First Record of the Rare Species Aeromonas lusitana from Rainbow Trout (Oncorhynchus mykiss, Walbaum): Comparative Analysis with the Existing Strains
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Phenotypic Characterization and Antimicrobial Susceptibility Testing
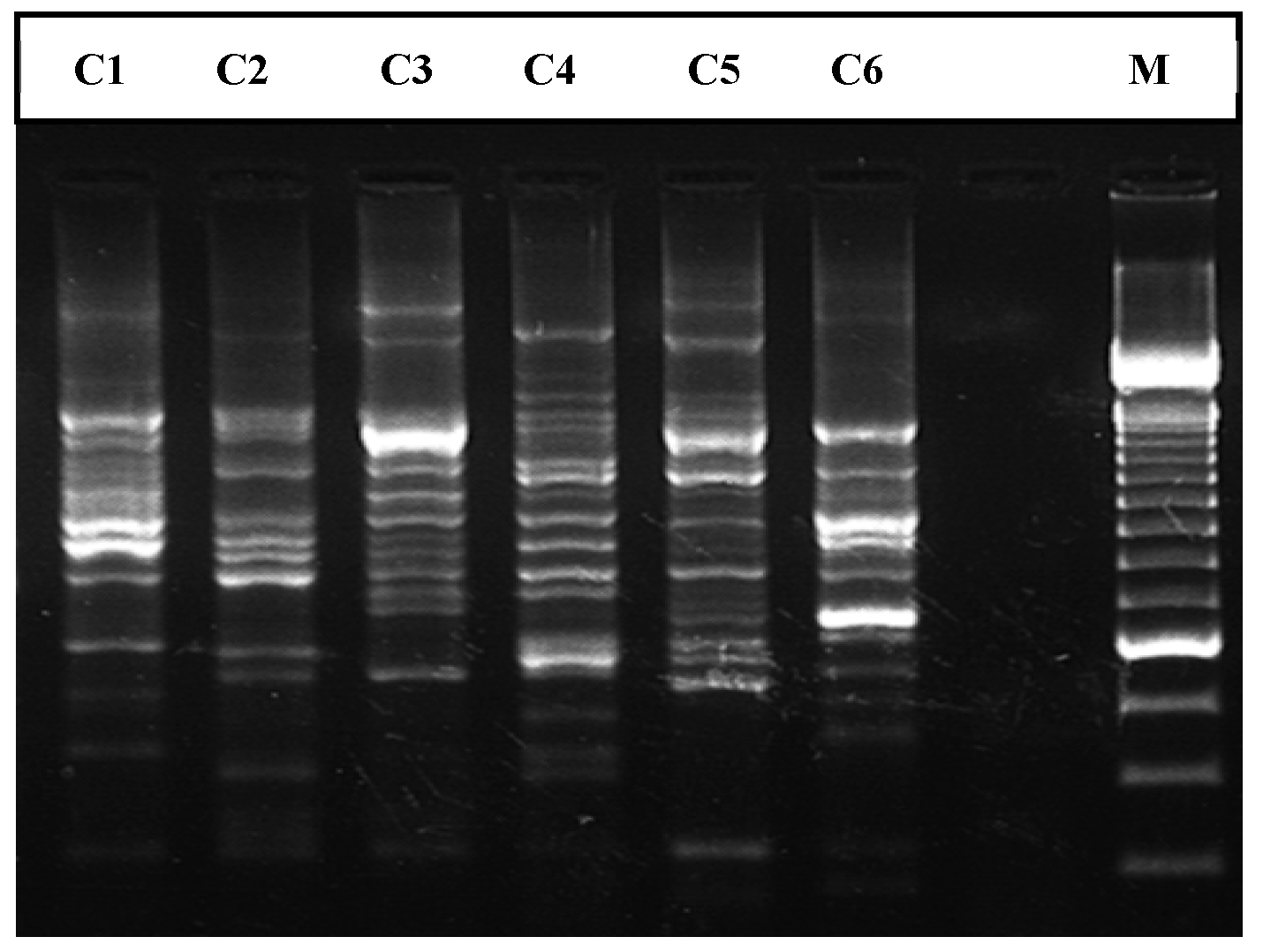
2.3. Fingerprinting by ERIC-PCR
2.4. Putative Virulence Genes Detection
2.5. Genome Sequencing, Assembling and Annotation
2.6. Phylogenetic Analysis Based on the Genome
2.7. Genome Information and Comparative Analysis
2.8. Genomic Indexes
2.9. Pangenome and Core Genes Analysis
2.10. Comparison of Predicted Protein Sequences
2.11. Protein Count and Distribution in Subsystems
2.12. Citrate Synthase Sequence Analysis in A. lusitana
3. Results and Discussion
3.1. Fingerprinting by ERIC-PCR
3.2. Phenotypic Characterization and Antimicrobial Susceptibility Testing
3.3. Putative Virulence Genes Detection
3.4. Phylogenetic Analysis with 7 Housekeeping Genes Extracted from the Genomes
3.5. Comparison of ANI and isDDH Values Obtained between A. lusitana and Its Closest Species
3.6. Genomic Information and Its Comparison to Other Related and Non-Related Species
3.7. Pangenome and Accessory Genome Analysis
3.8. Comparison of Predicted Protein Sequences
3.9. Protein Count and Distribution in Subsystems
3.10. Citrate Synthase Sequence Analysis in A. lusitana
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martin-Carnahan, A.; Joseph, S.W. Aeromonadales Ord. Nov. In Bergey’s Manual of Systematic Bacteriology; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Janda, J.M.; Abbott, S.L. The genus Aeromonas: Taxonomy, pathogenicity and infection. Clin. Microbiol. Rev. 2010, 23, 35–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beaz-Hidalgo, R.; Figueras, M.J. Aeromonas spp. whole genomes and virulence factors implicated in fish disease. J. Fish Dis. 2013, 36, 371–388. [Google Scholar] [CrossRef] [PubMed]
- Figueras, M.J.; Beaz-Hidalgo, R. Aeromonas infections in humans. In Aeromonas; Academic Press: Norfolk, UK, 2015; pp. 65–108. [Google Scholar]
- Figueras Salvat, M.J.; Ashbolt, N. Aeromonas. In Water and Sanitation for the 21st Century: Health and Microbiological Aspects of Excreta and Wastewater Management (Global Water Pathogen Project); Rose, J.B., Jiménez-Cisneros, B., Eds.; Publisher: Norfolk, UK, 2019. [Google Scholar]
- Barger, P.C.; Liles, M.R.; Newton, J.C. Type II secretion is essential for virulence of the emerging fish pathogen, hypervirulent Aeromonas hydrophila. Front. Vet. Sci. 2020, 706, 574113. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.T.; Fernández-Bravo, A.; Sanchis, M.; Mayayo, E.; Figueras, M.J.; Charette, S.J. Investigation of the virulence and genomics of Aeromonas salmonicida strains isolated from human patients. Infect. Genet. Evol. 2019, 68, 1–9. [Google Scholar] [CrossRef]
- Hossain, M.J.; Sun, D.; McGarey, D.J.; Wrenn, S.; Alexander, L.M.; Martino, M.E.; Xing, Y.; Terhune, J.S.; Liles, M.R. An Asian origin of virulent Aeromonas hydrophila responsible for disease epidemics in United States-farmed catfish. MBio 2014, 5, e00848-14. [Google Scholar] [CrossRef] [Green Version]
