Analysis of Nucleotide Sequence of Tax, miRNA and LTR of Bovine Leukemia Virus in Cattle with Different Levels of Persistent Lymphocytosis in Russia
Abstract
1. Introduction
2. Results
2.1. Selection of Cattle with High and Low Lymphocytosis
2.2. Variation among Tax Amino Acid Sequences
2.3. Identification of Single Nucleotide Polymorphisms in BLV miRNAs and Promoter Elements
2.4. Identification of SNPs in BLV LTR
2.5. An Association between BLV Sequence Variants and the Level of Persistent Lymphocytosis
3. Discussion
4. Materials and Methods
4.1. Sample Collection and Preparation
4.2. H3F3A Gene Quantitative Real-Time PCR Development
4.3. Quantification of BLV Provirus Copy Number
4.4. Amplification of LTR, pre-miRNA and Tax Gene Sequences
4.5. Sequence Data Analysis
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Shabeykin, A.A.; Gulyukin, A.M.; Stepanova, T.V.; Kozyreva, N.G.A. (Eds.) Risk Assessment for Interspecies Transmission of Enzootic Bovine Leukemia. In IOP Conference Series: Earth and Environmental Science; IOP Publishing Ltd.: Bristol, UK, 2019. [Google Scholar]
- Mukovnin, A.A.; Barsukov, Y.I.; Kapustin, S.I.; Kolomytsev, S.A. Epizootic Situation in Socially Significant and Especially Dangerous Animal Diseases in the Russian Federation for 2019 “TSENOVIK Agricultural Review”. 28.02.2019. Available online: https://www.tsenovik.ru/bizness/articles/mvet/epizooticheskaya-situatsiya-po-sotsialno-znachimym-i-osobo-opasnym-boleznyam-zhivotnykh-v-rossiyskoy____/ (accessed on 10 February 2021).
- Debacq, C.; Asquith, B.; Reichert, M.; Burny, A.; Kettmann, R.; Willems, L. Reduced cell turnover in bovine leukemia virus-infected, persistently lymphocytotic cattle. J. Virol. 2003, 77, 13073–13083. [Google Scholar] [CrossRef] [PubMed]
- Takeshima, S.-n.; Ohno, A.; Aida, Y. Bovine leukemia virus proviral load is more strongly associated with bovine major histocompatibility complex class II DRB3 polymorphism than with DQA1 polymorphism in Holstein cow in Japan. Retrovirology 2019, 16, 14. [Google Scholar] [CrossRef]
- Carignano, H.A.; Beribe, M.J.; Caffaro, M.E.; Amadio, A.; Nani, J.P.; Gutierrez, G.; Alvarez, I.; Trono, K.; Miretti, M.M.; Poli, M.A. BOLA-DRB3 gene polymorphisms influence bovine leukaemia virus infection levels in Holstein and Holstein × Jersey crossbreed dairy cattle. Anim. Genet. 2017, 48, 420–430. [Google Scholar] [CrossRef]
- Juliarena, M.A.; Barrios, C.N.; Ceriani, M.C.; Esteban, E.N. Hot topic: Bovine leukemia virus (BLV)-infected cows with low proviral load are not a source of infection for BLV-free cattle. J. Dairy Sci. 2016, 99, 4586–4589. [Google Scholar] [CrossRef]
- Kettmann, R.; Deschamps, J.; Cleuter, Y.; Couez, D.; Burny, A.; Marbaix, G. Leukemogenesis by bovine leukemia virus: Proviral DNA integration and lack of RNA expression of viral long terminal repeat and 3′ proximate cellular sequences. Proc. Natl. Acad. Sci. USA 1982, 79, 2465–2469. [Google Scholar] [CrossRef]
