The Carboxy Terminal Region on Spike Protein of Porcine Epidemic Diarrhea Virus (PEDV) Is Important for Evaluating Neutralizing Activity
Abstract
1. Introduction
2. Results
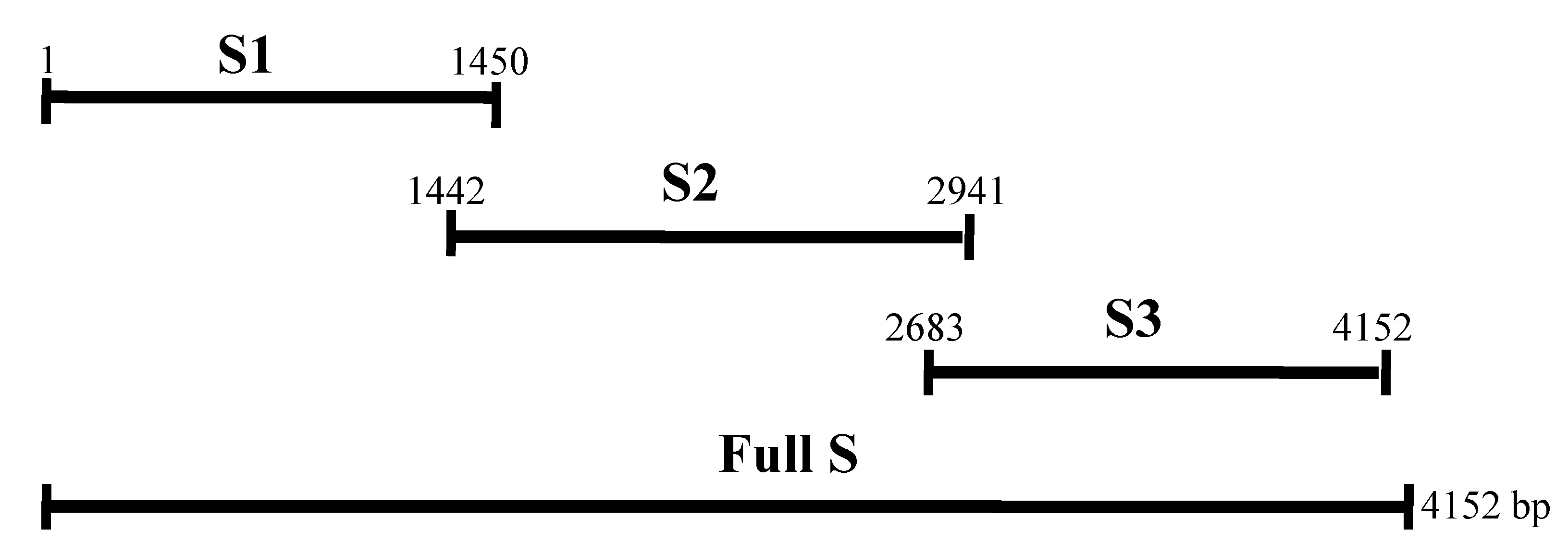
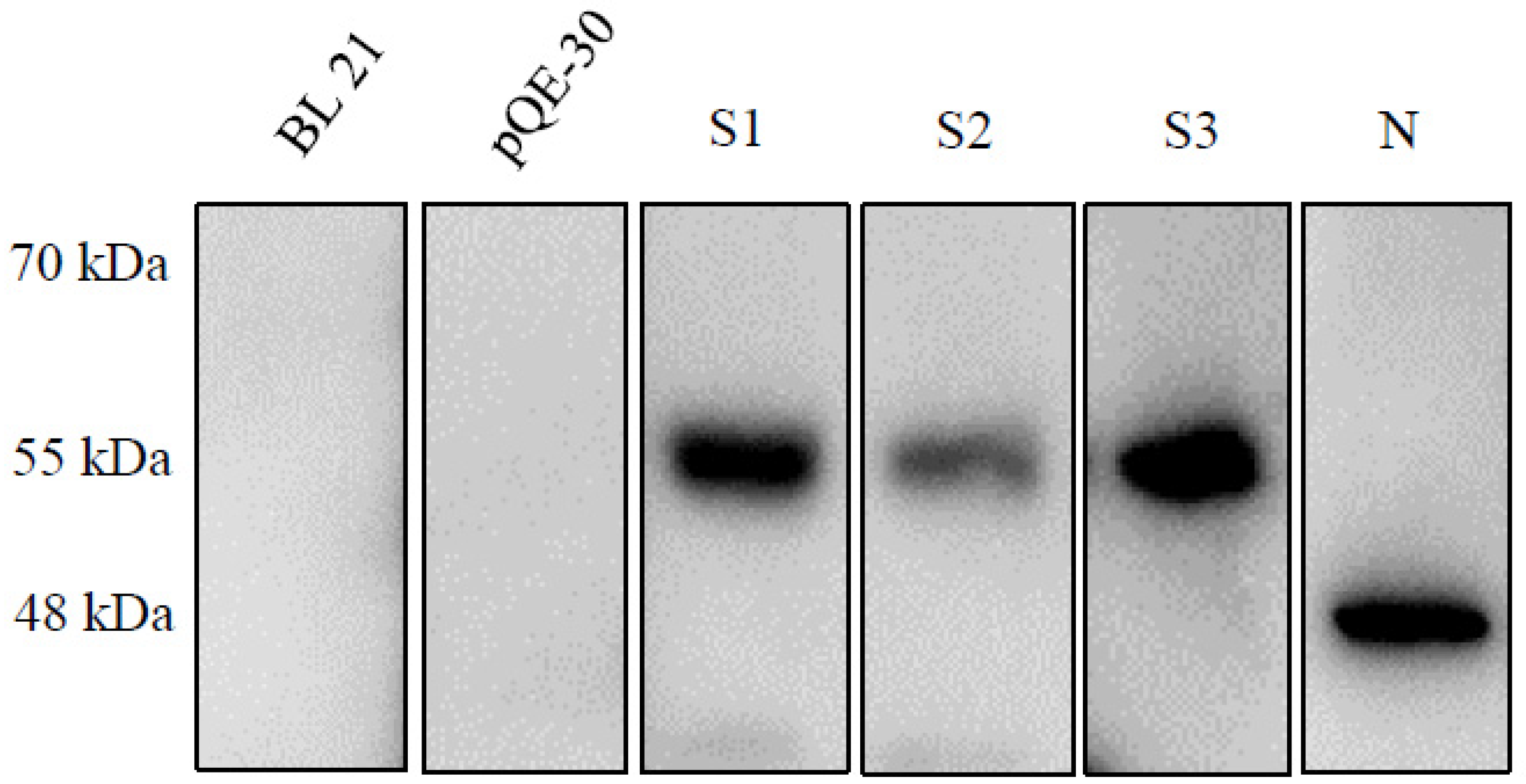
2.1. Expression of the Recombinant Spike and N Proteins
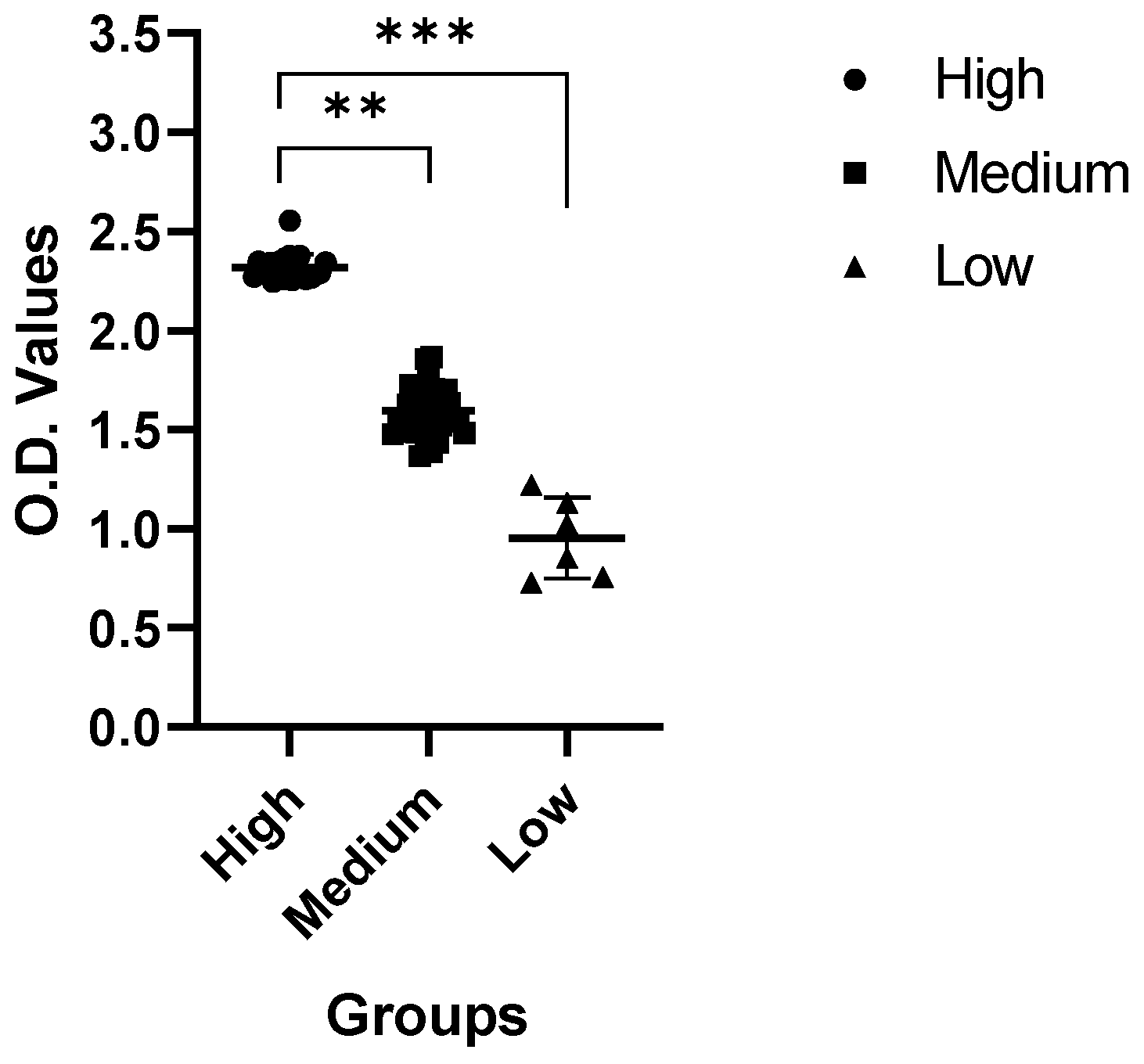
2.2. Serum Neutralization Test (SNT)
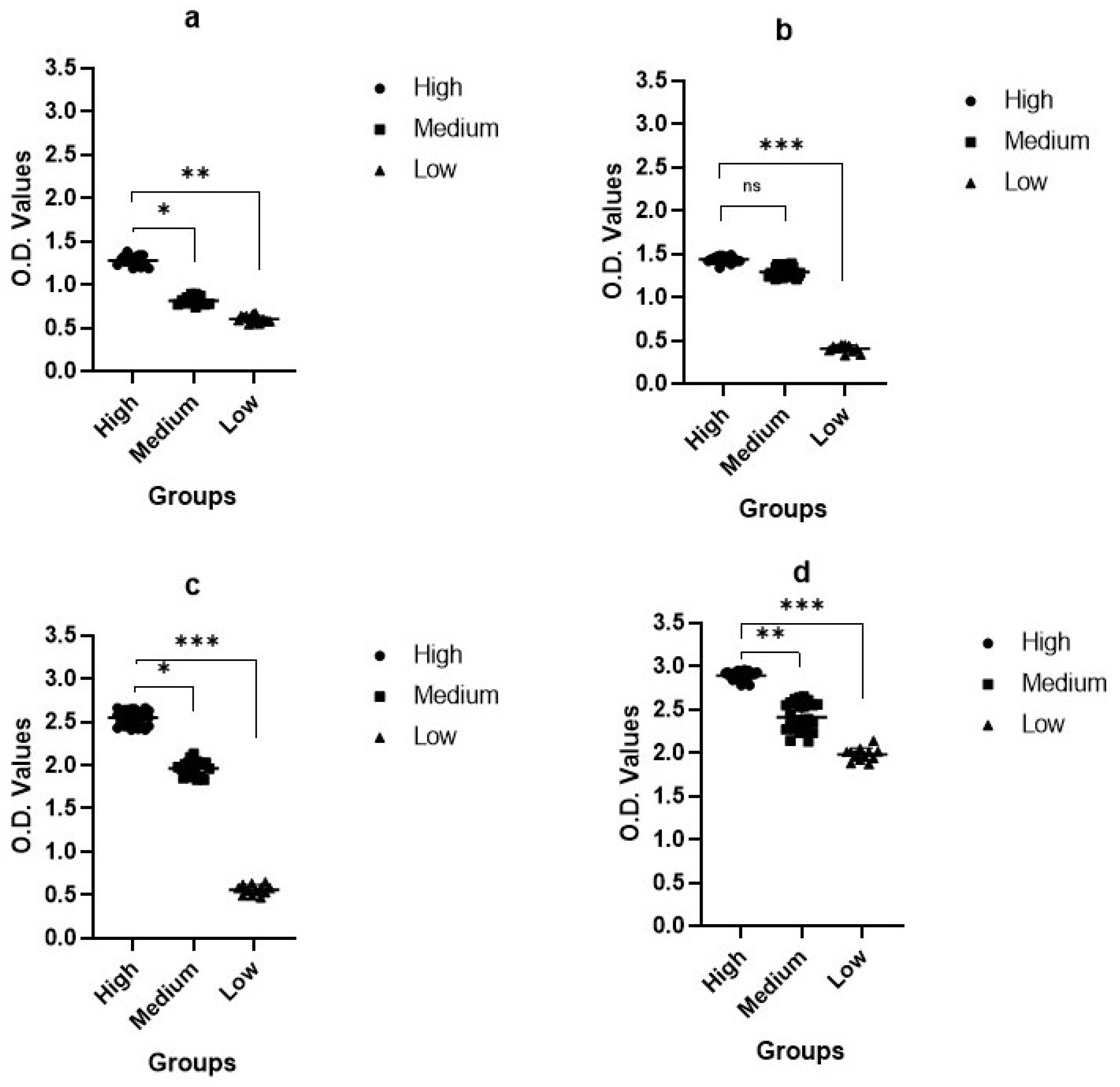
2.3. Enzyme-Linked Immunosorbent Assay (ELISA)
2.4. Protein ELISA (pELISA)
3. Discussion
4. Materials and Methods
4.1. Sampling
4.2. Viruses
4.3. RT-PCR for Amplification of Genes
4.4. Prokaryotic Expression
4.5. SDS-PAGE and Immunoblot Analysis
4.6. Serum Neutralization Test (SNT)
4.7. Enzyme-Linked Immunosorbent Assay (ELISA)
4.8. Protein ELISA (pELISA)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Debouck, P.; Pensaert, M. Experimental infection of pigs with a new porcine enteric coronavirus, CV 777. Am. J. Vet. Res. 1980, 41, 219–223. [Google Scholar] [PubMed]
- Wang, Q.; Vkasiva, A.; Kenney, S.; Saif, L. Emerging and re-emerging coronaviruses in pigs. Curr. Opin. Microbiol. 2019, 34, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Okada, K.; Ohshima, K. An outbreak of swine diarrhea of a new-type associated with coronavirus-like particles in Japan. Nihon Juigaku Zasshi 1983, 45, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Lee, C. Porcine epidemic diarrhea virus: An emerging and re-emerging epizootic swine virus. Virol. J. 2015, 12, 193. [Google Scholar] [CrossRef]
