Arboviral Disease Outbreaks in the Pacific Islands Countries and Areas, 2014 to 2020: A Systematic Literature and Document Review
Abstract
1. Introduction
2. Results
2.1. Dengue Outbreaks
Where Information about Dengue Outbreaks Was Found
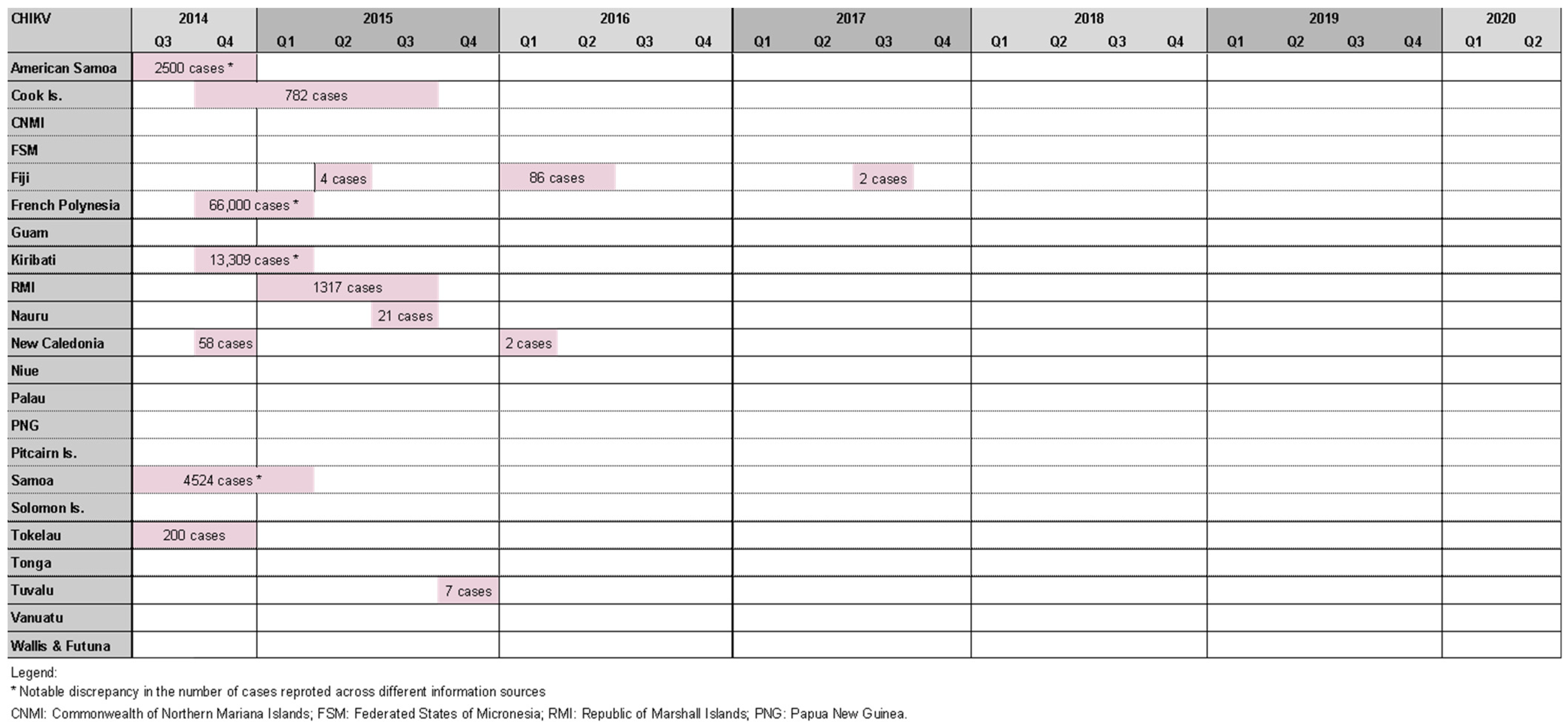
2.2. Chikungunya Outbreaks
Where Information about Chikungunya Outbreaks Was Found
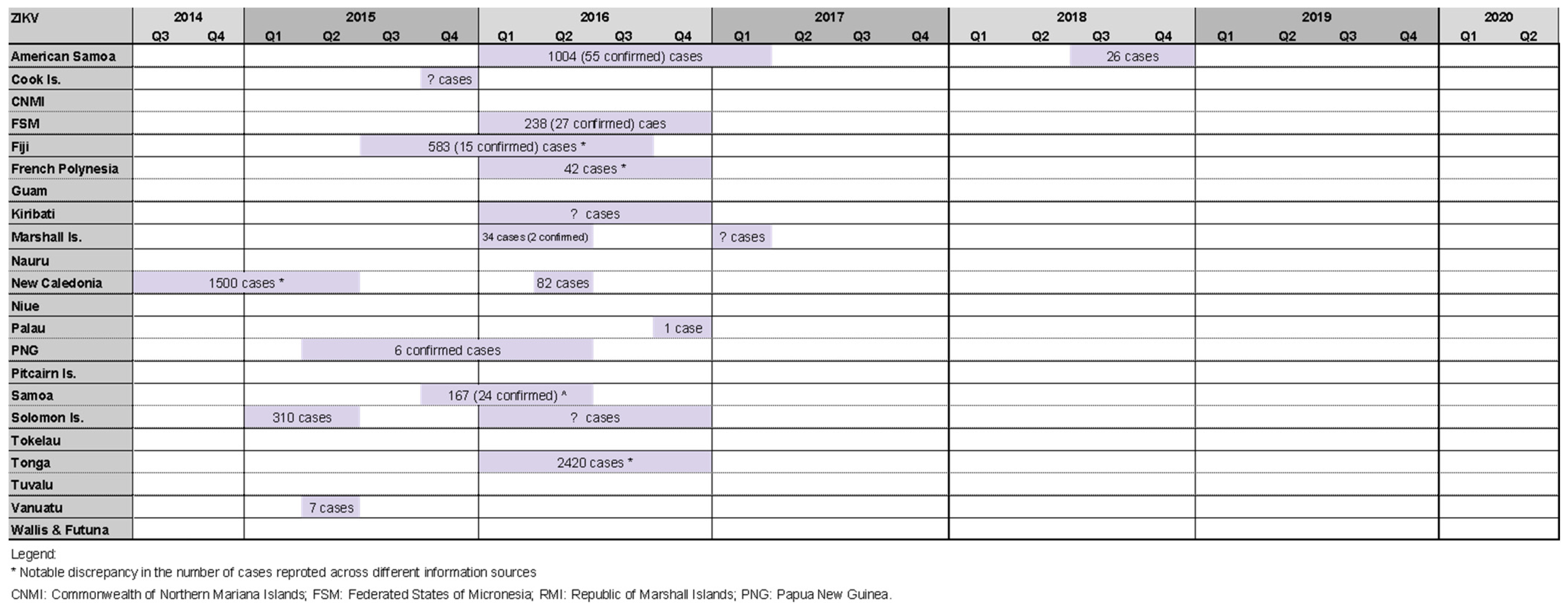
2.3. Zika Outbreaks
Where Information about Zika Outbreaks Was Found
2.4. Other Arboviral Disease Outbreaks
3. Discussion
4. Materials and Methods
4.1. Published Literature Search
4.2. Grey Literature Search
4.3. Consultation with Public Health Agency Focal Points
4.4. Data Extraction
4.5. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- World Health Organization. Vector-Borne Diseases. Available online: https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases (accessed on 30 May 2021).
- Heymann, D.L. Control of Communicable Diseases Manual: An Official Report of the American Public Health Association, 20th ed.; American Public Health Association: Washington, DC, USA, 2015. [Google Scholar]
- Heymann, D.L.; Hodgson, A.; Sall, A.A.; Freedman, D.O.; Staples, J.E.; Althabe, F.; Baruah, K.; Mahmud, G.; Kandun, N.; Vasconcelos, P.F.; et al. Zika virus and microcephaly: Why is this situation a PHEIC? Lancet 2016, 387, 719–721. [Google Scholar] [CrossRef]
- World Health Organization. WHO statement on the first meeting of the International Health Regulations (2005) (IHR 2005) Emergency Committee on Zika Virus and Observed Increase in Neurological Disorders and Neonatal Malformations. Available online: https://www.who.int/news/item/01-02-2016-who-statement-on-the-first-meeting-of-the-international-health-regulations-(2005)-(ihr-2005)-emergency-committee-on-zika-virus-and-observed-increase-in-neurological-disorders-and-neonatal-malformations (accessed on 30 May 2021).
