Quantitative Risk Assessment of African Swine Fever Introduction into Spain by Legal Import of Live Pigs
Abstract
:1. Introduction
2. Material and Methods
2.1. Definition of the Unit of Analysis and Data Sources
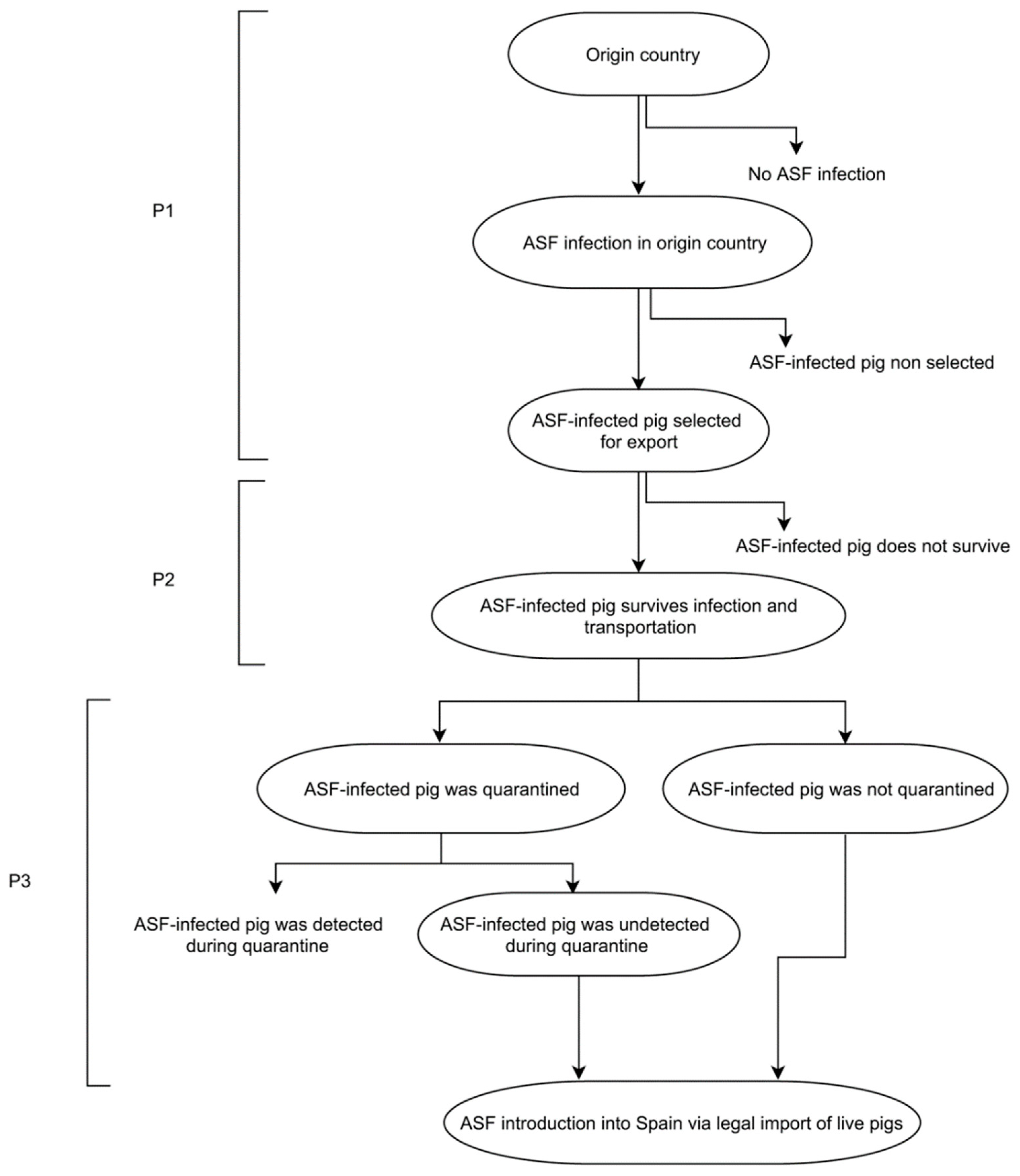
2.2. Model Formulation
2.3. Definition of Input Parameters
2.3.1. Probability of Selecting an Infected Pig (P1)
2.3.2. Probability of Survival of the Selected Pig (P2)
2.3.3. Probability That an Imported ASF-Infected Pig Establishes an Effective Contact with Other Domestic Pigs in Spain, Resulting in Disease Transmission (P3)
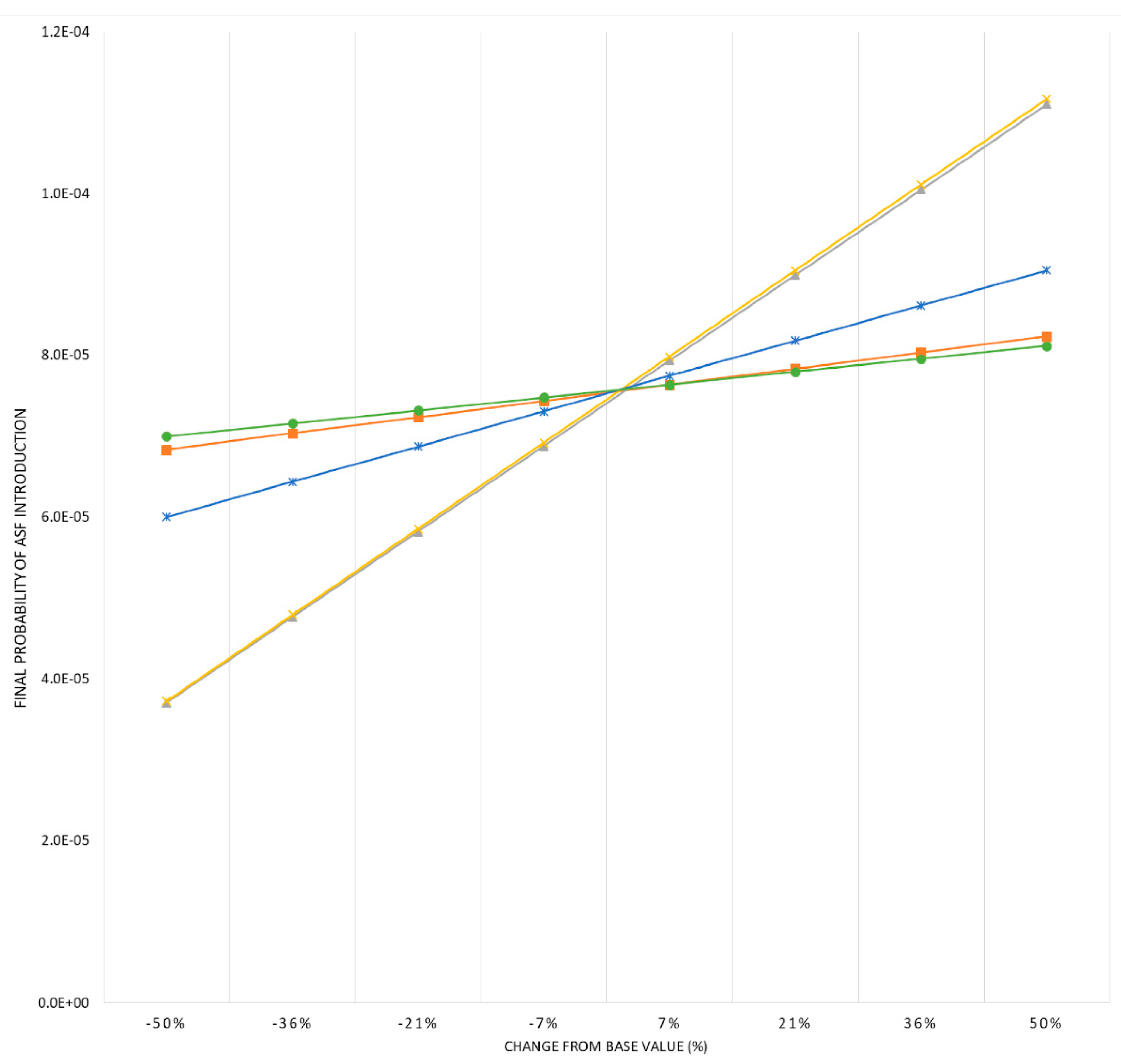
