Mediterranean Aquaculture in a Changing Climate: Temperature Effects on Pathogens and Diseases of Three Farmed Fish Species
Abstract
1. Introduction
2. Temperature Projections in the Mediterranean Sea
3. Host-Pathogen Interplay in the Context of Temperature Variation
3.1. The Host Perspective
3.1.1. Temperature Effect on Fish Physiology
3.1.2. Temperature Effect on the Fish Immune System
4. The Pathogen Perspective
4.1. Bacterial Pathogens
4.1.1. Major Bacterial Pathogens
4.1.2. Minor Bacterial Pathogens
4.2. Parasitic Pathogens
4.2.1. Major Parasitic Pathogens
4.2.2. Minor Parasitic Pathogens
4.3. Viral and Fungal Pathogens
4.3.1. Major Viral Pathogens
4.3.2. Minor Viral and Fungal Pathogens
5. Risk Assessment
6. Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FEAP. European Aquaculture Production Report 2014–2019; FEAP: Brussels, Belgium, 2020. [Google Scholar]
- Federation of Greek Mariculture. Aquaculture in Greece; Annual Report; Federation of Greek Mariculture: Athens, Greece, 2019. [Google Scholar]
- Seth-Smith, H.M.B.; Dourala, N.; Fehr, A.; Qi, W.; Katharios, P.; Ruetten, M.; Mateos, J.M.; Nufer, L.; Weilenmann, R.; Ziegler, U.; et al. Emerging pathogens of gilthead seabream: Characterisation and genomic analysis of novel intracellular β-proteobacteria. ISME J. 2016, 10, 1791–1803. [Google Scholar] [CrossRef]
- Bondad-Reantaso, M.; Subasinghe, R. Aquaculture development, health and wealth. In Proceedings of the Aquaculture in the Third Millennium, Bangkok, Thailand, 20–25 February 2000; pp. 167–191. [Google Scholar]
- Meyer, F.P. Aquaculture disease and health management. J. Anim. Sci. 1991, 69, 4201–4208. [Google Scholar] [CrossRef] [PubMed]
- Conte, F.S. Stress and the welfare of cultured fish. Appl. Anim. Behav. Sci. 2004, 86, 205–223. [Google Scholar] [CrossRef]
- Wedemeyer, G.A. Effects of rearing conditions on the health and physiological quality of fish in intensive culture. In Fish, Stress and Health in Aquaculture; Academic Press: Cambridge, MA, USA, 1997; pp. 35–71. [Google Scholar]
- Hastein, T.; Hjeltnes, B.; Lillehaug, A.; Utne Skare, J.; Berntssen, M.; Lundebye, A.K. Food safety hazards that occur during the production stage: Challenges for fish farming and the fishing industry. Rev. Sci. Tech. 2006, 25, 607–625. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.A.M.L.; Howgate, P. Fishborne zoonotic parasites and aquaculture: A review. Aquaculture 2011, 318, 253–261. [Google Scholar] [CrossRef]
- Gauthier, D.T. Bacterial zoonoses of fishes: A review and appraisal of evidence for linkages between fish and human infections. Vet. J. 2015, 203, 27–35. [Google Scholar] [CrossRef]
- Rivas, A.J.; Lemos, M.L.; Osorio, C.R. Photobacterium damselae subsp. damselae, a bacterium pathogenic for marine animals and humans. Front. Microbiol. 2013, 4, 283. [Google Scholar] [CrossRef]
- Gauthier, D.T.; Rhodes, M.W. Mycobacteriosis in fishes: A review. Vet. J. 2009, 180, 33–47. [Google Scholar] [CrossRef]
- Rigos, G.; Troisi, G.M. Antibacterial agents in Mediterranean finfish farming: A synopsis of drug pharmacokinetics in important euryhaline fish species and possible environmental implications. Rev. Fish Biol. Fish. 2005, 15, 53–73. [Google Scholar] [CrossRef]
- Rigos, G.; Kogiannou, D.; Padrós, F.; Cristòfol, C.; Florio, D.; Fioravanti, M.; Zarza, C. Best therapeutic practices for the use of antibacterial agents in finfish aquaculture: A particular view on European seabass (Dicentrarchus labrax) and gilthead seabream (Sparus aurata) in Mediterranean aquaculture. Rev. Aquac. 2020, 13, 1285–1323. [Google Scholar] [CrossRef]
- Cabello, F.C. Heavy use of prophylactic antibiotics in aquaculture: A growing problem for human and animal health and for the environment. Environ. Microbiol. 2006, 8, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Smith, P. Antimicrobial resistance in aquaculture. Rev. Sci. Tech. 2008, 27, 243. [Google Scholar] [CrossRef]
- Santos, L.; Ramos, F. Antimicrobial resistance in aquaculture: Current knowledge and alternatives to tackle the problem. Int. J. Antimicrob. Agents 2018, 52, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Watts, J.E.M.; Schreier, H.J.; Lanska, L.; Hale, M.S. The rising tide of antimicrobial resistance in aquaculture: Sources, sinks and solutions. Mar. Drugs 2017, 15, 158. [Google Scholar] [CrossRef] [PubMed]
- Heuer, O.E.; Kruse, H.; Grave, K.; Collignon, P.; Karunasagar, I.; Angulo, F.J. Human health consequences of use of antimicrobial agents in aquaculture. Clin. Infect. Dis. 2009, 49, 1248–1253. [Google Scholar] [CrossRef] [PubMed]
- Rosa, R.; Marques, A.; Nunes, M.L. Impact of climate change in Mediterranean aquaculture. Rev. Aquac. 2012, 4, 163–177. [Google Scholar] [CrossRef]
- Marras, S.; Cucco, A.; Antognarelli, F.; Azzurro, E.; Milazzo, M.; Bariche, M.; Butenschön, M.; Kay, S.; Di Bitetto, M.; Quattrocchi, G. Predicting future thermal habitat suitability of competing native and invasive fish species: From metabolic scope to oceanographic modelling. Conserv. Physiol. 2015, 3, cou059. [Google Scholar] [CrossRef]
- Stocker, T.F.; Qin, D.; Plattner, G.-K.; Tignor, M.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, V.; Midgley, P.M. 2013: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of IPCC the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Perkins, S.E.; Alexander, L.V.; Nairn, J.R. Increasing frequency, intensity and duration of observed global heatwaves and warm spells. Geophys. Res. Lett. 2012, 39, L20714. [Google Scholar] [CrossRef]
- Azzurro, E.; Sbragaglia, V.; Cerri, J.; Bariche, M.; Bolognini, L.; Ben Souissi, J.; Busoni, G.; Coco, S.; Chryssanthi, A.; Fanelli, E. Climate change, biological invasions, and the shifting distribution of Mediterranean fishes: A large-scale survey based on local ecological knowledge. Glob. Chang. Biol. 2019, 25, 2779–2792. [Google Scholar] [CrossRef]
- Belkin, I.M. Rapid warming of large marine ecosystems. Prog. Oceanogr. 2009, 81, 207–213. [Google Scholar] [CrossRef]
- Oliver, E.C.J.; Donat, M.G.; Burrows, M.T.; Moore, P.J.; Smale, D.A.; Alexander, L.V.; Benthuysen, J.A.; Feng, M.; Gupta, A.S.; Hobday, A.J. Longer and more frequent marine heatwaves over the past century. Nat. Commun. 2018, 9, 1324. [Google Scholar] [CrossRef]
- Van Vuuren, D.P.; Edmonds, J.; Kainuma, M.; Riahi, K.; Thomson, A.; Hibbard, K.; Hurtt, G.C.; Kram, T.; Krey, V.; Lamarque, J.-F. The representative concentration pathways: An overview. Clim. Chang. 2011, 109, 5. [Google Scholar] [CrossRef]
- Lotze, H.K.; Tittensor, D.P.; Bryndum-Buchholz, A.; Eddy, T.D.; Cheung, W.W.L.; Galbraith, E.D.; Barange, M.; Barrier, N.; Bianchi, D.; Blanchard, J.L. Ensemble projections of global ocean animal biomass with climate change. arXiv 2018, arXiv:467175. [Google Scholar]
- Sarà, G.; Gouhier, T.C.; Brigolin, D.; Porporato, E.M.D.; Mangano, M.C.; Mirto, S.; Mazzola, A.; Pastres, R. Predicting shifting sustainability trade-offs in marine finfish aquaculture under climate change. Glob. Chang. Biol. 2018, 24, 3654–3665. [Google Scholar] [CrossRef] [PubMed]
- Teske, S. Achieving the Paris Climate Agreement Goals; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Macias, D.; Garcia-Gorriz, E.; Piroddi, C.; Stips, A. Biogeochemical control of marine productivity in the Mediterranean Sea during the last 50 years. Glob. Biogeochem. Cycles 2014, 28, 897–907. [Google Scholar] [CrossRef] [PubMed]
- Le Traon, P.Y.; Reppucci, A.; Alvarez Fanjul, E.; Aouf, L.; Behrens, A.; Belmonte, M.; Bentamy, A.; Bertino, L.; Brando, V.E.; Kreiner, M.; et al. From observation to information and users: The Copernicus Marine Service perspective. Front. Mar. Sci. 2019, 6, 234. [Google Scholar] [CrossRef]
- Falconer, L.; Hjøllo, S.S.; Telfer, T.C.; McAdam, B.J.; Hermansen, Ø.; Ytteborg, E. The importance of calibrating climate change projections to local conditions at aquaculture sites. Aquaculture 2020, 514, 734487. [Google Scholar] [CrossRef]
- Hawkins, E.; Osborne, T.M.; Ho, C.K.; Challinor, A.J. Calibration and bias correction of climate projections for crop modelling: An idealised case study over Europe. Agric. For. Meteorol. 2013, 170, 19–31. [Google Scholar] [CrossRef]
- Barredo, J.I.; Mauri, A.; Caudullo, G.; Dosio, A. Assessing shifts of Mediterranean and arid climates under RCP4. 5 and RCP8. 5 climate projections in Europe. In Meteorology and Climatology of the Mediterranean and Black Seas; Springer: Berlin/Heidelberg, Germany, 2019; pp. 235–251. [Google Scholar]
- Garcias-Bonet, N.; Arrieta, J.M.; Duarte, C.M.; Marbà, N. Nitrogen-Fixing bacteria in Mediterranean seagrass (Posidonia oceanica) roots. Aquat. Bot. 2016, 131, 57–60. [Google Scholar] [CrossRef]
- Sakalli, A. Sea surface temperature change in the Mediterranean Sea under climate change: A linear model for simulation of the sea surface temperature up to 2100. Appl. Ecol. Environ. Res. 2017, 15, 707–716. [Google Scholar] [CrossRef]
- Adloff, F.; Somot, S.; Sevault, F.; Jordà, G.; Aznar, R.; Déqué, M.; Herrmann, M.; Marcos, M.; Dubois, C.; Padorno, E. Mediterranean Sea response to climate change in an ensemble of twenty first century scenarios. Clim. Dyn. 2015, 45, 2775–2802. [Google Scholar] [CrossRef]
- European MSP Platform. Available online: https://www.msp-platform.eu/projects/med-iamer-integrated-actions-mitigate-environmental-risks-mediterranean-sea (accessed on 2 September 2021).
- Hobday, A.J.; Alexander, L.V.; Perkins, S.E.; Smale, D.A.; Straub, S.C.; Oliver, E.C.J.; Benthuysen, J.A.; Burrows, M.T.; Donat, M.G.; Feng, M. A hierarchical approach to defining marine heatwaves. Prog. Oceanogr. 2016, 141, 227–238. [Google Scholar] [CrossRef]
- Frölicher, T.L.; Fischer, E.M.; Gruber, N. Marine heatwaves under global warming. Nature 2018, 560, 360–364. [Google Scholar] [CrossRef]
- Moyle, P.B.; Cech, J.J. Fishes: An Introduction to Ichthyology; Prentice Hall: Hoboken, NJ, USA, 2004; ISBN 0131008471. [Google Scholar]
- Little, A.G.; Loughland, I.; Seebacher, F. What do warming waters mean for fish physiology and fisheries? J. Fish. Biol. 2020, 97, 328–340. [Google Scholar] [CrossRef]
- Pörtner, H. Climate change and temperature-dependent biogeography: Oxygen limitation of thermal tolerance in animals. Naturwissenschaften 2001, 88, 137–146. [Google Scholar] [PubMed]
- Pörtner, H.-O. Climate variations and the physiological basis of temperature dependent biogeography: Systemic to molecular hierarchy of thermal tolerance in animals. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2002, 132, 739–761. [Google Scholar] [CrossRef]
- Johansson, D.; Juell, J.-E.; Oppedal, F.; Stiansen, J.-E.; Ruohonen, K. The influence of the pycnocline and cage resistance on current flow, oxygen flux and swimming behaviour of Atlantic salmon (Salmo salar L.) in production cages. Aquaculture 2007, 265, 271–287. [Google Scholar] [CrossRef]
- Sundh, H.; Kvamme, B.O.; Fridell, F.; Olsen, R.E.; Ellis, T.; Taranger, G.L.; Sundell, K. Intestinal barrier function of Atlantic salmon (Salmo salar L.) post smolts is reduced by common sea cage environments and suggested as a possible physiological welfare indicator. BMC Physiol. 2010, 10, 22. [Google Scholar] [CrossRef]
- Rombough, P.J. The effects of temperature on embryonic and larval development. In Global Warming: Implication of Fresh Water and Marine Fish; Cambridge University Press: Cambridge, UK, 1997; Volume 61, pp. 177–224. [Google Scholar]
- Blaxter, J.H.S. The effect of temperature on larval fishes. Neth. J. Zool. 1991, 42, 336–357. [Google Scholar] [CrossRef]
- Jobling, M. Temperature and growth: Modulation of growth rate via temperature change. In Global Warming: Implication of Fresh Water and Marine Fish; Cambridge University Press: Cambridge, UK, 1997; Volume 61, pp. 225–254. [Google Scholar]
- Pankhurst, N. Temperature effects on the reproductive performance of fish. Glob. Warm. Implic. Freshw. Mar. Fish 1997, 61, 159. [Google Scholar]
- Le Morvan, C.; Troutaud, D.; Deschaux, P. Differential effects of temperature on specific and nonspecific immune defences in fish. J. Exp. Biol. 1998, 201, 165–168. [Google Scholar] [CrossRef]
- Peterson, M.E.; Daniel, R.M.; Danson, M.J.; Eisenthal, R. The dependence of enzyme activity on temperature: Determination and validation of parameters. Biochem. J. 2007, 402, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Farkas, T.; Fodor, E.; Kitajka, K.; Halver, J.E. Response of fish membranes to environmental temperature. Aquac. Res. 2001, 32, 645–655. [Google Scholar] [CrossRef]
- Sandersfeld, T.; Davison, W.; Lamare, M.D.; Knust, R.; Richter, C. Elevated temperature causes metabolic trade-offs at the whole-organism level in the Antarctic fish Trematomus bernacchii. J. Exp. Biol. 2015, 218, 2373–2381. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, G.; Ray, A.K. The effect of seasonal temperature on endogenous gut enzyme activity in four air-breathing fish species. Croat. J. Fish. 2018, 76, 60–65. [Google Scholar] [CrossRef]
- Enders, E.C.; Boisclair, D. Effects of environmental fluctuations on fish metabolism: Atlantic salmon Salmo salar as a case study. J. Fish. Biol. 2016, 88, 344–358. [Google Scholar] [CrossRef] [PubMed]
- Korte, S.M.; Olivier, B.; Koolhaas, J.M. A new animal welfare concept based on allostasis. Physiol. Behav. 2007, 92, 422–428. [Google Scholar] [CrossRef]
- Pörtner, H.-O.; Bock, C.; Mark, F.C. Oxygen-and capacity-limited thermal tolerance: Bridging ecology and physiology. J. Exp. Biol. 2017, 220, 2685–2696. [Google Scholar] [CrossRef] [PubMed]
- Pörtner, H.O.; Knust, R. Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 2007, 315, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Iwama, G.K.; Vijayan, M.M.; Forsyth, R.B.; Ackerman, P.A. Heat shock proteins and physiological stress in fish. Am. Zool. 1999, 39, 901–909. [Google Scholar] [CrossRef]