- Gubler, D.J. The continuing spread of West Nile virus in the western hemisphere. Clin. Infect. Dis. 2007, 45, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Vanlandingham, D.L.; McGee, C.E.; Higgs, S. A single mutation in chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007, 3, e201. [Google Scholar] [CrossRef]
- Mayer, S.V.; Tesh, R.B.; Vasilakis, N. The emergence of arthropod-borne viral diseases: A global prospective on dengue, chikungunya and zika fevers. Acta Trop. 2017, 166, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Napoli, C.; Salcuni, P.; Pompa, M.G.; Declich, S.; Rizzo, C. Estimated imported infections of Chikungunya and Dengue in Italy, 2008 to 2011. J. Travel Med. 2012, 19, 294–297. [Google Scholar] [CrossRef] [PubMed]
- Messina, J.P.; Brady, O.J.; Golding, N.; Kraemer, M.U.G.; Wint, G.R.W.; Ray, S.E.; Pigott, D.M.; Shearer, F.M.; Johnson, K.; Earl, L.; et al. The current and future global distribution and population at risk of dengue. Nat. Microbiol. 2019, 4, 1508–1515. [Google Scholar] [CrossRef]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef]
- United Nations. World Populations Prospects 2019. Available online: https://population.un.org/wpp/ (accessed on 19 May 2021).
- Institute for Health Metrics and Evaluation. GBD Compare. Available online: http://vizhub.healthdata.org/gbd-compare (accessed on 19 May 2021).
- Craig, A.T.; Kaldor, J.; Schierhout, G.; Rosewell, A.E. Surveillance strategies for the detection of disease outbreaks in the Pacific islands: Meta-analysis of published literature, 2010–2019. Trop. Med. Int. Health 2020, 25, 906–918. [Google Scholar] [CrossRef]
- Craig, A.T.; Kama, M.; Samo, M.; Vaai, S.; Matanaicake, J.; Joshua, C.; Kolbe, A.; Durrheim, D.N.; Paterson, B.J.; Biaukula, V.; et al. Early warning epidemic surveillance in the Pacific island nations: An evaluation of the Pacific syndromic surveillance system. Trop. Med. Int. Health 2016, 21, 917–927. [Google Scholar] [CrossRef]
- Kool, J.L.; Paterson, B.; Pavlin, B.I.; Durrheim, D.; Musto, J.; Kolbe, A. Pacific-wide simplified syndromic surveillance for early warning of outbreaks. Glob. Public Health 2012, 7, 670–681. [Google Scholar] [CrossRef]
- World Economic Forum. The Global Risks Report 2016; World Economic Forum: Geneva, Switzerland, 2016. [Google Scholar]
- McIver, L.; Kim, R.; Woodward, A.; Hales, S.; Spickett, J.; Katscherian, D.; Hashizume, M.; Honda, Y.; Kim, H.; Iddings, S.; et al. Health impacts of climate change in Pacific island countries: A regional assessment of vulnerabilities and adaptation priorities. Environ. Health Perspect. 2016, 124, 1707–1714. [Google Scholar] [CrossRef] [PubMed]
- Perry, W.J. The mosquitoes and mosquito-borne diseases on New Caledonia, an historic account: 1885–1946. Am. J. Trop. Med. 1950, s1-30, 103–114. [Google Scholar] [CrossRef]
- Calvez, E.; Guillaumot, L.; Millet, L.; Marie, J.; Bossin, H.; Rama, V.; Faamoe, A.; Kilama, S.; Teurlai, M.; Mathieu-Daudé, F.; et al. Genetic diversity and phylogeny of aedes aegypti, the main arbovirus vector in the Pacific. PLoS Negl. Trop. Dis. 2016, 10, e0004374. [Google Scholar] [CrossRef] [PubMed]
- Roth, A.; Mercier, A.; Lepers, C.; Hoy, D.; Duituturaga, S.; Benyon, E.; Guillaumot, L.; Souares, Y. Concurrent outbreaks of dengue, chikungunya and Zika virus infections—An unprecedented epidemic wave of mosquito-borne viruses in the Pacific 2012–2014. Eurosurveillance 2014, 19, 20929. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.; Jamdar, S.F.; Alalowi, M.; Al Ageel Al Beaiji, S.M. Dengue virus: A global human threat: Review of literature. J. Int. Soc. Prev. Community Dent. 2016, 6, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Fredericks, A.C.; Fernandez-Sesma, A. The burden of dengue and chikungunya worldwide: Implications for the southern United States and California. Ann. Glob. Health 2014, 80, 466–475. [Google Scholar] [CrossRef]
