Limiting the Spread of Multidrug-Resistant Bacteria in Low-to-Middle-Income Countries: One Size Does Not Fit All
Abstract
1. General Introduction: The Threat of MDROs and Necessity of Limiting Their Spread
2. Transmission Pathways of MDROs in Hospitals and at the Community Level
3. Host Risk Factors for MDRO Acquisition
4. International Policies to Limit the Spread of MDROs
4.1. Surveillance Cultures
4.2. Isolation in Single-Bed Rooms
4.3. Hand Hygiene
4.4. Additional Contact Precautions
4.5. Cohorting
4.6. Antimicrobial Stewardship Programs
5. Constraints on the Application of International Protocols in Low-to-Middle-Income Countries
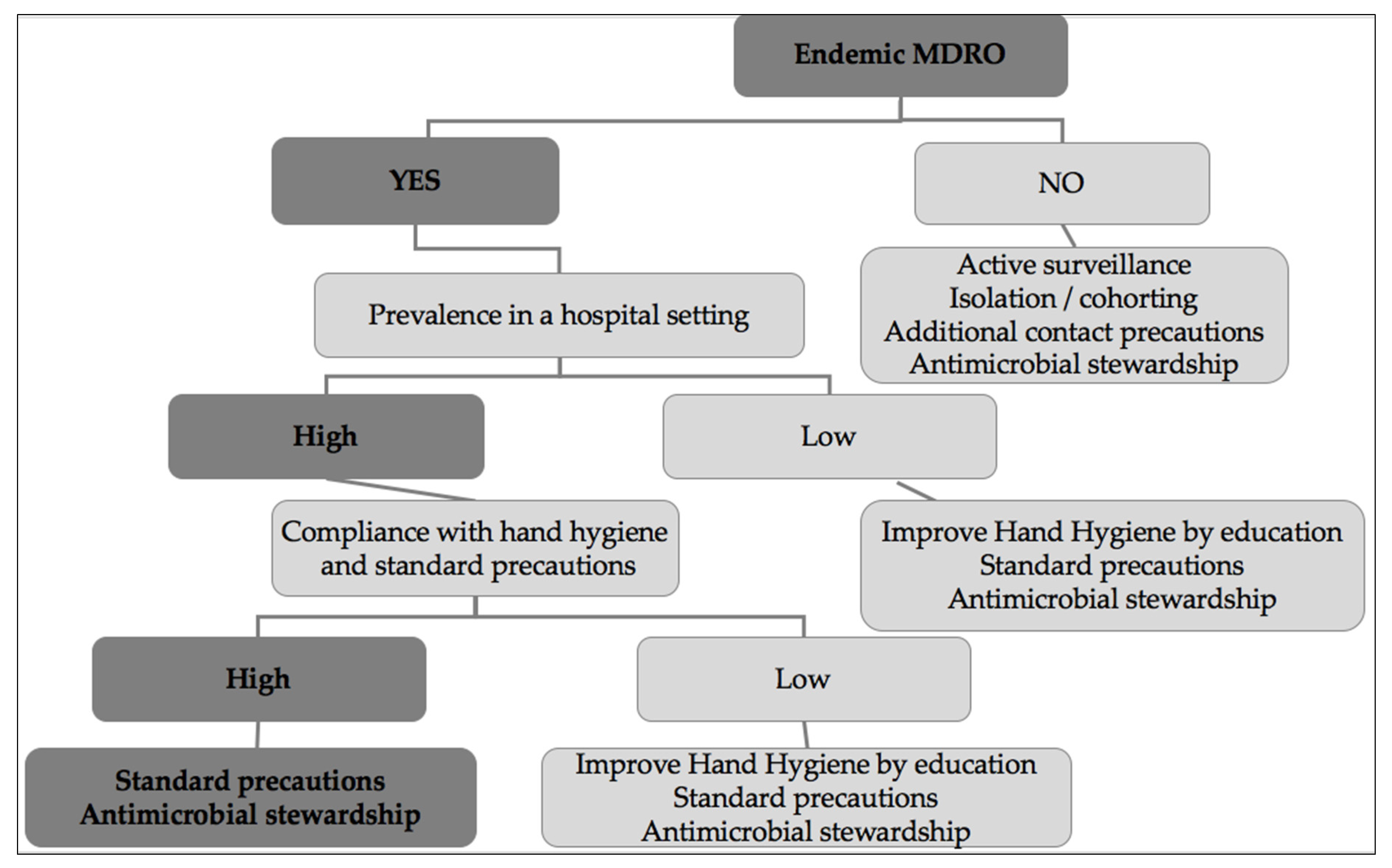
6. Possible Targeted Strategies Adapted to the Means of Low-to-Middle Income Countries
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Global Antimicrobial Resistance and Use Surveillance System (GLASS) Report: 2021. Available online: https://www.who.int/publications-detail-redirect/9789240027336 (accessed on 11 July 2021).
- Biggest Threats and Data|Antibiotic/Antimicrobial Resistance|CDC. Available online: https://www.cdc.gov/drugresistance/biggest_threats.html (accessed on 22 July 2019).