- Iwama, G.K.; Thomas, P.T.; Forsyth, R.B.; Vijayan, M.M. Heat shock protein expression in fish. Rev. Fish. Biol. Fish. 1998, 8, 35–56. [Google Scholar] [CrossRef]
- Madeira, D.; Narciso, L.; Cabral, H.N.; Vinagre, C.; Diniz, M.S. Influence of temperature in thermal and oxidative stress responses in estuarine fish. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2013, 166, 237–243. [Google Scholar] [CrossRef]
- Vinagre, C.; Madeira, D.; Narciso, L.; Cabral, H.N.; Diniz, M. Effect of temperature on oxidative stress in fish: Lipid peroxidation and catalase activity in the muscle of juvenile seabass, Dicentrarchus labrax. Ecol. Indic. 2012, 23, 274–279. [Google Scholar] [CrossRef]
- Almeida, J.R.; Gravato, C.; Guilhermino, L. Effects of temperature in juvenile seabass (Dicentrarchus labrax L.) biomarker responses and behaviour: Implications for environmental monitoring. Estuaries Coasts 2015, 38, 45–55. [Google Scholar] [CrossRef]
- Oomen, R.A.; Hutchings, J.A. Transcriptomic responses to environmental change in fishes: Insights from RNA sequencing. Facets 2017, 2, 610–641. [Google Scholar] [CrossRef]
- Logan, C.A.; Buckley, B.A. Transcriptomic responses to environmental temperature in eurythermal and stenothermal fishes. J. Exp. Biol. 2015, 218, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Padilla, D.K.; Adolph, S.C. Plastic inducible morphologies are not always adaptive: The importance of time delays in a stochastic environment. Evol. Ecol. 1996, 10, 105–117. [Google Scholar] [CrossRef]
- Deans, C.; Maggert, K.A. What do you mean, “epigenetic”? Genetics 2015, 199, 887–896. [Google Scholar] [CrossRef]
- Mousseau, T.A.; Fox, C.W. The adaptive significance of maternal effects. Trends Ecol. Evol. 1998, 13, 403–407. [Google Scholar] [CrossRef]
- van Straalen, N.M.; Feder, M.E. Ecological and evolutionary functional genomics. How can it contribute to the risk assessment of chemicals? Environ. Sci. Technol. 2012, 46, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Sgro, C.M. Climate change and evolutionary adaptation. Nature 2011, 470, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Norman, J.D.; Ferguson, M.M.; Danzmann, R.G. Transcriptomics of salinity tolerance capacity in Arctic charr (Salvelinus alpinus): A comparison of gene expression profiles between divergent QTL genotypes. Physiol. Genom. 2014, 46, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Oomen, R.A.; Hutchings, J.A. Genetic variability in reaction norms in fishes. Environ. Rev. 2015, 23, 353–366. [Google Scholar] [CrossRef]
- Zhang, R.; Ludwig, A.; Zhang, C.; Tong, C.; Li, G.; Tang, Y.; Peng, Z.; Zhao, K. Local adaptation of Gymnocypris przewalskii (Cyprinidae) on the Tibetan Plateau. Sci. Rep. 2015, 5, 9780. [Google Scholar] [CrossRef]
- Hu, P.; Liu, M.; Liu, Y.; Wang, J.; Zhang, D.; Niu, H.; Jiang, S.; Wang, J.; Zhang, D.; Han, B. Transcriptome comparison reveals a genetic network regulating the lower temperature limit in fish. Sci. Rep. 2016, 6, 28952. [Google Scholar] [CrossRef]
- Feidantsis, K.; Pörtner, H.O.; Lazou, A.; Kostoglou, B.; Michaelidis, B. Metabolic and molecular stress responses of the gilthead seabream Sparus aurata during long-term exposure to increasing temperatures. Mar. Biol. 2009, 156, 797–809. [Google Scholar] [CrossRef]
- Feidantsis, K.; Antonopoulou, E.; Lazou, A.; Pörtner, H.O.; Michaelidis, B. Seasonal variations of cellular stress response of the gilthead sea bream (Sparus aurata). J. Comp. Physiol. B 2013, 183, 625–639. [Google Scholar] [CrossRef]
- Feidantsis, K.; Pörtner, H.O.; Vlachonikola, E.; Antonopoulou, E.; Michaelidis, B. Seasonal changes in metabolism and cellular stress phenomena in the gilthead sea bream (Sparus aurata). Physiol. Biochem. Zool. 2018, 91, 878–895. [Google Scholar] [CrossRef]
- Person-Le Ruyet, J.; Mahe, K.; Le Bayon, N.; Le Delliou, H. Effects of temperature on growth and metabolism in a Mediterranean population of European sea bass, Dicentrarchus labrax. Aquaculture 2004, 237, 269–280. [Google Scholar] [CrossRef]
- Claireaux, G.; Lagardère, J.-P. Influence of temperature, oxygen and salinity on the metabolism of the European sea bass. J. Sea Res. 1999, 42, 157–168. [Google Scholar] [CrossRef]
- Islam, M.J.; Kunzmann, A.; Bögner, M.; Meyer, A.; Thiele, R.; Slater, M.J. Metabolic and molecular stress responses of European seabass, Dicentrarchus labrax at low and high temperature extremes. Ecol. Indic. 2020, 112, 106118. [Google Scholar] [CrossRef]
- Islam, M.J.; Slater, M.J.; Bögner, M.; Zeytin, S.; Kunzmann, A. Extreme ambient temperature effects in European seabass, Dicentrarchus labrax: Growth performance and hemato-biochemical parameters. Aquaculture 2020, 522, 735093. [Google Scholar] [CrossRef]
- Duncan, N.J.; Estévez, A.; Fernández-Palacios, H.; Gairin, I.; Hernández-Cruz, C.M.; Roo, J.; Schuchardt, D.; Vallés, R. 17—Aquaculture production of meagre (Argyrosomus regius): Hatchery techniques, ongrowing and market. In Advances in Aquaculture Hatchery Technology; Allan, G., Burnell, G., Eds.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Sawston, UK, 2013; pp. 519–541. ISBN 978-0-85709-119-2. [Google Scholar]
- Kir, M.; Sunar, M.C.; Altindag, B.C. Thermal tolerance and preferred temperature range of juvenile meagre acclimated to four temperatures. J. Therm. Biol. 2017, 65, 125–129. [Google Scholar] [CrossRef]
- Antonopoulou, E.; Chatzigiannidou, I.; Feidantsis, K.; Kounna, C.; Chatzifotis, S. Effect of water temperature on cellular stress responses in meagre (Argyrosomus regius). Fish. Physiol. Biochem. 2020, 46, 1075–1091. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Chacoff, L.; Arjona, F.J.; Ruiz-Jarabo, I.; Páscoa, I.; Gonçalves, O.; Martín del Río, M.P.; Mancera, J.M. Seasonal variation in osmoregulatory and metabolic parameters in earthen pond-cultured gilthead sea bream Sparus auratus. Aquac. Res. 2009, 40, 1279–1290. [Google Scholar] [CrossRef]
- Samaras, A.; Papandroulakis, N.; Costari, M.; Pavlidis, M. Stress and metabolic indicators in a relatively high (European sea bass, Dicentrarchus labrax) and a low (meagre, Argyrosomus regius) cortisol responsive species, in different water temperatures. Aquac. Res. 2016, 47, 3501–3515. [Google Scholar] [CrossRef]
- Pascoli, F.; Lanzano, G.S.; Negrato, E.; Poltronieri, C.; Trocino, A.; Radaelli, G.; Bertotto, D. Seasonal effects on hematological and innate immune parameters in sea bass Dicentrarchus labrax. Fish Shellfish Immunol. 2011, 31, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Planas, J.; Gutierrez, J.; Fernandez, J.; Carrillo, M.; Canals, P. Annual and daily variations of plasma cortisol in sea bass, Dicentrarchus labrax L. Aquaculture 1990, 91, 171–178. [Google Scholar] [CrossRef]
- Fanouraki, E.; Mylonas, C.C.; Papandroulakis, N.; Pavlidis, M. Species specificity in the magnitude and duration of the acute stress response in Mediterranean marine fish in culture. Gen. Comp. Endocrinol. 2011, 173, 313–322. [Google Scholar] [CrossRef]
- Pickering, A.D.; Pottinger, T.G. Stress responses and disease resistance in salmonid fish: Effects of chronic elevation of plasma cortisol. Fish. Physiol. Biochem. 1989, 7, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Pulsford, A.L.; Lemaire-Gony, S.; Tomlinson, M.; Collingwood, N.; Glynn, P.J. Effects of acute stress on the immune system of the dab, Limanda limanda. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1994, 109, 129–139. [Google Scholar] [CrossRef]
- Espelid, S.; Løkken, G.B.; Steiro, K.; Bøgwald, J. Effects of cortisol and stress on the immune system in Atlantic Salmon (Salmo salar L.). Fish Shellfish Immunol. 1996, 6, 95–110. [Google Scholar] [CrossRef]
- Wendelaar Bonga, S.E. The stress response in fish. Physiol. Rev. 1997, 77, 591–625. [Google Scholar] [CrossRef] [PubMed]
- Bly, J.E.; Clem, L.W. Temperature and teleost immune functions. Fish Shellfish Immunol. 1992, 2, 159–171. [Google Scholar] [CrossRef]
- Magnadóttir, B. Innate immunity of fish (overview). Fish Shellfish Immunol. 2006, 20, 137–151. [Google Scholar] [CrossRef]
- Magnadottir, B. Immunological control of fish diseases. Mar. Biotechnol. 2010, 12, 361–379. [Google Scholar] [CrossRef]
- Abram, Q.H.; Dixon, B.; Katzenback, B.A. Impacts of low temperature on the teleost immune system. Biology 2017, 6, 39. [Google Scholar] [CrossRef] [PubMed]
- Larsen, A.K.; Nymo, I.H.; Sørensen, K.K.; Seppola, M.; Rødven, R.; Jiménez de Bagüés, M.P.; Al Dahouk, S.; Godfroid, J. Concomitant temperature stress and immune activation may increase mortality despite efficient clearance of an intracellular bacterial infection in Atlantic cod. Front. Microbiol. 2018, 9, 2963. [Google Scholar] [CrossRef]
- Zanuzzo, F.S.; Beemelmanns, A.; Hall, J.R.; Rise, M.L.; Gamperl, A.K. The innate immune response of Atlantic salmon (Salmo salar) is not negatively affected by high temperature and moderate hypoxia. Front. Immunol. 2020, 11, 1009. [Google Scholar] [CrossRef]
- Ellis, A.E. Innate host defense mechanisms of fish against viruses and bacteria. Dev. Comp. Immunol. 2001, 25, 827–839. [Google Scholar] [CrossRef]
- Beck, B.H.; Peatman, E. Mucosal Health in Aquaculture; Academic Press: Cambridge, MA, USA, 2015; ISBN 0124171931. [Google Scholar]
- Wang, B.; Yao, M.; Lv, L.; Ling, Z.; Li, L. The human microbiota in health and disease. Engineering 2017, 3, 71–82. [Google Scholar] [CrossRef]
- Gosalbes, M.J.; Durbán, A.; Pignatelli, M.; Abellan, J.J.; Jiménez-Hernández, N.; Pérez-Cobas, A.E.; Latorre, A.; Moya, A. Metatranscriptomic approach to analyze the functional human gut microbiota. PLoS ONE 2011, 6, e17447. [Google Scholar] [CrossRef] [PubMed]
- Franzosa, E.A.; Morgan, X.C.; Segata, N.; Waldron, L.; Reyes, J.; Earl, A.M.; Giannoukos, G.; Boylan, M.R.; Ciulla, D.; Gevers, D. Relating the metatranscriptome and metagenome of the human gut. Proc. Natl. Acad. Sci. USA 2014, 111, E2329–E2338. [Google Scholar] [CrossRef]
- Egerton, S.; Culloty, S.; Whooley, J.; Stanton, C.; Ross, R.P. The gut microbiota of marine fish. Front. Microbiol. 2018, 9, 873. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.; Salinas, I. Under pressure: Interactions between commensal microbiota and the teleost immune system. Front. Immunol. 2017, 8, 559. [Google Scholar] [CrossRef]
- Magarinos, B.; Pazos, F.; Santos, Y.; Romalde, J.L.; Toranzo, A.E. Response of Pasteurella piscicida and Flexibacter maritimus to skin mucus of marine fish. Dis. Aquat. Organ. 1995, 21, 103–108. [Google Scholar] [CrossRef]
- Sundh, H.; Sundell, K.S. Environmental impacts on fish mucosa. In Mucosal Health in Aquaculture; Academic Press: Cambridge, MA, USA, 2015; ISBN 9780124171930. [Google Scholar]
- Harvell, C.D.; Kim, K.; Burkholder, J.M.; Colwell, R.R.; Epstein, P.R.; Grimes, D.J.; Hofmann, E.E.; Lipp, E.K.; Osterhaus, A.; Overstreet, R.M. Emerging marine diseases—Climate links and anthropogenic factors. Science 1999, 285, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, Y.S.; Bogwald, J. Influence of artificial wound and non-intact mucus layer on mortality of Atlantic salmon (Salmo salar L.) following a bath challenge with Vibrio anguillarum and Aeromonas salmonicida. Fish Shellfish Immunol. 1997, 7, 317–325. [Google Scholar] [CrossRef]
- Kiron, V. Fish immune system and its nutritional modulation for preventive health care. Anim. Feed Sci. Technol. 2012, 173, 111–133. [Google Scholar] [CrossRef]
- Bouck, G.R.; Smith, S.D. Mortality of experimentally descaled smolts of coho salmon (Oncorhynchus kisutch) in fresh and salt water. Trans. Am. Fish. Soc. 1979, 108, 67–69. [Google Scholar] [CrossRef]
- Stien, L.H.; Bracke, M.B.M.; Folkedal, O.; Nilsson, J.; Oppedal, F.; Torgersen, T.; Kittilsen, S.; Midtlyng, P.J.; Vindas, M.A.; Øverli, Ø. Salmon Welfare Index Model (SWIM 1.0): A semantic model for overall welfare assessment of caged Atlantic salmon: Review of the selected welfare indicators and model presentation. Rev. Aquac. 2013, 5, 33–57. [Google Scholar] [CrossRef]
- Sunyer, J.O.; Tort, L. Natural hemolytic and bactericidal activities of sea bream Sparus aurata serum are effected by the alternative complement pathway. Vet. Immunol. Immunopathol. 1995, 45, 333–345. [Google Scholar] [CrossRef]
- Angelidis, P.; Baudin-Laurencin, F.; Youinou, P. Effects of temperature on chemiluminescence of phagocytes from sea bass, Dicentrarchus labrax L. J. Fish Dis. 1988, 11, 281–288. [Google Scholar] [CrossRef]
- Tort, L.; Padros, F.; Rotllant, J.; Crespo, S. Winter syndrome in the gilthead sea bream Sparus aurata. Immunological and histopathological features. Fish Shellfish Immunol. 1998, 8, 37–47. [Google Scholar] [CrossRef]
- Ibarz, A.; Padrós, F.; Gallardo, M.Á.; Fernández-Borràs, J.; Blasco, J.; Tort, L. Low-Temperature challenges to gilthead sea bream culture: Review of cold-induced alterations and ‘Winter Syndrome’. Rev. fish Biol. Fish. 2010, 20, 539–556. [Google Scholar] [CrossRef]
- Rotllant, J.; Balm, P.H.M.; Wendelaar-Bonga, S.E.; Pérez-Sánchez, J.; Tort, L. A drop in ambient temperature results in a transient reduction of interrenal ACTH responsiveness in the gilthead sea bream (Sparus aurata, L.). Fish. Physiol. Biochem. 2000, 23, 265–273. [Google Scholar] [CrossRef]
- Tort, L.; Rotllant, J.; Rovira, L. Immunological suppression in gilthead sea bream Sparus aurata of the North-West Mediterranean at low temperatures. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 1998, 120, 175–179. [Google Scholar] [CrossRef]
- Hardie, L.J.; Fletcher, T.C.; Secombes, C.J. Effect of temperature on macrophage activation and the production of macrophage activating factor by rainbow trout (Oncorhynchus mykiss) leucocytes. Dev. Comp. Immunol. 1994, 18, 57–66. [Google Scholar] [CrossRef]
- Scott, A.L.; Rogers, W.A.; Klesius, P.H. Chemiluminescence by peripheral blood phagocytes from channel catfish: Function of opson in and temperature. Dev. Comp. Immunol. 1985, 9, 241–250. [Google Scholar] [CrossRef]
