Canine Distemper Virus in Tigers (Panthera tigris) and Leopards (P. pardus) in Nepal
Abstract
1. Introduction
2. Materials and Methods
3. Results
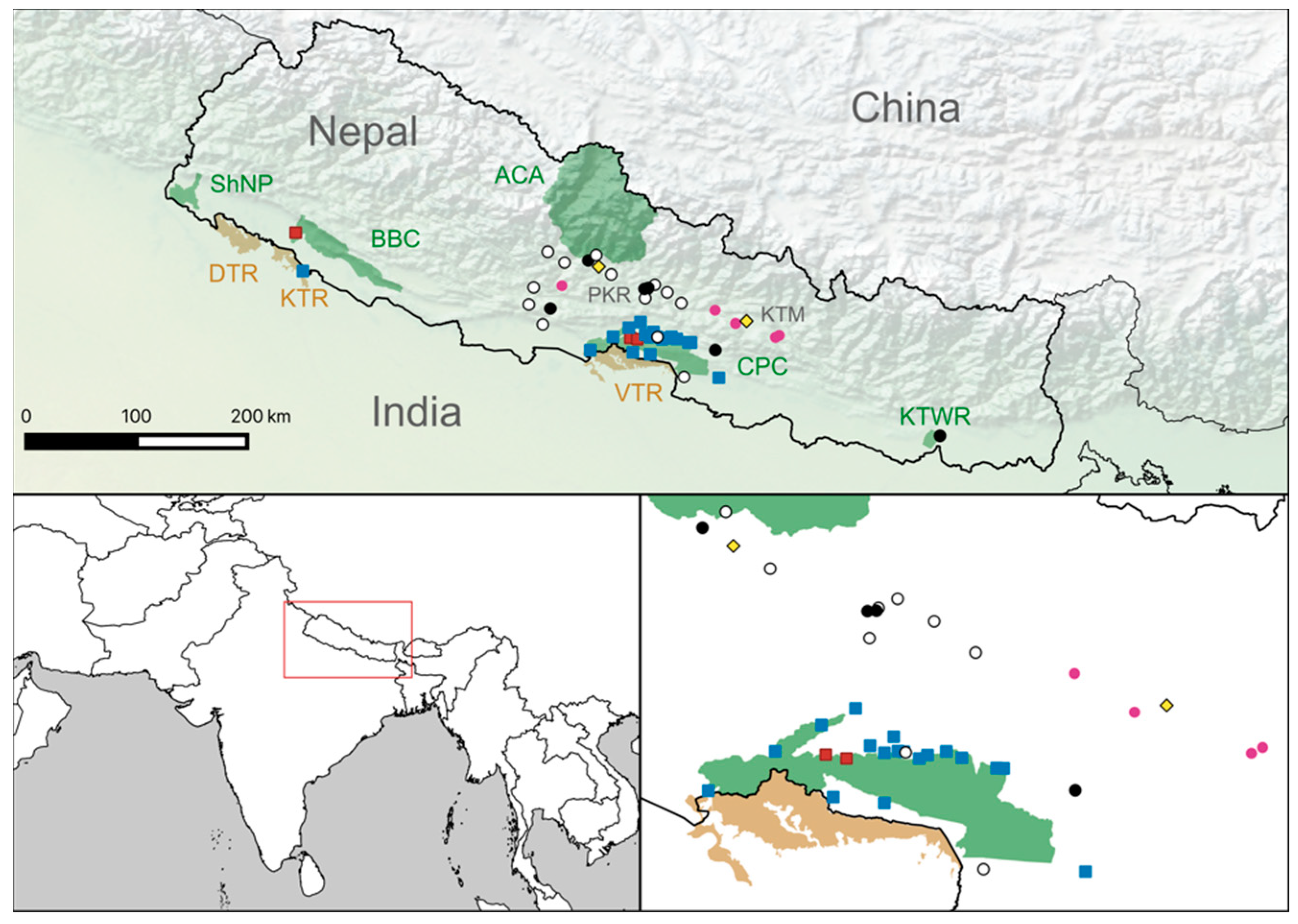
3.1. Serology Results
3.2. Clinical Findings for Animals That Tested Positive by SNT
3.3. Mortality Data
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martinez-Gutierrez, M.; Ruiz-Saenz, J. Diversity of susceptible hosts in canine distemper virus infection: A systematic review and data synthesis. BMC Vet. Res. 2016, 12, 78. [Google Scholar] [CrossRef]
- Munson, L.; Terio, K.A.; Kock, R.; Mlengeya, T.; Roelke, M.E.; Dubovi, E.; Summers, B.; Sinclair, A.R.E.; Packer, C. Climate extremes promote fatal co-infections during Canine Distemper epidemics in African lions. PLoS ONE 2008, 3, e2545. [Google Scholar] [CrossRef] [PubMed]
- Roelke-Parker, M.; Munson, L.; Packer, C.; Kock, R.; Cleaveland, S.; Carpenter, M.; O’Brien, S.J.; Pospischil, A.; Hofmann-Lehmann, R.; Lutz, H.; et al. A canine distemper virus epidemic in Serengeti lions (Panthera leo). Nature 1996, 379, 441–445. [Google Scholar] [CrossRef]
- Di Sabatino, D.; Lorusso, A.; Di Francesco, C.E.; Gentile, L.; Di Pirro, V.; Bellacicco, A.L.; Giovannini, A.; Di Francesco, G.; Marruchella, G.; Marsilio, F.; et al. Arctic lineage-Canine Distemper Virus as a cause of death in Apennine wolves (Canis lupus) in Italy. PLoS ONE 2014, 9, e82356. [Google Scholar] [CrossRef] [PubMed]
- Goller, K.V.; Fyumagwa, R.D.; Nikolin, V.; East, M.L.; Kilewo, M.; Speck, S.; Müller, T.; Matzke, M.; Wibbelt, G. Fatal canine distemper infection in a pack of African wild dogs in the Serengeti ecosystem, Tanzania. Vet. Microbiol. 2010, 146, 245–252. [Google Scholar] [CrossRef]
- Timm, S.F.; Munson, L.; Summers, B.A.; Terio, K.A.; Dubovi, E.J.; Rupprecht, C.E.; Kapil, S.; Garcelon, D.G. A suspected canine distemper epidemic as the cause of a catastrophic decline in Santa Catalina Island foxes (Urocyon littoralis catalinae). J. Wildl. Dis. 2009, 45, 333–343. [Google Scholar] [CrossRef]
- van de Bildt, M.W.G.; Kuiken, T.; Visee, A.V.; Lema, S.; Fitzjohn, T.R.; Osterhaus, A.D.M.E. Distemper outbreak and its effect on African wild dog conservation. Emerg. Infect. Dis. 2002, 8, 211–213. [Google Scholar] [CrossRef]
- Gordon, C.H.; Banyard, A.C.; Hussein, A.; Laurenson, M.K.; Malcolm, J.R.; Marino, J.; Regassa, F.; Stewart, A.E.; Fooks, A.R.; Sillero-Zubiri, C. Canine distemper in endangered Ethiopian wolves. Emerg. Infect. Dis. 2015, 21, 824–832. [Google Scholar] [CrossRef]