2.4. Sensitivity Analysis
3. Results
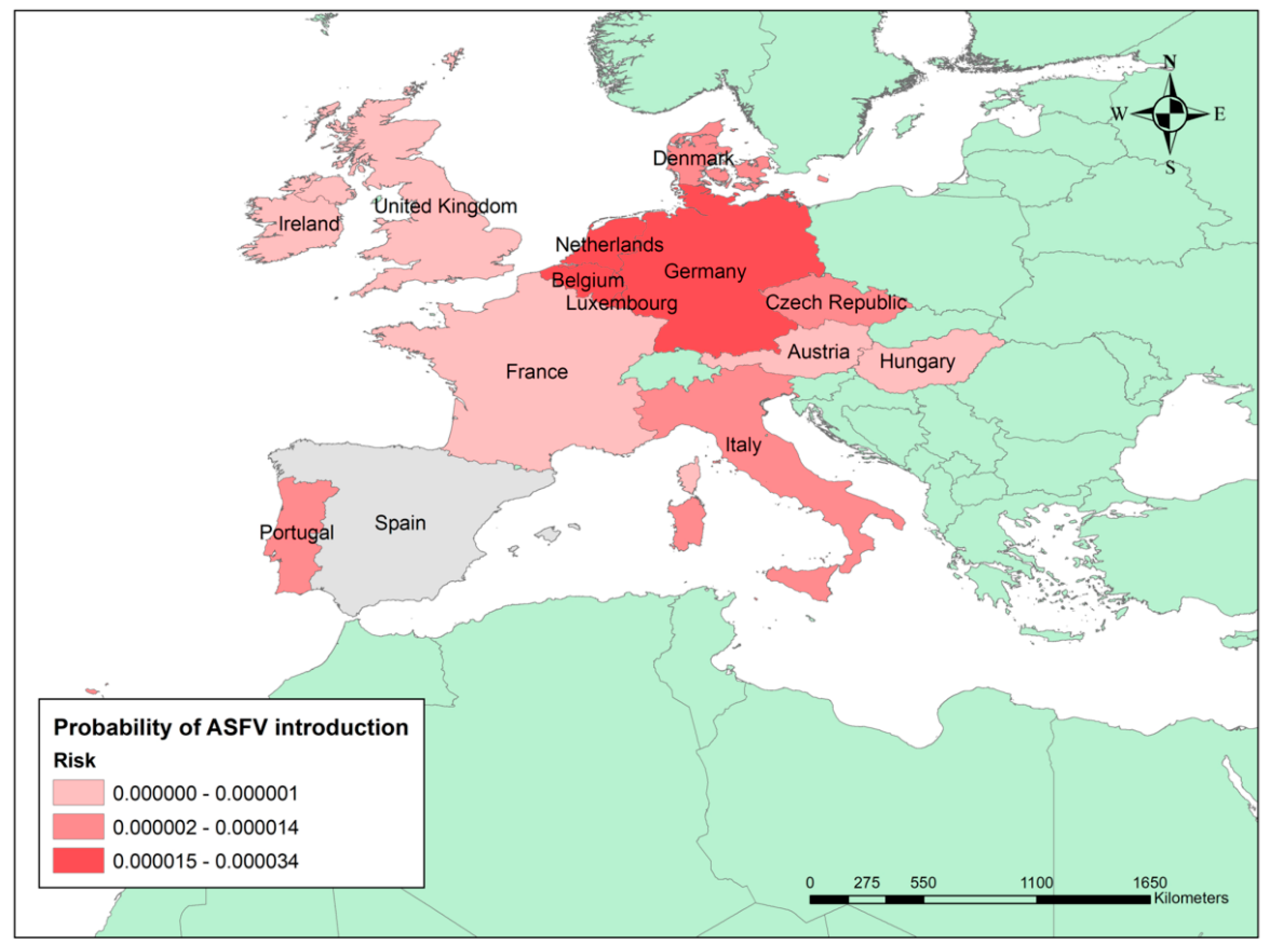
3.1. Probability of Having at Least One ASF Outbreak in Spain Due to the Import of Live Pigs
3.2. Sensitivity Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dixon, L.K.; Sun, H.; Roberts, H. African swine fever. Antiviral Res. 2019, 165, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Vizcaíno, J.M.; Mur, L.; Gomez-Villamandos, J.C.; Carrasco, L. An update on the epidemiology and pathology of African swine fever. J. Comp. Pathol. 2015, 152, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, E.R. On a form of swine fever occurring in British East Africa (Kenya Colony). J. Comp. Pathol. Ther. 1921, 34, 159–191. [Google Scholar] [CrossRef] [Green Version]
- Arias, M.; Sánchez-Vizcaíno, J.M. African swine fever eradication: The Spanish model. In Trends in Emerging Viral Infections of Swine; Morilla, A., Jin, K., Zimmerman, J., Eds.; Iowa State University Press: Ames, IA, USA, 2002; pp. 133–139. [Google Scholar]
- OIE–WAHIS (World Organization for Animal Health–World Animal Health Information System). Disease Situation. Available online: https://wahis.oie.int/#/analytics (accessed on 20 November 2021).