- Ainsworth, A.J.; Dexiang, C.; Waterstrat, P.R.; Greenway, T. Effect of temperature on the immune system of channel catfish (Ictalurus punctatus)—I. Leucocyte distribution and phagocyte function in the anterior kidney at 10 °C. Comp. Biochem. Physiol. A Physiol. 1991, 100, 907–912. [Google Scholar] [CrossRef]
- Bowden, T.J. Modulation of the immune system of fish by their environment. Fish Shellfish Immunol. 2008, 25, 373–383. [Google Scholar] [CrossRef]
- Nikoskelainen, S.; Kjellsen, O.; Lilius, E.-M.; Schrøder, M.B. Respiratory burst activity of Atlantic cod (Gadus morhua L.) blood phagocytes differs markedly from that of rainbow trout. Fish Shellfish Immunol. 2006, 21, 199–208. [Google Scholar] [CrossRef]
- Hrubec, T.C.; Robertson, J.L.; Smith, S.A.; Tinker, M.K. The effect of temperature and water quality on antibody response to Aeromonas salmonicida in sunshine bass (Morone chrysops × Morone saxatilis). Vet. Immunol. Immunopathol. 1996, 50, 157–166. [Google Scholar] [CrossRef]
- Eggset, G.; Mikkelsen, H.; Killie, J.-E.A. Immunocompetence and duration of immunity against Vibrio salmonicida and Aeromonas salmonicida after vaccination of Atlantic salmon (Salmo salar L.) at low and high temperatures. Fish Shellfish Immunol. 1997, 7, 247–260. [Google Scholar] [CrossRef]
- Steinhagen, D. Temperature modulation of the response of Ig-positive cells to Goussia carpelli (Protozoa: Apicomplexa) infections in carp, Cyprinus Carpio L. J. Parasitol. 1997, 83, 434–439. [Google Scholar] [CrossRef]
- Avtalion, R.R. Temperature effect on antibody production and immunological memory, in carp (Cyprinus carpio) immunized against bovine serum albumin (BSA). Immunology 1969, 17, 927. [Google Scholar]
- Avtalion, R.R.; Wojdani, A.; Duczyminer, M. Antibody production in carp (Cyprinus carpio), temperature effect and mechanism. In Proceedings of the Colloquium Organized by the French Society of Immunol (INSERM), Paris, France, 12–14 October 1972; pp. 75–86. [Google Scholar]
- Rijkers, G.T.; Frederix-Wolters, E.M.; Van Muiswinkel, W.B. The immune system of cyprinid fish. Kinetics and temperature dependence of antibody-producing cells in carp (Cyprinus carpio). Immunology 1980, 41, 91. [Google Scholar]
- Cone, R.E.; Marchalonis, J.J. Cellular and humoral aspects of the influence of environmental temperature on the immune response of poikilothermic vertebrates. J. Immunol. 1972, 108, 952–957. [Google Scholar]
- Avtalion, R.R.; Wojdani, A.; Malik, Z.; Shahrabani, R.; Duczyminer, M. Influence of environmental temperature on the immune response in fish. In Current Topics in Microbiology and Immunology/Ergebnisse der Mikrobiologie und Immunitätsforschung; Springer: Berlin/Heidelberg, Germany, 1973; pp. 1–35. [Google Scholar]
- Cecchini, S.; Saroglia, M. Antibody response in sea bass (Dicentrarchus labrax L.) in relation to water temperature and oxygenation. Aquac. Res. 2002, 33, 607–613. [Google Scholar] [CrossRef]
- Boltana, S.; Rey, S.; Roher, N.; Vargas, R.; Huerta, M.; Huntingford, F.A.; Goetz, F.W.; Moore, J.; Garcia-Valtanen, P.; Estepa, A. Behavioural fever is a synergic signal amplifying the innate immune response. Proc. R. Soc. B Biol. Sci. 2013, 280, 20131381. [Google Scholar] [CrossRef]
- Rakus, K.; Ronsmans, M.; Vanderplasschen, A. Behavioral fever in ectothermic vertebrates. Dev. Comp. Immunol. 2017, 66, 84–91. [Google Scholar] [CrossRef]
- Evans, S.S.; Repasky, E.A.; Fisher, D.T. Fever and the thermal regulation of immunity: The immune system feels the heat. Nat. Rev. Immunol. 2015, 15, 335–349. [Google Scholar] [CrossRef]
- Gräns, A.; Rosengren, M.; Niklasson, L.; Axelsson, M. Behavioural fever boosts the inflammatory response in rainbow trout Oncorhynchus mykiss. J. Fish. Biol. 2012, 81, 1111–1117. [Google Scholar] [CrossRef]
- Soares, M.P.; Teixeira, L.; Moita, L.F. Disease tolerance and immunity in host protection against infection. Nat. Rev. Immunol. 2017, 17, 83. [Google Scholar] [CrossRef] [PubMed]
- Lochmiller, R.L.; Deerenberg, C. Trade-offs in evolutionary immunology: Just what is the cost of immunity? Oikos 2000, 88, 87–98. [Google Scholar] [CrossRef]
- Colorni, A. A systemic mycobacteriosis in the European sea bass Dicentrarchus labrax cultured in Eilat (Red Sea). Isr. J. Aquac. 1992, 44, 75–81. [Google Scholar]
- Burge, C.A.; Mark Eakin, C.; Friedman, C.S.; Froelich, B.; Hershberger, P.K.; Hofmann, E.E.; Petes, L.E.; Prager, K.C.; Weil, E.; Willis, B.L. Climate change influences on marine infectious diseases: Implications for management and society. Ann. Rev. Mar. Sci. 2014, 6, 249–277. [Google Scholar] [CrossRef] [PubMed]
- Karvonen, A.; Rintamäki, P.; Jokela, J.; Valtonen, E.T. Increasing water temperature and disease risks in aquatic systems: Climate change increases the risk of some, but not all, diseases. Int. J. Parasitol. 2010, 40, 1483–1488. [Google Scholar] [CrossRef]
- Lafferty, K.D.; Harvell, C.D.; Conrad, J.M.; Friedman, C.S.; Kent, M.L.; Kuris, A.M.; Powell, E.N.; Rondeau, D.; Saksida, S.M. Infectious diseases affect marine fisheries and aquaculture economics. Ann. Rev. Mar. Sci. 2015, 7, 471–496. [Google Scholar] [CrossRef] [PubMed]
- Maynard, J.; Van Hooidonk, R.; Eakin, C.M.; Puotinen, M.; Garren, M.; Williams, G.; Heron, S.F.; Lamb, J.; Weil, E.; Willis, B. Projections of climate conditions that increase coral disease susceptibility and pathogen abundance and virulence. Nat. Clim. Chang. 2015, 5, 688. [Google Scholar] [CrossRef]
- Groner, M.L.; Maynard, J.; Breyta, R.; Carnegie, R.B.; Dobson, A.; Friedman, C.S.; Froelich, B.; Garren, M.; Gulland, F.M.D.; Heron, S.F. Managing marine disease emergencies in an era of rapid change. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150364. [Google Scholar] [CrossRef] [PubMed]
- Palm, H.W. Fish parasites as biological indicators in a changing world: Can we monitor environmental impact and climate change? In Progress in Parasitology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 223–250. [Google Scholar]
- Rigos, G.; Katharios, P. Pathological obstacles of newly-introduced fish species in Mediterranean mariculture: A review. Rev. Fish Biol. Fish. 2010, 20, 47–70. [Google Scholar] [CrossRef]
- Pasternak, Z.; Diamant, A.; Abelson, A. Co-Invasion of a Red Sea fish and its ectoparasitic monogenean, Polylabris cf. mamaevi into the Mediterranean: Observations on oncomiracidium behavior and infection levels in both seas. Parasitol. Res. 2007, 100, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Lokmer, A.; Wegner, K.M. Hemolymph microbiome of Pacific oysters in response to temperature, temperature stress and infection. ISME J. 2015, 9, 670–682. [Google Scholar] [CrossRef]
- Matanza, X.M.; Osorio, C.R. Exposure of the opportunistic marine pathogen Photobacterium damselae subsp. damselae to human body temperature is a stressful condition that shapes the transcriptome, viability, cell morphology, and virulence. Front. Microbiol. 2020, 11, 1771. [Google Scholar] [CrossRef] [PubMed]
- Matanza, X.M.; Osorio, C.R. Transcriptome changes in response to temperature in the fish pathogen Photobacterium damselae subsp. damselae: Clues to understand the emergence of disease outbreaks at increased seawater temperatures. PLoS ONE 2018, 13, e0210118. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.V.; Peixoto, P.G.; Ribeiro, D.D.; Araujo, M.C.; do Santos, C.T.B.; Hayashi, C.; Pedreira, M.M.; Pelli, A. Klebsiella pneumoniae as a main cause of infection in Nishikigoi Cyprinus carpio (carp) by inadequate handling. Braz. J. Vet. Pathol. 2014, 7, 86–88. [Google Scholar]
- Tripathy, S.; Sen, R.; Padhi, S.K.; Mohanty, S.; Maiti, N.K. Upregulation of transcripts for metabolism in diverse environments is a shared response associated with survival and adaptation of Klebsiella pneumoniae in response to temperature extremes. Funct. Integr. Genom. 2014, 14, 591–601. [Google Scholar] [CrossRef]
- Evans, T.G. Considerations for the use of transcriptomics in identifying the ‘genes that matter’ for environmental adaptation. J. Exp. Biol. 2015, 218, 1925–1935. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Hikida, M.; Masumura, K. Flexibacter maritimus sp. nov., a pathogen of marine fishes. Int. J. Syst. Evol. Microbiol. 1986, 36, 396–398. [Google Scholar] [CrossRef]
- Casadevall, A.; Pirofski, L. Host-Pathogen interactions: The attributes of virulence. J. Infect. Dis. 2001, 184, 337–344. [Google Scholar] [CrossRef]
- Woo, P.T.K.; Bruno, D.W. Fish, Diseases and Disorders. Viral, Bacterial and Fungal Infections; CABI: Wallingford, CT, USA, 1999; Volume 3. [Google Scholar]
- Smyrli, M.; Triga, A.; Dourala, N.; Varvarigos, P.; Pavlidis, M.; Quoc, V.H.; Katharios, P. Comparative Study on A Novel Pathogen of European Seabass. Diversity of Aeromonas veronii in the Aegean Sea. Microorganisms 2019, 7, 504. [Google Scholar]
- Smyrli, M.; Prapas, A.; Rigos, G.; Kokkari, C.; Pavlidis, M.; Katharios, P. Aeromonas veronii infection associated with high morbidity and mortality in farmed European seabass Dicentrarchus labrax in the Aegean Sea, Greece. Fish Pathol. 2017, 52, 68–81. [Google Scholar] [CrossRef]
- Crespo, S.; Zarza, C.; Padrós, F. Short communication Epitheliocystis hyperinfection in sea bass, Dicentrarchus labrax (L.): Light and electron microscope observations. J. Fish Dis. 2001, 24, 557–560. [Google Scholar] [CrossRef]
- Paperna, I.; Laurencin, F.B. Parasitic infections of sea bass, Dicentrarchus labrax, and gilt head sea bream, Sparus aurata, in mariculture facilities in France. Aquaculture 1979, 16, 173–175. [Google Scholar] [CrossRef]
- Paperna, I. Epitheliocystis infection in wild and cultured sea bream (Sparus aurata, Sparidae) and grey mullets (Liza ramada, Mugilidae). Aquaculture 1977, 10, 169–176. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P.; Crespo, S. New disease problems in Mediterranean aquaculture. In Proceedings of the Actas del 5 Congreso Nacional de Acuicultura, Barcelona, Spain, 10–13 May 1995; pp. 86–97. [Google Scholar]
- Padrós, F.; Crespo, S. Proliferative epitheliocystis associated with monogenean infection in juvenile seabream Sparus aurata in the north east of Spain. Bull. Eur. Assoc. Fish Pathol. 1995, 15, 42–44. [Google Scholar]
- Korun, J.; Olgac, V.; Akgun-Dars, K.; Colorni, A.; Diamant, A. Mycobacteriosis in European sea bass, Dicentrarchus labrax L., cultured in Turkey. Isr. J. Aquac. 2005, 57, 215–222. [Google Scholar]
- Timur, G.; Ürkü, Ç; Çanak, Ö.; G Genç, E.; Erturan, Z. Systemic mycobacteriosis caused by mycobacterium marinum in farmed meagre (Argyrosomus regius), in Turkey. Isr. J. Aquac Bamidgeh 2015, 67, 1–8. [Google Scholar]
- Avsever, M.L.; Çavuşoğlu, C.; Günen, M.Z.; Yazıcıoğlu, Ö.; Eskiizmirliler, S.; Didinen, B.I.; Tunalıgil, S.; Erdal, G.; Özden, M. The first report of Mycobacterium marinum isolated from cultured meagre, Argyrosomus regius. Bull. Eur. Ass. Fish. Pathol 2014, 34, 4. [Google Scholar]
- Mugetti, D.; Varello, K.; Gustinelli, A.; Pastorino, P.; Menconi, V.; Florio, D.; Fioravanti, M.L.; Bozzetta, E.; Zoppi, S.; Dondo, A. Mycobacterium pseudoshottsii in mediterranean fish farms: New trouble for European aquaculture? Pathogens 2020, 9, 610. [Google Scholar] [CrossRef]
- Elkesh, A.; Kantham, K.P.L.; Shinn, A.P.; Crumlish, M.; Richards, R.H. Systemic nocardiosis in a Mediterranean population of cultured meagre, Argyrosomus regius Asso (Perciformes: Sciaenidae). J. Fish Dis. 2013, 36, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Labella, A.; Manchado, M.; Alonso, M.C.; Castro, D.; Romalde, J.L.; Borrego, J.J. Molecular intraspecific characterization of Photobacterium damselae ssp. damselae strains affecting cultured marine fish. J. Appl. Microbiol. 2010, 108, 2122–2132. [Google Scholar]
- Labella, A.; Berbel, C.; Castro, D.; Borrego, J.J.; Manchado, M. Photobacterium damselae subsp. damselae, an Emerging Pathogen Affecting New Cultured Marine Fish Species in Southern Spain; IntechOpen: London, UK, 2011; ISBN 953307759X. [Google Scholar]
- Korun, J.; Timur, G. The first pasteurellosis case in cultured sea bass (Dicentrarchus labrax L.) at low marine water temperatures in Turkey. Isr. J. Aquac. 2005, 57, 197–206. [Google Scholar]
- Romalde, J.L. Photobacterium damselae subsp. piscicida: An integrated view of a bacterial fish pathogen. Int. Microbiol. 2002, 5, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Magariños, B.; Couso, N.; Noya, M.; Merino, P.; Toranzo, A.E.; Lamas, J. Effect of temperature on the development of pasteurellosis in carrier gilthead seabream (Sparus aurata). Aquaculture 2001, 195, 17–21. [Google Scholar] [CrossRef]
- Austin, B.; Austin, D.A.; Munn, C.B. Bacterial Fish Pathogens; Springer: Berlin/Heidelberg, Germany, 2012; ISBN 9400748841. [Google Scholar]
- Berthe, F.C.J.; Michel, C.; Bernardet, J.-F. Identification of Pseudomonas anguilliseptica isolated from several fish species in France. Dis. Aquat. Organ. 1995, 21, 151–155. [Google Scholar] [CrossRef]
- Doménech, A.; Fernández-Garayzábal, J.F.; Lawson, P.; García, J.A.; Cutuli, M.T.; Blanco, M.; Gibello, A.; Moreno, M.A.; Collins, M.D.; Domínguez, L. Winter disease outbreak in sea-bream (Sparus aurata) associated with Pseudomonas anguilliseptica infection. Aquaculture 1997, 156, 317–326. [Google Scholar] [CrossRef]
- Doménech, A.; Fernández-Garayzábal, J.F.; García, J.A.; Cutuli, M.T.; Blanco, M.; Gibello, A.; Moreno, M.A.; Domínguez, L. Association of Pseudomonas anguilliseptica infection with ‘winter disease’ in sea bream, Sparus aurata L. J. Fish Dis. 1999, 22, 69–71. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Egusa, S. Characteristics of a Pseudomonas sp. from an epizootic of pond-cultured eels (Anguilla japonica). Bull. Jpn. Soc. Sci. Fish. 1972, 38, 577–587. [Google Scholar] [CrossRef]
- Avendaño-Herrera, R.; Toranzo, A.E.; Magariños, B. Tenacibaculosis infection in marine fish caused by Tenacibaculum maritimum: A review. Dis. Aquat. Organ. 2006, 71, 255–266. [Google Scholar] [CrossRef]
