2000–2025: A Quarter of a Century of Studies on Pet Ownership in the Amazon—Epidemiological Implications for Public Health
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search Protocol
2.1.1. Study Eligibility Criteria
Inclusion Criteria
Exclusion Criteria
Data Extraction
2.2. Review Registration
2.3. Risk of Bias and Methodological Limitations
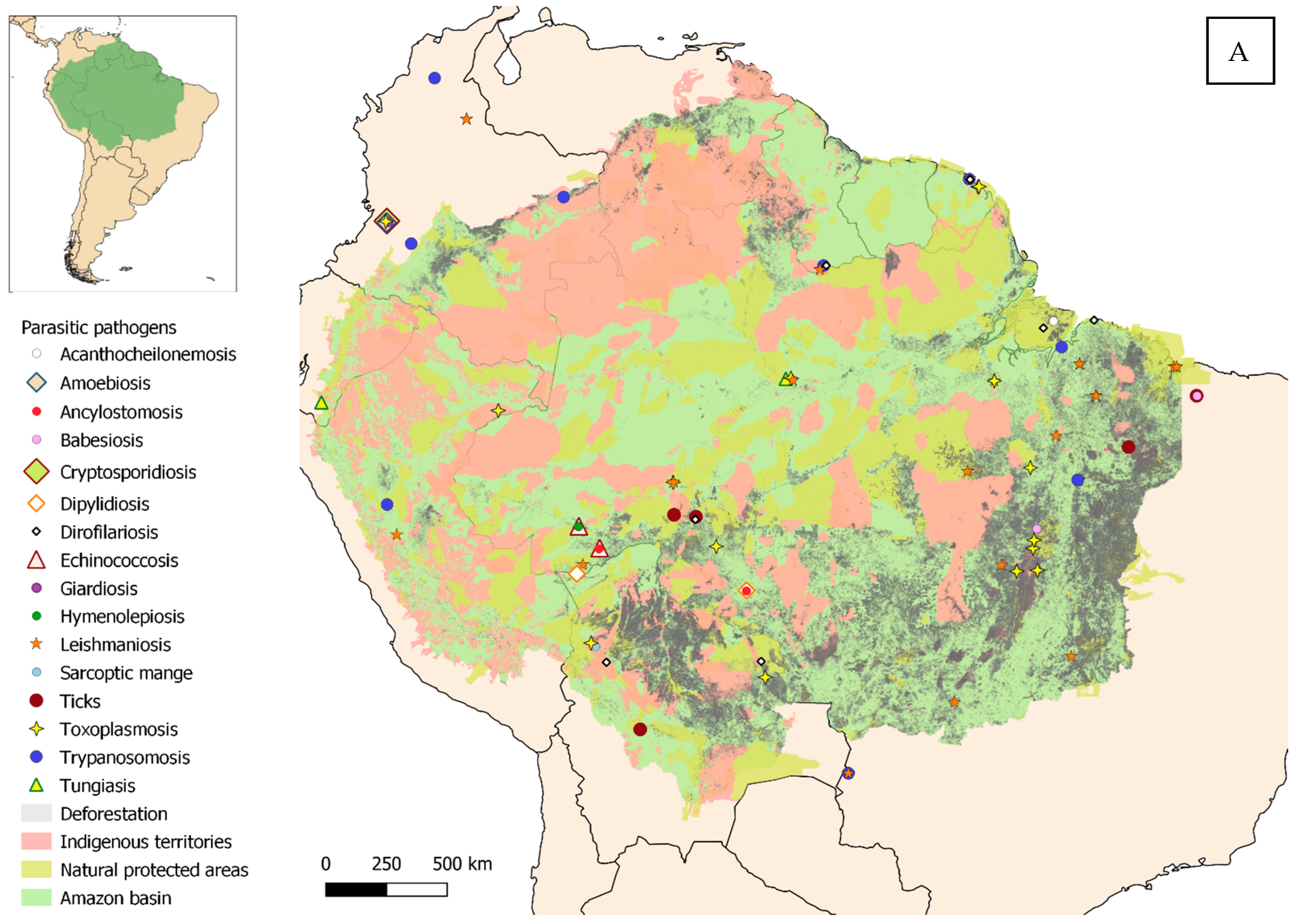
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Brazilian States | |
| (AC) | Acre State |
| (AM) | Amazonas State |
| (MA) | Maranhão State |
| (MS) | Mato Grosso do Sul State |
| (MT) | Mato Grosso State |
| (PA) | Pará State |
| (RO) | Rondônia State |
| (TO) | Tocantins State |
| Diagnostic test abbreviations | |
| AGID II | Agar Gel Immunodiffusion test (Type II) |
| BLASTn | Basic Local Alignment Search Tool–nucleotide |
| CF | Complement Fixation |
| ELISA | Enzyme-linked immunosorbent assay |
| ELISA-ICC | Enzyme-linked immunoassay with infected cultured cells as antigenic matrix |
| FITC | anti-rabies globulin: Fluorescein isothiocyanate-conjugated anti-rabies globulin |
| HRM | High-Resolution Melting Analysis |
| HWAT | Heartworm Antigen Test |
| ICT | Rapid immunochromatographic test |
| IFA | Immunofluorescence assay |
| IFAT | Indirect Fluorescent Antibody Test |
| IHA | Indirect Hemagglutination Test |
| mAT | Microscopic Agglutination Test |
| MAT | Modified Agglutination Test |
| MLMT | Microsatellite Marker Analysis |
| NNN Culture | Novy-Nicolle-MacNeal medium culture |
| nPCR | nested Polymerase Chain Reaction |
| PCR | Polymerase Chain Reaction |
| PCR-RFLP | Polymerase Chain Reaction–Restriction Fragment Length Polymorphism |
| qPCR | quantitative Polymerase Chain Reaction |
| RBT | Rose Bengal Test |
| RFFIT | Rapid Fluorescent Focus Inhibition Test |
| RFLP | Restriction Fragment Length Polymorphism |
| RICT | Rapid Immunochromatographic Test |
| RT-qPCR | Reverse Transcription quantitative Polymerase Chain Reaction |
| SAM | Serum Agglutination Microscopy |
| SAT | Slide agglutination test |
| TESA-blot | Trypomastigote Excreted-Secreted Antigen blot |
Appendix A
References
- RAISG. Amazonía Bajo Presión; RAISG: Sao Paulo, Brazil, 2020. [Google Scholar]
- Gardner, T.A.; Barlow, J.; Sodhi, N.S.; Peres, C.A. A Multi-Region Assessment of Tropical Forest Biodiversity in a Human-Modified World. Biol. Conserv. 2010, 143, 2293–2300. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Pimm, S.L.; Joppa, L.N. Global Patterns of Terrestrial Vertebrate Diversity and Conservation. Proc. Natl. Acad. Sci. USA 2013, 110, E2602–E2610. [Google Scholar] [CrossRef]
- Reid, W.V.; Mooney, H.A.; Cropper, A.; Capistrano, D.; Carpenter, S.R.; Chopra, K.; Dasgupta, P.; Dietz, T.; Duraiappah, A.K.; Hassan, R.; et al. Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005; ISBN 1597260401. [Google Scholar]
- Chivian, E.; Bernstein, A. (Eds.) Sustaining Life How Human Health Depends on Biodiversity; Oxford University Press: New York, NY, USA, 2008. [Google Scholar]
- Suffredini, I.B.; Sader, H.S.; Gonçalves, A.G.; Reis, A.O.; Gales, A.C.; Varella, A.D.; Younes, R.N. Screening of antibacterial extracts from plants native to the Brazilian Amazon Rain Forest and Atlantic Forest. Braz. J. Med. Biol. Res. 2004, 37, 379–384. [Google Scholar] [CrossRef]
- Olival, K.J.; Hosseini, P.R.; Zambrana-Torrelio, C.; Ross, N.; Bogich, T.L.; Daszak, P. Host and Viral Traits Predict Zoonotic Spillover from Mammals. Nature 2017, 546, 646–650. [Google Scholar] [CrossRef]
- Allen, T.; Murray, K.A.; Zambrana-Torrelio, C.; Morse, S.S.; Rondinini, C.; Di Marco, M.; Breit, N.; Olival, K.J.; Daszak, P. Global Hotspots and Correlates of Emerging Zoonotic Diseases. Nat. Commun. 2017, 8, 1124. [Google Scholar] [CrossRef]
- Burkett-Cadena, N.D.; Vittor, A.Y. Deforestation and Vector-Borne Disease: Forest Conversion Favors Important Mosquito Vectors of Human Pathogens. Basic Appl. Ecol. 2018, 26, 101–110. [Google Scholar] [CrossRef]
- MacDonald, A.J.; Mordecai, E.A. Amazon Deforestation Drives Malaria Transmission, and Malaria Burden Reduces Forest Clearing. Proc. Natl. Acad. Sci. USA 2019, 116, 22212–22218. [Google Scholar] [CrossRef]
- Morand, S.; Lajaunie, C. Outbreaks of Vector-Borne and Zoonotic Diseases Are Associated with Changes in Forest Cover and Oil Palm Expansion at Global Scale. Front. Vet. Sci. 2021, 8, 661063. [Google Scholar] [CrossRef]
- Ellwanger, J.H.; Kulmann-Leal, B.; Kaminski, V.L.; Valverde-Villegas, J.M.; Da Veiga, A.B.G.; Spilki, F.R.; Fearnside, P.M.; Caesar, L.; Giatti, L.L.; Wallau, G.L.; et al. Beyond Diversity Loss and Climate Change: Impacts of Amazon Deforestation on Infectious Diseases and Public Health. An. Acad. Bras. Ciências 2020, 92, e20191375. [Google Scholar] [CrossRef]
- Garnett, S.T.; Burgess, N.D.; Fa, J.E.; Fernández-Llamazares, Á.; Molnár, Z.; Robinson, C.J.; Watson, J.E.M.; Zander, K.K.; Austin, B.; Brondizio, E.S.; et al. A Spatial Overview of the Global Importance of Indigenous Lands for Conservation. Nat. Sustain. 2018, 1, 369–374. [Google Scholar] [CrossRef]
- Garza, M.; Abascal Miguel, L. Health Disparities among Indigenous Populations in Latin America: A Scoping Review. Int. J. Equity Health 2025, 24, 119. [Google Scholar] [CrossRef]
- Haslam, P.A.; Ary Tanimoune, N. The Determinants of Social Conflict in the Latin American Mining Sector: New Evidence with Quantitative Data. World Dev. 2016, 78, 401–419. [Google Scholar] [CrossRef]
- Lascano, S.M. Molecular Epidemiology of Trypanosoma (Herpetosoma) Rangeli (Kinetoplastida: Trypanosomatidae) in Ecuador, South America, and Study of the Parasite Cell Invasion Mechanism In Vitro; Ohio University: Columbus, OH, USA, 2009. [Google Scholar]
- Cohen, J.E.; Gurtler, R.E. Modeling Household Transmission of American Trypanosomiasis. Science 2001, 293, 694–698. [Google Scholar] [CrossRef]
- Carme, B.; Demar-Pierre, M. Toxoplasmosis in French Guiana. Atypical (Neo-)Tropical Features of a Cosmopolitan Parasitosis. Med. Trop. 2006, 66, 495–503. [Google Scholar]
- Capdevila-Argüelles, L.; Iglesias-García, A.; Orueta, J.F.; Zilletti, B. Especies Exóticas Invasoras: Diagnóstico y Bases Para La Prevención y El Manejo; Organismo Autónomo Parques Nacionales: Madrid, Spain; Ministerio de Medio Ambiente: Madrid, Spain, 2006; ISBN 9788480146678. [Google Scholar]
- Stahl, P.W. Early Dogs and Endemic South American Canids of the Spanish Main. J. Anthropol. Res. 2013, 69, 515–533. [Google Scholar]
- Mendonça, T.O.; Perin, P.P.; Zanini, D.D.S.; de Souza, H.L.; Pires, P.H.K.; Muniz, I.M.; Tebaldi, J.H.; Mathias, L.A.; Bürger, K.P.; Lux-Hoppe, E.G. Parasitosis in Pet Dogs from Rondônia, Amazon Biome, and Human Perception of Zoonoses. Int. J. Environ. Res. Public Health 2024, 21, 138. [Google Scholar] [CrossRef]
