Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators
Abstract
:1. Introduction
2. Results
2.1. Testing FFR Coupons with IAV and SARS-CoV-2
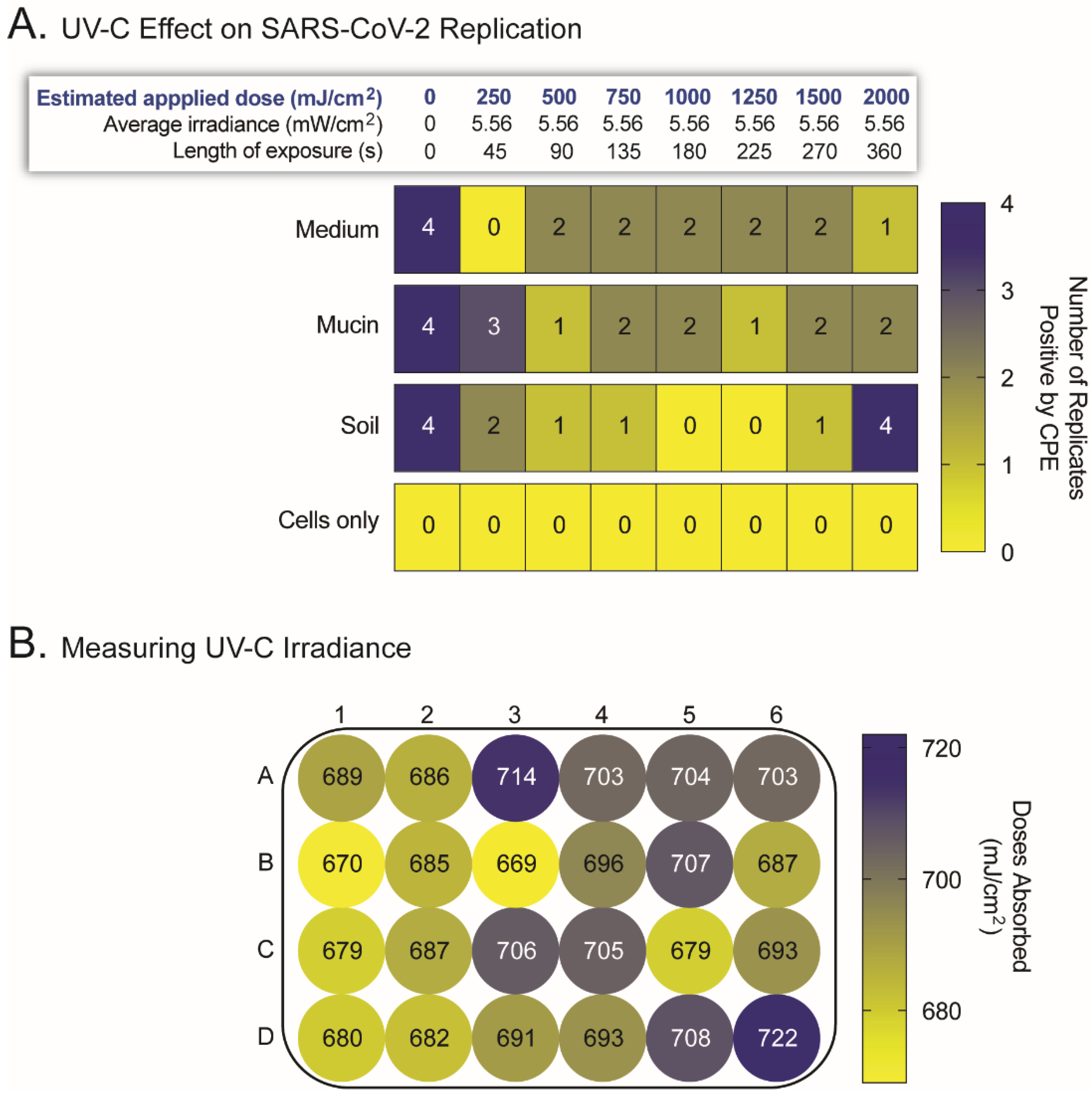
2.2. UV-C Irradation Inactivates SARS-CoV-2 but Its Efficacy Is Hampered by Surface Irregularities
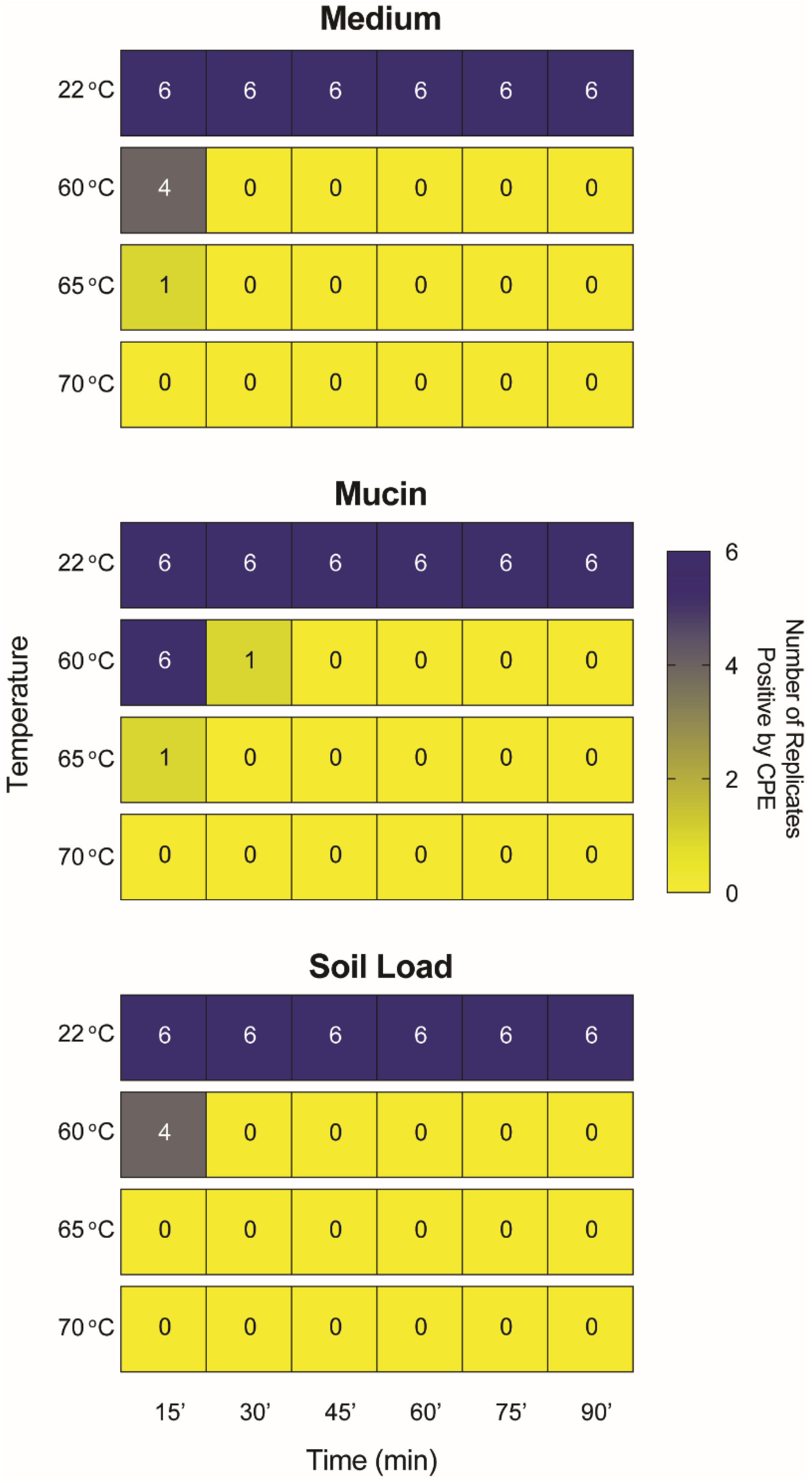
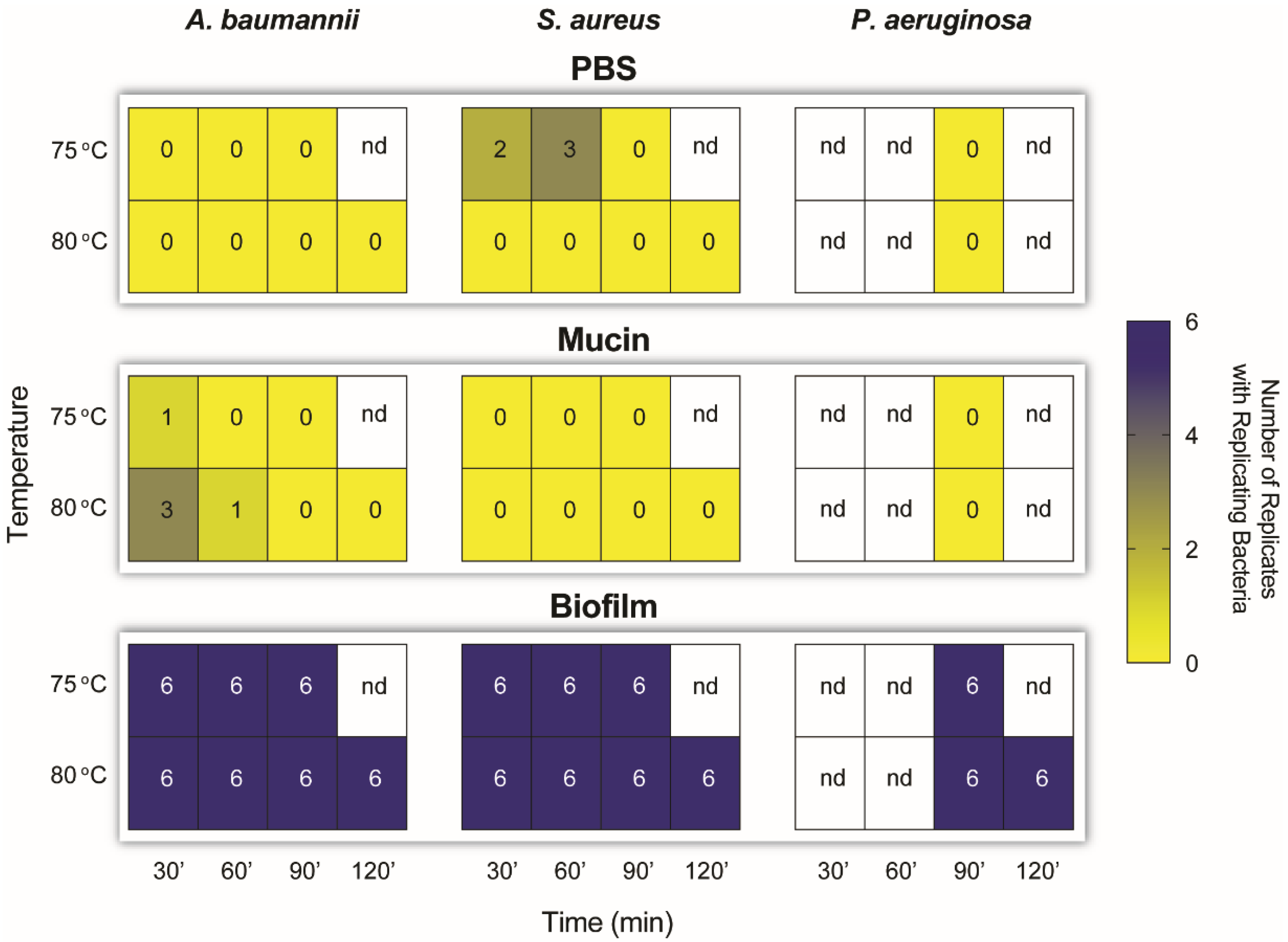
2.3. Dry Heat Inactivates SARS-CoV-2 and Planktonic Bacteria but Not Bacterial-Colony Films
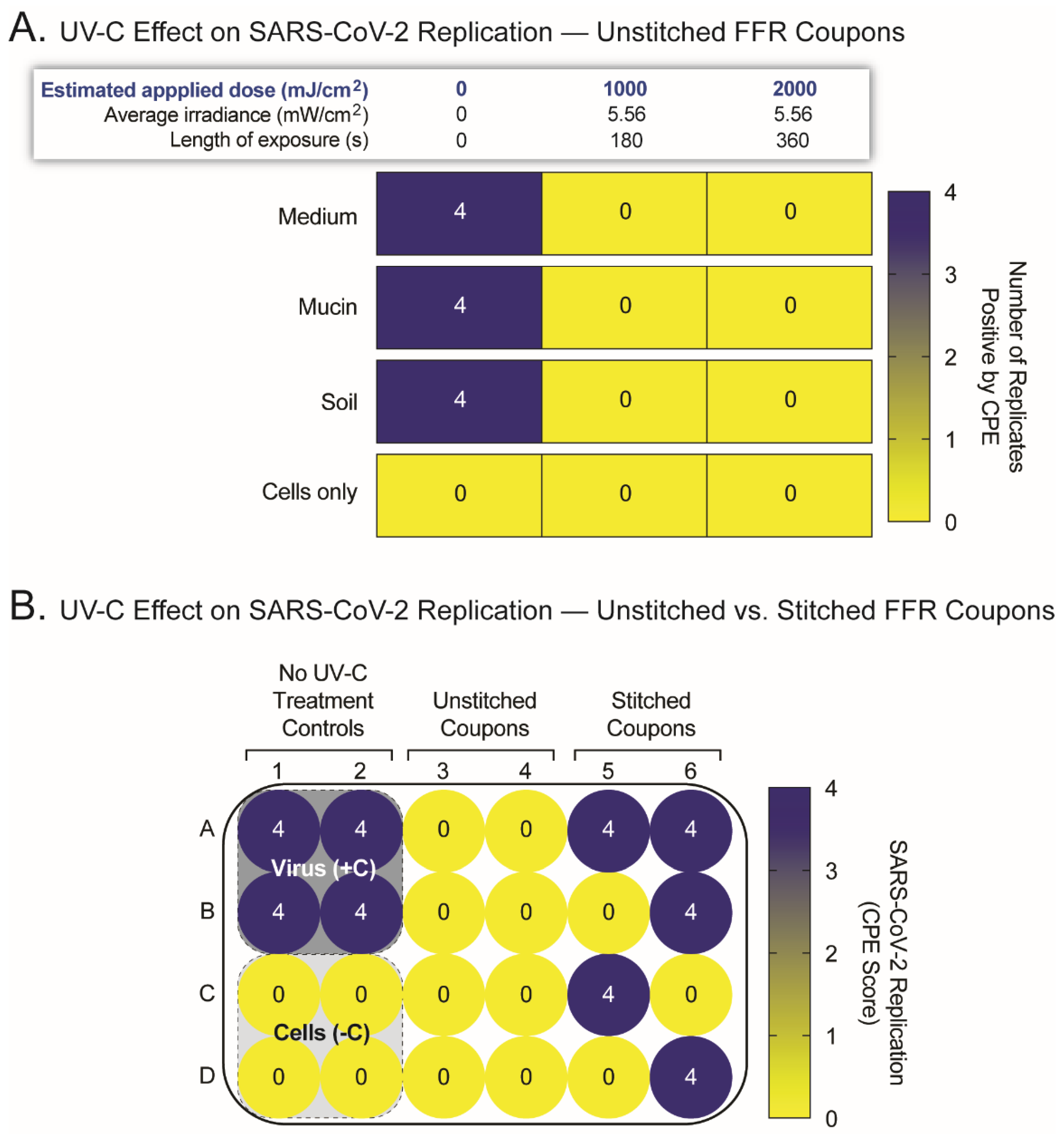
2.4. SARS-CoV-2 Remains Replication-Competent in FFR
3. Discussion
4. Materials and Methods
4.1. Cells and Viruses
4.2. Bacterial Strains and Growth Conditions
4.3. Mucin and Soil Media
4.4. FFR Coupon Preparation
4.5. Coupon Inoculation
4.6. Ultraviolet-C Irradiation Treatment
4.7. Chemical Actinometry
4.8. Dry Heat Treatment
4.9. SARS-CoV-2 Stability in FFR Coupons
4.10. SARS-CoV-2 Replication and Bacterial Growth Determination
4.10.1. SARS-CoV-2
4.10.2. Bacteria
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baker, M.G.; Wilson, N.; Anglemyer, A. Successful elimination of COVID–19 transmission in New Zealand. N. Engl. J. Med. 2020, 383, e56. [Google Scholar] [CrossRef]
- Basu, I.; Nagappan, R.; Fox-Lewis, S.; Muttaiyah, S.; McAuliffe, G. Evaluation of extraction and amplification assays for the detection of SARS-CoV-2 at Auckland Hospital laboratory during the COVID-19 outbreak in New Zealand. J. Virol. Methods 2021, 289, 114042. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Hale, M.; Reynolds, G.E. SARS-CoV-2 RT-PCR test results across symptomatic COVID-19 cases in Auckland, New Zealand, February–June 2020. Commun. Dis. Intell. 2021, 2018, 45. [Google Scholar] [CrossRef] [PubMed]
- Douglas, J.; Geoghegan, J.L.; Hadfield, J.; Bouckaert, R.; Storey, M.; Ren, X.; de Ligt, J.; French, N.; Welch, D. Real-Time Genomics for Tracking Severe Acute Respiratory Syndrome Coronavirus 2 Border Incursions after Virus Elimination, New Zealand. Emerg. Infect. Dis. 2021, 27, 2361–2368. [Google Scholar] [CrossRef]
- Jefferies, S.; French, N.; Gilkison, C.; Graham, G.; Hope, V.; Marshall, J.; McElnay, C.; McNeill, A.; Muellner, P.; Paine, S.; et al. COVID-19 in New Zealand and the impact of the national response: A descriptive epidemiological study. Lancet Public Health 2020, 5, e612–e623. [Google Scholar] [CrossRef]
- Geoghegan, J.L.; Douglas, J.; Ren, X.; Storey, M.; Hadfield, J.; Silander, O.K.; Freed, N.E.; Jelley, L.; Jefferies, S.; Sherwood, J.; et al. Use of Genomics to Track Coronavirus Disease Outbreaks, New Zealand. Emerg. Infect. Dis. 2021, 27, 1317–1322. [Google Scholar] [CrossRef]
- Burki, T. Global shortage of personal protective equipment. Lancet Infect. Dis. 2020, 20, 785–786. [Google Scholar] [CrossRef]