- Temkin, E.; Adler, A.; Lerner, A.; Carmeli, Y. Carbapenem-resistant Enterobacteriaceae: Biology, epidemiology, and management. Ann. N. Y. Acad. Sci. 2014, 1323, 22–42. [Google Scholar] [CrossRef]
- Weiner-Lastinger, L.M.; Abner, S.; Edwards, J.R.; Kallen, A.J.; Karlsson, M.; Magill, S.S.; Pollock, D.; See, I.; Soe, M.M.; Walters, M.S.; et al. Antimicrobial-resistant pathogens associated with adult healthcare-associated infections: Summary of data reported to the National Healthcare Safety Network, 2015–2017. Infect. Control Hosp. Epidemiol. 2020, 41, 1–18. [Google Scholar] [CrossRef]
- Lautenbach, E.; Patel, J.B.; Bilker, W.B.; Edelstein, P.H.; Fishman, N.O. Extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae: Risk factors for infection and impact of resistance on outcomes. Clin. Infect. Dis. 2001, 32, 1162–1171. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou-Olivgeris, M.; Drougka, E.; Fligou, F.; Kolonitsiou, F.; Liakopoulos, A.; Dodou, V.; Anastassiou, E.D.; Petinaki, E.; Marangos, M.; Filos, K.S.; et al. Risk factors for enterococcal infection and colonization by vancomycin-resistant enterococci in critically ill patients. Infection 2014, 42, 1013–1022. [Google Scholar] [CrossRef]
- Chiotos, K.; Tamma, P.D.; Flett, K.B.; Naumann, M.; Karandikar, M.V.; Bilker, W.B.; Zaoutis, T.; Han, J.H. Multicenter Study of the Risk Factors for Colonization or Infection with Carbapenem-Resistant Enterobacteriaceae in Children. Antimicrob. Agents Chemother. 2017, 61, e01440-17. [Google Scholar] [CrossRef] [PubMed]
- Yigit, H.; Queenan, A.M.; Anderson, G.J.; Domenech-Sanchez, A.; Biddle, J.W.; Steward, C.D.; Alberti, S.; Bush, K.; Tenover, F.C. Novel carbapenem-hydrolyzing beta-lactamase, KPC-1, from a carbapenem-resistant strain of Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2001, 45, 1151–1161. [Google Scholar] [CrossRef]
- Poirel, L.; Potron, A.; Nordmann, P. OXA-48-like carbapenemases: The phantom menace. J. Antimicrob. Chemother. 2012, 67, 1597–1606. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P. Carbapenemase-producing Enterobacteriaceae: Overview of a major public health challenge. Med. Mal. Infect. 2014, 44, 51–56. [Google Scholar] [CrossRef]
- Albiger, B.; Glasner, C.; Struelens, M.J.; Grundmann, H.; Monnet, D.L. European Survey of Carbapenemase-Producing Enterobacteriaceae (EuSCAPE) working group Carbapenemase-producing Enterobacteriaceae in Europe: Assessment by national experts from 38 countries, May 2015. Eurosurveillance 2015, 20. [Google Scholar] [CrossRef] [PubMed]
- Zowawi, H.M.; Balkhy, H.H.; Walsh, T.R.; Paterson, D.L. β-Lactamase production in key gram-negative pathogen isolates from the Arabian Peninsula. Clin. Microbiol. Rev. 2013, 26, 361–380. [Google Scholar] [CrossRef]
- Cattoir, V.; Leclercq, R. Twenty-five years of shared life with vancomycin-resistant enterococci: Is it time to divorce? J. Antimicrob. Chemother. 2013, 68, 731–742. [Google Scholar] [CrossRef]
- Data Collection and Analysis. European Centre for Disease Prevention and Control. Available online: https://www.ecdc.europa.eu/en/about-us/networks/disease-networks-and-laboratory-networks/ears-net-data (accessed on 14 October 2021).
- Guzman Prieto, A.M.; van Schaik, W.; Rogers, M.R.C.; Coque, T.M.; Baquero, F.; Corander, J.; Willems, R.J.L. Global Emergence and Dissemination of Enterococci as Nosocomial Pathogens: Attack of the Clones? Front. Microbiol. 2016, 7, 788. [Google Scholar] [CrossRef]
- Zhen, X.; Lundborg, C.S.; Sun, X.; Hu, X.; Dong, H. The Clinical and Economic Impact of Antibiotic Resistance in China: A Systematic Review and Meta-Analysis. Antibiot. Basel Switz. 2019, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.J.; Rogawski, E.; Thom, K.A.; Johnson, J.K.; Perencevich, E.N.; Shardell, M.; Leekha, S.; Harris, A.D. Transfer of multidrug-resistant bacteria to healthcare workers’ gloves and gowns after patient contact increases with environmental contamination. Crit. Care Med. 2012, 40, 1045–1051. [Google Scholar] [CrossRef]
- Creamer, E.; Shore, A.C.; Rossney, A.S.; Dolan, A.; Sherlock, O.; Fitzgerald-Hughes, D.; Sullivan, D.J.; Kinnevey, P.M.; O’Lorcain, P.; Cunney, R.; et al. Transmission of endemic ST22-MRSA-IV on four acute hospital wards investigated using a combination of spa, dru and pulsed-field gel electrophoresis typing. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 3151–3161. [Google Scholar] [CrossRef] [PubMed]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Tabah, A.; Koulenti, D.; Laupland, K.; Misset, B.; Valles, J.; Bruzzi de Carvalho, F.; Paiva, J.A.; Cakar, N.; Ma, X.; Eggimann, P.; et al. Characteristics and determinants of outcome of hospital-acquired bloodstream infections in intensive care units: The EUROBACT International Cohort Study. Intensive Care Med. 2012, 38, 1930–1945. [Google Scholar] [CrossRef] [PubMed]
