Effect of trans-Cinnamaldehyde on Adhesion and Other Virulence Factors of Methicillin-Resistant Staphylococcus aureus
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Isolates
2.2. Evaluation of Adhesion of MRSA Isolates Treated with TC to Proteins of the Host Plasma and Extracellular Matrix
2.3. Assessment of Virulence Factor Production by MRSA Treated with TC
2.3.1. Extracellular Protease Production
2.3.2. Extracellular DNase Production
2.3.3. Esterase Production
2.3.4. Hemolysis Assay
2.4. Cytotoxicity Study of TC on Human Fibroblast Cell Culture In Vitro
2.5. Statistical Analysis
3. Results
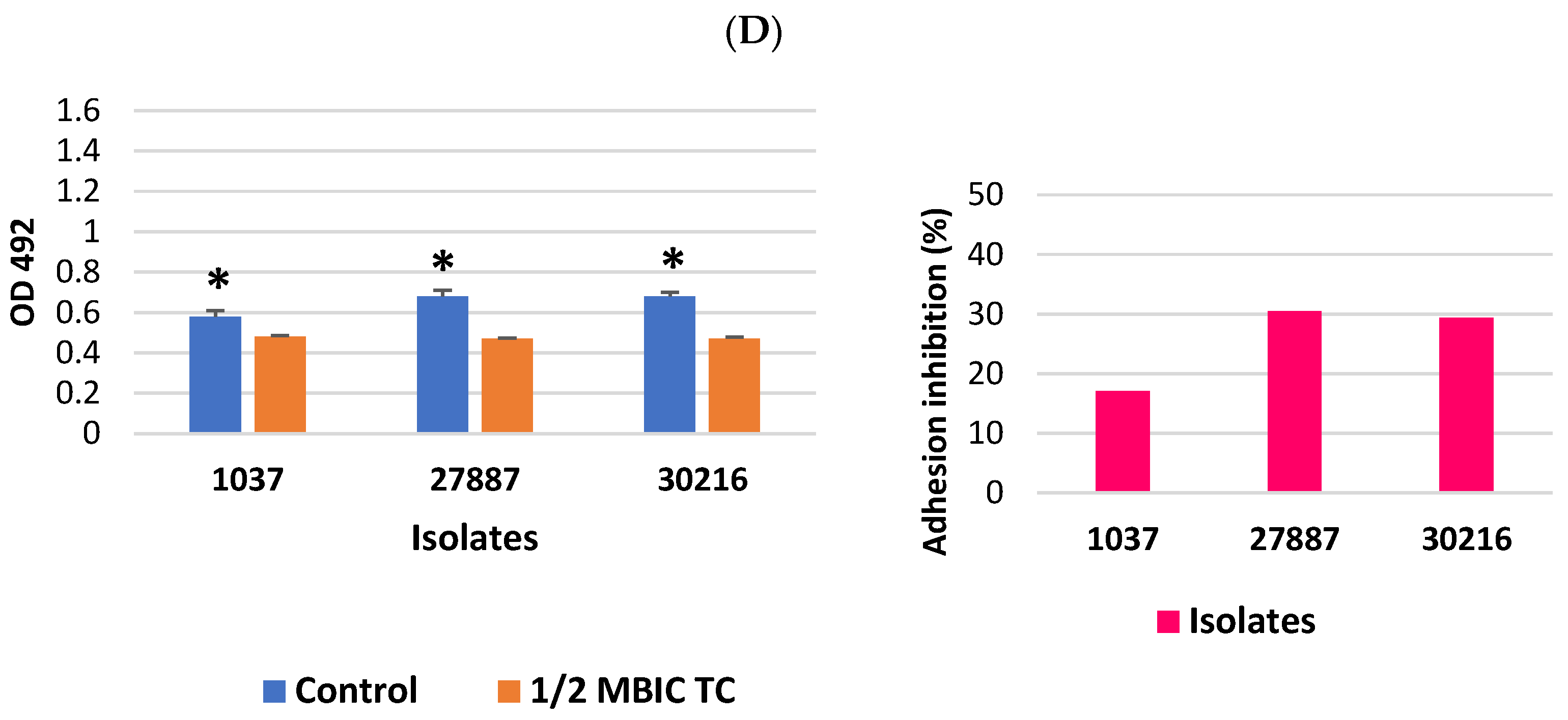
3.1. Inhibitory Effect of TC on Adhesion of MRSA Biofilm Cells to Proteins Present in the Host Plasma and Extracellular Matrix
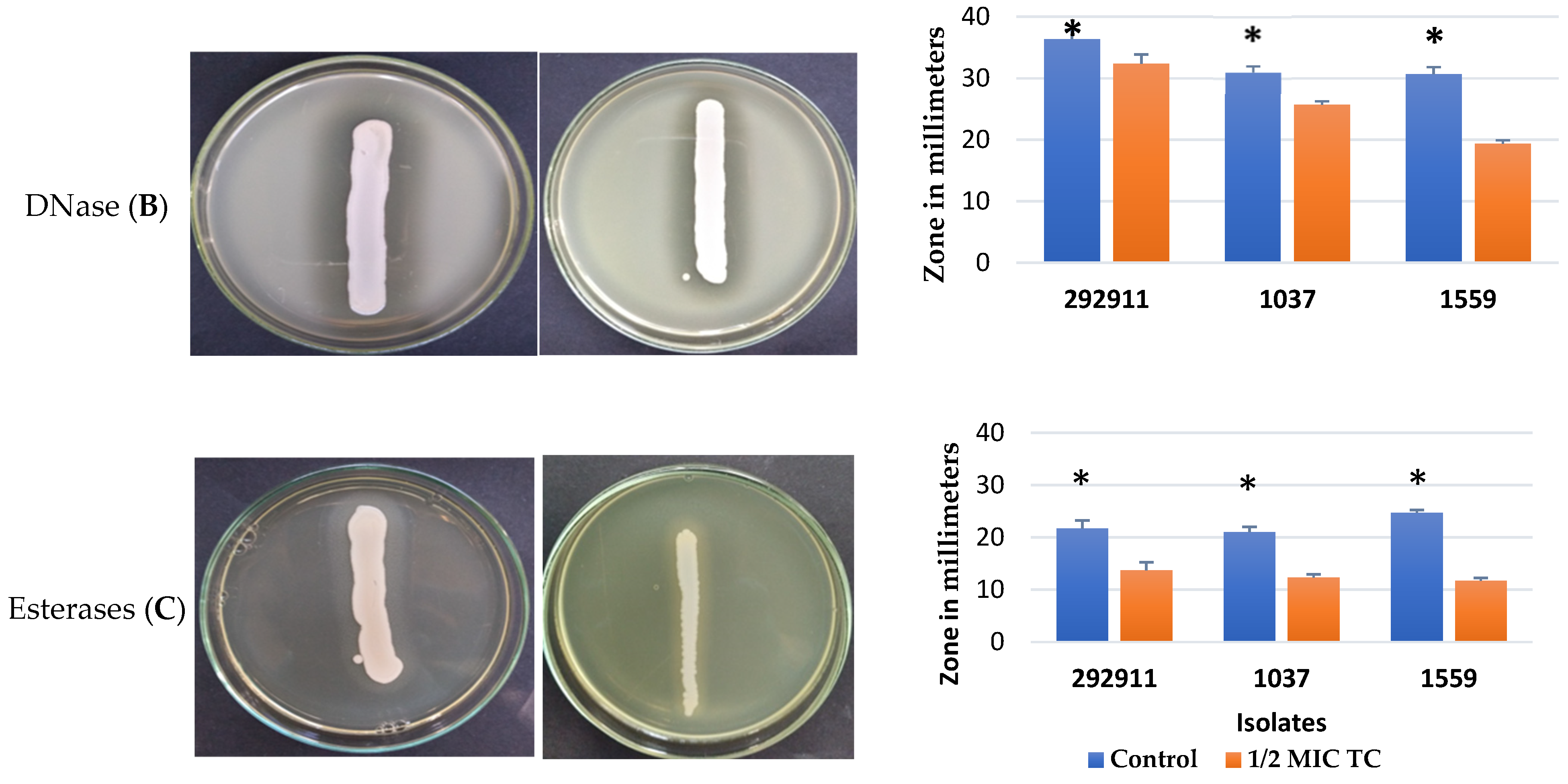
3.2. Inhibitory Effect of TC on Enzyme Production by MRSA Isolates
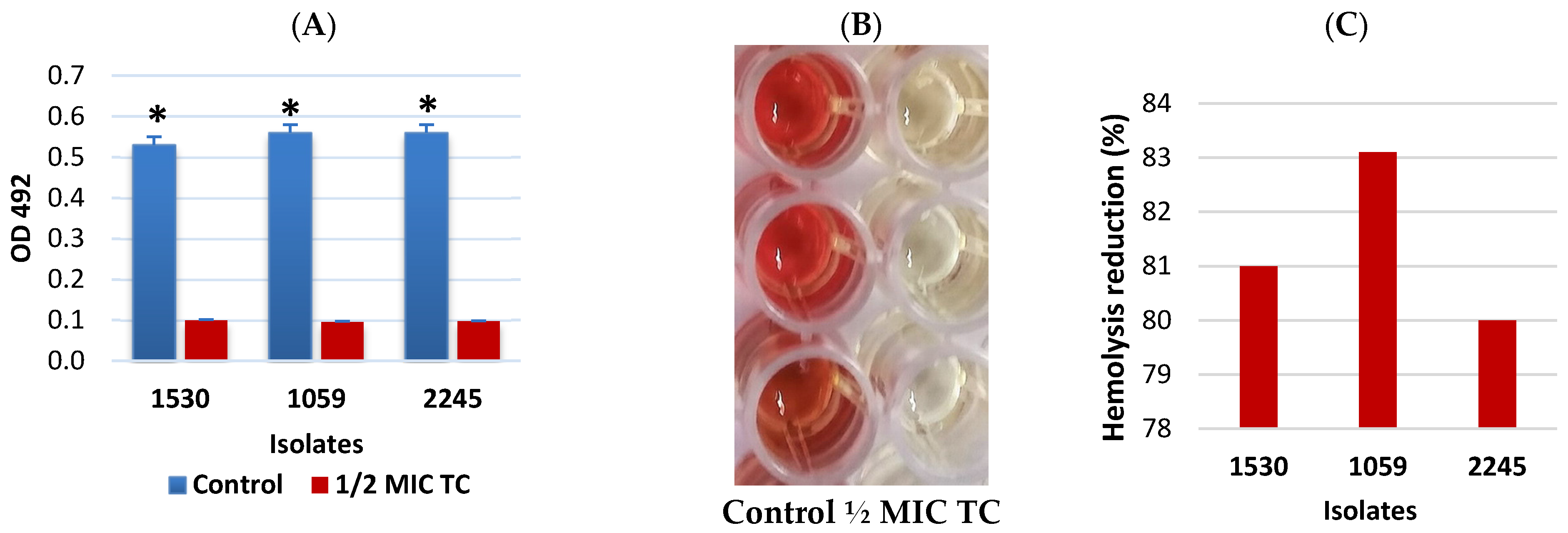
3.3. Inhibitory Effect of TC on Hemolytic Activity of MRSA Isolates
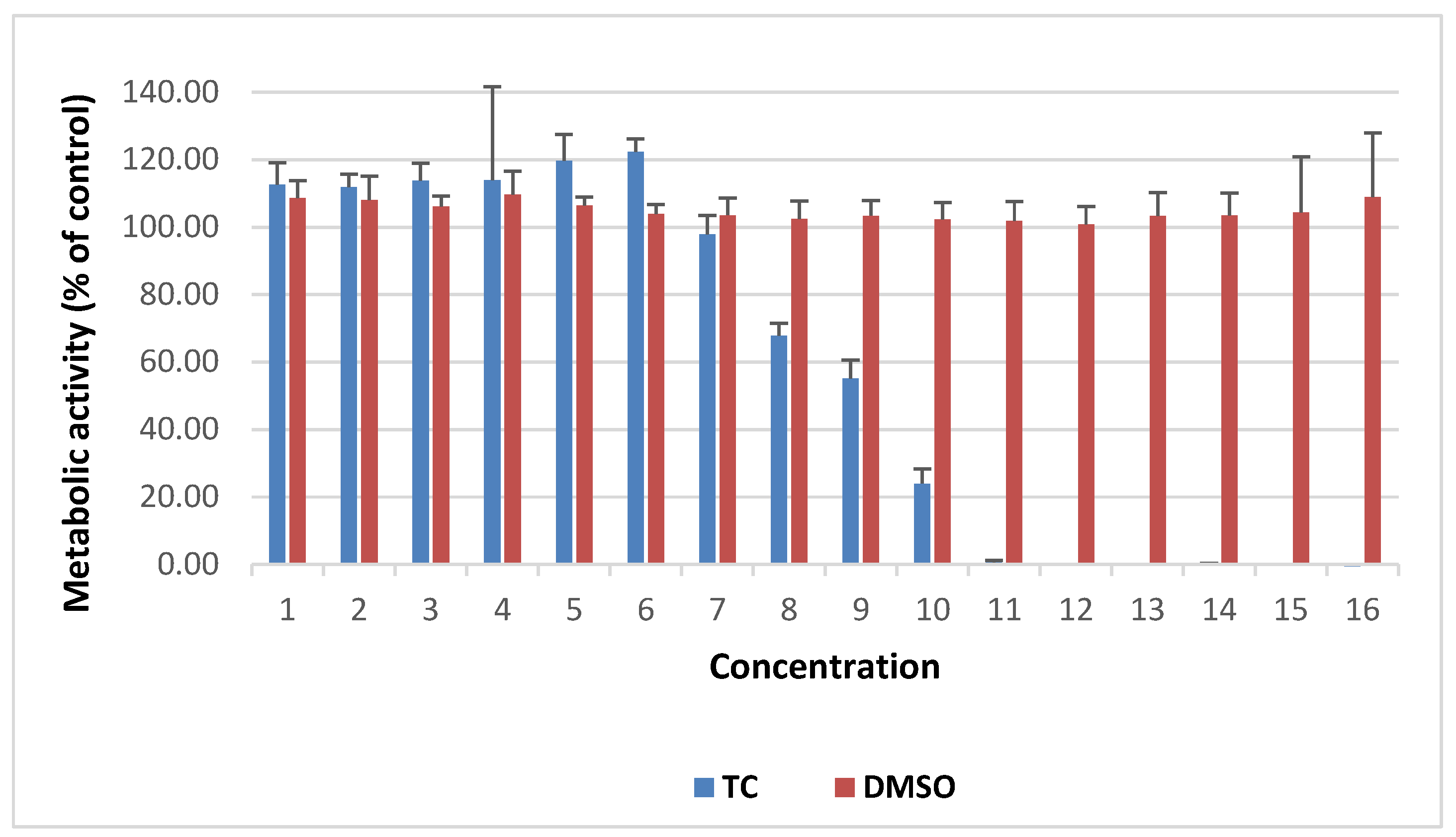
3.4. The Effect of TC on Fibroblast Viability In Vitro
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. Global Antibiotic Resistance Surveillance Report 2025: WHO Global Antimicrobial Resistance and Use Surveillance System (GLASS)—Summary; World Health Organization: Geneva, Switzerland, 2025. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). Antimicrobial Resistance in the EU/EEA (EARS-Net)—Annual Epidemiological Report for 2024; ECDC: Stockholm, Sweden, 2025. [Google Scholar]
- Katayama, Y.; Ito, T.; Hiramatsu, K. A new class of genetic element, staphylococcus cassette chromosome mec, encodes methicillin resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 2000, 44, 1549–1555. [Google Scholar] [CrossRef] [PubMed]