- Hoernke, K.; Djellouli, N.; Andrews, L.; Lewis-Jackson, S.; Manby, L.; Martin, S.; Vanderslott, S.; Vindrola-Padros, C. Frontline healthcare workers’ experiences with personal protective equipment during the COVID-19 pandemic in the UK: A rapid qualitative appraisal. BMJ Open 2021, 11, e046199. [Google Scholar] [CrossRef]
- Bauchner, H.; Fontanarosa, P.B.; Livingston, E.H. Conserving supply of personal protective equipment–a call for ideas. JAMA 2020, 323, 1911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nogee, D.; Tomassoni, A.J. COVID-19 and the N95 respirator shortage: Closing the gap. Infect. Control Hosp. Epidemiol. 2020, 41, 958. [Google Scholar] [CrossRef] [Green Version]
- Institute of Medicine. Reusability of Facemasks during an Influenza Pandemic: Facing the Flu; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Viscusi, D.J.; Bergman, M.S.; Eimer, B.C.; Shaffer, R.E. Evaluation of five decontamination methods for filtering facepiece respirators. Ann. Occup. Hyg. 2009, 53, 815–827. [Google Scholar]
- Bergman, M.S.; Viscusi, D.J.; Heimbuch, B.K.; Wander, J.D.; Sambol, A.R.; Shaffer, R.E. Evaluation of multiple (3-cycle) decontamination processing for filtering facepiece respirators. J. Eng. Fibers Fabr. 2010, 5, 33–41. [Google Scholar] [CrossRef] [Green Version]
- Heimbuch, B.K.; Wallace, W.H.; Kinney, K.; Lumley, A.E.; Wu, C.-Y.; Woo, M.-H.; Wander, J.D. A pandemic influenza preparedness study: Use of energetic methods to decontaminate filtering facepiece respirators contaminated with H1N1 aerosols and droplets. Am. J. Infect. Control 2011, 39, e1–e9. [Google Scholar] [CrossRef]
- Viscusi, D.J.; Bergman, M.S.; Novak, D.A.; Faulkner, K.A.; Palmiero, A.; Powell, J.; Shaffer, R.E. Impact of three biological decontamination methods on filtering facepiece respirator fit, odor, comfort, and donning ease. J. Occup. Environ. Hyg. 2011, 8, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Lore, M.B.; Heimbuch, B.K.; Brown, T.L.; Wander, J.D.; Hinrichs, S.H. Effectiveness of three decontamination treatments against influenza virus applied to filtering facepiece respirators. Ann. Occup. Hyg. 2012, 56, 92–101. [Google Scholar]
- Chughtai, A.A.; Seale, H.; Dung, T.C.; Maher, L.; Nga, P.T.; MacIntyre, C.R. Current practices and barriers to the use of facemasks and respirators among hospital-based health care workers in Vietnam. Am. J. Infect. Control 2015, 43, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Lindsley, W.G.; Martin, S.B., Jr.; Thewlis, R.E.; Sarkisian, K.; Nwoko, J.O.; Mead, K.R.; Noti, J.D. Effects of ultraviolet germicidal irradiation (UVGI) on N95 respirator filtration performance and structural integrity. J. Occup. Environ. Hyg. 2015, 12, 509–517. [Google Scholar] [CrossRef]
