Molecular Epidemiological Evidence Implicates Cattle as a Primary Reservoir of Campylobacter jejuni Infecting People via Contaminated Chickens
Abstract
:1. Introduction
2. Results
2.1. Characteristics of the Study Area
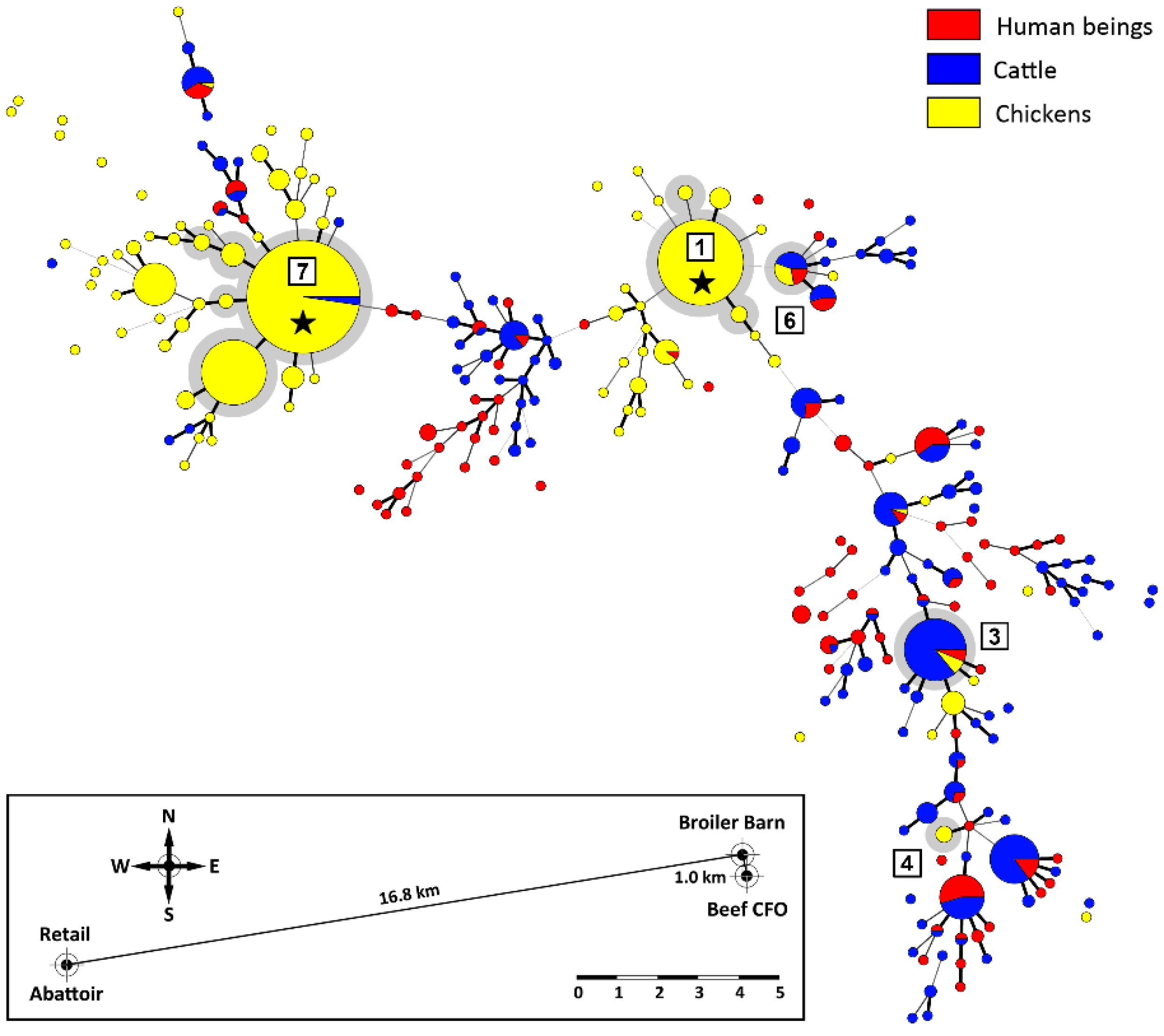
2.2. A High Degree of Campylobacter jejuni Strain Diversity Was Observed
2.3. Cattle Were the Dominant Source of Attributable Human Campylobacteriosis Cases
2.4. A High Proportion of Campylobacter jejuni Isolates Were in CGF40 Clusters Comprising Isolates from Multiple Sources
2.5. Cattle Was the Dominant Source of Clinically Relevant CGF40 Clusters
2.6. Additional Evidence Supports Beef Cattle as an Important Reservoir of Campylobacter jejuni Subtypes Infecting Chickens
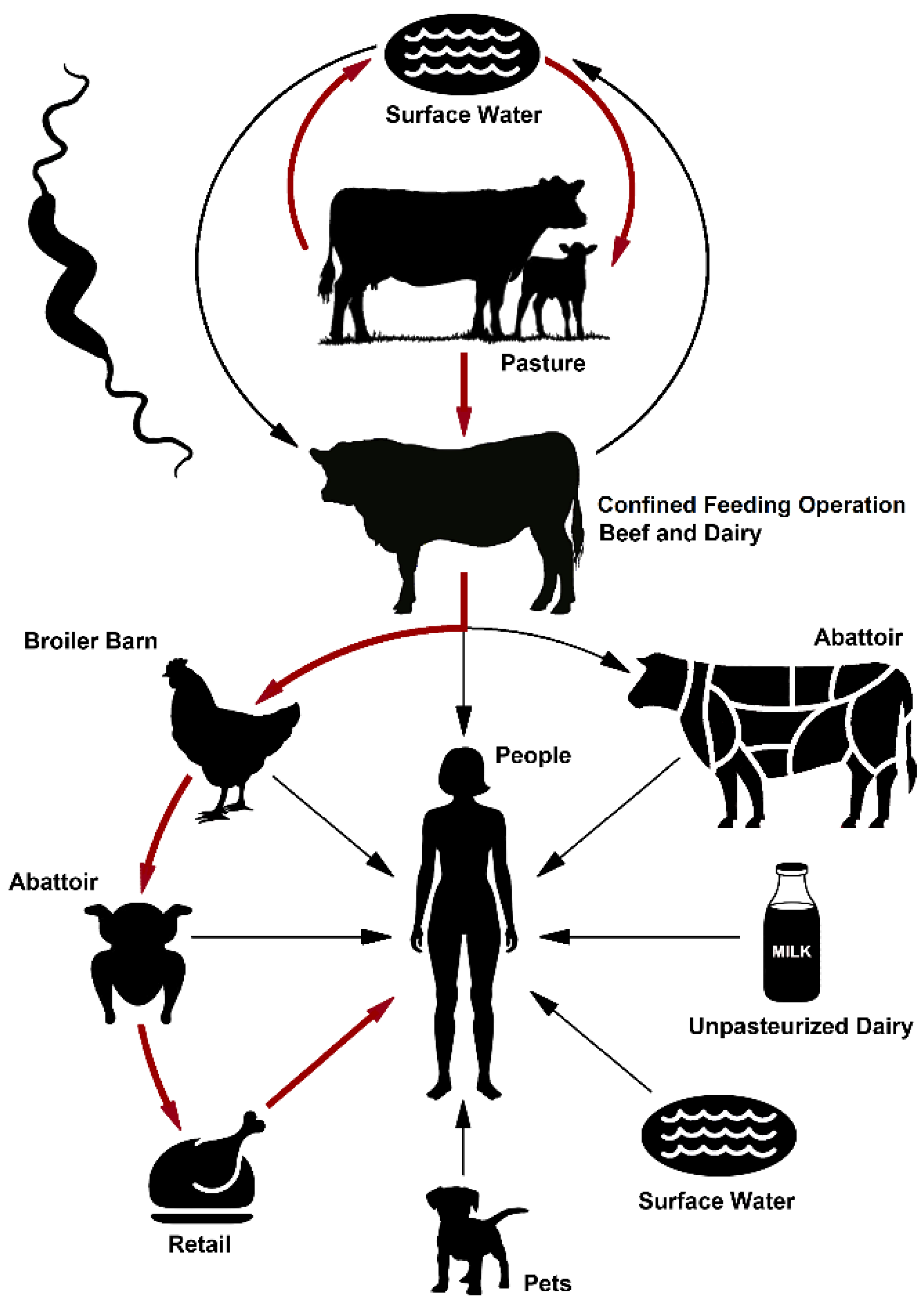
2.7. A Model for the Indirect Transmission of Bovine Campylobacter jejuni to People via Contamination of the Chicken Supply Chain
2.8. Global Production of Chickens and Cattle Occurs in the Same Geographical Space
3. Discussion
4. Materials and Methods
4.1. Campylobacteriosis Rates, and Livestock and Manure Production
4.2. Isolation of Campylobacter jejuni
4.3. Identification of Campylobacter jejuni
4.4. Subtyping of Campylobacter jejuni Isolates
4.5. Assignment of Sequence Type and Clonal Complex for CGF40 Subtypes
4.6. Source Attribution of Human Campylobacteriosis Cases
4.7. Statistical Analysis of Source Attribution Estimates and Hypothesis Testing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inglis, G.D.; Boras, V.F.; Webb, A.L.; Suttorp, V.V.; Hodgkinson, P.; Taboada, E.N. Enhanced microbiological surveillance reveals that temporal case clusters contribute to the high rates of campylobacteriosis in a model agroecosystem. Int. J. Med. Microbiol. 2019, 309, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Kaakoush, N.O.; Castano-Rodriguez, N.; Mitchell, H.M.; Man, S.M. Global epidemiology of Campylobacter infection. Clin. Microbiol. Rev. 2015, 28, 687–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, M.K.; Murray, R.; Flockhart, L.; Pintar, K.; Pollari, F.; Fazil, A.; Nesbitt, A.; Marshall, B. Estimates of the burden of foodborne illness in Canada for 30 specified pathogens and unspecified agents, circa 2006. Foodborne Pathog. Dis. 2013, 10, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Platts-Mills, J.A.; Kosek, M.N.; Allos, B.M.; Blaser, M.J. Campylobacter species. Antimicrobe 2020. Available online: http://www.antimicrobe.org/b91.asp (accessed on 4 November 2022).
- Johnson, T.J.; Shank, J.M.; Johnson, J.G. Current and potential treatments for reducing Campylobacter colonization in animal hosts and disease in humans. Front Microbiol. 2017, 8, 487. [Google Scholar] [CrossRef] [Green Version]
- Whiley, H.; van den Akker, B.; Giglio, S.; Bentham, R. The role of environmental reservoirs in human campylobacteriosis. Int. J. Environ. Res. Public Health 2013, 10, 5886–5907. [Google Scholar] [CrossRef] [Green Version]
