SARS-CoV-2 Infection in Children: Revisiting Host–Virus Interactions Through Post-Infection Immune Profiling
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Immune Assessment
2.3. Assessment of Specific Immune Responses to SARS-CoV-2 Antigens
2.4. Flow Cytometry Data Acquisition and Analysis
2.5. Statistics
3. Results
3.1. Characterization of the Study Population
3.2. Immune Profile After SARS-CoV-2 Infection in Pediatric Patients
3.3. Specific Humoral and Cellular Responses to SARS-CoV-2 in Children After Infection
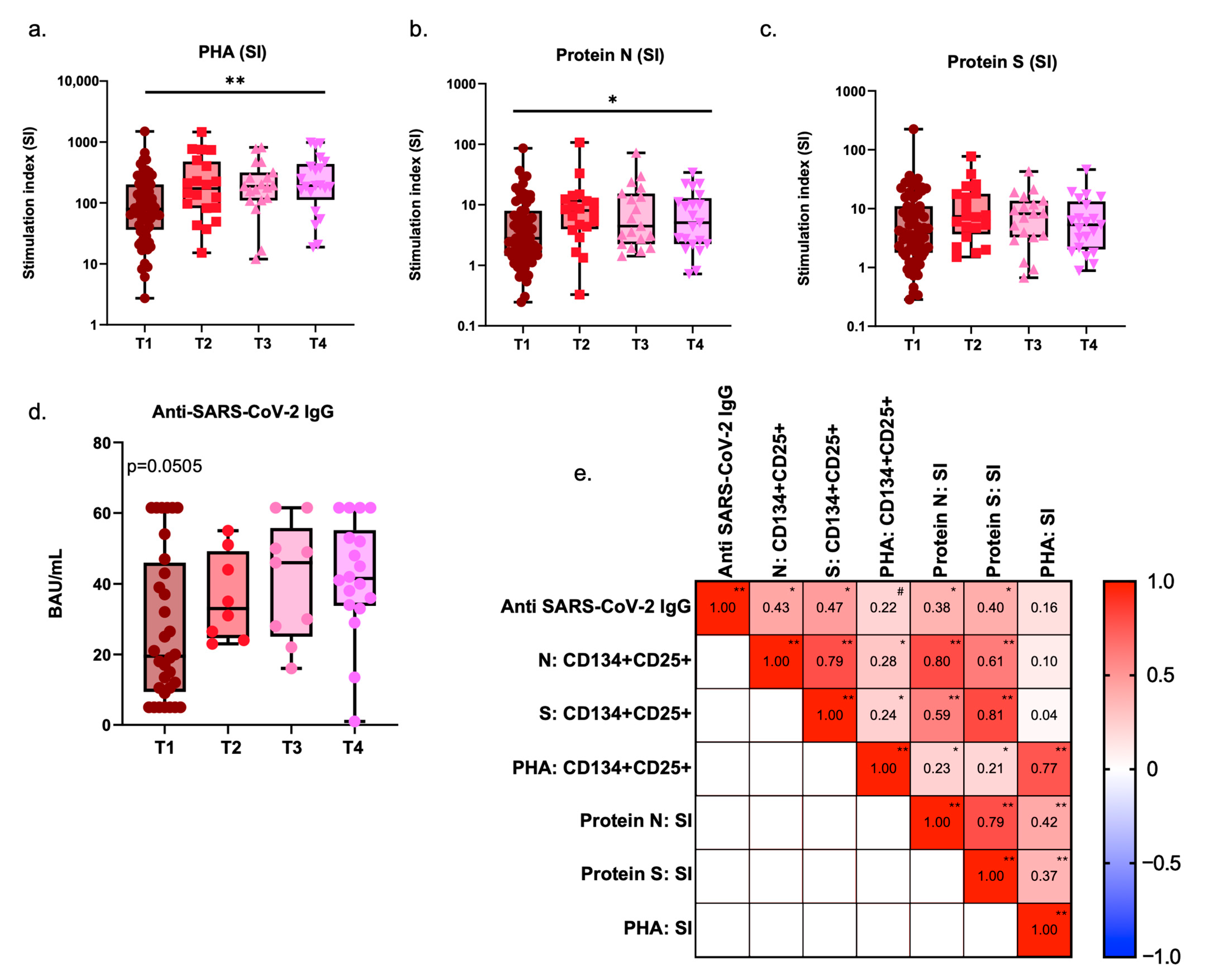
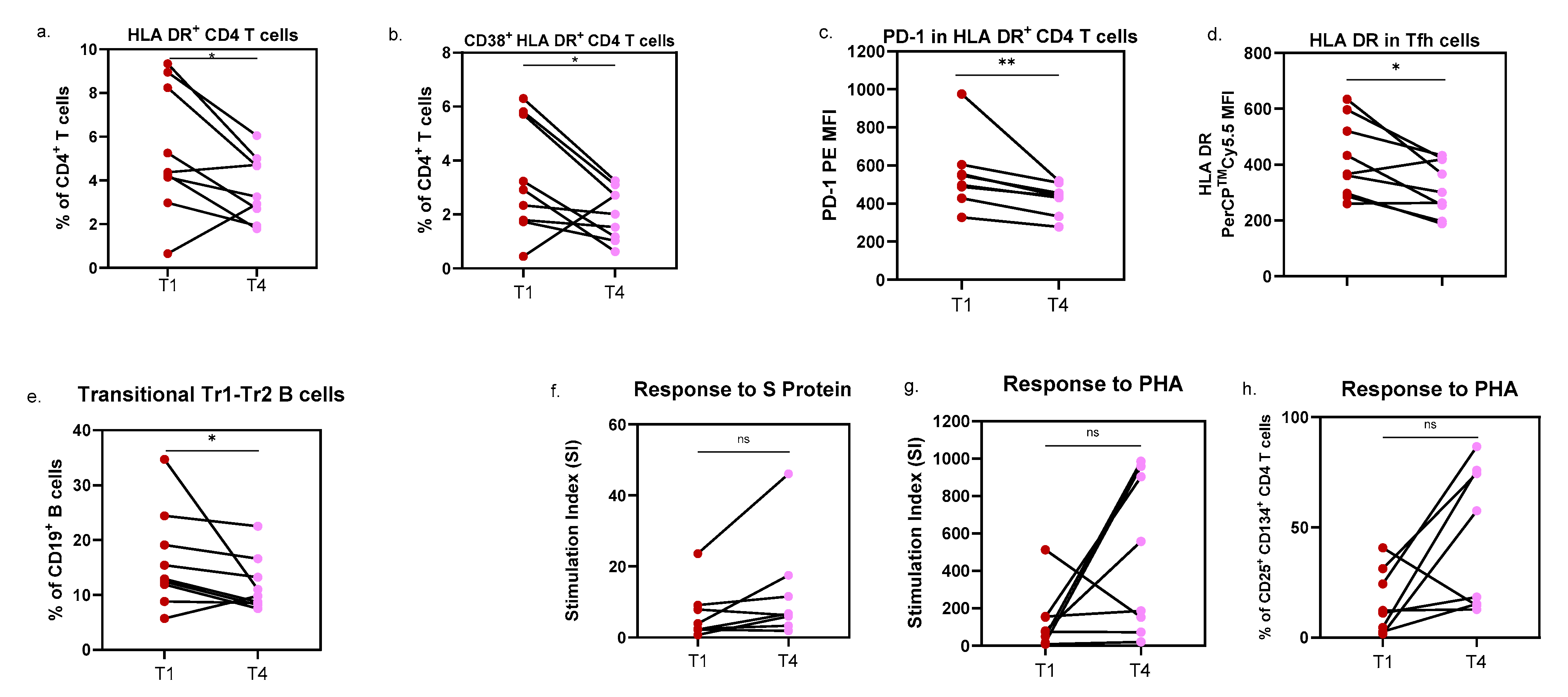
3.4. Longitudinal Analysis of Immune Dynamics Following SARS-CoV-2 Infection
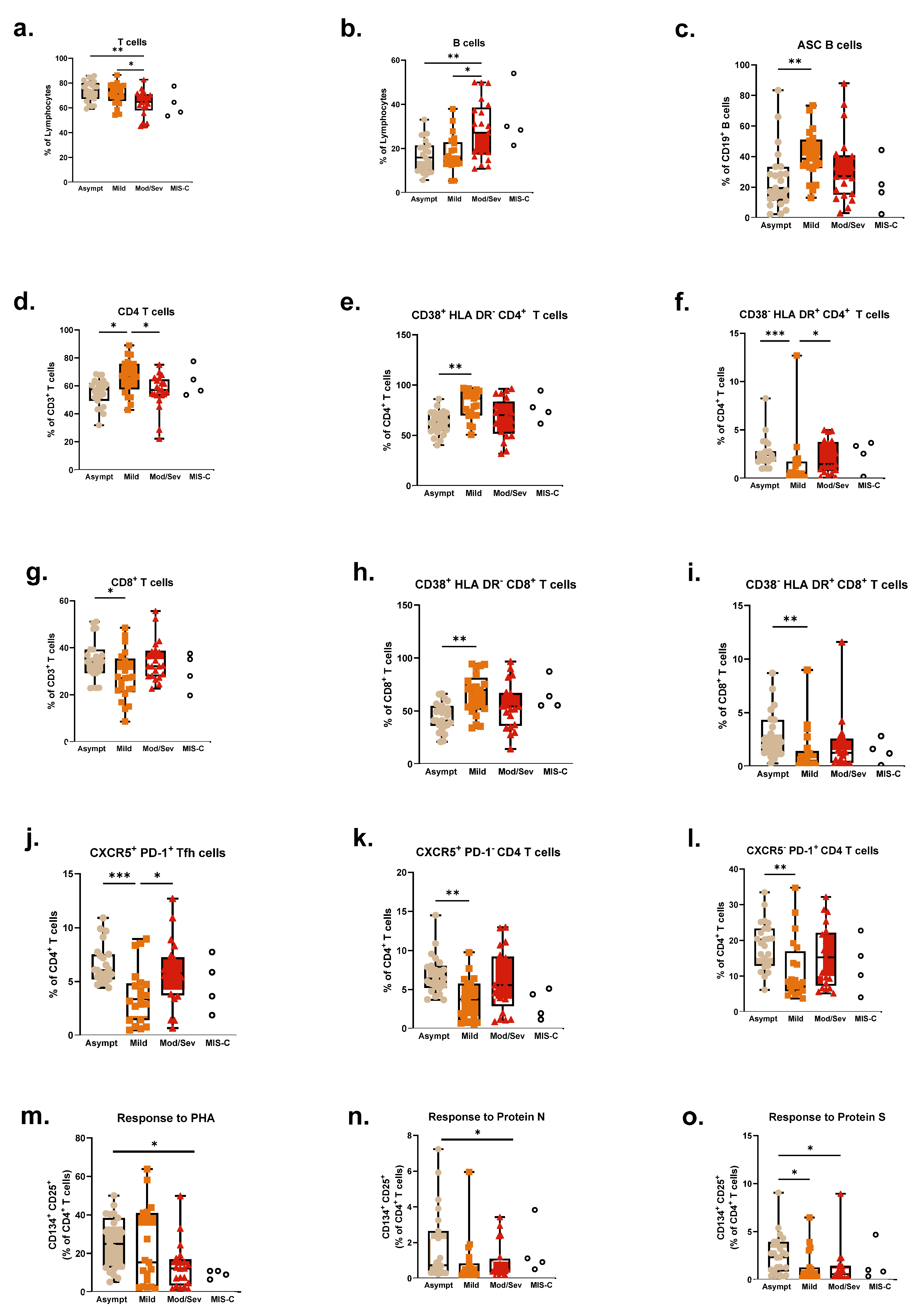
3.5. Immune Responses and Severity of Disease Symptoms
3.6. Impact of Age and Gender on the Immune Response to SARS-CoV-2 Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Abbreviations
| ACE-2 | Angiotensin-Converting Enzyme 2 |
| ASC | antibody-secreting cells |
| CHULC | Centro Hospitalar Universitário de Lisboa Central |
| COVID-19 | Coronavirus Disease 2019 |
| EDTA | Ethylenediaminetetraacetic acid |
| IFN | interferon |
| MFI | mean fluorescence intensity |
| MIS-C | Multisystem Inflammatory Syndrome in Children |
| NK | Natural killer cells |
| NS | non-stimulated |
| PCR | Polymerase Chain Reaction |
| PD-1 | Programmed cell Death protein 1 |
| PHA | Phytohemagglutinin |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 |
| SI | stimulation index |
| T1 | time point 1, (<14 d of infection) |
| T2 | time point 2, (14 d to 3 months after infection) |
| T3 | time point 3, (3 to 6 months after infection) |
