Silver Nanoparticles Conjugated with Contact Lens Solutions May Reduce the Risk of Acanthamoeba Keratitis
Abstract
1. Introduction
2. Results
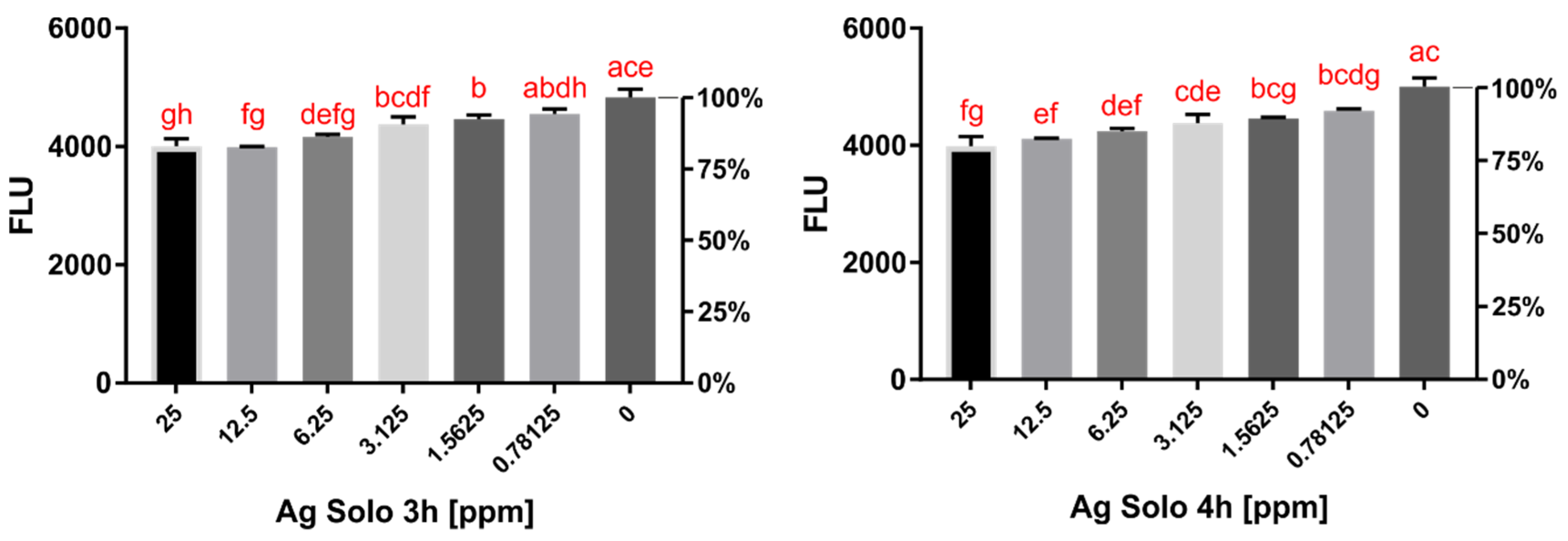
2.1. Activity Assay Results
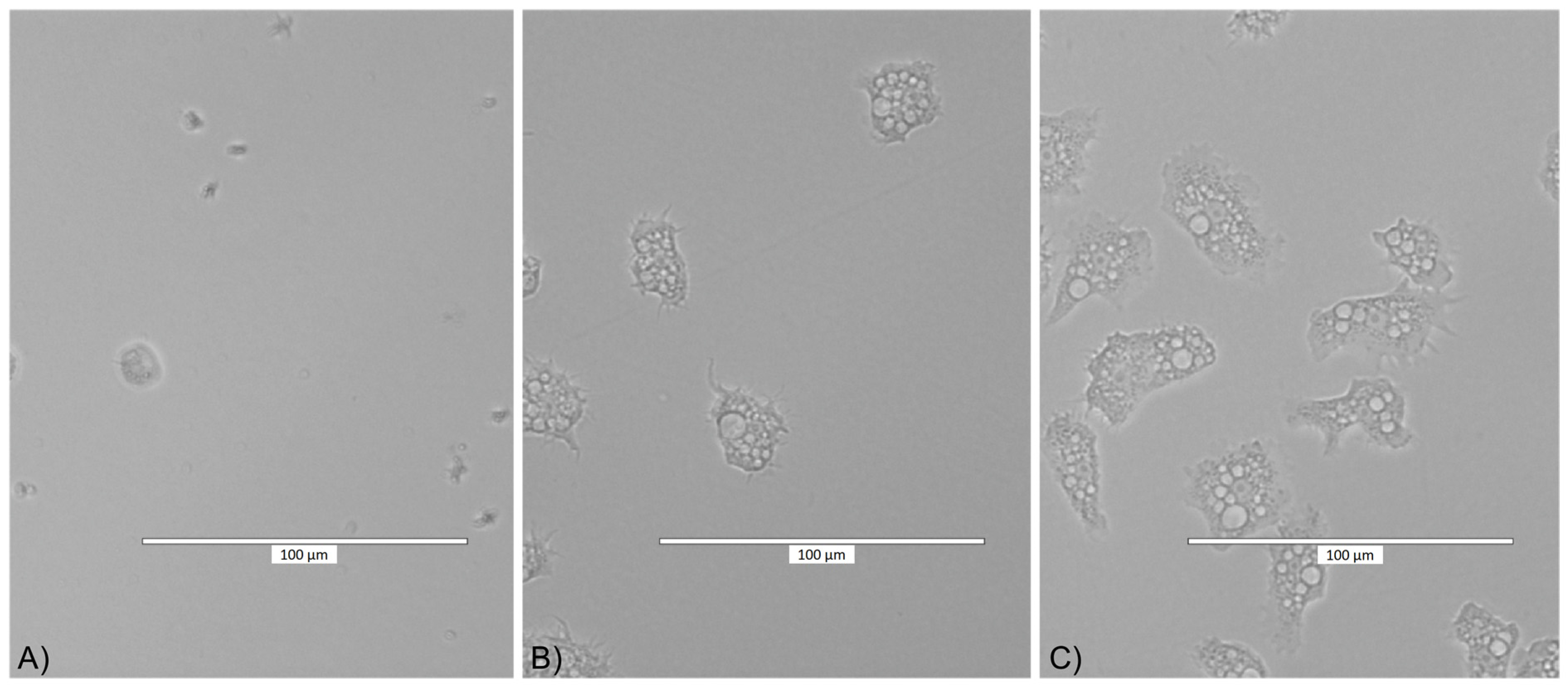
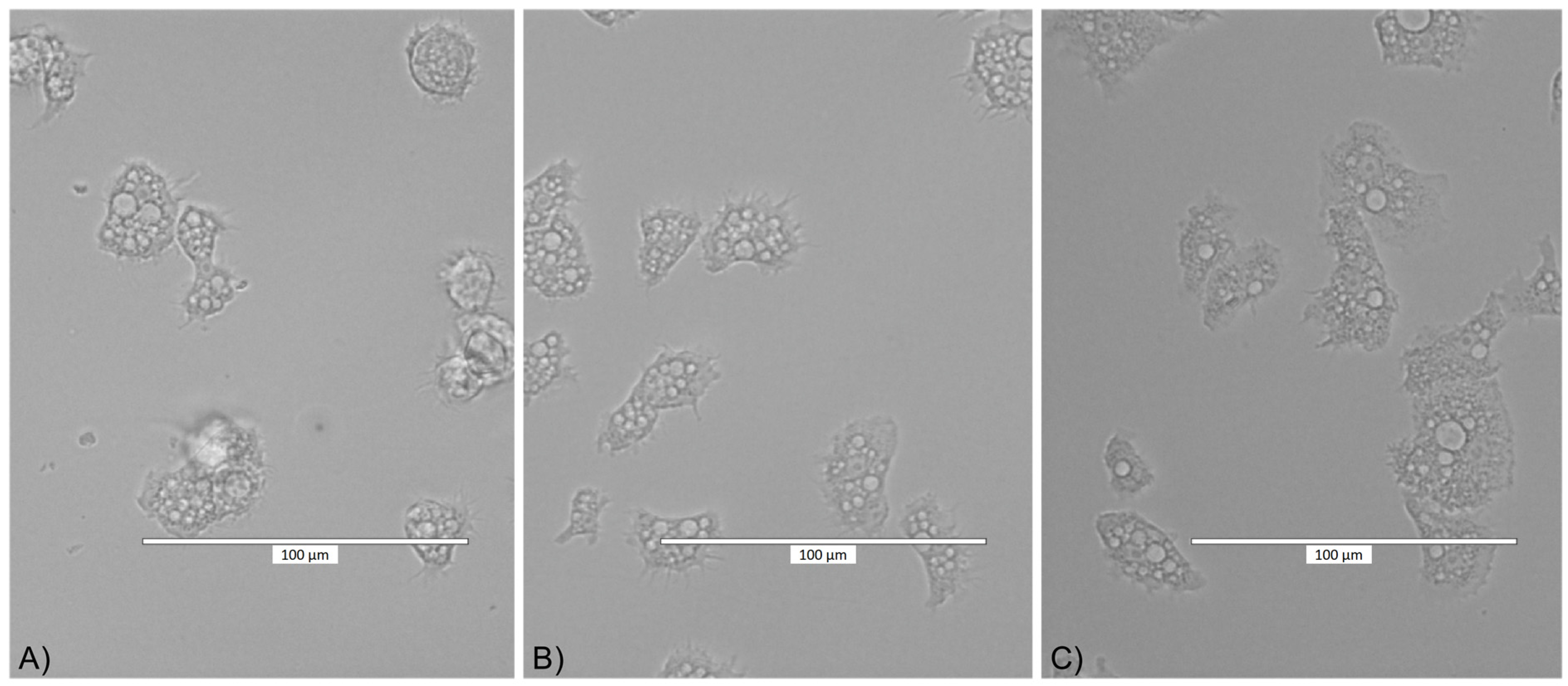
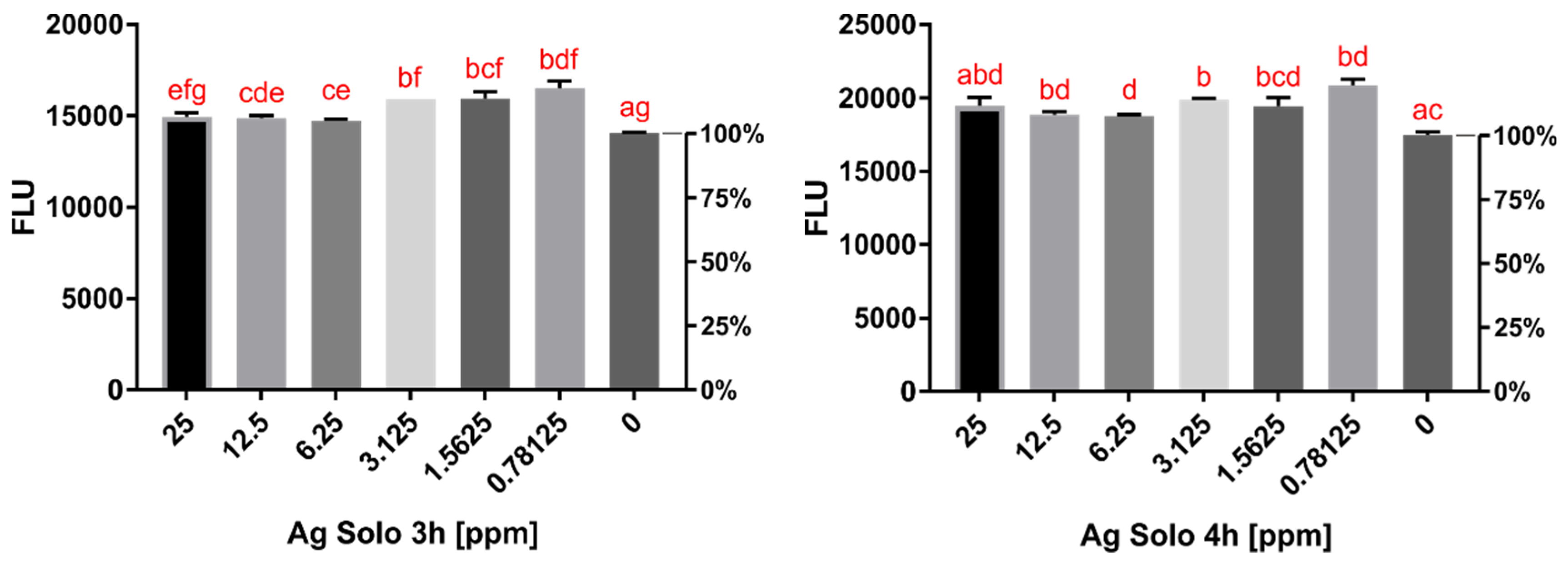
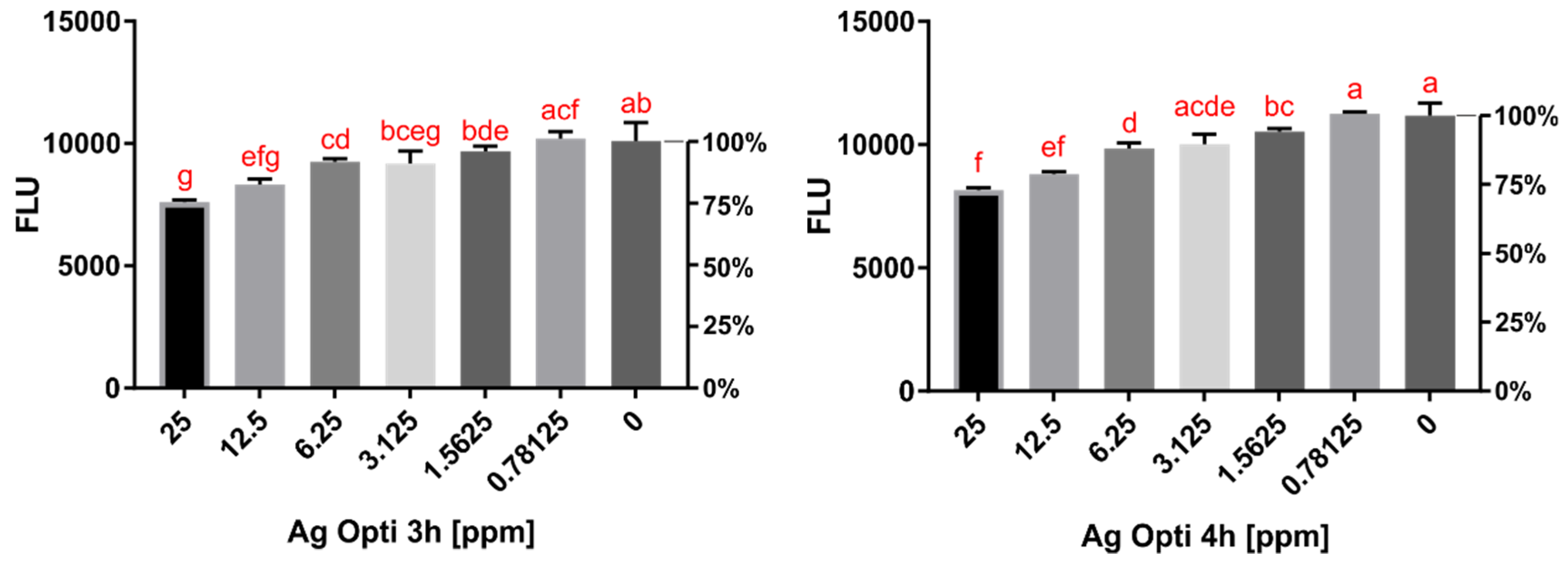
2.2. Cytotoxicity Assay Results
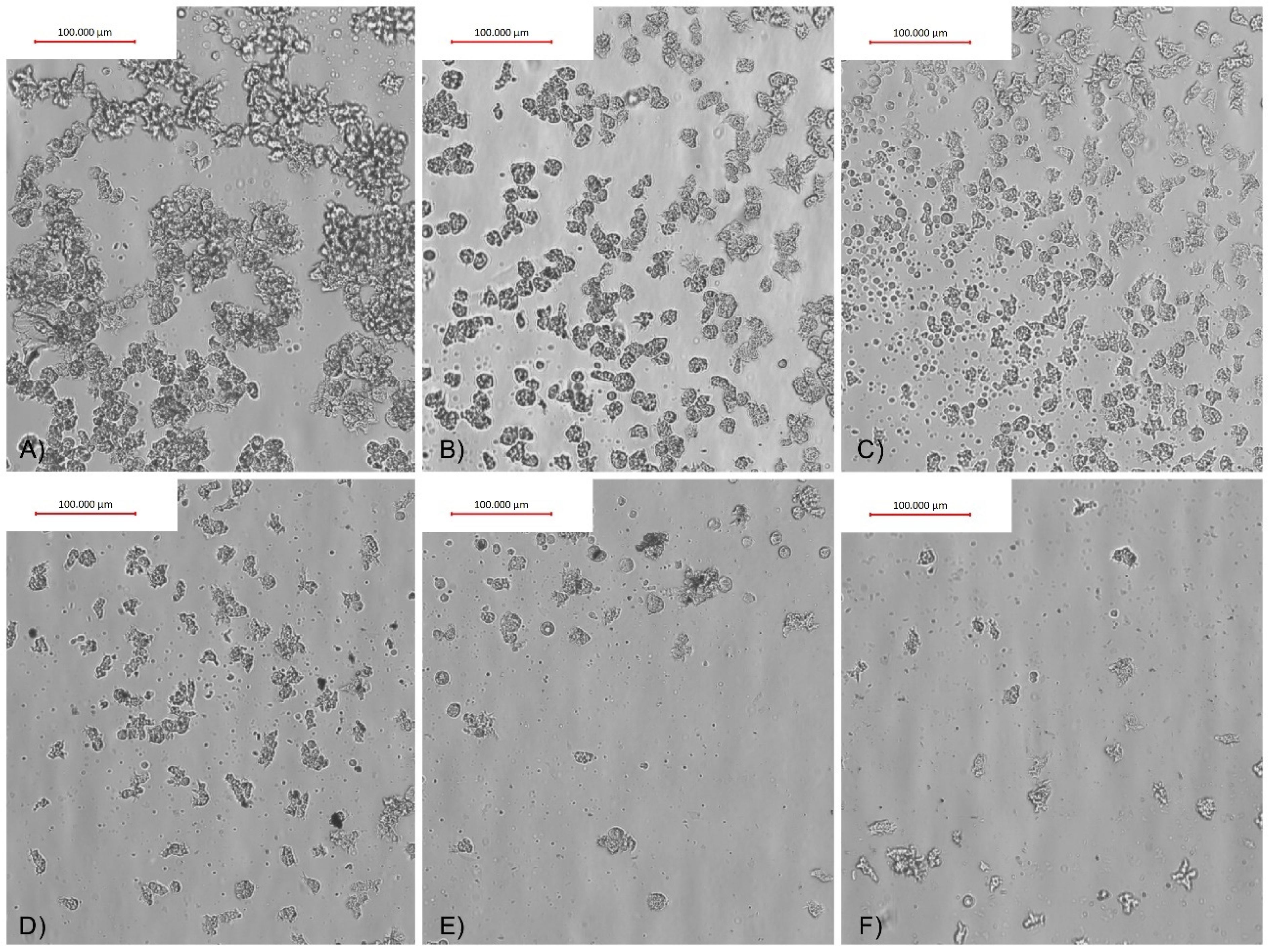
2.3. Adhesion Assay Results
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Acanthamoeba and Macrophage Cultivation
5.2. Nanoparticles
5.3. Contact Lens Solutions
5.4. Activity Assays
5.5. Cytotoxicity Assays
5.6. Adhesion Assays
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dart, J.K.; Saw, V.P.; Kilvington, S. Acanthamoeba keratitis: Diagnosis and treatment update 2009. Am. J. Ophthalmol. 2009, 148, 487–499. [Google Scholar] [CrossRef]
- Lorenzo-Morales, J.; Martin-Navarro, C.M.; Lopez-Arencibia, A.; Arnalich-Montiel, F.; Pinero, J.E.; Valladares, B. Acanthamoeba keratitis: An emerging disease gathering importance worldwide? Trends Parasitol. 2013, 29, 181–187. [Google Scholar] [CrossRef]
- Lorenzo-Morales, J.; Khan, N.A.; Walochnik, J. An update on Acanthamoeba keratitis: Diagnosis, pathogenesis and treatment. Parasite 2015, 22, 10. [Google Scholar] [CrossRef]
- Maycock, N.J.; Jayaswal, R. Update on Acanthamoeba Keratitis: Diagnosis, Treatment, and Outcomes. Cornea 2016, 35, 713–720. [Google Scholar] [CrossRef]
- Padzik, M.; Hendiger, E.B.; Zochowska, A.; Szczepaniak, J.; Baltaza, W.; Pietruczuk-Padzik, A.; Oledzka, G.; Chomicz, L. Evaluation of in vitro effect of selected contact lens solutions conjugated with nanoparticles in terms of preventive approach to public health risk generated by Acanthamoeba strains. Ann. Agric. Environ. Med. 2019, 26, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Kot, K.; Lanocha-Arendarczyk, N.A.; Kosik-Bogacka, D.I. Amoebas from the genus Acanthamoeba and their pathogenic properties. Ann. Parasitol. 2018, 64, 299–308. [Google Scholar] [CrossRef]