- Laxminarayan, R.; Matsoso, P.; Pant, S.; Brower, C.; Røttingen, J.-A.; Klugman, K.; Davies, S. Access to effective antimicrobials: A worldwide challenge. Lancet 2016, 387, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Allegranzi, B.; Bagheri Nejad, S.; Combescure, C.; Graafmans, W.; Attar, H.; Donaldson, L.; Pittet, D. Burden of endemic health-care-associated infection in developing countries: Systematic review and meta-analysis. Lancet 2011, 377, 228–241. [Google Scholar] [CrossRef]
- Ayobami, O.; Brinkwirth, S.; Eckmanns, T.; Markwart, R. Antibiotic resistance in hospital-acquired ESKAPE-E infections in low- and lower-middle-income countries: A systematic review and meta-analysis. Emerg. Microbes Infect. 2022, 11, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Saharman, Y.R.; Karuniawati, A.; Severin, J.A.; Verbrugh, H.A. Infections and antimicrobial resistance in intensive care units in lower-middle income countries: A scoping review. Antimicrob. Resist. Infect. Control 2021, 10, 22. [Google Scholar] [CrossRef]
- Schwaber, M.J.; Carmeli, Y. An ongoing national intervention to contain the spread of carbapenem-resistant enterobacteriaceae. Clin. Infect. Dis. 2014, 58, 697–703. [Google Scholar] [CrossRef]
- Fournier, S.; Desenfant, L.; Monteil, C.; Nion-Huang, M.; Richard, C.; Jarlier, V. Efficiency of different control measures for preventing carbapenemase-producing enterobacteria and glycopeptide-resistant Enterococcus faecium outbreaks: A 6-year prospective study in a French multihospital institution, January 2010 to December 2015. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Woerther, P.-L.; Burdet, C.; Chachaty, E.; Andremont, A. Trends in human fecal carriage of extended-spectrum β-lactamases in the community: Toward the globalization of CTX-M. Clin. Microbiol. Rev. 2013, 26, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Guet-Revillet, H.; Le Monnier, A.; Breton, N.; Descamps, P.; Lecuyer, H.; Alaabouche, I.; Bureau, C.; Nassif, X.; Zahar, J.-R. Environmental contamination with extended-spectrum β-lactamases: Is there any difference between Escherichia coli and Klebsiella spp? Am. J. Infect. Control 2012, 40, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Prospero, E.; Barbadoro, P.; Marigliano, A.; D’Errico, M.M. Health care workers, immunization and safety issues: Reflections from Italy. Am. J. Infect. Control 2013, 41, 664. [Google Scholar] [CrossRef]
- Eurosurveillance Editorial Team. ECDC’s latest publications. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef]
- Jones, K.; Bradshaw, S.B. Biofilm formation by the enterobacteriaceae: A comparison between salmonella enteritidis, Escherichia coli and a nitrogen-fixing strain of Klebsiella pneumoniae. J. Appl. Bacteriol. 1996, 80, 458–464. [Google Scholar] [CrossRef]
- Lerner, A.; Adler, A.; Abu-Hanna, J.; Meitus, I.; Navon-Venezia, S.; Carmeli, Y. Environmental contamination by carbapenem-resistant Enterobacteriaceae. J. Clin. Microbiol. 2013, 51, 177–181. [Google Scholar] [CrossRef]
- Weber, D.J.; Anderson, D.; Rutala, W.A. The role of the surface environment in healthcare-associated infections. Curr. Opin. Infect. Dis. 2013, 26, 338–344. [Google Scholar] [CrossRef]
- Eckstein, B.C.; Adams, D.A.; Eckstein, E.C.; Rao, A.; Sethi, A.K.; Yadavalli, G.K.; Donskey, C.J. Reduction of Clostridium Difficile and vancomycin-resistant Enterococcus contamination of environmental surfaces after an intervention to improve cleaning methods. BMC Infect. Dis. 2007, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Drees, M.; Snydman, D.R.; Schmid, C.H.; Barefoot, L.; Hansjosten, K.; Vue, P.M.; Cronin, M.; Nasraway, S.A.; Golan, Y. Prior environmental contamination increases the risk of acquisition of vancomycin-resistant enterococci. Clin. Infect. Dis. 2008, 46, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Nseir, S.; Blazejewski, C.; Lubret, R.; Wallet, F.; Courcol, R.; Durocher, A. Risk of acquiring multidrug-resistant Gram-negative bacilli from prior room occupants in the intensive care unit. Clin. Microbiol. Infect. 2011, 17, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.S.; Datta, R.; Platt, R. Risk of acquiring antibiotic-resistant bacteria from prior room occupants. Arch. Intern. Med. 2006, 166, 1945–1951. [Google Scholar] [CrossRef]
- Lerner, A.; Adler, A.; Abu-Hanna, J.; Cohen Percia, S.; Kazma Matalon, M.; Carmeli, Y. Spread of KPC-producing carbapenem-resistant Enterobacteriaceae: The importance of super-spreaders and rectal KPC concentration. Clin. Microbiol. Infect. 2015, 21, 470.e1–470.e7. [Google Scholar] [CrossRef]
- Boyce, J.M.; Havill, N.L.; Otter, J.A.; Adams, N.M.T. Widespread environmental contamination associated with patients with diarrhea and methicillin-resistant Staphylococcus aureus colonization of the gastrointestinal tract. Infect. Control Hosp. Epidemiol. 2007, 28, 1142–1147. [Google Scholar] [CrossRef]