- Skarp, C.P.A.; Hanninen, M.L.; Rautelin, H.I.K. Campylobacteriosis: The role of poultry meat. Clin. Microbiol. Infect. 2016, 22, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Ravel, A.; Hurst, M.; Petrica, N.; David, J.; Mutschall, S.K.; Pintar, K.; Taboada, E.N.; Pollari, F. Source attribution of human campylobacteriosis at the point of exposure by combining comparative exposure assessment and subtype comparison based on comparative genomic fingerprinting. PLoS ONE 2017, 12, e0183790. [Google Scholar] [CrossRef] [Green Version]
- Sheppard, S.K.; Dallas, J.F.; Strachan, N.J.; MacRae, M.; McCarthy, N.D.; Wilson, D.J.; Gormley, F.J.; Falush, D.; Ogden, I.D.; Maiden, M.C.; et al. Campylobacter genotyping to determine the source of human infection. Clin. Infect. Dis. 2009, 48, 1072–1078. [Google Scholar] [CrossRef] [Green Version]
- Mullner, P.; Spencer, S.E.; Wilson, D.J.; Jones, G.; Noble, A.D.; Midwinter, A.C.; Collins-Emerson, J.M.; Carter, P.; Hathaway, S.; French, N.P. Assigning the source of human campylobacteriosis in New Zealand: A comparative genetic and epidemiological approach. Infect. Genet. Evol. 2009, 9, 1311–1319. [Google Scholar] [CrossRef]
- Wilson, D.J.; Gabriel, E.; Leatherbarrow, A.J.; Cheesbrough, J.; Gee, S.; Bolton, E.; Fox, A.; Fearnhead, P.; Hart, C.A.; Diggle, P.J. Tracing the source of campylobacteriosis. PLoS Genet. 2008, 4, e1000203. [Google Scholar] [CrossRef]
- Inglis, G.D.; Taboada, E.N.; Boras, V.F. Rates of fluoroquinolone resistance in domestically acquired Campylobacter jejuni are increasing in people living within a model study location in Canada. Can. J. Microbiol. 2021, 67, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Inglis, G.D.; Teixeira, J.S.; Boras, V.F. Comparative prevalence and diversity of Campylobacter jejuni strains in water and human beings over a 1-year period in southwestern Alberta, Canada. Can. J. Microbiol. 2021, 67, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Inglis, G.D.; Gusse, J.G.; House, K.E.; Shelton, T.G.; Taboada, E.N. Clinically relevant Campylobacter jejuni subtypes are readily found and transmitted within the cattle production continuum but present a limited foodborne risk. Appl. Environ. Microbiol. 2020, 86, e02101-19. [Google Scholar] [CrossRef] [PubMed]
- Inglis, G.D.; Gusse, J.G.; House, K.E.; Shelton, T.G.; Taboada, E.N. Tetracycline resistant Campylobacter jejuni subtypes emanating from beef cattle administered non-therapeutic chlortetracycline are longitudinally transmitted within the production continuum but are not detected in ground beef. Microorganisms 2020, 8, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pires, S.M.; Evers, E.G.; van Pelt, W.; Ayers, T.; Scallan, E.; Angulo, F.J.; Havelaar, A.; Hald, T.; Med-Vet-Net Workpackage 28 Working, G. Attributing the human disease burden of foodborne infections to specific sources. Foodborne Pathog. Dis. 2009, 6, 417–424. [Google Scholar] [CrossRef] [Green Version]