- Gillet, N.; Florins, A.; Boxus, M.; Burteau, C.; Nigro, A.; Vandermeers, F.; Balon, H.; Bouzar, A.B.; Defoiche, J.; Burny, A.; et al. Mechanisms of leukemogenesis induced by bovine leukemia virus: Prospects for novel anti-retroviral therapies in human. Retrovirology 2007, 4, 18. [Google Scholar] [CrossRef]
- Safari, R.; Jacques, J.R.; Brostaux, Y.; Willems, L. Ablation of non-coding RNAs affects bovine leukemia virus B lymphocyte proliferation and abrogates oncogenesis. PLoS Pathog. 2020, 16, e1008502. [Google Scholar] [CrossRef]
- Panei, C.J.; Takeshima, S.-n.; Omori, T.; Nunoya, T.; Davis, W.C.; Ishizaki, H.; Matoba, K.; Aida, Y. Estimation of bovine leukemia virus (BLV) proviral load harbored by lymphocyte subpopulations in BLV-infected cattle at the subclinical stage of enzootic bovine leucosis using BLV-CoCoMo-qPCR. BMC Vet. Res. 2013, 9, 95. [Google Scholar] [CrossRef]
- Somura, Y.; Sugiyama, E.; Fujikawa, H.; Murakami, K. Comparison of the copy numbers of bovine leukemia virus in the lymph nodes of cattle with enzootic bovine leukosis and cattle with latent infection. Arch. Virol. 2014, 159, 2693–2697. [Google Scholar] [CrossRef] [PubMed]
- Jimba, M.; Takeshima, S.N.; Matoba, K.; Endoh, D.; Aida, Y. BLV-CoCoMo-qPCR: Quantitation of bovine leukemia virus proviral load using the CoCoMo algorithm. Retrovirology 2010, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Sagata, N.; Yasunaga, T.; Ogawa, Y.; Tsuzuku-Kawamura, J.; Ikawa, Y. Bovine leukemia virus: Unique structural features of its long terminal repeats and its evolutionary relationship to human T-cell leukemia virus. Proc. Natl. Acad. Sci. USA 1984, 81, 4741–4745. [Google Scholar] [CrossRef]
- Rosen, C.A.; Sodroski, J.G.; Kettman, R.; Haseltine, W.A. Activation of enhancer sequences in type II human T-cell leukemia virus and bovine leukemia virus long terminal repeats by virus-associated trans-acting regulatory factors. J. Virol. 1986, 57, 738–744. [Google Scholar] [CrossRef]
- Zanotti, M.; Poli, G.; Ponti, W.; Polli, M.; Rocchi, M.; Bolzani, E.; Longeri, M.; Russo, S.; Lewin, H.A.; van Eijk, M.J. Association of BoLA class II haplotypes with subclinical progression of bovine leukaemia virus infection in Holstein-Friesian cattle. Anim. Genet. 1996, 27, 337–341. [Google Scholar]
- Forletti, A.; Lützelschwab, C.M.; Cepeda, R.; Esteban, E.N.; Gutiérrez, S.E. Early events following bovine leukaemia virus infection in calves with different alleles of the major histocompatibility complex DRB3 gene. Vet. Res. 2020, 51, 4. [Google Scholar] [CrossRef]