- Panjwani, N. Pathogenesis of Acanthamoeba keratitis. Ocul. Surf. 2010, 8, 70–79. [Google Scholar] [CrossRef]
- Bunsuwansakul, C.; Mahboob, T.; Hounkong, K.; Laohaprapanon, S.; Chitapornpan, S.; Jawjit, S.; Yasiri, A.; Barusrux, S.; Bunluepuech, K.; Sawangjaroen, N.; et al. Acanthamoeba in Southeast Asia—Overview and Challenges. Korean J. Parasitol. 2019, 57, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Niederkorn, J.Y. The biology of Acanthamoeba keratitis. Exp. Eye Res. 2021, 202, 108365. [Google Scholar] [CrossRef]
- Kumar, R.; Lloyd, D. Recent advances in the treatment of Acanthamoeba keratitis. Clin. Infect. Dis. 2002, 35, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Clarke, B.; Sinha, A.; Parmar, D.N.; Sykakis, E. Advances in the diagnosis and treatment of Acanthamoeba keratitis. J. Ophthalmol. 2012, 2012, 484892. [Google Scholar] [CrossRef] [PubMed]
- Kokot, J.; Dobrowolski, D.; Lyssek-Boron, A.; Milka, M.; Smedowski, A.; Wojcik, L.; Wowra, B.; Wyligala, E. New approach to diagnosis and treatment of Acanthamoeba keratitis—Systematic review of literature. Klin Oczna 2012, 114, 311–316. [Google Scholar]
- Anwar, A.; Numan, A.; Siddiqui, R.; Khalid, M.; Khan, N.A. Cobalt nanoparticles as novel nanotherapeutics against Acanthamoeba castellanii. Parasites Vectors 2019, 12, 280–282. [Google Scholar] [CrossRef]
- Hendiger, E.B.; Padzik, M.; Sifaoui, I.; Reyes-Batlle, M.; Lopez-Arencibia, A.; Rizo-Liendo, A.; Bethencourt-Estrella, C.J.; Nicolas-Hernandez, D.S.; Chiboub, O.; Rodriguez-Exposito, R.L.; et al. Silver Nanoparticles as a Novel Potential Preventive Agent against Acanthamoeba Keratitis. Pathogens 2020, 9, 350. [Google Scholar] [CrossRef]