- Mills, D.; Harnish, D.A.; Lawrence, C.; Sandoval-Powers, M.; Heimbuch, B.K. Ultraviolet germicidal irradiation of influenza-contaminated N95 filtering facepiece respirators. Am. J. Infect. Control 2018, 46, e49–e55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Derraik, J.G.B.; Anderson, W.A.; Connelly, E.A.; Anderson, Y.C. Rapid Review of SARS-CoV-1 and SARS-CoV-2 Viability, Susceptibility to Treatment, and the Disinfection and Reuse of PPE, Particularly Filtering Facepiece Respirators. Int. J. Environ. Res. Public Health 2020, 17, 6117. [Google Scholar] [CrossRef]
- Kowalski, W.J. Ultraviolet Germicidal Irradiation Handbook UVGI for Air and Surface Disinfection; Springer: Berlin/Heidelberg, Germany; New York, NA, USA, 2009. [Google Scholar]
- Card, K.J.; Crozier, D.; Dhawan, A.; Dinh, M.; Dolson, E.; Farrokhian, N.; Gopalakrishnan, V.; Ho, E.; Jagdish, T.; King, E.S. UV sterilization of personal protective equipment with idle laboratory biosafety cabinets during the COVID-19 pandemic. MedRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Fischer, R.J.; Morris, D.H.; van Doremalen, N.; Sarchette, S.; Matson, M.J.; Bushmaker, T.; Yinda, C.K.; Seifert, S.N.; Gamble, A.; Williamson, B.N. Effectiveness of N95 respirator decontamination and reuse against SARS-CoV-2 virus. Emerg. Infect. Dis. 2020, 26, 2253. [Google Scholar] [CrossRef]
- Kumar, A.; Kasloff, S.B.; Leung, A.; Cutts, T.; Strong, J.E.; Hills, K.; Gu, F.X.; Chen, P.; Vazquez-Grande, G.; Rush, B. Decontamination of N95 masks for re-use employing 7 widely available sterilization methods. PLoS ONE 2020, 15, e0243965. [Google Scholar] [CrossRef]
- Ontiveros, C.C.; Sweeney, C.L.; Smith, C.; MacIsaac, S.; Munoz, S.; Davidson, R.; McCormick, C.; Thomas, N.; Davis, I.; Stoddart, A.K. Characterization of a commercially-available, low-pressure UV lamp as a disinfection system for decontamination of common nosocomial pathogens on N95 filtering facepiece respirator (FFR) material. Environ. Sci. Water Res. Technol. 2020, 6, 2089–2102. [Google Scholar] [CrossRef]
- Steinberg, B.E.; Aoyama, K.; McVey, M.; Levin, D.; Siddiqui, A.; Munshey, F.; Goldenberg, N.M.; Faraoni, D.; Maynes, J.T. Efficacy and safety of decontamination for N95 respirator reuse: A systematic literature search and narrative synthesis. Can. J. Anesth. 2020, 67, 1814–1823. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, Z.; Lanzarini-Lopes, M.; Sinha, S.; Rho, H.; Herckes, P.; Westerhoff, P. Germicidal ultraviolet light does not damage or impede performance of N95 masks upon multiple uses. Environ. Sci. Technol. Lett. 2020, 7, 600–605. [Google Scholar] [CrossRef]