- Thorne, E.T.; Williams, E.S. Disease and endangered species: The black-footed ferret as a recent example. Conserv. Biol. 1988, 2, 66–74. [Google Scholar] [CrossRef]
- Robinson, H.S.; Goodrich, J.M.; Miquelle, D.G.; Miller, C.S.; Seryodkin, I.V. 4 5 Mortality of Amur tigers: The more things change, the more they stay the same. Integr. Zool. 2015, 10, 344–353. [Google Scholar] [CrossRef]
- Gilbert, M.; Miquelle, D.G.; Goodrich, J.M.; Reeve, R.; Cleaveland, S.; Matthews, L.; Joly, D.O. Estimating the potential impact of canine distemper virus on the Amur tiger population (Panthera tigris altaica) in Russia. PLoS ONE 2014, 9, e110811. [Google Scholar]
- Mulia, B.H.; Mariya, S.; Bodgener, J.; Iskandriati, D.; Liwa, S.R.; Sumampau, T.; Manansang, J.; Darusman, H.S.; Osofsky, S.A.; Techakriengkrai, N.; et al. Exposure of Wild Sumatran Tiger (Panthera tigris sumatrae) to Canine Distemper Virus. J. Wildl. Dis. 2021, 57, 464–466. [Google Scholar] [CrossRef] [PubMed]
- Rahman, D.A.; Saepuloh, U.; Santosa, Y.; Darusman, H.S.; Pinondang, I.M.R.; Kindangen, A.S.; Pertiwi, A.P.; Sari, L.; Irawan, A.; Sultan, K.; et al. Molecular diagnosis with the corresponding clinical symptoms of canine distemper virus infection in Javan leopard (Panthera pardus ssp. melas). Heliyon 2022, 8, e11341. [Google Scholar] [CrossRef] [PubMed]
- Kadam, R.G.; Karikalan, M.; Siddappa, C.M.; Mahendran, K.; Srivastava, G.; Rajak, K.K.; Bhardwaj, Y.; Varshney, R.; War, Z.A.; Singh, R.; et al. Molecular and pathological screening of canine distemper virus in Asiatic lions, tigers, leopards, snow leopards, clouded leopards, leopard cats, jungle cats, civet cats, fishing cat, and jaguar of different states, India. Infect. Genet. Evol. 2022, 98, 105211. [Google Scholar] [CrossRef] [PubMed]
- Sulikhan, N.S.; Gilbert, M.; Blidchenko, E.Y.; Naidenko, S.V.; Ivanchuk, G.V.; Gorpenchenko, T.Y.; Alshinetskiy, M.V.; Shevtsova, E.I.; Goodrich, J.M.; Lewis, J.C.M.; et al. Canine Distemper Virus in a wild Far Eastern leopard (Panthera pardus orientalis). J. Wildl. Dis. 2018, 54, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Seimon, T.A.; Miquelle, D.G.; Chang, T.Y.; Newton, A.L.; Korotkova, I.; Ivanchuk, G.; Lyubchenko, E.; Tupikov, A.; Slabe, E.; McAloose, D. Canine distemper virus: An emerging disease in wild endangered Amur tigers (Panthera tigris altaica). MBio 2013, 4, e00410–e00413. [Google Scholar] [CrossRef] [PubMed]
- Sykes, J.E. Canine and Feline Infectious Diseases, 1st ed.; W.B. Saunders: Saint Louis, MO, USA, 2014; Chapter 15—Canine Distemper Virus Infection; pp. 152–165. ISBN 9780323241946. [Google Scholar]