- Dupont-Rouzeyrol, M.; Aubry, M.; O’Connor, O.; Roche, C.; Gourinat, A.-C.; Guigon, A.; Pyke, A.; Grangeon, J.-P.; Nilles, E.; Chanteau, S.; et al. Epidemiological and molecular features of dengue virus type-1 in New Caledonia, South Pacific, 2001–2013. Virol. J. 2014, 11, 61. [Google Scholar] [CrossRef]
- Singh, N.; Kiedrzynski, T.; Lepers, C.; Benyon, E.K. Dengue in the Pacific—An update of the current situation. Pac. Health Surveill. Response 2005, 12, 111–119. [Google Scholar]
- Soo, K.M.; Khalid, B.; Ching, S.M.; Chee, H.Y. Meta-analysis of dengue severity during infection by different dengue virus serotypes in primary and secondary infections. PLoS ONE 2016, 11, e0154760. [Google Scholar] [CrossRef]
- Guzman, M.G.; Vazquez, S. The complexity of antibody-dependent enhancement of dengue virus infection. Viruses 2010, 2, 2649–2662. [Google Scholar] [CrossRef]
- Pacific Public Health Surveillance Network. PacNet listserv. Various Posts, May 2014–October 2020. Available online: https://www.pphsn.net/Services/PacNet/intro.htm (accessed on 22 July 2020).
- International Society for Infectious Diseases. ProMed-Mail. Various Posts, May 2014–October 2020. Available online: www.promedmail.org (accessed on 22 July 2020).
- Aubry, M.; Cao-Lormeau, V.M. History of arthropod-borne virus infections in French Polynesia. New Microbes New Infect. 2019, 29, 100513. [Google Scholar] [CrossRef] [PubMed]
- Serie, M.; Clausse, D.; Noel, M.; Abraham, P.; Forfait, C.; Gaimard, S.; Betton, D. HepatoDengue: Severity and outcome of acute hepatitis in Dengue Fever in New Caledonia. Ann. Intensive Care 2020, 10, 16. [Google Scholar] [CrossRef]
- Craig, A.T.; Joshua, C.A.; Sio, A.R.; Teobasi, B.; Dofai, A.; Dalipanda, T.; Hardie, K.; Kaldor, J.; Kolbe, A. Enhanced surveillance during a public health emergency in a resource-limited setting: Experience from a large dengue outbreak in Solomon Islands, 2016–2017. PLoS ONE 2018, 13, e0198487. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.R.; van den Hurk, A.F.; Mackenzie, J.S.; Pyke, A.T. Dengue viruses in Papua New Guinea: Evidence of endemicity and phylogenetic variation, including the evolution of new genetic lineages. Emerg. Microbes Infect. 2017, 6, e114. [Google Scholar] [CrossRef]
- Mavian, C.; Dulcey, M.; Munoz, O.; Salemi, M.; Vittor, A.Y.; Capua, I. Islands as hotspots for emerging mosquito-borne viruses: A one-health perspective. Viruses 2018, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Pulsan, F.; Sobi, K.; Anga, G.; Vince, J.; Duke, T. An outbreak of dengue fever in children in the National Capital District of Papua New Guinea in 2016. Paediatr. Int. Child Health 2020, 40, 177–180. [Google Scholar] [CrossRef]
- Luang-Suarkia, D.; Mitja, O.; Ernst, T.; Bennett, S.; Tay, A.; Hays, R.; Smith, D.W.; Imrie, A. Hyperendemic dengue transmission and identification of a locally evolved DENV-3 lineage, Papua New Guinea 2007–2010. PLoS Negl. Trop. Dis. 2018, 12, e0006254. [Google Scholar] [CrossRef]
- Cotter, C.J.; Tufa, A.J.; Johnson, S.; Matai’a, M.; Sciulli, R.; Ryff, K.; Hancock, T.; Whelen, C.; Sharp, T.; Anesi, M. Outbreak of Dengue Virus Type 2—American Samoa, November 2016–October 2018. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 1319–1322. [Google Scholar] [CrossRef]
- World Health Organization. Zika Virus Infection—Papua New Guinea—Disease Outbreak News, 22 April 2016. 2016. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/22-april-2016-zika-png-en (accessed on 22 July 2020).
