Monitoring Therapeutic Treatments against Burkholderia Infections Using Imaging Techniques
Abstract
:1. Introduction
2. Results and Discussion
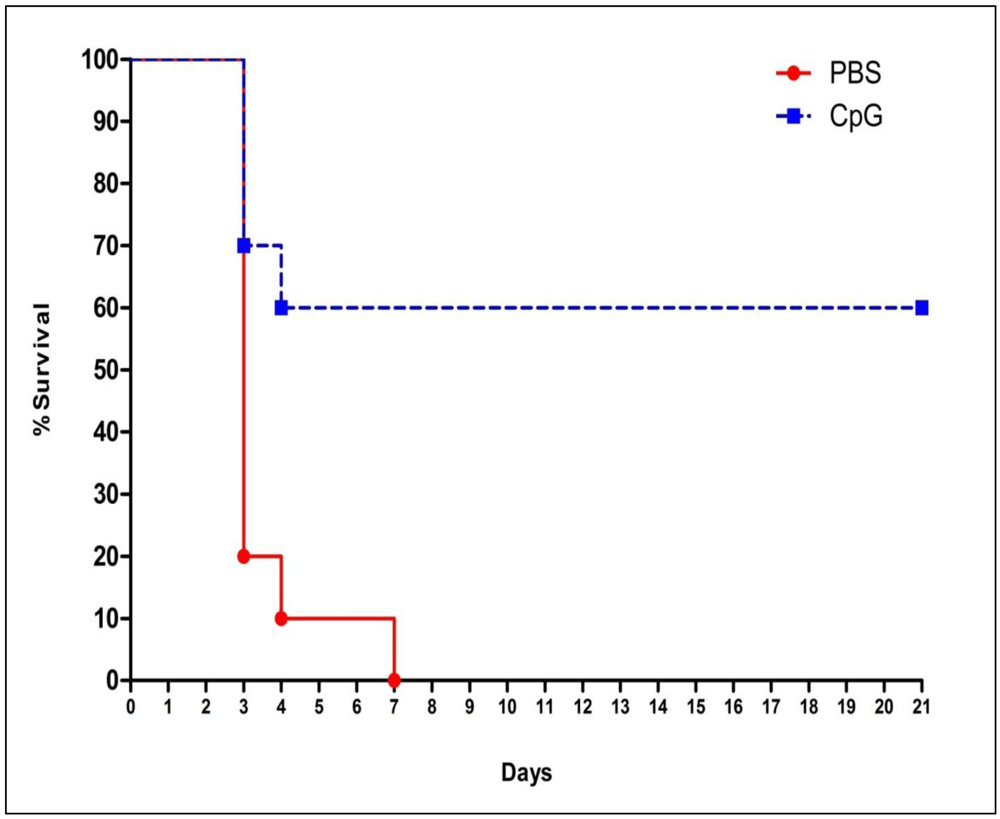
2.1. Class-C CpG ODNs Treatment Increases Percent Survival in BALB/c Mice
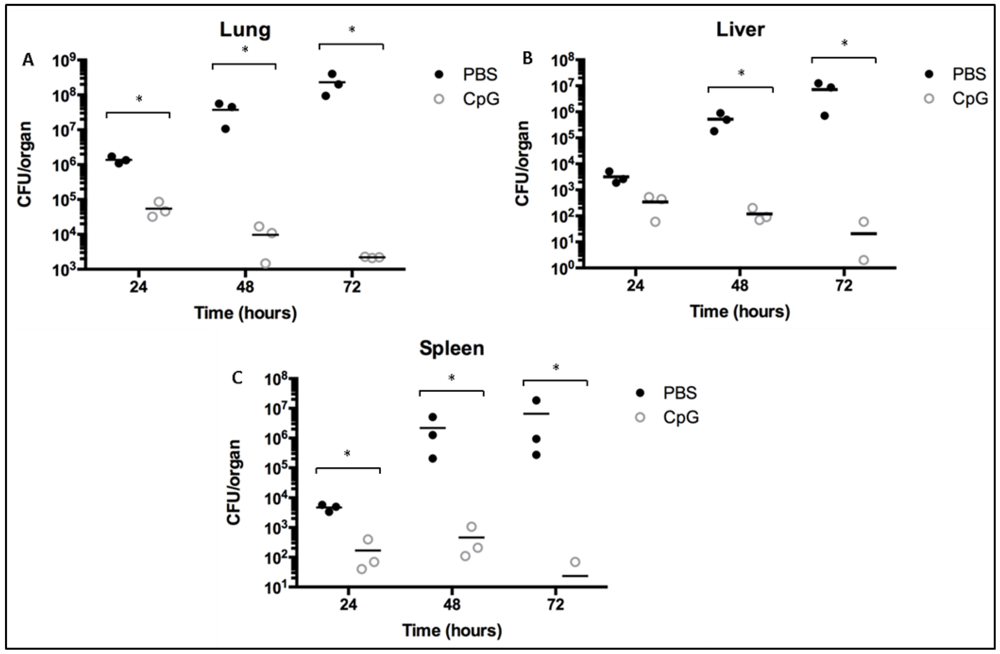
2.2. Class-C CpG ODNs Treatment Reduces Bacterial Load in BALB/c Mice
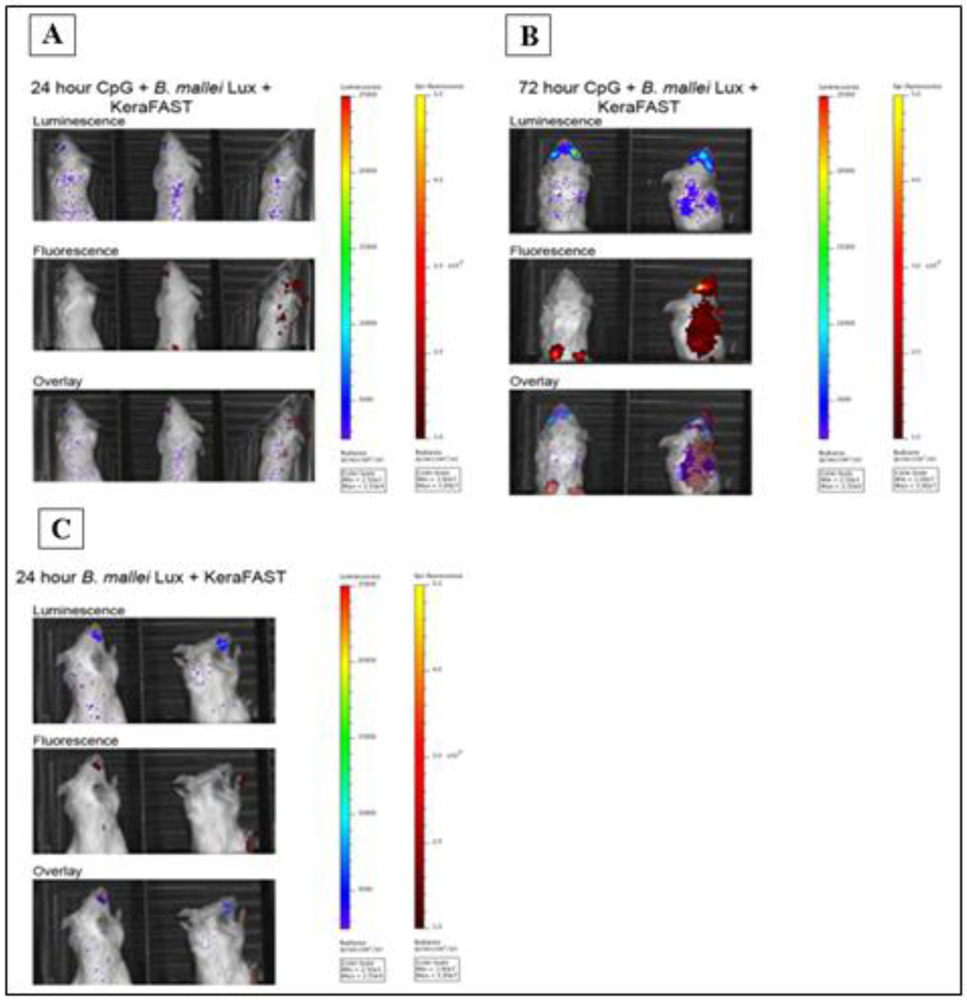
2.3. Class-C CpG ODNs Treatment Leads to Higher Neutrophil-Specific Fluorescence Signal in BALB/c Mice
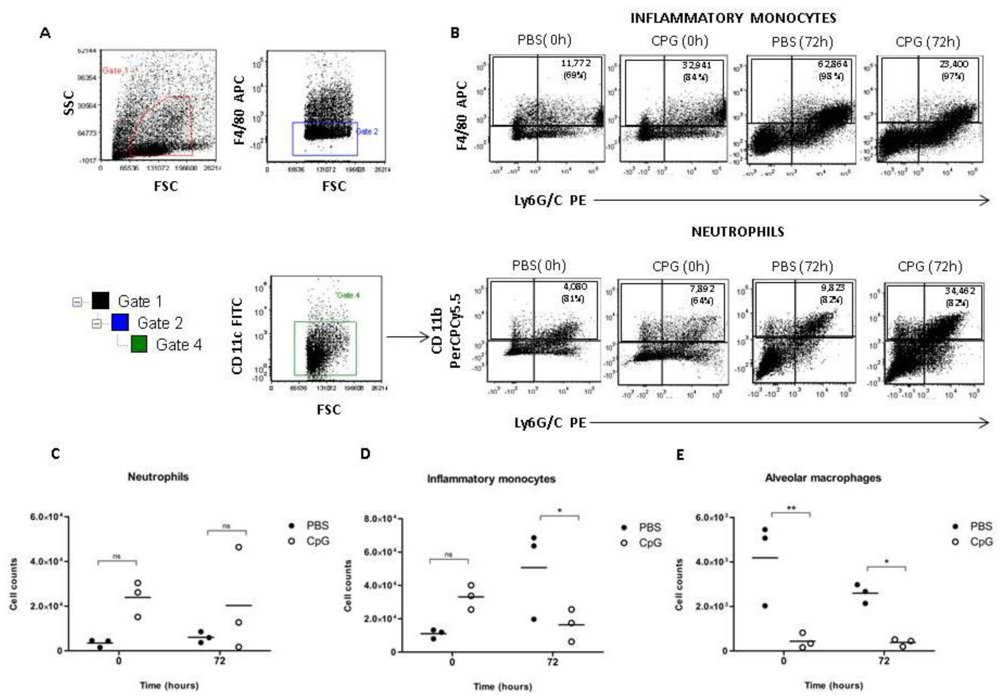
2.4. Class-C CpG ODNs Treatment Leads to Increase Levels of Inflammatory Monocytes during Early Infection of BALB/c Mice
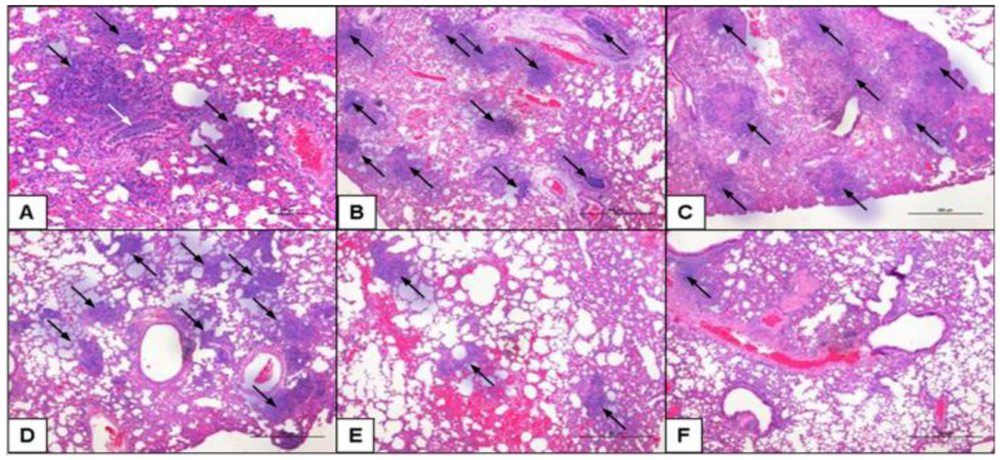
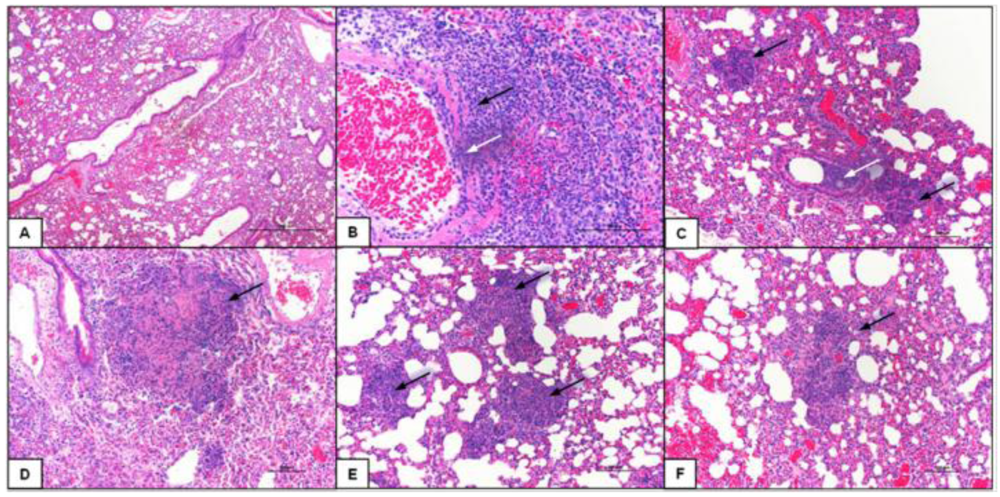
2.5. Class-C CpG ODNs Treatment Results in Decreased Tissue Damage in the Lungs
2.6. Discussion of Results
3. Experimental Section
3.1. Bacterial Strain
3.2. Mice
3.3. Ethics Statement
3.4. Survival Studies
3.5. Bacterial Burden
3.6. In vivo Imaging
3.7. Cell Preparation and Flow Cytometry
3.8. Histopathology of the Lungs
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Lehavi, O.; Aizenstien, O.; Katz, L.H.; Hourvitz, A. Glanders–a potential disease for biological warfare in humans and animals. Harefuah 2002, 141, 119. [Google Scholar]
- Neubauer, H.; Sprague, L.D.; Zacharia, R.; Tomaso, H.; Al Dahouk, S.; Wernery, R.; Wernery, U.; Scholz, H.C. Serodiagnosis of Burkholderia mallei infections in horses: State-of-the-art and perspectives. J. Vet. Med. B Infect. Dis Vet. Public Health 2005, 52, 201–205. [Google Scholar] [CrossRef]
- Whitlock, G.C.; Estes, D.M.; Torres, A.G. Glanders: Off to the races with Burkholderia mallei. FEMS Microbiol. Lett. 2007, 277, 115–122. [Google Scholar] [CrossRef]