- Ding, Z.; Fang, L.; Jing, H.; Zeng, S.; Wang, D.; Liu, L.; Zhang, H.; Luo, R.; Chen, H.; Xiao, S. Porcine Epidemic Diarrhea Virus Nucleocapsid Protein Antagonizes Beta Interferon Production by Sequestering the Interaction between IRF3 and TBK1. J. Virol. 2014, 88, 8936–8945. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Cruz, D.J.; Shin, H.J. Clathrin- and serine proteases-dependent uptake of porcine epidemic diarrhea virus into Vero cells. Virus Res. 2014, 191, 21–29. [Google Scholar] [CrossRef]
- Oh, J.S.; Lee, K.W.; Choi, H.W.; Lee, C.H. Immunogenicity and protective efficacy of recombinant S1 domain of the porcine epidemic diarrhea virus spike protein. Arch. Virol. 2014, 159, 2977–2987. [Google Scholar] [CrossRef]
- Li, C.H.; Li, W.T.; Esesarte, L.D.; Guo, H.B.; Elzen, P.V.D.; Aarts, E.; Born, E.V.D.; Rottier, P.J.M.; Bosch, B.J. Cell Attachment Domains of the Porcine Epidemic Diarrhea Virus Spike Protein Are Key Targets of Neutralizing Antibodies. J. Virol. 2017, 91, e00273-17. [Google Scholar] [CrossRef]
- Pijpers, A.; van Nieuwstadt, A.P.; Terpstra, C.; Verheijden, J.H. Porcine epidemic diarrhoea virus as a cause of persistent diarrhoea in a herd of breeding and finishing pigs. Vet. Rec. 1993, 132, 129–131. [Google Scholar] [CrossRef]
- Hofmann, M.; Wyler, R. Propagation of the virus of porcine epidemic diarrhea in cell culture. J. Clin. Microbiol. 1988, 26, 2235–2239. [Google Scholar] [CrossRef]