- Alberta Government. 2011 Census of Agriculture for Alberta. Alberta Agriculture and Rural Development 2014. Available online: https://www1.agric.gov.ab.ca/$Department/deptdocs.nsf/all/agdex4091/$FILE/852-6.pdf (accessed on 4 November 2022).
- Alberta Government. 2011 Municipal Affairs Population List. 2011. Available online: https://open.alberta.ca/opendata/alberta-municipal-affairs-population-list (accessed on 4 November 2022).
- Alberta Government. Agriculture Land Resource Atlas of Alberta. Alberta Agriculture, Food and Rural Development 2005. Available online: https://www.alberta.ca/agricultural-land-resource-atlas-of-alberta.aspx (accessed on 4 November 2022).
- Government of Canada. A Geographical Profile of Livestock Manure Production in Canada, 2006. Statistics Canada 2008. Available online: https://www150.statcan.gc.ca/n1/pub/16-002-x/2008004/article/10751-eng.htm (accessed on 4 November 2022).
- United States Department of Agriculture. Agricultural Waste Management Field Handbook. Chapter 4. Agricultural Waste Characteristics; 2008. Available online: https://msa.maryland.gov/megafile/msa/speccol/sc5300/sc5339/000113/003000/003072/unrestricted/20066421e-026.pdf (accessed on 4 November 2022).
- Inglis, G.D.; Ramezani, N.; Taboada, E.N.; Boras, V.F.; Uwiera, R.R.E. Analysis of Campylobacter jejuni subtype distribution in the chicken broiler production continuum: A longitudinal examination to identify primary contamination points. Appl. Environ. Microbiol. 2021, 87, e02001-20. [Google Scholar] [CrossRef]
- Garcia-Sanchez, L.; Melero, B.; Rovira, J. Campylobacter in the food chain. Adv. Food Nutr. Res. 2018, 86, 215–252. [Google Scholar] [CrossRef]
- Hansson, I.; Sandberg, M.; Habib, I.; Lowman, R.; Engvall, E.O. Knowledge gaps in control of Campylobacter for prevention of campylobacteriosis. Transbound. Emerg. Dis. 2018, 65, 30–48. [Google Scholar] [CrossRef] [Green Version]
- Barco, L.; Barrucci, F.; Olsen, J.E.; Ricci, A. Salmonella source attribution based on microbial subtyping. Int. J. Food Microbiol. 2013, 163, 193–203. [Google Scholar] [CrossRef]
- Taboada, E.N.; Ross, S.L.; Mutschall, S.K.; Mackinnon, J.M.; Roberts, M.J.; Buchanan, C.J.; Kruczkiewicz, P.; Jokinen, C.C.; Thomas, J.E.; Nash, J.H.; et al. Development and validation of a comparative genomic fingerprinting method for high-resolution genotyping of Campylobacter jejuni. J. Clin. Microbiol. 2012, 50, 788–797. [Google Scholar] [CrossRef]