- Willems, L.; Burny, A.; Collete, D.; Dangoisse, O.; Dequiedt, F.; Gatot, J.S.; Kerkhofs, P.; Lefèbvre, L.; Merezak, C.; Peremans, T.; et al. Genetic determinants of bovine leukemia virus pathogenesis. AIDS Res. Hum. Retrovir. 2000, 16, 1787–1795. [Google Scholar] [CrossRef] [PubMed]
- Tajima, S.; Tsukamoto, M.; Aida, Y. Latency of viral expression in vivo is not related to CpG methylation in the U3 region and part of the R region of the long terminal repeat of bovine leukemia virus. J. Virol. 2003, 77, 4423–4430. [Google Scholar] [CrossRef] [PubMed]
- Gillet, N.A.; Hamaidia, M.; de Brogniez, A.; Gutiérrez, G.; Renotte, N.; Reichert, M.; Trono, K.; Willems, L. Bovine Leukemia Virus Small Noncoding RNAs Are Functional Elements That Regulate Replication and Contribute to Oncogenesis In Vivo. PLoS Pathog. 2016, 12, e1005588. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, R.P.; Burke, J.M.; Sullivan, C.S. RNA virus microRNA that mimics a B-cell oncomiR. Proc. Natl. Acad. Sci. USA 2012, 109, 3077–3082. [Google Scholar] [CrossRef]
- Durkin, K.; Rosewick, N.; Artesi, M.; Hahaut, V.; Griebel, P.; Arsic, N.; Burny, A.; Georges, M.; Van den Broeke, A. Characterization of novel Bovine Leukemia Virus (BLV) antisense transcripts by deep sequencing reveals constitutive expression in tumors and transcriptional interaction with viral microRNAs. Retrovirology 2016, 13, 33. [Google Scholar] [CrossRef]
- Han, Y.C.; Park, C.Y.; Bhagat, G.; Zhang, J.; Wang, Y.; Fan, J.B.; Liu, M.; Zou, Y.; Weissman, I.L.; Gu, H. microRNA-29a induces aberrant self-renewal capacity in hematopoietic progenitors, biased myeloid development, and acute myeloid leukemia. J. Exp. Med. 2010, 207, 475–489. [Google Scholar] [CrossRef]
- Santanam, U.; Zanesi, N.; Efanov, A.; Costinean, S.; Palamarchuk, A.; Hagan, J.P.; Volinia, S.; Alder, H.; Rassenti, L.; Kipps, T.; et al. Chronic lymphocytic leukemia modeled in mouse by targeted miR-29 expression. Proc. Natl. Acad. Sci. USA 2010, 107, 12210–12215. [Google Scholar] [CrossRef] [PubMed]
- Frie, M.C.; Sporer, K.R.B.; Benitez, O.J.; Wallace, J.C.; Droscha, C.J.; Bartlett, P.C.; Coussens, P.M. Dairy Cows Naturally Infected with Bovine Leukemia Virus Exhibit Abnormal B- and T-Cell Phenotypes after Primary and Secondary Exposures to Keyhole Limpet Hemocyanin. Front. Vet. Sci. 2017, 4, 112. [Google Scholar] [CrossRef]
- Dube, S.; Abbott, L.; Dube, D.K.; Dolcini, G.; Gutierrez, S.; Ceriani, C.; Juliarena, M.; Ferrer, J.; Perzova, R.; Poiesz, B.J. The complete genomic sequence of an in vivo low replicating BLV strain. Virol. J. 2009, 6, 120. [Google Scholar] [CrossRef]
- Tajima, S.; Ikawa, Y.; Aida, Y. Complete bovine leukemia virus (BLV) provirus is conserved in BLV-infected cattle throughout the course of B-cell lymphosarcoma development. J. Virol. 1998, 72, 7569–7576. [Google Scholar] [CrossRef] [PubMed]