- Callebaut, P.; Debouck, P.; Pensaert, M. Enzyme-linked immunosorbent assay for the detection of the coronavirus-like agent and its antibodies in pigs with porcine epidemic diarrhea. Vet. Microbiol. 1982, 7, 295–306. [Google Scholar] [CrossRef]
- Carvajal, A.; Lanza, I.; Diego, R.; Rubio, P.; Carmenes, P. Evaluation of a blocking ELISA using monoclonal antibodies for the detection of porcine epidemic diarrhea virus and its antibodies. J. Vet. Diagn. Investig. 1995, 7, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Knuchel, M.; Ackermann, M.; Muller, H.K.; Kihm, U. An ELISA for detection of antibodies against porcine epidemic diarrhoea virus (PEDV) based on the specific solubility of the viral surface glycoprotein. Vet. Microbiol. 1992, 32, 117–134. [Google Scholar] [CrossRef]
- Kweon, C.H.; Lee, J.G.; Han, M.G.; Kang, Y.B. Rapid diagnosis of porcine epidemic diarrhea virus infection by polymerase chain reaction. J. Vet. Med. Sci. 1997, 59, 231–232. [Google Scholar] [CrossRef] [PubMed]
- van Nieuwstadt, A.P.; Zetstra, T. Use of two enzyme-linked immunosorbent assays to monitor antibody responses in swine with experimentally induced infection with porcine epidemic diarrhea virus. Am. J. Vet. Res. 1991, 52, 1044–1050. [Google Scholar] [PubMed]
- Sueyoshi, M.; Tsuda, T.; Yamazaki, K.; Yoshida, K.; Nakazawa, M.; Sato, K.; Minami, T.; Iwashita, K.; Watanabe, M.; Suzuki, Y.; et al. An immunohistochemical investigation of porcine epidemic diarrhoea. J. Comp. Pathol. 1995, 113, 59–67. [Google Scholar] [CrossRef]