- Uhl, E.W.; Kelderhouse, C.; Buikstra, J.; Blick, J.P.; Bolon, B.; and Hogan, R.J. New world origin of canine distemper: Interdisciplinary insights. Int. J. Paleopathol. 2019, 24, 266–278. [Google Scholar] [CrossRef]
- Viana, M.; Cleaveland, S.; Matthiopoulos, J.; Halliday, J.; Packer, C.; Craft, M.E.; Hampson, K.; Czupryna, A.; Dobson, A.P.; Dubovi, E.J.; et al. Dynamics of a morbillivirus at the domestic-wildlife interface: Canine distemper virus in domestic dogs and lions. Proc. Natl. Acad. Sci. USA 2015, 112, 1464–1469. [Google Scholar] [CrossRef]
- Almberg, E.S.; Cross, P.C.; Smith, D.W. Persistence of canine distemper virus in the Greater Yellowstone Ecosystem’s carnivore community. Ecol. Appl. 2010, 20, 2058–2074. [Google Scholar] [CrossRef]
- Butler, J.R.A.; Linnell, J.D.C.; Morrant, D.; Athreya, D.; Lescureux, V.; McKeown, N.A. Free-Ranging Dogs and Wildlife Conservation, 1st ed.; Gompper, M.E., Ed.; Oxford University press: Oxford, UK, 2014; Chapter 5—Dog eat dog, cat eat dog; pp. 117–243. ISBN 9780191810183. [Google Scholar]
- Acosta-Jamett, G.; Chalmers, W.S.K.; Cunningham, A.A.; Cleaveland, S.; Handel, I.G.; deC Bronsvoort, B.M. Urban domestic dog populations as a source of canine distemper virus for wild carnivores in the Coquimbo region of Chile. Vet. Microbiol. 2011, 152, 247–257. [Google Scholar] [CrossRef]
- Meli, M.L.; Simmler, P.; Cattori, P.; Martínez, F.; Vargas, A.; Palomares, F.; López-Bao, J.V.; Simón, M.A.; López, G.; León-Vizcaino, L.; et al. Importance of canine distemper virus (CDV) infection in free-ranging Iberian lynxes (Lynx pardinus). Vet. Microbiol. 2010, 146, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Nayak, R.M.; Jadav, K.K.; Rajput, N.; Gupta, S.; Rokde, A.; Singh, K.P. Surveillance of major canine pathogens in feral dogs and big cats at the domestic-wildlife interface in Panna tiger reserve, India. J. Anim. Res. 2020, 10, 303–308. [Google Scholar] [CrossRef]
- Haydon, D.T.; Cleaveland, S.; Taylor, L.H.; Laurenson, M.K. Identifying reservoirs of infection: A conceptual and practical challenge. Emerg. Infect. Dis. 2002, 8, 1468–1473. [Google Scholar] [PubMed]
- Oleaga, Á.; Vázquez, C.B.; Royo, L.J.; Barral, T.D.; Bonnaire, D.; Armenteros, J.A.; Rabanal, B.; Gortázar, C.; Balseiro, A. Canine distemper virus in wildlife in south-western Europe. Transbound. Emerg. Dis. 2022, 69, e473–e485. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.; Sulikhan, N.; Uphyrkina, O.; Goncharuk, M.; Kerley, L.; Castro, E.H.; Reeve, R.; Seimon, T.; McAloose, D.; Seryodkin, I.V.; et al. Distemper, extinction, and vaccination of the Amur tiger. Proc. Natl. Acad. Sci. USA 2020, 117, 31954–31962. [Google Scholar] [CrossRef]