| T4 | time point 4, (6 to 12 months after infection) |
| Tch | follicular CD8+ T cytotoxic cells |
| Tfh | follicular CD4+ T helper cells |
| Tr | transitional |
| UNICEF | United Nations International Children’s Emergency Fund |
| WHO | World Health Organization |
References
- Rotulo, G.A.; Palma, P. Understanding COVID-19 in children: Immune determinants and post-infection conditions. Pediatr. Res. 2023, 94, 434–442. [Google Scholar] [CrossRef]
- Nikolopoulou, G.B.; Maltezou, H.C. COVID-19 in Children: Where do we Stand? Arch. Med. Res. 2022, 53, 1–8. [Google Scholar] [CrossRef]
- Soriano, J.B.; Murthy, S.; Marshall, J.C.; Relan, P.; Diaz, J.V. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 2022, 22, e102–e107. [Google Scholar] [CrossRef]
- Ahmed, I.S.; Tapponi, S.L.; Widatallah, M.E.; Alakkad, Y.M.; Haider, M. Unmasking the enigma: An in-depth analysis of COVID-19 impact on the pediatric population. J. Infect. Public Health 2023, 16, 1346–1360. [Google Scholar] [CrossRef]
- Vieira de Melo, J.; Valsassina, R.; Garcia, A.M.; Silva, T.; Gouveia, C.; Brito, M.J. Multisystem Inflammatory Syndrome in Children Associated with COVID-19 in a Tertiary Level Hospital in Portugal. Acta Med. Port. 2022, 35, 881–890. [Google Scholar] [CrossRef]
- Silva, T.M.; Garcia, A.M.; Gouveia, C.; Candeias, F.; Brito, M.J. COVID-19 in a tertiary paediatric centre in Portugal: A single-centre retrospective study. BMJ Paediatr. Open 2022, 6, e001252. [Google Scholar] [CrossRef] [PubMed]