- Kot, B.; Wierzchowska, K.; Piechota, M.; Grużewska, A. Antimicrobial resistance patterns in methicillin-resistant Staphylococcus aureus from patients hospitalized during 2015–2017 in hospitals in Poland. Med. Princ. Pract. 2020, 29, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.H.; Dieter, M.P.; Bucher, J.R.; Jameson, C.W. Toxicokinetics of cinnamaldehyde in F344 rats. Food Chem. Toxicol. 1992, 30, 997–1004. [Google Scholar] [CrossRef]
- Ma, W.Y.; Shen, K.S.; Wang, Z.; Liu, Q.; Diao, X.J.; Liu, G.R. Synergistic antimicrobial effect and mechanism of enterocin Gr17 and cinnamaldehyde against Escherichia coli and Candida albicans. Arch. Microbiol. 2025, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, I.; Upadhyay, A.; Kollanoor-Johny, A.; Mooyottu, S.; Baskaran, S.A.; Yin, H.B.; Schreiber, D.T.; Khan, M.I.; Darre, M.J.; Curtis, P.A.; et al. In-feed supplementation of trans-cinnamaldehyde reduces layer-chicken eggborne transmission of Salmonella enterica serovar enteritidis. Appl. Environ. Microbiol. 2015, 81, 2985–2994. [Google Scholar] [CrossRef]
- Deng, J.; Wang, G.; Li, J.; Zhao, Y.; Wang, X. Effects of cinnamaldehyde on the cell wall of A. fumigatus and its application in treating mice with invasive pulmonary aspergillosis. Evid. Based Complement. Altern. Med. 2018, 2018, 5823209. [Google Scholar] [CrossRef]
- Xu, J.; Lin, Q.; Sheng, M.; Ding, T.; Li, B.; Gao, Y.; Tan, Y. Antibiofilm effect of cinnamaldehyde-chitosan nanoparticles against the biofilm of Staphylococcus aureus. Antibiotics 2022, 11, 1403. [Google Scholar] [CrossRef]