- Trebbien, R.; Chriel, M.; Struve, T.; Hjulsager, C.K.; Larsen, G.; Larsen, L.E. Wildlife reservoirs of canine distemper virus resulted in a major outbreak in Danish farmed mink (Neovison vison). PLoS ONE 2014, 9, e85598. [Google Scholar] [CrossRef]
- Greene, C.E.; Appel, M.J.G. Infectious Diseases in Dog and Cat, 3rd ed.; Greene, C.E., Ed.; Elsevier: St. Louis, MO, USA, 2006; pp. 25–41. ISBN 1-4160-3600-8. [Google Scholar]
- Appel, M.J.; Yates, R.A.; Foley, G.L.; Bernstein, J.J.; Santinelli, S.; Spelman, L.H.; Miller, L.D.; Arp, L.H.; Anderson, M.; Barr, M.; et al. Canine distemper epizootic in lions, tigers, and leopards in North America. J. Vet. Diagn. Invest. 1994, 6, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Konjević, D.; Sabočanec1, R.; Grabarević1, Ž.; Zurbriggen, A.; Bata, I.; Beck, A.; Kurilj, A.G.; Cvitković1, D. Canine distemper in Siberian tiger cubs from Zagreb ZOO: Case report. Acta. Veterinaria. Brno. 2011, 80, 47–50. [Google Scholar] [CrossRef]
- Nagao, Y.; Nishio, Y.; Shiomoda, H.; Tamaru, S.; Shimojima, M.; Goto, M.; Une, Y.; Sato, A.; Ikebe, Y.; Maeda, K. An outbreak of canine distemper virus in tigers (Panthera tigris): Possible transmission from wild animals to zoo animals. J. Vet. Med. Sci. 2012, 74, 699–705. [Google Scholar] [CrossRef]
- Gilbert, M.; Soutyrina, S.V.; Seryodkin, I.V.; Sulikhan, N.; Uphyrkina, O.V.; Goncharuk, M.; Matthews, L.; Cleaveland, S.; Miquelle, D.G. Canine distemper virus as a threat to wild tigers in Russia and across their range. Integr. Zool. 2015, 10, 329–343. [Google Scholar] [CrossRef]
- Myers, D.L.; Zurbriggen, A.; Lutz, H.; Pospischil, A. Distemper: Not a new disease in lions and tigers. Clin. Diagn. Lab. 1997, 4, 180–184. [Google Scholar] [CrossRef]
- Goodrich, J.M.; Quigley, K.S.; Lewis, J.C.M.; Astafiev, A.A.; Slabi, E.V.; Miquelle, D.G.; Smirnov, E.N.; Kerley, L.K.; Armstrong, D.L.; Quigley, H.B.; et al. Serosurvey of free-ranging Amur tigers in the Russian Far East. J. Wildl. Dis. 2012, 48, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Harder, T.C.; Kenter, M.; Vos, H.; Siebelink, K.; Huisman, W.; van Amerongen, G.; Orvell, C.; Barrett, T.; Appel, M.J.; Osterhaus, A.D. Canine distemper virus from diseased large felids: Biological properties and phylogenetic relationships. J. Gen. Virol. 1996, 77, 397–405. [Google Scholar] [CrossRef]