- Stepan, M.D.; Cioboata, R.; Vintilescu, Ş.; Vasile, C.M.; Osman, A.; Ciolofan, M.S.; Popescu, M.; Petrovici, I.L.; Zavate, A.C. Pediatric Functional Abdominal Pain Disorders following COVID-19. Life 2022, 12, 509. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Gao, L.; Wang, X.; Zhong, L.; Li, J.; Ding, S.; Zheng, Y.; Liu, J.; Cui, Y.; Wang, L.; et al. Epidemiological characteristics and clinical manifestations of pediatric patients with COVID-19 in China: A multicenter retrospective study. Pediatr. Investig. 2021, 5, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Gotzinger, F.; Santiago-Garcia, B.; Noguera-Julian, A.; Lanaspa, M.; Lancella, L.; Calo Carducci, F.I.; Gabrovska, N.; Velizarova, S.; Prunk, P.; Osterman, V.; et al. COVID-19 in children and adolescents in Europe: A multinational, multicentre cohort study. Lancet Child Adolesc. Health 2020, 4, 653–661. [Google Scholar] [CrossRef] [PubMed]
- King, J.A.; Whitten, T.A.; Bakal, J.A.; McAlister, F.A. Symptoms associated with a positive result for a swab for SARS-CoV-2 infection among children in Alberta. CMAJ 2021, 193, E1–E9. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Zhang, L.; Du, H.; Zhang, J.; Li, Y.Y.; Qu, J.; Zhang, W.; Wang, Y.; Bao, S.; Li, Y.; et al. SARS-CoV-2 Infection in Children. N. Engl. J. Med. 2020, 382, 1663–1665. [Google Scholar] [CrossRef]
- Kloc, M.; Ghobrial, R.M.; Kuchar, E.; Lewicki, S.; Kubiak, J.Z. Development of child immunity in the context of COVID-19 pandemic. Clin. Immunol. 2020, 217, 108510. [Google Scholar] [CrossRef]
- Dorjee, K.; Kim, H.; Bonomo, E.; Dolma, R. Prevalence and predictors of death and severe disease in patients hospitalized due to COVID-19: A comprehensive systematic review and meta-analysis of 77 studies and 38,000 patients. PLoS ONE 2020, 15, e0243191. [Google Scholar] [CrossRef]
