Development of a Vaccine against Escherichia coli Urinary Tract Infections
Abstract
:1. Introduction
2. Existing UTI Vaccines
| Clinical Setting | Outpatient (88%) | Inpatient (6.5%) | Emergency Room (5%) |
|---|---|---|---|
| Number of Visits | 970,000 | 72,000 | 55,000 |
| Cost per Visit | $241 | $64,824 | $2843 |
| Total Cost | $233 million | $4634 million | $156 million |
| Total Cost due to Recurrent UTI = $5.06 billion | |||
3. Progress towards Development of a Vaccine to Prevent UTI
3.1. What Secreted and Outer Membrane Proteins Can Be Predicted from the Genomic Sequence of E. coli CFT073?
3.2. What Proteins Are Synthesized by E. coli CFT073 in Urine?
3.3. What Genes Are Expressed by E. coli CFT073 during Experimental Infection?
3.4. Are the Same Genes Expressed by E. coli Infecting Women with UTI?
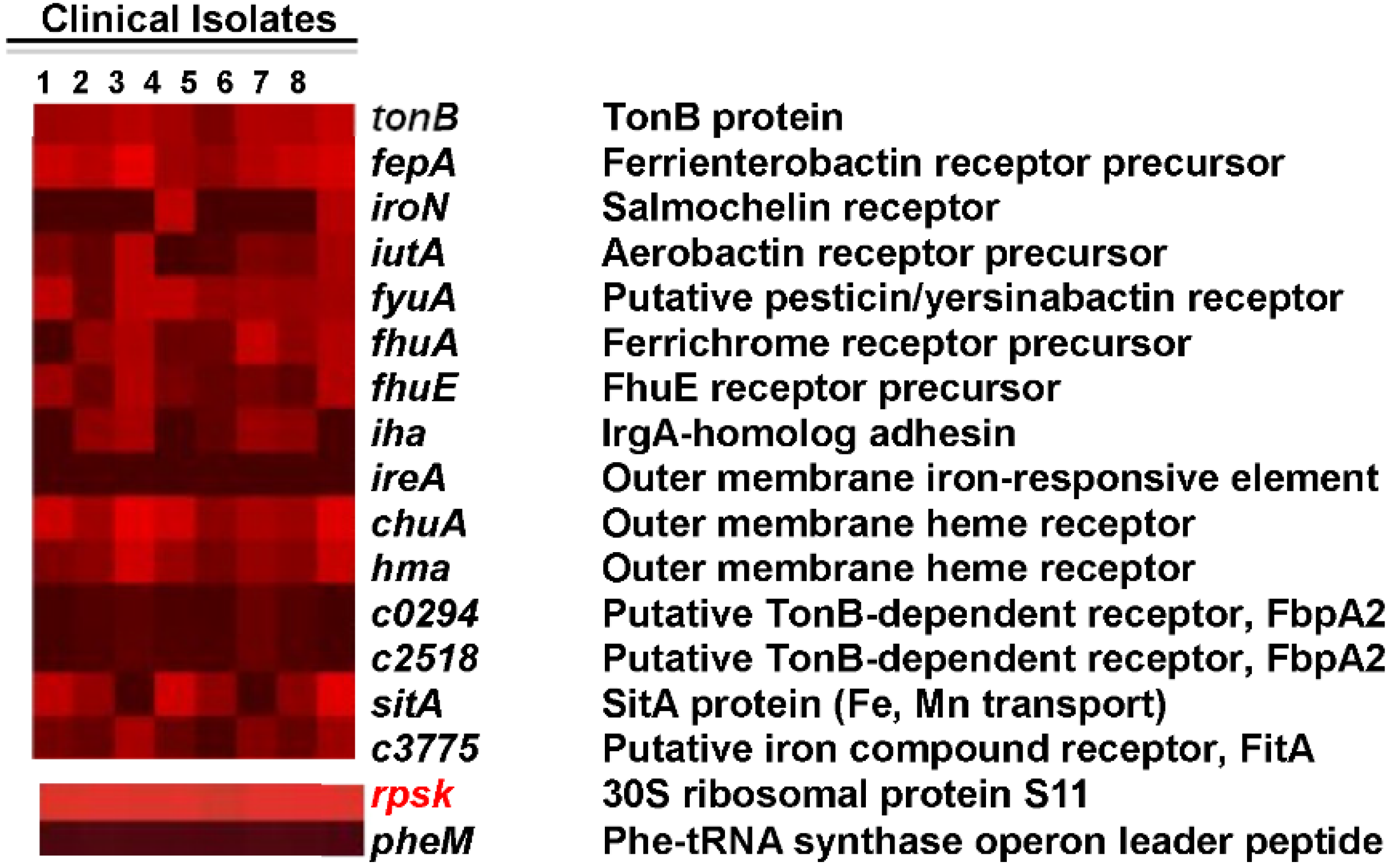
3.5. What E. coli CFT073 Proteins Are Recognized by an Antibody Response during Experimental UTI?
3.6. What Surface-Exposed Motifs of E. coli CFT073 Proteins Are Susceptible to Limited Proteolysis?
3.7. What Genes Are Common among UPEC Strains?
4. Rational Selection of Vaccine Candidates
Testing of Top-Scoring Antigens, all Representing Outer Membrane Protein Components of Iron Acquisition Systems, as an Intranasal Vaccine
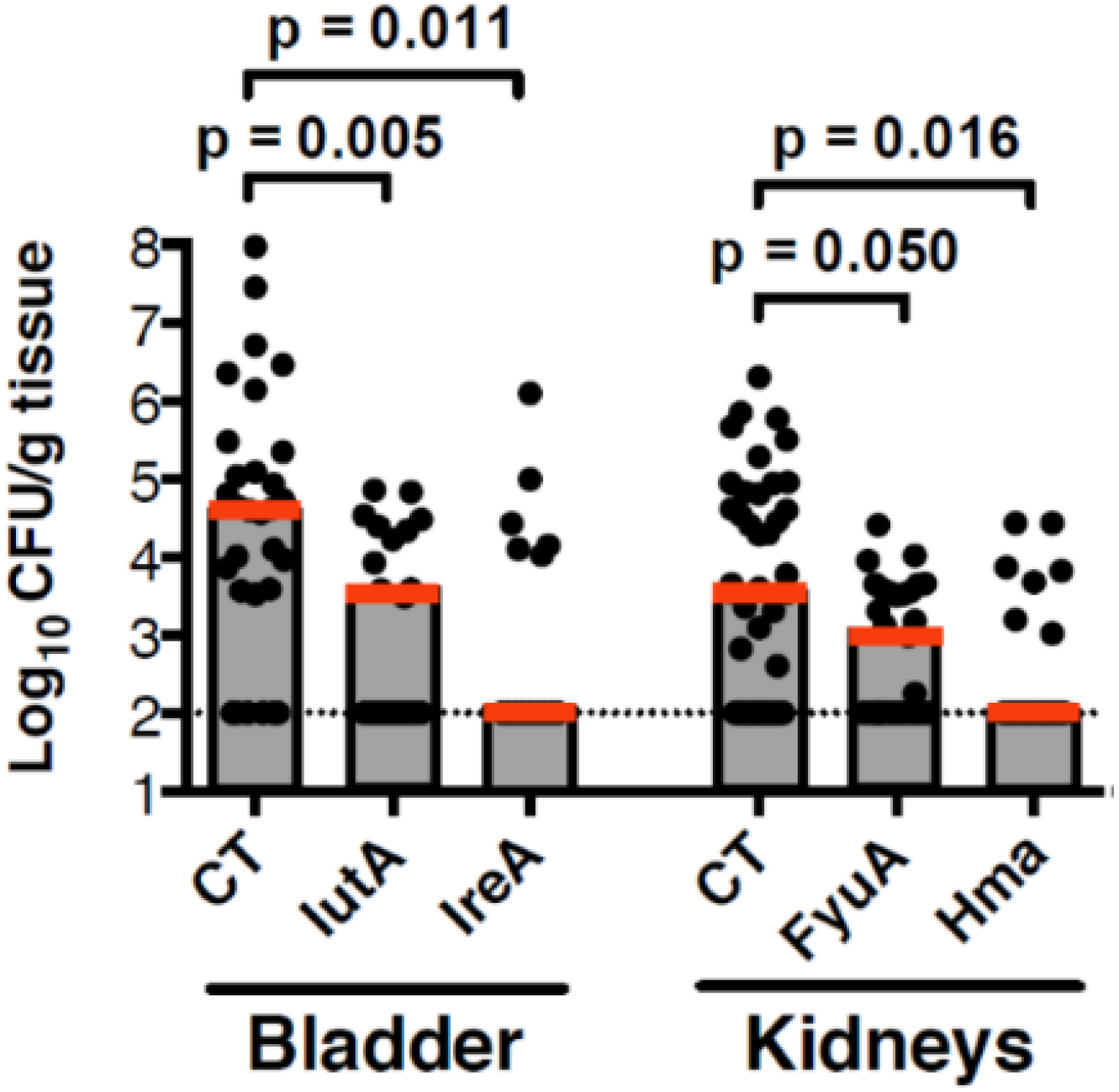
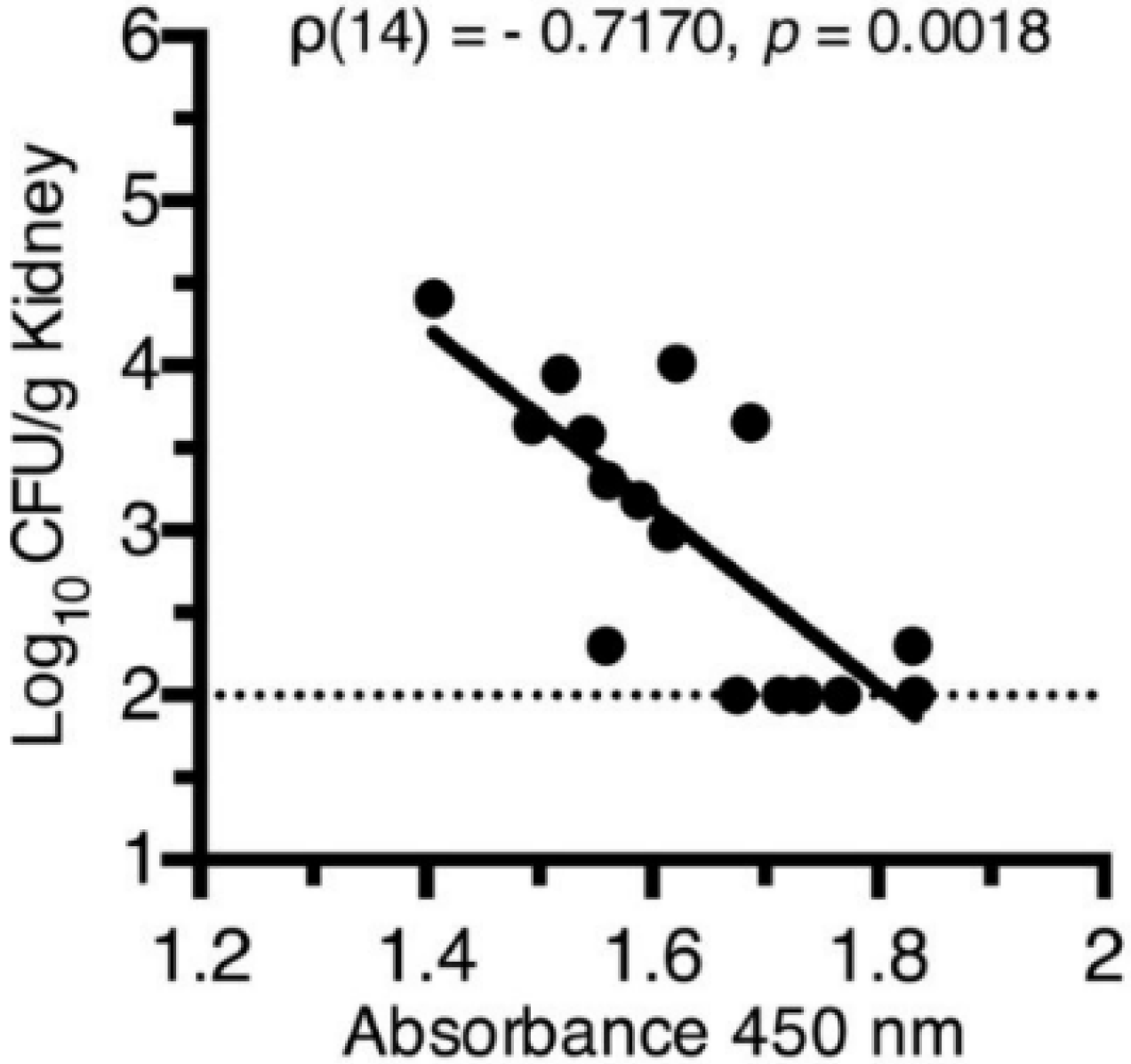
5. Summary