- Clark, C.G.; Taboada, E.; Grant, C.C.; Blakeston, C.; Pollari, F.; Marshall, B.; Rahn, K.; Mackinnon, J.; Daignault, D.; Pillai, D.; et al. Comparison of molecular typing methods useful for detecting clusters of Campylobacter jejuni and C. coli isolates through routine surveillance. J. Clin. Microbiol. 2012, 50, 798–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Food Safety Authority. Overview of methods for source attribution for human illness from food-borne microbiological hazards—scientific opinion of the panel on biological hazards. EFSA J. 2008, 764, 1–43. [Google Scholar]
- van Pelt, W.; van de Giessen, A.; van Leeuwen, W.; Wannet, W.J.; Henken, A.M.; Evers, E. Oorsprong, omvang en kosten van humane salmonellose. Deel 1. Oorsprong van human salmonellose met betrekking tot varken, rund, kip, ei en overige bronnen. Infect. Bull. 1999, 10, 233–240. [Google Scholar]
- Strachan, N.J.; Gormley, F.J.; Rotariu, O.; Ogden, I.D.; Miller, G.; Dunn, G.M.; Sheppard, S.K.; Dallas, J.F.; Reid, T.M.; Howie, H.; et al. Attribution of Campylobacter infections in northeast Scotland to specific sources by use of multilocus sequence typing. J. Infect. Dis. 2009, 199, 1205–1208. [Google Scholar] [CrossRef] [Green Version]
- Mullner, P.; Jones, G.; Noble, A.; Spencer, S.E.; Hathaway, S.; French, N.P. Source attribution of food-borne zoonoses in New Zealand: A modified Hald model. Risk Anal. 2009, 29, 970–984. [Google Scholar] [CrossRef] [PubMed]
- Guevremont, E.; Lamoureux, L.; Loubier, C.B.; Villeneuve, S.; Dubuc, J. Detection and characterization of Campylobacter spp. from 40 dairy cattle herds in Quebec, Canada. Foodborne Pathog. Dis. 2014, 11, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Inglis, G.D.; Kalischuk, L.D.; Busz, H.W. Chronic shedding of Campylobacter species in beef cattle. J. Appl. Microbiol. 2004, 97, 410–420. [Google Scholar] [CrossRef]
- Inglis, G.D.; Kalischuk, L.D.; Busz, H.W. A survey of Campylobacter species shed in faeces of beef cattle using polymerase chain reaction. Can. J. Microbiol. 2003, 49, 655–661. [Google Scholar] [CrossRef]
- Thepault, A.; Poezevara, T.; Quesne, S.; Rose, V.; Chemaly, M.; Rivoal, K. Prevalence of thermophilic Campylobacter in cattle production at slaughterhouse level in France and link between C. jejuni bovine strains and campylobacteriosis. Front. Microbiol. 2018, 9, 471. [Google Scholar] [CrossRef] [Green Version]
- Webb, A.L.; Selinger, L.B.; Taboada, E.N.; Inglis, G.D. Subtype-specific selection for resistance to fluoroquinolones but not to tetracyclines is evident in Campylobacter jejuni isolates from beef cattle in confined feeding operations in Southern Alberta, Canada. Appl. Environ. Microbiol. 2018, 84, e02713-17. [Google Scholar] [CrossRef] [Green Version]
- Narvaez-Bravo, C.; Taboada, E.N.; Mutschall, S.K.; Aslam, M. Epidemiology of antimicrobial resistant Campylobacter spp. isolated from retail meats in Canada. Int. J. Food Microbiol. 2017, 253, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Llarena, A.K.; Sivonen, K.; Hanninen, M.L. Campylobacter jejuni prevalence and hygienic quality of retail bovine ground meat in Finland. Lett. Appl. Microbiol. 2014, 58, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.; Jordan, D.; Morris, S.; Jenson, I.; Sumner, J. A national survey of the microbiological quality of retail raw meats in Australia. J. Food Prot. 2008, 71, 1232–1236. [Google Scholar] [CrossRef]