- Usai, F.; Di Sotto, A. trans-cinnamaldehyde as a novel candidate to overcome bacterial resistance: An overview of in vitro studies. Antibiotics 2023, 12, 254. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, S.; Cho, K.-H.; Lee, J.-H.; Lee, J. Antibiofilm activities of cinnamaldehyde analogs against uropathogenic Escherichia coli and Staphylococcus aureus. Int. J. Mol. Sci. 2022, 23, 7225. [Google Scholar] [CrossRef]
- Ferro, T.A.; Araújo, J.M.; Dos Santos Pinto, B.L.; Dos Santos, J.S.; Souza, E.B.; da Silva, B.L.; Colares, V.L.; Novais, T.M.; Filho, C.M.; Struve, C.; et al. Cinnamaldehyde inhibits Staphylococcus aureus virulence factors and protects against infection in a Galleria mellonella model. Front. Microbiol. 2016, 7, 2052, Erratum in Front. Microbiol. 2021, 11, 628074. https://doi.org/10.3389/fmicb.2020.628074. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kot, B.; Sytykiewicz, H.; Sprawka, I.; Witeska, M. Effect of trans-cinnamaldehyde on methicillin-resistant Staphylococcus aureus biofilm formation: Metabolic activity assessment and analysis of the biofilm-associated genes expression. Int. J. Mol. Sci. 2019, 21, 102. [Google Scholar] [CrossRef]
- Piechota, M.; Kot, B.; Frankowska-Maciejewska, A.; Grużewska, A.; Woźniak-Kosek, A. Biofilm formation by methicillin-resistant and methicillin-sensitive Staphylococcus aureus strains from hospitalized patients in Poland. Biomed Res. Int. 2018, 2018, 4657396. [Google Scholar] [CrossRef]
- Kong, C.; Chee, C.F.; Richter, K.; Thomas, N.; Abd Rahman, N.; Nathan, S. Suppression of Staphylococcus aureus biofilm formation and virulence by a benzimidazole derivative, UM-C162. Sci. Rep. 2018, 8, 2758. [Google Scholar] [CrossRef]
- Chapin, K.C.; Murray, P.R. Media. In Manual of Clinical Microbiology; Murray, P.R., Baron, E.J., Pfaller, M.A., Tenover, F.C., Yolken, R.H., Eds.; American Society for Microbiology: Washington, DC, USA, 1999; pp. 1687–1707. [Google Scholar]