- Sharma, G.; Kalra, S.K.; Tejan, N.; Ghoshal, U. Nanoparticles based therapeutic efficacy against Acanthamoeba: Updates and future prospect. Exp. Parasitol. 2020, 218, 108008. [Google Scholar] [CrossRef] [PubMed]
- Balashanmugam, P.; Kalaichelvan, P.T. Biosynthesis characterization of silver nanoparticles using Cassia roxburghii DC. aqueous extract, and coated on cotton cloth for effective antibacterial activity. Int. J. Nanomed. 2015, 10 (Suppl. 1), 87–97. [Google Scholar] [CrossRef]
- Baldi, F.; Daniele, S.; Gallo, M.; Paganelli, S.; Battistel, D.; Piccolo, O.; Faleri, C.; Puglia, A.M.; Gallo, G. Polysaccharide-based silver nanoparticles synthesized by Klebsiella oxytoca DSM 29614 cause DNA fragmentation in E. coli cells. Biometals 2016, 29, 321–331. [Google Scholar] [CrossRef]
- Emmanuel, R.; Palanisamy, S.; Chen, S.M.; Chelladurai, K.; Padmavathy, S.; Saravanan, M.; Prakash, P.; Ajmal Ali, M.; Al-Hemaid, F.M. Antimicrobial efficacy of green synthesized drug blended silver nanoparticles against dental caries and periodontal disease causing microorganisms. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 56, 374–379. [Google Scholar] [CrossRef]
- Shahverdi, A.R.; Fakhimi, A.; Shahverdi, H.R.; Minaian, S. Synthesis and effect of silver nanoparticles on the antibacterial activity of different antibiotics against Staphylococcus aureus and Escherichia coli. Nanomedicine 2007, 3, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Chen, X.; Song, W.; Kong, Z.; Li, P.; Liu, Y. Contribution of silver ions to the inhibition of infectivity of Schistosoma japonicum cercariae caused by silver nanoparticles. Parasitology 2013, 140, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Machado, L.F.; Sanfelice, R.A.; Bosqui, L.R.; Assolini, J.P.; Scandorieiro, S.; Navarro, I.T.; Depieri