- Khaki, P.; Mosavari, N.; Khajeh, N.S.; Emam, M.; Ahouran, M.; Hashemi, S.; Taheri, M.M.; Jahanpeyma, D.; Nikkhah, S. Glanders outbreak at Tehran Zoo, Iran. Iran J. Microbiol. 2012, 4, 3–7. [Google Scholar]
- Malik, P.; Singha, H.; Khurana, S.K.; Kumar, R.; Kumar, S.; Raut, A.A.; Riyesh, T.; Vaid, R.K.; Virmani, N.; Singh, B.K.; et al. Emergence and re-emergence of glanders in India: A description of outbreaks from 2006 to 2011. Vet. Ital. 2012, 48, 167–178. [Google Scholar]
- Wernery, U.; Wernery, R.; Joseph, M.; Al-Salloom, F.; Johnson, B.; Kinne, J.; Jose, S.; Jose, S.; Tappendorf, B.; Hornstra, H.; et al. Natural Burkholderia mallei infection in dromedary, Bahrain. Emerg. Infect. Dis. 2011, 17, 1277–1279. [Google Scholar] [CrossRef]
- De Carvalho Filho, M.B.; Ramos, R.M.; Fonseca, A.A.J.; de Lima Orzil, L.; Sales, M.L.; de Assis Santana, V.L.; de Souza, M.M.; Dos Reis Machado, E.; Filho, P.R.; Leite, R.C.; et al. Development and validation of a method for purification of mallein for the diagnosis of glanders in equines. BMC Vet. Res. 2012, 8, 154. [Google Scholar] [CrossRef]
- Ansari, M.; Minou, M. Two cases of chronic human glanders treated with sulfonamides. Ann. Inst. Pasteur (Paris) 1951, 81, 98–102. [Google Scholar]
- Deitchman, S.; Sokas, R. Glanders in a military research microbiologist. N. Engl. J. Med. 2001, 345, 1644. [Google Scholar]
- Anonymous. Acute glanders in the human subject. Prov. Med. J. Retrosp. Med. Sci. 1843, 6, 433–435. [Google Scholar]
- Coleman, W.; Ewing, J. A case of septicemic glanders in the human subject. J. Med. Res. 1903, 9, 223–240. [Google Scholar]