- Selvaraj, A.; Jayasree, T.; Valliammai, A.; Pandian, S.K. Myrtenol attenuates MRSA biofilm and virulence by suppressing sarA expression dynamism. Front. Microbiol. 2019, 10, 2027. [Google Scholar] [CrossRef]
- Figueiredo, C.S.S.E.S.; Oliveira, P.V.; Saminez, W.F.D.S.; Diniz, R.M.; Mendonça, J.S.P.; Silva, L.D.S.; Paiva, M.Y.M.; Nascimento, M.S.D.; Aliança, A.S.D.S.; Zagmignan, A.; et al. Immunomodulatory effects of cinnamaldehyde in Staphylococcus aureus-infected wounds. Molecules 2023, 28, 1204. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Heras, B.; Scanlon, M.J.; Martin, J.L. Targeting virulence not viability in the search for future antibacterials. Br. J. Clin. Pharmacol. 2015, 9, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Moormeier, D.E.; Bayles, K.W. Staphylococcus aureus bioflm: A complex developmental organism. Mol. Microbiol. 2017, 104, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, J.; Ji, Y. Environmental factors modulate biofilm formation by Staphylococcus aureus. Sci. Prog. 2020, 103, 1–14. [Google Scholar] [CrossRef]
- Foster, T.J.; Geoghegan, J.A.; Ganesh, V.K.; Höök, M. Adhesion, invasion and evasion: The many functions of the surface proteins of Staphylococcus aureus. Nat. Rev. Microbiol. 2014, 12, 49–62. [Google Scholar] [CrossRef]
- Hartford, O.M.; Wann, E.R.; Höök, M.; Foster, T.J. Identification of residues in the Staphylococcus aureus fibrinogen-binding MSCRAMM clumping factor A (ClfA) that are important for ligand binding. J. Biol. Chem. 2001, 276, 2466–2473. [Google Scholar] [CrossRef]