- Donskey, C.J.; Chowdhry, T.K.; Hecker, M.T.; Hoyen, C.K.; Hanrahan, J.A.; Hujer, A.M.; Hutton-Thomas, R.A.; Whalen, C.C.; Bonomo, R.A.; Rice, L.B. Effect of antibiotic therapy on the density of vancomycin-resistant enterococci in the stool of colonized patients. N. Engl. J. Med. 2000, 343, 1925–1932. [Google Scholar] [CrossRef]
- Bonten, M.J.; Hayden, M.K.; Nathan, C.; van Voorhis, J.; Matushek, M.; Slaughter, S.; Rice, T.; Weinstein, R.A. Epidemiology of colonisation of patients and environment with vancomycin-resistant enterococci. Lancet 1996, 348, 1615–1619. [Google Scholar] [CrossRef]
- Bonten, M.J.M.; Willems, R.J. Vancomycin-resistant enterococcus-chronicle of a foretold problem. Ned. Tijdschr. Geneeskd. 2012, 156, A5233. [Google Scholar]
- Saliba, R.; Ghelfenstein-Ferreira, T.; Lomont, A.; Pilmis, B.; Carbonnelle, E.; Seytre, D.; Nasser-Ayoub, E.; Zahar, J.-R.; Karam-Sarkis, D. Risk factors for the environmental spread of different multidrug-resistant organisms: A prospective cohort study. J. Hosp. Infect. 2021, 111, 155–161. [Google Scholar] [CrossRef]
- Blanco, N.; Pineles, L.; Lydecker, A.D.; Johnson, J.K.; Sorkin, J.D.; Morgan, D.J.; VA Gown and Glove Investigators; Roghmann, M.-C. Transmission of Resistant Gram-Negative Bacteria to Health Care Worker Gowns and Gloves during Care of Nursing Home Residents in Veterans Affairs Community Living Centers. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Ajao, A.O.; Harris, A.D.; Roghmann, M.-C.; Johnson, J.K.; Zhan, M.; McGregor, J.C.; Furuno, J.P. Systematic review of measurement and adjustment for colonization pressure in studies of methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, and clostridium difficile acquisition. Infect. Control Hosp. Epidemiol. 2011, 32, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Bonten, M.J.; Slaughter, S.; Ambergen, A.W.; Hayden, M.K.; van Voorhis, J.; Nathan, C.; Weinstein, R.A. The role of “colonization pressure” in the spread of vancomycin-resistant enterococci: An important infection control variable. Arch. Intern. Med. 1998, 158, 1127–1132. [Google Scholar] [CrossRef]
- Merrer, J.; Santoli, F.; Appéré de Vecchi, C.; Tran, B.; De Jonghe, B.; Outin, H. “Colonization pressure” and risk of acquisition of methicillin-resistant Staphylococcus aureus in a medical intensive care unit. Infect. Control Hosp. Epidemiol. 2000, 21, 718–723. [Google Scholar] [CrossRef]
- DalBen, M.F.; Basso, M.; Garcia, C.P.; Costa, S.F.; Toscano, C.M.; Jarvis, W.R.; Lobo, R.D.; Oliveira, M.S.; Levin, A.S. Colonization pressure as a risk factor for colonization by multiresistant Acinetobacter spp and carbapenem-resistant Pseudomonas aeruginosa in an intensive care unit. Clin. Sao Paulo Braz. 2013, 68, 1128–1133. [Google Scholar] [CrossRef]
- Williams, V.R.; Callery, S.; Vearncombe, M.; Simor, A.E. The role of colonization pressure in nosocomial transmission of methicillin-resistant Staphylococcus aureus. Am. J. Infect. Control 2009, 37, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, M.; Sharma, S.; Poliansky Blash, S.; Patel, G.; Banach, D.B.; Phillips, M.; LaBombardi, V.; Anderson, K.F.; Kitchel, B.; Srinivasan, A.; et al. Prevalence and risk factors for acquisition of carbapenem-resistant Enterobacteriaceae in the setting of endemicity. Infect. Control Hosp. Epidemiol. 2013, 34, 809–817. [Google Scholar] [CrossRef]
- Torres-Gonzalez, P.; Cervera-Hernandez, M.E.; Niembro-Ortega, M.D.; Leal-Vega, F.; Cruz-Hervert, L.P.; García-García, L.; Galindo-Fraga, A.; Martinez-Gamboa, A.; Bobadilla-Del Valle, M.; Sifuentes-Osornio, J.; et al. Factors Associated to Prevalence and Incidence of Carbapenem-Resistant Enterobacteriaceae Fecal Carriage: A Cohort Study in a Mexican Tertiary Care Hospital. PLoS ONE 2015, 10, e0139883. [Google Scholar] [CrossRef]
- Papadimitriou-Olivgeris, M.; Christofidou, M.; Fligou, F.; Bartzavali, C.; Vrettos, T.; Filos, K.S.; Marangos, M.; Anastassiou, E.D. The role of colonization pressure in the dissemination of colistin or tigecycline resistant KPC-producing Klebsiella pneumoniae in critically ill patients. Infection 2014, 42, 883–890. [Google Scholar] [CrossRef]
- DalBen, M.F. Transmission-Based Precautions for Multidrug-Resistant Organisms: What to Prioritize When Resources Are Limited. Curr. Treat. Options Infect. Dis. 2018, 10, 40–47. [Google Scholar] [CrossRef]
- Kim, S.; Covington, A.; Pamer, E.G. The intestinal microbiota: Antibiotics, colonization resistance, and enteric pathogens. Immunol. Rev. 2017, 279, 90–105. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef]
- Van der Waaij, D.; Berghuis-de Vries, J.M.; Lekkerkerk-van der Wees, J.E.C. Colonization resistance of the digestive tract in conventional and antibiotic-treated mice. J. Hyg. 1971, 69, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Stecher, B. The Roles of Inflammation, Nutrient Availability and the Commensal Microbiota in Enteric Pathogen Infection. Microbiol. Spectr. 2015, 3, 297–320. [Google Scholar] [CrossRef] [PubMed]