- Zhao, J.; Shi, N.; Sun, Y.; Martella, V.; Nikolin, V.; Zhu, C.; Zhang, H.; Hu, B.; Bai, X.; Yan, X. Pathogenesis of canine distemper virus in experimentally infected raccoon dogs, foxes, and minks. Antivir. Res. 2015, 122, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Böhm, M.; Thompson, H.; Weir, A.; Hasted, A.M.; Maxwell, N.S.; Herrtage, M.E. Serum antibody titres to canine parvovirus, adenovirus and distemper virus in dogs in the UK which had not been vaccinated for at least three years. Vet. Rec. 2004, 154, 457–463. [Google Scholar] [CrossRef]
- Bergmann, M.; Zablotski, Y.; Rieger, A.; Speck, S.; Truyen, U.; Hartmann, K. Comparison of four commercially available point-of-care tests to detect antibodies against canine distemper virus in dogs. Vet. J. 2021, 273, 105693. [Google Scholar] [CrossRef]
- Waner, T.; Mazar, S.; Nachmias, E.; Keren-Kornblatt, E.; Harrus, S. Evaluation of a dot ELISA kit for measuring immunoglobulin M antibodies to canine parvovirus and distemper virus. Vet. Rec. 2003, 152, 588–591. [Google Scholar] [CrossRef]
- DNPWC; DFSC. Status of tigers and prey in Nepal 2022; Ministry of Forests and Environment, Department of National Parks and Wildlife Conservation and Department of Forests and Soil Conservation: Kathmandu, Nepal, 2022.
- Goodrich, J.; Wibisono, H.; Miquelle, D.; Lynam, A.J.; Sanderson, E.; Chapman, S.; Gray, T.N.E.; Chanchani, P. Panthera tigris. 2022, The IUCN Red List of Threatened Species 2022: E.T15955A214862019. International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2022.
- Carter, N.; Pradhan, N.; Hengaju, K.; Sonawane, C.; Sage, A.H.; Grimm, V. 5 Forecasting effects of transport infrastructure on endangered tigers: A tool for conservation planning. PeerJ 2022, 17, e13472. [Google Scholar] [CrossRef]
- Thapa, K.; Malla, S.; Subba, S.A.; Thapa, G.J.; Lamichhane, B.R.; Subedi, N.; Dhakal, M.; Acharya, K.P.; Thapa, M.K.; Neupane, P.; et al. On the tiger trails: Leopard occupancy decline and leopard interaction with tigers in the forested habitat across the Terai Arc Landscape of Nepal. GECCO 2020, 25, e01412. [Google Scholar] [CrossRef]
- Stein, A.B.; Athreya, V.; Gerngross, P.; Balme, G.; Henschel, P.; Karanth, U.; Miquelle, D.; Rostro-Garcia, S.; Kamler, J.F.; Laguardia, A.; et al. Panthera pardus (amended version of 2019 assessment), 2020; The IUCN Red List of Threatened Species 2020: E.T15954A163991139; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2020. [Google Scholar]
- Jnawali, S.R.; Baral, H.S.; Lee, S.; Acharya, K.P.; Upadhyay, G.P.; Pandey, M.; Shrestha, R.; Joshi, D.; Lamichhane, B.R.; Griffiths, J.; et al. The Status of Nepal Mammals: The National Red List Series; Department of National Parks and Wildlife Conservation: Kathmandu, Nepal, 2011.
