epithelial Cells: Exploring Immunomodulatory Target Genes for Bovine Mastitis. Pathogens 2020, 9, 200. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, N.C.; Ficoseco, C.A.; Mansilla, F.A.; Melian, C.; Hebert, E.M.; Vignolo, G.M.; Nader-Macias, M.E.F. Identification, characterization and selection of autochthonous lactic acid bacteria as probiotic for feedlot cattle. Livest. Sci. 2018, 212, 99–110. [Google Scholar] [CrossRef]
- Aristimuño, F.C.; Mansilla, F.I.; Maldonado, N.C.; Miranda, H.; Nader-Macias, M.E.F.; Vignolo, G.M. Safety and Growth Optimization of Lactic Acid Bacteria Isolated from Feedlot Cattle for Probiotic Formula Design. Front. Microbiol. 2018, 9, 2220. [Google Scholar] [CrossRef]
- Mansilla, F.; Takagi, M.; Garcia-Castillo, V.; Aso, H.; Nader-Macias, M.E.; Vignolo, G.; Kitazawa, H.; Villena, J. Modulation of Toll-like receptor-mediated innate immunity in bovine intestinal epithelial cells by lactic acid bacteria isolated from feedlot cattle. Benef. Microbes 2020, in press. [Google Scholar] [CrossRef]
- Oliver, S.P.; Murinda, S.E. Antimicrobial resistance of mastitis pathogens. Vet. Clin. N. Am. Food Anim. Pract. 2012, 28, 165–185. [Google Scholar] [CrossRef]
- McGrath, M.F. A novel system for mammary epithelial cell culture. J. Dairy Sci. 1987, 70, 1967–1980. [Google Scholar] [CrossRef]
- Takanashi, N.; Tomosada, Y.; Villena, J.; Murata, K.; Takahashi, T.; Chiba, E.; Tohno, M.; Shimazu, T.; Aso, H.; Suda, Y.; et al. Advanced application of bovine intestinal epithelial cell line for evaluating regulatory effect of lactobacilli against heat-killed enterotoxigenic Escherichia coli-mediated inflammation. BMC Microbiol. 2013, 13, 54. [Google Scholar] [CrossRef]
- Kobayashi, H.; Kanmani, P.; Ishizuka, T.; Miyazaki, A.; Soma, J.; Albarracin, L.; Suda, Y.; Nochi, T.; Aso, H.; Iwabuchi, N.; et al. Development of an in vitro immunobiotic evaluation system against rotavirus infection in bovine intestinal epitheliocytes. Benef. Microbes 2017, 8, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Castillo, V.; Albarracin, L.; Kitazawa, H.; Villena, J. Screening and characterization of immunobiotic Lactic Acid bacteria with porcine immunoassay systems. In Lactic Acid Bacteria. Methods in Molecular Biology; Kanauchi, M., Ed.; Humana Press: New York, NY, USA, 2019; Volume 1887. [Google Scholar]
- Miyazawa, K.; Hondo, T.; Kanaya, T.; Tanaka, S.; Takakura, I.; Itani, W.; Rose, M.T.; Kitazawa, H.; Yamaguchi, T.; Aso, H. Characterization of newly established bovine intestinal epithelial cell line. Histochem. Cell Biol. 2010, 133, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Lie, M.-C.; Yang, J.; Wang, J.-F.; Zhu, W.-H. Lactobacillus rhamnosus GR-1 Ameliorates Escherichia coli-Induced Inflammation and Cell Damage via Attenuation of ASC-Independent NLRP3 Inflammasome Activation. Appl. Environ. Microbiol. 2016, 82, 1173–1182. [Google Scholar]
- Taranu, I.; Martin, D.E.; Braicu, C.; Pistol, G.C.; Sorescu, I.; Pruteanu, L.L.; Neagoe, I.B.; Vodnar, D.C. In Vitro Transcriptome Response to a Mixture of Lactobacilli Strains in Intestinal Porcine Epithelial Cell Line. Int. J. Mol. Sci. 2018, 19, 1923. [Google Scholar] [CrossRef] [PubMed]