- Bernardet, J.-F.; Kerouault, B.; Michel, C. Comparative study on Flexibacter maritimus strains isolated from farmed sea bass (Dicentrarchus labrax) in France. Fish Pathol. 1994, 29, 105–111. [Google Scholar] [CrossRef]
- Yardımcı, R.; Timur, G. Isolation and identification of Tenacibaculum maritimum, the causative agent of Tenacibaculosis in farmed sea bass (Dicentrarchus labrax) on the Aegean Sea coast of Turkey. Isr. J. Aquac. Bamidgeh 2015, 67, 1172. [Google Scholar]
- Bordas, M.A.; Balebona, M.C.; Rodriguez-Maroto, J.M.; Borrego, J.J.; Moriñigo, M.A. Chemotaxis of pathogenic vibrio strains towards mucus surfaces of Gilt-Head Sea Bream (Sparus aurata L.). Appl. Environ. Microbiol. 1998, 64, 1573–1575. [Google Scholar] [CrossRef]
- Colorni, A.; Paperna, I.; Gordin, H. Bacterial infections in gilt-head sea bream Sparus aurata cultured at Elat. Aquaculture 1981, 23, 257–267. [Google Scholar] [CrossRef]
- Bellos, G.; Angelidis, P.; Miliou, H. Effect of temperature and seasonality principal epizootiological risk factor on vibriosis and photobacteriosis outbreaks for European sea bass in Greece (1998–2013). J. Aquac. Res. Dev. 2015, 6, 5. [Google Scholar]
- García-Rosado, E.; Cano, I.; Martín-Antonio, B.; Labella, A.; Manchado, M.; Alonso, M.C.; Castro, D.; Borrego, J.J. Co-Occurrence of viral and bacterial pathogens in disease outbreaks affecting newly cultured sparid fish. Int. Microbiol. 2007, 10, 193. [Google Scholar] [PubMed]
- Ulitzur, S. Effect of temperature, salts, pH and other factors on the development of peritrichous flagella in Vibrio alginolyticus. Arch. Microbiol. 1975, 104, 285–288. [Google Scholar] [CrossRef]
- Du, M.; Chen, J.; Zhang, X.; Li, A.; Li, Y. Characterization and resuscitation of viable but nonculturable Vibrio alginolyticus VIB283. Arch. Microbiol. 2007, 188, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.L. Vibrio anguillarum: Influence of temperature, pH, NaCl concentration and incubation time on growth. J. Appl. Bacteriol. 1984, 57, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Larsen, M.H.; Blackburn, N.; Larsen, J.L.; Olsen, J.E. Influences of temperature, salinity and starvation on the motility and chemotactic response of Vibrio anguillarum. Microbiology 2004, 150, 1283–1290. [Google Scholar] [CrossRef]
- Bordas, M.A.; Balebona, M.C.; Zorrilla, I.; Borrego, J.J.; Morinigo, M.A. Kinetics of adhesion of selected fish-pathogenic Vibrio strains of skin mucus of gilt-head sea bream (Sparus aurata L.). Appl. Environ. Microbiol. 1996, 62, 3650–3654. [Google Scholar] [CrossRef]
- Nigrelli, R.F. Enzootics in the New York Aquarium caused by Cryptocaryon irritans Brown, 1951 (= Ichthyophthirius marinus Sikama, 1961), a histophagous ciliate in the skin, eyes and gills of marine fishes. Zoologica 1966, 51, 97–102. [Google Scholar]
- Cheung, P.J.; Nigrelli, R.F.; Ruggieri, G.D. Studies on cryptocaryoniasis in marine fi, 5 h: Effect of temperature and salinity on the reproductive cycle of Cryptocaryon irritans Brown, 1951. J. Fish Dis. 1979, 2, 93–97. [Google Scholar] [CrossRef]
- Wilkie, D.W. Outbreak of cryptocaryoniasis in marine aquaria at Scripps Institute of Oceanography. Calif. Fish. Game 1969, 55, 227–236. [Google Scholar]
- Iglesias, R.; Paramá, A.; Alvarez, M.F.; Leiro, J.; Aja, C.; Sanmartın, M.L. In vitro growth requirements for the fish pathogen Philasterides dicentrarchi (Ciliophora, Scuticociliatida). Vet. Parasitol. 2003, 111, 19–30. [Google Scholar] [CrossRef]
- Paperna, I. Reproduction cycle and tolerance to temperature and salinity of Amyloodinium ocellatum (Brown, 1931) (Dinoflagellida). Ann. Parasitol. Hum. Comp. 1984, 59, 7–30. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P.; Sitjà-Bobadilla, A.; Franco-Sierra, A.; Palenzuela, O. Protozoan parasites of gilthead sea bream, Sparus aurata L., from different culture systems in Spain. J. Fish Dis. 1995, 18, 105–115. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; Alvarez-Pellitero, P. Experimental transmission of Cryptosporidium molnari (Apicomplexa: Coccidia) to gilthead sea bream (Sparus aurata L.) and European sea bass (Dicentrarchus labrax L.). Parasitol. Res. 2003, 91, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Sitjà-Bobadilla, A.; Padrós, F.; Aguilera, C.; Alvarez-Pellitero, P. Epidemiology of Cryptosporidium molnari in Spanish gilthead sea bream (Sparus aurata L.) and European sea bass (Dicentrarchus labrax L.) cultures: From hatchery to market size. Appl. Environ. Microbiol. 2005, 71, 131–139. [Google Scholar] [CrossRef]
- Gjurčević, E.; Kužir, S.; Baždarić, B.; Matanović, K.; Debelić, I.; Marino, F.; Drašner, K.; Rosenthal, B.M. New data on Eimeria dicentrarchi (Apicomplexa: Eimeriidae), a common parasite of farmed European sea bass (Dicentrarchus labrax) from the mid-eastern Adriatic. Vet. Arh. 2017, 87, 77–86. [Google Scholar]
- Spanggaard, B. Ichthyophonus hoferi Epidemiology, Life Cycle, Taxonomy and Significance in Fish Products; Danmarks Fiskeriundersøgelser, Afdeling for Fiskeindustriel Forskning: Lingby, Denmark, 1996. [Google Scholar]
- Spanggaard, B.; Huss, H.H. Growth of the fish parasite Ichthyophonus hoferi under food relevant conditions. Int. J. Food Sci. Technol. 1996, 31, 427–432. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P.; Sitjà-Bobadilla, A. Ceratomyxa spp. (Protozoa: Myxosporea) infections in wild and cultured sea bass, Dicentrarchus labrax, from the Spanish Mediterranean area. J. Fish. Biol. 1993, 42, 889–901. [Google Scholar] [CrossRef]
- Katharios, P.; Garaffo, M.; Sarter, K.; Athanassopoulou, F.; Mylonas, C.C. A case of high mortality due to heavy infestation of Ceratomyxa diplodae in sharpsnout sea bream (Diplodus puntazzo) treated with reproductive steroids. Bull. Eur. Assoc. Fish Pathol. 2007, 27, 43. [Google Scholar]
- Palenzuela, O.; Sitjà-Bobadilla, A.; Álvarez-Pellitero, P. Ceratomyxa sparusaurati (Protozoa: Myxosporea) infections in cultured gilthead sea bream Sparus aurata (Pisces: Teleostei) from Spain: Aspects of the host-parasite relationship. Parasitol. Res. 1997, 83, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Alama-Bermejo, G.; Šíma, R.; Raga, J.A.; Holzer, A.S. Understanding myxozoan infection dynamics in the sea: Seasonality and transmission of Ceratomyxa puntazzi. Int. J. Parasitol. 2013, 43, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Picard-Sánchez, A.; Estensoro, I.; Del Pozo, R.; Palenzuela, O.R.; Piazzon, M.C.; Sitjà-Bobadilla, A. Water temperature, time of exposure and population density are key parameters in Enteromyxum leei fish-to-fish experimental transmission. J. Fish Dis. 2020, 43, 491–502. [Google Scholar] [CrossRef]
- Le Breton, A.; Marques, A. Occurrence of an histozoic Myxidium infection in two marine cultured species: Puntazzo puntazzo C. and Pagrus major. Bull. Eur. Assoc. Fish. Pathol. 1995, 15, 210. [Google Scholar]
- Rigos, G.; Christophilogiannis, P.; Yiagnisi, M.; Andriopoulou, A.; Koutsodimou, M.; Nengas, I.; Alexis, M. Myxosporean infections in Greek mariculture. Aquac. Int. 1999, 7, 361–364. [Google Scholar] [CrossRef]
- Yanagida, T.; Sameshima, M.; Nasu, H.; Yokoyama, H.; Ogawa, K. Temperature effects on the development of Enteromyxum spp. (Myxozoa) in experimentally infected tiger puffer, Takifugu rubripes (Temminck & Schlegel). J. Fish Dis. 2006, 29, 561–567. [Google Scholar]
- Estensoro, I.; Redondo, M.J.; Alvarez-Pellitero, P.; Sitjà-Bobadilla, A. Novel horizontal transmission route for Enteromyxum leei (Myxozoa) by anal intubation of gilthead sea bream Sparus aurata. Dis. Aquat. Organ. 2010, 92, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Sitja-Bobadilla, A.; Alvarez-Pellitero, P. Population dynamics of Sphaerospora dicentrarchi Sitja-Bobadilla et Alvarez-Pellitero, 1992 and S. testicularis Sitja-Bobadilla et Alvarez-Pellitero, 1990 (Myxosporea: Bivalvulida) infections in wild and cultured Mediterranean sea bass (Dicentrarchus labrax). Parasitology 1993, 106, 39–45. [Google Scholar]
- Fioravanti, M.L.; Caffara, M.; Florio, D.; Gustinelli, A.; Marcer, F. Sphaerospora dicentrarchi and S. testicularis (Myxozoa: Sphaerosporidae) in farmed European seabass (Dicentrarchus labrax) from Italy. Folia Parasitologica 2004, 51, 208. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; Alvarez-Pellitero, P. Light and electron microscopic description of Polysporoplasma n.g. (Myxosporea: Bivalvulida), polysporoplasma sparis n. sp. from Sparus aurata (L), and Polysporoplasma mugilis n. sp. from Liza aurata L. Eur. J. Protistol. 1995, 31, 77–89. [Google Scholar] [CrossRef]
- Mladineo, I. Myxosporidean infections in Adriatic cage-reared fish. Bull. Eur. Assoc. Fish Pathol. 2003, 23, 113–122. [Google Scholar]
- Colorni, A.; Diamant, A. Hyperparasitism of trichodinid ciliates on monogenean gill flukes of two marine fish. Dis. Aquat. Organ. 2005, 65, 177–180. [Google Scholar] [CrossRef]
- González-Lanza, C.; Alvarez-Pellitero, P.; Sitjà-Bobadilla, A. Diplectanidae (Monogenea) infestations of sea bass, Dicentrarchus labrax (L.), from the Spanish Mediterranean area. Parasitol. Res. 1991, 77, 307–314. [Google Scholar] [CrossRef]
- Cecchini, S.; Saroglia, M.; Berni, P.; Cognetti-Varriale, A.M. Influence of temperature on the life cycle of Diplectanum aequans (Monogenea, Diplectanidae), parasitic on sea bass, Dicentrarchus labrax (L.). J. Fish Dis. 1998, 21, 73–75. [Google Scholar] [CrossRef]
- Cecchini, S. Influence of temperature on the hatching of eggs of Diplectanum aequans, a parasite of sea bass. Aquac. Int. 1994, 2, 249–253. [Google Scholar]
- Cecchini, S.; Saroglia, M.; Cognetti-Varriale, A.M.; Terova, G.; Sabino, G. Effect of Low Environmental Temperature on Embryonic Development and Egg Hatching of Diplectanum aequans (Monogenea, Diplectanidae) Infecting European Sea Bass, Dicentrarchus Iabrax. Fish Pathol. 2001, 36, 33–34. [Google Scholar] [CrossRef]
- Mladineo, I. Monogenean parasites in Adriatic cage-reared fish. Acta Adriatica 2004, 45, 65–73. [Google Scholar]
- Sánchez-García, N.; Padrós, F.; Raga, J.A.; Montero, F.E. Comparative study of the three attachment mechanisms of diplectanid monogeneans. Aquaculture 2011, 318, 290–299. [Google Scholar] [CrossRef]
- Cruz e Silva, M.; Freitas, M.; Orge, M. Co-Infection by monogenetic trematodes of the genus Microcotyle V. Beneden and Hesse 1863, Lamellodiscus ignoratus Palombi, 1943, the protozoan Trichodina sp. Ehrenberg, 1838 and the presence of epitheliocystis, Vibrio algynoliticus and V. vulnificus in cultured seabream (Sparus aurata L.) in Portugal. Bull. Eur. Assoc. Fish Pathol. 1997, 17, 40–42. [Google Scholar]
- Vagianou, S.; Athanassopoulou, F.; Ragias, V.; Di Cave, D.; Leontides, L.; Golomazou, E. Prevalence and pathology of ectoparasites of Mediterranean Sea bream and sea bass reared under different environmental and aquaculture conditions. Isr. J. Aquac. 2006, 58, 78–88. [Google Scholar]
- Antonelli, L.; Quilichini, Y.; Marchand, B. Biological study of Furnestinia echeneis Euzet and Audouin 1959 (Monogenea: Monopisthocotylea: Diplectanidae), parasite of cultured gilthead sea bream Sparus aurata (Linnaeus 1758) (Pisces: Teleostei) from Corsica. Aquaculture 2010, 307, 179–186. [Google Scholar] [CrossRef]
- Silan, P.; Cabral, P.; Maillard, C. Enlargement of the host range of Polylabris tubicirrus (Monogenea, Polyopisthocotylea) under fish-farming conditions. Aquaculture 1985, 47, 267–270. [Google Scholar] [CrossRef]
- Merella, P.; Cherchi, S.; Garippa, G.; Fioravanti, M.L.; Gustinelli, A.; Salati, F. Outbreak of Sciaenacotyle panceri (Monogenea) on cage-reared meagre Argyrosomus regius (Osteichthyes) from the western Mediterranean Sea. Dis. Aquat. Organ. 2009, 86, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Ternengo, S.; Agostini, S.; Quilichini, Y.; Euzet, L.; Marchand, B. Intensive infestations of Sciaenocotyle pancerii (Monogenea, Microcotylidae) on Argyrosomus regius (Asso) under fish-farming conditions. J. Fish Dis. 2010, 33, 89–92. [Google Scholar] [CrossRef]
- Antonelli, L.; Quilichini, Y.; Marchand, B. Sparicotyle chrysophrii (Van Beneden and Hesse 1863) (Monogenea: Polyopisthocotylea) parasite of cultured Gilthead sea bream Sparus aurata (Linnaeus 1758) (Pisces: Teleostei) from Corsica: Ecological and morphological study. Parasitol. Res. 2010, 107, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Sanz, F. Mortality of cultured seabream (Sparus aurata) caused by an infection with a trematode of the genus Microcotyle. Bull. Eur. Assoc. Fish Pathol. 1992, 12, 186. [Google Scholar]
- Faisal, M.; Imam, E.A. Microcotyle chrysophrii (Monogenea, Polyopisthocotylea), a pathogen for cultured and wild gilthead seabream, Sparus aurata. Pathol. Mar. Sci. 1990, 283–290. [Google Scholar]
- Villar-Torres, M.; Montero, F.E.; Raga, J.A.; Repullés-Albelda, A. Come rain or come shine: Environmental effects on the infective stages of Sparicotyle chrysophrii, a key pathogen in Mediterranean aquaculture. Parasit. Vectors 2018, 11, 558. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; de Felipe, M.C.; Alvarez-Pellitero, P. In vivo and in vitro treatments against Sparicotyle chrysophrii (Monogenea: Microcotylidae) parasitizing the gills of gilthead sea bream (Sparus aurata L.). Aquaculture 2006, 261, 856–864. [Google Scholar] [CrossRef]
- Repullés-Albelda, A.; Holzer, A.S.; Raga, J.A.; Montero, F.E. Oncomiracidial development, survival and swimming behaviour of the monogenean Sparicotyle chrysophrii (Van Beneden and Hesse, 1863). Aquaculture 2012, 338–341, 47–55. [Google Scholar] [CrossRef]
- Euzet, L.; Noisy, D. Microcotyle Chrysophrii van Beneden et Hesse 1863 (Monogenea, Microcotylidae), Parasite du Teleosteen Sparus Aurata: Precisions Morpho-Anatomiques sur L’adulte et L’oncomiracidium. 1978. Available online: https://pascal-francis.inist.fr/vibad/index.php?action=getRecordDetail&idt=8852564 (accessed on 10 July 2021).