- Downer, R.; Roche, F.; Park, P.W.; Mecham, R.P.; Foster, T.J. The elastin-binding protein of Staphylococcus aureus (EbpS) is expressed at the cell surface as an integral membrane protein and not as a cell wall-associated protein. J. Biol. Chem. 2002, 277, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, C.R.; Postol, E.; Nomizo, R.; Reis, L.F.; Brentani, R.R. Identification of enolase as a laminin-binding protein on the surface of Staphylococcus aureus. Microbes. Infect. 2004, 6, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Madani, A.; Garakani, K.; Mofrad, M.R.K. Molecular mechanics of Staphylococcus aureus adhesin, CNA, and the inhibition of bacterial adhesion by stretching collagen. PLoS ONE 2017, 12, e0179601. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Dai, Y.; Chen, H.; He, X.; Ouyang, P.; Huang, X.; Sun, X.; Ai, Y.; Lai, S.; Zhu, L.; et al. Cinnamaldehyde resist Salmonella Typhimurium adhesion by inhibiting type I fimbriae. Molecules 2022, 10, 7753. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tam, K.; Torres, V.J. Staphylococcus aureus secreted toxins and extracellular enzymes. Microbiol. Spectr. 2019, 7, 10. [Google Scholar] [CrossRef]
- Faleye, O.S.; Sathiyamoorthi, E.; Lee, J.-H.; Lee, J. Inhibitory effects of cinnamaldehyde derivatives on biofilm formation and virulence factors in vibrio species. Pharmaceutics 2021, 13, 2176. [Google Scholar] [CrossRef]
- Li, T.; Wang, D.; Liu, N.; Ma, Y.; Ding, T.; Mei, Y.; Li, J. Inhibition of quorum sensing-controlled virulence factors and biofilm formation in Pseudomonas fluorescens by cinnamaldehyde. Int. J. Food Microbiol. 2018, 23, 98–106. [Google Scholar] [CrossRef]
- Ali, I.A.A.; Matinlinna, J.P.; Lévesque, C.M.; Neelakantan, P. trans-cinnamaldehyde attenuates Enterococcus faecalis virulence and inhibits biofilm formation. Antibiotics 2021, 10, 702. [Google Scholar] [CrossRef]