- Felsenstein, S.; Hedrich, C.M. COVID-19 in children and young people. Lancet Rheumatol. 2020, 2, e514–e516. [Google Scholar] [CrossRef]
- Chou, J.; Thomas, P.G.; Randolph, A.G. Immunology of SARS-CoV-2 infection in children. Nat. Immunol. 2022, 23, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Posfay-Barbe, K.M.; Wagner, N.; Gauthey, M.; Moussaoui, D.; Loevy, N.; Diana, A.; L’Huillier, A.G. COVID-19 in Children and the Dynamics of Infection in Families. Pediatrics 2020, 146, e20201576. [Google Scholar] [CrossRef]
- Jones, T.C.; Biele, G.; Muhlemann, B.; Veith, T.; Schneider, J.; Beheim-Schwarzbach, J.; Bleicker, T.; Tesch, J.; Schmidt, M.L.; Sander, L.E.; et al. Estimating infectiousness throughout SARS-CoV-2 infection course. Science 2021, 373, eabi5273. [Google Scholar] [CrossRef]
- Park, Y.J.; Choe, Y.J.; Park, O.; Park, S.Y.; Kim, Y.M.; Kim, J.; Kweon, S.; Woo, Y.; Gwack, J.; Kim, S.S.; et al. Contact Tracing during Coronavirus Disease Outbreak, South Korea, 2020. Emerg. Infect. Dis. 2020, 26, 2465–2468. [Google Scholar] [CrossRef] [PubMed]
- Seidel, A.; Jacobsen, E.M.; Fabricius, D.; Class, M.; Zernickel, M.; Blum, C.; Conzelmann, C.; Weil, T.; Gross, R.; Bode, S.F.N.; et al. Serum neutralizing capacity and T-cell response against the omicron BA.1 variant in seropositive children and their parents one year after SARS-CoV-2 infection. Front. Pediatr. 2023, 11, 1020865. [Google Scholar] [CrossRef] [PubMed]
- DGS. Norma 013/2022. Abordagem Das Pessoas Com Suspeita ou Confirmação de COVID-19. 2022. Available online: https://www.dgs.pt/normas-orientacoes-e-informacoes/normas-e-circulares-normativas/norma_013_2022-abordagem-das-pessoas-com-suspeita-ou-confirmacao-de-covid-19-pdf.aspx (accessed on 24 July 2025).