Cataneo, A.H.; Wowk, P.F.; Nakazato, G.; Bordignon, J.; et al. Biogenic silver nanoparticles reduce adherence, infection, and proliferation of toxoplasma gondii RH strain in HeLa cells without inflammatory mediators induction. Exp. Parasitol. 2020, 211, 107853. [Google Scholar] [CrossRef]
- Rahimi, M.T.; Ahmadpour, E.; Rahimi Esboei, B.; Spotin, A.; Kohansal Koshki, M.H.; Alizadeh, A.; Honary, S.; Barabadi, H.; Ali Mohammadi, M. Scolicidal activity of biosynthesized silver nanoparticles against Echinococcus granulosus protoscolices. Int. J. Surg. 2015, 19, 128–133. [Google Scholar] [CrossRef]
- Saad, H.A.; Soliman, M.I.; Azzam, A.M.; Mostafa, B. Antiparasitic Activity of Silver and Copper Oxide Nanoparticles Against Entamoeba Histolytica and Cryptosporidium Parvum Cysts. J. Egypt. Soc. Parasitol. 2015, 45, 593–602. [Google Scholar] [CrossRef]
- Said, D.E.; Elsamad, L.M.; Gohar, Y.M. Validity of silver, chitosan, and curcumin nanoparticles as anti-Giardia agents. Parasitol. Res. 2012, 111, 545–554. [Google Scholar] [CrossRef]
- Rahman, K.; Khan, S.U.; Fahad, S.; Chang, M.X.; Abbas, A.; Khan, W.U.; Rahman, L.; Haq, Z.U.; Nabi, G.; Khan, D. Nano-biotechnology: A new approach to treat and prevent malaria. Int. J. Nanomed. 2019, 14, 1401–1410. [Google Scholar] [CrossRef]
- Ullah, I.; Cosar, G.; Abamor, E.S.; Bagirova, M.; Shinwari, Z.K.; Allahverdiyev, A.M. Comparative study on the antileishmanial activities of chemically and biologically synthesized silver nanoparticles (AgNPs). 3 Biotech 2018, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Anwar, A.; Siddiqui, R.; Hussain, M.A.; Ahmed, D.; Shah, M.R.; Khan, N.A. Silver nanoparticle conjugation affects antiacanthamoebic activities of amphotericin B, nystatin, and fluconazole. Parasitol. Res. 2018, 117, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Borase, H.P.; Patil, C.D.; Sauter, I.P.; Rott, M.B.; Patil, S.V. Amoebicidal activity of phytosynthesized silver nanoparticles and their in vitro cytotoxicity to human cells. FEMS Microbiol. Lett. 2013, 345, 127–131. [Google Scholar] [CrossRef]