- Abdalrhman, A.S.; Wang, C.; Manalac, A.; Weersink, M.; Yassine, A.A.; Betz, V.; Barbeau, B.; Lilge, L.; Hofmann, R. Modeling the efficiency of UV at 254 nm for disinfecting the different layers within N95 respirators. J. Biophotonics 2021, 14, e202100135. [Google Scholar] [CrossRef]
- Cadnum, J.L.; Pearlmutter, B.S.; Li, D.F.; Jencson, A.L.; Scott, J.G.; Charnas, I.C.; Donskey, C.J. Evaluation of 2 Ultraviolet-C Light Boxes for Decontamination of N95 Respirators. Pathog. Immun. 2021, 6, 104. [Google Scholar] [CrossRef]
- Golladay, G.J.; Leslie, K.A.; Zuelzer, W.A.; Cassano, A.D.; Plauny, J.J.; Daniels, F.E.; Bearman, G.; Kates, S.L. Rationale and process for N95 respirator sanitation and re-use in the COVID-19 pandemic. Infect. Control Hosp. Epidemiol. 2021, 1–5. [Google Scholar] [CrossRef]
- Grist, S.M.; Geldert, A.; Gopal, A.; Su, A.; Balch, H.B.; Herr, A.E. Current Understanding of Ultraviolet-C Decontamination of N95 Filtering Facepiece Respirators. Appl. Biosaf. 2021, 26, 90–102. [Google Scholar] [CrossRef]
- Huber, T.; Goldman, O.; Epstein, A.E.; Stella, G.; Sakmar, T.P. Principles and practice for SARS-CoV-2 decontamination of N95 masks with UV-C. Biophys. J. 2021, 120, 2927–2942. [Google Scholar] [CrossRef]
- Jureka, A.S.; Williams, C.G.; Basler, C.F. Pulsed Broad-Spectrum UV Light Effectively Inactivates SARS-CoV-2 on Multiple Surfaces and N95 Material. Viruses 2021, 13, 460. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Comtois-Bona, M.; Cortes, D.; Cimenci, C.E.; Du, Q.; Thompson, C.; Figueroa, J.D.; Franklin, V.; Liu, P.; Alarcon, E.I. Integrated photothermal decontamination device for N95 respirators. Sci. Rep. 2021, 11, 1822. [Google Scholar] [CrossRef]
- Rohit, A.; Rajasekaran, S.; Shenoy, S.; Rai, S.; Iddya, K.; Dorairajan, S.K. Reprocessing of N95 masks: Experience from a resource-limited setting in India. Int. J. Infect. Dis. 2021, 104, 41–44. [Google Scholar] [CrossRef]
- Schumm, M.A.; Hadaya, J.E.; Mody, N.; Myers, B.A.; Maggard-Gibbons, M. Filtering Facepiece Respirator (N95 Respirator) Reprocessing: A Systematic Review. JAMA 2021, 325, 1296–1317. [Google Scholar] [CrossRef] [PubMed]
- Seresirikachorn, K.; Phoophiboon, V.; Chobarporn, T.; Tiankanon, K.; Aeumjaturapat, S.; Chusakul, S.; Snidvongs, K. Decontamination and reuse of surgical masks and N95 filtering facepiece respirators during the COVID-19 pandemic: A systematic review. Infect. Control Hosp. Epidemiol. 2021, 42, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Simmons, S.E.; Carrion, R.; Alfson, K.J.; Staples, H.M.; Jinadatha, C.; Jarvis, W.R.; Sampathkumar, P.; Chemaly, R.F.; Khawaja, F.; Povroznik, M. Deactivation of SARS-CoV-2 with pulsed-xenon ultraviolet light: Implications for environmental COVID-19 control. Infect. Control Hosp. Epidemiol. 2021, 42, 127–130. [Google Scholar] [CrossRef]