- Song, D.-S.; Kang, B.-K.; Lee, S.-S.; Yang, J.-S.; Moon, H.-J.; Oh, J.-S.; Ha, G.-W.; Jang, Y.-S.; Park, B.-K. An internal control in the quantitative RT-PCR assay for quantitation of porcine epidemic diarrhea virus shedding in pigs. J. Virol. Methods 2006, 133, 27–33. [Google Scholar] [CrossRef]
- To, T.L.; Ward, L.A.; Yuan, L.; Saif, L.J. Serum and intestinal isotype antibody responses and correlates of protective immunity to human rotavirus in a gnotobiotic pig model of disease. J. Gen. Virol. 1998, 79 Pt 11, 2661–2672. [Google Scholar] [CrossRef]
- Lin, H.; Zhou, H.; Gao, L.; Li, B.; He, K.; Fa, H. Development and application of an indirect ELISA for the detection of antibodies to porcine epidemic diarrhea virus based on a recombinant spike protein. BMC Vet. Res. 2018, 14, 243. [Google Scholar] [CrossRef]
- Lirola, L.G.G.; Zhang, J.; Avil, J.A.C.; Chen, Q.; Magtoto, R.; Poonsuk, K.; Baum, D.H.; Piñeyro, P.; Zimmerman, J. Reactivity of Porcine Epidemic Diarrhea Virus Structural Proteins to Antibodies against Porcine Enteric Coronaviruses: Diagnostic Implications. J. Clin. Microbiol. 2017, 55, 1426–1436. [Google Scholar] [CrossRef]
- Egberink, H.F.; Ederveen, J.; Callebaut, P.; Horzinek, M.C. Characterization of the structural proteins of porcine epizootic diarrhea virus, strain CV777. Am. J. Vet. Res. 1988, 49, 1320–1324. [Google Scholar]
- Duarte, M.; Laude, H. Sequence of the spike protein of the porcine epidemic diarrhoea virus. J. Gen. Virol. 