Neutrophils Do Not Express IL-17A in the Context of Acute Oropharyngeal Candidiasis
Abstract
:1. Introduction
2. Results
2.1. Neutrophils Do Not Produce IL-17 in Response to Oral C. albicans Infection
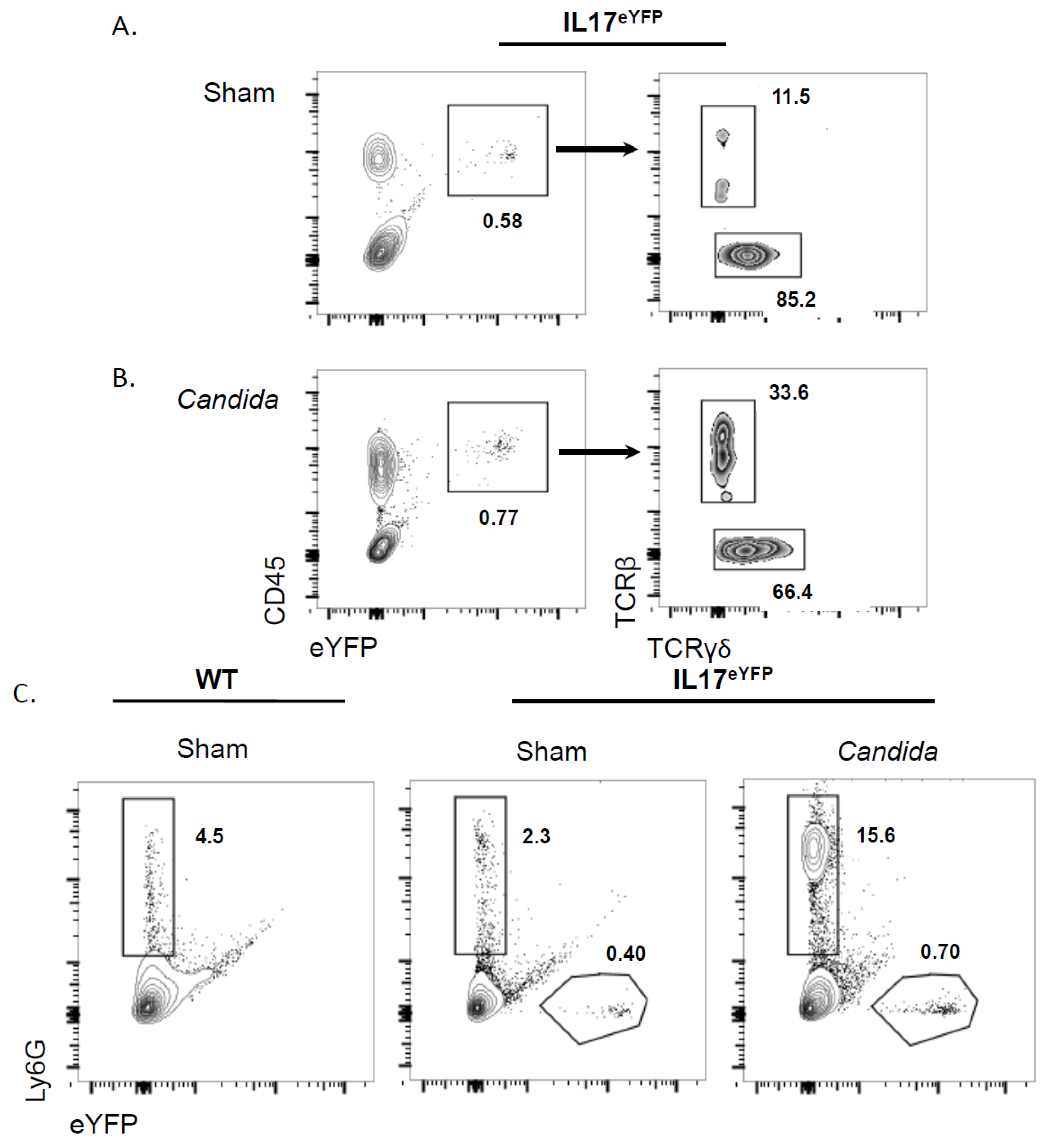
2.2. Neutrophil Depletion Is Not Required for IL-17 Production or Signaling in OPC
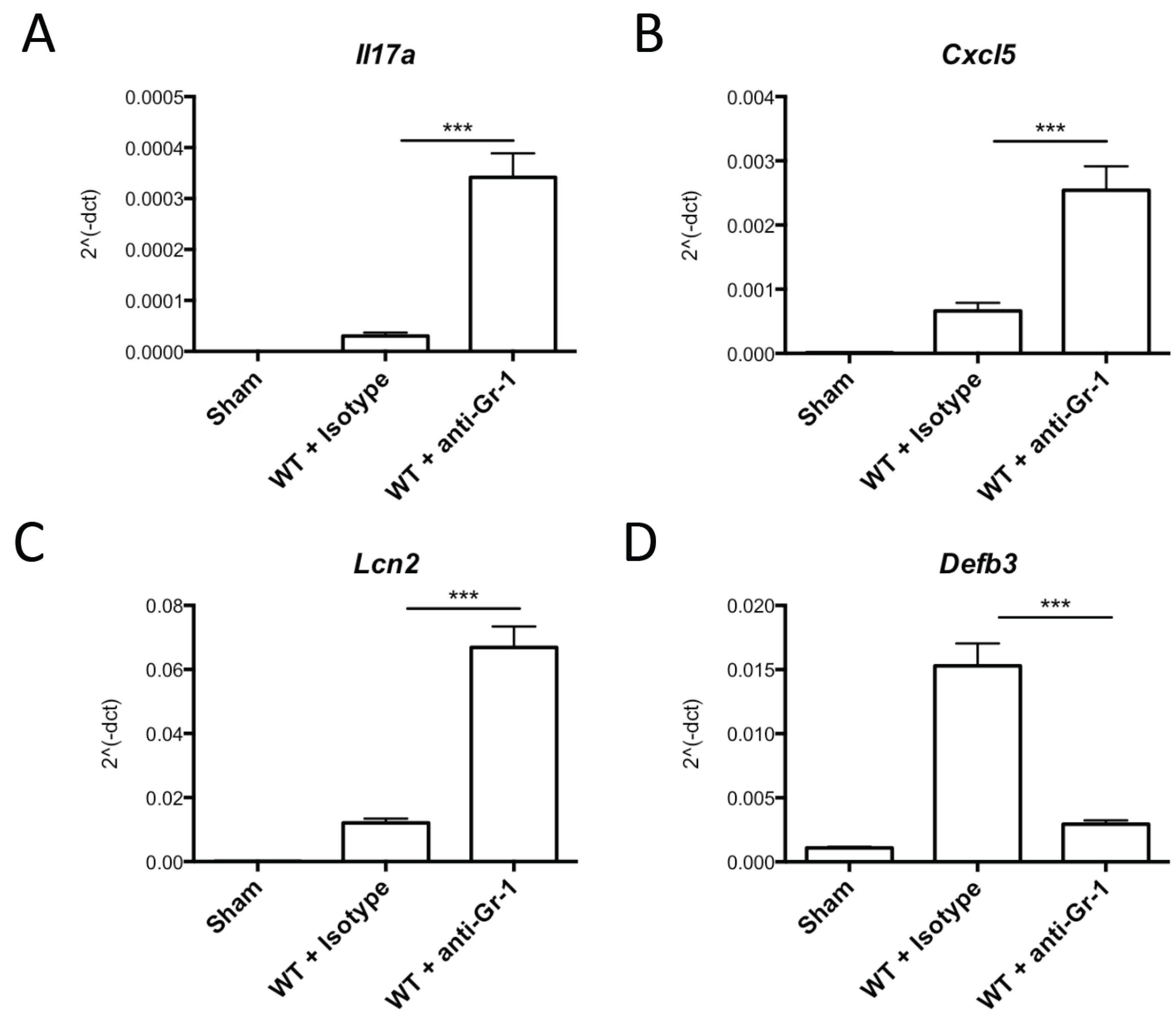
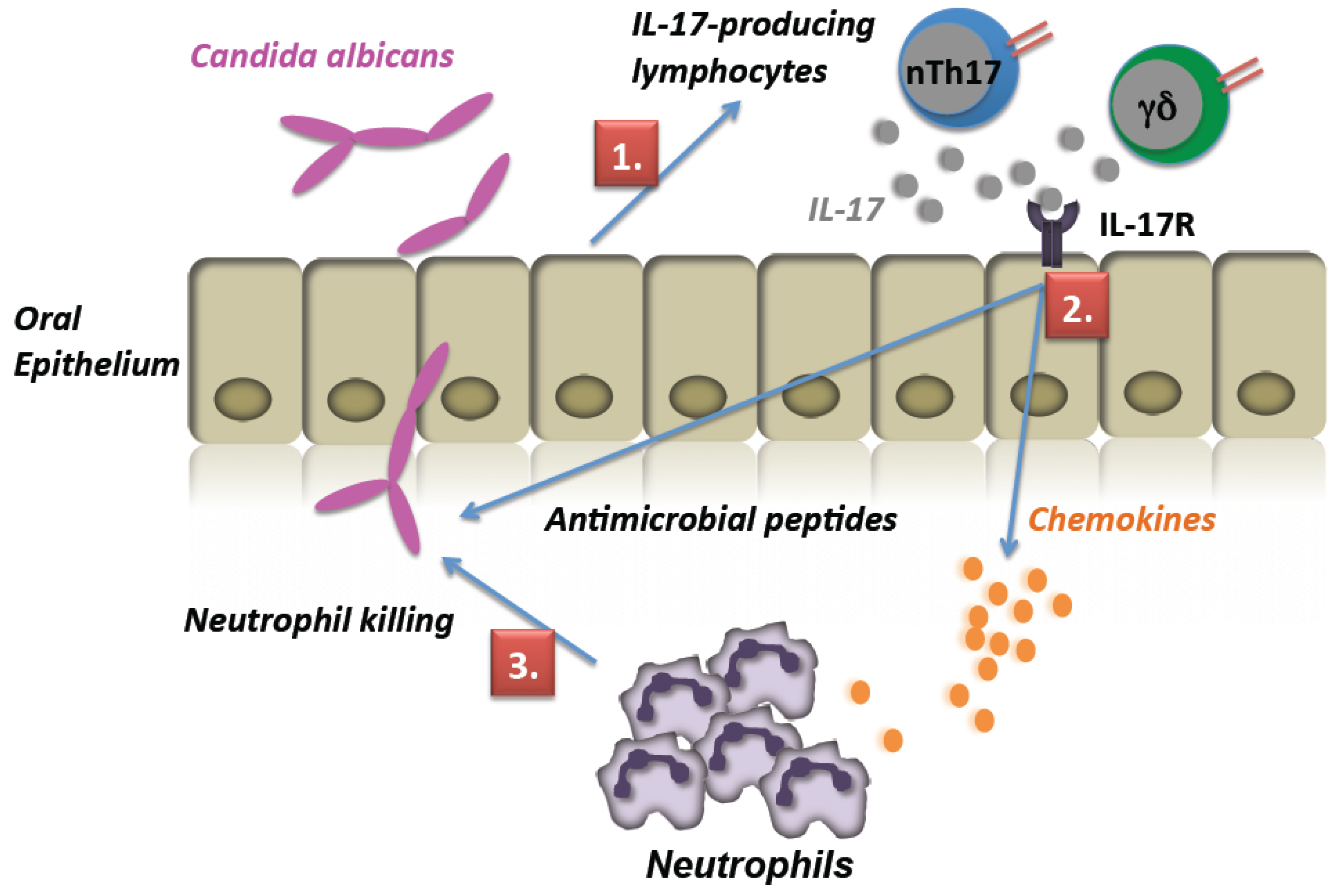
3. Discussion
4. Experimental Methods
4.1. Mice and Oral Candidiasis Model
4.2. Antibody-Mediated Neutrophil Depletion
4.3. Flow Cytometry
4.4. RNA and qPCR
4.5. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| OPC | oropharyngeal candidiasis |
| TCR | T cell receptor |
| CMC | chronic mucocutaneous candidiasis |
| YFP | yellow fluorescent protein |
| BD3 | β-defensin 3 |
| Ab | antibody |
| ICS | intracellular staining |
References and Notes
- Glocker, E.; Grimbacher, B. Chronic mucocutaneous candidiasis and congenital susceptibility to Candida. Curr. Opin. Allergy Clin. Immunol. 2010, 10, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Milner, J.D.; Brenchley, J.M.; Laurence, A.; Freeman, A.F.; Hill, B.J.; Elias, K.M.; Kanno, Y.; Spalding, C.; Elloumi, H.Z.; Paulson, M.L.; et al. Impaired T(h)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 2008, 452, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Huppler, A.R.; Bishu, S.; Gaffen, S.L. Mucocutaneous candidiasis: The IL-17 pathway and implications for targeted immunotherapy. Arthritis Res. Ther. 2012, 14, 217. [Google Scholar] [CrossRef] [PubMed]
- Conti, H.; Shen, F.; Nayyar, N.; Stocum, E.; Sun, J.N.; Lindemann, M.; Ho, A.; Hai, J.; Yu, J.; Jung, J.; et al. Th17 cells and IL-17 receptor signaling are essential for mucosal host defense against oral candidiasis. J. Exp. Med. 2009, 206, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Huppler, A.R.; Conti, H.R.; Hernandez-Santos, N.; Biswas, P.S.; Darville, T.; Gaffen, S.L. Role of neutrophils in IL-17-dependent immunity to mucosal candidiasis. J. Immunol. 2014, 192, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Fidel, P.L., Jr. Candida-host interactions in HIV disease: Implications for oropharyngeal candidiasis. Adv. Dent. Res. 2011, 23, 45–49. [Google Scholar] [CrossRef] [PubMed]
