A Three-Year Biocrime Sanitary Surveillance on Illegally Imported Companion Animals
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Samples
2.2. Laboratory Testing
2.2.1. Bacteriology
2.2.2. Mycology
2.2.3. Parasitology
2.2.4. Virological Investigations
2.2.5. Rabies Diagnosis and Rabies Antibody Titration
2.3. Statistics
3. Results
3.1. Seizures and Sampled Individuals
3.2. Pathogens Identified from Skin Brush and Skin Swabs
3.3. Pathogens Identified from Pooled Stools
3.4. Pathogens Identified from Animals Dead during the Observation Period
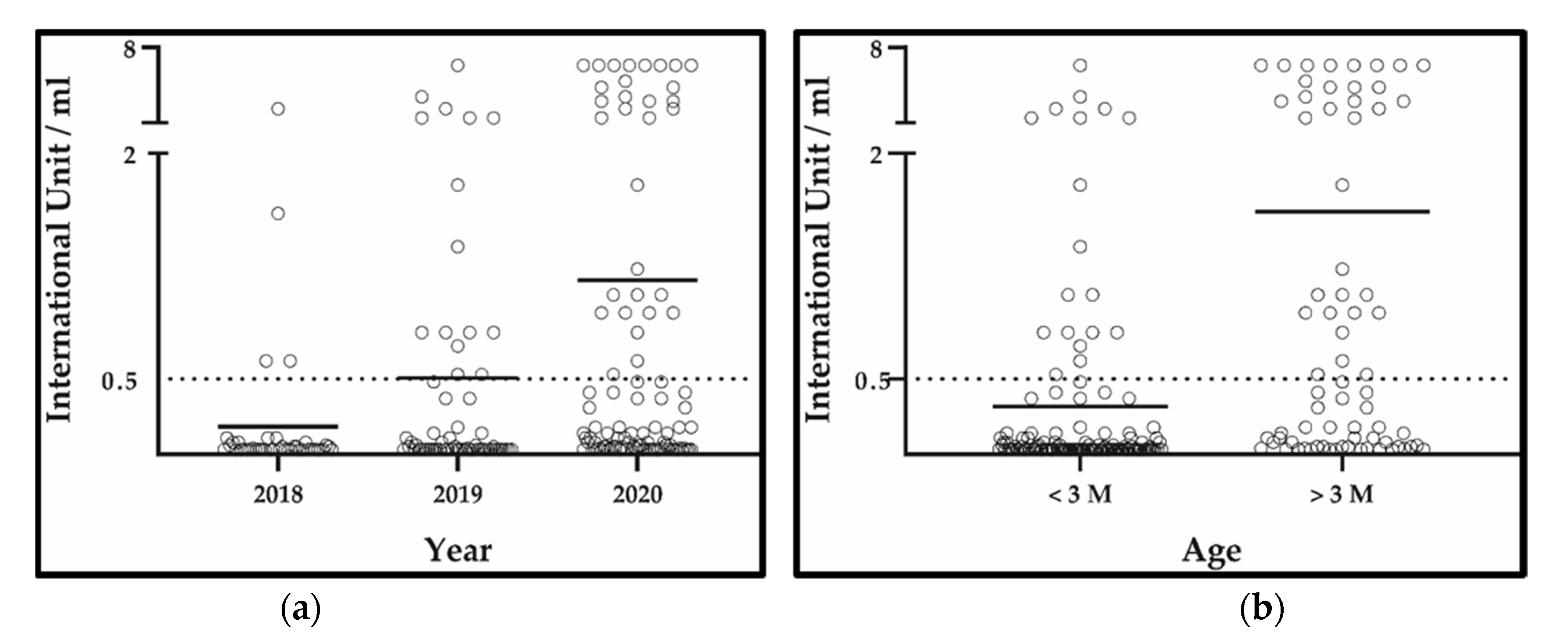
3.5. Rabies Post-Vaccination Assessment (Antibody Titration)
3.6. Salmonella Isolation and Characterization
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Acronym | Meaning |
|---|---|
| CaCoV | Canine Coronavirus |
| CI | Confidence Interval |
| CLSI | Clinical and Laboratory Standard Institute |
| COVID-19 | COronaVIrus Disease 2019 |
| CPV | Canine Parvovirus |
| ESBL | Extended-spectrum-beta-lactamase |
| EU | European Union |
| IU/mL | International Unit/mL |
| MLVA | Multilocus variable-tandem repeat analysis |
| MRSP | Methicillin restistant Staphilococcus pseudintermedius |
| MVC | Minute Virus of Canine |
References
- Zucca, P.; Rossmann, M.C.; Osorio, J.E.; Karem, K.; De Benedictis, P.; Haißl, J.; De Franceschi, P.; Calligaris, E.; Kohlweiß, M.; Meddi, G.; et al. The “Bio-Crime Model” of Cross-Border Cooperation Among Veterinary Public Health, Justice, Law Enforcements, and Customs to Tackle the Illegal Animal Trade/Bio-Terrorism and to Prevent the Spread of Zoonotic Diseases Among Human Population. Front. Vet. Sci. 2020, 7, 855. [Google Scholar] [CrossRef]
- Eurogroup for Animals. The Illegal Pet Trade: Game Over; Eurogroup for Animals: Brussels, Belgium, 2020. [Google Scholar]
- Johnson, N.; Fooks, A. Jet set pets: Examining the zoonosis risk in animal import and travel across the European Union. Vet. Med. Res. Rep. 2015, 6, 17–25. [Google Scholar] [CrossRef] [Green Version]
- Horigan, V.; De Nardi, M.; Simons, R.R.L.; Bertolini, S.; Crescio, M.I.; Estrada-Peña, A.; Léger, A.; Maurella, C.; Ru, G.; Schuppers, M.; et al. Using multi-criteria risk ranking methodology to select case studies for a generic risk assessment framework for exotic disease incursion and spread through Europe. Prev. Vet. Med. 2018, 153, 47–55. [Google Scholar] [CrossRef] [Green Version]