- Zhao, C.; Ge, B.; De Villena, J.; Sudler, R.; Yeh, E.; Zhao, S.; White, D.G.; Wagner, D.; Meng, J. Prevalence of Campylobacter spp., Escherichia coli, and Salmonella serovars in retail chicken, turkey, pork, and beef from the greater Washington, D.C., area. Appl. Environ. Microbiol. 2001, 67, 5431–5436. [Google Scholar] [CrossRef] [Green Version]
- Hong, J.; Kim, J.M.; Jung, W.K.; Kim, S.H.; Bae, W.; Koo, H.C.; Gil, J.; Kim, M.; Ser, J.; Park, Y.H. Prevalence and antibiotic resistance of Campylobacter spp. isolated from chicken meat, pork, and beef in Korea, from 2001 to 2006. J. Food Prot. 2007, 70, 860–866. [Google Scholar] [CrossRef]
- Clark, C.G.; Price, L.; Ahmed, R.; Woodward, D.L.; Melito, P.L.; Rodgers, F.G.; Jamieson, F.; Ciebin, B.; Li, A.; Ellis, A. Characterization of waterborne outbreak-associated Campylobacter jejuni, Walkerton, Ontario. Emerg. Infect. Dis. 2003, 9, 1232–1241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teunis, P.; Van den Brandhof, W.; Nauta, M.; Wagenaar, J.; Van den Kerkhof, H.; Van Pelt, W. A reconsideration of the Campylobacter dose-response relation. Epidemiol. Infect. 2005, 133, 583–592. [Google Scholar] [CrossRef]
- Revez, J.; Zhang, J.; Schott, T.; Kivisto, R.; Rossi, M.; Hanninen, M.L. Genomic variation between Campylobacter jejuni isolates associated with milk-borne-disease outbreaks. J. Clin. Microbiol. 2014, 52, 2782–2786. [Google Scholar] [CrossRef] [Green Version]
- Longenberger, A.H.; Palumbo, A.J.; Chu, A.K.; Moll, M.E.; Weltman, A.; Ostroff, S.M. Campylobacter jejuni infections associated with unpasteurized milk-multiple states, 2012. Clin. Infect. Dis. 2013, 57, 263–266. [Google Scholar] [CrossRef] [Green Version]
- Hasselback, P. Feedlot Alley and Enteric Illness: Are They Related or Is Southern Alberta Just a Wonderful Place for Humans, Cattle and Bugs to Live; Canadian Laboratory Medicine Congress: Calgary, AB, Canada, 2002. [Google Scholar]
- Pintar, K.D.M.; Thomas, K.M.; Christidis, T.; Otten, A.; Nesbitt, A.; Marshall, B.; Pollari, F.; Hurst, M.; Ravel, A. A comparative exposure assessment of Campylobacter in Ontario, Canada. Risk Anal. 2017, 37, 677–715. [Google Scholar] [CrossRef]
- Jolley, K.A.; Bray, J.E.; Maiden, M.C.J. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 2018, 3, 124. [Google Scholar] [CrossRef] [PubMed]
- Hermans, D.; Pasmans, F.; Messens, W.; Martel, A.; Van Immerseel, F.; Rasschaert, G.; Heyndrickx, M.; Van Deun, K.; Haesebrouck, F. Poultry as a host for the zoonotic pathogen Campylobacter jejuni. Vector Borne Zoonotic Dis. 2012, 12, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Hald, B.; Skovgard, H.; Pedersen, K.; Bunkenborg, H. Influxed insects as vectors for Campylobacter jejuni and Campylobacter coli in Danish broiler houses. Poult. Sci. 2008, 87, 1428–1434. [Google Scholar] [CrossRef] [PubMed]
