Rickettsia Detected in the Reptile Tick Bothriocroton hydrosauri from the Lizard Tiliqua rugosa in South Australia
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Sample Collection
3.2. DNA Extraction
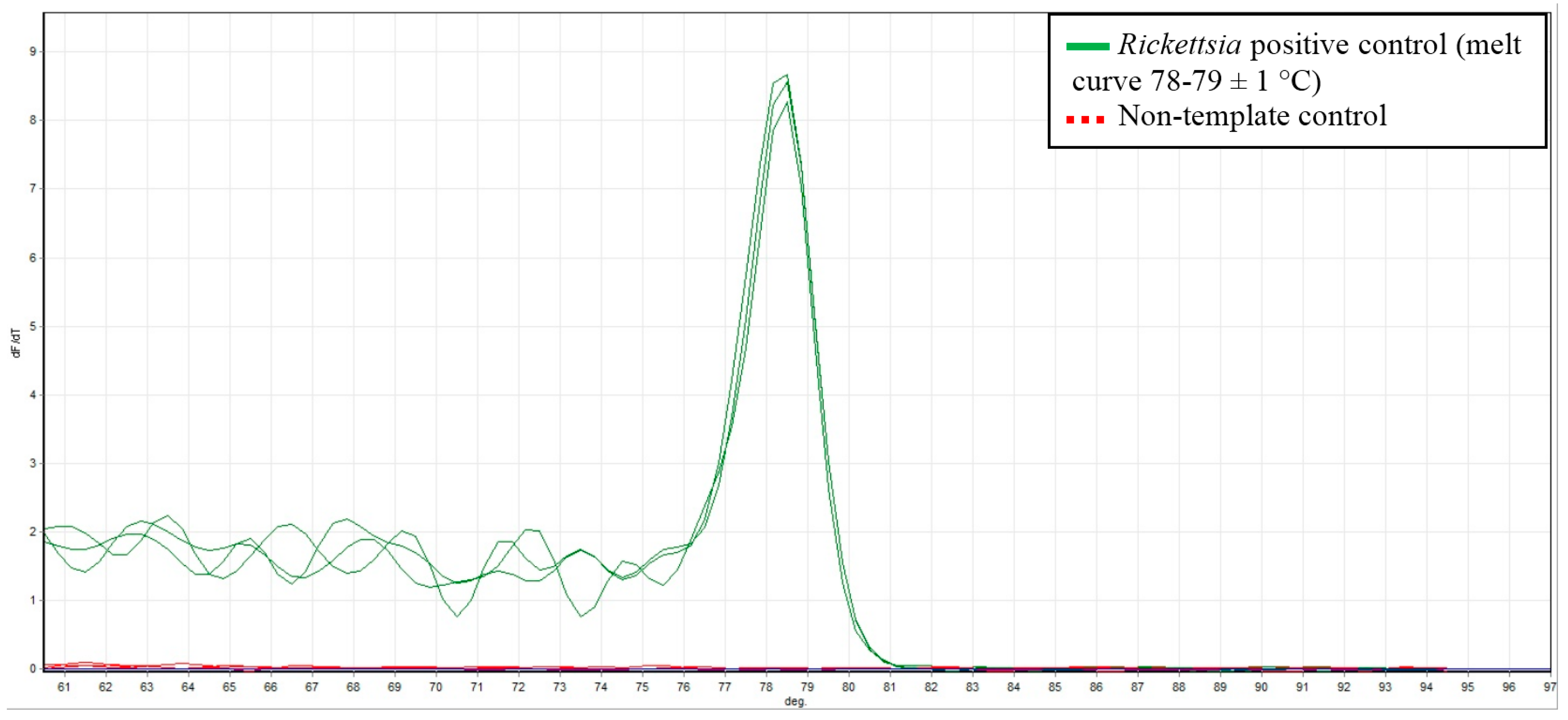
3.3. PCR Detection
3.4. Sequencing of Positive Samples
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and tick-borne diseases: A one health perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Parola, P.; Paddock, C.D.; Socolovschi, C.; Labruna, M.B.; Mediannikov, O.; Kernif, T.; Abdad, M.Y.; Stenos, J.; Bitam, I.; Fournier, P.-E.; et al. Update on tick-borne rickettsioses around the world: A geographic approach. Clin. Microbiol. Rev. 2013, 26, 657–702. [Google Scholar] [CrossRef] [PubMed]
- Derne, B.; Weinstein, P.; Musso, D.; Lau, C. Distribution of rickettsioses in oceania: Past patterns and implications for the future. Acta Trop. 2015, 143, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Fournier, P.E.; Raoult, D. Current knowledge on phylogeny and taxonomy of Rickettsia spp. Ann. N. Y. Acad. Sci. 2009, 1166, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Unsworth, N.B.; Stenos, J.; McGregor, A.R.; Dyer, J.R.; Graves, S.R. Not only ‘Flinders Island’spotted fever. Pathology 2005, 37, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Abdad, M.Y.; Cook, A.; Dyer, J.; Stenos, J.; Fenwick, S.G. Seroepidemiological study of outdoor recreationists’ exposure to spotted fever group Rickettsia in Western Australia. Am. J. Trop. Med. Hyg. 2014, 91, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Murphy, H.; Renvoisé, A.; Pandey, P.; Parola, P.; Raoult, D. Rickettsia honei infection in human, Nepal, 2009. Emerg. Infect. Dis. 2011, 17, 1865. [Google Scholar] [CrossRef] [PubMed]
- Graves, S.; Stenos, J. Rickettsia honei. Ann. N. Y. Acad. Sci. 2003, 990, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Stenos, J.; Graves, S.; Popov, V.L.; Walker, D.H. Aponomma hydrosauri, the reptile associated tick reservior of Rickettsia honei on Flinders Island, Australia. Am. J. Trop. Med. Hyg. 2003, 69, 314–317. [Google Scholar] [PubMed]
- Dyer, J.R.; Einsiedel, L.; Ferguson, P.E.; Lee, A.S.; Unsworth, N.B.; Graves, S.R.; Gordon, D.L. A new focus of Rickettsia honei spotted fever in South Australia. Med. J. Aust. 2005, 182, 231–234. [Google Scholar] [PubMed]