- Barger, P.C.; Liles, M.R.; Beck, B.H.; Newton, J.C. Correction to: Differential production and secretion of potentially toxigenic extracellular proteins from hypervirulent Aeromonas hydrophila under biofilm and planktonic culture. BMC Microbiol. 2021, 21, 8. [Google Scholar] [CrossRef]
- Hossain, M.J.; Waldbieser, G.C.; Sun, D.; Capps, N.K.; Hemstreet, W.B.; Carlisle, K.; Griffin, M.J.; Khoo, L.; Goodwin, A.E.; Sonstegard, T.S.; et al. Implication of lateral genetic transfer in the emergence of Aeromonas hydrophila isolates of epidemic outbreaks in channel catfish. PLoS ONE 2013, 8, e80943. [Google Scholar] [CrossRef] [Green Version]
- Romero, A.; Saraceni, P.R.; Merino, S.; Figueras, A.; Tomás, J.M.; Novoa, B. The animal model determines the results of Aeromonas virulence factors. Front. Microbiol. 2016, 7, 1574. [Google Scholar] [CrossRef] [Green Version]
- Galindo, C.; Sha, J.; Fadl, A.; Pillai, L.; Chopra, A. Host immune responses to Aeromonas virulence factors. Curr. Immunol. Rev. 2006, 2, 13–26. [Google Scholar] [CrossRef]
- Tomás, J.M. The main Aeromonas pathogenic factors. ISRN Microbiol. 2012, 2012, 256261. [Google Scholar] [CrossRef]
- Mendoza-Barberá, E.; Merino, S.; Tomás, J. Surface glucan structures in Aeromonas spp. Mar. Drugs 2021, 19, 649. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Bravo, A.; Figueras, M.J. An update on the genus Aeromonas: Taxonomy, epidemiology, and pathogenicity. Microorganisms 2020, 8, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-Murcia, A.; Beaz-Hidalgo, R.; Navarro, A.; Carvalho, M.J.; Aravena-Román, M.; Correia, A.; Figueras, M.J.; Saavedra, M.J. Aeromonas lusitana sp. nov., isolated from untreated water and vegetables. Curr. Microbiol. 2016, 72, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Colston, S.M.; Navarro, A.; Martinez-Murcia, A.J.; Graf, J. Draft genome sequence of Aeromonas lusitana sp. nov. strain DSM 24905T, isolated from a hot spring in Vila- Real, Portugal. Genome Announc. 2018, 6, e00226-18. [Google Scholar] [CrossRef] [Green Version]
- Vega-Sánchez, V.; Latif-Eugenín, F.; Soriano-Vargas, E.; Beaz-Hidalgo, R.; Figueras, M.J.; Aguilera-Arreola, M.G.; Castro-Escarpulli, G. Re-identification of Aeromonas isolates from rainbow trout and incidence of class 1 integron and β-lactamase genes. Vet. Microbiol. 2014, 172, 528–533. [Google Scholar] [CrossRef]
- Vega-Sánchez, V.; Acosta-Dibarrat, J.; Vega-Castillo, F.; Castro-Escarpulli, G.; Aguilera-Arreola, M.G.; Soriano-Vargas, E. Phenotypical characteristics, genetic identification, and antimicrobial sensitivity of Aeromonas species isolated from farmed rainbow trout (Onchorynchus mykiss) in Mexico. Acta Trop. 2014, 130, 76–79. [Google Scholar] [CrossRef]
- Carvalho, M.J.; Martínez-Murcia, A.; Esteves, A.C.; Correia, A.; Saavedra, M.J. Phylogenetic diversity, antibiotic resistance and virulence traits of Aeromonas spp. from untreated waters for human consumption. Int. J. Food Microbiol. 2012, 159, 230–239. [Google Scholar] [CrossRef]
- Abbott, S.L.; Cheung, W.K.W.; Janda, J.M. The genus Aeromonas: Biochemical characteristics, atypical reactions, and phenotypic identification schemes. J. Clin. Microbiol. 2003, 41, 2348–2357. [Google Scholar] [CrossRef] [Green Version]
- Beaz-Hidalgo, R.; Alperi, A.; Buján, N.; Romalde, J.L.; Figueras, M.J. Comparison of phenotypical and genetic identification of Aeromonas strains isolated from diseased fish. Syst. Appl. Microbiol. 2010, 33, 149–153. [Google Scholar] [CrossRef]