- Bier, J.W. Experimental anisakiasis: Cultivation and temperature tolerance determinations. J. Milk Food Technol. 1976, 39, 132–137. [Google Scholar] [CrossRef]
- Højgaard, D.P. Impact of temperature, salinity and light on hatching of eggs of Anisakis simplex (Nematoda, Anisakidae), isolated by a new method, and some remarks on survival of larvae. Sarsia 1998, 83, 21–28. [Google Scholar] [CrossRef]
- Kuhn, T.; Cunze, S.; Kochmann, J.; Klimpel, S. Environmental variables and definitive host distribution: A habitat suitability modelling for endohelminth parasites in the marine realm. Sci. Rep. 2016, 6, 30246. [Google Scholar] [CrossRef]
- Kalay, M.; Dönmez, A.E.; Koyuncu, C.E.; Genc, E.; Şahin, G. Seasonal variation of Hysterothylacium aduncum (Nematoda: Raphidascarididae) infestation in sparid fishes in the Northeast Mediterranean Sea. Turk. J. Vet. Anim. Sci. 2009, 33, 517–523. [Google Scholar]
- Manera, M.; Dezfuli, B.S. Lernanthropus kroyeri infections in farmed sea bass Dicentrarchus labrax: Pathological features. Dis. Aquat. Organ. 2003, 57, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Toksen, E. Lernanthropus kroyeri van Beneden, 1851 (Crustacea: Copepoda) infections of cultured sea bass (Dicentrarchus labrax L.). Bull. Assoc. Fish Pathol. 2007, 27, 49. [Google Scholar]
- Paperna, I. Study of Caligus minimus (Otto, 1821), (Caligidae Copepoda) infections of the sea Bass Dicentrarcbus labrax (L.) in, Bardawil lagoon. Ann. Parasitol. Hum. Comp. 1980, 55, 687–706. [Google Scholar] [CrossRef] [PubMed]
- Ragias, V.; Tontis, D.; Athanassopoulou, F. Incidence of an intense Caligus minimus Otto 1821, C. pageti Russel, 1925, C. mugilis Brian, 1935 and C. apodus Brian, 1924 infection in lagoon cultured sea bass (Dicentrarchus labrax L.) in Greece. Aquaculture 2004, 242, 727–733. [Google Scholar] [CrossRef]
- Papapanagiotou, E.P.; Trilles, J.P. Cymothoid parasite Ceratothoa parallela inflicts great losses on cultured gilthead sea bream Sparus aurata in Greece. Dis. Aquat. Organ. 2001, 45, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Horton, T.; Okamura, B. Cymothoid isopod parasites in aquaculture: A review and case study of a Turkish sea bass (Dicentrarchus labrax) and sea bream (Sparus auratus) farm. Dis. Aquat. Organ. 2001, 46, 181–188. [Google Scholar] [CrossRef]
- Moate, R.M.; Harris, J.E.; McMahon, S. Lymphocystis infections in cultured gilt-head sea bream (Sparus aurata) in the Aegean Sea. Bull. Eur. Assoc. Fish Pathol. 1992, 12, 134. [Google Scholar]
- de Canales, M.L.G.; Muñoz-Cueto, J.A.; Arellano, J.; Garcia-Garcia, A.; Sarasquete, C. Histological and histochemical characteristics of the lymphocystis disease in gilt-head sea bream, Sparus aurata L. from the South-Atlantic coast of Spain. Eur. J. Histochem. 1996, 40, 143–152. [Google Scholar]
- Paperna, I.; Ilana Sabnai, H.; Colorni, A. An outbreak of lymphocystis in Sparus aurata L. in the Gulf of Aqaba, Red Sea. J. Fish Dis. 1982, 5, 433–437. [Google Scholar] [CrossRef]
- Le Breton, A.; Grisez, L.; Sweetman, J.; Ollevier, F. Viral nervous necrosis (VNN) associated with mass mortalities in cage-reared sea bass, Dicentrarchus labrax (L.). J. Fish Dis. 1997, 20, 145–151. [Google Scholar] [CrossRef]
- Arimoto, M.; Sato, J.; Maruyama, K.; Mimura, G.; Furusawa, I. Effect of chemical and physical treatments on the inactivation of striped jack nervous necrosis virus (SJNNV). Aquaculture 1996, 143, 15–22. [Google Scholar] [CrossRef]
- Amigo, J.M.; Gracia, M.-P.; Rius, M.; Salvadó, H.; Maillo, P.A.; Vivarés, C.P. Longevity and effects of temperature on the viability and polar-tube extrusion of spores of Glugea stephani, a microsporidian parasite of commercial flatfish. Parasitol. Res. 1996, 82, 211–214. [Google Scholar] [CrossRef]
- Athanassopoulou, F. A case report of Pleistophora sp. infection in cultured sea bream (Sparus aurata L.) in Greece. Bull. Eur. Assoc. Fish Pathol. 1998, 18, 19–21. [Google Scholar]
- Sengupta, P.; Garrity, P. Sensing temperature. Curr. Biol. 2013, 23, R304–R307. [Google Scholar] [CrossRef] [PubMed]
- Konkel, M.E.; Tilly, K. Temperature-regulated expression of bacterial virulence genes. Microbes Infect. 2000, 2, 157–166. [Google Scholar] [CrossRef]
- Shapiro, R.S.; Cowen, L.E. Thermal control of microbial development and virulence: Molecular mechanisms of microbial temperature sensing. mBio 2012, 3, 5. [Google Scholar] [CrossRef]
- Falconi, M.; Colonna, B.; Prosseda, G.; Micheli, G.; Gualerzi, C.O. Thermoregulation of Shigella and Escherichia coli EIEC pathogenicity. A temperature-dependent structural transition of DNA modulates accessibility of virF promoter to transcriptional repressor H-NS. EMBO J. 1998, 17, 7033–7043. [Google Scholar] [CrossRef] [PubMed]
- Josenhans, C.; Suerbaum, S. The role of motility as a virulence factor in bacteria. Int. J. Med. Microbiol. 2002, 291, 605–614. [Google Scholar] [CrossRef]
- Kamp, H.D.; Higgins, D.E. Transcriptional and post-transcriptional regulation of the GmaR antirepressor governs temperature-dependent control of flagellar motility in Listeria monocytogenes. Mol. Microbiol. 2009, 74, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Chavant, P.; Martinie, B.; Meylheuc, T.; Bellon-Fontaine, M.-N.; Hebraud, M. Listeria monocytogenes LO28: Surface physicochemical properties and ability to form biofilms at different temperatures and growth phases. Appl. Environ. Microbiol. 2002, 68, 728–737. [Google Scholar] [CrossRef]
- Herald, P.J.; Zottola, E.A. Attachment of Listeria monocytogenes to stainless steel surfaces at various temperatures and pH values. J. Food Sci. 1988, 53, 1549–1562. [Google Scholar] [CrossRef]
- Garrett, T.R.; Bhakoo, M.; Zhang, Z. Bacterial adhesion and biofilms on surfaces. Prog. Nat. Sci. 2008, 18, 1049–1056. [Google Scholar] [CrossRef]
- Hasegawa, H.; Chatterjee, A.; Cui, Y.; Chatterjee, A.K. Elevated temperature enhances virulence of Erwinia carotovora subsp. carotovora strain EC153 to plants and stimulates production of the quorum sensing signal, N-acyl homoserine lactone, and extracellular proteins. Appl. Environ. Microbiol. 2005, 71, 4655–4663. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, M. The effects of culture concentration and age, time, and temperature on bacterial attachment to polystyrene. Can. J. Microbiol. 1977, 23, 1. [Google Scholar] [CrossRef]
- Maurelli, A.T.; Blackmon, B.; Curtiss, R. Temperature-Dependent expression of virulence genes in Shigella species. Infect. Immun. 1984, 43, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Mauchline, W.S.; James, B.W.; Fitzgeorge, R.B.; Dennis, P.J.; Keevil, C.W. Growth temperature reversibly modulates the virulence of Legionella pneumophila. Infect. Immun. 1994, 62, 2995–2997. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.F.; Dao, K.M.; Lenski, R.E. Rapid evolution in response to high-temperature selection. Nature 1990, 346, 79. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.F.; Lenski, R.E.; Mittler, J.E. Evolutionary adaptation to temperature. I. Fitness responses of Escherichia coli to changes in its thermal environment. Evolution 1992, 46, 16–30. [Google Scholar]
- Friman, V.-P.; Hiltunen, T.; Jalasvuori, M.; Lindstedt, C.; Laanto, E.; Örmälä, A.-M.; Laakso, J.; Mappes, J.; Bamford, J.K.H. High temperature and bacteriophages can indirectly select for bacterial pathogenicity in environmental reservoirs. PLoS ONE 2011, 6, e17651. [Google Scholar] [CrossRef]
- Yura, T.; Nagai, H.; Mori, H. Regulation of the heat-shock response in bacteria. Annu. Rev. Microbiol. 1993, 47, 321–350. [Google Scholar] [CrossRef]
- Brunn, A.F.; Heiberg, B. Wietere Untersuchungen uber die Rotsenche des Aales in den Dunischen Gewassern. Zentralbl. Fisch. 1935, 33, 379–382. [Google Scholar]
- Mahnken, C.V.W. Status report on commercial salmon culture in Puget Sound. Commer. Fish Farmer Aquac. News 1975, 2, 8–11. [Google Scholar]
- Ormonde, P.; Hörstedt, P.; O’Toole, R.; Milton, D.L. Role of motility in adherence to and invasion of a fish cell line by Vibrio anguillarum. J. Bacteriol. 2000, 182, 2326–2328. [Google Scholar] [CrossRef]
- O’Toole, R.; Milton, D.L.; Wolf-Watz, H. Chemotactic motility is required for invasion of the host by the fish pathogen Vibrio anguillarum. Mol. Microbiol. 1996, 19, 625–637. [Google Scholar] [CrossRef]
- Haldar, S.; Maharajan, A.; Chatterjee, S.; Hunter, S.A.; Chowdhury, N.; Hinenoya, A.; Asakura, M.; Yamasaki, S. Identification of Vibrio harveyi as a causative bacterium for a tail rot disease of sea bream Sparus aurata from research hatchery in Malta. Microbiol. Res. 2010, 165, 639–648. [Google Scholar] [CrossRef]
- Pujalte, M.J.; Sitja-Bobadilla, A.; Macián, M.C.; Belloch, C.; Alvarez-Pellitero, P.; Perez-Sanchez, J.; Uruburu, F.; Garay, E. Virulence and molecular typing of Vibrio harveyi strains isolated from cultured dentex, gilthead sea bream and European sea bass. Syst. Appl. Microbiol. 2003, 26, 284–292. [Google Scholar] [CrossRef]
- Firmino, J.; Furones, M.D.; Andree, K.B.; Sarasquete, C.; Ortiz-Delgado, J.B.; Asencio-Alcudia, G.; Gisbert, E. Contrasting outcomes of Vibrio harveyi pathogenicity in gilthead seabream, Sparus aurata and European seabass, Dicentrachus labrax. Aquaculture 2019, 511, 734210. [Google Scholar] [CrossRef]
- Paperna, I. Review of diseases affecting cultured Sparus aurata and Dicentrarchus labrax [sea bream, sea bass; Dinoflagellate, trichodines, bacterial diseases, metabolic disorders mainly]. In Proceedings of the L’aquaculture du bar (loup) et des Sparides, Sete, France, 15–17 March 1983. [Google Scholar]
- Balebona, M.C.; Zorrilla, I.; Moriñigo, M.A.; Borrego, J.J. Survey of bacterial pathologies affecting farmed gilt-head sea bream (Sparus aurata L.) in southwestern Spain from 1990 to 1996. Aquaculture 1998, 166, 19–35. [Google Scholar] [CrossRef]
- Zorrilla, I.; Chabrillón, M.; Arijo, S.; Dıaz-Rosales, P.; Martınez-Manzanares, E.; Balebona, M.C.; Morinigo, M.A. Bacteria recovered from diseased cultured gilthead sea bream (Sparus aurata L.) in southwestern Spain. Aquaculture 2003, 218, 11–20. [Google Scholar] [CrossRef]
- Balebona, M.C.; Andreu, M.J.; Bordas, M.A.; Zorrilla, I.; Moriñigo, M.A.; Borrego, J.J. Pathogenicity of Vibrio alginolyticus for Cultured Gilt-Head Sea Bream (Sparus aurata L.). Appl. Environ. Microbiol. 1998, 64, 4269–4275. [Google Scholar] [CrossRef] [PubMed]
- Bakhrouf, A.; Ben Ouada, H.; Oueslati, R. Essai de traitement des vibrioses du loup Dicentrarchus labrax dans une zone de pisciculture, à Monastir, Tunisie. Mar. Life 1995, 5, 47–53. [Google Scholar]
- Kahla-Nakbi, A.B.; Chaieb, K.; Besbes, A.; Zmantar, T.; Bakhrouf, A. Virulence and enterobacterial repetitive intergenic consensus PCR of Vibrio alginolyticus strains isolated from Tunisian cultured gilthead sea bream and sea bass outbreaks. Vet. Microbiol. 2006, 117, 321–327. [Google Scholar] [CrossRef]
- Castillo, D.; D’Alvise, P.; Kalatzis, P.G.; Kokkari, C.; Middelboe, M.; Gram, L.; Liu, S.; Katharios, P. Draft genome sequences of Vibrio alginolyticus strains V1 and V2, opportunistic marine pathogens. Genome Announc. 2015, 3, 4. [Google Scholar] [CrossRef]
- Yan, Q.; Chen, Q.; Ma, S.; Zhuang, Z.; Wang, X. Characteristics of adherence of pathogenic Vibrio alginolyticus to the intestinal mucus of large yellow croaker (Pseudosciaena crocea). Aquaculture 2007, 269, 21–30. [Google Scholar] [CrossRef]
- Gauthier, G.; Lafay, B.; Ruimy, R.; Breittmayer, V.; Nicolas, J.-L.; Gauthier, M.; Christen, R. Small-Subunit rRNA Sequences and Whole DNA Relatedness Concur for the Reassignment of Pasteurella piscicida (Snieszko et al.) Janssen and Surgalla to the Genus Photobacterium as Photobacterium damsela subsp. piscicida comb. nov. Int. J. Syst. Evol. Microbiol. 1995, 45, 139–144. [Google Scholar] [CrossRef]
- Toranzo, A.E.; Casal, J.F.; Figueras, A.; Magarin, B.; Barja, J.L. Pasteurellosis in cultured gilthead seabream (Sparus aurata): First report in Spain. Aquaculture 1991, 99, 1–15. [Google Scholar] [CrossRef]
- Baudin-Laurencin, F.; Pepin, J.F.; Raymond, J.C. First observation of an epizootic of pasteurellosis in farmed and wild fish of the French Mediterranean coasts. In Proceedings of the 5th International Conference of the European Association of Fish Pathology, Budapest, Hungary, 25–29 August 1991; p. 17. [Google Scholar]
- Love, M.; Teebken-Fisher, D.; Hose, J.E.; Farmer, J.J.; Hickman, F.W.; Fanning, G.R. Vibrio damsela, a marine bacterium, causes skin ulcers on the damselfish Chromis punctipinnis. Science 1981, 214, 1139–1140. [Google Scholar] [CrossRef]
- Mahmoud, S.A.; El-Bouhy, Z.M.; Hassanin, M.E.; Fadel, A.H. Vibrio alginolyticus and Photobacterium damselae subsp. damselae: Prevalence, histopathology and treatment in sea bass Dicentrarchus labrax. J. Pharm. Chem. Biol. Sci. 2017, 5, 354–364. [Google Scholar]
- Essam, H.M.; Abdellrazeq, G.S.; Tayel, S.I.; Torky, H.A.; Fadel, A.H. Pathogenesis of Photobacterium damselae subspecies infections in sea bass and sea bream. Microb. Pathog. 2016, 99, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Uzun, E.; Ogut, H. The isolation frequency of bacterial pathogens from sea bass (Dicentrarchus labrax) in the Southeastern Black Sea. Aquaculture 2015, 437, 30–37. [Google Scholar] [CrossRef]
- Vera, P.; Navas, J.I.; Fouz, B. First isolation of Vibrio damsela from seabream (Sparus aurata). Bull. Eur. Assoc. Fish Pathol. 1991, 11, 112. [Google Scholar]
- Toranzo, A.E.; Magariños, B.; Romalde, J.L. A review of the main bacterial fish diseases in mariculture systems. Aquaculture 2005, 246, 37–61. [Google Scholar] [CrossRef]
- Pepin, J.-F.; Emery, E. Marine Cytophaga-like bacteria (CLB) isolated from diseased reared sea bass (Dicentrarchus labrax L.) from French Mediterranean coast. Bull. Eur. Assoc. Fish. Pathol. 1993, 13, 165. [Google Scholar]
- Kolygas, M.N.; Gourzioti, E.; Vatsos, I.N.; Athanassopoulou, F. Identification of Tenacibaculum maritimum strains from marine farmed fish in Greece. Vet. Rec. 2012, 170, 623. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.M.; Stine, C.B.; Baya, A.M.; Kent, M.L. A review of mycobacteriosis in marine fish. J. Fish Dis. 2009, 32, 119–130. [Google Scholar] [CrossRef]
- Ucko, M.; Colorni, A.; Kvitt, H.; Diamant, A.; Zlotkin, A.; Knibb, W.R. Strain variation in Mycobacterium marinum fish isolates. Appl. Environ. Microbiol. 2002, 68, 5281–5287. [Google Scholar] [CrossRef]
- Batra, P.; Mathur, P.; Misra, M.C. Aeromonas spp.: An Emerging Nosocomial Pathogen. J. Lab. Physicians 2016, 8, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Groberg, W.J., Jr.; McCoy, R.H.; Pilcher, K.S.; Fryer, J.L. Relation of water temperature to infections of coho salmon (Oncorhynchus kisutch), chinook salmon (O. tshawytscha), and steelhead trout (Salmo gairdneri) with Aeromonas salmonicida and A. hydrophila. J. Fish. Board Can. 1978, 35, 1. [Google Scholar] [CrossRef]
- Yiagnisis, M.; Athanassopoulou, F. Bacteria isolated from diseased wild and farmed marine fish in Greece. In Recent Advances in Fish Farms; IntechOpen: London, UK, 2011. [Google Scholar]
- Blandford, M.I.; Taylor-Brown, A.; Schlacher, T.A.; Nowak, B.; Polkinghorne, A. Epitheliocystis in fish: An emerging aquaculture disease with a global impact. Transbound. Emerg. Dis. 2018, 65, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- Nowak, B.F.; LaPatra, S.E. Epitheliocystis in fish. J. Fish Dis. 2006, 29, 573–588. [Google Scholar] [CrossRef] [PubMed]
- Paperna, I.; Sabnai, I.; Zachary, A. Ultrastructural studies in piscine epitheliocystis: Evidence for a pleomorphic developmental cycle. J. Fish Dis. 1981, 4, 459–472. [Google Scholar] [CrossRef]
- Crespo, S.; Zarza, C.; Padros, F.; de Mateo, M.M. Epitheliocystis agents in sea bream Sparus aurata: Morphological evidence for two distinct chlamydia-like developmental cycles. Dis. Aquat. Organ. 1999, 37, 61–72. [Google Scholar] [CrossRef]
- Qi, W.; Vaughan, L.; Katharios, P.; Schlapbach, R.; Seth-Smith, H.M.B. Host-Associated Genomic Features of the Novel Uncultured Intracellular Pathogen Ca. Ichthyocystis Revealed by Direct Sequencing of Epitheliocysts. Genome Biol. Evol. 2016, 8, 1672–1689. [Google Scholar] [CrossRef]
- Tsertou, M.I.; Smyrli, M.; Kokkari, C.; Antonopoulou, E.; Katharios, P. The aetiology of systemic granulomatosis in meagre (Argyrosomus regius): The “Nocardia” hypothesis. Aquac. Rep. 2018, 12, 5–11. [Google Scholar] [CrossRef]
- Cone, D.K. Monogenea (Phylum Platyhelminthes). PTK Fish. Dis. Disord. 1995, 1, 289–327. [Google Scholar]
- Silan, P.; Maillard, C. Biologie comparée du développement et discrimination des Diplectanidae ectoparasites du Bar (Teleostei). Ann. Sci. Nat. Zool. Biol. Anim. 1989, 10, 31–45. [Google Scholar]
- Kim, K.H.; Ahn, K.J.; Kim, C.S. Seasonal abundances of Prosomicrocotyla gotoi (Monogenea) and Opecoelus sphaericus (Digenea) from greenlings Hexagrammos otakii in a southern coastal area in Korea. Aquaculture 2001, 192, 147–153. [Google Scholar] [CrossRef]
- Gannicott, A.M.; Tinsley, R.C. Larval survival characteristics and behaviour of the gill monogenean Discocotyle sagittata. Parasitology 1998, 117, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, L.; Foata, J.; Quilichini, Y.; Marchand, B. Influence of season and site location on European cultured sea bass parasites in Corsican fish farms using indicator species analysis (IndVal). Parasitol. Res. 2016, 115, 561–568. [Google Scholar] [CrossRef]
- Hoffman, G.L.; Putz, R.E. Host susceptibility and the effect of aging, freezing, heat, and chemicals on spores of Myxosoma cerebralis. Progress. Fish-Cult. 1969, 31, 35–37. [Google Scholar] [CrossRef]
- Byers, J.E. Marine Parasites and Disease in the Era of Global Climate Change. Ann. Rev. Mar. Sci. 2021, 13, 397–470. [Google Scholar] [CrossRef] [PubMed]
- Marcogliese, D.J. The impact of climate change on the parasites and infectious diseases of aquatic animals. Rev. Sci. Tech. 2008, 27, 467–484. [Google Scholar] [CrossRef]
- Marcogliese, D.J. The distribution and abundance of parasites in aquatic ecosystems in a changing climate: More than just temperature. Integr. Comp. Biol. 2016, 56, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Lõhmus, M.; Björklund, M. Climate change: What will it do to fish-parasite interactions? Biol. J. Linn. Soc. 2015, 116, 397–411. [Google Scholar] [CrossRef]
- Marcogliese, D.J. Implications of climate change for parasitism of animals in the aquatic environment. Can. J. Zool. 2001, 79, 1331–1352. [Google Scholar] [CrossRef]
- Lafferty, K.D.; Holt, R.D. How should environmental stress affect the population dynamics of disease? Ecol. Lett. 2003, 6, 654–664. [Google Scholar] [CrossRef]
- Studer, A.; Thieltges, D.W.; Poulin, R. Parasites and global warming: Net effects of temperature on an intertidal host-parasite system. Mar. Ecol. Prog. 2010, 415, 11–22. [Google Scholar] [CrossRef]
- Soares, F.; Roque, A.; Gavaia, P.J. Review of the principal diseases affecting cultured meagre (Argyrosomus regius). Aquac. Res. 2018, 49, 1373–1382. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P. Report about Fish Parasitic Diseases; Options Méditerranéennes B Etudes et Recherches: Zaragoza, Spain, 2004; pp. 103–130. Available online: http://om.ciheam.org/article.php?IDPDF=4600222 (accessed on 20 July 2021).