- Smith, J.S.; Hanseler, H.; Welle, J.; Rattray, R.; Campbell, M.; Brotherton, T.; Moudgil, T.; Pack, T.F.; Wegmann, K.; Jensen, S. Effect of various decontamination procedures on disposable N95 mask integrity and SARS-CoV-2 infectivity. J. Clin. Transl. Sci. 2021, 5, e10. [Google Scholar] [CrossRef]
- Wanner, G.K.; Ader, D.; Caplan, R.; Padaki, A.S.; Ravert, D.; Drees, M. Evaluation of N95 Respirator Ultraviolet Decontamination and Clinical Reuse with Quantitative Fit Testing. Infect. Control Hosp. Epidemiol. 2021, 1–3. [Google Scholar] [CrossRef]
- Biasin, M.; Bianco, A.; Pareschi, G.; Cavalleri, A.; Cavatorta, C.; Fenizia, C.; Galli, P.; Lessio, L.; Lualdi, M.; Tombetti, E.; et al. UV-C irradiation is highly effective in inactivating SARS-CoV-2 replication. Sci. Rep. 2021, 11, 6260. [Google Scholar] [CrossRef]
- Ozog, D.M.; Sexton, J.Z.; Narla, S.; Pretto-Kernahan, C.D.; Mirabelli, C.; Lim, H.W.; Hamzavi, I.H.; Tibbetts, R.J.; Mi, Q.-S. The effect of ultraviolet C radiation against different N95 respirators inoculated with SARS-CoV-2. Int. J. Infect. Dis. 2020, 100, 224–229. [Google Scholar] [CrossRef]
- Delanghe, L.; Cauwenberghs, E.; Spacova, I.; de Boeck, I.; van Beeck, W.; Pepermans, K.; Claes, I.; Vandenheuvel, D.; Verhoeven, V.; Lebeer, S. Cotton and Surgical Face Masks in Community Settings: Bacterial Contamination and Face Mask Hygiene. Front. Med. 2021, 8, 732047. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K.; Rhee, Y.; Schoeny, M.; Lolans, K.; Cheng, J.; Reddy, S.; Weinstein, R.A.; Hayden, M.K.; Popovich, K.J.; Centers for Disease Control and Prevention Epicenters Program. Impact of doffing errors on healthcare worker self-contamination when caring for patients on contact precautions. Infect. Control Hosp. Epidemiol. 2019, 40, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Larrimer, A.; Curtiss, T.; Kim, J.; Jones, A.; Baird-Tomlinson, H.; Pekosz, A.; Olivo, P.D. Influenza virus assays based on virus-inducible reporter cell lines. Influenza Other Respir. Viruses 2009, 3, 241–251. [Google Scholar] [CrossRef]
- Kasloff, S.B.; Leung, A.; Strong, J.E.; Funk, D.; Cutts, T. Stability of SARS-CoV-2 on critical personal protective equipment. Sci. Rep. 2021, 11, 984. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Cousins, S. New Zealand eliminates COVID-19. Lancet 2020, 395, 1474. [Google Scholar] [CrossRef]
- Baker, M.; Kvalsvig, A.; Verrall, A.J.; Telfar-Barnard, L.; Wilson, N. New Zealand’s elimination strategy for the COVID-19 pandemic and what is required to make it work. N. Z. Med. J. 2020, 133, 10–14. [Google Scholar] [PubMed]
