Cryptosporidium: Still Open Scenarios
Abstract
:1. Introduction
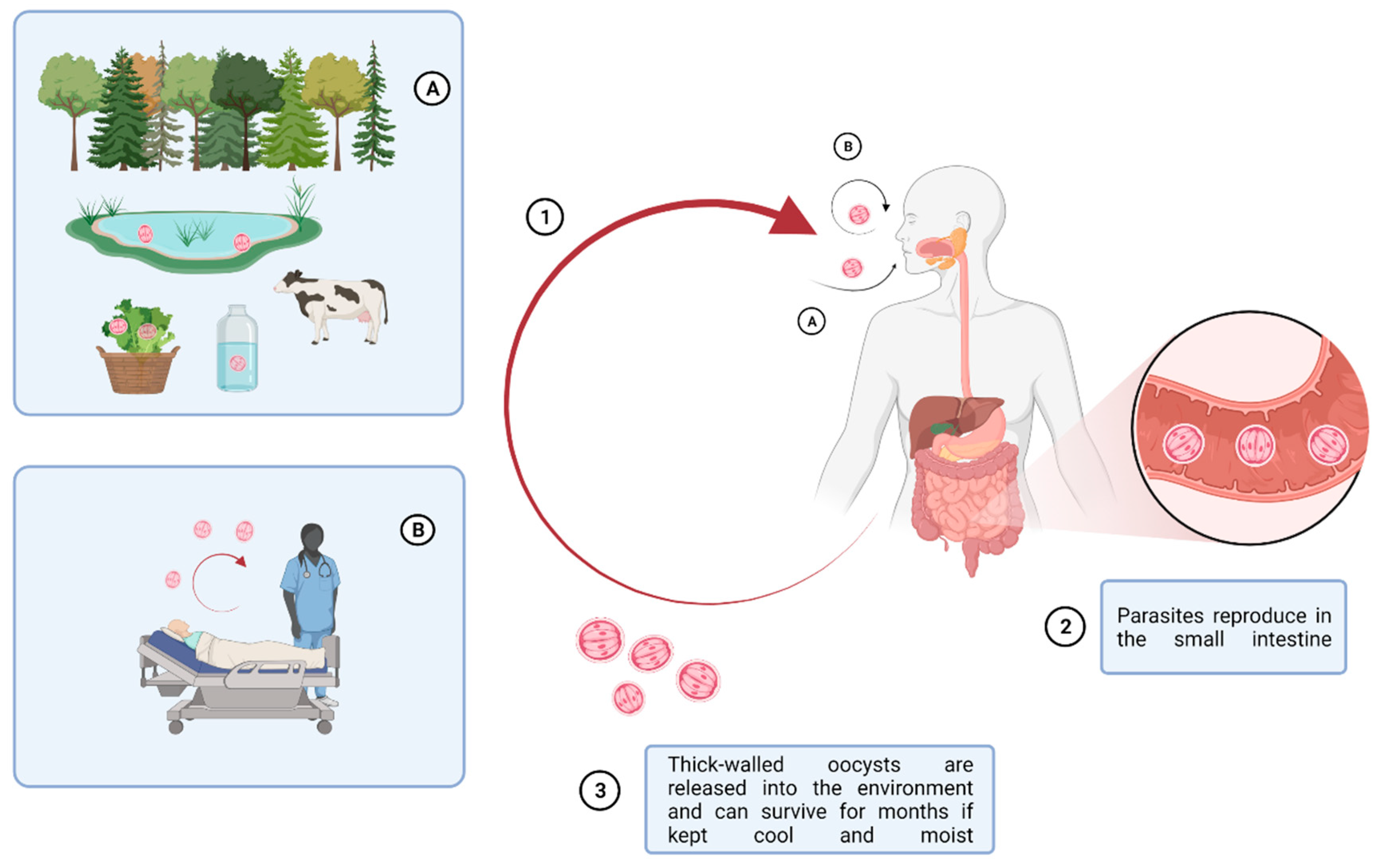
2. Epidemiology of Cryptosporidium: Recent Updates
3. Genotyping and Spreading
4. Geographical Spreading
5. Cryptosporidium Treatments
6. Microbiota and Cryptosporidium spp.
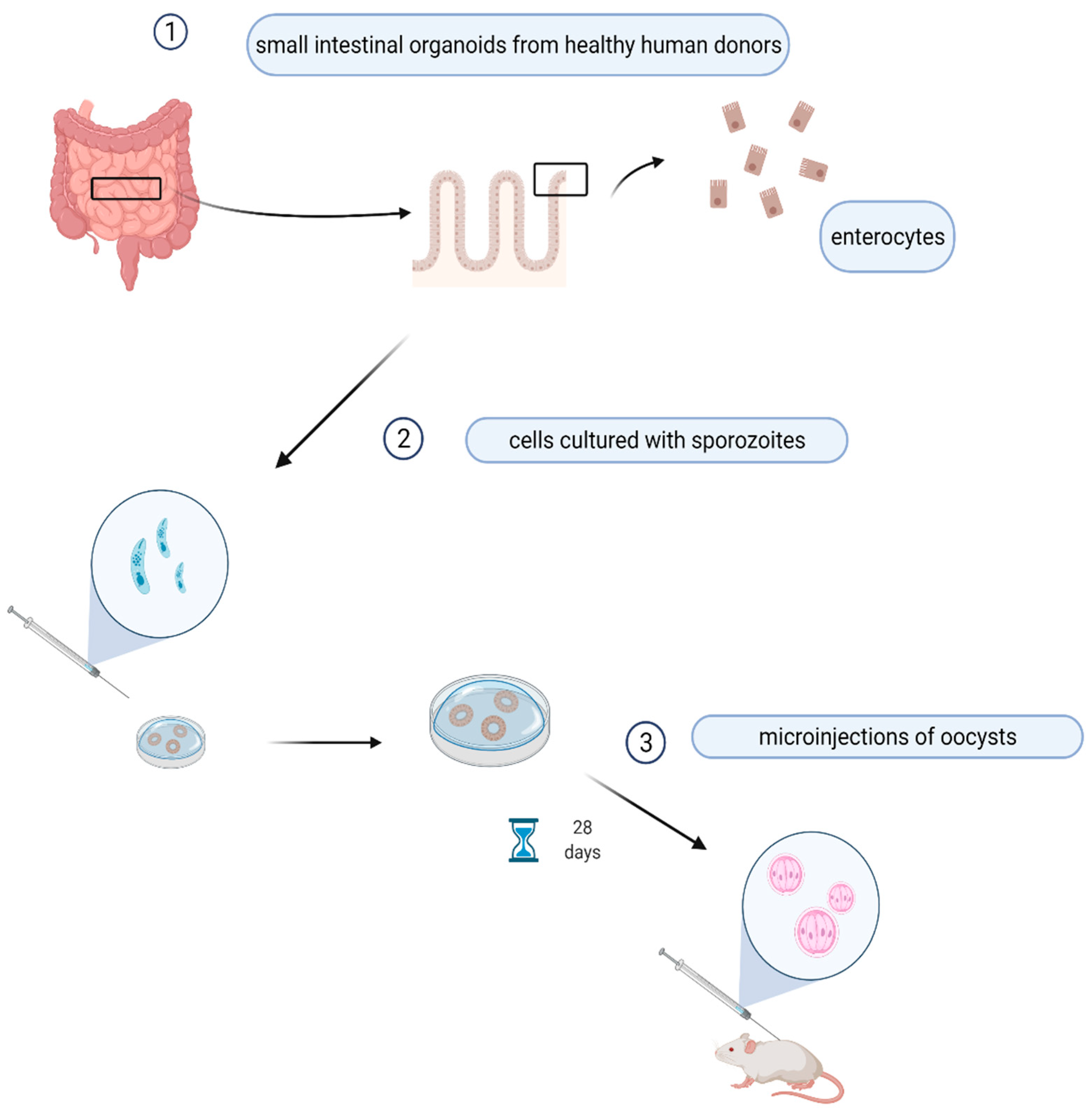
7. Study Models
8. Materials and Methods
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Putignani, L. Cryptosporidium. In Book Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar] [CrossRef]
- Putignani, L.; Menichella, D. Global Distribution, Public Health and Clinical Impact of the Protozoan Pathogen Cryptosporidium. Interdiscip. Perspect. Infect. Dis. 2010, 2010, 753512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cacciò, S.M.; Putignani, L. Epidemiology of Human Cryptosporidiosis. In Cryptosporidium: Parasite and Disease; Cacciò, S.M., Widmer, G., Eds.; Springer: Vienna, Austria, 2014; pp. 43–79. ISBN 978-3-7091-1561-9. [Google Scholar]
- Checkley, W.; White, A.C.; Jaganath, D.; Arrowood, M.J.; Chalmers, R.M.; Chen, X.-M.; Fayer, R.; Griffiths, J.K.; Guerrant, R.L.; Hedstrom, L.; et al. A Review of the Global Burden, Novel Diagnostics, Therapeutics, and Vaccine Targets for Cryptosporidium. Lancet Infect. Dis. 2015, 15, 85–94. [Google Scholar] [CrossRef] [Green Version]
- Rossignol, J.F.; Ayoub, A.; Ayers, M.S. Treatment of Diarrhea Caused by Cryptosporidium Parvum: A Prospective Randomized, Double-Blind, Placebo-Controlled Study of Nitazoxanide. J. Infect. Dis. 2001, 184, 103–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amadi, B.; Mwiya, M.; Musuku, J.; Watuka, A.; Sianongo, S.; Ayoub, A.; Kelly, P. Effect of Nitazoxanide on Morbidity and Mortality in Zambian Children with Cryptosporidiosis: A Randomised Controlled Trial. Lancet Lond. Engl. 2002, 360, 1375–1380. [Google Scholar] [CrossRef]
- Bones, A.J.; Jossé, L.; More, C.; Miller, C.N.; Michaelis, M.; Tsaousis, A.D. Past and Future Trends of Cryptosporidium in Vitro Research. Exp. Parasitol. 2019, 196, 28–37. [Google Scholar] [CrossRef]
- O’ Leary, J.K.; Blake, L.; Corcoran, G.D.; Sleator, R.D.; Lucey, B. A Novel Genotyping Method for Cryptosporidium Hominis. Exp. Parasitol. 2021, 225, 108113. [Google Scholar] [CrossRef]
- King, P.; Tyler, K.M.; Hunter, P.R. Anthroponotic Transmission of Cryptosporidium Parvum Predominates in Countries with Poorer Sanitation: A Systematic Review and Meta-Analysis. Parasit. Vectors 2019, 12, 16. [Google Scholar] [CrossRef]
- Widmer, G.; Carmena, D.; Kváč, M.; Chalmers, R.M.; Kissinger, J.C.; Xiao, L.; Sateriale, A.; Striepen, B.; Laurent, F.; Lacroix-Lamandé, S.; et al. Update on Cryptosporidium Spp.: Highlights from the Seventh International Giardia and Cryptosporidium Conference. Parasite Paris Fr. 2020, 27, 14. [Google Scholar] [CrossRef] [Green Version]
- Koehler, A.V.; Whipp, M.J.; Haydon, S.R.; Gasser, R.B. Cryptosporidium Cuniculus--New Records in Human and Kangaroo in Australia. Parasit. Vectors 2014, 7, 492. [Google Scholar] [CrossRef]
- Feng, Y.; Ryan, U.M.; Xiao, L. Genetic Diversity and Population Structure of Cryptosporidium. Trends Parasitol. 2018, 34, 997–1011. [Google Scholar] [CrossRef]
- Fayer, R. Taxonomy and Species Delimitation in Cryptosporidium. Exp. Parasitol. 2010, 124, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Ryan, U.; Fayer, R.; Xiao, L. Cryptosporidium Species in Humans and Animals: Current Understanding and Research Needs. Parasitology 2014, 141, 1667–1685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tyzzer, E.E. An Extracellular Coccidium, Cryptosporidium Muris (Gen. Et Sp. Nov.), of the Gastric Glands of the Common Mouse. J. Med. Res. 1910, 23, 487–510.3. [Google Scholar] [PubMed]
- Vetterling, J.M.; Jervis, H.R.; Merrill, T.G.; Sprinz, H. Cryptosporidium Wrairi Sp. n. from the Guinea Pig Cavia Porcellus, with an Emendation of the Genus. J. Protozool. 1971, 18, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Fayer, R.; Trout, J.M.; Xiao, L.; Morgan, U.M.; Lal, A.A.; Dubey, J.P. CRYPTOSPORIDIUM CANIS N. SP. FROM DOMESTIC DOGS. J. Parasitol. 2001, 87, 1415–1422. [Google Scholar] [CrossRef]