- Reversat, J.; Silan, P.; Maillard, C. Structure of monogenean populations, ectoparasites of the gilthead sea bream Sparus aurata. Mar. Biol. 1992, 112, 43–47. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; Palenzuela, O. Enteromyxum species. In Fish Parasites. Pathobiology and Protection; CABI: Wallingford, CT, USA, 2012; Volume 163, p. 76. [Google Scholar]
- Fioravanti, M.L.; Mladineo, I.; Palenzuela, O.; Beraldo, P.; Massimo, M.; Gustinelli, A.; Sitjà-Bobadilla, A. Fish. Farmer’s Guide to Combating Parasitic Infections in European Sea Bass and Gilthead Sea Bream Aquaculture; A Series of ParaFishControl Guides to Combating Fish Parasite Infections in Aquaculture. Guide, 4; Sitjà-Bobadilla, A., Bello-Gómez, E., Eds.; Consejo Superior de Investigaciones Científicas: Madrid, Spain, 2020; e-NIPO: 833-20-104-5. [Google Scholar]
- Šarušic, G. Preliminary report of infestation by isopod Ceratothoa oestroides (Risso, 1826), in marine cultured fish. Bull. Eur. Assoc. Fish Pathol. 1999, 19, 110–112. [Google Scholar]
- Čolak, S.; Kolega, M.; Mejdandžić, D.; Župan, I.; Šarić, T.; Piplović, E.; Mustać, B. Prevalence and effects of the cymothoid isopod (Ceratothoa oestroides, Risso 1816) on cultured meagre (Argyrosomus regius, Asso 1801) in the Eastern Adriatic Sea. Aquac. Res. 2018, 49, 1001–1007. [Google Scholar] [CrossRef]
- Öktener, A.; Trilles, J.-P. Report on cymothoids (Crustacea, Isopoda) collected from marine fishes in Turkey. Acta Adriatica 2004, 45, 145–154. [Google Scholar]
- Mladineo, I. Life cycle of Ceratothoa oestroides, a cymothoid isopod parasite from sea bass Dicentrarchus labrax and sea bream Sparus aurata. Dis. Aquat. Organ. 2003, 57, 97–101. [Google Scholar] [CrossRef]
- Mladineo, I.; Hrabar, J.; Vidjak, O.; Bočina, I.; Čolak, S.; Katharios, P.; Cascarano, M.C.; Keklikoglou, K.; Volpatti, D.; Beraldo, P. Host-Parasite Interaction between Parasitic Cymothoid Ceratothoa oestroides and its Host, Farmed European Sea Bass (Dicentrarchus labrax). Pathogens 2020, 9, 230. [Google Scholar] [CrossRef]
- Arechavala-Lopez, P.; Sanchez-Jerez, P.; Bayle-Sempere, J.T.; Uglem, I.; Mladineo, I. Reared fish, farmed escapees and wild fish stocks—A triangle of pathogen transmission of concern to Mediterranean aquaculture management. Aquac. Environ. Interact. 2013, 3, 153–161. [Google Scholar] [CrossRef]
- Mladineo, I.; Hrabar, J.; Trumbić, Ž.; Manousaki, T.; Tsakogiannis, A.; Taggart, J.B.; Tsigenopoulos, C.S. Community Parameters and Genome-Wide RAD-Seq Loci of Ceratothoa oestroides Imply its Transfer between Farmed European Sea Bass and Wild Farm-Aggregating Fish. Pathogens 2021, 10, 100. [Google Scholar] [CrossRef]
- Woo, P.T.K. Fish, Diseases and Disorders. Protozoan and Metazoan infections; CABI: Wallingford, CT, USA, 1995; Volume 1, ISBN 0851990150. [Google Scholar]
- Colorni, A. Biology of Cryptocaryon irritans and strategies for its control. Aquaculture 1987, 67, 236–237. [Google Scholar] [CrossRef]
- Rigos, G.; Pavlidis, M.; Divanach, P. Host susceptibility to Cryptocaryon sp. infection of Mediterranean marine broodfish held under intensive culture conditions: A case report. Bull. Assoc. Fish Pathol. 2001, 21, 33–36. [Google Scholar]
- Colorni, A.; Burgess, P. Cryptocaryon irritans Brown 1951, the cause of ‘white spot disease’ in marine fish: An update. Aquar. Sci. Conserv. 1997, 1, 217–238. [Google Scholar] [CrossRef]
- Paperna, I. Amyloodinium ocellatum (Brown, 1931) (Dinoflagellida) infestations in cultured marine fish at Eilat, Red Sea: Epizootiology and pathology. J. Fish Dis. 1980, 3, 363–372. [Google Scholar] [CrossRef]
- Francis-Floyd, R.; Floyd, M.R. Amyloodinium ocellatum, An Important Parasite of Cultured Marine Fish; Southern Regional Aquaculture Center: Stoneville, MS, USA, 2011. [Google Scholar]
- Moreira, M.; Schrama, D.; Soares, F.; Wulff, T.; Pousão-Ferreira, P.; Rodrigues, P. Physiological responses of reared sea bream (Sparus aurata Linnaeus, 1758) to an Amyloodinium ocellatum outbreak. J. Fish Dis. 2017, 40, 1545–1560. [Google Scholar] [CrossRef] [PubMed]
- Soares, F.; Quental-Ferreira, H.; Moreira, M.; Cunha, E.; Ribeiro, L.; Pousão-Ferreira, P. First report of Amyloodinium ocellatum in farmed meagre (Argyrosomus regius). Bull. Eur. Assoc. Fish Pathol. 2012, 32, 30–33. [Google Scholar]
- Woo, P.T.; Bruno, D.W.; Lim, L.H.S. Diseases and Disorders of Finfish in Cage Culture; CABI: Wallingford, CT, USA, 2002; ISBN 1780642075. [Google Scholar]
- Fioravanti, M.L.; Caffara, M.; Florio, D.; Gustinelli, A.; Marcer, F. A parasitological survey of European sea bass (Dicentrarchus labrax) and gilthead sea bream (Sparus aurata) cultured in Italy. Vet. Res. Commun. 2006, 30, 249–252. [Google Scholar] [CrossRef]
- Alvárez-Pellitero, P.; Sitjà-Bobadilla, A.; Franco-Sierra, A. Protozoan parasites of wild and cultured sea bass, Dicentrarchus labrax (L.), from the Mediterranean area. Aquac. Res. 1993, 24, 101–108. [Google Scholar] [CrossRef]
- Brazik, D.C.; Bullis, R.A. The Effect of Temperature on the Relationship between a Ciliated Protozoan, Trichodina cottidarum, and the Longhorn Sculpin, Myoxocephalus octodecemspinosus. Biol. Bull. 1995, 189, 239. [Google Scholar] [CrossRef]
- Lom, J.; Laird, M. Parasitic protozoa from marine and euryhaline fish of Newfoundland and New Brunswick. I. Peritrichous ciliates. Can. J. Zool. 1969, 47, 1367–1380. [Google Scholar] [CrossRef]
- Dragesco, A.; Dragesco, J.; Coste, F.; Gasc, C.; Romestand, B.; Raymond, J.-C.; Bouix, G. Philasterides dicentrarchi, n. sp.,(Ciliophora, Scuticociliatida), a histophagous opportunistic parasite of Dicentrarchus labrax (Linnaeus, 1758), a reared marine fish. Eur. J. Protistol. 1995, 31, 327–340. [Google Scholar] [CrossRef]
- Post, G.W. Textbook of Fish Health; TFH Publications: Neptune City, NJ, USA, 1983; ISBN 0876665997. [Google Scholar]
- Isaksen, T.E. Ichthyobodo Infections on Farmed and wild Fish—Methods for Detection and Identification of Ichthyobodo spp. Ph. D. Thesis, University of Bergen, Bergen, Norway, 2013. [Google Scholar]
- Schäperclaus, W. Fish Diseases; CRC Press: Boca Raton, FL, USA, 1992; Volume 2. ISBN 906191 9517.