- Sahin, O.; Kassem, I.I.; Shen, Z.; Lin, J.; Rajashekara, G.; Zhang, Q. Campylobacter in poultry: Ecology and potential interventions. Avian. Dis. 2015, 59, 185–200. [Google Scholar] [CrossRef]
- Bahrndorff, S.; Rangstrup-Christensen, L.; Nordentoft, S.; Hald, B. Foodborne disease prevention and broiler chickens with reduced Campylobacter infection. Emerg. Infect. Dis. 2013, 19, 425–430. [Google Scholar] [CrossRef] [Green Version]
- Hald, B.; Sommer, H.M.; Skovgard, H. Use of fly screens to reduce Campylobacter spp. introduction in broiler houses. Emerg. Infect. Dis. 2007, 13, 1951–1953. [Google Scholar] [CrossRef]
- Karenlampi, R.; Rautelin, H.; Schonberg-Norio, D.; Paulin, L.; Hanninen, M.L. Longitudinal study of Finnish Campylobacter jejuni and C. coli isolates from humans, using multilocus sequence typing, including comparison with epidemiological data and isolates from poultry and cattle. Appl. Environ. Microbiol. 2007, 73, 148–155. [Google Scholar] [CrossRef] [Green Version]
- Nylen, G.; Dunstan, F.; Palmer, S.R.; Andersson, Y.; Bager, F.; Cowden, J.; Feierl, G.; Galloway, Y.; Kapperud, G.; Megraud, F.; et al. The seasonal distribution of Campylobacter infection in nine European countries and New Zealand. Epidemiol. Infect. 2002, 128, 383–390. [Google Scholar] [CrossRef]
- Hastings, R.; Colles, F.M.; McCarthy, N.D.; Maiden, M.C.; Sheppard, S.K. Campylobacter genotypes from poultry transportation crates indicate a source of contamination and transmission. J. Appl. Microbiol. 2011, 110, 266–276. [Google Scholar] [CrossRef] [Green Version]
- Oh, E.; Chui, L.; Bae, J.; Li, V.; Ma, A.; Mutschall, S.K.; Taboada, E.N.; McMullen, L.M.; Jeon, B. Frequent implication of multistress-tolerant Campylobacter jejuni in human infections. Emerg. Infect. Dis. 2018, 24, 1037–1044. [Google Scholar] [CrossRef] [Green Version]
- Sheppard, S.K.; Cheng, L.; Meric, G.; de Haan, C.P.; Llarena, A.K.; Marttinen, P.; Vidal, A.; Ridley, A.; Clifton-Hadley, F.; Connor, T.R.; et al. Cryptic ecology among host generalist Campylobacter jejuni in domestic animals. Mol. Ecol. 2014, 23, 2442–2451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheppard, S.K.; Colles, F.; Richardson, J.; Cody, A.J.; Elson, R.; Lawson, A.; Brick, G.; Meldrum, R.; Little, C.L.; Owen, R.J.; et al. Host association of Campylobacter genotypes transcends geographic variation. Appl. Environ. Microbiol. 2010, 76, 5269–5277. [Google Scholar] [CrossRef] [PubMed]
- Ontario Government. Water Requirements for Livestock. 2019. Available online: http://omafra.gov.on.ca/english/livestock/beef/news/vbn0708a5.htm (accessed on 4 November 2022).