- Geoghegan, J.L.; Ren, X.; Storey, M.; Hadfield, J.; Jelley, L.; Jefferies, S.; Sherwood, J.; Paine, S.; Huang, S.; Douglas, J.; et al. Genomic epidemiology reveals transmission patterns and dynamics of SARS-CoV-2 in Aotearoa New Zealand. Nat. Commun. 2020, 11, 6351. [Google Scholar] [CrossRef]
- Harfoot, R.; Lawley, B.; Hernandez, L.C.; Kuang, J.; Grant, J.; Treece, J.M.; Lequeux, S.; Day, R.; Stanton, J.-A.L.; Bostina, M.; et al. First Isolation and Characterization of SARS-CoV-2 in Aotearoa New Zealand as Part of a Rapid response to the COVID-19 Pandemic in Early 2020. Viruses 2022, in press. [Google Scholar]
- Lee, H.C.; Goh, C.L. Occupational dermatoses from personal protective equipment during the COVID-19 pandemic in the tropics–a review. J. Eur. Acad. Dermatol. Venereol. 2021, 35, 589–596. [Google Scholar] [CrossRef]
- Calderwood, M.S.; Deloney, V.M.; Anderson, D.J.; Cheng, V.C.; Gohil, S.; Kwon, J.H.; Mody, L.; Monsees, E.; Vaughn, V.M.; Wiemken, T.L.; et al. Policies and practices of SHEA Research Network hospitals during the COVID-19 pandemic. Infect. Control Hosp. Epidemiol. 2020, 41, 1127–1135. [Google Scholar] [CrossRef]
- Cadnum, J.L.; Li, D.F.; Redmond, S.N.; John, A.R.; Pearlmutter, B.; Donskey, C.J. Effectiveness of ultraviolet-C light and a high-level disinfection cabinet for decontamination of N95 respirators. Pathog. Immun. 2020, 5, 52. [Google Scholar] [CrossRef]
- Fisher, E.M.; Shaffer, R.E. A method to determine the available UV-C dose for the decontamination of filtering facepiece respirators. J. Appl. Microbiol. 2011, 110, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Woo, M.-H.; Grippin, A.; Anwar, D.; Smith, T.; Wu, C.-Y.; Wander, J.D. Effects of relative humidity and spraying medium on UV decontamination of filters loaded with viral aerosols. Appl. Environ. Microbiol. 2012, 78, 5781–5787. [Google Scholar] [CrossRef] [Green Version]
- Bentancor, M.; Fernández, S.; Viera, F.; Etcheverry, S.; Poradosú, C.; D’Angelo, P.; Montemuiño, H.; Mirazo, S.; Irigoyen, Á.; Sanabría, A.; et al. LUCIA: An open source device for disinfection of N95 masks using UV-C radiation. HardwareX 2021, 9, e00181. [Google Scholar] [CrossRef]
- Rathnasinghe, R.; Karlicek, R.F., Jr.; Schotsaert, M.; Koffas, M.; Arduini, B.L.; Jangra, S.; Wang, B.; Davis, J.L.; Alnaggar, M.; Costa, A.; et al. Scalable, effective, and rapid decontamination of SARS-CoV-2 contaminated N95 respirators using germicidal ultraviolet C (UVC) irradiation device. Sci. Rep. 2021, 11, 19970. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.; Bodurtha, P.; Bourgeois, C.; Dickson, E.; Jensen, C.; Naqvi, S. The impact of extreme reuse and extended wear conditions on protection provided by a surgical-style N95 filtering facepiece respirator. J. Occup. Environ. Hyg. 2020, 17, 546–559. [Google Scholar] [CrossRef]