- Simons, R.R.L.; Horigan, V.; Ip, S.; Taylor, R.A.; Crescio, M.I.; Maurella, C.; Mastrantonio, G.; Bertolini, S.; Ru, G.; Cook, C.; et al. A spatial risk assessment model framework for incursion of exotic animal disease into the European Union Member States. Microb. Risk Anal. 2019, 13, 100075. [Google Scholar] [CrossRef]
- Zucca, P.; Rossmann, M.C.; Dodic, M.; Ramma, Y.; Matsushima, T.; Seet, S.; Holtze, S.; Bremini, A.; Fischinger, I.; Morosetti, G.; et al. What Do Adolescents Know About One-Health and Zoonotic Risks? A School-Based Survey in Italy, Austria, Germany, Slovenia, Mauritius, and Japan. Front. Public Health 2021, 9, 658876. [Google Scholar] [CrossRef]
- CLSI. CLSI Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals; CLSI: Wayne, PA, USA, 2018. [Google Scholar]
- Arroyo, L.G.; Rousseau, J.; Willey, B.M.; Low, D.E.; Staempfli, H.; McGeer, A.; Weese, J.S. Use of a selective enrichment broth to recover Clostridium difficile from stool swabs stored under different conditions. J. Clin. Microbiol. 2005, 43, 5341–5343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- OIE—World Organization for Animal Health OIE. Manual of Diagnostic Tests and Vaccines for Terrestiral Animals 2021. Chapt. 3.10.07. Salmonellosis (version adopted in May 2016). Biol. Stand. Comm. World Organ. Anim. Health Paris Fr. 2021, 1, 1738–1741. [Google Scholar]
- ECDC. Laboratory Standard Operating Procedure for MLVA of Salmonella Enterica Serotype Typhimurium; ECDC (European Centre for Disease Prevention and Control): Stockholm, Sweden, 2011.
- ECDC. Laboratory Standard Operating Procedure for Multiple-Locus Variable-Number Tandem Repeat Analysis of Salmonella Enterica Serotype Enteritidis; ECDC (European Centre for Disease Prevention and Control): Stockholm, Sweden, 2016.
- Irinyi, L.; Serena, C.; Garcia-Hermoso, D.; Arabatzis, M.; Desnos-Ollivier, M.; Vu, D.; Cardinali, G.; Arthur, I.; Normand, A.C.; Giraldo, A.; et al. International Society of Human and Animal Mycology (ISHAM)-ITS reference DNA barcoding database—The quality controlled standard tool for routine identification of human and animal pathogenic fungi. Med. Mycol. 2015, 53, 313–337. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture Großbritannien. Manual of Veterinary Parasitological Laoratory Techniques; Her Majesty’s Stationary Office: London, UK, 1986.