- Costard, S.; Jones, B.A.; Martínez-López, B.; Mur, L.; De la Torre, A.; Martínez, M.; Sánchez-Vizcaíno, F.; Sánchez-Vizcaíno, J.M.; Pfeiffer, D.U.; Wieland, B. Introduction of African swine fever into the European Union through illegal importation of pork and pork products. PLoS ONE 2013, 8, e61104. [Google Scholar] [CrossRef] [Green Version]
- Neumann, E.; Hall, W.; Dahl, J.; Hamilton, D.; Kurian, A. Is transportation a risk factor for African swine fever transmission in Australia: A review. Aust. Vet. J. 2021, 99, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Horst, H.S.; Dijkhuizen, A.A.; Huirne, R.B.M.; De Leeuw, P.W. Introduction of contagious animal diseases into the Netherlands: Elicitation of Expert Opinions. Livest. Prod. Sci. 1998, 53, 253–264. [Google Scholar] [CrossRef]
- Mur, L.; Martínez-López, B.; Sánchez-Vizcaíno, J.M. Risk of African swine fever introduction into the European Union through transport-associated routes: Returning trucks and waste from international ships and planes. BMC Vet. Res. 2012, 8, 149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- OIE (World Organization for Animal Health). Listed Diseases. 2021. Available online: https://www.oie.int/en/what-we-do/animal-health-and-welfare/animal-diseases (accessed on 31 August 2021).
- De Vos, C.J.; Saatkamp, H.W.; Nielen, M.; Huirne, R.B.M. Scenario Tree modeling to analyze the probability of Classical swine fever virus introduction into Member States of the European Union. Risk Anal. 2004, 24, 237–253. [Google Scholar] [CrossRef] [PubMed]
- EUROSTAT (Statistics of External Commercial Trade). Database. Available online: https://ec.europa.eu/eurostat/web/main/data/database (accessed on 18 October 2021).
- Mur, L.; Martínez-López, B.; Martínez-Avilés, M.; Costard, S.; Wieland, B.; Pfeiffer, D.U.; Sánchez-Vizcaíno, J.M. Quantitative risk assessment for the introduction of African swine fever virus into the European Union by legal import of live pigs. Transbound. Emerg. Dis. 2012, 59, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Ibatá, D.M.; Martínez-López, B.; Quijada, D.; Burton, K.; Mur, L. Quantitative approach for the risk assessment of African swine fever and Classical swine fever introduction into the United States through legal imports of pigs and swine products. PLoS ONE 2017, 12, e0182850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- FAOSTAT (Statistical Database of the Food and Agriculture Organization of the United Nations). Database. Available online: https://www.fao.org/faostat/en/#data (accessed on 24 October 2021).
- Murray, A.; Johnson, C. Impact of the halothane gene on muscle quality and pre-slaughter deaths in Western Canadian pigs. Can. J. Anim. Sci. 1998, 78, 543–548. [Google Scholar] [CrossRef]
- Martínez-López, B.; Perez, A.M.; Sánchez-Vizcaíno, J.M. A stochastic model to quantify the risk of introduction of Classical swine fever virus through import of domestic and wild boars. Epidemiol. Infect. 2009, 137, 1505–1515. [Google Scholar] [CrossRef] [PubMed]
- Risk Analysis and OIE. Available online: http://www.fao.org/docrep/003/x7354e/x7354e12.htm (accessed on 20 October 2021).
- Handbook on Import Risk Analysis for Animals and Animal Products. Available online: https://rr-africa.oie.int/wp-content/uploads/2018/03/handbook_on_import_risk_analysis_-_oie_-_vol__i.pdf (accessed on 10 October 2021).
- EC (European Commission). Commission Implementing Decision. Available online: https://eur-lex.europa.eu/legal-content/GA/TXT/?uri=CELEX:32018D0834 (accessed on 18 November 2021).
- OIE (World Organization for Animal Health). Self-Declaration Disease Status. Available online: https://www.oie.int/fileadmin/Home/eng/Animal_Health_in_the_World/docs/pdf/Self-declarations/2020_12_Belgium_ASF_self-declaration_ENG.pdf (accessed on 11 October 2021).