- Mendoza Roldan, J.A.; Otranto, D. Zoonotic Parasites Associated with Predation by Dogs and Cats. Parasites Vectors 2023, 16, 55. [Google Scholar] [CrossRef]
- Salvarani, F.M.; Oliveira, H.G.D.S.; Correa, L.Y.S.; Soares, A.A.L.; Ferreira, B.C. The Importance of Studying Infectious and Parasitic Diseases of Wild Animals in the Amazon Biome with a Focus on One Health. Vet. Sci. 2025, 12, 100. [Google Scholar] [CrossRef]
- Lambin, E.F.; Gesit, H.J. Proximate Causes and Underlying Driving Forces of Tropical Deforestation: Tropical Forests Are Disappearing as the Result of Many Pressures, Both Local and Regional, Acting in Various Combinations in Different Geographical Locations. BioScience 2002, 52, 143–150. [Google Scholar]
- Une Seule Santé Par l’Organisation Mondiale de La Santé. Available online: https://www.who.int/fr/news-room/fact-sheets/detail/one-health (accessed on 10 October 2025).
- Medina-Vogel, G. Ecología de Enfermedades Infecciosas Emergentes y Conservación de Especies Silvestres Ecology of Emerging Infectious Diseases and Wild Species Conservation. Arch. Med. Vet. 2010, 42, 11–24. [Google Scholar] [CrossRef]
- Malzoni Furtado, M.; Domingues de Ramos Filho, J.; Corrêa Scheffer, K.; Coelho, C.J.; Cruz, P.S.; Ikuta, C.Y.; de Almeida Jácomo, A.T.; de Oliveira Porfírio, G.E.; Silveira, L.; Sollmann, R.; et al. Serosurvey for Selected Viral Infections in Freeranging Jaguars (Panthera onca) and Domestic Carnivores in Brazilian Cerrado, Pantanal, and Amazon. J. Wildl. Dis. 2013, 49, 510–521. [Google Scholar] [CrossRef]
- Nava, A.F.D.; Cullen, L.; Sana, D.A.; Nardi, M.S.; Ramos Filho, J.D.; Lima, T.F.; Abreu, K.C.; Ferreira, F. First Evidence of Canine Distemper in Brazilian Free-Ranging Felids. Ecohealth 2008, 5, 513–518. [Google Scholar] [CrossRef]
- Abad-Franch, F.; Grimmer, G.H.; de Paula, V.S.; Figueiredo, L.T.M.; Braga, W.S.M.; Luz, S.L.B. Mayaro Virus Infection in Amazonia: A Multimodel Inference Approach to Risk Factor Assessment. PLoS Negl. Trop. Dis. 2012, 6, e1846. [Google Scholar] [CrossRef]
- Meynard, J.-B.; Flamand, C.; Dupuy, C.; Mahamat, A.; Eltges, F.; Queuche, F.; Renner, J.; Fontanella, J.-M.; Hommel, D.; Dussart, P.; et al. First Human Rabies Case in French Guiana, 2008: Epidemiological Investigation and Control. PLoS Negl. Trop. Dis. 2012, 6, e1537. [Google Scholar] [CrossRef]
- Gaspari, M.M.F. Estudo Epidemiológico de Patógenos Circulantes Nas Populações de Onça-Pintada e Animais Domésticos Em Áreas Preservadas de Três Biomas Brasileiros: Cerrado, Pantanal e Amazônia; Universidade de São Paulo: São Paulo, Brazil, 2010. [Google Scholar]
- Bronson, E.; Emmons, L.H.; Murray, S.; Dubovi, E.J.; Deem, S.L. Serosurvey of Pathogens in Domestic Dogs on the Border of Noël Kempff Mercado National Park, Bolivia. J. Zoo Wildl. Med. 2008, 39, 28–36. [Google Scholar] [CrossRef]
- Widdowson, M.-A.; Morales, G.J.; Chaves, S.; McGrane, J. Epidemiology of Urban Canine Rabies, Santa Cruz, Bolivia, 1972–1997. Emerg. Infect. Dis. 2002, 8, 458–461. [Google Scholar] [CrossRef]
- Zambrano-Mila, M.S.; Freire-Paspuel, B.; Orlando, S.A.; Garcia-Bereguiain, M.A. SARS-CoV-2 Infection in Free Roaming Dogs from the Amazonian Jungle. One Health 2022, 14, 100387. [Google Scholar] [CrossRef]
- Mesquita, V.A.; Talhari, S.; Leturiondo, A.L.; de Souza, G.C.; de Brito, E.M.; de Andrade, S.L.; Fernandes, D.C.D.L.; Frota, M.Z.M.; Cruz, R.C.D.S.; Guimarães, J.D.A.R.; et al. Zoonotic Sporotrichosis Outbreak: Emerging Public Health Threat in the Amazon State, Brazil. PLoS Negl. Trop. Dis. 2024, 18, e0012328. [Google Scholar] [CrossRef]
- Milstein, M.S.; Shaffer, C.A.; Suse, P.; Marawanaru, A.; Heinrich, D.A.; Larsen, P.A.; Wolf, T.M. A Mixed-Methods Approach to Understanding Domestic Dog Health and Disease Transmission Risk in an Indigenous Reserve in Guyana, South America. PLoS Negl. Trop. Dis. 2022, 16, e0010469. [Google Scholar] [CrossRef]
- Furtado, M.M.; Gennari, S.M.; Ikuta, C.Y.; Jácomo, A.T.D.A.; de Morais, Z.M.; Pena, H.F.D.J.; Porfírio, G.E.D.O.; Silveira, L.; Sollmann, R.; de Souza, G.O.; et al. Serosurvey of Smooth Brucella, Leptospira Spp. and Toxoplasma Gondii in Free-Ranging Jaguars (Panthera onca) and Domestic Animals from Brazil. PLoS ONE 2015, 10, e0143816. [Google Scholar] [CrossRef]
- Rodriguez-Pazmiño, A.S.; Brito, C.M.; Salas-Rueda, M.; Orlando, S.A.; Garcia-Bereguiain, M.A. A First Insight into Seropositivity and Risk Factors for Brucella spp. and Coxiella burnetii in Free-Roaming Dogs in Ecuador. One Health 2024, 19, 100909. [Google Scholar] [CrossRef]
- Gardon, J.; Héraud, J.; Laventure, S.; Ladam, A.; Capot, P.; Fouquet, E.; Favre, J.; Weber, S.; Hommel, D.; Hulin, A.; et al. Suburban Transmission of Q Fever in French Guiana: Evidence of a Wild Reservoir. J. Infect. Dis. 2001, 184, 278–284. [Google Scholar] [CrossRef]
- Costa, A.P.D.; Costa, F.B.; Labruna, M.B.; Silveira, I.; Moraes-Filho, J.; Soares, J.F.; Spolidorio, M.G.; Guerra, R.d.M.S.N.d.C. A Serological and Molecular Survey of Babesia Vogeli, Ehrlichia Canis and Rickettsia Spp. among Dogs in the State of Maranhão, Northeastern Brazil. Rev. Bras. Parasitol. Veterinária 2015, 24, 28–35. [Google Scholar] [CrossRef]
- Guzmán, D.A.; Diaz, E.; Sáenz, C.; Álvarez, H.; Cueva, R.; Zapata-Ríos, G.; Prado-Vivar, B.; Falconí, M.; Pearson, T.; Barragan, V. Domestic Dogs in Indigenous Amazonian Communities: Key Players in Leptospira Cycling and Transmission? PLoS Negl. Trop. Dis. 2024, 18, e0011671. [Google Scholar] [CrossRef]
- Fiorello, C.V.; Deem, S.L.; Gompper, M.E.; Dubovi, E.J. Seroprevalence of Pathogens in Domestic Carnivores on the Border of Madidi National Park, Bolivia. Anim. Conserv. 2004, 7, 45–54. [Google Scholar] [CrossRef]
- Costa, F.B. Soroepidemiologia e Epidemiologia Molecular Das Infecções Por Rickettsia Spp Em Cães e Carrapatos de Ambientes Urbano e Rural Do Estado Do Maranhão; Universidade de São Paulo: São Paulo, Brazil, 2014. [Google Scholar]
- Tomassone, L.; Conte, V.; Parrilla, G.; De Meneghi, D. Rickettsia Infection in Dogs and Rickettsia Parkeri in Amblyomma Tigrinum Ticks, Cochabamba Department, Bolivia. Vector-Borne Zoonotic Dis. 2010, 10, 953–958. [Google Scholar] [CrossRef]
- Forshey, B.M.; Stewart, A.; Morrison, A.C.; Gálvez, H.; Rocha, C.; Astete, H.; Eza, D.; Chen, H.-W.; Chao, C.-C.; Montgomery, J.M.; et al. Epidemiology of Spotted Fever Group and Typhus Group Rickettsial Infection in the Amazon Basin of Peru. Am. Soc. Trop. Med. Hyg. 2010, 82, 683–690. [Google Scholar] [CrossRef]
- Costa, I.N.D.; Garcia, M.V.; Santos, V.P.D.; Costa, N.V.C.; Carioca, A.L.P.M.; Andreotti, R.; Medeiros, J.F.; Aguirre, A.D.A.R. Simultaneous Infestations in Dogs by Different Tick Species (Acari: Ixodidae) in Two Areas of the Western Brazilian Amazon. Rev. Bras. Parasitol. Veterinária 2025, 34, e020324. [Google Scholar] [CrossRef]