- Inglis, G.D.; McAllister, T.A.; Larney, F.J.; Topp, E. Prolonged survival of Campylobacter species in bovine manure compost. Appl. Environ. Microbiol. 2010, 76, 1110–1119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alberta Government. Beneficial Management Practices—Environmental Manual for Feedlot Producers in Alberta. 2002. Available online: https://www.cattlefeeders.ca/wp-content/uploads/2015/04/Feedlot-Environmental-BMP-Manual.pdf (accessed on 4 November 2022).
- van de Giessen, A.W.; Bloemberg, B.P.; Ritmeester, W.S.; Tilburg, J.J. Epidemiological study on risk factors and risk reducing measures for campylobacter infections in Dutch broiler flocks. Epidemiol. Infect. 1996, 117, 245–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van de Giessen, A.W.; Tilburg, J.J.; Ritmeester, W.S.; van der Plas, J. Reduction of campylobacter infections in broiler flocks by application of hygiene measures. Epidemiol. Infect. 1998, 121, 57–66. [Google Scholar] [CrossRef]
- Ellis-Iversen, J.; Jorgensen, F.; Bull, S.; Powell, L.; Cook, A.J.; Humphrey, T.J. Risk factors for Campylobacter colonisation during rearing of broiler flocks in Great Britain. Prev. Vet. Med. 2009, 89, 178–184. [Google Scholar] [CrossRef]
- Ridley, A.M.; Morris, V.K.; Cawthraw, S.A.; Ellis-Iversen, J.; Harris, J.A.; Kennedy, E.M.; Newell, D.G.; Allen, V.M. Longitudinal molecular epidemiological study of thermophilic campylobacters on one conventional broiler chicken farm. Appl. Environ. Microbiol. 2011, 77, 98–107. [Google Scholar] [CrossRef] [Green Version]
- Ellis-Iversen, J.; Ridley, A.; Morris, V.; Sowa, A.; Harris, J.; Atterbury, R.; Sparks, N.; Allen, V. Persistent environmental reservoirs on farms as risk factors for Campylobacter in commercial poultry. Epidemiol. Infect. 2012, 140, 916–924. [Google Scholar] [CrossRef]
- Sommer, H.M.; Heuer, O.E.; Sorensen, A.I.; Madsen, M. Analysis of factors important for the occurrence of Campylobacter in Danish broiler flocks. Prev. Vet. Med. 2013, 111, 100–111. [Google Scholar] [CrossRef]
- Thepault, A.; Meric, G.; Rivoal, K.; Pascoe, B.; Mageiros, L.; Touzain, F.; Rose, V.; Beven, V.; Chemaly, M.; Sheppard, S.K. Genome-wide identification of host-segregating epidemiological markers for source attribution in Campylobacter jejuni. Appl. Environ. Microbiol. 2017, 83, 100–111. [Google Scholar] [CrossRef] [Green Version]
- Thepault, A.; Rose, V.; Quesne, S.; Poezevara, T.; Beven, V.; Hirchaud, E.; Touzain, F.; Lucas, P.; Meric, G.; Mageiros, L.; et al. Ruminant and chicken: Important sources of campylobacteriosis in France despite a variation of source attribution in 2009 and 2015. Sci. Rep. 2018, 8, 9305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Public Health Agency of Canada. Notifiable diseases online. 2018. Available online: http://diseases.canada.ca/notifiable/ (accessed on 4 November 2022).