- CLSI Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing, 30th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2020. [Google Scholar]
- Versalovic, J.; Koeuth, T.; Lupski, R. Distribution of repetitive DNA sequences in eubacteria and application to fingerpriting of bacterial genomes. Nucleic Acids Res. 1991, 19, 6823–6831. [Google Scholar] [CrossRef]
- Merino, S.; Gavín, R.; Vilches, S.; Shaw, J.G.; Tomás, J.M. A Colonization factor (production of lateral flagella) of mesophilic Aeromonas spp. is inactive in Aeromonas salmonicida strains. Appl. Environ. Microbiol. 2003, 69, 663–667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soler, L.; Figueras, M.J.; Chacón, M.R.; Vila, J.; Marco, F.; Martinez-Murcia, A.J.; Guarro, J. Potential virulence and antimicrobial susceptibility of Aeromonas popoffii recovered from freshwater and seawater. FEMS Immunol. Med. Microbiol. 2002, 32, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Kingombe, C.I.B.; Huys, G.; Tonolla, M.; Albert, M.J.; Swings, J.; Peduzzi, R.; Jemmi, T. PCR Detection, characterization, and distribution of virulence genes in Aeromonas spp. Appl. Environ. Microbiol. 1999, 65, 5293–5302. [Google Scholar] [CrossRef] [Green Version]
- Aguilera-Arreola, M.G.; Hernández-Rodríguez, C.; Zúñiga, G.; Figueras, M.J.; Castro-Escarpulli, G. Aeromonas hydrophila clinical and environmental ecotypes as revealed by genetic diversity and virulence genes. FEMS Microbiol. Lett. 2005, 242, 231–240. [Google Scholar] [CrossRef] [Green Version]
- Chopra, A.K.; Peterson, J.W.; Xu, X.J.; Coppenhaver, D.H.; Houston, C.W. Molecular and biochemical characterization of a heat-labile cytotonic enterotoxin from Aeromonas hydrophila. Microb. Pathog. 1996, 21, 357–377. [Google Scholar] [CrossRef]
- Chacón, M.R.; Figueras, M.J.; Castro-Escarpulli, G.; Soler, L.; Guarro, J. Distribution of virulence genes in clinical and environmental isolates of Aeromonas spp. Antonie Van Leeuwenhoek 2003, 84, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Castro-Escarpulli, G.; Figueras, M.J.; Aguilera-Arreola, G.; Soler, L.; Fernández-Rendón, E.; Aparicio, G.O.; Guarro, J.; Chacón, M.R. Characterization of Aeromonas spp. isolated from frozen fish intended for human consumption in Mexico. Int. J. Food Microbiol. 2003, 84, 41–49. [Google Scholar] [CrossRef]
- Chacón, M.R.; Soler, L.; Groisman, E.A.; Guarro, J.; Figueras, M.J. Type III secretion system genes in clinical Aeromonas isolates. J. Clin. Microbiol. 2004, 42, 1285–1287. [Google Scholar] [CrossRef] [Green Version]
- Braun, M.; Stuber, K.; Schlatter, Y.; Wahli, T.; Kuhnert, P.; Frey, J. Characterization of an ADP-Ribosyltransferase toxin (AexT) from Aeromonas salmonicida subsp. salmonicida. J. Bacteriol. 2002, 42, 1851–1858. [Google Scholar] [CrossRef] [Green Version]
- Fehr, D.; Casanova, C.; Liverman, A.; Blazkova, H.; Orth, K.; Dobbelaere, D.; Frey, J.; Burr, S.E. AopP, a type III effector protein of Aeromonas salmonicida, inhibits the NF-ΚB signalling pathway. Microbiology 2006, 152, 2809–2818. [Google Scholar] [CrossRef]