- Le Guern, R.; Stabler, S.; Gosset, P.; Pichavant, M.; Grandjean, T.; Faure, E.; Karaca, Y.; Faure, K.; Kipnis, E.; Dessein, R. Colonisation resistance against multidrug-resistant bacteria: A narrative review. J. Hosp. Infect. 2021, 118, 48–58. [Google Scholar] [CrossRef]
- Keith, J.W.; Pamer, E.G. Enlisting commensal microbes to resist antibiotic-resistant pathogens. J. Exp. Med. 2019, 216, 10–19. [Google Scholar] [CrossRef]
- Ruppé, E.; Burdet, C.; Grall, N.; de Lastours, V.; Lescure, F.-X.; Andremont, A.; Armand-Lefèvre, L. Impact of antibiotics on the intestinal microbiota needs to be re-defined to optimize antibiotic usage. Clin. Microbiol. Infect. 2018, 24, 3–5. [Google Scholar] [CrossRef]
- Dethlefsen, L.; Huse, S.; Sogin, M.L.; Relman, D.A. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol. 2008, 6, e280. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; Relman, D.A. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4554–4561. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.C.; Buckley, A.M.; Spittal, W.; Ewin, D.; Clark, E.; Altringham, J.; Bentley, K.; Moura, I.B.; Wilcox, M.H.; Woodford, N.; et al. The effect of intestinal microbiota dysbiosis on growth and detection of carbapenemase-producing Enterobacterales within an in vitro gut model. J. Hosp. Infect. 2021, 113, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ruppé, E.; Armand-Lefèvre, L.; Estellat, C.; Consigny, P.-H.; El Mniai, A.; Boussadia, Y.; Goujon, C.; Ralaimazava, P.; Campa, P.; Girard, P.-M.; et al. High Rate of Acquisition but Short Duration of Carriage of Multidrug-Resistant Enterobacteriaceae after Travel to the Tropics. Clin. Infect. Dis. 2015, 61, 593–600. [Google Scholar] [CrossRef]
- Savard, P.; Carroll, K.C.; Wilson, L.E.; Perl, T.M. The challenges of carbapenemase-producing Enterobacteriaceae and infection prevention: Protecting patients in the chaos. Infect. Control Hosp. Epidemiol. 2013, 34, 730–739. [Google Scholar] [CrossRef]
- French, C.E.; Coope, C.; Conway, L.; Higgins, J.P.T.; McCulloch, J.; Okoli, G.; Patel, B.C.; Oliver, I. Control of carbapenemase-producing Enterobacteriaceae outbreaks in acute settings: An evidence review. J. Hosp. Infect. 2017, 95, 3–45. [Google Scholar] [CrossRef]
- Systematic Review of the Effectiveness of Infection Control Measures to Prevent the Transmission of Carbapenemase-Producing Enterobacteriaceae through Cross-Border Transfer of Patients. European Centre for Disease Prevention and Control. 2014. Available online: https://www.ecdc.europa.eu/en/publications-data/systematic-review-effectiveness-infection-control-measures-prevent-transmission (accessed on 30 October 2021).
- Debby, B.D.; Ganor, O.; Yasmin, M.; David, L.; Nathan, K.; Ilana, T.; Dalit, S.; Smollan, G.; Galia, R. Epidemiology of carbapenem resistant Klebsiella pneumoniae colonization in an intensive care unit. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1811–1817. [Google Scholar] [CrossRef]
- Kurup, A.; Chlebicka, N.; Tan, K.Y.; Chen, E.X.; Oon, L.; Ling, T.A.; Ling, M.L.; Hong, J.L.G. Active surveillance testing and decontamination strategies in intensive care units to reduce methicillin-resistant Staphylococcus aureus infections. Am. J. Infect. Control 2010, 38, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Magiorakos, A.P.; Burns, K.; Rodríguez Baño, J.; Borg, M.; Daikos, G.; Dumpis, U.; Lucet, J.C.; Moro, M.L.; Tacconelli, E.; Simonsen, G.S.; et al. Infection prevention and control measures and tools for the prevention of entry of carbapenem-resistant Enterobacteriaceae into healthcare settings: Guidance from the European Centre for Disease Prevention and Control. Antimicrob. Resist. Infect. Control 2017, 6, 113. [Google Scholar] [CrossRef]
- Huijbers, P.M.C.; Blaak, H.; de Jong, M.C.M.; Graat, E.A.M.; Vandenbroucke-Grauls, C.M.J.E.; de Roda Husman, A.M. Role of the Environment in the Transmission of Antimicrobial Resistance to Humans: A Review. Environ. Sci. Technol. 2015, 49, 11993–12004. [Google Scholar] [CrossRef]
- Ostholm-Balkhed, A.; Tärnberg, M.; Nilsson, M.; Nilsson, L.E.; Hanberger, H.; Hällgren, A.; Travel Study Group of Southeast Sweden. Travel-associated faecal colonization with ESBL-producing Enterobacteriaceae: Incidence and risk factors. J. Antimicrob. Chemother. 2013, 68, 2144–2153. [Google Scholar] [CrossRef]
- Lucet, J.-C.; Paoletti, X.; Demontpion, C.; Degrave, M.; Vanjak, D.; Vincent, C.; Andremont, A.; Jarlier, V.; Mentré, F.; Nicolas-Chanoine, M.-H.; et al. Carriage of methicillin-resistant Staphylococcus aureus in home care settings: Prevalence, duration, and transmission to household members. Arch. Intern. Med. 2009, 169, 1372–1378. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cattoen, C. Persistance du portage de bactéries multirésistantes après la réanimation. Réanimation 2015, 24, 249–255. [Google Scholar] [CrossRef]
- One Health. Available online: https://www.who.int/health-topics/one-health (accessed on 2 January 2023).