- Lom, J.; Dyková, I. Protozoan Parasites of Fishes; Elsevier: Amsterdam, The Netherlands, 1992; ISBN 0444894349. [Google Scholar]
- Santos, M.J. Observations on the parasitofauna of wild sea bass (Dicentrarchus labrax L.) from Portugal. Bull. Assoc. Fish Pathol. 1996, 16, 77–79. [Google Scholar]
- Yardimci, B.; Pekmezci, G.Z.; Behire, I.D.; Metin, S. Ichthyobodo spp. Infection in Meagre (Argyrosomus regius) from Turkey: Parasitological and Pathological Findings. Türkiye Parazitolojii Derg. 2016, 40, 48. [Google Scholar] [CrossRef]
- Tokșen, E. First detection of Ichthyobodo spp. infection and its treatment in a sea bream (Sparus aurata L.) farm in Izmir. Türkiye Parazitoloji Derg. 2000, 24, 321–325. [Google Scholar]
- Sitjà-Bobadilla, A.; Palenzuela, O.; Alvarez-Pellitero, P. Light microscopic description of Eimeria sparis sp. nov. and Goussia sparis sp. nov.(Protozoa: Apicomplexa) from Sparus aurata L.(Pisces: Teleostei). Parasitol. Res. 1996, 82, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Merella, P.; Garippa, G.; Salati, F. Parasites of cage cultured European seabass Dicentrarchus Labrax and gilthead seabream Sparus aurata from Sardinia (western Mediterranean): First results. Parrasitologia 2006, 48, 290. [Google Scholar]
- Alvarez-Pellitero, P.; Sitjà-Bobadilla, A. Cryptosporidium molnari n. sp. (Apicomplexa: Cryptosporidiidae) infecting two marine fish species, Sparus aurata L. and Dicentrarchus labrax L. Int. J. Parasitol. 2002, 32, 1007–1021. [Google Scholar] [CrossRef]
- Athanassopoulou, F. Ichthyophoniasis in sea bream, Sparus aurata (L.), and rainbow trout, Oncorhynchus mykiss (Walbaum), from Greece. J. Fish Dis. 1992, 15, 437–441. [Google Scholar] [CrossRef]
- Ragan, M.A.; Goggin, C.L.; Cawthorn, R.J.; Cerenius, L.; Jamieson, A.V.; Plourde, S.M.; Rand, T.G.; Söderhäll, K.; Gutell, R.R. A novel clade of protistan parasites near the animal-fungal divergence. Proc. Natl. Acad. Sci. USA 1996, 93, 11907–11912. [Google Scholar] [CrossRef] [PubMed]
- Spanggaard, B.; Huss, H.H.; Bresciani, J. Morphology of Ichthyophonus hoferi assessed by light and scanning electron microscopy. J. Fish Dis. 1995, 18, 567–577. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; Alvarez-Pellitero, P. Light and electron microscopic description of Sphaerospora dicentrarchi n. sp.(Myxosporea: Sphaerosporidae) from wild and cultured sea bass, Dicentrarchus labrax L. J. Protozool. 1992, 39, 273–281. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; Alvarez-Pellitero, P. Sphaerospora testicularis sp. nov. (Myxosporea: Sphaerosporidae) in wild and cultured sea bass, Dicentrarchus labrax (L.), from the Spanish Mediterranean area. J. Fish Dis. 1990, 13, 193–203. [Google Scholar] [CrossRef]
- Sitjà-Bobadilla, A.; Alvarez-Pellitero, P. Pathologic effects of Sphaerospora dicentrarchi Sitjà-Bobadilla and Alvarez-Pellitero, 1992 and S. testicularis Sitjà-Bobadilla and Alvarez-Pellitero, 1990 (Myxosporea: Bivalvulida) parasitic in the Mediterranean sea bass Dicentrarchus labrax L. (Teleostei: Serranidae) and the cell-mediated immune reaction: A light and electron microscopy study. Parasitol. Res. 1993, 79, 119–129. [Google Scholar] [PubMed]
- Palenzuela, O.; Alvarez-Pellitero, P.; Sitjà-Bobadilla, A. Glomerular disease associated with Polysporoplasma sparis (Myxozoa) infections in cultured gilthead sea bream, Sparus aurata L. (Pisces: Teleostei). Parasitology 1999, 118, 245–256. [Google Scholar] [CrossRef]
- Paling, J.E. The attachment of the monogenean Diplectanum aequans (Wagener) Diesing to the gills of Morone labrax L. Parasitology 1966, 56, 493–503. [Google Scholar] [CrossRef]
- Oliver, G. Effect pathogène de la fixation de Diplectanum aequans (Wagener, 1857) Diesing, 1858 (Monogenea, Monopisthocotylea, Diplectanidae) sur les branchies de Dicentrarchus labrax (Linnaeus, 1758), (Pisces, Serranidae). Zeitschrift für Parasitenkunde 1977, 53, 7–11. [Google Scholar] [CrossRef]
- Dezfuli, B.; Giari, L.; Simoni, E.; Menegatti, R.; Shinn, A.; Manera, M. Gill histopathology of cultured European sea bass, Dicentrarchus labrax (L.), infected with Diplectanum aequans (Wagener 1857) Diesing 1958 (Diplectanidae: Monogenea). Parasitol. Res. 2007, 100, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Andree, K.B.; Roque, A.; Duncan, N.; Gisbert, E.; Estevez, A.; Tsertou, M.I.; Katharios, P. Diplectanum sciaenae (Van Beneden & Hesse, 1863) (Monogenea) infecting meagre, Argyrosomus regius (Asso, 1801) broodstock in Catalonia, Spain. A case report. Vet. Parasitol. Reg. Stud. Rep. 2015, 1-2, 75–79. [Google Scholar] [CrossRef]
- Mahmoud, N.E.; Mahmoud, A.M.; Fahmy, M.M. Parasitological and Comparative Pathological Studies on Monogenean Infestation of Cultured Sea Bream (Sparus aurata, Spariidae) in Egypt. Oceanography 2014, 2, 1000129. [Google Scholar]
- Mladineo, I.; Maršić-Lučić, J. Host switch of Lamellodiscus elegans (Monogenea: Monopisthocotylea) and Sparicotyle chrysophrii (Monogenea: Polyopisthocotylea) between cage-reared sparids. Vet. Res. Commun. 2007, 31, 153. [Google Scholar] [CrossRef]
- Desdevises, Y. The phylogenetic position of Furnestinia echeneis (Monogenea, Diplectanidae) based on molecular data: A case of morphological adaptation? Int. J. Parasitol. 2001, 31, 205–208. [Google Scholar] [CrossRef]
- Hayward, C.J. Revision of the monogenean genus Polylabris (Microcotylidae). Invertebr. Syst. 1996, 10, 995–1039. [Google Scholar] [CrossRef]
- Athanassopoulou, F.; Ragias, V.; Vagianou, S.; Di Cave, D.; Rigos, G.; Papathanasiou, G.; Georgoulakis, J. Report of Sparicotyle (Microcotyle) chrysophrii Van Beneden and Hesse 1863, Atrispinum seminalis Euzet and Maillard 1973 and Polylabris tubicirrus Paperna and Kohn 1964 (Monogenea) on captive sea bream (Sparus aurata) and sharp snout sea bream (Diplodus p.). Bull. Eur. Assoc. Fish Pathol. 2005, 25, 256–261. [Google Scholar]
- Bernardi, C. Preliminary study on prevalence of larvae of Anisakidae family in European sea bass (Dicentrarchus labrax). Food Control 2009, 20, 433–434. [Google Scholar] [CrossRef]
- Peñalver, J.; Dolores, E.M.; Muñoz, P. Absence of anisakid larvae in farmed European sea bass (Dicentrarchus labrax L.) and gilthead sea bream (Sparus aurata L.) in Southeast Spain. J. Food Prot. 2010, 73, 1332–1334. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, M.L.; Gustinelli, A.; Rigos, G.; Buchmann, K.; Caffara, M.; Pascual, S.; Pardo, M.Á. Negligible risk of zoonotic anisakid nematodes in farmed fish from European mariculture, 2016 to 2018. Eurosurveillance 2021, 26, 1900717. [Google Scholar] [CrossRef]
- Cammilleri, G.; Costa, A.; Graci, S.; Buscemi, M.D.; Collura, R.; Vella, A.; Pulvirenti, A.; Cicero, A.; Giangrosso, G.; Schembri, P. Presence of Anisakis pegreffii in farmed sea bass (Dicentrarchus labrax L.) commercialized in Southern Italy: A first report. Vet. Parasitol. 2018, 259, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Sakanari, J.A.; Mckerrow, J.H. Anisakiasis. Clin. Microbiol. Rev. 1989, 2, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Mladineo, I.; Hrabar, J. Anisakis pegreffii. Trends Parasitol. 2020, 36, 717–718. [Google Scholar] [CrossRef] [PubMed]
- Fiorenza, E.A.; Wendt, C.A.; Dobkowski, K.A.; King, T.L.; Pappaionou, M.; Rabinowitz, P.; Samhouri, J.F.; Wood, C.L. It’s a wormy world: Meta-Analysis reveals several decades of change in the global abundance of the parasitic nematodes Anisakis spp. and Pseudoterranova spp. in marine fishes and invertebrates. Glob. Chang. Biol. 2020, 26, 2854–2866. [Google Scholar] [CrossRef]
- Sterud, E. Parasites of wild sea bass Dicentrarchus labrax from Norway. Dis. Aquat. Organ. 2002, 48, 209–212. [Google Scholar] [CrossRef]
- Deardorff, T.L.; Overstreet, R.M. Review of Hysterothylacium and Iheringascaris (both previously= Thynnascaris) (Nematoda: Anisakidae) from the northern Gulf of Mexico. Proc. Biol. Soc. Washingt. 1980, 93, 1035–1079. [Google Scholar]
- González, L. The life cycle of Hysterothylacium aduncum (Nematoda: Anisakidae) in Chilean marine farms. Aquaculture 1998, 162, 173–186. [Google Scholar] [CrossRef]
- Samak, O.A.A. Mode of attachment and histopathological impacts associated with the parasitic copepod Lernanthropus kroyeri infesting gills of the sea bass fish, Dicentrarchus labrax in Egypt. J. Egypt. Ger. Soc. Zool. 2005, 48, 1. [Google Scholar]
- Horton, T.; Okamura, B. Post-Haemorrhagic anaemia in sea bass, Dicentrarchus labrax (L.), caused by blood feeding of Ceratothoa oestroides (Isopoda: Cymothoidae). J. Fish Dis. 2003, 26, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-S.; O’Bryen, P.J.; Marcus, N.H. Copepods in Aquaculture; Blackwell Publishing: Hoboken, NJ, USA, 2008; ISBN 0470276304. [Google Scholar]
- Breitbart, M. Marine viruses: Truth or dare. Ann. Rev. Mar. Sci. 2012, 4, 425–448. [Google Scholar] [CrossRef]
- Danovaro, R.; Corinaldesi, C.; Dell’Anno, A.; Fuhrman, J.A.; Middelburg, J.J.; Noble, R.T.; Suttle, C.A. Marine viruses and global climate change. FEMS Microbiol. Rev. 2011, 35, 993–1034. [Google Scholar] [CrossRef] [PubMed]
- Rohwer, F.; Thurber, R.V. Viruses manipulate the marine environment. Nature 2009, 459, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.M.; Becnel, J.J. Microsporidia: Pathogens of Opportunity; John Wiley & Sons: Hoboken, NJ, USA, 2014; ISBN 1118395239. [Google Scholar]
- Munday, B.L.; Kwang, J.; Moody, N. Betanodavirus infections of teleost fish: A review. J. Fish Dis. 2002, 25, 127–142. [Google Scholar] [CrossRef]
- Gomez, D.K.; Sato, J.; Mushiake, K.; Isshiki, T.; Okinaka, Y.; Nakai, T. PCR-Based detection of betanodaviruses from cultured and wild marine fish with no clinical signs. J. Fish Dis. 2004, 27, 603–608. [Google Scholar] [CrossRef]
- Mori, K.; Nakai, T.; Nagahara, M.; Muroga, K.; Mekuchi, T.; Kanno, T. A viral disease in hatchery-reared larvae and juveniles of redspotted grouper. Fish. Pathol. 1991, 26, 209–210. [Google Scholar] [CrossRef]
- Frerichs, G.N.; Rodger, H.D.; Peric, Z. Cell culture isolation of piscine neuropathy nodavirus from juvenile sea bass, Dicentrarchus labrax. J. Gen. Virol. 1996, 77, 2067–2071. [Google Scholar] [CrossRef]
- Munday, B.L.; Nakai, T. Nodaviruses as pathogens in larval and juvenile marine finfish. World J. Microbiol. Biotechnol. 1997, 13, 375–381. [Google Scholar] [CrossRef]
- Castri, J.; Thiéry, R.; Jeffroy, J.; de Kinkelin, P.; Raymond, J.C. Sea bream Sparus aurata, an asymptomatic contagious fish host for nodavirus. Dis. Aquat. Organ. 2001, 47, 33–38. [Google Scholar] [CrossRef]