- Howe, C.; Miller, W.R. Human glanders; report of six cases. Ann. Intern. Med. 1947, 26, 93–115. [Google Scholar] [CrossRef]
- Estes, D.M.; Dow, S.W.; Schweizer, H.P.; Torres, A.G. Present and future therapeutic strategies for melioidosis and glanders. Exp. Rev. Anti Infect. Ther. 2010, 8, 325–338. [Google Scholar] [CrossRef]
- Easton, A.; Haque, A.; Chu, K.; Patel, N.; Lukaszewski, R.A.; Krieg, A.M.; Titball, R.W.; Bancroft, G.J. Combining vaccination and postexposure CpG therapy provides optimal protection against lethal sepsis in a biodefense model of human melioidosis. J. Infect. Dis. 2011, 204, 636–644. [Google Scholar]
- Judy, B.M.; Taylor, K.; Deeraksa, A.; Johnston, R.K.; Endsley, J.J.; Vijayakumar, S.; Aronson, J.F.; Estes, D.M.; Torres, A.G. Prophylactic application of CpG oligonucleotides augments the early host response and confers protection in acute melioidosis. PLoS One 2012, 7, e34176. [Google Scholar] [CrossRef]
- Rozak, D.A.; Gelhaus, H.C.; Smith, M.; Zadeh, M.; Huzella, L.; Waag, D.; Adamovicz, J.J. CpG oligodeoxyribonucleotides protect mice from Burkholderia pseudomallei but not Francisella tularensis Schu S4 aerosols. J. Immune Based Ther. Vaccines 2010, 8, 2. [Google Scholar] [CrossRef]
- Waag, D.M.; McCluskie, M.J.; Zhang, N.; Krieg, A.M. A CpG oligonucleotide can protect mice from a low aerosol challenge dose of Burkholderia mallei. Infect. Immun. 2006, 74, 1944–1948. [Google Scholar] [CrossRef]
- Wongratanacheewin, S.; Kespichayawattana, W.; Intachote, P.; Pichyangkul, S.; Sermswan, R.W.; Krieg, A.M.; Sirisinha, S. Immunostimulatory CpG oligodeoxynucleotide confers protection in a murine model of infection with Burkholderia pseudomallei. Infect. Immun. 2004, 72, 4494–4502. [Google Scholar] [CrossRef]