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schappert, S.M.; Rechtsteiner, E.A. Ambulatory medical care utilization estimates for 2006. Available online: http://198.246.124.22/nchs/data/nhsr/nhsr008.pdf (accessed on 30 December 2015).
- Foxman, B. Recurring urinary tract infection: Incidence and risk factors. Am. J. Public Health 1990, 80, 331–333. [Google Scholar] [PubMed]
- O’Hanley, P. Prospects for urinary tract infection vaccines. In Urinary Tract Infections: Molecular Pathogenesis and Clinical Management; Mobley, H.L.T., Warren, J.W., Eds.; ASM Press: Washington, DC, USA, 1996; pp. 405–425. [Google Scholar]
- Litwin, M.S.; Saigal, C.S. Introduction; GPO: Washington, DC, USA, 2007.
- Foxman, B. The epidemiology of urinary tract infection. Nat. Rev. Urol. 2010, 7, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Litwin, M.S.; Saigal, C.S.; Yano, E.M.; Avila, C.; Geschwind, S.A.; Hanley, J.M.; Joyce, G.F.; Madison, R.; Pace, J.; Polich, S.M.; et al. Urologic diseases in America Project: Analytical methods and principal findings. J. Urol. 2005, 173, 933–937. [Google Scholar] [CrossRef] [PubMed]
- Kunin, C.M. Detection, Prevention and Management of Urinary Tract Infections, 4th ed.; Lea & Febiger: Philadelphia, PA, USA, 1987. [Google Scholar]
- DeFrances, C.J.; Lucas, C.A.; Buie, V.C.; Golosinskiy, A. 2006 National Hospital Discharge Survey. Available online: http://198.246.124.22/nchs/data/nhsr/nhsr005.pdf (accessed on 30 December 2015).