- Sun, E.; Huang, L.; Zhang, X.; Zhang, J.; Shen, D.; Zhang, Z.; Wang, Z.; Huo, H.; Wang, W.; Huangfu, H.; et al. Genotype I African swine fever viruses emerged in domestic pigs in China and caused chronic infection. Emerg. Microbes Infect. 2021, 10, 2183–2193. [Google Scholar] [CrossRef] [PubMed]
- Sun, E.; Zhang, Z.; Wang, Z.; He, X.; Zhang, X.; Wang, L.; Wang, W.; Huang, L.; Xi, F.; Huangfu, H.; et al. Emergence and prevalence of naturally occurring lower virulent African swine fever viruses in domestic pigs in China in 2020. Sci. China Life Sci. 2021, 64, 752–765. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Pérez, C.; Jurado, C.; Sánchez-Vizcaíno, J.M. African swine fever vaccine: Turning a dream into reality. Transbound. Emerg. Dis. 2021, 68, 2657–2668. [Google Scholar] [CrossRef] [PubMed]
| Notation | Definition | Parametrization | Source | Values |
|---|---|---|---|---|
| P1 | Probability of selecting an ASF-infected pig from the country of origin (c) at month (m) before detection of ASFV infection | Beta (α1,α2) α1p = NI + 1 α2p = No – (NI + 1) | NI = Pc × Ou × Tc × Hp No = pig population in c | |
| Pc | Probability of undetected infection in country c | Beta (α1,α2) α1q = X + 1 α2q = M – (X + 1) | OIE-WAHIS [5] X: Number of months with at least one undetected ASF outbreak in c M: Number of months considered in the analysis | |
| Ou | Expected number of undetected outbreaks before official notification | Pert (min, most likely, max) | Herrera-Ibatá et al. [14] | Pert (1, 1.28, 6) |
| Tc | Average herd size in country c | Normal = No/So | ||
| No | Pig population size in country c | Normal (µ,σ) | FAOSTAT (Statistical Database of the Food and Agriculture Organization of the United Nations) [15] | |
| So | Number of pig farms in country c | Normal (µ,σ) | FAOSTAT [15] | |
| Hp | Intra-herd prevalence | Pert (min, most likely, max) | Herrera-Ibatá et al. [14] | Pert (0.05, 0.15, 0.32) |
| P2 | Probability of survival of the selected ASF-infected pig | Ps × Pt | ||
| Ps | Probability that an ASF-infected pig survives the ASF infection | Pert (min, most likely, max) | Sánchez-Vizcaíno et al. [2] | Pert (0.05, 0.2, 0,1) |
| Pt | Probability that an ASF-infected pig survives transportation | Pert (min, most likely, max) | Murray and Johnson [16] | Pert (0.0005, 0.0027, 0.092) |
| P3 | Probability that an imported ASF-infected pig establishes an effective contact with other domestic pigs in Spain, resulting in disease transmission | Pq + [(1 − Pq) × Pu] | ||
| Pq | Probability that imported pigs were not quarantined | Beta (α1,α2) | Martínez-López et al. [17] | Beta (130.7, 15.4) |
| Pu | Probability that an ASF-infected pig was not detected during quarantine | Beta (α1,α2) | Martínez-López et al. [17] | Beta (1.3, 34.2) |
| ncm | Import of live pigs from country c to Spain during month m (2012–2020) | Normal (µ,σ) | EUROSTAT [12] | |
| pcm | Probability that an ASF-infected pig from country c was introduced into a Spanish farm during month m and effectively contacted a susceptible pig | Binomial (n, p) | n = ncm p = P1× P2 × P3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Pérez, C.; Bosch, J.; Ito, S.; Martínez-Avilés, M.; Sánchez-Vizcaíno, J.M. Quantitative Risk Assessment of African Swine Fever Introduction into Spain by Legal Import of Live Pigs. Pathogens 2022, 11, 76. https://doi.org/10.3390/pathogens11010076
Muñoz-Pérez C, Bosch J, Ito S, Martínez-Avilés M, Sánchez-Vizcaíno JM. Quantitative Risk Assessment of African Swine Fever Introduction into Spain by Legal Import of Live Pigs. Pathogens. 2022; 11(1):76. https://doi.org/10.3390/pathogens11010076
Chicago/Turabian StyleMuñoz-Pérez, Carolina, Jaime Bosch, Satoshi Ito, Marta Martínez-Avilés, and José Manuel Sánchez-Vizcaíno. 2022. "Quantitative Risk Assessment of African Swine Fever Introduction into Spain by Legal Import of Live Pigs" Pathogens 11, no. 1: 76. https://doi.org/10.3390/pathogens11010076
APA StyleMuñoz-Pérez, C., Bosch, J., Ito, S., Martínez-Avilés, M., & Sánchez-Vizcaíno, J. M. (2022). Quantitative Risk Assessment of African Swine Fever Introduction into Spain by Legal Import of Live Pigs. Pathogens, 11(1), 76. https://doi.org/10.3390/pathogens11010076