- Twizere, J.C.; Kerkhofs, P.; Burny, A.; Portetelle, D.; Kettmann, R.; Willems, L. Discordance between bovine leukemia virus tax immortalization in vitro and oncogenicity in vivo. J. Virol. 2000, 74, 9895–9902. [Google Scholar] [CrossRef]
- Inoue, E.; Matsumura, K.; Soma, N.; Hirasawa, S.; Wakimoto, M.; Arakaki, Y.; Yoshida, T.; Osawa, Y.; Okazaki, K. L233P mutation of the Tax protein strongly correlated with leukemogenicity of bovine leukemia virus. Vet. Microbiol. 2013, 167, 364–371. [Google Scholar] [CrossRef]
- Van Den Broeke, A.; Bagnis, C.; Ciesiolka, M.; Cleuter, Y.; Gelderblom, H.; Kerkhofs, P.; Griebel, P.; Mannoni, P.; Burny, A. In vivo rescue of a silent tax-deficient bovine leukemia virus from a tumor-derived ovine B-cell line by recombination with a retrovirally transduced wild-type tax gene. J. Virol. 1999, 73, 1054–1065. [Google Scholar] [CrossRef] [PubMed]
- Willems, L.; Kettmann, R.; Dequiedt, F.; Portetelle, D.; Voneche, V.; Cornil, I.; Kerkhofs, P.; Burny, A.; Mammerickx, M. In vivo infection of sheep by bovine leukemia virus mutants. J. Virol. 1993, 67, 4078–4085. [Google Scholar] [CrossRef]
- Zyrianova, I.M.; Kovalchuk, S.N. Bovine leukemia virus tax gene/Tax protein polymorphism and its relation to Enzootic Bovine Leukosis. Virulence 2020, 11, 80–87. [Google Scholar] [CrossRef]
- Zyrianova, I.M.; Koval’chuk, S.N. Bovine leukemia virus pre-miRNA genes’ polymorphism. RNA Biol. 2018, 15, 1440–1447. [Google Scholar] [CrossRef]
- Pluta, A.; Jaworski, J.P.; Douville, R.N. Regulation of Expression and Latency in BLV and HTLV. Viruses 2020, 12, 1079. [Google Scholar] [CrossRef]
- Murakami, H.; Todaka, H.; Uchiyama, J.; Sato, R.; Sogawa, K.; Sakaguchi, M.; Tsukamoto, K. A point mutation to the long terminal repeat of bovine leukemia virus related to viral productivity and transmissibility. Virology 2019, 537, 45–52. [Google Scholar] [CrossRef]
- Murakami, H.; Uchiyama, J.; Suzuki, C.; Nikaido, S.; Shibuya, K.; Sato, R.; Maeda, Y.; Tomioka, M.; Takeshima, S.N.; Kato, H.; et al. Variations in the viral genome and biological properties of bovine leukemia virus wild-type strains. Virus Res. 2018, 253, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Pluta, A.; Willems, L.; Douville, R.N.; Kuźmak, J. Effects of Naturally Occurring Mutations in Bovine Leukemia Virus 5′-LTR and Tax Gene on Viral Transcriptional Activity. Pathogens 2020, 9, 836. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Watanuki, S.; Murakami, H.; Sato, R.; Ishizaki, H.; Aida, Y. Development of a luminescence syncytium induction assay (LuSIA) for easily detecting and quantitatively measuring bovine leukemia virus infection. Arch. Virol. 2018, 163, 1519–1530. [Google Scholar] [CrossRef]
- Iwanaga, M.; Watanabe, T.; Utsunomiya, A.; Okayama, A.; Uchimaru, K.; Koh, K.R.; Ogata, M.; Kikuchi, H.; Sagara, Y.; Uozumi, K.; et al. Human T-cell leukemia virus type I (HTLV-1) proviral load and disease progression in asymptomatic HTLV-1 carriers: A nationwide prospective study in Japan. Blood 2010, 116, 1211–1219. [Google Scholar] [CrossRef] [PubMed]