- Durand, A.; Dupré, C.; Robriquet, L. Faut-il isoler les patients porteurs de BMR? Réanimation 2016, 25, 318–327. [Google Scholar] [CrossRef]
- Brun-Buisson, C. Le dépistage des porteurs de bactéries multirésistantes: Chez quels patients? Réanimation 2015, 24, 304–314. [Google Scholar] [CrossRef]
- Friedmann, R.; Raveh, D.; Zartzer, E.; Rudensky, B.; Broide, E.; Attias, D.; Yinnon, A.M. Prospective evaluation of colonization with extended-spectrum beta-lactamase (ESBL)-producing enterobacteriaceae among patients at hospital admission and of subsequent colonization with ESBL-producing enterobacteriaceae among patients during hospitalization. Infect. Control Hosp. Epidemiol. 2009, 30, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Kluytmans-van den Bergh, M.F.Q.; Verhulst, C.; Willemsen, L.E.; Verkade, E.; Bonten, M.J.M.; Kluytmans, J.A.J.W. Rectal Carriage of Extended-Spectrum-Beta-Lactamase-Producing Enterobacteriaceae in Hospitalized Patients: Selective Preenrichment Increases Yield of Screening. J. Clin. Microbiol. 2015, 53, 2709–2712. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kluytmans-van den Bergh, M.F.Q.; Bruijning-Verhagen, P.C.J.; Vandenbroucke-Grauls, C.M.J.E.; de Brauwer, E.I.G.B.; Buiting, A.G.M.; Diederen, B.M.; van Elzakker, E.P.M.; Friedrich, A.W.; Hopman, J.; Al Naiemi, N.; et al. Contact precautions in single-bed or multiple-bed rooms for patients with extended-spectrum β-lactamase-producing Enterobacteriaceae in Dutch hospitals: A cluster-randomised, crossover, non-inferiority study. Lancet Infect. Dis. 2019, 19, 1069–1079. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.J.; Pineles, L.; Shardell, M.; Graham, M.M.; Mohammadi, S.; Forrest, G.N.; Reisinger, H.S.; Schweizer, M.L.; Perencevich, E.N. The effect of contact precautions on healthcare worker activity in acute care hospitals. Infect. Control Hosp. Epidemiol. 2013, 34, 69–73. [Google Scholar] [CrossRef]
- Zahar, J.R.; Garrouste-Orgeas, M.; Vesin, A.; Schwebel, C.; Bonadona, A.; Philippart, F.; Ara-Somohano, C.; Misset, B.; Timsit, J.F. Impact of contact isolation for multidrug-resistant organisms on the occurrence of medical errors and adverse events. Intensive Care Med. 2013, 39, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- Tschudin-Sutter, S.; Frei, R.; Dangel, M.; Stranden, A.; Widmer, A.F. Rate of transmission of extended-spectrum beta-lactamase-producing enterobacteriaceae without contact isolation. Clin. Infect. Dis. 2012, 55, 1505–1511. [Google Scholar] [CrossRef]
- Morgan, D.J.; Diekema, D.J.; Sepkowitz, K.; Perencevich, E.N. Adverse outcomes associated with Contact Precautions: A review of the literature. Am. J. Infect. Control 2009, 37, 85–93. [Google Scholar] [CrossRef]
- Tschudin-Sutter, S.; Pargger, H.; Widmer, A.F. Hand hygiene in the intensive care unit. Crit. Care Med. 2010, 38, S299–S305. [Google Scholar] [CrossRef] [PubMed]
- Kaier, K.; Frank, U.; Hagist, C.; Conrad, A.; Meyer, E. The impact of antimicrobial drug consumption and alcohol-based hand rub use on the emergence and spread of extended-spectrum beta-lactamase-producing strains: A time-series analysis. J. Antimicrob. Chemother. 2009, 63, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Su, D.; Hu, B.; Rosenthal, V.D.; Li, R.; Hao, C.; Pan, W.; Tao, L.; Gao, X.; Liu, K. Impact of the International Nosocomial Infection Control Consortium (INICC) Multidimensional Hand Hygiene Approach in five intensive care units in three cities of China. Public Health 2015, 129, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Trick, W.E.; Vernon, M.O.; Welbel, S.F.; Demarais, P.; Hayden, M.K.; Weinstein, R.A.; Chicago Antimicrobial Resistance Project. Multicenter intervention program to increase adherence to hand hygiene recommendations and glove use and to reduce the incidence of antimicrobial resistance. Infect. Control Hosp. Epidemiol. 2007, 28, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Derde, L.P.G.; Cooper, B.S.; Goossens, H.; Malhotra-Kumar, S.; Willems, R.J.L.; Gniadkowski, M.; Hryniewicz, W.; Empel, J.; Dautzenberg, M.J.D.; Annane, D.; et al. Interventions to reduce colonisation and transmission of antimicrobial-resistant bacteria in intensive care units: An interrupted time series study and cluster randomised trial. Lancet Infect. Dis. 2014, 14, 31–39. [Google Scholar] [CrossRef] [PubMed]
- De Freitas DalBen, M.; Teixeira Mendes, E.; Moura, M.L.; Abdel Rahman, D.; Peixoto, D.; Alves Dos Santos, S.; Barcelos de Figueiredo, W.; Vitale Mendes, P.; Utino Taniguchi, L.; Bezerra Coutinho, F.A.; et al. A Model-Based Strategy to Control the Spread of Carbapenem-Resistant Enterobacteriaceae: Simulate and Implement. Infect. Control Hosp. Epidemiol. 2016, 37, 1315–1322. [Google Scholar] [CrossRef]