- de Argôlo, E.G.G.; Reis, T.; Fontes, D.A.T.; Gonçalves, E.C.; Giese, E.G.; Melo, F.T.D.V.; dos Santos, J.N.; Furtado, A.P. Canine Filariasis in the Amazon: Species Diversity and Epidemiology of These Emergent and Neglected Zoonoses. PLoS ONE 2018, 13, e0200419. [Google Scholar] [CrossRef]
- Dias-Correia, T.P.; Neves, L.B.D.; Bittencourt-Oliveira, F.; Giglio, G.C.B.; Pereira, T.C.; Almeida, F.B.D.; Rodrigues-Silva, R. Diversity of Helminths with Zoonotic Potential and Molecular Characterization of Toxocara Canis Infecting Domestic Dogs from Locations of Amazon and Atlantic Forest Brazilian Biomes. Rev. Bras. Parasitol. Veterinária 2023, 32, e012723. [Google Scholar] [CrossRef]
- Potes-Morales, C.; Crespo-Ortiz, M.D.P. Molecular Diagnosis of Intestinal Protozoa in Young Adults and Their Pets in Colombia, South America. PLoS ONE 2023, 18, e0283824. [Google Scholar] [CrossRef]
- Morais, A.N.; Sousa, M.G.; Meireles, L.R.; Kesper, N., Jr.; Umezawa, E.S. Canine Visceral Leishmaniasis and Chagas Disease among Dogs in Araguaína, Tocantins. Rev. Bras. Parasitol. Veterinária 2013, 22, 225–229. [Google Scholar] [CrossRef]
- Soares, H.S.; Camargo, L.M.A.; Gennari, S.M.; Labruna, M.B. Survey of Canine Tick-Borne Diseases in Lábrea, Brazilian Amazon: ‘Accidental’ Findings of Dirofilaria Immitis Infection. Rev. Bras. Parasitol. Veterinária 2014, 23, 473–480. [Google Scholar] [CrossRef]
- Barbosa, U.C.; Nava, A.F.D.; Ferreira Neto, J.V.; Dias, C.A.; Silva, V.C.D.; Mesquita, H.G.D.; Sampaio, R.T.D.; Barros, W.G.; Farias, E.D.S.; Silva, T.R.R.D.; et al. Dirofilaria Immitis Is Endemic in Rural Areas of the Brazilian Amazonas State Capital, Manaus. Rev. Bras. Parasitol. Veterinária 2023, 32, e000223. [Google Scholar] [CrossRef]
- Moreira, H.R.; Madeira, E.A.O.; Cunha, D.N.L.; Scofield, A.; Góes-Cavalcante, G.; Abel, I.; Guimarães, R.J.P.S.; Fernandes, J.I. Dirofilaria Immitis Infection in Dogs in Algodoal Island, Brazilian Amazon. Pesqui. Veterinária Bras. 2019, 39, 510–515. [Google Scholar] [CrossRef]
- Ogawa, G.M.; Cruz, E.N.D.; Cunha, P.N.A.; Camargo, L.M.A. Canine Heartworm Disease in Porto Velho: First Record, Distribution Map and Occurrence of Positive Mosquitoes. Rev. Bras. Parasitol. Veterinária 2013, 22, 559–564. [Google Scholar] [CrossRef]
- Gomes Zanfagnini, L.; Carvalho Bento, G.K.; Fernandes Nunes da Silva Malavazi, P.; Figueiredo Souza, S.; Duarte Pacheco, A. Primeira Descrição de Dirofilariose Canina Alóctone Em Rio Branco, XAcre: Relato de Caso. Rev. Med. Vet. 2024, 48, e1497. [Google Scholar] [CrossRef]
- Laidoudi, Y.; Marié, J.-L.; Tahir, D.; Watier-Grillot, S.; Mediannikov, O.; Davoust, B. Detection of Canine Vector-Borne Filariasis and Their Wolbachia Endosymbionts in French Guiana. Microorganisms 2020, 8, 770. [Google Scholar] [CrossRef] [PubMed]
- das Neves, L.B.; Teixeira, P.E.F.; Silva, S.; de Oliveira, F.B.; Garcia, D.D.; de Almeida, F.B.; Rodrigues-Silva, R.; Machado-Silva, J.R. First Molecular Identification of Echinococcus vogeli and Echinococcus granulosus (Sensu stricto) G1 Revealed in Feces of Domestic Dogs (Canis familiaris) from Acre, Brazil. Parasites Vectors 2017, 10, 28. [Google Scholar] [CrossRef]
- Carneiro, L.A.; Vasconcelos dos Santos, T.; Lima, L.V.D.R.; Ramos, P.K.S.; Campos, M.B.; Silveira, F.T. First Report on Feline Leishmaniasis Caused by Leishmania (Leishmania) Amazonensis in Amazonian Brazil. Vet. Parasitol. Reg. Stud. Rep. 2020, 19, 100360. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.C.O. Investigação Do Perfil Sorológico e Detecção Do DNA de Leishmania Spp. Em Cães de Áreas Endêmicas Para Leishmaniose Tegumentar Americana No Estado Do Pará, Brasil; Universidade Federal do Pará: Belém, Brazil, 2012. [Google Scholar]
- Castillo-Castañeda, A.C.; Patiño, L.H.; Zuñiga, M.F.; Cantillo-Barraza, O.; Ayala, M.S.; Segura, M.; Bautista, J.; Urbano, P.; Jaimes-Dueñez, J.; Ramírez, J.D. An Overview of the Trypanosomatid (Kinetoplastida: Trypanosomatidae) Parasites Infecting Several Mammal Species in Colombia. Parasites Vectors 2022, 15, 471. [Google Scholar] [CrossRef]
- Santos, F.J.A.; Nascimento, L.C.S.; Silva, W.B.; Oliveira, L.P.; Santos, W.S.; Aguiar, D.C.F.; Garcez, L.M. First Report of Canine Infection by Leishmania (Viannia) Guyanensis in the Brazilian Amazon. Int. J. Environ. Res. Public Health 2020, 17, 8488. [Google Scholar] [CrossRef]
- Cupolillo, E.; Cavalcanti, A.S.; Ferreira, G.E.M.; Boité, M.C.; Morgado, F.N.; Porrozzi, R. Occurrence of Multiple Genotype Infection Caused by Leishmania Infantum in Naturally Infected Dogs. PLoS Negl. Trop. Dis. 2020, 14, e0007986. [Google Scholar] [CrossRef]
- Basano, S.D.A.; Tarso, P.; Soares, H.S.; Costa, A.P.; Marcili, A.; Labruna, M.B.; Dias, R.A.; Camargo, L.M.A.; Gennari, S.M. Toxoplasma Gondii, Neospora Caninum and Leishmania Amazonensis Antibodies in Domestic Dogs in the Western Brazilian Amazon Region. Braz. J. Vet. Res. Anim. Sci. 2016, 53, 1. [Google Scholar] [CrossRef]
- Nascimento de Campos, H.G.; Gennari, S.M.; da Silva, R.E.; Soares, H.S.; Costa, J.D.O.J.; de Azevedo, S.S.; Marcili, A. Molecular and Serological Detection of Leishmania Infantum (Trypanosomatida: Trypanosomatidae) in Domestic Dogs in Manaus City, Amazonas, Brazil. J. Med. Entomol. 2024, 61, 1519–1523. [Google Scholar] [CrossRef]
- Campolongo, C.; Silva, R.E.D.; Azevedo, R.C.D.F.E.; Pesenato, I.P.; Carioca, A.C.F.; Alves, B.F.; Castelli, G.S.N.; Onofrio, V.C.; Marcili, A. Prevalence of Leishmania Infantum in Dogs from Deforested Areas of the Amazon Biome. Vector-Borne Zoonotic Dis. 2022, 22, 108–113. [Google Scholar] [CrossRef]
- Lima, J.T.R.D.; Gennari, S.M.; Soares, H.S.; Minervino, A.H.H.; Malheiros, A.F.; Marques, F.S.; Laurenti, M.D.; Machado, R.Z.; Marcili, A.; Labruna, M.B.; et al. Serodiagnosis of Visceral and Cutaneous Leishmaniasis in Human and Canine Populations Living in Indigenous Reserves in the Brazilian Amazon Region. Rev. Soc. Bras. Med. Trop. 2017, 50, 61–66. [Google Scholar] [CrossRef]
- Rocha, A.V.V.O.; Moreno, B.F.S.; Cabral, A.D.; Louzeiro, N.M.; Miranda, L.M.; Santos, V.M.B.D.; Costa, F.B.; Nogueira, R.D.M.S.; Marcili, A.; Sperança, M.A.; et al. Diagnosis and Epidemiology of Leishmania Infantum in Domestic Cats in an Endemic Area of the Amazon Region, Brazil. Vet. Parasitol. 2019, 273, 80–85. [Google Scholar] [CrossRef]
- Valadas, S.; Minervino, A.H.H.; Lima, V.M.F.; Soares, R.M.; Ortolani, E.L.; Gennari, S.M. Occurrence of Antibodies Anti-Neospora Caninum, Anti-Toxoplasma Gondii, and Anti-Leishmania Chagasi in Serum of Dogs from Pará State, Amazon, Brazil. Parasitol. Res. 2010, 107, 453–457. [Google Scholar] [CrossRef]
- de Oliveira Porfirio, G.E.; Santos, F.M.; de Macedo, G.C.; Barreto, W.T.G.; Campos, J.B.V.; Meyers, A.C.; André, M.R.; Perles, L.; de Oliveira, C.E.; Xavier, S.C.D.C.; et al. Maintenance of Trypanosoma Cruzi, T. Evansi and Leishmania Spp. by Domestic Dogs and Wild Mammals in a Rural Settlement in Brazil-Bolivian Border. Int. J. Parasitol. Parasites Wildl. 2018, 7, 398–404. [Google Scholar] [CrossRef]