- Hills, S.L.; Fischer, M.; Petersen, L.R. Epidemiology of Zika Virus Infection. J. Infect. Dis. 2017, 216, S868–S874. [Google Scholar] [CrossRef]
- Craig, A.T.; Butler, M.T.; Pastore, R.; Paterson, B.J.; Durrheim, D.N. Acute flaccid paralysis incidence and Zika virus surveillance, Pacific Islands. Bull. World Health Organ. 2017, 95, 69–75. [Google Scholar] [CrossRef]
- Gilotra, S.K.; Shah, K.V. Laboratory studies on transmission of Chikungunya virus by mosquitoes. Am. J. Epidemiol. 1967, 86, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Caglioti, C.; Lalle, E.; Castilletti, C.; Carletti, F.; Capobianchi, M.R.; Bordi, L. Chikungunya virus infection: An overview. New Microbiol. 2013, 36, 211–227. [Google Scholar] [PubMed]
- Aubry, M.; Kama, M.; Henderson, A.D.; Teissier, A.; Vanhomwegen, J.; Mariteragi-Helle, T.; Paoaafaite, T.; Manuguerra, J.C.; Christi, K.; Watson, C.H.; et al. Low chikungunya virus seroprevalence two years after emergence in Fiji. Int. J. Infect. Dis. 2020, 90, 223–225. [Google Scholar] [CrossRef]
- Aubry, M.; Teissier, A.; Roche, C.; Richard, V.; Yan, A.S.; Zisou, K.; Rouault, E.; Maria, V.; Lastère, S.; Cao-Lormeau, V.M.; et al. Chikungunya outbreak, French Polynesia, 2014. Emerg. Infect. Dis. 2015, 21, 724–726. [Google Scholar] [CrossRef] [PubMed]
- Koeltz, A.; Lastere, S.; Jean-Baptiste, S. Intensive care admissions for severe Chikungunya virus infection, French Polynesia. Emerg. Infect. Dis. 2018, 24, 794–796. [Google Scholar] [CrossRef] [PubMed]
- Oehler, E.; Fournier, E.; Leparc-Goffart, I.; Larre, P.; Cubizolle, S.; Sookhareea, C.; Lastère, S.; Ghawche, F. Increase in cases of Guillain-Barré syndrome during a Chikungunya outbreak, French Polynesia, 2014 to 2015. Eurosurveillance 2015, 20, 30079. [Google Scholar] [CrossRef]
- United States Centers for Disease Control and Prevention. Chikunguya Virus: 2014 Data. Available online: https://www.cdc.gov/chikungunya/geo/united-states-2014.html (accessed on 30 July 2021).