- Instituto Nacional De Saúde Doutor Ricardo Jorge. Diversidade Genética do Novo Coronavírus SARS-CoV-2 (COVID-19) em Portugal. Relatório de 31 May 2022. Available online: https://insaflu.insa.pt/covid19/. (accessed on 31 December 2023).
- Morris, S.B. Estimating effect sizes from pretest-posttest-control group designs. Organ. Res. Methods 2008, 11, 364–386. [Google Scholar] [CrossRef]
- Wolfgang, L.; Lenhard, A. Computation of effect sizes. Psychometrica. 2022. Available online: https://www.psychometrica.de/effect_size.html (accessed on 24 July 2025).
- Brodin, P. Immune responses to SARS-CoV-2 infection and vaccination in children. Semin. Immunol. 2023, 69, 101794. [Google Scholar] [CrossRef]
- Rybkina, K.; Bell, J.N.; Bradley, M.C.; Wohlbold, T.; Scafuro, M.; Meng, W.; Korenberg, R.C.; Davis-Porada, J.; Anderson, B.R.; Weller, R.J.; et al. SARS-CoV-2 infection and recovery in children: Distinct T cell responses in MIS-C compared to COVID-19. J. Exp. Med. 2023, 220, e20221518. [Google Scholar] [CrossRef]
- Casabianca, M.; Caula, C.; Titomanlio, L.; Lenglart, L. Neurological consequences of SARS-CoV-2 infections in the pediatric population. Front. Pediatr. 2023, 11, 1123348. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.Y.; Day-Lewis, M.; Henderson, L.A.; Friedman, K.G.; Lo, J.; Roberts, J.E.; Lo, M.S.; Platt, C.D.; Chou, J.; Hoyt, K.J.; et al. Distinct clinical and immunological features of SARS-CoV-2-induced multisystem inflammatory syndrome in children. J. Clin. Investig. 2020, 130, 5942–5950. [Google Scholar] [CrossRef]
- Vella, L.A.; Giles, J.R.; Baxter, A.E.; Oldridge, D.A.; Diorio, C.; Kuri-Cervantes, L.; Alanio, C.; Pampena, M.B.; Wu, J.E.; Chen, Z.; et al. Deep immune profiling of MIS-C demonstrates marked but transient immune activation compared to adult and pediatric COVID-19. Sci. Immunol. 2021, 6, eabf7570. [Google Scholar] [CrossRef]
- Butters, C.; Benede, N.; Moyo-Gwete, T.; Richardson, S.I.; Rohlwink, U.; Shey, M.; Ayres, F.; Manamela, N.P.; Makhado, Z.; Balla, S.R.; et al. Comparing the immune abnormalities in MIS-C to healthy children and those with inflammatory disease reveals distinct inflammatory cytokine production and a monofunctional T cell response. Clin. Immunol. 2023, 259, 109877. [Google Scholar] [CrossRef] [PubMed]
- Govender, M.; Hopkins, F.R.; Goransson, R.; Svanberg, C.; Shankar, E.M.; Hjorth, M.; Nilsdotter-Augustinsson, A.; Sjowall, J.; Nystrom, S.; Larsson, M. T cell perturbations persist for at least 6 months following hospitalization for COVID-19. Front. Immunol. 2022, 13, 931039. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Wei, L.; Li, G.; Hua, M.; Sun, Y.; Wang, D.; Han, K.; Yan, Y.; Song, C.; Song, R.; et al. Persistent High Percentage of HLA-DR(+)CD38(high) CD8(+) T Cells Associated With Immune Disorder and Disease Severity of COVID-19. Front. Immunol. 2021, 12, 735125. [Google Scholar] [CrossRef]
- Khoo, W.H.; Jackson, K.; Phetsouphanh, C.; Zaunders, J.J.; Alquicira-Hernandez, J.; Yazar, S.; Ruiz-Diaz, S.; Singh, M.; Dhenni, R.; Kyaw, W.; et al. Tracking the clonal dynamics of SARS-CoV-2-specific T cells in children and adults with mild/asymptomatic COVID-19. Clin. Immunol. 2023, 246, 109209. [Google Scholar] [CrossRef]
- Vazquez-Alejo, E.; Tarancon-Diez, L.; Espinar-Buitrago, M.S.; Genebat, M.; Calderon, A.; Perez-Cabeza, G.; Magro-Lopez, E.; Leal, M.; Munoz-Fernandez, M.A. Persistent Exhausted T-Cell Immunity after Severe COVID-19: 6-Month Evaluation in a Prospective Observational Study. J. Clin. Med. 2023, 12, 3539. [Google Scholar] [CrossRef]