- Brilhante, A.F.; Lima, L.; Zampieri, R.A.; Nunes, V.L.B.; Dorval, M.E.C.; Malavazi, P.F.N.D.S.; Melchior, L.A.K.; Ishikawa, E.A.Y.; Cardoso, C.D.O.; Floeter-Winter, L.M.; et al. Leishmania (Viannia) Braziliensis Type 2 as Probable Etiological Agent of Canine Cutaneous Leishmaniasis in Brazilian Amazon. PLoS ONE 2019, 14, e0216291. [Google Scholar] [CrossRef] [PubMed]
- Fornazari, F.; Sevá, A.D.P.; Oliveira, K.M.M.; Assunçao, P.C.F.; Guimaraes, V.Y. Exposure of Dogs and Wild Carnivores to Canine Distemper Virus, Canine Parvovirus, Leishmania Infantum, and Toxoplasma Gondii in the Xingu River Basin, Brazilian Amazon: Prevalence, Spatial Distribution, and Association with Land Cover Types. Acta Amazon. 2023, 53, 325–335. [Google Scholar] [CrossRef]
- Moreno, B.F.S. Avaliação Sorológica Para Detecção de Anticorpos Anti-Leishmania Sp. Em Gatos de Uma Área Endêmica Para Leishmaniose Visceral; Universidad Estatal de Maranhão: São Luís, Brazil, 2017. [Google Scholar]
- Medkour, H.; Davoust, B.; Dulieu, F.; Maurizi, L.; Lamour, T.; Marié, J.-L.; Mediannikov, O. Potential Animal Reservoirs (Dogs and Bats) of Human Visceral Leishmaniasis Due to Leishmania Infantum in French Guiana. PLoS Negl. Trop. Dis. 2019, 13, e0007456. [Google Scholar] [CrossRef]
- Rotureau, B.; Ravel, C.; Aznar, C.; Carme, B.; Dedet, J.-P. First Report of Leishmania Infantum in French Guiana: Canine Visceral Leishmaniasis Imported from the Old World. J. Clin. Microbiol. 2006, 44, 1120–1122. [Google Scholar] [CrossRef]
- Reithinger, R. The Epidemiology and Control of Canine Leishmaniasis in Peru and Brazil; University of London: London, UK, 2004. [Google Scholar]
- Ziemniczak, H.M.; Maia, M.O.; Maia, M.O.; Ferreira, E.; Vieira, N.T.; Saturnino, K.C.; Bresciani, K.D.S.; Gomes, A.A.D.; Pacheco, R.D.C.; Santos-Doni, T.R. High Seroprevalence of Toxoplasma Gondii and Neospora Spp. in Stray Dogs from Rolim de Moura, Rondônia State, Western Brazilian Amazon. Semin. Cienc. Agrar. 2021, 42, 3535–3542. [Google Scholar] [CrossRef]
- Minervino, A.H.H.; Cassinelli, A.B.M.; de Lima, J.T.R.; Soares, H.S.; Malheiros, A.F.; Marcili, A.; Gennari, S.M. Prevalence of Anti-Neospora Caninum and Anti-Toxoplasma Gondii Antibodies in Dogs from Two Different Indigenous Communities in the Brazilian Amazon Region. J. Parasitol. 2012, 98, 1276–1278. [Google Scholar] [CrossRef]
- Campos, H.G.N.D.; Soares, H.S.; Azevedo, S.S.D.; Gennari, S.M. Occurrence of Toxoplasma Gondii and Neospora Caninum Antibodies and Risk Factors in Domiciliated Dogs of Manaus, Amazonas, Brazil. Rev. Bras. Parasitol. Veterinária 2022, 31, e020321. [Google Scholar] [CrossRef]
- Mercier, A.; Ajzenberg, D.; Devillard, S.; Demar, M.P.; de Thoisy, B.; Bonnabau, H.; Collinet, F.; Boukhari, R.; Blanchet, D.; Simon, S.; et al. Human Impact on Genetic Diversity of Toxoplasma Gondii: Example of the Anthropized Environment from French Guiana. Infect. Genet. Evol. 2011, 11, 1378–1387. [Google Scholar] [CrossRef]
- Tahir, D.; Davoust, B.; Heu, K.; Lamour, T.; Demar, M.; Marié, J.-L.; Blanchet, D. Molecular and Serological Investigation of Trypanosoma Cruzi Infection in Dogs in French Guiana. Vet. Parasitol. Reg. Stud. Rep. 2018, 12, 106–109. [Google Scholar] [CrossRef]
- Garces Quintero, E.Y. Caracterización Ecoepidemiológica de La Transmisión Peridoméstica de Trypanosoma Cruzi En El Municipio de Talaigua Nuevo; Departamento de Bolívar, Costa Caribe Colombiana, Universidad de Antioquía: Medellín, Colombia, 2012. [Google Scholar]
- Oliveira, M.J.D.; Anadão, L.D.S.; Souza, F.D.A.; Santos, R.S.D.; Fermino, B.R.; Rodrigues, C.M.F.; Carvalho, J.S.; Gasparotto, P.H.G.; Silva, H.S.D.; Santos, F.G.D.A. Infecção Natural Por Trypanosoma Evansi Em Cão No Estado de Rondônia, Brasil: Relato de Caso. Cad. Pedagógico 2024, 21, e4369. [Google Scholar] [CrossRef]
- Cantillo-Barraza, O.; Solis, C.; Zamora, A.; Herazo, R.; Osorio, M.I.; Garcés, E.; Xavier, S.; Mejía-Jaramillo, A.M.; Triana-Chávez, O. Enzootic Trypanosoma Cruzi Infection by Rhodnius Prolixus Shows Transmission to Humans and Dogs in Vichada, Colombia. Front. Cell. Infect. Microbiol. 2022, 12, 999082. [Google Scholar] [CrossRef]
- Roque, A.L.R.; Xavier, S.C.C.; Gerhardt, M.; Silva, M.F.O.; Lima, V.S.; D’Andrea, P.S.; Jansen, A.M. Trypanosoma Cruzi among Wild and Domestic Mammals in Different Areas of the Abaetetuba Municipality (Pará State, Brazil), an Endemic Chagas Disease Transmission Area. Vet. Parasitol. 2013, 193, 71–77. [Google Scholar] [CrossRef]
- Villena, F.E.; Puicón, V.H.; López, A.M.; Rivera, K.; Pannebaker, D.; Valdivia, H.O.; Arévalo, H. Parasitological and Molecular Detection of Trypanosoma Evansi in a Dog from Tocache, San Martin, Peru. Vet. Parasitol. Reg. Stud. Rep. 2023, 42, 100895. [Google Scholar] [CrossRef]
- Corrêa, R.D.S.; Araujo, J.A.S.D.; Leite, J.M.B.; Silva Filho, L.E.D.; da Silva, N.M. Tungiasis in Dogs Residing in the Community Nossa Senhora in the Livramento, Sustainable Development Reserve Tupé, Amazonas. Rev. Bras. Hig. E Sanidade Anim. 2014, 8, 79–87. [Google Scholar] [CrossRef]
- Chaguay Villamar, K.M.; Hernández Copello, V.M.; Toro-Valdivieso, C.; Rivera Gomez-Barris, B.M. First Report of Tunga Trimamillata Infection in a Dog. BMC Vet. Res. 2025, 21, 100. [Google Scholar] [CrossRef] [PubMed]
- Jansen, A.M.; Xavier, S.C.D.C.; Roque, A.L.R. Trypanosoma Cruzi Transmission in the Wild and Its Most Important Reservoir Hosts in Brazil. Parasites Vectors 2018, 11, 502. [Google Scholar] [CrossRef] [PubMed]
- Herrera, L. Una Revisión Sobre Reservorios de Trypanosoma (Schizotrypanum) Cruzi (Chagas, 1909), Agente Etiológico de La Enfermedad de Chagas. Bol. Malariol. Salud Ambient. 2010, 50, 3–15. [Google Scholar]
- Herrera, L.; Urdaneta-Morales, S. Didelphis marsupialis: A Primary Reservoir of Trypanosoma Cruzi in Urban Areas of Caracas, Venezuela. Ann. Trop. Med. Parasitol. 1992, 86, 607–612. [Google Scholar] [CrossRef]
- Yeo, M.; Acosta, N.; Llewellyn, M.; Sánchez, H.; Adamson, S.; Miles, G.A.J.; López, E.; González, N.; Patterson, J.S.; Gaunt, M.W.; et al. Origins of Chagas Disease: Didelphis Species Are Natural Hosts of Trypanosoma Cruzi I and Armadillos Hosts of Trypanosoma Cruzi II, Including Hybrids. Int. J. Parasitol. 2005, 35, 225–233. [Google Scholar] [CrossRef]
- Gürtler, R.E.; Cecere, M.C.; Lauricella, M.A.; Cardinal, M.V.; Kitron, U.; Cohen, J.E. Domestic Dogs and Cats as Sources of Trypanosoma Cruzi Infection in Rural Northwestern Argentina. Parasitology 2007, 134, 69–82. [Google Scholar] [CrossRef]
- Dantas-Torres, F. Canine Leishmaniosis in South America. Parasites Vectors 2009, 2, S1. [Google Scholar] [CrossRef]