- Fayer, R.; Santín, M.; Macarisin, D. Cryptosporidium Ubiquitum n. Sp. in Animals and Humans. Vet. Parasitol. 2010, 172, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Fayer, R.; Santín, M. Cryptosporidium Xiaoi n. Sp. (Apicomplexa: Cryptosporidiidae) in Sheep (Ovis Aries). Vet. Parasitol. 2009, 164, 192–200. [Google Scholar] [CrossRef]
- Li, J.; Guo, Y.; Roellig, D.M.; Li, N.; Feng, Y.; Xiao, L. Cryptosporidium Felis Differs from Other Cryptosporidium Spp. in Codon Usage. Microb. Genom. 2021, 7, 000711. [Google Scholar] [CrossRef]
- Ryan, U.; Zahedi, A.; Feng, Y.; Xiao, L. An Update on Zoonotic Cryptosporidium Species and Genotypes in Humans. Animals 2021, 11, 3307. [Google Scholar] [CrossRef]
- Levine, N.D. Taxonomy and Review of the Coccidian Genus Cryptosporidium (Protozoa, Apicomplexa). J. Protozool. 1984, 31, 94–98. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P.; Sitjà-Bobadilla, A. Cryptosporidium Molnari n. Sp. (Apicomplexa: Cryptosporidiidae) Infecting Two Marine Fish Species, Sparus aurata L. and Dicentrarchus labrax L. Int. J. Parasitol. 2002, 32, 1007–1021. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P.; Quiroga, M.; Sitjà-Bobadilla, A.; Redondo, M.; Palenzuela, O.; Padrós, F.; Vázquez, S.; Nieto, J. Cryptosporidium Scophthalmi n. Sp. (Apicomplexa: Cryptosporidiidae) from Cultured Turbot Scophthalmus Maximus. Light and Electron Microscope Description and Histopathological Study. Dis. Aquat. Organ. 2004, 62, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Kváč, M.; Kestřánová, M.; Pinková, M.; Květoňová, D.; Kalinová, J.; Wagnerová, P.; Kotková, M.; Vítovec, J.; Ditrich, O.; McEvoy, J.; et al. Cryptosporidium Scrofarum n. Sp. (Apicomplexa: Cryptosporidiidae) in Domestic Pigs (Sus Scrofa). Vet. Parasitol. 2013, 191, 218–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kváč, M.; Květoňová, D.; Sak, B.; Ditrich, O. Cryptosporidium Pig Genotype II in Immunocompetent Man. Emerg. Infect. Dis. 2009, 15, 982–983. [Google Scholar] [CrossRef] [PubMed]
- Current, W.L.; Upton, S.J.; Haynes, T.B. The Life Cycle of Cryptosporidium Baileyi n. Sp. (Apicomplexa, Cryptosporidiidae) Infecting Chickens. J. Protozool. 1986, 33, 289–296. [Google Scholar] [CrossRef]
- Ditrich, O.; Palkovič, L.; Štěrba, J.; Prokopič, J.; Loudová, J.; Giboda, M. The First Finding OfCryptosporidium Baileyi in Man. Parasitol. Res. 1991, 77, 44–47. [Google Scholar] [CrossRef]
- da Silva, D.C.; Homem, C.G.; Nakamura, A.A.; Teixeira, W.F.P.; Perri, S.H.V.; Meireles, M.V. Physical, Epidemiological, and Molecular Evaluation of Infection by Cryptosporidium Galli in Passeriformes. Parasitol. Res. 2010, 107, 271–277. [Google Scholar] [CrossRef]
- Jirků, M.; Valigurová, A.; Koudela, B.; Krízek, J.; Modrý, D.; Slapeta, J. New Species of Cryptosporidium Tyzzer, 1907 (Apicomplexa) from Amphibian Host: Morphology, Biology and Phylogeny. Folia Parasitol. 2008, 55, 81–94. [Google Scholar] [CrossRef] [Green Version]
- da Silva, D.C.; Paiva, P.R.S.O.; Nakamura, A.A.; Homem, C.G.; de Souza, M.S.; Grego, K.F.; Meireles, M.V. The Detection of Cryptosporidium Serpentis in Snake Fecal Samples by Real-Time PCR. Vet. Parasitol. 2014, 204, 134–138. [Google Scholar] [CrossRef]
- Pedraza-Díaz, S.; Ortega-Mora, L.M.; Carrión, B.A.; Navarro, V.; Gómez-Bautista, M. Molecular Characterisation of Cryptosporidium Isolates from Pet Reptiles. Vet. Parasitol. 2009, 160, 204–210. [Google Scholar] [CrossRef]
- Fayer, R.; Santín, M.; Trout, J.M. Cryptosporidium Ryanae n. Sp. (Apicomplexa: Cryptosporidiidae) in Cattle (Bos Taurus). Vet. Parasitol. 2008, 156, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Fayer, R.; Santín, M.; Xiao, L. Cryptosporidium Bovis n. Sp. (Apicomplexa: Cryptosporidiidae) in Cattle (Bos Taurus). J. Parasitol. 2005, 91, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Higuera, A.; Villamizar, X.; Herrera, G.; Giraldo, J.C.; Vasquez-A, L.R.; Urbano, P.; Villalobos, O.; Tovar, C.; Ramírez, J.D. Molecular Detection and Genotyping of Intestinal Protozoa from Different Biogeographical Regions of Colombia. PeerJ 2020, 8, e8554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryan, U.M.; Power, M.; Xiao, L. Cryptosporidium Fayeri n. Sp. (Apicomplexa: Cryptosporidiidae) from the Red Kangaroo (Macropus Rufus). J. Eukaryot. Microbiol. 2008, 55, 22–26. [Google Scholar] [CrossRef]