- Elkins, K.L.; Rhinehart-Jones, T.R.; Stibitz, S.; Conover, J.S.; Klinman, D.M. Bacterial DNA containing CpG motifs stimulates lymphocyte-dependent protection of mice against lethal infection with intracellular bacteria. J. Immunol. 1999, 162, 2291–2298. [Google Scholar]
- Becker, Y. CpG ODNs treatments of HIV-1 infected patients may cause the decline of transmission in high risk populations–a review, hypothesis and implications. Virus Genes 2005, 30, 251–266. [Google Scholar] [CrossRef]
- Deng, J.C.; Moore, T.A.; Newstead, M.W.; Zeng, X.; Krieg, A.M.; Standiford, T.J. CpG oligodeoxynucleotides stimulate protective innate immunity against pulmonary Klebsiella infection. J. Immunol. 2004, 173, 5148–5155. [Google Scholar]
- Hickey, A.J.; Lin, J.S.; Kummer, L.W.; Szaba, F.M.; Duso, D.K.; Tighe, M.; Parent, M.A.; Smiley, S.T. Intranasal prophylaxis with CpG oligodeoxynucleotide can protect against Yersinia pestis infection. Infect. Immun. 2013, 81, 2123–2132. [Google Scholar] [CrossRef]
- Klinman, D.M.; Conover, J.; Coban, C. Repeated administration of synthetic oligodeoxynucleotides expressing CpG motifs provides long-term protection against bacterial infection. Infect. Immun. 1999, 67, 5658–5663. [Google Scholar]
- Pun, P.B.; Bhat, A.A.; Mohan, T.; Kulkarni, S.; Paranjape, R.; Rao, D.N. Intranasal administration of peptide antigens of HIV with mucosal adjuvant CpG ODN coentrapped in microparticles enhances the mucosal and systemic immune responses. Int. Immunopharmacol. 2009, 9, 468–477. [Google Scholar] [CrossRef]