- Dedet, J.-P. Cutaneous Leishmaniasis in French Guiana: A Review. Am. J. Trop. Med. Hyg. 1990, 43, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Massey, A.L.; Ferreira da Silva, D.J.; Vieira, C.J.D.S.P.; Allen, J.M.; Canale, G.R.; Bernardo, C.S.S.; de Morais Bronzoni, R.V.; Peres, C.A.; Levi, T. Using IDNA to Determine Impacts of Amazonian Deforestation on Leishmania Hosts, Vectors, and Their Interactions. PLoS Negl. Trop. Dis. 2025, 19, e0012925. [Google Scholar] [CrossRef]
- Barragan, V.; Chiriboga, J.; Miller, E.; Olivas, S.; Birdsell, D.; Hepp, C.; Hornstra, H.; Schupp, J.M.; Morales, M.; Gonzalez, M.; et al. High Leptospira Diversity in Animals and Humans Complicates the Search for Common Reservoirs of Human Disease in Rural Ecuador. PLoS Negl. Trop. Dis. 2016, 10, e0004990. [Google Scholar] [CrossRef]
- Giraldo de León, G.; Orrego Uribe, A.; Betancurth, A.M. Los Roedores Como Reservorios de Leptospiras En Planteles Porcinos de La Zona Central Cafetera de Colombia. Arch. Med. Vet. 2002, 34, 69–78. [Google Scholar] [CrossRef]
- Ricardo, T.; Monje, L.D.; Landolt, N.; Chiani, Y.T.; Schmeling, M.F.; Beldoménico, P.M.; Vanasco, N.B.; Previtali, M.A. Primer Informe de Leptospira interrogans En El Roedor Sigmodontino Scapteromys aquaticus. Rev. Panam. Salud Pública 2018, 42, e83. [Google Scholar] [CrossRef] [PubMed]
- Díaz, E.A.; Arroyo, G.; Sáenz, C.; Mena, L.; Barragán, V. Leptospirosis in Horses: Sentinels for a Neglected Zoonosis? A Systematic Review. Vet. World 2023, 16, 2110–2119. [Google Scholar] [CrossRef]
- Orlando, S.A.; Perez, A.; Sanchez, E.; de la Cruz, C.; Rugel, O.; Garcia-Bereguiain, M.A. High Seroprevalence of Anti-Leptospira Spp. Antibodies in Domestic and Wild Mammals from a Mixed Use Rescue Center in Ecuador: Lessons for “One Health” Based Conservation Strategies. One Health 2020, 10, 100140. [Google Scholar] [CrossRef]
- Hartmann, K.; Egberink, H.; Pennisi, M.G.; Lloret, A.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Frymus, T.; Gruffydd-Jones, T.; Hosie, M.J.; et al. Leptospira Species Infection in Cats. J. Feline Med. Surg. 2013, 15, 576–581. [Google Scholar] [CrossRef]
- Elmore, S.A.; Jones, J.L.; Conrad, P.A.; Patton, S.; Lindsay, D.S.; Dubey, J.P. Toxoplasma Gondii: Epidemiology, Feline Clinical Aspects, and Prevention. Trends Parasitol. 2010, 26, 190–196. [Google Scholar] [CrossRef]
- Cañon-Franco, W.A.; Araújo, F.A.P.D.; Gennari, S.M. Toxoplasma Gondii Em Pequenos Felinos Silvestres Neotropicais. Braz. J. Vet. Res. Anim. Sci. 2013, 50, 50. [Google Scholar] [CrossRef]
- Demar, M.; Ajzenberg, D.; Maubon, D.; Djossou, F.; Panchoe, D.; Punwasi, W.; Valery, N.; Peneau, C.; Daigre, J.-L.; Aznar, C.; et al. Fatal Outbreak of Human Toxoplasmosis along the Maroni River: Epidemiological, Clinical, and Parasitological Aspects. Clin. Infect. Dis. 2007, 45, e88–e95. [Google Scholar] [CrossRef]
- Gómez-Marín, J.E.; de-la-Torre, A.; Barrios, P.; Cardona, N.; Álvarez, C.; Herrera, C. Toxoplasmosis in Military Personnel Involved in Jungle Operations. Acta Trop. 2012, 122, 46–51. [Google Scholar] [CrossRef]
- do Carmo, E.L.; Póvoa, M.M.; Monteiro, N.S.; Marinho, R.R.; Nascimento, J.M.; Freitas, S.N.; Bichara, C.N.C. Surto de Toxoplasmose Humana No Distrito de Monte Dourado, Município de Almeirim, Pará, Brasil. Rev. Pan-Amaz. Saúde 2010, 1. [Google Scholar] [CrossRef]
- Souza, N.F.; Benigno, R.N.M.; Figueiredo, M.; Salim, S.K.; Silva, D.; Gonçalves, R.; Peixoto, P.C.; Serra-Freire, N.M. Prevalência de Dirofilaria Immitis Em Cães No Município de Belém/PA, Com Base Na Microfilaremia. Rev. Bras. Parasitol. Veterinária 1997, 6, 83–86. [Google Scholar]
- Vieira, C.; Vélez, I.D.; Montoya, M.N.; Agudelo, S.; Genchi, C.; Simón, F. Dirofilaria Immitis in Tikuna Indians and Their Dogs in the Colombian Amazon. Ann. Trop. Med. Parasitol. 1998, 92, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Portela, C.S.; Mendes de Araújo, C.P.; Moura Sousa, P.; Gomes Simão, C.L.; Silva de Oliveira, J.C.; Crainey, J.L. Filarial Disease in the Brazilian Amazon and Emerging Opportunities for Treatment and Control. Curr. Res. Parasitol. Vector-Borne Dis. 2024, 5, 100168. [Google Scholar] [CrossRef]
- Moroni, B.; Rossi, L.; Bernigaud, C.; Guillot, J. Zoonotic Episodes of Scabies: A Global Overview. Pathogens 2022, 11, 213. [Google Scholar] [CrossRef]
- Rudd, J.L.; Clifford, D.L.; Cypher, B.L.; Hull, J.M.; Jane Riner, A.; Foley, J.E. Molecular Epidemiology of a Fatal Sarcoptic Mange Epidemic in Endangered San Joaquin Kit Foxes (Vulpes Macrotis Mutica). Parasites Vectors 2020, 13, 456. [Google Scholar] [CrossRef] [PubMed]
- Wallén, J.; Erlandsson, R.; Larm, M.; Meijer, T.; Norén, K.; Angerbjörn, A. Consequences of Repeated Sarcoptic Mange Outbreaks in an Endangered Mammal Population. Ecography 2024, 2024, e07291. [Google Scholar] [CrossRef]
- Unterköfler, M.S.; Schausberger, M.; Deutz, A.; Gressmann, G.; Kübber-Heiss, A.; Ferroglio, E.; Joachim, A. Sarcoptic Mange in Wild Ungulates in the European Alps—A Systematic Review. Int. J. Parasitol. Parasites Wildl. 2023, 22, 121–125. [Google Scholar] [CrossRef]
- Díaz Luque, J.A.; Müller, H.; González, L.; Berkunsky, I. Clinical Signs Suggestive of Mange Infestation in a Free-Ranging Maned Wolf (Chrysocyon Brachyurus) in the Moxos Savannahs of Beni, Bolivia. Mastozool. Neotrop. 2014, 21, 135–138. [Google Scholar]
- Fiori, F.; de Paula, R.C.; Boulhosa, R.L.P.; Dias, R.A. Clinical Evaluation of Sarcoptic Mange (Sarcoptes scabiei) in Maned Wolves (Chrysocyon brachyurus). Vet. Res. Commun. 2025, 49, 202. [Google Scholar] [CrossRef] [PubMed]
- Fiori, F.; de Paula, R.C.; Navas-Suárez, P.E.; Boulhosa, R.L.P.; Dias, R.A. The Sarcoptic Mange in Maned Wolf (Chrysocyon brachyurus): Mapping an Emerging Disease in the Largest South American Canid. Pathogens 2023, 12, 830. [Google Scholar] [CrossRef] [PubMed]
- Teodoro, T.G.W.; Lima, P.A.; Stehling, P.C.; Oliveira Junior, I.M.; Varaschin, M.S.; Wouters, F.; Wouters, A.T.B. Sarcoptic Mange (Sarcoptes scabiei) in Wild Canids (Cerdocyon thous). Pesqui. Veterinária Bras. 2018, 38, 1444–1448. [Google Scholar] [CrossRef]
- Silva Paranhos, L.; Alves Soares Vaz de Castro, P.; Rotondo de Araújo, G.; Do Carmo Magalhães, F.; Trindade Bezerra, J.M. Prevalence of Tungiasis in Humans in Brazil and in Its Federative Units: A Systematic Review. Rev. Patol. Trop./J. Trop. Pathol. 2022, 51, 31–50. [Google Scholar] [CrossRef]
- Veraldi, S.; Valsecchi, M. Imported Tungiasis: A Report of 19 Cases and Review of the Literature. Int. J. Dermatol. 2007, 46, 1061–1066. [Google Scholar] [CrossRef]
- Buhariwalla, F.; Cann, B.; Marrie, T.J. A Dog-Related Outbreak of Q Fever. Clin. Infect. Dis. 1996, 23, 753–755. [Google Scholar] [CrossRef]
- Pinsky, R.L.; Fishbein, D.B.; Greene, C.R.; Gensheimer, K.F. An Outbreak of Cat-Associated Q Fever in the United States. J. Infect. Dis. 1991, 164, 202–204. [Google Scholar] [CrossRef]