- Jaeger, K.E.; Ransac, S.; Dijkstra, B.W.; Colson, C.; van Heuvel, M.; Misset, O. Bacterial lipases. FEMS Microbiol. Rev. 1994, 15, 29–63. [Google Scholar] [CrossRef]
- Rollof, J.; Vinge, E.; Nilsson-Ehle, P.; Braconier, J.H. Aggregation of human granulocytes by Staphylococcus aureus lipase. J. Med. Microbiol. 1992, 36, 52–55. [Google Scholar] [CrossRef][Green Version]
- Rollof, J.; Hedström, S.A.; Nilsson-Ehle, P. Lipolytic activity of Staphylococcus aureus strains from disseminated and localized infections. Acta Pathol. Microbiol. Immunol. Scand. B 1987, 95, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Xiong, N.; Zhang, Y.; Rayner, S.; Chen, S. Functional characterization of lipase in the pathogenesis of Staphylococcus aureus. Biochem. Biophys. Res. Commun. 2012, 419, 617–620. [Google Scholar] [CrossRef]
- Li, S.; Zhou, S.; Yang, Q.; Liu, Y.; Yang, Y.; Xu, N.; Ai, X.; Dong, J. Cinnamaldehyde decreases the pathogenesis of Aeromonas hydrophila by inhibiting quorum sensing and biofilm formation. Fishes 2023, 8, 122. [Google Scholar] [CrossRef]
- Tang, J.; Zhou, R.; Shi, X.; Kang, M.; Wang, H.; Chen, H. Two thermostable nucleases coexisted in Staphylococcus aureus: Evidence from mutagenesis and in vitro expression. FEMS Microbiol. Lett. 2008, 284, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Berends, E.T.; Horswill, A.R.; Haste, N.M.; Monestier, M.; Nizet, V.; von Köckritz-Blickwede, M. Nuclease expression by Staphylococcus aureus facilitates escape from neutrophil extracellular traps. J. Innate. Immun. 2010, 2, 576–586. [Google Scholar] [CrossRef]
- Thammavongsa, V.; Missiakas, D.M.; Schneewind, O. Staphylococcus aureus degrades neutrophil extracellular traps to promote immune cell death. Science 2013, 342, 863–866. [Google Scholar] [CrossRef]