- Ray, N.B.; Krieg, A.M. Oral pretreatment of mice with CpG DNA reduces susceptibility to oral or intraperitoneal challenge with virulent Listeria monocytogenes. Infect. Immun. 2003, 71, 4398–4404. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Harker, J.A.; Wang, B.; Openshaw, P.J.; Tregoning, J.S.; Culley, F.J. Preexposure to CpG protects against the delayed effects of neonatal respiratory syncytial virus infection. J. Virol. 2012, 86, 10456–10461. [Google Scholar]
- Liu, Y.; Luo, X.; Yang, C.; Yu, S.; Xu, H. Three CpG oligodeoxynucleotide classes differentially enhance antigen-specific humoral and cellular immune responses in mice. Vaccine 2011, 29, 5778–5784. [Google Scholar] [CrossRef]
- Cooper, C.L.; Ahluwalia, N.K.; Efler, S.M.; Vollmer, J.; Krieg, A.M.; Davis, H.L. Immunostimulatory effects of three classes of CpG oligodeoxynucleotides on PBMC from HCV chronic carriers. J. Immune Based Ther. Vaccines 2008, 6, 3. [Google Scholar] [CrossRef]
- Ballas, Z.K.; Rasmussen, W.L.; Krieg, A.M. Induction of NK activity in murine and human cells by CpG motifs in oligodeoxynucleotides and bacterial DNA. J. Immunol. 1996, 157, 1840–1845. [Google Scholar]
- Asselin-Paturel, C.; Brizard, G.; Chemin, K.; Boonstra, A.; O'Garra, A.; Vicari, A.; Trinchieri, G. Type I interferon dependence of plasmacytoid dendritic cell activation and migration. J. Exp. Med. 2005, 201, 1157–1167. [Google Scholar] [CrossRef]
- Gursel, M.; Verthelyi, D.; Gürsel, I.; Ishii, K.J.; Klinman, D.M. Differential and competitive activation of human immune cells by distinct classes of CpG oligodeoxynucleotide. J. Leukoc Biol. 2002, 71, 813–820. [Google Scholar]
- Krug, A.; Rothenfusser, S.; Hornung, V.; Jahrsdörfer, B.; Blackwell, S.; Ballas, Z.K.; Endres, S.; Krieg, A.M.; Hartmann, G. Identification of CpG oligonucleotide sequences with high induction of IFN-alpha/beta in plasmacytoid dendritic cells. Eur. J. Immunol. 2001, 31, 2154–2163. [Google Scholar] [CrossRef]