- Celina, S.S.; Cerný, J. Coxiella burnetii in Ticks, Livestock, Pets and Wildlife: A Mini-Review. Front. Vet. Sci. 2022, 9, 1068129. [Google Scholar] [CrossRef]
- Chacón-Díaz, C.; Altamirano-Silva, P.; González-Espinoza, G.; Medina, M.-C.; Alfaro-Alarcón, A.; Bouza-Mora, L.; Jiménez-Rojas, C.; Wong, M.; Barquero-Calvo, E.; Rojas, N.; et al. Brucella Canis Is an Intracellular Pathogen That Induces a Lower Proinflammatory Response than Smooth Zoonotic Counterparts. Infect. Immun. 2015, 83, 4861–4870. [Google Scholar] [CrossRef]
- Djokic, V.; Freddi, L.; de Massis, F.; Lahti, E.; van den Esker, M.H.; Whatmore, A.; Haughey, A.; Ferreira, A.C.; Garofolo, G.; Melzer, F.; et al. The Emergence of Brucella Canis as a Public Health Threat in Europe: What We Know and What We Need to Learn. Emerg. Microbes Infect. 2023, 12, 2249126. [Google Scholar] [CrossRef]
- Lucero, N.E.; Escobar, G.I.; Ayala, S.M.; Jacob, N. Diagnosis of Human Brucellosis Caused by Brucella Canis. J. Med. Microbiol. 2005, 54, 457–461. [Google Scholar] [CrossRef]
- Davis, G.H.N.G. Estudos Epidemiológicos Sobre Arbovírus Em Populações Rurais e Urbanas Do Estado Do Amazonas; Universidade Federal do Amazonas: Manaus, Brazil, 2009. [Google Scholar]
- Ortiz-Prado, E.; Rivera-Olivero, I.A.; Freire-Paspuel, B.; Lowe, R.; Lozada, T.; Henriquez-Trujillo, A.R.; Garcia-Bereguiain, M.A. Testing for SARS-CoV-2 at the Core of Voluntary Collective Isolation: Lessons from the Indigenous Populations Living in the Amazon Region in Ecuador. Int. J. Infect. Dis. 2021, 105, 234–235. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, J.D.; Sordillo, E.M.; Gotuzzo, E.; Zavaleta, C.; Caplivski, D.; Navarro, J.C.; Crainey, J.L.; Bessa Luz, S.L.; Noguera, L.A.D.; Schaub, R.; et al. SARS-CoV-2 in the Amazon Region: A Harbinger of Doom for Amerindians. PLoS Negl. Trop. Dis. 2020, 14, e0008686. [Google Scholar] [CrossRef]
- Morales-Jadán, D.; Vallejo-Janeta, A.P.; Freire-Paspuel, B.; Rodriguez-Pazmiño, Á.S.; Rivera-Olivero, I.; Henriquez-Trujillo, A.R.; Lozada, T.; Tapia, A.; Orlando, S.A.; Ortiz-Prado, E.; et al. COVID-19 Outbreaks in Endangered Indigenous Groups from the Amazonia during the First Wave of the COVID-19 Pandemic in Ecuador: A Retrospective Cross-Sectional Study. Rural. Remote Health 2024, 24, 1–9. [Google Scholar] [CrossRef]
- Muñoz-del-Carpio-Toia, A.; Bartolo-Marchena, M.; Benites-Zapata, V.A.; Herrera-Añazco, P. Mortality from COVID-19 in Amazonian and Andean Original Indigenous Populations of Peru. Travel. Med. Infect. Dis. 2023, 56, 102658. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.T.; Nascimento, L.F.D.J.; Barbosa, A.A.T.; Martins, M.P.; Tunon, G.I.L.; Santos, P.O.M.; Dantas-Torres, F.; Dolabella, S.S. The Rising Incidence of Feline and Cat-transmitted Sporotrichosis in Latin America. Zoonoses Public Health 2024, 71, 609–619. [Google Scholar] [CrossRef]
- Destoumieux-Garzón, D.; Mavingui, P.; Boetsch, G.; Boissier, J.; Darriet, F.; Duboz, P.; Fritsch, C.; Giraudoux, P.; Le Roux, F.; Morand, S.; et al. The One Health Concept: 10 Years Old and a Long Road Ahead. Front. Vet. Sci. 2018, 5, 14. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Jeggo, M. The One Health Approach—Why Is It So Important? Trop. Med. Infect. Dis. 2019, 4, 88. [Google Scholar] [CrossRef]
- Bonilla-Aldana, D.K.; Dhama, K.; Rodriguez-Morales, A.J. Revisiting the One Health Approach in the Context of COVID-19: A Look into the Ecology of This Emerging Disease. Adv. Anim. Vet. Sci. 2020, 8, 234–237. [Google Scholar] [CrossRef]
- Eisenberg, J.N.S.; Cevallos, W.; Ponce, K.; Levy, K.; Bates, S.J.; Scott, J.C.; Hubbard, A.; Vieira, N.; Endara, P.; Espinel, M.; et al. Environmental Change and Infectious Disease: How New Roads Affect the Transmission of Diarrheal Pathogens in Rural Ecuador. Proc. Natl. Acad. Sci. USA 2006, 103, 19460–19465. [Google Scholar] [CrossRef]
- Ferrante, L. A Road to the next Pandemic: The Consequences of Amazon Highway BR-319 for Planetary Health. Lancet Planet. Health 2024, 8, e524–e525. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.P.C.; Silva, E.A.R.T.; Gama, H.S.S.D.; Cordeiro, J.S.M.; Oliveira, A.P.S.; Araújo, J.A.; Dávila, R.N.; Amazonas Júnior, H.A.; Farias, A.S.; Sachett, J.A.G.; et al. Participatory Research towards the Control of Snakebite Envenoming and Other Illnesses in a Riverine Community of the Western Brazilian Amazon. PLoS Negl. Trop. Dis. 2025, 19, e0012840. [Google Scholar] [CrossRef]
- Garnelo, L.; Parente, R.C.P.; Puchiarelli, M.L.R.; Correia, P.C.; Torres, M.V.; Herkrath, F.J. Barriers to Access and Organization of Primary Health Care Services for Rural Riverside Populations in the Amazon. Int. J. Equity Health 2020, 19, 54. [Google Scholar] [CrossRef]
- Correa, L.L.; Pinheiro, A.D.S.F. Dynamics of Parasitic Diseases and the Environmental and Sanitation Context in Cities of the Brazilian Amazon. J. Parasit. Dis. Diagn. Ther. 2017, 2. [Google Scholar]
- Rodríguez-Prieto, V.; Vicente-Rubiano, M.; Sánchez-Matamoros, A.; Rubio-Guerri, C.; Melero, M.; Martínez-López, B.; Martínez-Avilés, M.; Hoinville, L.; VERGNE, T.; Comin, A.; et al. Systematic Review of Surveillance Systems and Methods for Early Detection of Exotic, New and Re-Emerging Diseases in Animal Populations. Epidemiol. Infect. 2015, 143, 2018–2042. [Google Scholar] [CrossRef] [PubMed]
- Sangat, S.S.; Rosero, M.; Olsson, E.; Nowakowski, A.J.; Drescher-Lehman, J.; Roehrdanz, P.R.; Noon, M.L.; McManus, N.; Perz, S.G.; Angel, M.; et al. Afro-Descendant Lands in South America Contribute to Biodiversity Conservation and Climate Change Mitigation. Commun. Earth Environ. 2025, 6, 458. [Google Scholar] [CrossRef]
- Nolte, C.; Agrawal, A.; Silvius, K.M.; Soares-Filho, B.S. Governance Regime and Location Influence Avoided Deforestation Success of Protected Areas in the Brazilian Amazon. Proc. Natl. Acad. Sci. USA 2013, 110, 4956–4961. [Google Scholar] [CrossRef] [PubMed]
- Robertson, I.D.; Irwin, P.J.; Lymbery, A.J.; Thompson, R.C.A. The Role of Companion Animals in the Emergence of Parasitic Zoonoses. Int. J. Parasitol. 2000, 30, 1369–1377. [Google Scholar] [CrossRef]


| Pathogen | Location | Prevalence | Diagnostic Technique | Host Species | Reference |
|---|---|---|---|---|---|
| Mayaro virus | Brazil | ||||
| Settlements 150 km northern Manaus | 60.5% (52/86) | ELISA-ICC | Dog | [29] | |
| 46.1% (95/206) | ELISA-ICC | Cat | [29] | ||
| Rabies virus | French Guiana | ||||
| Cayenne | 0% (0/1) | Not specified | Dog | [30] | |
| 0% (0/5) | Not specified | Cat | [30] | ||
| Brazil | |||||
| Cantão State Park (TO) | 8% (4/50) | RFFIT | Dog | [31] | |
| 11.1% (1/9) | RFFIT | Cat | [31] | ||
| Bolivia | |||||
| Nöel Kempff Mercado NP Boundaries | 56% (22/39) | RFFIT | Dog | [32] | |
| Santa Cruz de la Sierra | 50.4% (4694/9308) | FITC–anti-rabies globulin | Dog | [33] | |
| Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) | Ecuador Ecuadorian Amazonia (exact location not provided) | 66.6% (2/3) | RT-qPCR | Dog | [34] |
| Sporothrix spp. (Sporothrix brasilensis, S. schenkii, S. globosa) | Brazil | ||||
| Several locations in Amazonas Estate (mostly in Manaus) | 2798 clinical cases | Culture + qPCR | Cat | [35] |
| Pathogen | Location | Prevalence | Diagnostic Technique | Host Species | Reference |
|---|---|---|---|---|---|
| Guyana | |||||
| Anaplasma phagocytophylum/A. platys | Konashen Community | 0% (0/20) | ICT | Dog | [36] |
| Borrelia burgdorferi | Guyana | ||||
| Konashen Community | 0% (0/20) | ICT | Dog | [36] | |
| Bolivia | |||||
| Nöel Kempff Mercado NP Boundaries | 0% (0/22) | IFA | Dog | [32] | |
| Brucella canis | Guyana | ||||
| Konashen Community | 0% (0/20) | ICT | Dog | [36] | |
| Bolivia | |||||
| Nöel Kempff Mercado NP Boundaries | 10% (4/40) | SAT, AGID II | Dog | [32] | |
| Brazil | |||||
| Brucella “smooth” (B. abortus, B. melitensis, B. suis, B. neotomae) | Cantão State Park (TO) | 0% (0/39) | RBT with Acidified Buffered Antigen | Dog | [37] |
| Brucella spp. | Ecuador | ||||
| Tena, Napo | 2.6% (1/39) | ELISA | Dog | [38] | |
| Brazil | |||||
| Cantão State Park (TO) | 0% (0/39) | RBT | Dog | [37] | |
| Coxiella burnetti (Q fever) | French Guiana | ||||
| Cayenne | 12.3% (7/57) | CF | Dog | [39] | |
| 0% (0/6) | CF | Cat | [39] | ||
| Ecuador | |||||
| Tena, Napo | 0% (0/39) | ELISA | Dog | [38] | |
| Ehrlichia canis/E. ewingii | Guyana | ||||
| Konashen Community | 10% (2/20) | ICT | Dog | [36] | |
| Brazil | |||||
| Chapadinha (MA) | 14.6% (47/322) | IFAT | Dog | [40] | |
| Bolivia | |||||
| Nöel Kempff Mercado NP Boundaries | 86% (19/22) | IFA | Dog | [32] | |
| Leptospira spp. | Ecuador | ||||
| Nueva Providencia, Orellana | 75% (36/48) | mAT + PCR | Dog | [41] | |
| Brazil | |||||
| Cantão State Park (TO) | 0% (0/10) | mAT | Cat | [37] | |
| 16.1% (9/56) | mAT | Dog | [37] | ||
| Bolivia | |||||
| Nöel Kempff Mercado NP Boundaries | 20% (8/40) | mAT | Dog | [32] | |
| Leptospira borgpetersenii serovar Hardjo | Brazil | ||||
| Cantão State Park (TO) | 16.1% (9/56) | SAM | Dog | [31] | |
| 0% (0/10) | SAM | Cat | [31] | ||
| Leptospira interrogans | Bolivia | ||||
| San Buenaventura (Madidi NP border) | 31% (8/26) | mAT | Dog | [42] | |
| 7% (1/14) | mAT | Cat | [42] | ||
| Leptospira interrogans serovars bratislava, canicola, grippotyphosa, hardjo, icterohemorrhagica, pomona | Konashen Community | 0% (0/20) | mAT | Dog | [36] |
| Rickettsia amblyommii | Brazil | ||||
| Several locations in Maranhão state | 10.2% (160/1560) | IFAT | Dog | [43] | |
| Rickettsia spp. (R. rickettsii, R. parkeri, R. rhipicephali & R. bellii) | Brazil | ||||
| Chapadinha (MA) | 18.9% (61/322) | IFAT | Dog | [40] | |
| 0% (0/322) | PCR | Dog | [40] | ||
| Several locations in Maranhão state | 12.6% (196/1560) | IFAT | Dog | [43] | |
| 4.1% (64/1560) | IFAT | Dog | [43] | ||
| 4.2% (66/1560) | IFAT | Dog | [43] | ||
| Bolivia | |||||
| Nöel Kempff Mercado NP Boundaries | 86% (19/22) | IFA | Dog | [32] | |
| Peru | |||||
| Cochabamba | 2.3% (1/44) | PCR | Dog | [44] | |
| 68% (30/44) | ELISA | Dog | [44] | ||
| Spotted fever group Rickettsia (R. rickettsii, R. parkeri, and R. peacockii) | Peru | ||||
| Iquitos | 59.2% (42/71) | ELISA | Dog | [45] | |
| 7.7% (1/13) | ELISA | Cat | [45] | ||
| Typhus group Rickettsia (Rickettsia typhi and Reckettsia prowazekii) | Peru | ||||
| Iquitos | 2.8% (2/71) | ELISA | Dog | [45] | |
| 0% (0/13) | ELISA | Cat | [45] |
| Pathogen | Location | Prevalence | Diagnostic Technique | Host Species | Reference |
|---|---|---|---|---|---|
| Brazil | |||||
| Amblyomma spp. (A. ovale, A. scalpturatum, A. oblongoguttatum; A. latepunctatum; A. coelebs; A. naponense) | Municipal Nat. Park of Porto Velho (RO) | 4.9% (9/184) | Stereomicroscopy and dichotomous keys (16S RNA gene sequencing for larvae) | Dog | [46] |
| Mapinguari National Park (RO) | 22.15% (35/158) | Dog | |||
| Amblyomma tigrinum | Peru | ||||
| Cochabamba | 22.7% (10/44) | Taxonomic key + PCR | Dog | [44] | |
| Acanthocheilonema reconditum | Brazil | ||||
| Marajó (PA) | 7.18% (30/418) | PCR and sequencing | Dog | [47] | |
| Ancylostoma spp. | Brazil | ||||
| Several locations in SE Acre (AC) | 42.1% (72/171) | Coprological flotation | Dog | [48] | |
| Rolim de Moura (RO) | 68.71% (112/163) | Coprology | Dog | [21] | |
| Colombia | |||||
| Cali | 11.1% (3/27) | Microscopy + PCR | Dog | [49] | |
| Babesia canis | Brazil | ||||
| Chapadinha (MA) | 16.1% (52/322) | IFAT | Dog | [40] | |
| Cantão (TO) | 10.6% (5/47) | PCR | Dog | [31] | |
| 0% (0/5) | PCR | Cat | [31] | ||
| Colombia | |||||
| Cryptosporidium spp. | Cali | 53.3% (8/15) | Microscopy + PCR | Cat | [49] |
| 55.6% (14/27) | Microscopy + PCR | Dog | [49] | ||
| Brazil | |||||
| Dirofilaria immitis | Marajó (PA) | 2.15% (9/418) | PCR and sequencing | Dog | [47] |
| Araguaína (TO) | 4.5% (5/111) | TESA-blot | Dog | [50] | |
| Lábrea (AM) | 44.4% (44/99) | PCR | Dog | [51] | |
| Manaus (AM) | 3.7% (28/766) | Blood smear | Dog | [52] | |
| Ilha do Algodoal (AC) | 35.8% (24/67) | Knott’s method + PCR | Dog | [53] | |
| Porto Velho (RO) | 12.8% (93/727) | Immunochromatography | Dog | [54] | |
| Rio Branco (AC) | Case report (1/1) | Microscopy + ICT + Echocardiography | Dog | [55] | |
| French Guiana | |||||
| Cayenne & Kourou | 15.3% (15/98) | HWAT | Dog | [56] | |
| 11.2% (11/98) | qPCR | Dog | [56] | ||
| Guyana | |||||
| Konashen Community | 10% (2/20) | IFAT | Dog | [36] | |
| Bolivia | |||||
| San Buenaventura (Madidi NP border) | 39% (11/28) | ELISA (Antigen) | Dog | [42] | |
| 93% (13/14) | kinetic ELISA | Cat | [42] | ||
| Nöel Kempff Mercado NP Boundaries | 33% (13/40) | Occult Heartworm | Dog | [32] | |
| Brazil | |||||
| Dipylidium caninum | Rolim de Moura (RO) | 1.23% (2/163) | Coprology | Dog | [21] |
| Several locations in SE Acre (AC) | 6.4% (11/171) | Coprological flotation | Dog | [48] | |
| Brazil | |||||
| Echinococcus vogeli | Southern Acre (AC) | 1.54% (1/65) | Coprology (sedimentation) + PCR + sequencing | Dog | [57] |
| Brazil | |||||
| Echinococcus granulosus | Southern Acre (AC) | 1.54% (1/65) | Coprology (sedimentation) + PCR + sequencing | Dog | [57] |
| Endolimax nana | Colombia | ||||
| Cali | 13.3% (2/15) | Microscopy + PCR | Cat | [49] | |
| Colombia | |||||
| Giardia spp. | Cali | 3.7% (1/27 | Microscopy + PCR | Dog | [49] |
| 20% (3/15) | Microscopy + PCR | Cat | [49] | ||
| Leishmania amazonensis | Brazil | ||||
| Belem (PA) | Case report (1/1) | Blood smear + Giemsa Microscopy + PCR-RFLP | Cat | [58] | |
| Ulianópolis (PA) | 45% (101/224) | IFAT | Dog | [59] | |
| 1.8% (4/224) | PCR | Dog | [59] | ||
| Colombia | |||||
| Several locations in N and W Colombia | 22.2% (10/45) | PCR | Dog | [60] | |