- Viguier, C.; Arora, S.; Gilmartin, N.; Welbeck, K.; O’Kennedy, R. Mastitis detection: Current trends and future perspectives. Trends Biotechnol. 2009, 27, 486–493. [Google Scholar] [CrossRef]
- Rainard, P.; Riollet, C. Innate immunity of the bovine mammary gland. Vet. Res. 2006, 37, 369–400. [Google Scholar] [CrossRef]
- Kopp, E.; Medzhitov, R. Recognition of microbial infection by Toll-like receptors. Curr. Opin. Immunol. 2003, 15, 396–401. [Google Scholar] [CrossRef]
- Paape, M.; Mehrzad, J.; Zhao, X.; Detilleux, J.; Burvenich, C. Defense of the bovine mammary gland by polymorphonuclear neutrophil leukocytes. J. Mammary Gland Biol. 2002, 7, 109–121. [Google Scholar] [CrossRef]
- De Galdeano, C.M.; LeBlanc, A.d.M.; Vinderola, G.; Bonet, M.E.B.; Perdigon, G. Minireview. Proposed model: Mechanisms of immunomodulation induced by probiotic bacteria. Clin. Vaccine Immunol. 2007, 14, 485–492. [Google Scholar] [CrossRef]
- Liew, F.Y.; Xu, D.; Brint, E.K.; O’Neill, L.A. Negative regulation of toll-like receptor-mediated immune responses. Nat. Rev. Immunol. 2005, 5, 446–458. [Google Scholar] [CrossRef]
- Liu, M.; Song, S.; Li, H.; Jiang, X.; Yin, P.; Wan, C.; Liu, X.; Liu, F.; Xu, J. The protective effect of caffeic acid against inflammation injury of primary bovine mammary epithelial cells induced by lipopolysaccharide. J. Dairy Sci. 2014, 97, 2856–2865. [Google Scholar] [CrossRef] [PubMed]
- Oviedo-Boyso, J.; Valdez-Alarcón, J.J.; Cajero-Juárez, M.; Ochoa-Zarzosa, A.; López-Meza, J.E.; Bravo-Pantino, A.; Baizabal-Aguirre, V.M. Innate immune response of bovine mammary gland to pathogenic bacteria responsible for mastitis. J. Infect. 2007, 54, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.Y.; Li, Y.; Kumagai, Y.; Xu, Y.; Weinstein, J.S.; Kellner, E.S.; Nacionales, D.C.; Butfiloski, E.J.; van-Rooijen, N.; Akira, S.; et al. Type I interferon modulates monocyte recruitment and maturation in chronic inflammation. Am. J. Pathol. 2009, 175, 2023–2033. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Lim, W.; Bae, H.; Bazer, F.W.; Song, G. C-C motif chemokine ligand 2 induces proliferation and prevents lipopolysaccharide-induced inflammatory responses in bovine mammary epithelial cells. J. Dairy Sci. 2018, 101, 4527–4541. [Google Scholar] [CrossRef]
- Vereecke, L.; Sze, M.; Mc Guire, C.; Rogiers, B.; Chu, Y.; Schmidt-Supprian, M.; Pasparakis, M.; Beyaert, R.; van Loo, G. Enterocyte-specific A20 deficiency sensitizes to tumor necrosis factor-induced toxicity and experimental colitis. J. Exp. Med. 2010, 207, 1513–1523. [Google Scholar] [CrossRef]
- Tomosada, Y.; Villena, J.; Murata, K.; Chiba, E.; Shimazu, T.; Aso, H.; Iwabuchi, N.; Xiao, J.-X.; Saito, T.; Kitazawa, H. Immunoregulatory Effect of Bifidobacteria Strains in Porcine Intestinal Epithelial Cells through Modulation of Ubiquitin-Editing Enzyme A20 Expression. PLoS ONE 2013, 8, e59259. [Google Scholar] [CrossRef]
- Ibeagha-Awemu, E.M.; Lee, J.W.; Ibeagha, A.E.; Bannerman, D.D.; Paape, M.J.; Zhao, X. Bacterial lipopolysaccharide induces increased expression of toll-like receptor (TLR) 4 and downstream TLR signaling molecules in bovine mammary epithelial cells. Vet. Res. 2008, 39, 11. [Google Scholar] [CrossRef]