- Bhakdi, S.; Muhly, M.; Füssle, R. Correlation between toxin binding and hemolytic activity in membrane damage by staphylococcal-toxin. Infect. Immun. 1984, 46, 318–323. [Google Scholar] [CrossRef]
- Nygaard, T.K.; Pallister, K.B.; DuMont, A.L.; DeWald, M.; Watkins, R.L.; Pallister, E.Q.; Malone, C.; Griffith, S.; Horswill, A.R.; Torres, V.J.; et al. Alpha-toxin induces programmed cell death of human T cells, B cells, and monocytes during USA 300 infection. PLoS ONE 2012, 7, e36532. [Google Scholar] [CrossRef]
- Zhu, L.; He, Z.; Li, M.; Xu, J.; Ding, W.; Zeng, W.; Jiang, X. Antimicrobial and antivirulence function of cinnamaldehyde against Streptococcus suis type 2. Microbiol. Spectr. 2025, 13, e0256124. [Google Scholar] [CrossRef]
- Bickers, D.; Calow, P.; Greim, H.; Hanifin, J.M.; Rogers, A.E.; Saurat, J.H.; Sipes, I.G.; Smith, R.L.; Tagami, H.; RIFM expert panel. A toxicologic and dermatologic assessment of cinnamyl alcohol, cinnamaldehyde and cinnamic acid when used as fragrance ingredients. Food Chem. Toxicol. 2005, 43, 799–836. [Google Scholar] [CrossRef]
- ISO 10993-5:2009; Biological Evaluation of Medical devicesPart 5: Tests for in Vitro Cytotoxicity. International Organization for Standardization (ISO): Geneva, Switzerland, 2009.
- Dugoua, J.J.; Seely, D.; Perri, D.; Cooley, K.; Forelli, T.; Mills, E.; Koren, G. From type 2 diabetes to antioxidant activity: A systematic review of the safety and efficacy of common and cassia cinnamon bark. Can. J. Physiol. Pharmacol. 2007, 85, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Wang, S.-M.; Lu, Q.; Luo, J.; Cheng, Y.-X. Identification of compounds from the water soluble extract of cinnamomum cassia barks and their inhibitory effects against high-glucose-induced mesangial cells. Molecules 2013, 18, 10930–10943. [Google Scholar] [CrossRef]