- Beckmann, N.D.; Comella, P.H.; Cheng, E.; Lepow, L.; Beckmann, A.G.; Tyler, S.R.; Mouskas, K.; Simons, N.W.; Hoffman, G.E.; Francoeur, N.J.; et al. Downregulation of exhausted cytotoxic T cells in gene expression networks of multisystem inflammatory syndrome in children. Nat. Commun. 2021, 12, 4854. [Google Scholar] [CrossRef] [PubMed]
- Kreutmair, S.; Unger, S.; Nunez, N.G.; Ingelfinger, F.; Alberti, C.; De Feo, D.; Krishnarajah, S.; Kauffmann, M.; Friebel, E.; Babaei, S.; et al. Distinct immunological signatures discriminate severe COVID-19 from non-SARS-CoV-2-driven critical pneumonia. Immunity 2021, 54, 1578–1593.e1575. [Google Scholar] [CrossRef]
- Zhang, Y.; Yan, Q.; Luo, K.; He, P.; Hou, R.; Zhao, X.; Wang, Q.; Yi, H.; Liang, H.; Deng, Y.; et al. Analysis of B Cell Receptor Repertoires Reveals Key Signatures of the Systemic B Cell Response after SARS-CoV-2 Infection. J. Virol. 2022, 96, e0160021. [Google Scholar] [CrossRef]
- Peart Akindele, N.; Pieterse, L.; Suwanmanee, S.; Griffin, D.E. B-Cell Responses in Hospitalized Severe Acute Respiratory Syndrome Coronavirus 2-Infected Children With and Without Multisystem Inflammatory Syndrome. J. Infect. Dis. 2022, 226, 822–832. [Google Scholar] [CrossRef]
- Bartlett, M.L.; Suwanmanee, S.; Peart Akindele, N.; Ghimire, S.; Chan, A.K.P.; Guo, C.; Gould, S.J.; Cox, A.L.; Griffin, D.E. Continued Virus-Specific Antibody-Secreting Cell Production, Avidity Maturation and B Cell Evolution in Patients Hospitalized with COVID-19. Viral. Immunol. 2022, 35, 259–272. [Google Scholar] [CrossRef]
- Nehar-Belaid, D.; Mejías, A.; Xu, Z.; Marches, R.; Yerrabelli, R.; Chen, G.; Mertz, S.; Ye, F.; Sánchez, P.J.; Tsang, J.S.; et al. SARS-CoV-2 induced immune perturbations in infants vary with disease severity and differ from adults’ responses. Nat. Commun. 2025, 16, 4562. [Google Scholar] [CrossRef]
- Mansourabadi, A.H.; Aghamajidi, A.; Dorfaki, M.; Keshavarz, F.; Shafeghat, Z.; Moazzeni, A.; Arab, F.L.; Rajabian, A.; Roozbehani, M.; Falak, R.; et al. B lymphocytes in COVID-19: A tale of harmony and discordance. Arch. Virol. 2023, 168, 148. [Google Scholar] [CrossRef]
- Fiandrino, S.; Donà, D.; Giaquinto, C.; Poletti, P.; Tira, M.D.; Di Chiara, C.; Paolotti, D. Clinical characteristics of COVID-19 in children and adolescents: Insights from an Italian paediatric cohort using a machine-learning approach. BMJ Public Health 2025, 3, e001888. [Google Scholar] [CrossRef] [PubMed]
- Andersen, K.M.; McColgan, M.D.; Cane, A.; Moran, M.M.; Reimbaeva, M.; Lopez, S.M.C. Comparison of COVID-19-associated inpatient burden by variant period in hospitalized children age <5 years in the United States. Front. Pediatr. 2025, 13, 1573933. [Google Scholar] [CrossRef] [PubMed]
- Zayou, L.; Prakash, S.; Vahed, H.; Dhanushkodi, N.R.; Quadiri, A.; Belmouden, A.; Lemkhente, Z.; Chentoufi, A.; Gil, D.; Ulmer, J.B.; et al. Dynamics of spike-specific neutralizing antibodies across five-year emerging SARS-CoV-2 variants of concern reveal conserved epitopes that protect against severe COVID-19. Front. Immunol. 2025, 16, 1503954. [Google Scholar] [CrossRef]
- Kharroubi, S.A.; Diab-El-Harake, M. Sex-differences in COVID-19 diagnosis, risk factors and disease comorbidities: A large US-based cohort study. Front. Public Health 2022, 10, 1029190. [Google Scholar] [CrossRef]
- Dong, Y.; Mo, X.; Hu, Y.; Qi, X.; Jiang, F.; Jiang, Z.; Tong, S. Epidemiology of COVID-19 Among Children in China. Pediatrics 2020, 145, e20200702. [Google Scholar] [CrossRef]