- Ohno, A.; Takeshima, S.-n.; Matsumoto, Y.; Aida, Y. Risk factors associated with increased bovine leukemia virus proviral load in infected cattle in Japan from 2012 to 2014. Virus Res. 2015, 210, 283–290. [Google Scholar] [CrossRef]
- Frie, M.C.; Droscha, C.J.; Greenlick, A.E.; Coussens, P.M. MicroRNAs Encoded by Bovine Leukemia Virus (BLV) Are Associated with Reduced Expression of B Cell Transcriptional Regulators in Dairy Cattle Naturally Infected with BLV. Front. Vet. Sci. 2017, 4, 245. [Google Scholar] [CrossRef]
- Willems, L.; Grimonpont, C.; Kerkhofs, P.; Capiau, C.; Gheysen, D.; Conrath, K.; Roussef, R.; Mamoun, R.; Portetelle, D.; Burny, A.; et al. Phosphorylation of bovine leukemia virus Tax protein is required for in vitro transformation but not for transactivation. Oncogene 1998, 16, 2165–2176. [Google Scholar] [CrossRef]
- Szynal, M.; Cleuter, Y.; Beskorwayne, T.; Bagnis, C.; Van Lint, C.; Kerkhofs, P.; Burny, A.; Martiat, P.; Griebel, P.; Van den Broeke, A. Disruption of B-cell homeostatic control mediated by the BLV-Tax oncoprotein: Association with the upregulation of Bcl-2 and signaling through NF-κB. Oncogene 2003, 22, 4531–4542. [Google Scholar] [CrossRef]
- Kettmann, R.; Cleuter, Y.; Gregoire, D.; Burny, A. Role of the 3′ long open reading frame region of bovine leukemia virus in the maintenance of cell transformation. J. Virol. 1985, 54, 899–901. [Google Scholar] [CrossRef]
- Brooks, P.A.; Cockerell, G.L.; Nyborg, J.K. Activation of BLV Transcription by NF-κB and Tax. Virology 1998, 243, 94–98. [Google Scholar] [CrossRef][Green Version]
- Arainga, M.; Takeda, E.; Aida, Y. Identification of bovine leukemia virus tax function associated with host cell transcription, signaling, stress response and immune response pathway by microarray-based gene expression analysis. BMC Genom. 2012, 13, 121. [Google Scholar] [CrossRef] [PubMed]
- Willems, L.; Grimonpont, C.; Heremans, H.; Rebeyrotte, N.; Chen, G.; Portetelle, D.; Burny, A.; Kettmann, R. Mutations in the bovine leukemia virus Tax protein can abrogate the long terminal repeat-directed transactivating activity without concomitant loss of transforming potential. Proc. Natl. Acad. Sci. USA 1992, 89, 3957–3961. [Google Scholar] [CrossRef] [PubMed]
- Stone, D.M.; Norton, L.K.; Chambers, J.C.; Meek, W.J. CD4 T lymphocyte activation in BLV-induced persistent B lymphocytosis in cattle. Clin. Immunol. 2000, 96, 280–288. [Google Scholar] [CrossRef]
- Bai, L.; Takeshima, S.N.; Isogai, E.; Kohara, J.; Aida, Y. Novel CD8(+) cytotoxic T cell epitopes in bovine leukemia virus with cattle. Vaccine 2015, 33, 7194–7202. [Google Scholar] [CrossRef]