- Parola, P.; Davoust, B.; Raoult, D. Tick-and flea-borne rickettsial emerging zoonoses. Vet. Res. 2005, 36, 469–492. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.C.; Walker, A.R. Ticks of Australia. The species that infest domestic animals and humans. Zootaxa 2014, 3816, 1–144. [Google Scholar] [CrossRef] [PubMed]
- Vilcins, I.-M.E.; Fournier, P.-E.; Old, J.M.; Deane, E. Evidence for the presence of Francisella and spotted fever group Rickettsia DNA in the tick Amblyomma fimbriatum (acari: Ixodidae), Northern Territory, Australia. J. Med. Entomol. 2009, 46, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, M.L.; Gardner, M.G.; Fitch, A.J.; Ansari, T.H.; Smyth, A.K. A direct benefit of native saltbush revegetation for an endemic lizard (Tiliqua rugosa) in southern Australia. Aust. J. Zool. 2012, 60, 192–198. [Google Scholar] [CrossRef]
- Regnery, R.L.; Spruill, C.L.; Plikaytis, B.D. Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. J. Bacteriol. 1991, 173, 1576–1589. [Google Scholar] [PubMed]
- Rogall, T.; Wolters, J.; Flohr, T.; Bottger, E.C. Towards a phylogeny and definition of species at the molecular level within the genus Mycobacterium. Int. J. Syst. Evol. Microbiol. 1990, 40, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whiley, H.; Custance, G.; Graves, S.; Stenos, J.; Taylor, M.; Ross, K.; Gardner, M.G. Rickettsia Detected in the Reptile Tick Bothriocroton hydrosauri from the Lizard Tiliqua rugosa in South Australia. Pathogens 2016, 5, 41. https://doi.org/10.3390/pathogens5020041
Whiley H, Custance G, Graves S, Stenos J, Taylor M, Ross K, Gardner MG. Rickettsia Detected in the Reptile Tick Bothriocroton hydrosauri from the Lizard Tiliqua rugosa in South Australia. Pathogens. 2016; 5(2):41. https://doi.org/10.3390/pathogens5020041
Chicago/Turabian StyleWhiley, Harriet, Georgie Custance, Stephen Graves, John Stenos, Michael Taylor, Kirstin Ross, and Michael G. Gardner. 2016. "Rickettsia Detected in the Reptile Tick Bothriocroton hydrosauri from the Lizard Tiliqua rugosa in South Australia" Pathogens 5, no. 2: 41. https://doi.org/10.3390/pathogens5020041
APA StyleWhiley, H., Custance, G., Graves, S., Stenos, J., Taylor, M., Ross, K., & Gardner, M. G. (2016). Rickettsia Detected in the Reptile Tick Bothriocroton hydrosauri from the Lizard Tiliqua rugosa in South Australia. Pathogens, 5(2), 41. https://doi.org/10.3390/pathogens5020041