- Frola, I.D.; Pellegrino, M.S.; Espeche, M.C.; Giraudo, J.A.; Nader-Macias, M.E.F.; Bogni, C.I. Effects of intramammary inoculation of Lactobacillus perolens CRL1724 in lactating cows udders. J. Dairy Res. 2011, 78, 1–9. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]


| Species | Strain ID | Source of Origin |
|---|---|---|
| Lactobacillus mucosae | CRL2069 | cattle feces/pen soil/feed rations |
| Lactobacillus acidophilus | CRL2074 | cattle feces/pen soil/feed rations |
| Lactobacillus rhamnosus | CRL2084 | cattle feed rations |
| Lactobacillus fermentum | CRL2085 | cattle feed rations |
| Gene Symbol | Sequence (3′ to 5′) * | Amplicon Size | Accession Number |
|---|---|---|---|
| ACTB | F: TGG ATT GGC GGC TCC AT R: GCT GAT CCA CAT CTG CTG GAA | 57 | NM_173979.3 |
| IL-1α | F: CAG TTG CCC ATC CAA AGT TGT T R: TGC CAT GTG CAC CAA TTT TT | 59 | NM_174092.1 |
| IL-1β | F: GAG CCT GTC ATC TTC GAA ACG R: GCA CGG GTG CGT CAC A | 55 | NM_174093.1 |
| CCL2 | F: CAC CAG CAG CAA GTG TCC TAA A R: CAC ATA ACT CCT TGC CCA GGA T | 65 | NM_174006.2 |
| CXCL2 | F: CTA GGC CAG CTC TAA CTG AC R: TGG TGA TTC CTC TTT TCC CT | 107 | NM_174299.3 |
| CXCL3 | F: GAC AGT TCC TGA AAA GTG GT R: ATA GTC CAG CAC ATC AAG TC | 104 | NM_001046513.2 |
| IL-8 | F: TGC TCT CTT GGC AGC TTT CC R: TCT TGA CAG AAC TGC AGC TTC AC | 61 | NM_173925.2 |
| TLR2 | F: GGG TGC TGT GTC ACC GTT TC R: GCC ACG CCC ACA TCA TCT | 57 | NM_174197.2 |
| TLR4 | F: AGC ACC TAT GAT GCC TTT GTC A R: GTT CAT TCC GCA CCC AGT CT | 61 | NM_174198.6 |
| A20 | F: AAAGTGGGCTGCATGTACTTTGG R: AGGCTGTGGGACTGGCTTTC | 121 | NM_001192170.1 |
| SIGIRR | F: GGCAGTGAAGTGGATGTGTCA R: TCCGTGCGGGCACTGTA | 56 | NM_001082443 |
| Tollip | F: CGGGCGTGGACTCTTTCTAC R: GATGCGGTCGTCCATGGA | 65 | NM_001039961 |
| Bcl3 | F: CATGGAACACCCCCTGTCA R: GGCGTATCTCCATCCTCATCA | 66 | NM_001205993 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukuyama, K.; Islam, M.A.; Takagi, M.; Ikeda-Ohtsubo, W.; Kurata, S.; Aso, H.; Vignolo, G.; Villena, J.; Kitazawa, H. Evaluation of the Immunomodulatory Ability of Lactic Acid Bacteria Isolated from Feedlot Cattle Against Mastitis Using a Bovine Mammary Epithelial Cells In Vitro Assay. Pathogens 2020, 9, 410. https://doi.org/10.3390/pathogens9050410
Fukuyama K, Islam MA, Takagi M, Ikeda-Ohtsubo W, Kurata S, Aso H, Vignolo G, Villena J, Kitazawa H. Evaluation of the Immunomodulatory Ability of Lactic Acid Bacteria Isolated from Feedlot Cattle Against Mastitis Using a Bovine Mammary Epithelial Cells In Vitro Assay. Pathogens. 2020; 9(5):410. https://doi.org/10.3390/pathogens9050410
Chicago/Turabian StyleFukuyama, Kohtaro, Md. Aminul Islam, Michihiro Takagi, Wakako Ikeda-Ohtsubo, Shoichiro Kurata, Hisashi Aso, Graciela Vignolo, Julio Villena, and Haruki Kitazawa. 2020. "Evaluation of the Immunomodulatory Ability of Lactic Acid Bacteria Isolated from Feedlot Cattle Against Mastitis Using a Bovine Mammary Epithelial Cells In Vitro Assay" Pathogens 9, no. 5: 410. https://doi.org/10.3390/pathogens9050410
APA StyleFukuyama, K., Islam, M. A., Takagi, M., Ikeda-Ohtsubo, W., Kurata, S., Aso, H., Vignolo, G., Villena, J., & Kitazawa, H. (2020). Evaluation of the Immunomodulatory Ability of Lactic Acid Bacteria Isolated from Feedlot Cattle Against Mastitis Using a Bovine Mammary Epithelial Cells In Vitro Assay. Pathogens, 9(5), 410. https://doi.org/10.3390/pathogens9050410