- Almaguer-Leyva, M.; Mendoza-Flores, L.; Medina-Torres, A.G.; Salinas-Caballero, A.G.; Vidaña-Amaro, J.A.; Garza-Gonzalez, E.; Camacho-Ortiz, A. Hand hygiene compliance in patients under contact precautions and in the general hospital population. Am. J. Infect. Control 2013, 41, 976–978. [Google Scholar] [CrossRef]
- Golan, Y.; Doron, S.; Griffith, J.; El Gamal, H.; Tanios, M.; Blunt, K.; Barefoot, L.; Bloom, J.; Gamson, K.; Snydman, L.K.; et al. The impact of gown-use requirement on hand hygiene compliance. Clin. Infect. Dis. 2006, 42, 370–376. [Google Scholar] [CrossRef]
- Han, J.H.; Bilker, W.B.; Nachamkin, I.; Zaoutis, T.E.; Coffin, S.E.; Linkin, D.R.; Hu, B.; Tolomeo, P.; Fishman, N.O.; Lautenbach, E. The effect of a hospital-wide urine culture screening intervention on the incidence of extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella species. Infect. Control Hosp. Epidemiol. 2013, 34, 1160–1166. [Google Scholar] [CrossRef]
- Harris, A.D.; Pineles, L.; Belton, B.; Johnson, J.K.; Shardell, M.; Loeb, M.; Newhouse, R.; Dembry, L.; Braun, B.; Perencevich, E.N.; et al. Universal glove and gown use and acquisition of antibiotic-resistant bacteria in the ICU: A randomized trial. JAMA 2013, 310, 1571–1580. [Google Scholar] [CrossRef]
- Huskins, W.C.; Huckabee, C.M.; O’Grady, N.P.; Murray, P.; Kopetskie, H.; Zimmer, L.; Walker, M.E.; Sinkowitz-Cochran, R.L.; Jernigan, J.A.; Samore, M.; et al. Intervention to reduce transmission of resistant bacteria in intensive care. N. Engl. J. Med. 2011, 364, 1407–1418. [Google Scholar] [CrossRef]
- Hilliquin, D.; Lomont, A.; Zahar, J.-R. Cohorting for preventing the nosocomial spread of Carbapenemase-Producing Enterobacterales, in non-epidemic settings: Is it mandatory? J. Hosp. Infect. 2020, 105, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Palmore, T.N.; Henderson, D.K. Managing transmission of carbapenem-resistant enterobacteriaceae in healthcare settings: A view from the trenches. Clin. Infect. Dis. 2013, 57, 1593–1599. [Google Scholar] [CrossRef]
- Austin, D.J.; Bonten, M.J.; Weinstein, R.A.; Slaughter, S.; Anderson, R.M. Vancomycin-resistant enterococci in intensive-care hospital settings: Transmission dynamics, persistence, and the impact of infection control programs. Proc. Natl. Acad. Sci. USA 1999, 96, 6908–6913. [Google Scholar] [CrossRef] [PubMed]
- Schwaber, M.J.; Lev, B.; Israeli, A.; Solter, E.; Smollan, G.; Rubinovitch, B.; Shalit, I.; Carmeli, Y. Israel Carbapenem-Resistant Enterobacteriaceae Working Group Containment of a country-wide outbreak of carbapenem-resistant Klebsiella pneumoniae in Israeli hospitals via a nationally implemented intervention. Clin. Infect. Dis. 2011, 52, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, S.; Zanichelli, V.; Grayson, M.L.; Twyman, A.; Abbas, M.; Pires, D.; Allegranzi, B.; Harbarth, S. Control of Carbapenem-resistant Enterobacteriaceae, Acinetobacter baumannii, and Pseudomonas aeruginosa in Healthcare Facilities: A Systematic Review and Reanalysis of Quasi-experimental Studies. Clin. Infect. Dis. 2019, 68, 873–884. [Google Scholar] [CrossRef]
- Lepelletier, D.; Lucet, J.C.; Astagneau, P.; Coignard, B.; Vaux, S.; Rabaud, C.; Grandbastien, B.; Berthelot, P. Control of emerging extensively drug-resistant organisms (eXDRO) in France: A survey among infection preventionists from 286 healthcare facilities. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1615–1620. [Google Scholar] [CrossRef]
- Atamna-Mawassi, H.; Huberman-Samuel, M.; Hershcovitz, S.; Karny-Epstein, N.; Kola, A.; Cortés, L.E.L.; Leibovici, L.; Yahav, D. Interventions to reduce infections caused by multidrug resistant Enterobacteriaceae (MDR-E): A systematic review and meta-analysis. J. Infect. 2021, 83, 156–166. [Google Scholar] [CrossRef]
- Ayukekbong, J.A.; Ntemgwa, M.; Atabe, A.N. The threat of antimicrobial resistance in developing countries: Causes and control strategies. Antimicrob. Resist. Infect. Control 2017, 6, 47. [Google Scholar] [CrossRef]
- Hay, S.I.; Rao, P.C.; Dolecek, C.; Day, N.P.J.; Stergachis, A.; Lopez, A.D.; Murray, C.J.L. Measuring and mapping the global burden of antimicrobial resistance. BMC Med. 2018, 16, 78. [Google Scholar] [CrossRef]
- Birgand, G.; Leroy, C.; Nerome, S.; Luong Nguyen, L.B.; Lolom, I.; Armand-Lefevre, L.; Ciotti, C.; Lecorre, B.; Marcade, G.; Fihman, V.; et al. Costs associated with implementation of a strict policy for controlling spread of highly resistant microorganisms in France. BMJ Open 2016, 6, e009029. [Google Scholar] [CrossRef]
- Otter, J.A.; Burgess, P.; Davies, F.; Mookerjee, S.; Singleton, J.; Gilchrist, M.; Parsons, D.; Brannigan, E.T.; Robotham, J.; Holmes, A.H. Counting the cost of an outbreak of carbapenemase-producing Enterobacteriaceae: An economic evaluation from a hospital perspective. Clin. Microbiol. Infect. 2017, 23, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.M.; Mathema, B.; Larson, E.L. Carbapenem-resistant Enterobacteriaceae in the community: A scoping review. Int. J. Antimicrob. Agents 2017, 50, 127–134. [Google Scholar] [CrossRef]