| Isolate ID | Source of Isolation | TC Concentrations (µg/mL) |
|---|---|---|
| Adhesion assay | ||
| 1037 | Anus | 60 |
| 27887 | Wound | 240 |
| 30216 | Wound | 60 |
| Assay of protease, DNase and esterase production | ||
| 292911 | Nose | 30 |
| 1037 | Anus | 30 |
| 1559 | Wound | 30 |
| Hemolysis assay | ||
| 1530 | Wound | 30 |
| 1059 | Nose | 30 |
| 2245 | Wound | 30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kot, B.; Wierzchowska, K.; Grużewska, A.; Trafny, E.A.; Stępińska, M.; Witeska, M. Effect of trans-Cinnamaldehyde on Adhesion and Other Virulence Factors of Methicillin-Resistant Staphylococcus aureus. Pathogens 2026, 15, 271. https://doi.org/10.3390/pathogens15030271
Kot B, Wierzchowska K, Grużewska A, Trafny EA, Stępińska M, Witeska M. Effect of trans-Cinnamaldehyde on Adhesion and Other Virulence Factors of Methicillin-Resistant Staphylococcus aureus. Pathogens. 2026; 15(3):271. https://doi.org/10.3390/pathogens15030271
Chicago/Turabian StyleKot, Barbara, Kamila Wierzchowska, Agata Grużewska, Elżbieta Anna Trafny, Małgorzata Stępińska, and Małgorzata Witeska. 2026. "Effect of trans-Cinnamaldehyde on Adhesion and Other Virulence Factors of Methicillin-Resistant Staphylococcus aureus" Pathogens 15, no. 3: 271. https://doi.org/10.3390/pathogens15030271
APA StyleKot, B., Wierzchowska, K., Grużewska, A., Trafny, E. A., Stępińska, M., & Witeska, M. (2026). Effect of trans-Cinnamaldehyde on Adhesion and Other Virulence Factors of Methicillin-Resistant Staphylococcus aureus. Pathogens, 15(3), 271. https://doi.org/10.3390/pathogens15030271