- de Repentigny, L.; Lewandowski, D.; Joliceur, P. Immunopathogenesis of oropharyngeal candidiasis in human immunodeficiency virus infection. Clin. Microbiol. Rev. 2004, 17, 729–759. [Google Scholar] [CrossRef] [PubMed]
- Goupil, M.; Cousineau-Cote, V.; Aumont, F.; Senechal, S.; Gaboury, L.; Hanna, Z.; Jolicoeur, P.; de Repentigny, L. Defective IL-17- and IL-22-dependent mucosal host response to Candida albicans determines susceptibility to oral candidiasis in mice expressing the HIV-1 transgene. BMC Immunol. 2014, 15, 49. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Rodriguez, E.V.; Rivino, L.; Geginat, J.; Jarrossay, D.; Gattorno, M.; Lanzavecchia, A.; Sallusto, F.; Napolitani, G. Surface phenotype and antigenic specificity of human interleukin 17-producing T helper memory cells. Nature Immunol. 2007, 8, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Bär, E.; Gladiator, A.; Bastidas, S.; Roschitzki, B.; Acha-Orbea, H.; Oxenius, A.; LeibundGut-Landmann, S. A novel Th cell epitope of Candida albicans mediates protection from fungal infection. J. Immunol 2012, 188, 5636–5643. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Santos, N.; Huppler, A.R.; Peterson, A.C.; Khader, S.A.; McKenna, K.C.; Gaffen, S.L. Th17 cells confer long term adaptive immunity to oral mucosal Candida albicans infections. Mucosal Immunol. 2013, 6, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Bishu, S.; Hernandez-Santos, N.; Simpson-Abelson, M.; Huppler, A.R.; Conti, H.R.; Ghilardi, N.; Mamo, A.; Gaffen, S.L. CARD9 is required for adaptive but not innate immunity to oral mucosal Candida albicans infections. Infect. Immun. 2014, 82, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Ho, A.; Shen, F.; Conti, H.; Patel, N.; Childs, E.; Peterson, A.; Hernandez-Santos, N.; Kolls, J.; Kane, L.; Ouyang, W.; et al. IL-17RC is required for immune signaling via an extended SEFIR domain in the cytoplasmic tail. J. Immunol. 2010, 185, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Puel, A.; Cypowji, S.; Bustamante, J.; Wright, J.; Liu, L.; Lim, H.; Migaud, M.; Israel, L.; Chrabieh, M.; Audry, M.; et al. Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science 2011, 332, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.C.; Whibley, N.; Mamo, A.J.; Siebenlist, U.; Chan, Y.R.; Gaffen, S.L. Interleukin-17-induced protein Lipocalin 2 is dispensable for immunity to oral candidiasis. Infect. Immun. 2014, 82, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Boisson, B.; Wang, C.; Pedergnana, V.; Wu, L.; Cypowyj, S.; Rybojad, M.; Belkadi, A.; Picard, C.; Abel, L.; Fieschi, C.; et al. A biallelic Act1 mutation selectively abolishes interleukin-17 responses in humans with chronic mucocutaneous candidiasis. Immunity 2013, 39, 676–686. [Google Scholar] [CrossRef] [PubMed]
- Iliev, I.D.; Funari, V.A.; Taylor, K.D.; Nguyen, Q.; Reyes, C.N.; Strom, S.P.; Brown, J.; Becker, C.A.; Fleshner, P.R.; Dubinsky, M.; et al. Interactions between commensal fungi and the C-type lectin receptor dectin-1 influence colitis. Science 2012, 336, 1314–1317. [Google Scholar] [CrossRef] [PubMed]
- Cua, D.J.; Tato, C.M. Innate IL-17-producing cells: The sentinels of the immune system. Nat. Rev. Immunol. 2010, 10, 479–489. [Google Scholar] [CrossRef]
- Conti, H.; Peterson, A.; Huppler, A.; Brane, L.; Hernández-Santos, N.; Whibley, N.; Garg, A.; Simpson-Abelson, M.; Gibson, G.; Mamo, A.; et al. Oral-resident ‘natural’ Th17 cells and γδ-T cells control opportunistic Candida albicans infections. J. Exp. Med. 2014, 211, 2075–2084. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Duarte, J.H.; Veldhoen, M.; Hornsby, E.; Li, Y.; Cua, D.J.; Ahlfors, H.; Wilhelm, C.; Tolaini, M.; Menzel, U.; et al. Fate mapping of IL-17-producing T cells in inflammatory responses. Nature Immunol. 2011, 12, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Kagami, S.; Rizzo, H.L.; Kurtz, S.E.; Miller, L.S.; Blauvelt, A. IL-23 and IL-17A, but not IL-12 and IL-22, are required for optimal skin host defense against Candida albicans. J. Immunol. 2010, 185, 5453–5462. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, R.S.; Vareechon, C.; Prajna, N.V.; Dharmalingam, K.; Pearlman, E.; Lalitha, P. Interleukin 17 expression in peripheral blood neutrophils from fungal keratitis patients and healthy cohorts in southern India. J. Infect. Dis. 2015, 211, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Werner, J.L.; Gessner, M.A.; Lilly, L.M.; Nelson, M.P.; Metz, A.E.; Horn, D.; Dunaway, C.W.; Deshane, J.; Chaplin, D.D.; Weaver, C.T.; et al. Neutrophils produce interleukin 17A (IL-17A) in a dectin-1- and IL-23-dependent manner during invasive fungal infection. Infect. Immun. 2011, 79, 3966–3977. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.R.; Leal, S.M., Jr.; Sun, Y.; Pearlman, E. Aspergillus and fusarium corneal infections are regulated by Th17 cells and IL-17-producing neutrophils. J. Immunol. 2014, 192, 3319–3327. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.R.; Roy, S.; Leal, S.M., Jr.; Sun, Y.; Howell, S.J.; Cobb, B.A.; Li, X.; Pearlman, E. Activation of neutrophils by autocrine IL-17A-IL-17RC interactions during fungal infection is regulated by IL-6, IL-23, RORγt and dectin-2. Nature Immunol. 2014, 15, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Gladiator, A.; Wangler, N.; Trautwein-Weidner, K.; Leibundgut-Landmann, S. Cutting edge: IL-17-secreting innate lymphoid cells are essential for host defense against fungal infection. J. Immunol. 2013, 190, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Solis, N.V.; Filler, S.G. Mouse model of oropharyngeal candidiasis. Nat. Protoc. 2012, 7, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Mehrad, B.; Strieter, R.M.; Moore, T.A.; Tsai, W.C.; Lira, S.A.; Standiford, T.J. CXC chemokine receptor-2 ligands are necessary components of neutrophil-mediated host defense in invasive pulmonary aspergillosis. J. Immunol. 1999, 163, 6086–6094. [Google Scholar] [PubMed]
- Moutsopoulos, N.M.; Konkel, J.; Sarmadi, M.; Eskan, M.A.; Wild, T.; Dutzan, N.; Abusleme, L.; Zenobia, C.; Hosur, K.B.; Abe, T.; et al. Defective neutrophil recruitment in leukocyte adhesion deficiency type I disease causes local IL-17-driven inflammatory bone loss. Sci. Transl. Med. 2014, 6, 229ra240. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Santos, N.; Gaffen, S.L. Th17 cells in immunity to Candida albicans. Cell. Host Microbe 2012, 11, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.Y.; Chen, Y.; Thai, P.; Wachi, S.; Huang, F.; Kim, C.; Harper, R.W.; Wu, R. IL-17 markedly up-regulates beta-defensin-2 expression in human airway epithelium via JAK and NF-κB signaling pathways. J. Immunol. 2004, 173, 3482–3491. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.Y.; Kim, C.; Huang, F.; Wu, R. Requirements for two proximal NF-κB binding sites and IκBζ in IL-17A-induced human beta-defensin 2 expression by conducting airway epithelium. J. Biol. Chem. 2008, 283, 15309–15318. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.C.; Tan, X.Y.; Luxenberg, D.P.; Karim, R.; Dunussi-Joannopoulos, K.; Collins, M.; Fouser, L.A. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J. Exp. Med. 2006, 203, 2271–2279. [Google Scholar] [CrossRef] [PubMed]
- Naglik, J.; Moyes, D. Epithelial cell innate response to Candida albicans. Adv. Dent. Res. 2011, 23, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Conti, H.R.; Gaffen, S.L. Host responses to Candida albicans: Th17 cells and mucosal candidiasis. Microbes Infect. 2010, 12, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, C.; Micheletti, A.; Cassatella, M.A. Neutrophil-derived cytokines: Facts beyond expression. Front. Immunol. 2014, 5, 508. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, Y.; Ishigame, H.; Saijo, S.; Nakae, S. Functional specialization of interleukin-17 family members. Immunity 2011, 34, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Ruddy, M.; Wong, G.; Sfintescu, C.; Baker, P.; Smith, J.; Evans, R.; Gaffen, S. An essential role for IL-17 in preventing pathogen-initiated bone destruction: Recruitment of neutrophils to inflamed bone requires IL-17 receptor-dependent signals. Blood 2007, 109, 3794–3802. [Google Scholar] [CrossRef] [PubMed]
- Trautwein-Weidner, K.; Gladiator, A.; Nur, S.; Diethelm, P.; LeibundGut-Landmann, S. IL-17-mediated antifungal defense in the oral mucosa is independent of neutrophils. Mucosal Immunol. 2015, 8, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Eskan, M.A.; Jotwani, R.; Abe, T.; Chmelar, J.; Lim, J.H.; Liang, S.; Ciero, P.A.; Krauss, J.L.; Li, F.; Rauner, M.; et al. The leukocyte integrin antagonist Del-1 inhibits IL-17-mediated inflammatory bone loss. Nature Immunol. 