1994, 75 Pt 5, 1195–1200. [Google Scholar] [CrossRef]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.; Rottier, P.J. The coronavirus spike protein is a class I virus fusion protein: Structural and functional characterization of the fusion core complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Han, J.H.; Kwon, H.M. Partial sequence of the spike glycoprotein gene of transmissible gastroenteritis viruses isolated in Korea. Vet. Microbiol. 2003, 94, 195–206. [Google Scholar] [CrossRef]
- Cruz, D.J.; Kim, C.J.; Shin, H.J. Phage-displayed peptides having antigenic similarities with porcine epidemic diarrhea virus (PEDV) neutralizing epitopes. Virology 2006, 354, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.H.; Bae, J.L.; Kang, T.J.; Kim, J.; Chung, G.H.; Lim, C.W.; Laude, H.; Yang, M.S.; Jang, Y.S. Identification of the epitope region capable of inducing neutralizing antibodies against the porcine epidemic diarrhea virus. Mol. Cells 2002, 14, 295–299. [Google Scholar] [PubMed]
- Kang, T.J.; Han, S.C.; Yang, M.S.; Jang, Y.S. Expression of synthetic neutralizing epitope of porcine epidemic diarrhea virus fused with synthetic B subunit of Escherichia coli heat-labile enterotoxin in tobacco plants. Protein Expr. Purif. 2006, 46, 16–22. [Google Scholar] [CrossRef]
- Godeke, G.J.; de Haan, C.A.; Rossen, J.W.; Vennema, H.; Rottier, P.J. Assembly of spikes into coronavirus particles is mediated by the carboxy-terminal domain of the spike protein. J. Virol. 2000, 74, 1566–1571. [Google Scholar] [CrossRef] [PubMed]