- Vollmer, J.; Weeratna, R.; Payette, P.; Jurk, M.; Schetter, C.; Laucht, M.; Wader, T.; Tluk, S.; Liu, M.; Davis, H.L.; et al. Characterization of three CpG oligodeoxynucleotide classes with distinct immunostimulatory activities. Eur. J. Immunol. 2004, 34, 251–262. [Google Scholar] [CrossRef]
- Bernasconi, N.L.; Onai, N.; Lanzavecchia, A. A role for toll-like receptors in acquired immunity: Up-regulation of TLR9 by BDR triggering in naive B cells and constitutive expression in memory B cells. Blood 2003, 101, 4500–4504. [Google Scholar] [CrossRef]
- Hartmann, G.; Battiany, J.; Poeck, H.; Wagner, M.; Kerkmann, M.; Lubenow, N.; Rothenfusser, S.; Endres, S. Rational design of new CpG oligonucleotides that combine B cell activation with high IFN-alpha induction in plasmacytoid dendritic cells. Eur. J. Immunol. 2003, 33, 1633–1641. [Google Scholar] [CrossRef]
- Marshall, J.D.; Fearon, K.; Abbate, C.; Subramanian, S.; Yee, P.; Gregorio, J.; Coffman, R.L.; Van Nest, G. Identification of a novel CpG DNA class and motif that optimally stimulate B cell and plasmacytoid dendritic cell functions. J. Leukoc Biol. 2003, 73, 781–792. [Google Scholar] [CrossRef]
- Galyov, E.E.; Brett, P.J.; Deshazer, D. Molecular insights into Burkholderia pseudomallei and Burkholderia mallei pathogenesis. Annu. Rev. Microbiol. 2010, 64, 495–517. [Google Scholar] [CrossRef]
- Massey, S.; Johnston, K.; Mott, T.M.; Judy, B.M.; Kvitko, B.H.; Schweizer, H.P.; Estes, D.M.; Torres, A.G. In vivo bioluminescence imaging of Burkholderia mallei respiratory infection and treatment in the mouse model. Front. Microbiol. 2011, 2, 174. [Google Scholar]