- Braima, K.; Zahedi, A.; Oskam, C.; Austen, J.; Egan, S.; Reid, S.; Ryan, U. Zoonotic Infection by Cryptosporidium Fayeri IVgA10G1T1R1 in a Western Australian Human. Zoonoses Public Health 2021, 68, 358–360. [Google Scholar] [CrossRef]
- Waldron, L.S.; Cheung-Kwok-Sang, C.; Power, M.L. Wildlife-Associated Cryptosporidium Fayeri in Human, Australia. Emerg. Infect. Dis. 2010, 16, 2006–2007. [Google Scholar] [CrossRef]
- Power, M.L.; Ryan, U.M. A New Species of Cryptosporidium (Apicomplexa: Cryptosporidiidae) from Eastern Grey Kangaroos (Macropus Giganteus). J. Parasitol. 2008, 94, 1114–1117. [Google Scholar] [CrossRef]
- Ren, X.; Zhao, J.; Zhang, L.; Ning, C.; Jian, F.; Wang, R.; Lv, C.; Wang, Q.; Arrowood, M.J.; Xiao, L. Cryptosporidium Tyzzeri n. Sp. (Apicomplexa: Cryptosporidiidae) in Domestic Mice (Mus Musculus). Exp. Parasitol. 2012, 130, 274–281. [Google Scholar] [CrossRef]
- Garcia-R, J.C.; Pita, A.B.; Velathanthiri, N.; French, N.P.; Hayman, D.T.S. Species and Genotypes Causing Human Cryptosporidiosis in New Zealand. Parasitol. Res. 2020, 119, 2317–2326. [Google Scholar] [CrossRef]
- Elwin, K.; Hadfield, S.J.; Robinson, G.; Crouch, N.D.; Chalmers, R.M. Cryptosporidium Viatorum n. Sp. (Apicomplexa: Cryptosporidiidae) among Travellers Returning to Great Britain from the Indian Subcontinent, 2007–2011. Int. J. Parasitol. 2012, 42, 675–682. [Google Scholar] [CrossRef]
- Ong, C.S.L.; Eisler, D.L.; Alikhani, A.; Fung, V.W.K.; Tomblin, J.; Bowie, W.R.; Isaac-Renton, J.L. Novel Cryptosporidium Genotypes in Sporadic Cryptosporidiosis Cases: First Report of Human Infections with a Cervine Genotype. Emerg. Infect. Dis. 2002, 8, 263–268. [Google Scholar] [CrossRef]
- Holubová, N.; Zikmundová, V.; Limpouchová, Z.; Sak, B.; Konečný, R.; Hlásková, L.; Rajský, D.; Kopacz, Z.; McEvoy, J.; Kváč, M. Cryptosporidium Proventriculi Sp. n. (Apicomplexa: Cryptosporidiidae) in Psittaciformes Birds. Eur. J. Protistol. 2019, 69, 70–87. [Google Scholar] [CrossRef]
- Holubová, N.; Tůmová, L.; Sak, B.; Hejzlarová, A.; Konečný, R.; McEvoy, J.; Kváč, M. Description of Cryptosporidium Ornithophilus n. Sp. (Apicomplexa: Cryptosporidiidae) in Farmed Ostriches. Parasit. Vectors 2020, 13, 340. [Google Scholar] [CrossRef] [PubMed]
- Ježková, J.; Prediger, J.; Holubová, N.; Sak, B.; Konečný, R.; Feng, Y.; Xiao, L.; Rost, M.; McEvoy, J.; Kváč, M. Cryptosporidium Ratti n. Sp. (Apicomplexa: Cryptosporidiidae) and Genetic Diversity of Cryptosporidium Spp. in Brown Rats (Rattus Norvegicus) in the Czech Republic. Parasitology 2021, 148, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Kváč, M.; Hofmannová, L.; Hlásková, L.; Květoňová, D.; Vítovec, J.; McEvoy, J.; Sak, B. Cryptosporidium Erinacei n. Sp. (Apicomplexa: Cryptosporidiidae) in Hedgehogs. Vet. Parasitol. 2014, 201, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Prediger, J.; Ježková, J.; Holubová, N.; Sak, B.; Konečný, R.; Rost, M.; McEvoy, J.; Rajský, D.; Kváč, M. Cryptosporidium Sciurinum n. Sp. (Apicomplexa: Cryptosporidiidae) in Eurasian Red Squirrels (Sciurus Vulgaris). Microorganisms 2021, 9, 2050. [Google Scholar] [CrossRef]
- Ježková, J.; Limpouchová, Z.; Prediger, J.; Holubová, N.; Sak, B.; Konečný, R.; Květoňová, D.; Hlásková, L.; Rost, M.; McEvoy, J.; et al. Cryptosporidium Myocastoris n. Sp. (Apicomplexa: Cryptosporidiidae), the Species Adapted to the Nutria (Myocastor Coypus). Microorganisms 2021, 9, 813. [Google Scholar] [CrossRef]
- Jezkova, J.; Horcickova, M.; Hlaskova, L.; Sak, B.; Kvetonova, D.; Novak, J.; Hofmannova, L.; McEvoy, J.; Kvac, M. Cryptosporidium Testudinis Sp. n., Cryptosporidium Ducismarci Traversa, 2010 and Cryptosporidium Tortoise Genotype III (Apicomplexa: Cryptosporidiidae) in Tortoises. Folia Parasitol. 2016, 63, 35. [Google Scholar] [CrossRef] [Green Version]
- Holubová, N.; Sak, B.; Horčičková, M.; Hlásková, L.; Květoňová, D.; Menchaca, S.; McEvoy, J.; Kváč, M. Cryptosporidium Avium n. Sp. (Apicomplexa: Cryptosporidiidae) in Birds. Parasitol. Res. 2016, 115, 2243–2251. [Google Scholar] [CrossRef]