- Ye, R.; Montalto-Morrison, C.; Masters, P.S. Genetic analysis of determinants for spike glycoprotein assembly into murine coronavirus virions: Distinct roles for charge-rich and cysteine-rich regions of the endodomain. J. Virol. 2004, 78, 9904–9917. [Google Scholar] [CrossRef] [PubMed]
- Okda, F.A.; Lawson, S.; Singrey, A.; Nelson, J.; Hain, K.S.; Joshi, L.R.; Christopher-Hennings, J.; Nelson, E.A.; Diel, D.G. The S2 glycoprotein subunit of porcine epidemic diarrhea virus contains immunodominant neutralizing epitopes. Virology 2017, 509, 185–194. [Google Scholar] [CrossRef]
- Buratti, E.; Tisminetzky, S.G.; D’Agaro, P.; Baralle, F.E. A neutralizing monoclonal antibody previously mapped exclusively on human immunodeficiency virus type 1 gp41 recognizes an epitope in p17 sharing the core sequence IEEE. J. Virol. 1997, 71, 2457–2462. [Google Scholar] [CrossRef] [PubMed]
- Boucher, C.A.; Krone, W.J.; Goudsmit, J.; Meloen, R.H.; Naylor, P.H.; Goldstein, A.L.; Sun, D.K.; Sarin, P.S. Immune response and epitope mapping of a candidate HIV-1 p17 vaccine HGP30. J. Clin. Lab. Anal. 1990, 4, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Cruz, D.J.; Kim, C.J.; Shin, H. The GPRLQPY motif located at the carboxy-terminal of the spike protein induces antibodies that neutralize Porcine epidemic diarrhea virus. Virus Res. 2008, 132, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]

| Proteins (Gene Size) | Primer | Sequence |
|---|---|---|
| Spike S1 (1450 bp) | PED S1-S | 5′ GGA TCC ATG AGG TCT TTA ATT TAC TTC 3′ |
| PED S1-AS | 5′ GGT ACC ATG GGG TAAA AAC CAT CGT CAA GGT CAA AAG 3′ | |