- Di Cesare, A.; Castagna, G.; Meloni, S.; Otranto, D.; Traversa, D. Mixed trichuroid infestation in a dog from Italy. Parasites Vectors 2012, 5, 2–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sloss, M.W.; Kemp, R.L.; Zajac, A.M. Veterinary Clinical Parassitology, 6th ed.; Iowa State University Press: Ames, IA, USA, 1994. [Google Scholar]
- Decaro, N.; Elia, G.; Martella, V.; Desario, C.; Campolo, M.; Di Trani, L.; Tarsitano, E.; Tempesta, M.; Buonavoglia, C. A real-time PCR assay for rapid detection and quantitation of canine parvovirus type 2 in the feces of dogs. Vet. Microbiol. 2005, 105, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Elia, G.; Decaro, N.; Martella, V.; Cirone, F.; Lucente, M.S.; Lorusso, E.; Di Trani, L.; Buonavoglia, C. Detection of canine distemper virus in dogs by real-time RT-PCR. J. Virol. Methods 2006, 136, 171–176. [Google Scholar] [CrossRef]
- Toffan, A.; Mazzariol, S.; Povinelli, M.; Trovò, G.; Terregino, C.; Bernardini, D.; Corrò, M. Episodio di grave enterite ad eziologia virale multipla in cuccioli di cane importati dall’Est Europa. In Proceedings of the XIV SIDILV Conference Abstract Book, Sorrento, Italy, 24–26 October 2012; pp. 504–505. [Google Scholar]
- OIE—World Organization for Animal Health OIE. Manual of Diagnostic Tests and Vaccines for Terrestiral Animals 2021. Chapt. 3.01.17. Rabies (infection with rabies virus and other lyssaviruses) (version adopted in May 2018). Biol. Stand. Comm. World Organ. Anim. Health Paris Fr. 2018, 1, 580–585. [Google Scholar]
- De Benedictis, P.; De Battisti, C.; Dacheux, L.; Marciano, S.; Ormelli, S.; Salomoni, A.; Caenazzo, S.T.; Lepelletier, A.; Bourhy, H.; Capua, I.; et al. Lyssavirus detection and typing using pyrosequencing. J. Clin. Microbiol. 2011, 49, 1932–1938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Union Regulation (EU). No 576/2013 of the European Parliament and of the Council of 12 June 2013 on the non-commercial movement of pet animals and repealing Regulation (EC) No 998/2003. Off. J. Eur. Union 2013, 1, 1–26. [Google Scholar]
- Yaoyu, F.; Xiao, L. Zoonotic potential and molecular epidemiology of Giardia species and giardiasis. Clin. Microbiol. Rev. 2011, 24, 110–140. [Google Scholar] [CrossRef] [Green Version]
- Traversa, D.; Di Regalbono, A.F.; Di Cesare, A.; La Torre, F.; Drake, J.; Pietrobelli, M. Environmental contamination by canine geohelminths. Parasites Vectors 2014, 7, 67. [Google Scholar] [CrossRef] [Green Version]
- Deplazes, P.; van Knapen, F.; Schweiger, A.; Overgaauw, P.A.M. Role of pet dogs and cats in the transmission of helminthic zoonoses in Europe, with a focus on echinococcosis and toxocarosis. Vet. Parasitol. 2011, 182, 41–53. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Centre for Disease Prevention and Control. The European Union One Health 2019 Zoonoses Report. EFSA J. 2021, 19, e06406. [Google Scholar] [CrossRef]
- ECDC. Salmonellosis. In ECDC Annual Epidemiological Report for 2017; ECDC: Stockholm, Sweden, 2020. [Google Scholar]
- Hopkins, K.L.; de Pinna, E.; Wain, J. Prevalence of Salmonella enterica serovar 4,[5],12:I:-in England and Wales, 2010. Eurosurveillance 2012, 17, 20275. [Google Scholar] [CrossRef]
- Travis, D.A.; Sriramarao, P.; Cardona, C.; Steer, C.J.; Kennedy, S.; Sreevatsan, S.; Murtaugh, M.P. One Medicine One Science: A framework for exploring challenges at the intersection of animals, humans, and the environment. Ann. N. Y. Acad. Sci. 2014, 1334, 26–44. [Google Scholar] [CrossRef] [Green Version]