| 26.6% (12/45) | PCR | Dog | [60] | ||
| Leishmania (V.) braziliensis | Brazil | ||||
| Ulianópolis (PA) | 40.6% (91/224) | IFAT | Dog | [59] | |
| 30.3% (68/224) | IFAT | Dog | [59] | ||
| Several locations in N and W Colombia | 17.7% (8/45) | PCR | Dog | [60] | |
| Tomé-Açu (PA) | 14.2% (3/21) | PCR + sequencing (BLASTn) | Dog | [61] | |
| Leishmania chagasi | Brazil | ||||
| Araguaína (TO) | 54.95% (61/111) | IFAT | Dog | [50] | |
| 51.35% (57/111) | ELISA | Dog | [50] | ||
| Brazil | |||||
| Leishmania (V.) guyanensis | Tomé-Açu (PA) | 23.8% (5/21) | PCR + sequencing (BLASTn) | Dog | [61] |
| Colombia | |||||
| Leishmania panamensis | Several locations in N and W Colombia | 13.3% (6/45) | PCR | Dog | [60] |
| Colombia | |||||
| Leishmania infantum | Several locations in N and W Colombia | 6.6% (3/45) | PCR | Dog | [60] |
| Guyana | |||||
| Konashen Community | 5% (1/20) | IFAT | Dog | [36] | |
| Brazil | |||||
| Southern Mato Grosso (MT) | Analysis on 46 known positive dogs | MLMT | Dog | [62] | |
| Labréa (AM) | 8% (8/99) | IFAT | Dog | [63] | |
| Manaus (AM) | 39% (60/154) | PCR | Dog | [64] | |
| 20.8% (32/154) | Serology | Dog | [64] | ||
| Marabá (PA) | 75.5% (302/400) | Serology with chromatography | Dog | [65] | |
| 59.25% (237/400) | PCR | Dog | [65] | ||
| Urubú Branco, Confresa (MT) | 4.4% (5/114) | ELISA | Dog | [66] | |
| São Luís (MA) | 30.4% (32/105) | IFAT | Cat | [67] | |
| 8.5% (9/105) | PCR | Cat | [67] | ||
| Several locations at central Pará (PA) | 23.2% (30/129) | ELISA | Dog | [68] | |
| Tomé-Açu (PA) | 57.1% (12/21) | PCR + sequencing (BLASTn) | Dog | [61] | |
| Leishmania shawi | Brazil | ||||
| Ulianópolis (PA) | 43.3% (97/224) | IFAT | Dog | [59] | |
| Brazil | |||||
| Leishmania spp. | Corumbá (MS) | 50% (31/62) | IFAT + ELISA | Dog | [69] |
| Xapuri (AC) | Detected, but not specific prevalence | NNN Culture + direct examination, PCR + sequencing, RFLP and HRM | Dog | [70] | |
| Xingú river (PA) | 15.4% (46/298) | IFAT + ELISA | Dog | [71] | |
| São Luís (MA) | 26.25% (21/80) | IFAT | Cat | [72] | |
| Tomé-Açu (PA) | 83% (30/36) | PCR + sequencing (BLASTn) | Dog | [61] | |
| 4.7% (1/21) | |||||
| French Guiana | |||||
| Cayenne & Kourou | (autochthon) 1.7% (3/179), (military working dogs) 5.1% (4/78) | qPCR + sequencing | Dog | [73] | |
| Cayenne | Clinical case (3 dogs) | ICT + PCR | Dog | [74] | |
| Peru | |||||
| Huánuco Department | 26% (251/953) | ELISA + PCR | Dog | [75] | |
| Rodentolepis (Hymenolepis) nana | Brazil | ||||
| Several locations in SE Acre (AC) | 0.58% (1/171) | Coprological flotation | Dog | [48] | |
| Brazil | |||||
| Rhipicephalus sanguineus | Municipal Nat. Park of Porto Velho (RO) | 15.8% (29/184) | stereomicroscopy and dichotomous keys (16S RNA gene sequencing for larvae) | Dog | [46] |
| Mapinguari National Park (RO) | 16.6% (26/158) | Dog | [46] | ||
| Several locations in Maranhão state (MA) | 9.6% (150/1560) | Dichotomous keys | Dog | [43] | |
| Brazil | |||||
| Toxocara spp. | Several locations in SE Acre (AC) | 18.1% (15/83) | Coprological flotation | Dog | [48] |
| Brazil | |||||
| Toxoplasma gondii | Xingú river (PA) | 48.8% (124/245) | IFAT | Dog | [71] |
| Cantão State Park (TO) | 47.8% (22/46) | MAT | Dog | [37] | |
| 80% (8/10) | MAT | Cat | [37] | ||
| Rolim de Moura (RO) | 82.2% (376/458) | IFAT | Dog | [76] | |
| Labréa (AM) | 61.6% (61/99) | IFAT | Dog | [63] | |
| Several locations at central Pará (PA) | 69.8% (90/129) | IFAT | Dog | [68] | |
| Tapirapé comm. (MT) | 42.22% (47/114) | IFAT | Dog | [77] | |
| Karajá comm. (PA) | 52.83% (112/212) | IFAT | Dog | [77] | |
| Manaus (AM) | 12.3% (19/154) | IFAT | Dog | [78] | |
| Rolim de Moura (RO) | 82.2% (376/458) | IFAT | Dog | [76] | |
| Labréa (AM) | 61.6% (61/99) | IFAT | Dog | [63] | |
| Peru | |||||
| Nueva Esperanza, Yavari-Mirin basin | 94.1% (16/17) | ELISA | Dog | [38] | |
| 100% (4/4) | ELISA | Cat | [38] | ||
| Colombia | |||||
| Cali | 6.6% (1/15) | Microscopy + PCR | Cat | [49] | |
| French Guiana | |||||
| Cayenne & Kourou | 55.7% (49/88) | MAT (>1/20) | Mostly dogs & cats (but also a non-specified number of other species) | [79] | |
| Bolivia | |||||
| San Buenaventura (Madidi NP border) | 62% (16/26) | IHA | Dog | [42] | |
| Nöel Kempff Mercado NP Boundaries | 80% (32/40) | IHA | Dog | [32] | |
| Trichuris vulpis | Brazil | ||||
| Rolim de Moura (RO) | 11.66% (19/163) | Coprology | Dog | [21] | |
| Acre | 5.26% (9/171) | Coprological flotation | Dog | [48] | |
| Brazil | |||||
| Trypanosoma cruzi | Corumbá (MS) | 76% (47/62) | IFAT, ELISA and nPCR | Dog | [69] |
| French Guiana | |||||
| Cayenne & Kourou | 5.8% (9/153) | RICT + PCR | Dog | [80] | |
| Guyana | |||||
| Konashen Community | 0% (0/20) | IFAT | Dog | [36] | |
| Colombia | |||||
| Talaigua nuevo | 18.9 (31/164) | ELISA + IFAT | Dog | [81] | |
| Several locations in N and W Colombia | 13.3% (6/45) | PCR | Dog | [60] | |
| Brazil | |||||
| Trypanosoma evansi | Corumbá (MS) | 73% (45/62) | IFAT and nPCR | Dog | [69] |
| Ariquemes (RO) | Clinical case 1 dog | Blood smears and PCR | Dog | [82] | |
| Colombia | |||||
| Vichada | 10.5% (49/465). | ELISA + IFAT | Dog | [83] | |
| Trypanosoma spp. | Brazil | ||||
| Abaetetuba (PA) | 0% (0/11) | Blood culture | Dog | [84] | |
| Peru | |||||
| Tocache, San Martin | 100% (1/1) | Blood smear + PCR | Dog | [85] | |
| Tunga penetrans | Brazil | ||||
| Nossa Senhora do Livramento (AM) | 75.6% (59/78) | Taxonomic key | Dog | [86] | |
| Ecuador | |||||
| Tunga trimamillata | Guayaquil (imported from Loja) | Clinical case (1/1) | Morphological diagnosis (characteristic nodule) + histopathology | Dog | [87] |
| Bolivia | |||||
| Sarcoptes scabiei | San Buenaventura (Madidi NP border) | 55% (22/40) | ELISA | Dog | [42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Vanderhooft, C.J.; Díaz, E.A.; Sáenz, C.; Lizana, V. 2000–2025: A Quarter of a Century of Studies on Pet Ownership in the Amazon—Epidemiological Implications for Public Health. Pathogens 2026, 15, 77. https://doi.org/10.3390/pathogens15010077
Vanderhooft CJ, Díaz EA, Sáenz C, Lizana V. 2000–2025: A Quarter of a Century of Studies on Pet Ownership in the Amazon—Epidemiological Implications for Public Health. Pathogens. 2026; 15(1):77. https://doi.org/10.3390/pathogens15010077
Chicago/Turabian StyleVanderhooft, Coline J., Eduardo A. Díaz, Carolina Sáenz, and Victor Lizana. 2026. "2000–2025: A Quarter of a Century of Studies on Pet Ownership in the Amazon—Epidemiological Implications for Public Health" Pathogens 15, no. 1: 77. https://doi.org/10.3390/pathogens15010077
APA StyleVanderhooft, C. J., Díaz, E. A., Sáenz, C., & Lizana, V. (2026). 2000–2025: A Quarter of a Century of Studies on Pet Ownership in the Amazon—Epidemiological Implications for Public Health. Pathogens, 15(1), 77. https://doi.org/10.3390/pathogens15010077