| Spike S2 (1499 bp) | PED S2-S | 5′ GGA TCC TTA CCC CAT CTC TTC TAG AAA CCT 3′ |
| PED S2-AS | 5′ GGT ACC TAC AAA CAT ATG TAG CAC AAT CAA CAA CAC AC 3′ | |
| Spike S3 (1469 bp) | PED S3-S | 5′ GGA TCC AAG ACT TGC TTT TAA ATA 3′ |
| PED S3-AS | 5′ GGT ACC TTA GTG ATG GTG ATG GTG ATG CTG CAC GTG AAC CT3′ | |
| Nucleocapsid (1326 bp) | PED N-S | 5′ GGA TTC GCA TCT GTC AGC TTT CAG 3′ |
| PED N-AS | 5′ GGT ACC TTA ATT TCC TGT ATC GAA 3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, K.-J.; Kim, D.-H.; Hong, E.-J.; Shin, H.-J. The Carboxy Terminal Region on Spike Protein of Porcine Epidemic Diarrhea Virus (PEDV) Is Important for Evaluating Neutralizing Activity. Pathogens 2021, 10, 683. https://doi.org/10.3390/pathogens10060683
Kang K-J, Kim D-H, Hong E-J, Shin H-J. The Carboxy Terminal Region on Spike Protein of Porcine Epidemic Diarrhea Virus (PEDV) Is Important for Evaluating Neutralizing Activity. Pathogens. 2021; 10(6):683. https://doi.org/10.3390/pathogens10060683
Chicago/Turabian StyleKang, Ki-Jong, Dong-Hwan Kim, Eui-Ju Hong, and Hyun-Jin Shin. 2021. "The Carboxy Terminal Region on Spike Protein of Porcine Epidemic Diarrhea Virus (PEDV) Is Important for Evaluating Neutralizing Activity" Pathogens 10, no. 6: 683. https://doi.org/10.3390/pathogens10060683
APA StyleKang, K.-J., Kim, D.-H., Hong, E.-J., & Shin, H.-J. (2021). The Carboxy Terminal Region on Spike Protein of Porcine Epidemic Diarrhea Virus (PEDV) Is Important for Evaluating Neutralizing Activity. Pathogens, 10(6), 683. https://doi.org/10.3390/pathogens10060683