- Gupta, K.; Hooton, T.M.; Stamm, W.E. Increasing antimicrobial resistance and the management of uncomplicated community-acquired urinary tract infections. Ann. Intern. Med. 2001, 135, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Moura, A.; Nicolau, A.; Hooton, T.; Azeredo, J. Antibiotherapy and pathogenesis of uncomplicated UTI: Difficult relationships. J. Appl. Microbiol. 2009, 106, 1779–1791. [Google Scholar] [CrossRef] [PubMed]
- Zhanel, G.G.; Hisanaga, T.L.; Laing, N.M.; DeCorby, M.R.; Nichol, K.A.; Palatnik, L.P.; Johnson, J.; Noreddin, A.; Harding, G.K.; Nicolle, L.E.; et al. Antibiotic resistance in outpatient urinary isolates: Final results from the North American Urinary Tract Infection Collaborative Alliance (NAUTICA). Int. J. Antimicrob. Agents 2005, 26, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Karlowsky, J.A.; Hoban, D.J.; Decorby, M.R.; Laing, N.M.; Zhanel, G.G. Fluoroquinolone-resistant urinary isolates of Escherichia coli from outpatients are frequently multidrug resistant: Results from the North American Urinary Tract Infection Collaborative Alliance-Quinolone Resistance study. Antimicrob. Agents Chemother. 2006, 50, 2251–2254. [Google Scholar] [CrossRef] [PubMed]
- Billips, B.K.; Schaeffer, A.J.; Klumpp, D.J. Molecular basis of uropathogenic Escherichia coli evasion of the innate immune response in the bladder. Infect. Immun. 2008, 76, 3891–3900. [Google Scholar] [CrossRef] [PubMed]
- Sivick, K.E.; Schaller, M.A.; Smith, S.N.; Mobley, H.L. The innate immune response to uropathogenic Escherichia coli involves IL-17A in a murine model of urinary tract infection. J. Immunol. 2010, 184, 2065–2075. [Google Scholar] [CrossRef] [PubMed]
- Alteri, C.J.; Hagan, E.C.; Sivick, K.E.; Smith, S.N.; Mobley, H.L. Mucosal immunization with iron receptor antigens protects against urinary tract infection. PLoS Pathog 2009, 5, e1000586. [Google Scholar] [CrossRef] [PubMed]
- Kruze, D.; Biro, K.; Holzbecher, K.; Andrial, M.; Bossart, W. Protection by a polyvalent vaccine against challenge infection and pyelonephritis. Urol. Res. 1992, 20, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Kruze, D.; Holzbecher, K.; Andrial, M.; Bossart, W. Urinary antibody response after immunisation with a vaccine against urinary tract infection. Urol. Res. 1989, 17, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Thumbikat, P.; Waltenbaugh, C.; Schaeffer, A.J.; Klumpp, D.J. Antigen-specific responses accelerate bacterial clearance in the bladder. J. Immunol. 2006, 176, 3080–3086. [Google Scholar] [CrossRef] [PubMed]
- Uehling, D.T.; James, L.J.; Hopkins, W.J.; Balish, E. Immunization against urinary tract infection with a multi-valent vaginal vaccine. J. Urol. 1991, 146, 223–226. [Google Scholar] [PubMed]
- Langermann, S.; Mollby, R.; Burlein, J.E.; Palaszynski, S.R.; Auguste, C.G.; DeFusco, A.; Strouse, R.; Schenerman, M.A.; Hultgren, S.J.; Pinkner, J.S.; et al. Vaccination with FimH adhesin protects cynomolgus monkeys from colonization and infection by uropathogenic Escherichia coli. J. Infect. Dis. 2000, 181, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Brumbaugh, A.R.; Mobley, H.L. Preventing urinary tract infection: progress toward an effective Escherichia coli vaccine. Expert Rev. Vaccines 2012, 11, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Bauer, H.W.; Rahlfs, V.W.; Lauener, P.A.; Blessmann, G.S. Prevention of recurrent urinary tract infections with immuno-active E. coli fractions: A meta-analysis of five placebo-controlled double-blind studies. Int. J. Antimicrob. Agents 2002, 19, 451–456. [Google Scholar] [CrossRef]