- Dortet, L.; Cuzon, G.; Ponties, V.; Nordmann, P. Trends in carbapenemase-producing Enterobacteriaceae, France, 2012 to 2014. Eurosurveillance 2017, 22, 30461. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, P.M. Multidrug-resistant Gram-negative bacteria: A product of globalization. J. Hosp. Infect. 2015, 89, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Jakovljevic, M.; Al Ahdab, S.; Jurisevic, M.; Mouselli, S. Antibiotic Resistance in Syria: A Local Problem Turns Into a Global Threat. Front. Public Health 2018, 6, 212. [Google Scholar] [CrossRef]
- Collin, S.M.; Farra, A. Antimicrobial resistance, infection prevention and control, and conflict in the Middle East. Int. J. Infect. Dis. 2021, 111, 326–327. [Google Scholar] [CrossRef]
- Safdar, N.; Sengupta, S.; Musuuza, J.S.; Juthani-Mehta, M.; Drees, M.; Abbo, L.M.; Milstone, A.M.; Furuno, J.P.; Varman, M.; Anderson, D.J.; et al. Status of the Prevention of Multidrug-Resistant Organisms in International Settings: A Survey of the Society for Healthcare Epidemiology of America Research Network. Infect. Control Hosp. Epidemiol. 2017, 38, 53–60. [Google Scholar] [CrossRef]
- Maechler, F.; Schwab, F.; Hansen, S.; Fankhauser, C.; Harbarth, S.; Huttner, B.D.; Diaz-Agero, C.; Lopez, N.; Canton, R.; Ruiz-Garbajosa, P.; et al. Contact isolation versus standard precautions to decrease acquisition of extended-spectrum β-lactamase-producing Enterobacterales in non-critical care wards: A cluster-randomised crossover trial. Lancet 2020, 20, 575–584. [Google Scholar] [CrossRef]
- Kirkland, K.B. Taking off the gloves: Toward a less dogmatic approach to the use of contact isolation. Clin. Infect. Dis. 2009, 48, 766–771. [Google Scholar] [CrossRef]
- Harris, A.D.; Morgan, D.J.; Pineles, L.; Magder, L.; O’Hara, L.M.; Johnson, J.K. Acquisition of Antibiotic-Resistant Gram-negative Bacteria in the Benefits of Universal Glove and Gown (BUGG) Cluster Randomized Trial. Clin. Infect. Dis. 2021, 72, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Teerawattanapong, N.; Kengkla, K.; Dilokthornsakul, P.; Saokaew, S.; Apisarnthanarak, A.; Chaiyakunapruk, N. Prevention and Control of Multidrug-Resistant Gram-Negative Bacteria in Adult Intensive Care Units: A Systematic Review and Network Meta-analysis. Clin. Infect. Dis. 2017, 64, S51–S60. [Google Scholar] [CrossRef] [PubMed]
| High Compliance with Hand Hygiene | Antimicrobial Stewardship | Active Surveillance | Patient Isolation | Additional Contact Precautions | Cohorting | ||
|---|---|---|---|---|---|---|---|
| Multi-drug resistant bacterial species | Escherichia coli | ✓ | ✓ | × | × | × | × |
| Other Enterobacterales | ✓ | ✓ | × | × | × | × | |
| VRE | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Pseudomonas aeruginosa producing carbapenemase | ✓ | ✓ | × | × | × | × | |
| Acinetobacter baumannii resistant to carbapenems | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Hospital wards | Long term care facilities | ✓ | ✓ | × | × | × | × |
| Intensive care units and reanimation | ✓ | ✓ | ✓ | ✓ | ✓ | ✓* | |
| Other wards | ✓ | ✓ | × | × | × | × | |
| Patient clinical characteristics | Diarrhea and or fecal incontinence | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| High Katz score | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Other patients | ✓ | ✓ | × | × | × | × | |
| Variables | Target |
|---|---|
| Incidence of MDRO | <national median incidence |
| Predominant species | Escherichiacoli |
| Compliance with hand hygiene | >70% |
| Regional prevalence | low |
| Antimicrobial stewardship program | Applied |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saliba, R.; Zahar, J.-R.; Dabar, G.; Riachy, M.; Karam-Sarkis, D.; Husni, R. Limiting the Spread of Multidrug-Resistant Bacteria in Low-to-Middle-Income Countries: One Size Does Not Fit All. Pathogens 2023, 12, 144. https://doi.org/10.3390/pathogens12010144
Saliba R, Zahar J-R, Dabar G, Riachy M, Karam-Sarkis D, Husni R. Limiting the Spread of Multidrug-Resistant Bacteria in Low-to-Middle-Income Countries: One Size Does Not Fit All. Pathogens. 2023; 12(1):144. https://doi.org/10.3390/pathogens12010144
Chicago/Turabian StyleSaliba, Rindala, Jean-Ralph Zahar, Georges Dabar, Moussa Riachy, Dolla Karam-Sarkis, and Rola Husni. 2023. "Limiting the Spread of Multidrug-Resistant Bacteria in Low-to-Middle-Income Countries: One Size Does Not Fit All" Pathogens 12, no. 1: 144. https://doi.org/10.3390/pathogens12010144
APA StyleSaliba, R., Zahar, J.-R., Dabar, G., Riachy, M., Karam-Sarkis, D., & Husni, R. (2023). Limiting the Spread of Multidrug-Resistant Bacteria in Low-to-Middle-Income Countries: One Size Does Not Fit All. Pathogens, 12(1), 144. https://doi.org/10.3390/pathogens12010144