- Wahid, B.; Ali, A.; Rafique, S.; Idrees, M. Global expansion of chikungunya virus: Mapping the 64-year history. Int. J. Infect. Dis. 2017, 58, 69–76. [Google Scholar] [CrossRef]
- Kama, M.; Aubry, M.; Naivalu, T.; Vanhomwegen, J.; Mariteragi-Helle, T.; Teissier, A.; Paoaafaite, T.; Hué, S.; Hibberd, M.L.; Manuguerra, J.C.; et al. Sustained low-level transmission of Zika and Chikungunya viruses after emergence in the Fiji Islands. Emerg. Infect. Dis. 2019, 25, 1535–1538. [Google Scholar] [CrossRef]
- Nhan, T.X.; Musso, D. The burden of chikungunya in the Pacific. Clin. Microbiol. Infect. 2015, 21, e47–e48. [Google Scholar] [CrossRef]
- Dick, G.W.; Kitchen, S.F.; Haddow, A.J. Zika virus. I. Isolations and serological specificity. Trans. R. Soc. Trop. Med. Hyg. 1952, 46, 509–520. [Google Scholar] [CrossRef]
- Duffy, M.R.; Chen, T.H.; Hancock, W.T.; Powers, A.M.; Kool, J.L.; Lanciotti, R.S.; Pretrick, M.; Marfel, M.; Holzbauer, S.; Dubray, C.; et al. Zika virus outbreak on Yap Island, Federated States of Micronesia. N. Engl. J. Med. 2009, 360, 2536–2543. [Google Scholar] [CrossRef]
- Simon, O.; Acket, B.; Forfait, C.; Girault, D.; Gourinat, A.C.; Millon, P.; Daures, M.; Vanhomwegen, J.; Billot, S.; Biron, A.; et al. Zika virus outbreak in New Caledonia and Guillain-Barré syndrome: A case-control study. J. Neurovirol. 2018, 24, 362–368. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Rapid Risk Assessment: Zika Virus Infection Outbreak, Brazil and the Pacific Region, 26 May 2015; ECDC: Solna, Sweden, 2015. [Google Scholar]
- Healy, J.M.; Burgess, M.C.; Chen, T.-H.; Hancock, W.T.; Toews, K.-A.E.; Anesi, M.S.; Tulafono, R.T., Jr.; Mataia, M.A.; Sili, B.; Solaita, J.; et al. Notes from the Field: Outbreak of Zika Virus Disease—American Samoa, 2016. Morb. Mortal. Wkly. Rep. 2016, 65, 1146–1147. [Google Scholar] [CrossRef]
- Samarasekera, U.; Triunfol, M. Concern over Zika virus grips the world. Lancet 2016, 387, 521–524. [Google Scholar] [CrossRef]
- Sheel, M.; Collins, J.; Kama, M.; Nand, D.; Faktaufon, D.; Samuela, J.; Biaukula, V.; Haskew, C.; Flint, J.; Roper, K.; et al. Evaluation of the early warning, alert and response system after Cyclone Winston, Fiji, 2016. Bull. World Health Organ. 2019, 97, 178–189. [Google Scholar] [CrossRef]
- Musso, D.; Gubler, D.J. Zika Virus. Clin. Microbiol. Rev. 2016, 29, 487–524. [Google Scholar] [CrossRef]
- Paixão, E.S.; Barreto, F.; Teixeira, M.d.G.; Costa, M.d.C.; Rodrigues, L.C. History, epidemiology, and clinical manifestations of Zika: A systematic review. Am. J. Public Health 2016, 106, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Bierlaire, D.; Mauguin, S.; Broult, J.; Musso, D. Zika virus and blood transfusion: The experience of French Polynesia. Transfusion 2017, 57, 729–733. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aubry, M.; Finke, J.; Teissier, A.; Roche, C.; Broult, J.; Paulous, S.; Desprès, P.; Cao-Lormeau, V.M.; Musso, D. Silent circulation of Ross River virus in French Polynesia. Int. J. Infect. Dis. 2015, 37, 19–24. [Google Scholar] [CrossRef]
- Caly, L.; Horwood, P.F.; Vijaykrishna, D.; Lynch, S.; Greenhill, A.R.; Pomat, W.; Rai, G.; Kisa, D.; Bande, G.; Druce, J.; et al. Divergent Barmah Forest virus from Papua New Guinea. Emerg. Infect. Dis. 2019, 25, 2266–2269. [Google Scholar] [CrossRef] [PubMed]
- Aubry, M.; Teissier, A.; Huart, M.; Merceron, S.; Vanhomwegen, J.; Roche, C.; Vial, A.-L.; Teururai, S.; Sicard, S.; Paulous, S.; et al. Ross River virus seroprevalence, French Polynesia, 2014–2015. Emerg. Infect. Dis. 2017, 23, 1751. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.; Aubry, M.; Musso, D.; Teissier, A.; Paulous, S.; Desprès, P.; de-Lamballerie, X.; Pastorino, B.; Cao-Lormeau, V.M.; Weinstein, P. New evidence for endemic circulation of Ross River virus in the Pacific Islands and the potential for emergence. Int. J. Infect. Dis. 2017, 57, 73–76. [Google Scholar] [CrossRef]