- Horčičková, M.; Čondlová, Š.; Holubová, N.; Sak, B.; Květoňová, D.; Hlásková, L.; Konečný, R.; Sedláček, F.; Clark, M.; Giddings, C.; et al. Diversity of Cryptosporidium in Common Voles and Description of Cryptosporidium Alticolis Sp. n. and Cryptosporidium Microti Sp. n. (Apicomplexa: Cryptosporidiidae). Parasitology 2019, 146, 220–233. [Google Scholar] [CrossRef]
- Zahedi, A.; Bolland, S.J.; Oskam, C.L.; Ryan, U. Cryptosporidium Abrahamseni n. Sp. (Apicomplexa: Cryptosporidiiae) from Red-Eye Tetra (Moenkhausia Sanctaefilomenae). Exp. Parasitol. 2021, 223, 108089. [Google Scholar] [CrossRef] [PubMed]
- Bolland, S.J.; Zahedi, A.; Oskam, C.; Murphy, B.; Ryan, U. Cryptosporidium Bollandi n. Sp. (Apicomplexa: Cryptosporidiiae) from Angelfish (Pterophyllum Scalare) and Oscar Fish (Astronotus Ocellatus). Exp. Parasitol. 2020, 217, 107956. [Google Scholar] [CrossRef] [PubMed]
- Čondlová, Š.; Horčičková, M.; Sak, B.; Květoňová, D.; Hlásková, L.; Konečný, R.; Stanko, M.; McEvoy, J.; Kváč, M. Cryptosporidium Apodemi Sp. n. and Cryptosporidium Ditrichi Sp. n. (Apicomplexa: Cryptosporidiidae) in Apodemus spp. Eur. J. Protistol. 2018, 63, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Beser, J.; Bujila, I.; Wittesjö, B.; Lebbad, M. From Mice to Men: Three Cases of Human Infection with Cryptosporidium Ditrichi. Infect. Genet. Evol. 2020, 78, 104120. [Google Scholar] [CrossRef] [PubMed]
- Kváč, M.; Havrdová, N.; Hlásková, L.; Daňková, T.; Kanděra, J.; Ježková, J.; Vítovec, J.; Sak, B.; Ortega, Y.; Xiao, L.; et al. Cryptosporidium Proliferans n. Sp. (Apicomplexa: Cryptosporidiidae): Molecular and Biological Evidence of Cryptic Species within Gastric Cryptosporidium of Mammals. PLoS ONE 2016, 11, e0147090. [Google Scholar] [CrossRef]
- Li, J.; Wang, Z.; Karim, M.R.; Zhang, L. Detection of Human Intestinal Protozoan Parasites in Vegetables and Fruits: A Review. Parasit. Vectors 2020, 13, 380. [Google Scholar] [CrossRef]
- Hlavsa, M.C.; Aluko, S.K.; Miller, A.D.; Person, J.; Gerdes, M.E.; Lee, S.; Laco, J.P.; Hannapel, E.J.; Hill, V.R. Outbreaks Associated with Treated Recreational Water—United States, 2015–2019. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 733–738. [Google Scholar] [CrossRef]
- Boschi-Pinto, C. Estimating Child Mortality Due to Diarrhoea in Developing Countries. Bull. World Health Organ. 2008, 86, 710–717. [Google Scholar] [CrossRef]
- Xiao, L.; Ryan, U.M. Cryptosporidiosis: An Update in Molecular Epidemiology. Curr. Opin. Infect. Dis. 2004, 17, 483–490. [Google Scholar] [CrossRef] [Green Version]
- Pumipuntu, N.; Piratae, S. Cryptosporidiosis: A Zoonotic Disease Concern. Vet. World 2018, 11, 681–686. [Google Scholar] [CrossRef] [Green Version]
- Vanathy, K.; Parija, S.C.; Mandal, J.; Hamide, A.; Krishnamurthy, S. Cryptosporidiosis: A Mini Review. Trop. Parasitol. 2017, 7, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Shaik, J.S.; Grigg, M.E. Genomics and Molecular Epidemiology of Cryptosporidium Species. Acta Trop. 2018, 184, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sponseller, J.K.; Griffiths, J.K.; Tzipori, S. The Evolution of Respiratory Cryptosporidiosis: Evidence for Transmission by Inhalation. Clin. Microbiol. Rev. 2014, 27, 575–586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bogan, J.E. Disinfection Techniques for Cryptosporidium. J. Dairy Vet. Sci. 2018, 7, 001–003. [Google Scholar] [CrossRef] [Green Version]
- Yang, K.; LeJeune, J.; Alsdorf, D.; Lu, B.; Shum, C.K.; Liang, S. Global Distribution of Outbreaks of Water-Associated Infectious Diseases. PLoS Negl. Trop. Dis. 2012, 6, e1483. [Google Scholar] [CrossRef] [Green Version]
- O’ Leary, J.K.; Blake, L.; Corcoran, G.D.; Sleator, R.D.; Lucey, B. Increased Diversity and Novel Subtypes among Clinical Cryptosporidium Parvum and Cryptosporidium Hominis Isolates in Southern Ireland. Exp. Parasitol. 2020, 218, 107967. [Google Scholar] [CrossRef]
- Duranti, A.; Cacciò, S.M.; Pozio, E.; Di Egidio, A.; De Curtis, M.; Battisti, A.; Scaramozzino, P. Risk Factors Associated with Cryptosporidium Parvum Infection in Cattle. Zoonoses Public Health 2009, 56, 176–182. [Google Scholar] [CrossRef]