- Alteri, C.J.; Mobley, H.L. Quantitative profile of the uropathogenic Escherichia coli outer membrane proteome during growth in human urine. Infect. Immun. 2007, 75, 2679–2688. [Google Scholar] [CrossRef] [PubMed]
- Hagan, E.C.; Mobley, H.L. Haem acquisition is facilitated by a novel receptor Hma and required by uropathogenic Escherichia coli for kidney infection. Mol. Microbiol. 2009, 71, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Snyder, J.A.; Haugen, B.J.; Buckles, E.L.; Lockatell, C.V.; Johnson, D.E.; Donnenberg, M.S.; Welch, R.A.; Mobley, H.L. Transcriptome of uropathogenic Escherichia coli during urinary tract infection. Infect. Immun. 2004, 72, 6373–6381. [Google Scholar] [CrossRef] [PubMed]
- Hagan, E.C.; Lloyd, A.L.; Rasko, D.A.; Faerber, G.J.; Mobley, H.L. Escherichia coli global gene expression in urine from women with urinary tract infection. PLoS Pathog 2010, 6, e1001187. [Google Scholar] [CrossRef] [PubMed]
- Subashchandrabose, S.; Hazen, T.H.; Brumbaugh, A.R.; Himpsl, S.D.; Smith, S.N.; Ernst, R.D.; Rasko, D.A.; Mobley, H.L. Host-specific induction of Escherichia coli fitness genes during human urinary tract infection. Proc. Natl. Acad. Sci. USA 2014, 111, 18327–18332. [Google Scholar] [CrossRef] [PubMed]
- Spurbeck, R.R.; Dinh, P.C., Jr.; Walk, S.T.; Stapleton, A.E.; Hooton, T.M.; Nolan, L.K.; Kim, K.S.; Johnson, J.R.; Mobley, H.L. Escherichia coli isolates that carry vat, fyuA, chuA, and yfcV efficiently colonize the urinary tract. Infect. Immun. 2012, 80, 4115–4122. [Google Scholar] [CrossRef] [PubMed]
- Hagan, E.C.; Mobley, H.L. Uropathogenic Escherichia coli outer membrane antigens expressed during urinary tract infection. Infect. Immun. 2007, 75, 3941–3949. [Google Scholar] [CrossRef] [PubMed]
- Walters, M.S.; Mobley, H.L. Identification of uropathogenic Escherichia coli surface proteins by shotgun proteomics. J. Microbiol. Methods 2009, 78, 131–135. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lloyd, A.L.; Rasko, D.A.; Mobley, H.L. Defining genomic islands and uropathogen-specific genes in uropathogenic Escherichia coli. J. Bacteriol. 2007, 189, 3532–3546. [Google Scholar] [CrossRef] [PubMed]
- Welch, R.A.; Burland, V.; Plunkett, G., 3rd; Redford, P.; Roesch, P.; Rasko, D.; Buckles, E.L.; Liou, S.R.; Boutin, A.; Hackett, J.; et al. Extensive mosaic structure revealed by the complete genome sequence of uropathogenic Escherichia coli. Proc. Natl. Acad. Sci. USA 2002, 99, 17020–17024. [Google Scholar] [CrossRef] [PubMed]
- Brumbaugh, A.R.; Smith, S.N.; Mobley, H.L. Immunization with the yersiniabactin receptor, FyuA, protects against pyelonephritis in a murine model of urinary tract infection. Infect. Immun. 2013, 81, 3309–3316. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mobley, H.L.T.; Alteri, C.J. Development of a Vaccine against Escherichia coli Urinary Tract Infections. Pathogens 2016, 5, 1. https://doi.org/10.3390/pathogens5010001
Mobley HLT, Alteri CJ. Development of a Vaccine against Escherichia coli Urinary Tract Infections. Pathogens. 2016; 5(1):1. https://doi.org/10.3390/pathogens5010001
Chicago/Turabian StyleMobley, Harry L. T., and Christopher J. Alteri. 2016. "Development of a Vaccine against Escherichia coli Urinary Tract Infections" Pathogens 5, no. 1: 1. https://doi.org/10.3390/pathogens5010001
APA StyleMobley, H. L. T., & Alteri, C. J. (2016). Development of a Vaccine against Escherichia coli Urinary Tract Infections. Pathogens, 5(1), 1. https://doi.org/10.3390/pathogens5010001