- Dickson, A.; Smith, M.; Smith, F.; Park, J.; King, C.; Currie, K.; Langdridge, D.; Davis, M.; Flowers, P. Understanding the relationship between pet owners and their companion animals as a key context for antimicrobial resistance-related behaviours: An interpretative phenomenological analysis. Health Psychol. Behav. Med. 2019, 7, 45–61. [Google Scholar] [CrossRef] [Green Version]
- Gandolfi-Decristophoris, P.; Petrini, O.; Ruggeri-Bernardi, N.; Schelling, E. Extended-spectrum β-lactamase-producing Enterobacteriaceae in healthy companion animals living in nursing homes and in the community. Am. J. Infect. Control 2013, 41, 831–835. [Google Scholar] [CrossRef]
- Guardabassi, L.; Larsen, J.; Weese, J.S.; Butaye, P.; Battisti, A.; Kluytmans, J.; Lloyd, D.H.; Skov, R.L. Public health impact and antimicrobial selection of meticillin-resistant staphylococci in animals. J. Glob. Antimicrob. Resist. 2013, 1, 55–62. [Google Scholar] [CrossRef]
- Rubin, J.E.; Ball, K.R.; Chirino-Trejo, M. Antimicrobial susceptibility of Staphylococcus aureus and Staphylococcus pseudintermedius isolated from various animals. Can. Vet. J. 2011, 52, 162–164. [Google Scholar] [PubMed]
- Paul, N.C.; Moodley, A.; Ghibaudo, G.; Guardabassi, L. Carriage of methicillin-resistant Staphylococcus pseudintermedius in small animal veterinarians: Indirect evidence of zoonotic transmission. Zoonoses Public Health 2011, 58, 533–539. [Google Scholar] [CrossRef] [PubMed]
- van Duijkeren, E.; Kamphuis, M.; van der Mije, I.C.; Laarhoven, L.M.; Duim, B.; Wagenaar, J.A.; Houwers, D.J. Transmission of methicillin-resistant Staphylococcus pseudintermedius between infected dogs and cats and contact pets, humans and the environment in households and veterinary clinics. Vet. Microbiol. 2011, 150, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.; Kadlec, K.; Strommenger, B. Methicillin-resistant Staphylococcus aureus and Staphylococcus pseudintermedius detected in the BfT-GermVet monitoring programme 2004–2006 in Germany. J. Antimicrob. Chemother. 2008, 61, 282–285. [Google Scholar] [CrossRef] [Green Version]
- Kjellman, E.E.; Slettemeås, J.S.; Small, H.; Sunde, M. Methicillin-resistant Staphylococcus pseudintermedius (MRSP) from healthy dogs in Norway-occurrence, genotypes and comparison to clinical MRSP. Microbiologyopen 2015, 4, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Stegmann, R.; Burnens, A.; Maranta, C.A.; Perreten, V. Human infection associated with methicillin-resistant Staphylococcus pseudintermedius ST71. J. Antimicrob. Chemother. 2010, 65, 2047–2048. [Google Scholar] [CrossRef] [PubMed]
- Riegel, P.; Jesel-Morel, L.; Laventie, B.; Boisset, S.; Vandenesch, F.; Prévost, G. Coagulase-positive Staphylococcus pseudintermedius from animals causing human endocarditis. Int. J. Med. Microbiol. 2011, 301, 237–239. [Google Scholar] [CrossRef]
- Servas, V.; Mailles, A.; Neau, D.; Castor, C.; Manetti, A.; Fouquet, E.; Ragnaud, J.-M.; Bourhy, H.; Paty, M.-C.; Melik, N.; et al. An imported case of canine rabies in Aquitaine: Investigation and management of the contacts at risk, August 2004–March 2005. Eurosurveillance 2005, 10, 578. [Google Scholar] [CrossRef]
- Ribadeau-Dumas, F.; Cliquet, F.; Gautret, P.; Robardet, E.; Le Pen, C.; Bourhy, H. Travel-associated rabies in pets and residual rabies risk, Western Europe. Emerg. Infect. Dis. 2016, 22, 1268–1271. [Google Scholar] [CrossRef] [Green Version]