2012, 13, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Stark, M.A.; Huo, Y.; Burcin, T.L.; Morris, M.A.; Olson, T.S.; Ley, K. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 2005, 22, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Le Jan, S.; Plee, J.; Vallerand, D.; Dupont, A.; Delanez, E.; Durlach, A.; Jackson, P.L.; Blalock, J.E.; Bernard, P.; Antonicelli, F. Innate immune cell-produced IL-17 sustains inflammation in bullous pemphigoid. J. Invest. Derm. 2014, 134, 2908–2917. [Google Scholar] [CrossRef] [PubMed]
- Milanova, V.; Ivanovska, N.; Dimitrova, P. TLR2 elicits IL-17-mediated RANKL expression, IL-17, and OPG production in neutrophils from arthritic mice. Mediators Inflamm. 2014, 2014, 643406. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Huang, L.; Vergis, A.L.; Ye, H.; Bajwa, A.; Narayan, V.; Strieter, R.M.; Rosin, D.L.; Okusa, M.D. IL-17 produced by neutrophils regulates IFN-γ-mediated neutrophil migration in mouse kidney ischemia-reperfusion injury. J. Clin. Invest. 2010, 120, 331–342. [Google Scholar] [CrossRef] [PubMed]
- van Baarsen, L.G.; Lebre, M.C.; van der Coelen, D.; Aarrass, S.; Tang, M.W.; Ramwadhdoebe, T.H.; Gerlag, D.M.; Tak, P.P. Heterogeneous expression pattern of interleukin 17A (IL-17A), IL-17F and their receptors in synovium of rheumatoid arthritis, psoriatic arthritis and osteoarthritis: Possible explanation for nonresponse to anti-IL-17 therapy? Arthritis Res. Ther. 2014, 16, 426. [Google Scholar] [CrossRef] [PubMed]
- Noordenbos, T.; Yeremenko, N.; Gofita, I.; van de Sande, M.; Tak, P.P.; Canete, J.D.; Baeten, D. Interleukin-17-positive mast cells contribute to synovial inflammation in spondylarthritis. Arth. Rheum. 2012, 64, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Hueber, A.J.; Asquith, D.L.; Miller, A.M.; Reilly, J.; Kerr, S.; Leipe, J.; Melendez, A.J.; McInnes, I.B. Mast cells express IL-17A in rheumatoid arthritis synovium. J. Immunol. 2010, 184, 3336–3340. [Google Scholar] [CrossRef] [PubMed]