- Padzik, M.; Hendiger, E.B.; Chomicz, L.; Grodzik, M.; Szmidt, M.; Grobelny, J.; Lorenzo-Morales, J. Tannic acid-modified silver nanoparticles as a novel therapeutic agent against Acanthamoeba. Parasitol. Res. 2018, 117, 3519–3525. [Google Scholar] [CrossRef]
- Hendiger, E.B.; Padzik, M.; Zochowska, A.; Baltaza, W.; Oledzka, G.; Zyskowska, D.; Bluszcz, J.; Jarzynka, S.; Chomicz, L.; Grodzik, M.; et al. Tannic acid-modified silver nanoparticles enhance the anti-Acanthamoeba activity of three multipurpose contact lens solutions without increasing their cytotoxicity. Parasites Vectors 2020, 13, 1–8. [Google Scholar] [CrossRef]
- Anwar, A.; Siddiqui, R.; Raza Shah, M.; Khan, N.A. Antidiabetic Drugs and Their Nanoconjugates Repurposed as Novel Antimicrobial Agents against Acanthamoeba castellanii. J. Microbiol. Biotechnol. 2019, 29, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Grün, A.; Scheid, P.; Hauröder, B.; Emmerling, C.; Manz, W. Assessment of the effect of silver nanoparticles on the relevant soil protozoan genus Acanthamoeba. J. Plant Nutr. Soil Sci. 2017, 180, 602–613. [Google Scholar] [CrossRef]
- Anwar, A.; Soomaroo, A.; Anwar, A.; Siddiqui, R.; Khan, N.A. Metformin-coated silver nanoparticles exhibit anti-acanthamoebic activities against both trophozoite and cyst stages. Exp. Parasitol. 2020, 215, 107915. [Google Scholar] [CrossRef] [PubMed]