- Klevar, S.; Høgåsen, H.R.; Davidson, R.K.; Hamnes, I.S.; Berndtsson, L.T.; Lund, A. Cross-border transport of rescue dogs may spread rabies in Europe. Vet. Rec. 2015, 176, 672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rota Nodari, E.; Alonso, S.; Mancin, M.; De Nardi, M.; Hudson-Cooke, S.; Veggiato, C.; Cattoli, G.; De Benedictis, P. Rabies vaccination: Higher failure rates in imported dogs than in those vaccinated in Italy. Zoonoses Public Health 2016, 64, 146–155. [Google Scholar] [CrossRef] [PubMed]
- De Benedictis, P.; Veggiato, C.; Selli, L.; Dundon, W.G.; Capua, I. Frontier control sampling indicates insufficient compliance to international rabies regulation. In Proceedings of the Med Vet Net 4th Annual Scientific Meeting Abstract Book, St Malo, France, 11–14 June 2008; p. 29. [Google Scholar]
- Fèvre, E.M.; Bronsvoort, B.M.D.C.; Hamilton, K.A.; Cleaveland, S. Animal movements and the spread of infectious diseases. Trends Microbiol. 2006, 14, 125–131. [Google Scholar] [CrossRef]
- Tietjen, S.; Kaufhold, B.; Mueller, T.; Freuling, C.M. Case report: Illegal puppy trade and transport, Germany. Rabies Bull. Eur. 2011, 35, 8–10. [Google Scholar]
- Altman, K.D.; Kelman, M.; Ward, M.P. Are vaccine strain, type or administration protocol risk factors for canine parvovirus vaccine failure? Vet. Microbiol. 2017, 210, 8–16. [Google Scholar] [CrossRef]
- Behdenna, A.; Lembo, T.; Calatayud, O.; Cleaveland, S.; Halliday, J.E.B.; Packer, C.; Lankester, F.; Hampson, K.; Craft, M.E.; Czupryna, A.; et al. Transmission ecology of canine parvovirus in a multi-host, multi-pathogen system. Proc. Biol. Sci. 2019, 286, 20182772. [Google Scholar] [CrossRef] [Green Version]
- Weckworth, J.K.; Davis, B.W.; Dubovi, E.; Fountain-Jones, N.; Packer, C.; Cleaveland, S.; Craft, M.E.; Eblate, E.; Schwartz, M.; Mills, L.S.; et al. Cross-species transmission and evolutionary dynamics of canine distemper virus during a spillover in African lions of Serengeti National Park. Mol. Ecol. 2020, 29, 4308–4321. [Google Scholar] [CrossRef]
- Bianco, A.; Zecchin, B.; Fusaro, A.; Schivo, A.; Ormelli, S.; Bregoli, M.; Citterio, C.V.; Obber, F.; Dellamaria, D.; Trevisiol, K.; et al. Two waves of canine distemper virus showing different spatio-temporal dynamics in Alpine wildlife (2006–2018). Infect. Genet. Evol. 2020, 84, 104359. [Google Scholar] [CrossRef]
- Chang, Y.C.; Lin, Z.Y.; Lin, Y.X.; Lin, K.H.; Chan, F.T.; Hsiao, S.T.; Liao, J.W.; Chiou, H.Y. Canine Parvovirus Infections in Taiwanese Pangolins (Manis pentadactyla pentadactyla). Vet. Pathol. 2021. [Google Scholar] [CrossRef]
- Canuti, M.; Todd, M.; Monteiro, P.; Van Osch, K.; Weir, R.; Schwantje, H.; Britton, A.P.; Lang, A.S. Ecology and infection dynamics of multi-host amdoparvoviral and protoparvoviral carnivore pathogens. Pathogens 2020, 9, 124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Benedictis, P.; Mutinelli, F.; Veggiato, C.; Capua, I.; Squecco, G.; Coassin, R.; Ferri, G. Rabies in a vaccinated dog in Italy. Vet. Rec. 2009, 165, 216. [Google Scholar] [CrossRef] [PubMed]
- Campalto, M.; Carrino, M.; Tassoni, L.; Rizzo, G.; Rossmann, M.C.; Cocchi, M.; De Benedictis, P.; Beato, M.S. Divergent minute virus of canines strains identified in illegally imported puppies in Italy. Arch. Virol. 2020, 165, 2945–2951. [Google Scholar] [CrossRef] [PubMed]