- Wang, G.; Clark, C.G.; Rodgerst, F.G. Detection in Escherichia coli of the genes encoding the major virulence factors, the genes defining the O157:H7 serotype, and components of the type 2 shiga toxin family by multiplex PCR. J. Clin. Microbiol. 2002, 40, 3613–3619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muniesa, M.; Hammerl, J.A.; Hertwig, S.; Appel, B.; Brüssow, H. Shiga toxin-producing Escherichia Coli O104:H4: A new challenge for microbiology. Appl. Environ. Microbiol. 2012, 78, 4065–4073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larsen, M.V.; Cosentino, S.; Rasmussen, S.; Friis, C.; Hasman, H.; Marvig, R.L.; Jelsbak, L.; Sicheritz-Pontén, T.; Ussery, D.W.; Aarestrup, F.M.; et al. Multilocus sequence typing of total-genome-sequenced bacteria. J. Clin. Microbiol. 2012, 50, 1355–1361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aziz, R.K.; Bartels, D.; Best, A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef] [Green Version]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res. 2014, 42, D206–D214. [Google Scholar] [CrossRef]
- Lowe, T.M.; Chan, P.P. tRNAscan-SE on-line: Integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res. 2016, 44, W54–W57. [Google Scholar] [CrossRef]
- Colston, S.M.; Fullmer, M.S.; Beka, L.; Lamy, B.; Peter Gogarten, J.; Graf, J. Bioinformatic genome comparisons for taxonomic and phylogenetic assignments using Aeromonas as a test case. MBio 2014, 5, e02136-14. [Google Scholar] [CrossRef] [Green Version]
- Beaz-Hidalgo, R.; Latif-Eugenín, F.; Hossain, M.J.; Berg, K.; Niemi, R.M.; Rapala, J.; Lyra, C.; Liles, M.R.; Figueras, M.J. Aeromonas aquatica sp. nov., Aeromonas finlandiensis sp. nov. and Aeromonas lacus sp. nov. isolated from finnish waters associated with cyanobacterial blooms. Syst. Appl. Microbiol. 2015, 38, 161–168. [Google Scholar] [CrossRef]
- Esteve, C.; Gutierrez, M.C.; Ventosa, A. Aeromonas encheleia sp. nov., isolated from European eels. Int. J. Syst. Bacteriol. 1995, 45, 462–466. [Google Scholar] [CrossRef] [Green Version]
- Seshadri, R.; Joseph, S.W.; Chopra, A.K.; Sha, J.; Shaw, J.; Graf, J.; Haft, D.; Wu, M.; Ren, Q.; Rosovitz, M.J.; et al. Genome sequence of Aeromonas hydrophila ATCC 7966T: Jack of all trades. J. Bacteriol. 2006, 188, 8272–8282. [Google Scholar] [CrossRef]
- Reith, M.E.; Singh, R.K.; Curtis, B.; Boyd, J.M.; Bouevitch, A.; Kimball, J.; Munholland, J.; Murphy, C.; Sarty, D.; Williams, J.; et al. The genome of Aeromonas salmonicida subsp. salmonicida A449: Insights into the evolution of a fish pathogen. BMC Genom. 2008, 9, 427. [Google Scholar] [CrossRef]
- Charette, S.J.; Brochu, F.; Boyle, B.; Filion, G.; Tanaka, K.H.; Derome, N. Draft genome sequence of the virulent strain 01-B526 of the fish pathogen Aeromonas salmonicida. J. Bacteriol. 2012, 194, 722–723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, C.J.; Wang, H.C.; Chen, C.S.; Shu, H.Y.; Kao, A.W.; Chen, P.L.; Ko, W.C. Genome sequence of a novel human pathogen, Aeromonas aquariorum. J. Bacteriol. 2012, 194, 4114–4115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Liu, Y.; Zhou, Z.; Huang, H.; Ren, Y.; Zhang, Y.; Li, G.; Zhou, Z.; Wang, L. Complete genome sequence of Aeromonas veronii strain B565. J. Bacteriol. 2011, 193, 3389–3390. [Google Scholar] [CrossRef] [Green Version]