- Anwar, A.; Mungroo, M.R.; Anwar, A.; Sullivan, W.J.; Khan, N.A.; Siddiqui, R. Repositioning of Guanabenz in Conjugation with Gold and Silver Nanoparticles against Pathogenic Amoebae Acanthamoeba castellanii and Naegleria fowleri. ACS Infect. Dis. 2019, 5, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Anwar, A.; Yi, Y.P.; Fatima, I.; Khan, K.M.; Siddiqui, R.; Khan, N.A.; Anwar, A. Antiamoebic activity of synthetic tetrazoles against Acanthamoeba castellanii belonging to T4 genotype and effects of conjugation with silver nanoparticles. Parasitol. Res. 2020, 119, 1943–1954. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Lee, J.E.; Lee, D.I.; Yu, H.S. Adhesion of Acanthamoeba on Cosmetic Contact Lenses. J. Korean Med. Sci. 2018, 33, e26. [Google Scholar] [CrossRef]
- Omana-Molina, M.A.; Gonzalez-Robles, A.; Salazar-Villatoro, L.; Bernal-Escobar, A.; Duran-Diaz, A.; Mendez-Cruz, A.R.; Martinez-Palomo, A. Silicone hydrogel contact lenses surface promote Acanthamoeba castellanii trophozoites adherence: Qualitative and quantitative analysis. Eye Contact Lens 2014, 40, 132–139. [Google Scholar] [CrossRef]
- Bakay, B.B.; Polat, Z.A. In Vitro Evaluation of Adhesion of Two Acanthamoeba Strains to Cosmetic Contact Lenses. Eye Contact Lens 2018, 44 (Suppl. 2), S241–S246. [Google Scholar] [CrossRef]
- Lee, G.H.; Lee, J.E.; Park, M.K.; Yu, H.S. Adhesion of Acanthamoeba on Silicone Hydrogel Contact Lenses. Cornea 2016, 35, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Lee, D.I.; Park, S.H.; Yu, H.S.; Lee, J.E.; Lee, J.S. Effect of Multipurpose Solution Combined with Autophagy Inhibitors on Adhesion of Acanthamoeba trophozoites to Silicone Hydrogel Contact Lenses. Cornea 2017, 36, 1538–1543. [Google Scholar] [CrossRef] [PubMed]
- Mitsuwan, W.; Sangkanu, S.; Romyasamit, C.; Kaewjai, C.; Jimoh, T.O.; de Lourdes Pereira, M.; Siyadatpanah, A.; Kayesth, S.; Nawaz, M.; Rahmatullah, M.; et al. Curcuma longa rhizome extract and Curcumin reduce the adhesion of Acanthamoeba triangularis trophozoites and cysts in polystyrene plastic surface and contact lens. Int. J. Parasitol. Drugs Drug. Resist. 2020, 14, 218–229. [Google Scholar] [CrossRef]