- Sakakibara, N.; Kabeya, H.; Ohashi, K.; Sugimoto, C.; Onuma, M. Epitope mapping of bovine leukemia virus transactivator protein Tax. J. Vet. Med. Sci. 1998, 60, 599–605. [Google Scholar] [CrossRef]
- Niewiesk, S.; Daenke, S.; Parker, C.E.; Taylor, G.; Weber, J.; Nightingale, S.; Bangham, C.R. Naturally occurring variants of human T-cell leukemia virus type I Tax protein impair its recognition by cytotoxic T lymphocytes and the transactivation function of Tax. J. Virol. 1995, 69, 2649–2653. [Google Scholar] [CrossRef]
- Rosewick, N.; Momont, M.; Durkin, K.; Takeda, H.; Caiment, F.; Cleuter, Y.; Vernin, C.; Mortreux, F.; Wattel, E.; Burny, A.; et al. Deep sequencing reveals abundant noncanonical retroviral microRNAs in B-cell leukemia/lymphoma. Proc. Natl. Acad. Sci. USA 2013, 110, 2306–2311. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Buehring, G.C. In vivo protein binding and functional analysis of cis-acting elements in the U3 region of the bovine leukemia virus long terminal repeat. J. Virol. 1998, 72, 5994–6003. [Google Scholar] [CrossRef]
- Calomme, C.; Nguyen, T.L.; de Launoit, Y.; Kiermer, V.; Droogmans, L.; Burny, A.; Van Lint, C. Upstream stimulatory factors binding to an E box motif in the R region of the bovine leukemia virus long terminal repeat stimulates viral gene expression. J. Biol. Chem. 2002, 277, 8775–8789. [Google Scholar] [CrossRef]
- Calomme, C.; Dekoninck, A.; Nizet, S.; Adam, E.; Nguyên, T.L.-A.; Van Den Broeke, A.; Willems, L.; Kettmann, R.; Burny, A.; Van Lint, C. Overlapping CRE and E Box Motifs in the Enhancer Sequences of the Bovine Leukemia Virus 5′ Long Terminal Repeat Are Critical for Basal and Acetylation-Dependent Transcriptional Activity of the Viral Promoter: Implications for Viral Latency. J. Virol. 2004, 78, 13848–13864. [Google Scholar] [CrossRef]
- Gaynor, E.M.; Mirsky, M.L.; Lewin, H.A. Use of flow cytometry and RT-PCR for detecting gene expression by single cells. Biotechniques 1996, 21, 286–291. [Google Scholar] [CrossRef]
- Radke, K.; Sigala, T.J.; Grossman, D. Transcription of bovine leukemia virus in peripheral blood cells obtained during early infection in vivo. Microb. Pathog. 1992, 12, 319–331. [Google Scholar] [CrossRef]
- Gupta, P.; Ferrer, J.F. Expression of bovine leukemia virus genome is blocked by a nonimmunoglobulin protein in plasma from infected cattle. Science 1982, 215, 405–407. [Google Scholar] [CrossRef]
- Lagarias, D.M.; Radke, K. Transcriptional activation of bovine leukemia virus in blood cells from experimentally infected, asymptomatic sheep with latent infections. J. Virol. 1989, 63, 2099–2107. [Google Scholar] [CrossRef]
- Merezak, C.; Pierreux, C.; Adam, E.; Lemaigre, F.; Rousseau, G.G.; Calomme, C.; Van Lint, C.; Christophe, D.; Kerkhofs, P.; Burny, A.; et al. Suboptimal enhancer sequences are required for efficient bovine leukemia virus propagation in vivo: Implications for viral latency. J. Virol. 2001, 75, 6977–6988. [Google Scholar] [CrossRef]