- PacIfic Public Health Surveillance Network. Pacific Outbreak Manual; Pacific Community (SPC): Noumea, New Caledonia, 2016. [Google Scholar]
- Rosewell, A.; Ropa, B.; Randall, H.; Dagina, R.; Hurim, S.; Bieb, S.; Datta, S.; Ramamurthy, S.; Mola, G.; Zwi, A.B.; et al. Mobile phone-based syndromic surveillance system, Papua New Guinea. Emerg. Infect. Dis. 2013, 19, 1811–1818. [Google Scholar] [CrossRef]
- Ayanore, M.A.; Amuna, N.; Aviisah, M.; Awolu, A.; Kipo-Sunyehzi, D.D.; Mogre, V.; Ofori-Asenso, R.; Gmanyami, J.M.; Kugbey, N.; Gyapong, M. Towards resilient health systems in sub-Saharan Africa: A systematic review of the English language literature on health workforce, surveillance, and health governance issues for health systems strengthening. Ann. Glob. Health 2019, 85, 113. [Google Scholar] [CrossRef] [PubMed]
- Grantz, K.H.; Meredith, H.R.; Cummings, D.A.T.; Metcalf, C.J.E.; Grenfell, B.T.; Giles, J.R.; Mehta, S.; Solomon, S.; Labrique, A.; Kishore, N.; et al. The use of mobile phone data to inform analysis of COVID-19 pandemic epidemiology. Nat. Commun. 2020, 11, 4961. [Google Scholar] [CrossRef]
- Ropa, B.; Flint, J.; O’Reilly, M.; Pavlin, B.I.; Dagina, R.; Peni, B.; Bauri, M.; Pukienei, A.; Merritt, T.; Terrell-Perica, S.; et al. Lessons from the first 6 years of an intervention-based field epidemiology training programme in Papua New Guinea, 2013–2018. BMJ Glob. Health 2019, 4, e001969. [Google Scholar] [CrossRef]
- Hoy, D.; Durand, A.M.; Hancock, T.; Cash, H.L.; Hardie, K.; Paterson, B.; Paulino, Y.; White, P.; Merritt, T.; Fitzgibbons, D.; et al. Lessons learnt from a three-year pilot field epidemiology training programme. West. Pac. Surveill. Response J. 2017, 8, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Worwor, G.; Harries, A.D.; Merilles, O.E., Jr.; Viney, K.; Rory, J.J.; Taleo, G.; Guyant, P. Syndromic surveillance in Vanuatu since Cyclone Pam: A descriptive study. West. Pac. Surveill. Response J. 2016, 7, 6–11. [Google Scholar] [CrossRef][Green Version]
- Khormi, H.M.; Kumar, L. Climate change and the potential global distribution of Aedes aegypti: Spatial modelling using GIS and CLIMEX. Geospat. Health 2014, 8, 405–415. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matthews, R.J.; Kaluthotage, I.; Russell, T.L.; Knox, T.B.; Horwood, P.F.; Craig, A.T. Arboviral Disease Outbreaks in the Pacific Islands Countries and Areas, 2014 to 2020: A Systematic Literature and Document Review. Pathogens 2022, 11, 74. https://doi.org/10.3390/pathogens11010074
Matthews RJ, Kaluthotage I, Russell TL, Knox TB, Horwood PF, Craig AT. Arboviral Disease Outbreaks in the Pacific Islands Countries and Areas, 2014 to 2020: A Systematic Literature and Document Review. Pathogens. 2022; 11(1):74. https://doi.org/10.3390/pathogens11010074
Chicago/Turabian StyleMatthews, Rosie J., Ishani Kaluthotage, Tanya L. Russell, Tessa B. Knox, Paul F. Horwood, and Adam T. Craig. 2022. "Arboviral Disease Outbreaks in the Pacific Islands Countries and Areas, 2014 to 2020: A Systematic Literature and Document Review" Pathogens 11, no. 1: 74. https://doi.org/10.3390/pathogens11010074
APA StyleMatthews, R. J., Kaluthotage, I., Russell, T. L., Knox, T. B., Horwood, P. F., & Craig, A. T. (2022). Arboviral Disease Outbreaks in the Pacific Islands Countries and Areas, 2014 to 2020: A Systematic Literature and Document Review. Pathogens, 11(1), 74. https://doi.org/10.3390/pathogens11010074