- Thapa, T. Human caused mortality in the leopard (Panthera pardus) population of Nepal. JIST 2015, 19, 155. [Google Scholar] [CrossRef]
- Paudel, P.; Acharya, K.P.; Baral, H.S.; Heinen, J.T.; Jnawali, S.R. Trends, patterns, and networks of illicit wildlife trade in Nepal: A national synthesis. Conserv. Sci. Pract. 2020, 2, e247. [Google Scholar] [CrossRef]
- Odden, M.; Wegge, P.; Fredriksen, T. Do tigers displace leopards? If so, why? Ecol. Res. 2010, 25, 875–881. [Google Scholar] [CrossRef]
- Lamichhane, B.R.; Leirs, H.; Persoon, G.A.; Subedi, N.; Dhakal, M.; Oli, B.N.; Reynaert, S.; Sluydts, V.; Pokheral, C.P.; Poudyal, L.P.; et al. Factors associated with co-occurrence of large carnivores in a human-dominated landscape. Biodivers. Conserv. 2019, 28, 1473–1491. [Google Scholar] [CrossRef]
- Daszak, P.; Cunningham, A.; Hyatt, A. Emerging infectious diseases of wildlife—Threats to biodiversity and human health. Science 2000, 21, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Logan, N.; McMonagle, E.; Drew, A.A.; Takahashi, E.; McDonald, M.; Baron, M.D.; Gilbert, M.; Cleaveland, S.; Haydon, D.T.; Hosie, M.J.; et al. Efficient generation of vesicular stomatitis virus (VSV)-pseudotypes bearing morbilliviral glycoproteins and their use in quantifying virus neutralising antibodies. Vaccine 2016, 34, 814–822. [Google Scholar] [CrossRef]
- Lorenz, R.J.; Bögel, K. Laboratory Techniques in Rabies: Methods of Calculation. Monogr. Ser. World Health Organ. 1973, 23, 321–335. [Google Scholar]
- R: CoreTeam. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: http://www.R-project.org/ (accessed on 23 June 2022).
- Wang, D.; Accatino, F.; Smith, J.L.D.; Wang, T. Contributions of distemper control and habitat expansion to the Amur leopard viability. Commun. Biol. 2022, 5, 1153. [Google Scholar] [CrossRef]
- Kubo, T.; Kagawa, Y.; Taniyama, H.; Hasegawa, A. Retrospective serological analysis of spontaneous CDV infection in 192 dogs. JVMS 2008, 70, 475–477. [Google Scholar] [CrossRef] [PubMed]
- Winters, K.; Mathes, L.E.; Krakowka, S.; Olsen, R.G. Immunoglobulin class response to canine distemper virus in gnotobiotic dogs. Vet. Immunol.; Immunopathol. 1984, 5, 209–215. [Google Scholar] [CrossRef]
- Phuentshok, Y.; Choden, K.; Alvarez Rojas, C.A.; Deplazes, P.; Wangdi, S.; Gyeltshen, K.; Rinzin, K.; Thapa, N.K.; Tenzinla, T.; Dorjee, D.; et al. Cerebral cysticercosis in a wild Bengal tiger (Panthera tigris tigris) in Bhutan: A first report in non-domestic felids. Int. J. Parasitol. Parasites Wildl. 2021, 14, 150–156. [Google Scholar] [CrossRef]
- Akin, E.Y.; Baumgartner, W.A.; Lee, J.K.; Beasley, M.J. Meningioma in a Bengal tiger (Panther tigris tigris). J. Zoo Wildl. Med. 2013, 44, 761–764. [Google Scholar] [CrossRef]
- Crook, E.K.; Carpenter, N.A. Acute lymphocytic cholangitis and liver failure in an Amur tiger (Panthera tigris altaica). J. Zoo Wildl. Med. 2014, 45, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Saqib, M.; Abbas, G.; Mughal, M.N. Successful management of ivermectin-induced blindness in an African lion (Panthera leo) by intravenous administration of a lipid emulsion. BMC Vet. Res. 2015, 11, 287. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, R.; Nakamura, S.; Hori, H.; Kato, Y.; Une, Y. Purulent meningoventriculitis caused by Streptococcus equi subspecies zooepidemicus in a Snow Leopard (Panthera uncia). J. Comp. Pathol. 