- Aps, J.K.; Martens, L.C. Review: The physiology of saliva and transfer of drugs into saliva. Forensic Sci. Int. 2005, 150, 119–131. [Google Scholar] [CrossRef] [PubMed]
- American Society for Testing and Materials. ASTM E2197–11 Standard Quantitative Disk Carrier Test Method for Determining the Bactericidal, Virucidal, Fungicidal, Mycobactericidal and Sporicidal Activities of Liquid Chemical Germicides; ASTM International: West Conshohocken, PA, USA, 2018. [Google Scholar]
- Pascoe, M.J.; Robertson, A.; Crayford, A.; Durand, E.; Steer, J.; Castelli, A.; Wesgate, R.; Evans, S.L.; Porch, A.; Maillard, J.Y. Dry heat and microwave generated steam protocols for the rapid decontamination of respiratory personal protective equipment in response to COVID-19-related shortages. J. Hosp. Infect. 2020, 106, 10–19. [Google Scholar] [CrossRef]
- Costa, D.M.; Johani, K.; Melo, D.S.; Lopes, L.K.O.; Lopes Lima, L.K.O.; Tipple, A.F.V.; Hu, H.; Vickery, K. Biofilm contamination of high-touched surfaces in intensive care units: Epidemiology and potential impacts. Lett. Appl. Microbiol. 2019, 68, 269–276. [Google Scholar] [CrossRef] [Green Version]
- Almatroudi, A.; Tahir, S.; Hu, H.; Chowdhury, D.; Gosbell, I.B.; Jensen, S.O.; Whiteley, G.S.; Deva, A.K.; Glasbey, T.; Vickery, K. Staphylococcus aureus dry-surface biofilms are more resistant to heat treatment than traditional hydrated biofilms. J. Hosp. Infect. 2018, 98, 161–167. [Google Scholar] [CrossRef]
- Kolter, R. Biofilms in lab and nature: A molecular geneticist’s voyage to microbial ecology. Int. Microbiol. 2010, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Berlanga, M.; Guerrero, R. Living together in biofilms: The microbial cell factory and its biotechnological implications. Microb. Cell Fact. 2016, 15, 165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rempel, D.; Henneman, J.; Agalloco, J.; Crittenden, J.; N95DECON Consortium. Hydrogen peroxide methods for decontaminating N95 filtering facepiece respirators. Appl. Biosaf. 2021, 26, 71–79. [Google Scholar] [CrossRef]
- Yamada, S.; Fukushi, S.; Kinoshita, H.; Ohnishi, M.; Suzuki, T.; Fujimoto, T.; Saijo, M.; Maeda, K. Assessment of SARS-CoV-2 infectivity of upper respiratory specimens from COVID-19 patients by virus isolation using VeroE6/TMPRSS2 cells. BMJ Open Respir. Res. 2021, 8, e000830. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Centers for Disease Control and Prevention. Outbreaks of community-associated methicillin-resistant Staphylococcus aureus skin infections-Los Angeles County, California, 2002–2003. MMWR Morb. Mortal. Wkly. Rep. 2003, 52, 88. [Google Scholar]