- Di Piazza, F.; Di Benedetto, M.A.; Maida, C.M.; Glorioso, S.; Adamo, G.; Mazzola, T.; Firenze, A. A Study on Occupational Exposure of Sicilian Farmers to Giardia and Cryptosporidium. J. Prev. Med. Hyg. 2013, 54, 212–217. [Google Scholar]
- Mor, S.M.; Tumwine, J.K.; Ndeezi, G.; Srinivasan, M.G.; Kaddu-Mulindwa, D.H.; Tzipori, S.; Griffiths, J.K. Respiratory Cryptosporidiosis in HIV-Seronegative Children in Uganda: Potential for Respiratory Transmission. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2010, 50, 1366–1372. [Google Scholar] [CrossRef] [Green Version]
- Waldron, L.S.; Dimeski, B.; Beggs, P.J.; Ferrari, B.C.; Power, M.L. Molecular Epidemiology, Spatiotemporal Analysis, and Ecology of Sporadic Human Cryptosporidiosis in Australia. Appl. Environ. Microbiol. 2011, 77, 7757–7765. [Google Scholar] [CrossRef] [Green Version]
- Waldron, L.S.; Ferrari, B.C.; Power, M.L. Glycoprotein 60 Diversity in C. Hominis and C. Parvum Causing Human Cryptosporidiosis in NSW, Australia. Exp. Parasitol. 2009, 122, 124–127. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, A.; Monis, P.; Aucote, S.; King, B.; Paparini, A.; Jian, F.; Yang, R.; Oskam, C.; Ball, A.; Robertson, I.; et al. Zoonotic Cryptosporidium Species in Animals Inhabiting Sydney Water Catchments. PLoS ONE 2016, 11, e0168169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Razakandrainibe, R.; Diawara, E.H.I.; Costa, D.; Le Goff, L.; Lemeteil, D.; Ballet, J.J.; Gargala, G.; Favennec, L. Common Occurrence of Cryptosporidium Hominis in Asymptomatic and Symptomatic Calves in France. PLoS Negl. Trop. Dis. 2018, 12, e0006355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varcasia, A.; Capelli, G.; Ruiu, A.; Ladu, M.; Scala, A.; Bjorkman, C. Prevalence of Neospora Caninum Infection in Sardinian Dairy Farms (Italy) Detected by Iscom ELISA on Tank Bulk Milk. Parasitol. Res. 2006, 98, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, R.M.; Robinson, G.; Elwin, K.; Hadfield, S.J.; Xiao, L.; Ryan, U.; Modha, D.; Mallaghan, C. Cryptosporidium Sp. Rabbit Genotype, a Newly Identified Human Pathogen. Emerg. Infect. Dis. 2009, 15, 829–830. [Google Scholar] [CrossRef]
- Lv, C.; Zhang, L.; Wang, R.; Jian, F.; Zhang, S.; Ning, C.; Wang, H.; Feng, C.; Wang, X.; Ren, X.; et al. Cryptosporidium Spp. in Wild, Laboratory, and Pet Rodents in China: Prevalence and Molecular Characterization. Appl. Environ. Microbiol. 2009, 75, 7692–7699. [Google Scholar] [CrossRef] [Green Version]
- Bodager, J.R.; Parsons, M.B.; Wright, P.C.; Rasambainarivo, F.; Roellig, D.; Xiao, L.; Gillespie, T.R. Complex Epidemiology and Zoonotic Potential for Cryptosporidium Suis in Rural Madagascar. Vet. Parasitol. 2015, 207, 140–143. [Google Scholar] [CrossRef]
- Montecino-Latorre, D.; Li, X.; Xiao, C.; Atwill, E.R. Elevation and Vegetation Determine Cryptosporidium Oocyst Shedding by Yellow-Bellied Marmots (Marmota Flaviventris) in the Sierra Nevada Mountains. Int. J. Parasitol. Parasites Wildl. 2015, 4, 171–177. [Google Scholar] [CrossRef] [Green Version]
- Zahedi, A.; Paparini, A.; Jian, F.; Robertson, I.; Ryan, U. Public Health Significance of Zoonotic Cryptosporidium Species in Wildlife: Critical Insights into Better Drinking Water Management. Int. J. Parasitol. Parasites Wildl. 2016, 5, 88–109. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Xiao, L.; Alderisio, K.; Elwin, K.; Cebelinski, E.; Chalmers, R.; Santin, M.; Fayer, R.; Kvac, M.; Ryan, U.; et al. Subtyping Cryptosporidium Ubiquitum, a Zoonotic Pathogen Emerging in Humans. Emerg. Infect. Dis. 2014, 20, 217–224. [Google Scholar] [CrossRef]
- Dyachenko, V.; Kuhnert, Y.; Schmaeschke, R.; Etzold, M.; Pantchev, N.; Daugschies, A. Occurrence and Molecular Characterization of Cryptosporidium Spp. Genotypes in European Hedgehogs (Erinaceus europaeus L.) in Germany. Parasitology 2010, 137, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Power, M.L.; Cheung-Kwok-Sang, C.; Slade, M.; Williamson, S. Cryptosporidium Fayeri: Diversity within the GP60 Locus of Isolates from Different Marsupial Hosts. Exp. Parasitol. 2009, 121, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, A.; Monis, P.; Gofton, A.W.; Oskam, C.L.; Ball, A.; Bath, A.; Bartkow, M.; Robertson, I.; Ryan, U. Cryptosporidium Species and Subtypes in Animals Inhabiting Drinking Water Catchments in Three States across Australia. Water Res. 2018, 134, 327–340. [Google Scholar] [CrossRef] [Green Version]