- Keijsers, R.R.; Joosten, I.; van Erp, P.E.; Koenen, H.J.; van de Kerkhof, P.C. Cellular sources of IL-17 in psoriasis: A paradigm shift? Exp. Derm. 2014, 23, 799–803. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.M.; Rubin, C.J.; Khandpur, R.; Wang, J.Y.; Riblett, M.; Yalavarthi, S.; Villanueva, E.C.; Shah, P.; Kaplan, M.J.; Bruce, A.T. Mast cells and neutrophils release IL-17 through extracellular trap formation in psoriasis. J. Immunol. 2011, 187, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Noordenbos, T.; Paramarta, J.; Blijdorp, I.; Baeten, D. Human mast cells engulf and store exogenous IL-17A. Clin. Exp. Rheumatol. 2014, 32, 815. [Google Scholar] [CrossRef]
- Baeten, D.; (Academic Medical Center/University of Amsterdam, Amsterdam, The Netherlands). Personal communication, 2015.
- Ishigame, H.; Kakuta, S.; Nagai, T.; Kadoki, M.; Nambu, A.; Komiyama, Y.; Fujikado, N.; Tanahashi, Y.; Akitsu, A.; Kotaki, H.; et al. Differential roles of interleukin-17A and -17F in host defense against mucoepithelial bacterial infection and allergic responses. Immunity 2009, 30, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Kamai, Y.; Kubota, M.; Kamai, Y.; Hosokawa, T.; Fukuoka, T.; Filler, S. New model of oropharyngeal candidiasis in mice. Anti-microb. Agents Chemo. 2001, 45, 3195–3197. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huppler, A.R.; Verma, A.H.; Conti, H.R.; Gaffen, S.L. Neutrophils Do Not Express IL-17A in the Context of Acute Oropharyngeal Candidiasis. Pathogens 2015, 4, 559-572. https://doi.org/10.3390/pathogens4030559
Huppler AR, Verma AH, Conti HR, Gaffen SL. Neutrophils Do Not Express IL-17A in the Context of Acute Oropharyngeal Candidiasis. Pathogens. 2015; 4(3):559-572. https://doi.org/10.3390/pathogens4030559
Chicago/Turabian StyleHuppler, Anna R., Akash H. Verma, Heather R. Conti, and Sarah L. Gaffen. 2015. "Neutrophils Do Not Express IL-17A in the Context of Acute Oropharyngeal Candidiasis" Pathogens 4, no. 3: 559-572. https://doi.org/10.3390/pathogens4030559
APA StyleHuppler, A. R., Verma, A. H., Conti, H. R., & Gaffen, S. L. (2015). Neutrophils Do Not Express IL-17A in the Context of Acute Oropharyngeal Candidiasis. Pathogens, 4(3), 559-572. https://doi.org/10.3390/pathogens4030559