- Beatson, S.A.; De Luna, M.D.G.; Bachmann, N.L.; Alikhan, N.F.; Hanks, K.R.; Sullivan, M.J.; Wee, B.A.; Freitas-Almeida, A.C.; Dos Santos, P.A.; De Melo, J.T.B.; et al. Genome sequence of the emerging pathogen Aeromonas caviae. J. Bacteriol. 2011, 193, 1286–1287. [Google Scholar] [CrossRef] [Green Version]
- Meier-Kolthoff, J.P.; Auch, A.F.; Klenk, H.P.; Göker, M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform. 2013, 14, 60. [Google Scholar] [CrossRef] [Green Version]
- Lee, I.; Kim, Y.O.; Park, S.C.; Chun, J. OrthoANI: An improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol. 2016, 66, 1100–1103. [Google Scholar] [CrossRef]
- Brockhurst, M.A.; Harrison, E.; Hall, J.P.J.; Richards, T.; McNally, A.; MacLean, C. The ecology and evolution of pangenomes. Curr. Biol. 2019, 29, R1094–R1103. [Google Scholar] [CrossRef]
- Hassan, A.; Naz, A.; Obaid, A.; Paracha, R.Z.; Naz, K.; Awan, F.M.; Muhmmad, S.A.; Janjua, H.A.; Ahmad, J.; Ali, A. Pangenome and immuno-proteomics analysis of Acinetobacter baumannii strains revealed the core peptide vaccine targets. BMC Genom. 2016, 17, 732. [Google Scholar] [CrossRef] [Green Version]
- Lukjancenko, O.; Wassenaar, T.M.; Ussery, D.W. Comparison of 61 sequenced Escherichia coli genomes. Microb. Ecol. 2010, 60, 708–720. [Google Scholar] [CrossRef]
- Hyatt, D.; Chen, G.L.; LoCascio, P.F.; Land, M.L.; Larimer, F.W.; Hauser, L.J. Prodigal: Prokaryotic gene recognition and translation initiation site identification. BMC Bioinform. 2010, 11, 119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vesth, T.; Lagesen, K.; Acar, Ö.; Ussery, D. CMG-Biotools, a free workbench for basic comparative microbial genomics. PLoS ONE 2013, 8, e60120. [Google Scholar] [CrossRef] [PubMed]
- Ussery, D.W.; Kiil, K.; Lagesen, K.; Sicheritz-Pontén, T.; Bohlin, J.; Wassenaar, T.M. The genus Burkholderia: Analysis of 56 genomic sequences. Genome Dyn. 2009, 6, 140–157. [Google Scholar] [PubMed]
- Soler, L.; Figueras, M.J.; Chacón, M.R.; Guarro, J.; Martinez-Murcia, A.J. Comparison of three molecular methods for typing Aeromonas popoffii isolates. Antonie Van Leeuwenhoek 2003, 83, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Beaz-Hidalgo, R.; Shakèd, T.; Laviad, S.; Halpern, M.; Figueras, M.J. Chironomid egg masses harbour the clinical species Aeromonas taiwanensis and Aeromonas sanarellii. FEMS Microbiol. Lett. 2012, 337, 48–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dias, C.; Mota, V.; Martinez-Murcia, A.; Saavedra, M.J. Antimicrobial resistance patterns of Aeromonas spp. isolated from ornamental fish. J. Aquac. Res. Dev. 2012, 3, 3. [Google Scholar] [CrossRef] [Green Version]
- Aravena-Román, M.; Inglis, T.J.J.; Henderson, B.; Riley, T.V.; Chang, B.J. Antimicrobial susceptibilities of Aeromonas strains isolated from clinical and environmental sources to 26 antimicrobial agents. Antimicrob. Agents Chemother. 2012, 56, 1110–1112. [Google Scholar] [CrossRef] [Green Version]
- Lye, D.J. Gastrointestinal colonization rates for human clinical isolates of Aeromonas veronii using a mouse model. Curr. Microbiol. 2011, 63, 332. [Google Scholar] [CrossRef]
- Merino, S.; Aguilar, A.; Nogueras, M.M.; Regue, M.; Swift, S.; Tomás, J.M. Cloning, sequencing, and role in virulence of two phospholipases (A1 and C) from mesophilic Aeromonas sp. serogroup O:34. Infect. Immun. 1999, 67, 4008–4013. [Google Scholar] [CrossRef] [Green Version]