- Lopez-Jimena, B.; Cherif, N.; Garcia-Rosado, E.; Infante, C.; Cano, I.; Castro, D.; Hammami, S.; Borrego, J.J.; Alonso, M.D. A combined RT-PCR and dot-blot hybridization method reveals the coexistence of SJNNV and RGNNV betanodavirus genotypes in wild meagre (Argyrosomus regius). J. Appl. Microbiol. 2010, 109, 1361–1369. [Google Scholar] [CrossRef]
- Arimoto, M.; Maruyama, K.; Furusawa, I. Epizootiology of viral nervous necrosis (VNN) in striped jack. Fish Pathol. 1994, 29, 19–24. [Google Scholar] [CrossRef][Green Version]
- Tanaka, S.; Aoki, H.; Nakai, T. Pathogenicity of the nodavirus detected from diseased sevenband grouper Epinephelus septemfasciatus. Fish Pathol. 1998, 33, 31–36. [Google Scholar] [CrossRef]
- Wolf, K. Fish Viruses and Fish Viral Diseases; Cornell University Press: Ithaca, NY, USA, 1988; ISBN 0801412595. [Google Scholar]
- Kanakoudis, G.; Vlemmas, I.; Papaioannou, N.; Fotis, G.; Pnevmatikos, G. Study of Lymphocystis disease in Sparus auratus in Greece. Deltio Ellinikis Ktiniatrikis Etair. 1992, 43, 109–115. [Google Scholar]
- Wolf, K.; Gravell, M.; Malsberger, R.G. Lymphocystis virus: Isolation and propagation in centrarchid fish cell lines. Science 1966, 151, 1004–1005. [Google Scholar] [CrossRef] [PubMed]
- Mathieu-Daude, F.; Faye, N.; Coste, F.; Manier, J.; Marques, A.; Bouix, G. Occurence of a microsporidiosis in marine cultured gilt-head sea bream from the Languedoc coast: A problem of specificity in the genus Glugea (Protozoa, Microspora). Bull. Eur. Assoc. Fish Pathol. 1992, 12, 67–70. [Google Scholar]
- Morsy, K.; Bashtar, A.R.; Abdel-Ghaffar, F.; Al-Quraishy, S. Morphological and phylogenetic description of a new xenoma-inducing microsporidian, Microsporidium aurata nov. sp., parasite of the gilthead seabream Sparus aurata from the Red Sea. Parasitol. Res. 2013, 112, 3905–3915. [Google Scholar] [CrossRef]
- Faye, N.; Toguebaye, B.S.; Bouix, G. Ultrastructure and development of Pleistophora senegalensis sp. nov. (Protozoa, Microspora) from the gilt-head sea bream, Sparus aurata L. (Teleost, Sparidae) from the coast of Senegal. J. Fish Dis. 1990, 13, 179–192. [Google Scholar] [CrossRef]
- Kou, G.-H.; Wang, C.-H.; Hung, H.-W.; Jang, Y.-S.; Chou, C.-M.; Lo, C.-F. A cell line (EP-1 cell line) derived from “Beko disease” affected Japanese eel elver (Anguilla japonica) persistently infected with Pleistophora anguillarum. Aquaculture 1995, 132, 161–173. [Google Scholar] [CrossRef]
- Katharios, P.; Kalatzis, P.G.; Kokkari, C.; Pavlidis, M.; Wang, Q. Characterization of a highly virulent Edwardsiella anguillarum strain isolated from Greek aquaculture, and a spontaneously induced prophage therein. Front. Microbiol. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Katharios, P.; Kokkari, C.; Dourala, N.; Smyrli, M. First report of Edwardsiellosis in cage-cultured sharpsnout sea bream, Diplodus puntazzo from the Mediterranean. BMC Vet. Res. 2015, 11. [Google Scholar] [CrossRef]
- Shao, S.; Lai, Q.; Liu, Q.; Wu, H.; Xiao, J.; Shao, Z.; Wang, Q.; Zhang, Y. Phylogenomics characterization of a highly virulent Edwardsiella strain ET080813T encoding two distinct T3SS and three T6SS gene clusters: Propose a novel species as Edwardsiella anguillarum sp. nov. Syst. Appl. Microbiol. 2015, 38, 36–47. [Google Scholar] [CrossRef]
- Dolan, P.T.; Whitfield, Z.J.; Andino, R. Mapping the evolutionary potential of RNA viruses. Cell Host Microbe 2018, 23, 435–446. [Google Scholar] [CrossRef]
- Janssen, K.; Chavanne, H.; Berentsen, P.; Komen, H. Impact of selective breeding on European aquaculture. Aquaculture 2017, 472, 8–16. [Google Scholar] [CrossRef]
- Hammond, J. Animal breeding in relation to nutrition and environmental conditions. Biol. Rev. 1947, 22, 195–213. [Google Scholar] [CrossRef] [PubMed]
- Sae-Lim, P.; Gjerde, B.; Nielsen, H.M.; Mulder, H.; Kause, A. A review of genotype-by-environment interaction and micro-environmental sensitivity in aquaculture species. Rev. Aquac. 2016, 8, 369–393. [Google Scholar] [CrossRef]
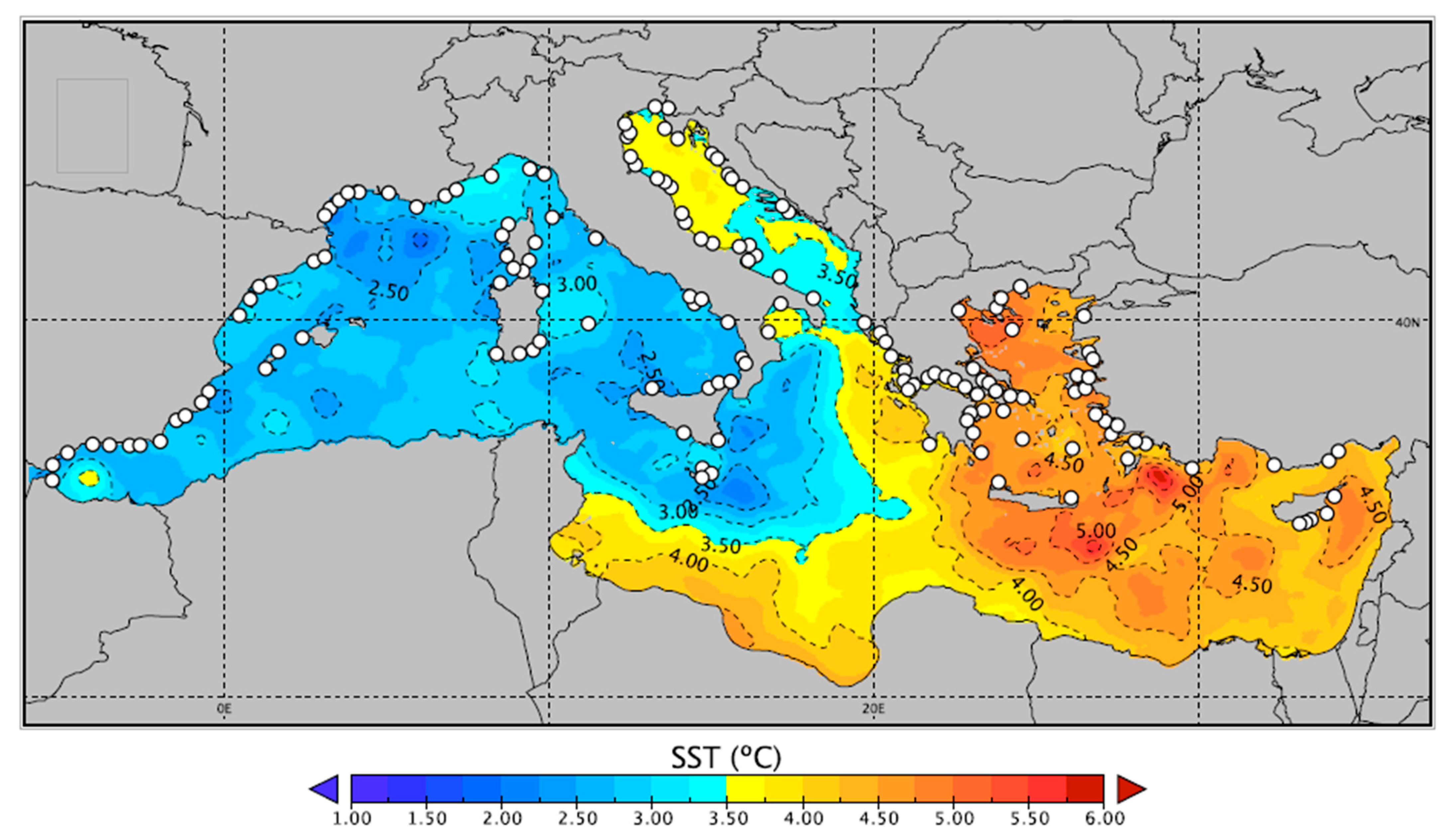
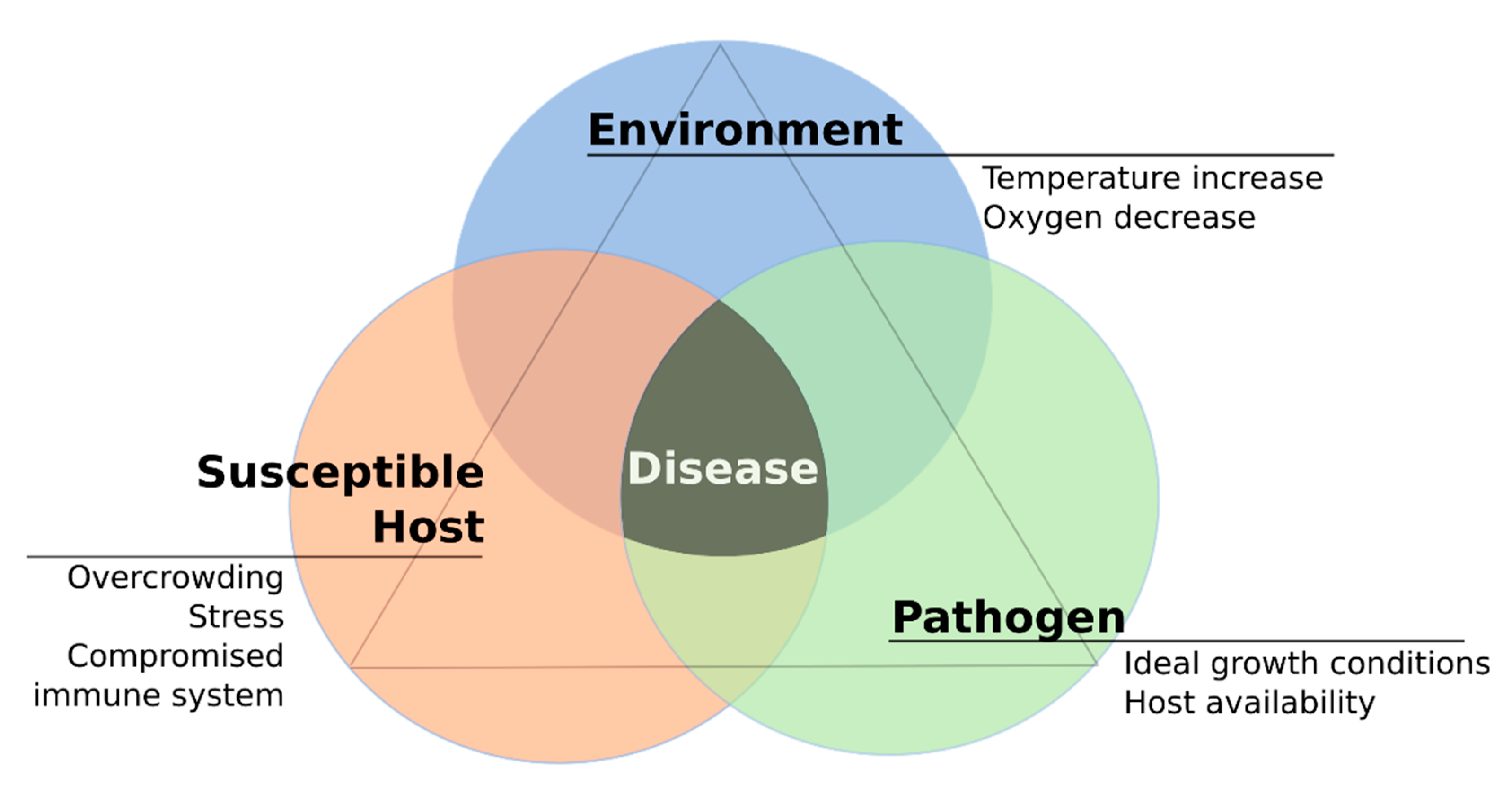
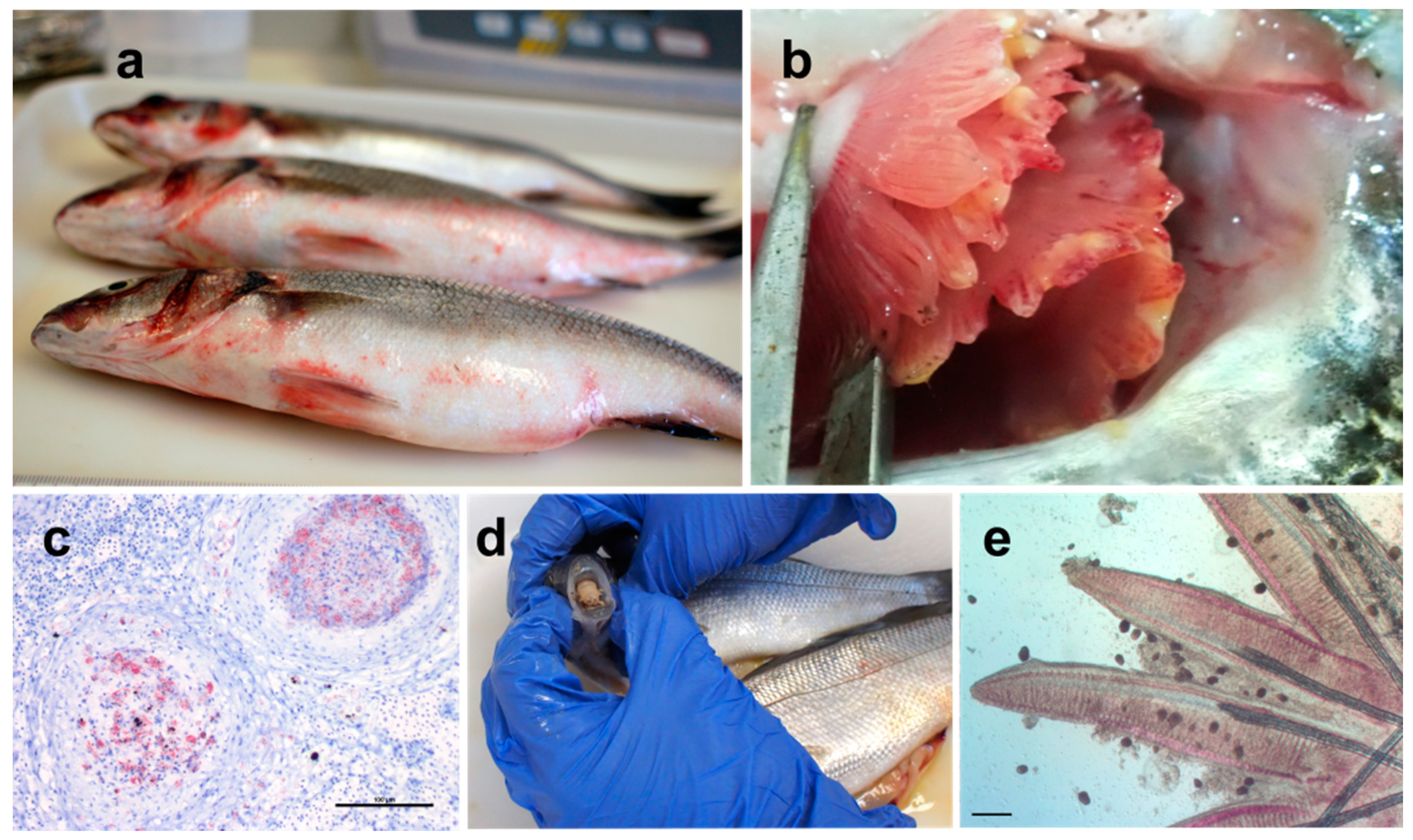

| Fish Host | Tested Parameters | Temperature (°C) | References |
|---|---|---|---|
| gilthead seabream | Thermal stress | ≥22 | [77] |
| mortality | ≥30 | [77] | |
| Haemolytic activity (Optimum) | 20–25 | [116,142] | |
| Immunosuppression | ≤15 | [121] | |
| European seabass | metabolic rate/enzimatic activity (Optimum) | 20–25 | [64,80,81] |
| Oxidative stress | ≥25 | [65] | |
| mortality | ≥32 | [82,83] | |
| Phagocytic activity | ↑ 5–20; ↓ 25–40 | [117] | |
| Antibody response | ↑ 24–30; ↓ 12–18 | [135] | |
| Increasing cortisol levels | ≥25 | [89,90] | |
| meagre | Optimum rearing (juveniles) | 26–30 | [85] |
| Increasing cortisol levels | ≤15 | [88] |
| Group | Pathogen | Disease Name | Host | Zoonotic | Refererence on Temperature or Seasonality | |
|---|---|---|---|---|---|---|
| Bacteria | Aeromonas sp. | Aeromoniasis | 1,2 | [159] | ||
| Aeromonas hydrophila | Aeromoniasis | 2 | yes | [160] | ||
| Aeromonas veronii bv sobria | Aeromoniasis | 2 | [161] | |||
| Ca. Ichthyocystis spp. | Epitheliocystis | 1,2 | [3,162,163,164,165,166] | |||
| Mycobacterium spp. | Mycobacteriosis | 2,3 | yes | [142,167,168,169,170] | ||
| Nocardia spp. | Nocardiosis | 3 | [171] | |||
| Photobacterium damselae subsp. damselae | 1,2,3 | yes | [11,152,153,172,173] | |||
| Photobacterium damselae subsp. piscicida | Pseudoturbeculosis | 1,2 | [174,175,176] | |||
| Pseudomonas anguilliseptica | 1 | [177,178,179,180,181] | ||||
| Tenacibaculum maritimum | Tenacibaculosis (myxobacteriosis) | 1,2 | [182,183,184] | |||
| Vibrio alginolyticus | Vibriosis | 1,2 | [177,185,186,187,188,189,190] | |||
| Vibrio anguillarum | Vibriosis | 1,2,3 | [159,191,192,193] | |||
| Vibrio harveyi | Vibriosis | 1,2 | ||||
| Vibrio splendidus | Vibriosis | 1 | [188] | |||
| Parasites | Ciliate | Cryptocaryon irritans | Marine Ich | 1,2,3 | [194,195,196] | |
| Trichodina spp. | Trochodiniasis | 1,2 | ||||
| Philasterides dicentrarchi (Scuticociliates) | 2 | [197] | ||||
| Dinoflagellate | Amyloodinium ocelatum | Velvet disease | 1,2,3 | [198] | ||
| Flagellate | Ichthyobodo sp. | Costiasis | 1,2,3 | [199] | ||
| Apicomplexa (Myzozoa) | Cryptosporidium molnari | 1 | [200,201] | |||
| Eimeria sp. | 1,2 | [199,202] | ||||
| Goussia sp. | 1 | |||||
| Mesomycetozoa (protozoa) | Ichthyophonus hopheri | Ichthyophoniasis | 1,2 | [203,204] | ||
| Myxosporea (Myxozoa) | Ceratomyxa spp. | 1,2 | [205,206,207,208] | |||
| Enteromyxum leii | Enteromyxosis | 1 | [209,210,211,212,213] | |||
| Kudoa spp. | 1 | |||||
| Kudoa dicentrarchi | 2 | [214,215] | ||||
| Sphaerospora testicularis | 2 | [214,215] | ||||
| Sphaerospora sparis | 1 | [216,217] | ||||
| Monogenean | Diplectanum aequans | 2 | [218,219,220,221,222] | |||
| Diplectanum sciaenae | 3 | |||||
| Encotyllabe spari | 1 | [222] | ||||
| Lamellodiscus spp. | 1 | [166,223,224,225] | ||||
| Lamellodiscus echeneis | 1 | [226,227] | ||||
| Polylabris sp. | 1 | [228] | ||||
| Sparicotyle chrysophrii | 3 | [229,230] | ||||
| Sparicotyle chrysophrii | Sparocotylosis | 1 | [231,232,233,234,235,236,237] | |||
| Nematoda | Anisakis sp. | 2 | yes | [238,239,240] | ||
| Hysterothylacium sp. | 1,2 | yes | [241] | |||
| Crustacea (Copepoda) | Lernanthropus kroyeri | 2 | [242,243] | |||
| Crustacea (Isopoda) | Caligus | 2 | [244,245] | |||
| Ceratothoa oestroides | 1,2,3 | [246,247] | ||||
| Viruses | Lymphocystis | 1,2 | [248,249,250] | |||
| Nodavirus | Viral Encephalopathy and Retinopathy | 1,2 | [251,252] | |||
| Fungi | Microsporidia | Glugea sp. | 1 | [253] | ||
| Microsporidum aurata | 1 | |||||
| Pleistophora sp. | 1 | [254] | ||||
| Pathogen | Tested Temperatures (°C) | Min | Optimum | Max | References |
|---|---|---|---|---|---|
| Aeromonas hydropila | ND | 28 | 37 | Reviewed in [159] | |
| Aeromonas veronii | 4, 12, 22, 30, 37 | 12 | 30 | ND | [161] |
| Mycobacterium spp. | 15, 20, 24, 30, 35, 45 | <15 | 24 | 30 | [142] |
| Photobacterium damselae subsp. damselae | 15, 25 | <15 | 25 | [153] | |
| 25, 37 | 25 | 37 | [152] | ||
| Photobacterium damselae subsp. piscicida | 15 | 22.5 to 30 | 32.5 | Reviewed in [175] | |
| Pseudomonas anguilliseptica | 5 | 5 to 30 | <37 | Reviewed in [177] | |
| Tenacibacilum maritimum | 15 | 30 | 34 | Reviewed in [182] | |
| Vibrio alginolyticus | <12 | 30 | 37 | Reviewed in [177] | |
| Vibrio anguillarum | 5, 10, 15, 20,25, 30 | 5 | 25 | >30 | [191] |
| Temperature | Effects on Host | Effects on Parasites | Effect on Transmission |
|---|---|---|---|
| + | Altered feeding | Faster embryonic development and hatching | Earlier reproduction in spring |
| ++ | Altered behaviour | Faster rates of development and maturation | More generations per year |
| +++ | Weakening of immune defences | Decreased longevity of larvae and adults | Prolonged transmission in the fall |
| ++++ | Reduced host resistance | Increased mortality of all stages | Potential transmission year-round |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cascarano, M.C.; Stavrakidis-Zachou, O.; Mladineo, I.; Thompson, K.D.; Papandroulakis, N.; Katharios, P. Mediterranean Aquaculture in a Changing Climate: Temperature Effects on Pathogens and Diseases of Three Farmed Fish Species. Pathogens 2021, 10, 1205. https://doi.org/10.3390/pathogens10091205
Cascarano MC, Stavrakidis-Zachou O, Mladineo I, Thompson KD, Papandroulakis N, Katharios P. Mediterranean Aquaculture in a Changing Climate: Temperature Effects on Pathogens and Diseases of Three Farmed Fish Species. Pathogens. 2021; 10(9):1205. https://doi.org/10.3390/pathogens10091205
Chicago/Turabian StyleCascarano, Maria Chiara, Orestis Stavrakidis-Zachou, Ivona Mladineo, Kim D. Thompson, Nikos Papandroulakis, and Pantelis Katharios. 2021. "Mediterranean Aquaculture in a Changing Climate: Temperature Effects on Pathogens and Diseases of Three Farmed Fish Species" Pathogens 10, no. 9: 1205. https://doi.org/10.3390/pathogens10091205
APA StyleCascarano, M. C., Stavrakidis-Zachou, O., Mladineo, I., Thompson, K. D., Papandroulakis, N., & Katharios, P. (2021). Mediterranean Aquaculture in a Changing Climate: Temperature Effects on Pathogens and Diseases of Three Farmed Fish Species. Pathogens, 10(9), 1205. https://doi.org/10.3390/pathogens10091205