- Pomier, C.; Alcaraz, M.T.; Debacq, C.; Lançon, A.; Kerkhofs, P.; Willems, L.; Wattel, E.; Mortreux, F. Early and transient reverse transcription during primary deltaretroviral infection of sheep. Retrovirology 2008, 5, 16. [Google Scholar] [CrossRef] [PubMed]
- Mortreux, F.; Leclercq, I.; Gabet, A.S.; Leroy, A.; Westhof, E.; Gessain, A.; Wain-Hobson, S.; Wattel, E. Somatic mutation in human T-cell leukemia virus type 1 provirus and flanking cellular sequences during clonal expansion in vivo. J. Natl. Cancer Inst. 2001, 93, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Moulés, V.; Pomier, C.; Sibon, D.; Gabet, A.S.; Reichert, M.; Kerkhofs, P.; Willems, L.; Mortreux, F.; Wattel, E. Fate of premalignant clones during the asymptomatic phase preceding lymphoid malignancy. Cancer Res. 2005, 65, 1234–1243. [Google Scholar] [CrossRef][Green Version]
- Afonso, P.V.; Cassar, O.; Gessain, A. Molecular epidemiology, genetic variability and evolution of HTLV-1 with special emphasis on African genotypes. Retrovirology 2019, 16, 39. [Google Scholar] [CrossRef]
- Niewiesk, S.; Bangham, C.R. Evolution in a chronic RNA virus infection: Selection on HTLV-I tax protein differs between healthy carriers and patients with tropical spastic paraparesis. J. Mol. Evol. 1996, 42, 452–458. [Google Scholar] [CrossRef]
- Bendixen, H.J. Preventive measures in cattle leukemia: Leukosis enzootica bovis. Ann. N. Y. Acad. Sci. 1963, 108, 1241–1267. [Google Scholar] [CrossRef]
- Alvarez, I.; Gutiérrez, G.; Gammella, M.; Martínez, C.; Politzki, R.; González, C.; Caviglia, L.; Carignano, H.; Fondevila, N.; Poli, M.; et al. Evaluation of total white blood cell count as a marker for proviral load of bovine leukemia virus in dairy cattle from herds with a high seroprevalence of antibodies against bovine leukemia virus. Am. J. Vet. Res. 2013, 74, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Rola-Łuszczak, M.; Finnegan, C.; Olech, M.; Choudhury, B.; Kuźmak, J. Development of an improved real time PCR for the detection of bovine leukaemia provirus nucleic acid and its use in the clarification of inconclusive serological test results. J. Virol. Methods 2013, 189, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Assi, W.; Hirose, T.; Wada, S.; Matsuura, R.; Takeshima, S.N.; Aida, Y. PRMT5 Is Required for Bovine Leukemia Virus Infection In Vivo and Regulates BLV Gene Expression, Syncytium Formation, and Glycosylation In Vitro. Viruses 2020, 12, 650. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Wingender, E.; Karas, H.; Knüppel, R. TRANSFAC database as a bridge between sequence data libraries and biological function. Pac. Symp. Biocomput. 1997, 2, 477–485. [Google Scholar]