| FDA 1 | FDA 2 | FDA 3 | FDA 4 | |
|---|---|---|---|---|
| SCA | 8.8 ± 4.2 | no activity | no activity | 63.5 ± 14.0 |
| SCA + 25 ppm AgNPs | 90.0 ± 2.2 | no activity | 51.6 ± 9.1 | 95.2 ± 1.5 |
| SCA + 12.5 ppm AgNPs | 74.4 ± 0.8 | no activity | 50.9 ± 6.2 | 93.1 ± 2.3 |
| SCA + 6.25 ppm AgNPs | 36.9 ± 0.4 | no activity | 21.1 ± 20.5 | 89.0 ± 2.4 |
| SCA + 3.125 ppm AgNPs | unreliable data | no activity | 37.9 ± 5.0 | 80.3 ± 6.1 |
| O-F | 13.2 ± 23.2 | unreliable data | no activity | no activity |
| O-F + 25 ppm AgNPs | 94.7 ± 3.7 | unreliable data | 95.3 ± 0.5 | 94.7 ± 0.7 |
| O-F + 12.5 ppm AgNPs | 91.9 ± 2.1 | unreliable data | 69.8 ± 2.9 | 82.2 ± 0.3 |
| O-F + 6.25 ppm AgNPs | 76.7 ± 3.3 | unreliable data | 55.6 ± 13.5 | 62.7 ± 0.7 |
| O-F + 3.125 ppm AgNPs | 56.4 ± 0.4 | unreliable data | 6.1 ± 55.8 | 55.7 ± 19.0 |
| Manufacturer | Solution | Ingredients | Minimum Disinfection Time (h) |
|---|---|---|---|
| Menicon | Solo Care Aqua (SCA) | Polyhexanide 0.0001%, Hydrolock (dexpanthenol, sorbitol), sodium phosphate, tromethamine, poloxamer 407, disodium edetate | 4 |
| Alcon | Opti Free (O-F) | TearGlyde (Tetronic 1304, nonanoyl ethylenediaminetriacetic acid), Polyquad (polyquaternium-1) 0.001%, Aldox (myristamidopropyl dimethylamine) 0.0005% | 6 |
| Bausch + Lomb | ReNu Multiplus (ReNu) | Hydranate (hydroxyalkylphosphonate) 0.03%, boric acid, edetate disodium, poloxamine 1%, sodium borate, sodium chloride, preserved with Dymed (polyaminopropyl biguanide 0.0001%) | 4 |
| B-Lens | B-Lens (BL) | Polyheksametylene biguanide (PHMB) 0.0001%, Pluronic F 1%, Panthenol 1.5%. | 4 |
| Best View | Best View (BV) | PHMB 0.0001%, Ethylenediaminetetraacetic acid (EDTA), 0.01%, Pluronic | 6 |