- Jex, A.R.; Smith, H.V.; Monis, P.T.; Campbell, B.E.; Gasser, R.B. Cryptosporidium—Biotechnological Advances in the Detection, Diagnosis and Analysis of Genetic Variation. Biotechnol. Adv. 2008, 26, 304–317. [Google Scholar] [CrossRef]
- Klotz, C.; Aebischer, T.; Seeber, F. Stem Cell-Derived Cell Cultures and Organoids for Protozoan Parasite Propagation and Studying Host-Parasite Interaction. Int. J. Med. Microbiol. IJMM 2012, 302, 203–209. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Guo, F.; Zhu, G. Cryptosporidium Lactate Dehydrogenase Is Associated with the Parasitophorous Vacuole Membrane and Is a Potential Target for Developing Therapeutics. PLoS Pathog. 2015, 11, e1005250. [Google Scholar] [CrossRef] [PubMed]
- Kelly, P. Treatment and Prevention of Cryptosporidiosis: What Options Are There for a Country like Zambia? Parasitology 2011, 138, 1488–1491. [Google Scholar] [CrossRef]
- Diawara, E.H.; François, A.; Stachulski, A.V.; Razakandrainibe, R.; Costa, D.; Favennec, L.; Rossignol, J.-F.; Gargala, G. Systemic Efficacy on Cryptosporidium Parvum Infection of Aminoxanide (RM-5061), a New Amino-Acid Ester Thiazolide Prodrug of Tizoxanide. Parasitology 2021, 148, 975–984. [Google Scholar] [CrossRef]
- Hussien, S.M.M.; Abdella, O.H.; Abu-Hashim, A.H.; Aboshiesha, G.A.; Taha, M.A.A.; El-Shemy, A.S.; El-Bader, M.M. Comparative Study between the Effect of Nitazoxanide and Paromomycine in Treatment of Cryptosporidiosis in Hospitalized Children. J. Egypt. Soc. Parasitol. 2013, 43, 463–470. [Google Scholar]
- Huang, M.-Z.; Li, J.; Guan, L.; Li, D.-Q.; Nie, X.-M.; Gui, R.; Chen, X. Therapeutic Effects of Acetylspiramycin and Garlicin on Cryptosporidiosis among Drug Users. Int. J. Parasitol. Drugs Drug Resist. 2015, 5, 185–190. [Google Scholar] [CrossRef] [Green Version]
- Diptyanusa, A.; Sari, I.P. Treatment of Human Intestinal Cryptosporidiosis: A Review of Published Clinical Trials. Int. J. Parasitol. Drugs Drug Resist. 2021, 17, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Nachipo, P.; Hermann, D.; Quinnan, G.; Gordon, M.A.; Van Voorhis, W.C.; Iroh Tam, P.-Y. Evaluating the Safety, Tolerability, Pharmacokinetics and Efficacy of Clofazimine in Cryptosporidiosis (CRYPTOFAZ): Study Protocol for a Randomized Controlled Trial. Trials 2018, 19, 456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahman, S.U.; Mi, R.; Zhou, S.; Gong, H.; Ullah, M.; Huang, Y.; Han, X.; Chen, Z. Advances in Therapeutic and Vaccine Targets for Cryptosporidium: Challenges and Possible Mitigation Strategies. Acta Trop. 2021, 226, 106273. [Google Scholar] [CrossRef] [PubMed]
- Vinayak, S.; Pawlowic, M.C.; Sateriale, A.; Brooks, C.F.; Studstill, C.J.; Bar-Peled, Y.; Cipriano, M.J.; Striepen, B. Genetic Modification of the Diarrhoeal Pathogen Cryptosporidium Parvum. Nature 2015, 523, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Ashigbie, P.G.; Shepherd, S.; Steiner, K.L.; Amadi, B.; Aziz, N.; Manjunatha, U.H.; Spector, J.M.; Diagana, T.T.; Kelly, P. Use-Case Scenarios for an Anti-Cryptosporidium Therapeutic. PLoS Negl. Trop. Dis. 2021, 15, e0009057. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, K.N.C.; Sowmyanarayanan, T.V.; Paul, A.; Babji, S.; Ajjampur, S.S.R.; Priyadarshini, S.; Sarkar, R.; Balasubramanian, K.A.; Wanke, C.A.; Ward, H.D.; et al. Immune Response and Intestinal Permeability in Children with Acute Gastroenteritis Treated with Lactobacillus Rhamnosus GG: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2014, 58, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Pickerd, N.; Tuthill, D. Resolution of Cryptosporidiosis with Probiotic Treatment. Postgrad. Med. J. 2004, 80, 112–113. [Google Scholar] [CrossRef] [Green Version]