- Aravena-Román, M.; Inglis, T.J.J.; Riley, T.V.; Chang, B.J. Distribution of 13 virulence genes among clinical and environmental Aeromonas spp. in Western Australia. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1889–1895. [Google Scholar] [CrossRef]
- Pessoa, R.B.G.; de Oliveira, W.F.; Marques, D.S.C.; dos Santos Correia, M.T.; de Carvalho, E.V.M.M.; Coelho, L.C.B.B. The genus Aeromonas: A general approach. Microb. 2019, 130, 81–94. [Google Scholar] [CrossRef]
- Pablos, M.; Rodríguez-Calleja, J.M.; Santos, J.A.; Otero, A.; García-López, M.L. Occurrence of motile Aeromonas in municipal drinking water and distribution of genes encoding virulence factors. Int. J. Food Microbiol. 2009, 135, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Figueras, M.J.; Alperi, A.; Saavedra, M.J.; Ko, W.C.; Gonzalo, N.; Navarro, M.; Martínez-Murcia, A.J. Clinical relevance of the recently described species Aeromonas aquariorum. J. Clin. Microbiol. 2009, 47, 3742–3746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puthucheary, S.D.; Puah, S.M.; Chua, K.H. Molecular characterization of clinical isolates of Aeromonas species from Malaysia. PLoS ONE 2012, 7, e30205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, C.J.; Wu, J.J.; Yan, J.J.; Lee, H.C.; Lee, N.Y.; Chang, C.M.; Shih, H.-I.; Wu, H.M.; Wang, L.R.; Ko, W.C. Clinical significance and distribution of putative virulence markers of 116 consecutive clinical Aeromonas isolates in Southern Taiwan. J. Infect. 2007, 54, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Burr, S.E.; Pugovkin, D.; Wahli, T.; Segner, H.; Frey, J. Attenuated virulence of an Aeromonas salmonicida subsp. salmonicida type III secretion mutant in a rainbow trout model. Microbiology 2005, 151, 2111–2118. [Google Scholar] [CrossRef] [Green Version]
- Dacanay, A.; Knickle, L.; Solanky, K.S.; Boyd, J.M.; Walter, J.A.; Brown, L.L.; Johnson, S.C.; Reith, M. Contribution of the type III secretion system (ttss) to virulence of Aeromonas salmonicida subsp. salmonicida. Microbiology 2006, 152, 1847–1856. [Google Scholar] [CrossRef] [Green Version]
- Konstantinidis, K.T.; Tiedje, J.M. Towards a genome-based taxonomy for prokaryotes. J. Bacteriol. 2005, 187, 6258–6264. [Google Scholar] [CrossRef] [Green Version]
- Pang, M.; Jiang, J.; Xie, X.; Wu, Y.; Dong, Y.; Kwok, A.H.Y.; Zhang, W.; Yao, H.; Lu, C.; Leung, F.C.; et al. Novel insights into the pathogenicity of epidemic Aeromonas hydrophila ST251 clones from comparative genomics. Sci. Rep. 2015, 5, 9833. [Google Scholar] [CrossRef] [Green Version]
- Lee, Z.M.P.; Bussema, C.; Schmidt, T.M. Rrn DB: Documenting the number of rRNA and tRNA genes in bacteria and archaea. Nucleic Acids Res. 2009, 37, D489–D493. [Google Scholar] [CrossRef]
- Bartkova, S.; Kokotovic, B.; Dalsgaard, I. Infection routes of Aeromonas salmonicida in rainbow trout monitored in vivo by real-time bioluminescence imaging. J. Fish Dis. 2017, 40, 73–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demarta, A.; Küpfer, M.; Riegel, P.; Harf-Monteil, C.; Tonolla, M.; Peduzzi, R.; Monera, A.; José Saavedra, M.; Martínez-Murcia, A. Aeromonas tecta sp. nov., isolated from clinical and environmental sources. Syst. Appl. Microbiol. 2008, 31, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Lai, Q.; Dong, C.; Wang, W.; Shao, Z. Complete genome sequence of Alcanivorax xenomutans P40, an alkane-degrading bacterium isolated from deep seawater. Mar. Genom. 2018, 38, 1–4. [Google Scholar] [CrossRef]