2012, 147, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Elia, G.; Camero, M.; Losurdo, M.; Lucente, M.S.; Larocca, V.; Martella, V.; Decaro, N.; Buonavoglia, C. Virological and serological findings in dogs with naturally occurring distemper. J. Virol. Methods 2015, 213, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Elia, G.; Decaro, N.; Martella, V.; Cirone, F.; Lucente, M.S.; Lorusso, E.; Di Trani, L.; Buonavoglia, C. Detection of canine distemper virus in dogs by real-time RT-PCR. J. Virol. Methods. 2006, 136, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Koirala, R.; Aryal, A.; Amiot, C.; Adhikari, B.; Karmacharya, D.; Raubenheimer, D. Genetic identification of carnivore scat: Implication of dietary information for human-carnivore conflict in the Annapurna Conservation Area, Nepal. Zool. Ecol. 2012, 22, 137–143. [Google Scholar] [CrossRef]
- Bhattarai, B.P.; Kindlmann, P. Interactions between Bengal tiger (Panthera tigris) and leopard (Panthera pardus): Implications for their conservation. Biodivers. Conserv. 2012, 21, 2075–2094. [Google Scholar] [CrossRef]
- Lovari, S.; Pokheral, C.P.; Jnawali, S.R.; Fusani, L.; Ferretti, F. Coexistence of the tiger and the common leopard in a prey-rich area: The role of prey partitioning. J. Zool. 2015, 295, 122–131. [Google Scholar] [CrossRef]
- Kandel, S. Panthera pardus fusca (Family: Felidae) diet composition from Lamjung, Nepal. Environ. Ecol. Res. 2019, 7, 253–258. [Google Scholar] [CrossRef]
- Kandel, S.R.; Lamichhane, B.R.; Subedi, N. Leopard (Panthera pardus) density and diet in a forest corridor of Terai: Implications for conservation and conflict management. Wildl. Res. 2020, 47, 460–467. [Google Scholar] [CrossRef]
- Lamichhane, S.; Jha, B. Prey selection by Bengal Tiger Panthera tigris tigris (Mammalia: Carnivora: Felidae) of Chitwan National Park, Nepal. J. Threat. Taxa. 2015, 7, 8081–8088. [Google Scholar] [CrossRef]
- Wegge, P.; Yadav, S.K.; Lamichhane, B.R. Are corridors good for tigers (Panthera tigris) but bad for people? An assessment of the Khata corridor in lowland Nepal. Oryx 2018, 52, 35–45. [Google Scholar] [CrossRef]
- Bhandari, S.; Chalise, M.K.; Pokharel, C.P. Diet of Bengal tigers (Panthera tigris tigris) in Chitwan National Park, Nepal. Eur. J. Ecol. 2017, 3, 80–84. [Google Scholar] [CrossRef]
- Baniya, R.K.; Baniya, C.B.; Mou, P.; Ge, J. Prey selection by tiger (Panthera tigris tigris) in Shuklaphanta Wildlife Reserve Nepal. Int. J. Sci. 2017, 3, 90–99. [Google Scholar]
- Upadhyaya, S.K.; Musters, C.J.M.; Lamichhane, B.R.; de Snoo, G.R.; Thapa, P.; Dhakal, M.; Karmacharya, D.; Shrestha, P.M.; de Iongh, H.H. An insight into the diet and prey preference of tigers in Bardia National Park, Nepal. Trop. Conserv. Sci. 2018, 11, 1940082918799476. [Google Scholar] [CrossRef]
- Lamichhane, S.; Thanet, D.R.; Regmi, P.R.; Maharjan, A.; Lamichhane, B.R. Dietary composition and prey preference of Royal Bengal Tiger (Panthera tigris tigris, Linnaeus 1758) of Parsa National Park, Nepal. Eur. J. Ecol. 2022, 8, 34–38. [Google Scholar] [CrossRef]
- Bhattarai, B.; Kindlmann, P. Human Disturbance is the Major Determinant of the Habitat and Prey Preference of the Bengal Tiger (Panthera tigris tigris) in the Chitwan National Park, Nepal. Eur. J. Ecol. 2018, 4, 13–21. [Google Scholar] [CrossRef]
- Ng, D.; Carver, S.; Gotame, M.; Karmasharya, D.; Karmacharya, D.; Pradhan, S.M.; Rana, A.N.; Johnson, C.N. Canine distemper in Nepal’s Annapurna Conservation Area—Implications of dog husbandry and human behaviour for wildlife disease. PLoS ONE 2019, 14, e0220874. [Google Scholar] [CrossRef]