- Sebastián Domingo, J.J. Review of the Role of Probiotics in Gastrointestinal Diseases in Adults. Gastroenterol. Hepatol. 2017, 40, 417–429. [Google Scholar] [CrossRef] [Green Version]
- Partida-Rodríguez, O.; Serrano-Vázquez, A.; Nieves-Ramírez, M.E.; Moran, P.; Rojas, L.; Portillo, T.; González, E.; Hernández, E.; Finlay, B.B.; Ximenez, C. Human Intestinal Microbiota: Interaction Between Parasites and the Host Immune Response. Arch. Med. Res. 2017, 48, 690–700. [Google Scholar] [CrossRef]
- Ichikawa-Seki, M.; Motooka, D.; Kinami, A.; Murakoshi, F.; Takahashi, Y.; Aita, J.; Hayashi, K.; Tashibu, A.; Nakamura, S.; Iida, T.; et al. Specific Increase of Fusobacterium in the Faecal Microbiota of Neonatal Calves Infected with Cryptosporidium Parvum. Sci. Rep. 2019, 9, 12517. [Google Scholar] [CrossRef] [Green Version]
- Carey, M.A.; Medlock, G.L.; Alam, M.; Kabir, M.; Uddin, M.J.; Nayak, U.; Papin, J.; Faruque, A.S.G.; Haque, R.; Petri, W.A.; et al. Megasphaera in the Stool Microbiota Is Negatively Associated With Diarrheal Cryptosporidiosis. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021, 73, e1242–e1251. [Google Scholar] [CrossRef] [PubMed]
- Brosschot, T.P.; Reynolds, L.A. The Impact of a Helminth-Modified Microbiome on Host Immunity. Mucosal Immunol. 2018, 11, 1039–1046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bednarska, M.; Jankowska, I.; Pawelas, A.; Piwczyńska, K.; Bajer, A.; Wolska-Kuśnierz, B.; Wielopolska, M.; Welc-Falęciak, R. Prevalence of Cryptosporidium, Blastocystis, and Other Opportunistic Infections in Patients with Primary and Acquired Immunodeficiency. Parasitol. Res. 2018, 117, 2869–2879. [Google Scholar] [CrossRef] [Green Version]
- Mohteshamuddin, K.; Hamdan, L.; AlKaabi, A.B.; Barigye, R. Cryptosporidium Parvum and Other Enteric Pathogens in Scouring Neonatal Dairy Calves from the Al Ain Region, United Arab Emirates. Vet. Parasitol. Reg. Stud. Rep. 2020, 21, 100435. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Yoon, K.-J. An Overview of Calf Diarrhea—Infectious Etiology, Diagnosis, and Intervention. J. Vet. Sci. 2014, 15, 1. [Google Scholar] [CrossRef] [Green Version]
- Garro, C.J.; Morici, G.E.; Tomazic, M.L.; Vilte, D.; Encinas, M.; Vega, C.; Bok, M.; Parreño, V.; Schnittger, L. Occurrence of Cryptosporidium and Other Enteropathogens and Their Association with Diarrhea in Dairy Calves of Buenos Aires Province, Argentina. Vet. Parasitol. Reg. Stud. Rep. 2021, 24, 100567. [Google Scholar] [CrossRef]
- Charania, R.; Wade, B.E.; McNair, N.N.; Mead, J.R. Changes in the Microbiome of Cryptosporidium-Infected Mice Correlate to Differences in Susceptibility and Infection Levels. Microorganisms 2020, 8, 879. [Google Scholar] [CrossRef]
- Mammeri, M.; Obregón, D.A.; Chevillot, A.; Polack, B.; Julien, C.; Pollet, T.; Cabezas-Cruz, A.; Adjou, K.T. Cryptosporidium Parvum Infection Depletes Butyrate Producer Bacteria in Goat Kid Microbiome. Front. Microbiol. 2020, 11, 548737. [Google Scholar] [CrossRef]
- Pane, S.; Ristori, M.V.; Gardini, S.; Russo, A.; Del Chierico, F.; Putignani, L. Clinical Parasitology and Parasitome Maps as Old and New Tools to Improve Clinical Microbiomics. Pathogens 2021, 10, 1550. [Google Scholar] [CrossRef]
- Karanis, P. The Truth about in Vitro Culture of Cryptosporidium Species. Parasitology 2018, 145, 855–864. [Google Scholar] [CrossRef]
- Karanis, P.; Aldeyarbi, H.M. Evolution of Cryptosporidium in Vitro Culture. Int. J. Parasitol. 2011, 41, 1231–1242. [Google Scholar] [CrossRef] [PubMed]
- Bhalchandra, S.; Lamisere, H.; Ward, H. Intestinal Organoid/Enteroid-Based Models for Cryptosporidium. Curr. Opin. Microbiol. 2020, 58, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Chieppa, M.; Rescigno, M.; Huang, A.Y.C.; Germain, R.N. Dynamic Imaging of Dendritic Cell Extension into the Small Bowel Lumen in Response to Epithelial Cell TLR Engagement. J. Exp. Med. 2006, 203, 2841–2852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heo, I.; Dutta, D.; Schaefer, D.A.; Iakobachvili, N.; Artegiani, B.; Sachs, N.; Boonekamp, K.E.; Bowden, G.; Hendrickx, A.P.A.; Willems, R.J.L.; et al. Modelling Cryptosporidium Infection in Human Small Intestinal and Lung Organoids. Nat. Microbiol. 2018, 3, 814–823. [Google Scholar] [CrossRef] [PubMed]