- Cvjetanovic, B.; Barua, D. The seventh pandemic of cholera. Nature 1972, 239, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Duong-Nu, T.M.; Jeong, K.; Hong, S.H.; Nguyen, H.V.; Ngo, V.H.; Min, J.J.; Lee, S.E.; Rhee, J.H. All three TonB systems are required for Vibrio vulnificus CMCP6 tissue invasiveness by controlling flagellum expression. Infect. Immun. 2015, 84, 254–265. [Google Scholar] [CrossRef] [Green Version]
- Deng, W.; Burland, V.; Plunkett, G.; Boutin, A.; Mayhew, G.F.; Liss, P.; Perna, N.T.; Rose, D.J.; Mau, B.; Zhou, S.; et al. Genome sequence of Yersinia pestis KIM. J. Bacteriol. 2002, 184, 4601–4611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suksomtip, M.; Liu, P.; Anderson, T.; Tungpradabkul, S.; Wood, D.W.; Nester, E.W. Citrate synthase mutants of Agrobacterium are attenuated in virulence and display reduced Vir gene induction. J. Bacteriol. 2005, 187, 4844–4852. [Google Scholar] [CrossRef] [Green Version]
- Quandt, E.M.; Gollihar, J.; Blount, Z.D.; Ellington, A.D.; Georgiou, G.; Barrick, J.E. Fine-tuning citrate synthase flux potentiates and refines metabolic innovation in the Lenski evolution experiment. eLife 2015, 4, e09696. [Google Scholar] [CrossRef]
- Ding, Y.; Liu, X.; Chen, F.; Di, H.; Xu, B.; Zhou, L.; Deng, X.; Wu, M.; Yang, C.G.; Lan, L. Metabolic sensor governing bacterial virulence in Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2014, 111, E4981–E4990. [Google Scholar] [CrossRef]







| ESV-351 | CECT 7828T | Isolates (4) | |
|---|---|---|---|
| β-hemolysis | + | + | + |
| Indole | + b | - | - |
| MR | + | + | + |
| LDC | + | + | + |
| Glucose (gas) | + | + | + |
| Citrate | + | + | + |
| Hydrolysis of: | |||
| SDS | - | - | V |
| Aesculin | + | + | + |
| Starch | + | + | + |
| Elastase | ND | + | + |
| Acid from: | |||
| Glycerol | + | + | + |
| L-arabinose | - | - | - |
| Salicin | + | + | + |
| Utilization of: | |||
| DL-lactate | - | - | - |
| L-arabinose | - | - | - |
| Growth at: 4.5% NaCl | + | + | + |
| Antimicrobial Agent (μg) a | Results (MIC) | Breakpoints | |||
|---|---|---|---|---|---|
| ESV-351 | CECT 7828T | S | I | R | |
| Amikacin (30 μg) | S (≤8) | S (≤8) | ≤16 | 32 | ≥64 |
| Ampicillin (10 μg) | I (>16) | I (>16) | ≤8 | 16 | ≥32 |
| Amoxicillin-clavulanic acid (20/10 μg) | I (>16/8) | I (>16/8) | ≤8/4 | 16/8 | ≥32/16 |
| Aztreonam (30 μg) | S (≤1) | S (≤1) | ≤8 | 16 | ≥32 |
| Ceftazidime (30 μg) | S (≤1) | S (≤1) | ≤8 | 16 | ≥32 |
| Ceftriaxone (30 μg) | S (33) b | S (33) b | ≥21 | 14–20 | ≤13 |
| Cephalothin (30 μg) | I (>16) | I (>16) | ≤8 | 16 | ≥32 |
| Ciprofloxacin (30 μg) | S (42) b | S (42) b | ≥21 | 16–20 | ≤15 |
| Cefotaxime (30 μg) | S (≤1) | S (≤1) | ≤8 | 16–32 | ≥64 |
| Cefoxitin (30 μg) | S (≤8) | S (≤8) | ≤8 | 16 | ≥32 |
| Ciprofloxacin (5 μg) | S (≤0.5) | S (≤0.5) | ≤1 | 2 | ≥4 |
| Cefepime (30 μg) | S (≤1) | S (≤1) | ≤8 | 16 | ≥32 |
| Cefuroxime (30 μg) | S (8) b | S (8) b | ≤8 | 16 | ≥32 |
| Ertapenem (10 μg) | S (≤0.5) | S (≤0.5) | ≤2 | 4 | ≥8 |
| Gentamicin (10 μg) | S (≤2) | S (≤2) | ≤4 | 8 | ≥16 |
| Imipenem (10 μg) | S (≤1) | S (≤1) | ≤4 | 8 | ≥16 |