- Cristiani, L.; Mancino, E.; Matera, L.; Nenna, R.; Pierangeli, A.; Scagnolari, C.; Midulla, F. Will children reveal their secret? The coronavirus dilemma. Eur. Respir. J. 2020, 55, 2000749. [Google Scholar] [CrossRef] [PubMed]
- Rydyznski Moderbacher, C.; Ramirez, S.I.; Dan, J.M.; Grifoni, A.; Hastie, K.M.; Weiskopf, D.; Belanger, S.; Abbott, R.K.; Kim, C.; Choi, J.; et al. Antigen-Specific Adaptive Immunity to SARS-CoV-2 in Acute COVID-19 and Associations with Age and Disease Severity. Cell 2020, 183, 996–1012.e19. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Mentzer, A.J.; Liu, G.; Yao, X.; Yin, Z.; Dong, D.; Dejnirattisai, W.; Rostron, T.; Supasa, P.; Liu, C.; et al. Broad and strong memory CD4(+) and CD8(+) T cells induced by SARS-CoV-2 in UK convalescent individuals following COVID-19. Nat. Immunol. 2020, 21, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Sekine, T.; Perez-Potti, A.; Rivera-Ballesteros, O.; Stralin, K.; Gorin, J.B.; Olsson, A.; Llewellyn-Lacey, S.; Kamal, H.; Bogdanovic, G.; Muschiol, S.; et al. Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19. Cell 2020, 183, 158–168.e14. [Google Scholar] [CrossRef]
- Cohen, C.A.; Li, A.P.Y.; Hachim, A.; Hui, D.S.C.; Kwan, M.Y.W.; Tsang, O.T.Y.; Chiu, S.S.; Chan, W.H.; Yau, Y.S.; Kavian, N.; et al. SARS-CoV-2 specific T cell responses are lower in children and increase with age and time after infection. Nat. Commun. 2021, 12, 4678. [Google Scholar] [CrossRef]
- Lin, J.; Law, R.; Korosec, C.S.; Zhou, C.; Koh, W.H.; Ghaemi, M.S.; Samaan, P.; Ooi, H.K.; Matveev, V.; Yue, F.; et al. Longitudinal Assessment of SARS-CoV-2-Specific T Cell Cytokine-Producing Responses for 1 Year Reveals Persistence of Multicytokine Proliferative Responses, with Greater Immunity Associated with Disease Severity. J. Virol. 2022, 96, e0050922. [Google Scholar] [CrossRef]
- Nelson, R.W.; Chen, Y.; Venezia, O.L.; Majerus, R.M.; Shin, D.S.; MGH COVID-19 Collection & Processing Team; Carrington, M.N.; Yu, X.G.; Wesemann, D.R.; Moon, J.J.; et al. SARS-CoV-2 epitope-specific CD4(+) memory T cell responses across COVID-19 disease severity and antibody durability. Sci. Immunol. 2022, 7, eabl9464. [Google Scholar] [CrossRef]
- Sieber, J.; Mayer, M.; Schmidthaler, K.; Kopanja, S.; Camp, J.V.; Popovitsch, A.; Dwivedi, V.; Hoz, J.; Schoof, A.; Weseslindtner, L.; et al. Long-Lived Immunity in SARS-CoV-2-Recovered Children and Its Neutralizing Capacity Against Omicron. Front. Immunol. 2022, 13, 882456. [Google Scholar] [CrossRef]
- Zaunders, J.J.; Munier, M.L.; Seddiki, N.; Pett, S.; Ip, S.; Bailey, M.; Xu, Y.; Brown, K.; Dyer, W.B.; Kim, M.; et al. High levels of human antigen-specific CD4+ T cells in peripheral blood revealed by stimulated coexpression of CD25 and CD134 (OX40). J. Immunol. 2009, 183, 2827–2836. [Google Scholar] [CrossRef]
- Almendro-Vazquez, P.; Laguna-Goya, R.; Paz-Artal, E. Defending against SARS-CoV-2: The T cell perspective. Front. Immunol. 2023, 14, 1107803. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.T.; Linster, M.; Tan, C.W.; Le Bert, N.; Chia, W.N.; Kunasegaran, K.; Zhuang, Y.; Tham, C.Y.L.; Chia, A.; Smith, G.J.D.; et al. Early induction of functional SARS-CoV-2-specific T cells associates with rapid viral clearance and mild disease in COVID-19 patients. Cell Rep. 2021, 34, 108728. [Google Scholar] [CrossRef] [PubMed]
- Joshi, D.; Nyhoff, L.E.; Zarnitsyna, V.I.; Moreno, A.; Manning, K.; Linderman, S.; Burrell, A.R.; Stephens, K.; Norwood, C.; Mantus, G.; et al. Infants and young children generate more durable antibody responses to SARS-CoV-2 infection than adults. iScience 2023, 26, 107967. [Google Scholar] [CrossRef] [PubMed]