- Eurogroup for Animals. Communication on an EU Strategy to tackle organised crime (2021–2025): The Need to Include Illegal Animal Trade; Eurogroup for Animals: Brussels, Belgium, 2021. [Google Scholar]
| Target | Positive/Analysed | Prevalence% (CI%) |
|---|---|---|
| Giardia spp. | 123/256 | 48.05 (42.00–54.15) |
| Cryptosporidium spp. | 9/250 | 3.60 (1.91–6.70) |
| Salmonella | 11/226 | 4.87 (2.74–8.50) |
| Canine Parvovirus | 186/227 | 81.94 (76.41–86.40) |
| Canine Coronavirus | 78/189 | 41.27 (34.49–48.39) |
| Minute Virus of Canine | 77/232 | 33.19 (27.45–39.48) |
| Canine Adenovirus type 1 | 24/188 | 12.77 (8.73–18.29) |
| Canine Adenovirus type 2 | 10/153 | 6.54 (3.59–11.61) |
| Target | Positive/Analysed | Prevalence% (CI%) |
|---|---|---|
| Giardia spp. | 6/69 | 8.70 (4.05–17.70) |
| Cryptosporidium spp. | 0/69 | 0.00 (0.00–3.77) |
| Salmonella | 4/75 | 5.33 (2.09–12.92) |
| Canine Parvovirus | 68/73 | 93.15 (84.95–97.04) |
| Canine Coronavirus | 9/71 | 12.68 (6.81–22.37) |
| Minute Virus of Canine | 13/75 | 17.33 (10.42–27.43) |
| Canine Adenovirus type 1 | 6/74 | 8.11 (3.46–16.89) |
| Canine Adenovirus type 2 | 1/74 | 1.35 (0.00–7.27) |
| Year | Origin | Host Species | Characterisation | MLVA Profile |
|---|---|---|---|---|
| 2018 | Feaces | Dog | S. Infantis | n.p. |
| 2018 | Feaces | Dog | S. Infantis | n.p. |
| 2018 | Feaces | Cat | S. Enteritidis | 2-10-7-3-2 |
| 2018 | Feaces | Cat | monophasic S. Typhimurium | 3-12-10-NA-0211 |
| 2020 | Feaces | Dog | S. Enteritidis | 2-11-7-3-2 |
| 2020 | Intestines | Dog | S. Enteritidis | 3-9-4-4-1 |
| 2020 | Intestines | Dog | S. Enteritidis | 3-9-4-4-1 |
| 2020 | Intestines | Dog | S. Enteritidis | 3-9-4-4-1 |
| 2019 | Feaces | Cat | monophasic S. Typhimurium | 3-12-10-NA-0211 |
| 2019 | Feaces | Cat | S. Enteritidis | 2-10-7-3-2 |
| 2019 | Feaces | Cat | S. Enteritidis | 2-10-7-3-2 |
| 2019 | Feaces | Cat | S. Enteritidis | 2-10-7-3-2 |
| 2020 | Feaces | Dog | S. Corvallis | n.p. |
| 2020 | Feaces | Dog | S. Debou | n.p. |
| 2020 | Feaces | Dog | S. Hadar | n.p. |
| 2020 | Feaces | Dog | S. Bredeney | n.p. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cocchi, M.; Danesi, P.; De Zan, G.; Leati, M.; Gagliazzo, L.; Ruggeri, M.; Palei, M.; Bremini, A.; Rossmann, M.-C.; Lippert-Petscharnig, M.; et al. A Three-Year Biocrime Sanitary Surveillance on Illegally Imported Companion Animals. Pathogens 2021, 10, 1047. https://doi.org/10.3390/pathogens10081047
Cocchi M, Danesi P, De Zan G, Leati M, Gagliazzo L, Ruggeri M, Palei M, Bremini A, Rossmann M-C, Lippert-Petscharnig M, et al. A Three-Year Biocrime Sanitary Surveillance on Illegally Imported Companion Animals. Pathogens. 2021; 10(8):1047. https://doi.org/10.3390/pathogens10081047
Chicago/Turabian StyleCocchi, Monia, Patrizia Danesi, Gabrita De Zan, Marta Leati, Laura Gagliazzo, Margherita Ruggeri, Manlio Palei, Alessandro Bremini, Marie-Christin Rossmann, Melanie Lippert-Petscharnig, and et al. 2021. "A Three-Year Biocrime Sanitary Surveillance on Illegally Imported Companion Animals" Pathogens 10, no. 8: 1047. https://doi.org/10.3390/pathogens10081047
APA StyleCocchi, M., Danesi, P., De Zan, G., Leati, M., Gagliazzo, L., Ruggeri, M., Palei, M., Bremini, A., Rossmann, M.-C., Lippert-Petscharnig, M., Mansfeld, M.-D., Deotto, S., Leardini, S., Gobbo, F., Zucca, P., & De Benedictis, P. (2021). A Three-Year Biocrime Sanitary Surveillance on Illegally Imported Companion Animals. Pathogens, 10(8), 1047. https://doi.org/10.3390/pathogens10081047