| Nalidixic acid (30 μg) | S (≤16) | S (≤16) | ≤8 | 16 | ≥16 |
| Piperacillin (100 μg) | S (29) b | S (29) b | ≥21 | 18–20 | ≤17 |
| Piperacillin-tazobactam (100/10 μg) | S (≤8) | S (≤8) | ≤16/4 | 32/4–64/4 | ≥128/4 |
| Tetracycline (30 μg) | S (27) b | S (27) b | ≥19 | 15–18 | ≤14 |
| Trimethoprim-sulfamethoxazole (25 μg) | S (≤2/38) | S (≤2/38) | ≤2/38 | - | ≥4/76 |
| Tigecycline (15 μg) | S (≤1) | S (≤1) | ≤1 | 2 | ≥2 |
| Tobramycin (10 μg) | S (≤2) | S (≤2) | ≤2 | 4 | ≥4 |
| Strain | Source | laf | act | ast | alt | aerA | Lipase Genes | Serine Protease Genes | ascF-G | ascV | aexT | stx1 | stx2 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ESV-351 | Gills of rainbow trout | - | + | - | - | + | + | + | - | - | - | - | - |
| CECT 7828T | Untreated water | + | - | - | - | + | + | + | + | + | + | - | - |
| A.136/15 | Untreated water | + | - | - | - | + | + | + | + | + | - | - | - |
| L8-3 | Vegetables | + | - | - | - | + | + | - | + | + | - | - | - |
| L10-4 | Vegetables | + | + | - | - | + | + | + | - | - | - | - | - |
| A.2876 | Untreated water | - | + | - | - | + | + | + | - | - | - | - | - |
| Species | Size (Mb) | Average | Protein Coding Genes | Average | tRNAs | Average | G+C (%) | Average |
|---|---|---|---|---|---|---|---|---|
| A. lusitana ESV-351 A. lusitana CECT 7828T | 4.74 4.55 | 4.64 | 4251 4069 | 4160 | 110 98 | 104 | 60.7 61.0 | 60.8 |
| Closest species | ||||||||
| A. tecta CECT 7028T A. eucrenophila CECT 4224T A. aquatica CECT 8025T A. encheleia CECT 4342T | 4.75 4.54 4.58 4.47 | 4.56 | 4267 4098 4091 4051 | 4094 | 96 101 82 94 | 95 | 60.1 61.2 61.2 62.0 | 61.2 |
| Closed genomes available at Genbank | ||||||||
| A. salmonicida A449 A. hydrophila ATCC 7966T A. caviae Ae398 A. veronii B565 A. salmonicida 01-B526 A. dhakensis AAK1 | 4.70 4.74 4.43 4.55 4.75 4.81 | 4.72 | 4388 4128 3690 4057 4179 4214 | 4153 | 110 128 ND 102 111 ND | 110 | 58.5 61.5 61.4 58.7 58.5 61.8 | 60.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Bravo, A.; Vega-Sánchez, V.; Pérez-Cataluña, A.; Latif-Eugenín, F.; Beaz-Hidalgo, R.; Martínez-Murcia, A.; Soriano-Vargas, E.; Cabrero-Martínez, O.A.; Castro-Escarpulli, G.; Figueras, M.J. First Record of the Rare Species Aeromonas lusitana from Rainbow Trout (Oncorhynchus mykiss, Walbaum): Comparative Analysis with the Existing Strains. Pathogens 2022, 11, 1299. https://doi.org/10.3390/pathogens11111299
Fernández-Bravo A, Vega-Sánchez V, Pérez-Cataluña A, Latif-Eugenín F, Beaz-Hidalgo R, Martínez-Murcia A, Soriano-Vargas E, Cabrero-Martínez OA, Castro-Escarpulli G, Figueras MJ. First Record of the Rare Species Aeromonas lusitana from Rainbow Trout (Oncorhynchus mykiss, Walbaum): Comparative Analysis with the Existing Strains. Pathogens. 2022; 11(11):1299. https://doi.org/10.3390/pathogens11111299
Chicago/Turabian StyleFernández-Bravo, Ana, Vicente Vega-Sánchez, Alba Pérez-Cataluña, Fadua Latif-Eugenín, Roxana Beaz-Hidalgo, Antonio Martínez-Murcia, Edgardo Soriano-Vargas, Omar Alejandro Cabrero-Martínez, Graciela Castro-Escarpulli, and Maria José Figueras. 2022. "First Record of the Rare Species Aeromonas lusitana from Rainbow Trout (Oncorhynchus mykiss, Walbaum): Comparative Analysis with the Existing Strains" Pathogens 11, no. 11: 1299. https://doi.org/10.3390/pathogens11111299