- Cimolai, N. COVID-19 among infants: Key clinical features and remaining controversies. Clin. Exp. Pediatr. 2024, 67, 1–16. [Google Scholar] [CrossRef]



| Asymptomatic Infection | |
| Mild disease | Mild symptoms, including anosmia, ageusia, or dysgeusia of sudden onset |
| Moderate disease | Fever lasting 3 or more days; or, Dyspnea 1 but with SpO2 ≥ 90% (or ≥ 93% in pediatric age) on room air, and without hemodynamic instability |
| Severe disease | Pneumonia 2 with respiratory distress, respiratory rate > 30 cpm, or SpO2 < 90% on air room; or with hemodynamic instability |
| Group | T1 n = 74 | T2 n = 23 | T3 n = 20 | T4 n = 28 | p-Value |
|---|---|---|---|---|---|
| Gender, n (%) | 0.0196 a | ||||
| Girls | 35 (47.3) | 8 (34.7) | 2 (10.0) | 13 (46.4) | |
| Boys | 39 (52.7) | 15 (65.2) | 18 (90.0) | 15 (53.6) | |
| # vs. T3 | # vs. T4 | ||||
| Age, y | 0.5293 b | ||||
| Median | 6.3 | 8.4 | 2.8 | 6.7 | |
| [IQR] | [1.3–13.6] | [3.9–13.3] | [0.7–12.5] | [1.7–10.6] | |
| [Range] | [0.0–17.8] * | [0.3–17.9] | [0.4–17.6] | [0.7–17.8] | |
| Time post-infection | <0.0001 b | ||||
| Months, | 0.1 (3.0 days) | 2.2 | 4.4 | 8.6 | |
| Median [IQR] | [0.0–0.2] | [1.2–2.5] | [3.5–5.3] | [7.1–10.6] | |
| Disease symptoms, n (%) | (n = 73) | 0.5682 a | |||
| Asymptomatic | 25 (34.2) | 7 (30.4) | 4 (20.0) | 8 (28.6) | |
| Mild | 23 (31.5) | 7 (30.4) | 5 (25.0) | 6 (21.4) | |
| Moderate | 7 (9.5) | 0 (0.0) | 2 (10.0) | 2 (7.1) | |
| Severe | 18 (24.3) | 9 (39.1) | 9 (45.0) | 12 (42.9) |
| Symptoms at T1 | Asymptomatic n = 25 | Mild n = 23 | Mod-sev n = 21 | MIS-C n = 4 | p-Value * |
| Gender, n (%) | 0.0250 a | ||||
| Girls | 17 (68.0) | 8 (34.8) | 7 (34.6) | 2 (50.0) | |
| Boys | 8 (32.0) | 15 (65.2) | 14 (65.4) | 2 (50.0) | |
| # vs. Mild # vs. Mod-Sev | n.s. vs. Mod-sev | ||||
| Age, years Median [IQR] | 11.9 [5.5–15.7] p = 0.0020 vs. Mild | 1.7 [0.2–10.1] | 5.9 [1.3–12.9] | 1.4 [0.5–8.4] | 0.0023 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, C.G.; Ângelo-Dias, M.; Chasqueira, M.d.J.; Brito, M.J.; Silva, T.M.; Matos, M.V.; Lopes, M.T.; Crespo, H.; Mata, M.; Borrego, L.M.; et al. SARS-CoV-2 Infection in Children: Revisiting Host–Virus Interactions Through Post-Infection Immune Profiling. Pathogens 2025, 14, 838. https://doi.org/10.3390/pathogens14090838
Martins CG, Ângelo-Dias M, Chasqueira MdJ, Brito MJ, Silva TM, Matos MV, Lopes MT, Crespo H, Mata M, Borrego LM, et al. SARS-CoV-2 Infection in Children: Revisiting Host–Virus Interactions Through Post-Infection Immune Profiling. Pathogens. 2025; 14(9):838. https://doi.org/10.3390/pathogens14090838
Chicago/Turabian StyleMartins, Catarina Gregório, Miguel Ângelo-Dias, Maria de Jesus Chasqueira, Maria João Brito, Tiago Milheiro Silva, Maria Vitória Matos, Maria Teresa Lopes, Hélio Crespo, Mariana Mata, Luís Miguel Borrego, and et al. 2025. "SARS-CoV-2 Infection in Children: Revisiting Host–Virus Interactions Through Post-Infection Immune Profiling" Pathogens 14, no. 9: 838. https://doi.org/10.3390/pathogens14090838
APA StyleMartins, C. G., Ângelo-Dias, M., Chasqueira, M. d. J., Brito, M. J., Silva, T. M., Matos, M. V., Lopes, M. T., Crespo, H., Mata, M., Borrego, L. M., & Paixão, P. (2025). SARS-CoV-2 Infection in Children: Revisiting Host–Virus Interactions Through Post-Infection Immune Profiling. Pathogens, 14(9), 838. https://doi.org/10.3390/pathogens14090838