- Alberta Health and Wellness. Health Trends in Alberta. 2007. Available online: https://open.alberta.ca/dataset/a4731b3e-9848-42a2-be69-eeae8f9bcf41/resource/9d244198-8fcc-43ee-bfee-f859ec7e1545/download/trends-2007-health.pdf (accessed on 4 November 2022).
- Food and Agricultural Organization of the United Nations. Live Animals. 2019. Available online: http://www.fao.org/faostat/en/#data/QA (accessed on 4 November 2022).
- Inglis, G.D.; Boras, V.F.; Houde, A. Enteric campylobacteria and RNA viruses associated with healthy and diarrheic humans in the Chinook Health Region of southwestern Alberta, Canada. J. Clin. Microbiol. 2011, 49, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Klena, J.D.; Parker, C.T.; Knibb, K.; Ibbitt, J.C.; Devane, P.M.; Horn, S.T.; Miller, W.G.; Konkel, M.E. Differentiation of Campylobacter coli, Campylobacter jejuni, Campylobacter lari, and Campylobacter upsaliensis by a multiplex PCR developed from the nucleotide sequence of the lipid A gene lpxA. J. Clin. Microbiol. 2004, 42, 5549–5557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inglis, G.D.; Kalischuk, L.D. Use of PCR for direct detection of Campylobacter species in bovine feces. Appl. Environ. Microbiol. 2003, 69, 3435–3447. [Google Scholar] [CrossRef] [Green Version]
- Inglis, G.D.; Zaytsoff, S.J.M.; Selinger, L.B.; Taboada, E.N.; Uwiera, R.R.E. Therapeutic administration of enrofloxacin in mice does not select for fluoroquinolone resistance in Campylobacter jejuni. Can. J. Microbiol. 2018, 64, 681–694. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI). Blastn; 2021. Available online: https://www.ncbi.nlm.nih.gov/ (accessed on 4 November 2022).
- Ribosomal Database Project. RDP Seqmatch. 2021. Available online: http://rdp.cme.msu.edu/ (accessed on 4 November 2022).
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria 2021. Available online: https://www.R-project.org/ (accessed on 4 November 2022).
- Government of Canada. Agricultural operation characteristics Canada. Statistics Canada2016. Available online: https://www150.statcan.gc.ca/n1/pub/95-634-x/2017001/article/54899-eng.htm (accessed on 4 November 2022).







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© Her Majesty the Queen in Right of Canada as represented by the Minister of Agriculture and Agri-Food, 2021 and the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira, J.S.; Boras, V.F.; Hetman, B.M.; Taboada, E.N.; Inglis, G.D. Molecular Epidemiological Evidence Implicates Cattle as a Primary Reservoir of Campylobacter jejuni Infecting People via Contaminated Chickens. Pathogens 2022, 11, 1366. https://doi.org/10.3390/pathogens11111366
Teixeira JS, Boras VF, Hetman BM, Taboada EN, Inglis GD. Molecular Epidemiological Evidence Implicates Cattle as a Primary Reservoir of Campylobacter jejuni Infecting People via Contaminated Chickens. Pathogens. 2022; 11(11):1366. https://doi.org/10.3390/pathogens11111366
Chicago/Turabian StyleTeixeira, Januana S., Valerie F. Boras, Benjamin M. Hetman, Eduardo N. Taboada, and G. Douglas Inglis. 2022. "Molecular Epidemiological Evidence Implicates Cattle as a Primary Reservoir of Campylobacter jejuni Infecting People via Contaminated Chickens" Pathogens 11, no. 11: 1366. https://doi.org/10.3390/pathogens11111366