- Jacobs, A.C.; Thompson, M.G.; Black, C.C.; Kessler, J.L.; Clark, L.P.; McQueary, C.N.; Gancz, H.Y.; Corey, B.W.; Moon, J.K.; Si, Y.; et al. AB5075, a highly virulent isolate of Acinetobacter baumannii, as a model strain for the evaluation of pathogenesis and antimicrobial treatments. MBio 2014, 5, e01076-14. [Google Scholar] [CrossRef] [Green Version]
- Cheng, K.; Smyth, R.L.; Govan, J.R.; Doherty, C.; Winstanley, C.; Denning, N.; Heaf, D.P.; van Saene, H.; Hart, C.A. Spread of beta-lactam-resistant Pseudomonas aeruginosa in a cystic fibrosis clinic. Lancet 1996, 348, 639–642. [Google Scholar] [CrossRef]
- Freeman, D.J.; Falkiner, F.R.; Keane, C.T. New method for detecting slime production by coagulase negative staphylococci. J. Clin. Pathol. 1989, 42, 872–874. [Google Scholar] [CrossRef] [Green Version]
- American Society for Testing and Materials. ASTM D412–16: Standard Test Method for Vulcanized Rubber and Thermoplastic Elastomers—Tension; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- Rahn, R.O. Potassium Iodide as a Chemical Actinometer for 254 nm Radiation: Use of lodate as an Electron Scavenger. Photochem. Photobiol. 1997, 66, 450–455. [Google Scholar] [CrossRef]
- Rahn, R.O.; Stefan, M.I.; Bolton, J.R.; Goren, E.; Shaw, P.-S.; Lykke, K.R. Quantum Yield of the Iodide-Iodate Chemical Actinometer: Dependence on Wavelength and Concentration. Photochem. Photobiol. 2007, 78, 146–152. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harfoot, R.; Yung, D.B.Y.; Anderson, W.A.; Wild, C.E.K.; Coetzee, N.; Hernández, L.C.; Lawley, B.; Pletzer, D.; Derraik, J.G.B.; Anderson, Y.C.; et al. Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators. Pathogens 2022, 11, 83. https://doi.org/10.3390/pathogens11010083
Harfoot R, Yung DBY, Anderson WA, Wild CEK, Coetzee N, Hernández LC, Lawley B, Pletzer D, Derraik JGB, Anderson YC, et al. Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators. Pathogens. 2022; 11(1):83. https://doi.org/10.3390/pathogens11010083
Chicago/Turabian StyleHarfoot, Rhodri, Deborah B. Y. Yung, William A. Anderson, Cervantée E. K. Wild, Nicolene Coetzee, Leonor C. Hernández, Blair Lawley, Daniel Pletzer, José G. B. Derraik, Yvonne C. Anderson, and et al. 2022. "Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators" Pathogens 11, no. 1: 83. https://doi.org/10.3390/pathogens11010083
APA StyleHarfoot, R., Yung, D. B. Y., Anderson, W. A., Wild, C. E. K., Coetzee, N., Hernández, L. C., Lawley, B., Pletzer, D., Derraik, J. G. B., Anderson, Y. C., & Quiñones-Mateu, M. E. (2022). Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators. Pathogens, 11(1), 83. https://doi.org/10.3390/pathogens11010083