- Luka, G.; Samiei, E.; Tasnim, N.; Dalili, A.; Najjaran, H.; Hoorfar, M. Comprehensive Review of Conventional and State-of-the-Art Detection Methods of Cryptosporidium. J. Hazard. Mater. 2022, 421, 126714. [Google Scholar] [CrossRef] [PubMed]
- Manjunatha, U.H.; Chao, A.T.; Leong, F.J.; Diagana, T.T. Cryptosporidiosis Drug Discovery: Opportunities and Challenges. ACS Infect. Dis. 2016, 2, 530–537. [Google Scholar] [CrossRef]
| Species Name | Major Host(s) | Human Host | Author(s) |
|---|---|---|---|
| Cryptosporidium cuniculus | rabbits | yes | [11] |
| Cryptosporidium hominis | equine | yes | [12,13] |
| Cryptosporidium parvum | sheep, cattle | yes | [13,14] |
| Cryptosporidium muris | rodents and farm animals | yes | [13,15] |
| Cryptosporidium andersoni | cattle | yes | [13,16,17] |
| Cryptosporidium suis | pigs | yes | [13,18,19] |
| Cryptosporidium wrairi | guinea pigs | yes | [13,16] |
| Cryptosporidium meleagridis | birds | yes | [13] |
| Cryptosporidium canis | dogs | yes | [13,17] |
| Cryptosporidium ubiquitum | ruminants and rodents | yes | [18] |
| Cryptosporidium xiaoi | sheep and goats | yes | [18,19] |
| Cryptosporidium felis | cats | yes | [20,21] |
| Cryptosporidium nasorum | fish | not | [22] |
| Cryptosporidium molnari | fish | not | [23] |
| Cryptosporidium scophthalmi | fish | not | [24] |
| Cryptosporidium scrofarum | pigs | yes | [25,26] |
| Cryptosporidium baileyi | chicken | yes | [27,28] |
| Cryptosporidium galli | chicken | not | [29] |
| Cryptosporidium fragile | amphibia | not | [30] |
| Cryptosporidium serpentis | snakes | not | [31] |
| Cryptosporidium varanii | pet reptiles | not | [32] |
| Cryptosporidium ryanae | cattle | not | [33] |
| Cryptosporidium bovis | cattle | yes | [34,35] |
| Cryptosporidium fayeri | marsupials | yes | [36,37,38] |
| Cryptosporidium macropodum | marsupials | not | [39] |
| Cryptosporidium tyzzeri | mice | yes | [40,41] |
| Cryptosporidium viatorum | rodents | yes | [42] |
| Cryptosporidium occultus | rodents | yes | [43] |
| Cryptosporidium proventriculi | birds | not | [44] |
| Cryptosporidium ornithophilus | ostrich | not | [45] |
| Cryptosporidium ratti | rats | not | [46] |
| Cryptosporidium erinacei | hedgehogs | yes | [41,47] |
| Cryptosporidium sciurinum | red squirrels | not | [48] |
| Cryptosporidium myocastoris | nutria | not | [49] |
| Cryptosporidium testudinis | tortoises | not | [50] |
| Cryptosporidium avium | birds | not | [51] |
| Cryptosporidium alticolis | common voles | not | [52] |
| Cryptosporidium microti | common voles | not | [52] |
| Cryptosporidium abrahamseni | fish | not | [53] |
| Cryptosporidium bollandi | fish | not | [54] |
| Cryptosporidium apodemi | rats | not | [55] |
| Cryptosporidium ditrichi | rodents | yes | [55,56] |
| Cryptosporidium ducismarci | tortoises | not | [50] |
| Cryptosporidium proliferans | rodents | not | [57] |
| Treatment | Clinical Improvement | Protocols | Author(s) |
|---|---|---|---|
| Nitazoxanide | inhibits oocyst excretion | clinical trials | [90] |
| Paromomycin | no clinical benefits | clinical trials | [91] |
| Spiramycin | oocyst reduction | in vitro and in vivo studies | [92] |
| Macrolides (Azithromycin, Erythromycin, Roxithromycin) | no clear clinical benefits | in vitro and in vivo studies | [93] |
| Clofazimine | oocyst reduction | clinical trials | [94] |
| Benzoxaboroles, Pyrazolopyridine | oocyst reduction | in vivo studies | [95] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pane, S.; Putignani, L. Cryptosporidium: Still Open Scenarios. Pathogens 2022, 11, 515. https://doi.org/10.3390/pathogens11050515
Pane S, Putignani L. Cryptosporidium: Still Open Scenarios. Pathogens. 2022; 11(5):515. https://doi.org/10.3390/pathogens11050515
Chicago/Turabian StylePane, Stefania, and Lorenza Putignani. 2022. "Cryptosporidium: Still Open Scenarios" Pathogens 11, no. 5: 515. https://doi.org/10.3390/pathogens11050515
APA StylePane, S., & Putignani, L. (2022). Cryptosporidium: Still Open Scenarios. Pathogens, 11(5), 515. https://doi.org/10.3390/pathogens11050515