- Deng, J.C.; Moore, T.A.; Newstead, M.W.; Zeng, X.; Krieg, A.M.; Standiford, T.J. Cpg oligodeoxynucleotides stimulate protective innate immunity against pulmonary Klebsiella infection. J. Immunol. 2004, 173, 5148–5155. [Google Scholar]
- Puangpetch, A.; Anderson, R.; Huang, Y.Y.; Sermswan, R.W.; Chaicumpa, W.; Sirisinha, S.; Wongratanacheewin, S. Cationic liposomes extend the immunostimulatory effect of CpG oligodeoxynucleotide against Burkholderia pseudomallei infection in BALB/c mice. Clin. Vaccine Immunol. 2012, 19, 675–683. [Google Scholar] [CrossRef]
- Utaisincharoen, P.; Kespichayawattana, W.; Anuntagool, N.; Chaisuriya, P.; Pichyangkul, S.; Krieg, A.M.; Sirisinha, S. CpG ODN enhances uptake of bacteria by mouse macrophages. Clin. Exp. Immunol. 2003, 132, 70–75. [Google Scholar] [CrossRef]
- Krieg, A.M. CpG motifs in bacterial DNA and their immune effects. Annu. Rev. Immunol. 2002, 20, 709–760. [Google Scholar] [CrossRef]
- Bondi, S.K.; Goldberg, J.B. Strategies toward vaccines against Burkholderia mallei and Burkholderia pseudomallei. Expert Rev. Vaccines 2008, 7, 1357–1365. [Google Scholar] [CrossRef]
- Wedlock, D.N.; Denis, M.; Skinner, M.A.; Koach, J.; de Lisle, G.W.; Vordermeier, H.M.; Hewinson, R.G.; van Drunen Littel-van den Hurk, S.; Babiuk, L.A.; Hecker, R.; et al. Vaccination of cattle with a CpG oligodeoxynucleotide-formulated mycobacterial protein vaccine and Mycobacterium bovis BCG induces levels of protection against bovine tuberculosis superior to those induced by vaccination with BCG alone. Infect. Immun. 2005, 73, 3540–3546. [Google Scholar] [CrossRef]
- Ren, T.; Liang, Y.J.; Cai, Y.Y.; Li, C.Y.; Mei, J.; Yuan, Z.H.; Tao, M.F.; Tian, M.; Zhao, B. A study on the mechanisms of mycobacterial clearance induced by CpG-oligodeoxynucleotides in mice. Zhonghua Jie He He Hu Xi Za Zhi 2008, 31, 46–50. [Google Scholar]