- Sadaula, A.; Dev Joshi, J.; Lamichhane, B.R.; Gairhe, K.P.; Subedi, N.; Pokheral, C.P.; Thapaliya, S.; Pandey, G.; Riijal, K.R.; Pandey, P. Seroprevalence of Canine Distemper and Canine Parvovirus Among Domestic Dogs in Buffer Zone of Chitwan National Park, Nepal. SSRN J. 2022, preprint. [Google Scholar] [CrossRef]
- Harihar, A.; Pandav, B.; Goyal, S.P. Responses of leopard (Panthera pardus) to the recovery of a tiger (Panthera tigris) population. J. Appl. Ecol. 2011, 48, 806–814. [Google Scholar] [CrossRef]
- Athreya, V.; Odden, M.; Linnell, J.D.C.; Krishnaswamy, J.; Karanth, U.K. A cat among the dogs: Leopard Panthera pardus diet in a human-dominated landscape in western Maharashtra, India. Oryx 2016, 50, 156–162. [Google Scholar] [CrossRef]
- Kumbhojkar, S.; Yosef, R.; Kosicki, J.Z.; Kwiatkowska, P.K.; Tryjanowski, P. Dependence of the leopard Panthera pardus fusca in Jaipur, India, on domestic animals. Oryx 2021, 55, 692–698. [Google Scholar] [CrossRef]
- Thapa, K.; Manandhar, S.; Bista, M.; Shakya, J.; Sah, G.; Dhakal, M.; Sharma, N.; Llewellyn, B.; Wultsch, C.; Waits, L.P.; et al. Assessment of genetic diversity, population structure, and gene flow of tigers (Panthera tigris tigris) across Nepal’s Terai Arc Landscape. PLoS ONE 2018, 13, e0193495. [Google Scholar] [CrossRef]
- Lamichhane, B.R.; Lamichhane, S.; Regmi, R.; Dhungana, M.; Thapa, S.K.; Prasai, A.; Gurung, A.; Bhattarai, S.; Paudel, R.P.; Subedi, N. Leopard (Panthera pardus) occupancy in the Chure range of Nepal. Ecol. Evol. 2021, 11, 13641–13660. [Google Scholar] [CrossRef]

| ID | Year | Titer | Healthy | Underweight | Ocular Signs | Respiratory Signs | Gastro-Intestinal Signs | Thickened Pads | Neurological Signs | Mortality | Recovery |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pt 12 | 2016 | 32 | ✓ | ✓ | ✓ | ✓ | |||||
| Pt 23 | 2019 | 128 | ✓ | ||||||||
| Pt26 | 2021 | 91 | ✓ | ||||||||
| Pp5 | 2017 | 362 | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| Pp7 | 2019 | 23,168 | ✓ | ✓ | ✓ | ||||||
| Pp8 | 2019 | 23 | ✓ | ||||||||
| Pp11 | 2019 | 181 | ✓ | ||||||||
| Pp12 | 2019 | 128 | ✓ | ||||||||
| Pp17 | 2020 | 2896 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bodgener, J.; Sadaula, A.; Thapa, P.J.; Shrestha, B.K.; Gairhe, K.P.; Subedi, S.; Rijal, K.R.; Pandey, P.; Joshi, J.D.; Kandel, P.; et al. Canine Distemper Virus in Tigers (Panthera tigris) and Leopards (P. pardus) in Nepal. Pathogens 2023, 12, 203. https://doi.org/10.3390/pathogens12020203
Bodgener J, Sadaula A, Thapa PJ, Shrestha BK, Gairhe KP, Subedi S, Rijal KR, Pandey P, Joshi JD, Kandel P, et al. Canine Distemper Virus in Tigers (Panthera tigris) and Leopards (P. pardus) in Nepal. Pathogens. 2023; 12(2):203. https://doi.org/10.3390/pathogens12020203
Chicago/Turabian StyleBodgener, Jessica, Amir Sadaula, Parbat Jung Thapa, Bhijay Kumar Shrestha, Kamal Prasad Gairhe, Suraj Subedi, Kiran Raj Rijal, Purushotam Pandey, Janardan Dev Joshi, Prakriti Kandel, and et al. 2023. "Canine Distemper Virus in Tigers (Panthera tigris) and Leopards (P. pardus) in Nepal" Pathogens 12, no. 2: 203. https://doi.org/10.3390/pathogens12020203
APA StyleBodgener, J., Sadaula, A., Thapa, P. J., Shrestha, B. K., Gairhe, K. P., Subedi, S., Rijal, K. R., Pandey, P., Joshi, J. D., Kandel, P., Lamichane, B. R., Pokheral, C. P., Subedi, N., Kandel, R. C., Luitel, H., Techakriengkrai, N., & Gilbert, M. (2023). Canine Distemper Virus in Tigers (Panthera tigris) and Leopards (P. pardus) in Nepal. Pathogens, 12(2), 203. https://doi.org/10.3390/pathogens12020203