| Polymer | FDA Group | Water Content | Ionic | Silicon Content | Manufacturer |
|---|---|---|---|---|---|
| Senofilcon A | 1 | 38% | no | yes | ACUVUE oasys |
| Nelfilcon A | 2 | 69% | no | no | Dailies |
| Balafilcon A | 3 | 36% | yes | yes | Baush + Lomb PureVision |
| Methafilcon A | 4 | 55% | yes | no | FitView |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hendiger, E.B.; Padzik, M.; Sifaoui, I.; Reyes-Batlle, M.; López-Arencibia, A.; Zyskowska, D.; Grodzik, M.; Pietruczuk-Padzik, A.; Hendiger, J.; Olędzka, G.; et al. Silver Nanoparticles Conjugated with Contact Lens Solutions May Reduce the Risk of Acanthamoeba Keratitis. Pathogens 2021, 10, 583. https://doi.org/10.3390/pathogens10050583
Hendiger EB, Padzik M, Sifaoui I, Reyes-Batlle M, López-Arencibia A, Zyskowska D, Grodzik M, Pietruczuk-Padzik A, Hendiger J, Olędzka G, et al. Silver Nanoparticles Conjugated with Contact Lens Solutions May Reduce the Risk of Acanthamoeba Keratitis. Pathogens. 2021; 10(5):583. https://doi.org/10.3390/pathogens10050583
Chicago/Turabian StyleHendiger, Edyta B., Marcin Padzik, Inés Sifaoui, María Reyes-Batlle, Atteneri López-Arencibia, Diana Zyskowska, Marta Grodzik, Anna Pietruczuk-Padzik, Jacek Hendiger, Gabriela Olędzka, and et al. 2021. "Silver Nanoparticles Conjugated with Contact Lens Solutions May Reduce the Risk of Acanthamoeba Keratitis" Pathogens 10, no. 5: 583. https://doi.org/10.3390/pathogens10050583
APA StyleHendiger, E. B., Padzik, M., Sifaoui, I., Reyes-Batlle, M., López-Arencibia, A., Zyskowska, D., Grodzik, M., Pietruczuk-Padzik, A., Hendiger, J., Olędzka, G., Chomicz, L., Piñero, J. E., & Lorenzo-Morales, J. (2021). Silver Nanoparticles Conjugated with Contact Lens Solutions May Reduce the Risk of Acanthamoeba Keratitis. Pathogens, 10(5), 583. https://doi.org/10.3390/pathogens10050583