- Appelberg, R. Neutrophils and intracellular pathogens: Beyond phagocytosis and killing. Trends Microbiol. 2007, 15, 87–92. [Google Scholar] [CrossRef]
- Kumar, V.; Sharma, A. Neutrophils: Cinderella of innate immune system. Int. Immunopharmacol. 2010, 10, 1325–1334. [Google Scholar] [CrossRef]
- Xiao, L.; Zhang, Y.; Liu, Z.; Yang, M.; Pu, L.; Pan, D. Synthesis of the cyanine 7 labeled neutrophil-specific agents for noninvasive near infrared fluorescence imaging. Bioorg. Med. Chem. Lett. 2010, 20, 3515–3517. [Google Scholar] [CrossRef]
- Blasi, F.; Tarsia, P.; Aliberti, S. Strategic targets of essential host-pathogen interactions. Respiration 2005, 72, 9–25. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mott, T.M.; Johnston, R.K.; Vijayakumar, S.; Estes, D.M.; Motamedi, M.; Sbrana, E.; Endsley, J.J.; Torres, A.G. Monitoring Therapeutic Treatments against Burkholderia Infections Using Imaging Techniques. Pathogens 2013, 2, 383-401. https://doi.org/10.3390/pathogens2020383
Mott TM, Johnston RK, Vijayakumar S, Estes DM, Motamedi M, Sbrana E, Endsley JJ, Torres AG. Monitoring Therapeutic Treatments against Burkholderia Infections Using Imaging Techniques. Pathogens. 2013; 2(2):383-401. https://doi.org/10.3390/pathogens2020383
Chicago/Turabian StyleMott, Tiffany M., R. Katie Johnston, Sudhamathi Vijayakumar, D. Mark Estes, Massoud Motamedi, Elena Sbrana, Janice J. Endsley, and Alfredo G. Torres. 2013. "Monitoring Therapeutic Treatments against Burkholderia Infections Using Imaging Techniques" Pathogens 2, no. 2: 383-401. https://doi.org/10.3390/pathogens2020383