- Rice, P.; Longden, I.; Bleasby, A. EMBOSS: The European Molecular Biology Open Software Suite. Trends Genet. 2000, 16, 276–277. [Google Scholar] [CrossRef]
- Burke, J.M.; Bass, C.R.; Kincaid, R.P.; Sullivan, C.S. Identification of tri-phosphatase activity in the biogenesis of retroviral microRNAs and RNAP III-generated shRNAs. Nucleic Acids Res. 2014, 42, 13949–13962. [Google Scholar] [CrossRef] [PubMed]
- Burke, J.M.; Kincaid, R.P.; Aloisio, F.; Welch, N.; Sullivan, C.S. Expression of short hairpin RNAs using the compact architecture of retroviral microRNA genes. Nucl. Acids Res. 2017, 45, e154. [Google Scholar] [CrossRef] [PubMed]
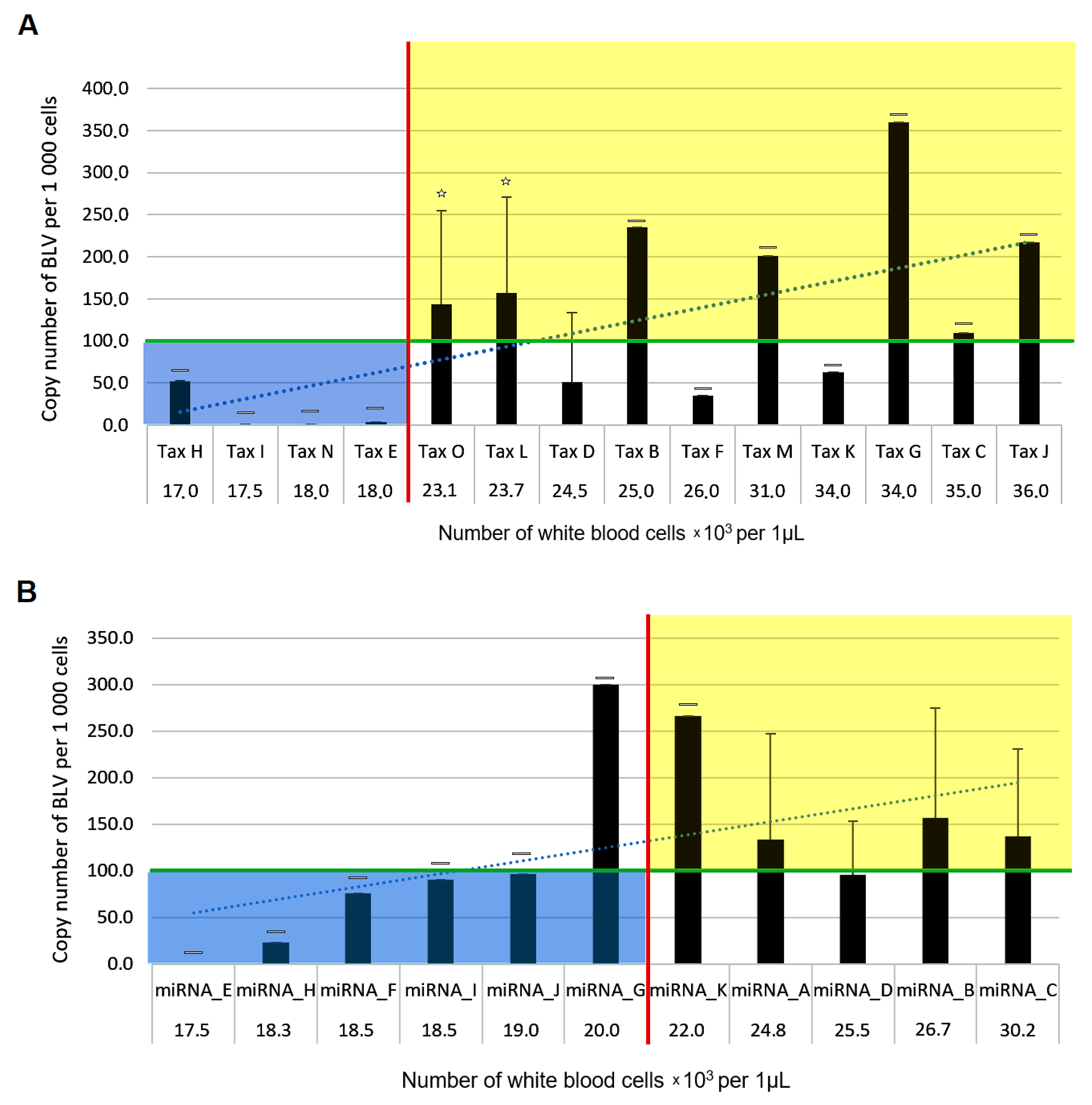
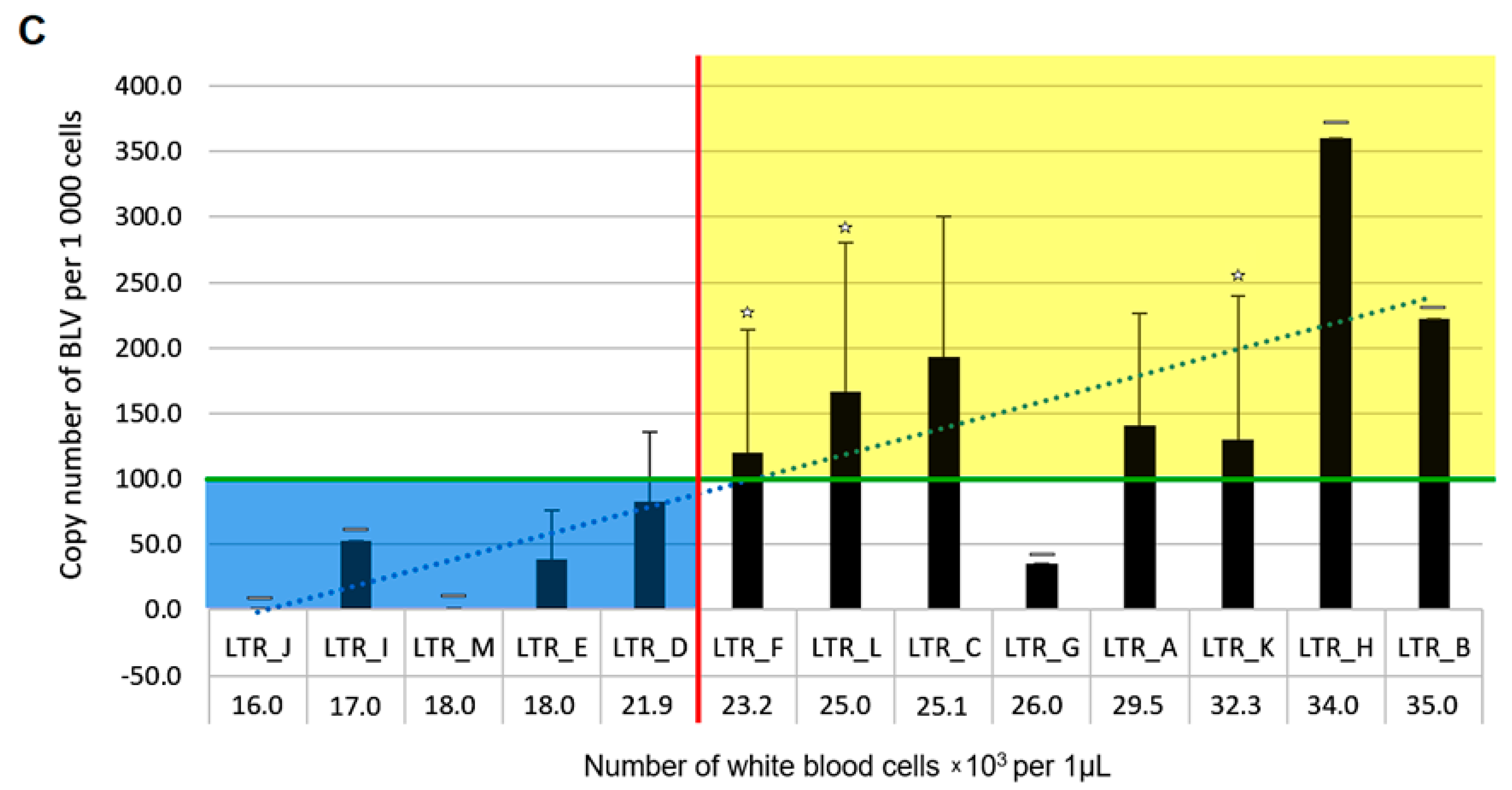
| Variant Name | Position | Amino Acid | Domain | Respective Number of Cattle | Genotype |
|---|---|---|---|---|---|
| Tax_A | 40 | Q -> L | Zn finger domain | LE48 | 4 |
| 142 | V -> A | T-cell epitope | |||
| 233, 281 | L -> P, S -> P | -* | |||
| Tax_B | 42 | E -> K | Zn finger domain | PL33 | 4 |
| 140 | N -> I | T-cell epitope | |||
| 142 | V -> A | T-cell epitope | |||
| 233, 281 | L -> P, S -> P | - | |||
| Tax_C | 51 | E -> K | Zn finger domain | PL42 | 4 |
| 131 | I -> V | T-cell epitope | |||
| 186 | I -> V | CTL epitope & Leucine-rich domain | |||
| 69, 102, 214, 221, 228 | T -> M, V -> I, I -> T, T -> P, E -> A | - | |||
| Tax_D | 52 | T -> I | Zn finger domain | PL20, PL30, PL32, PL44 | 4 |
| 183 | R -> K | CTL epitope & Leucine-rich domain | |||
| 278 | L -> I | B-cell epitope | |||
| Tax_E | 52 | T -> I | Zn finger domain | PL13 | 4 |
| 140 | N -> K | T-cell epitope | |||
| 183 | R -> K | CTL epitope & Leucine-rich domain | |||
| 278 | L -> I | B-cell epitope | |||
| 69 | T -> M | - | |||
| Tax_F | 52 | T -> I | Zn finger domain | PL35 | 4 |
| 124 | S -> F | T-cell epitope | |||
| 183 | R -> K | CTL epitope & Leucine-rich domain | |||
| 278 | L -> I | B-cell epitope | |||
| 2, 22, 73 | S -> A, N -> S, R -> Q | - | |||
| Tax_G | 52 | T -> I | Zn finger domain | PL41 | 4 |
| 183 | R -> K | CTL epitope & Leucine-rich domain | |||
| 245 | A -> T | Multifunctional domain | |||
| 278 | L -> I | B-cell epitope | |||
| 235 | Q -> R | - | |||
| Tax_H | 178 | F -> Y | CTL epitope & Leucine-rich domain | PL5 | 4 |
| 246 | I -> T | Multifunctional domain | |||
| 249 | F -> L | Multifunctional domain | |||
| Tax_I | 245 | A -> T | Multifunctional domain | PL9 | 4 |
| 102, 214, 221 | V -> I, I -> T, T -> P | - | |||
| Tax_J | 117 | Q -> R | T-cell epitope | PL45 | 4 |
| 102 | V -> I | - | |||
| Tax_K | 141 | L -> S | T-cell epitope | PL40 | 4 |
| 186 | I -> V | CTL epitope & Leucine-rich domain | |||
| Tax_L | 167 | T -> I | CTL epitope & Leucine-rich domain | PL14, PL15, PL21, PL25, PL27, PL28, PL36, PL39, PL46 | 7 |
| 173 | L -> P | CTL epitope & Leucine-rich domain | |||
| 257 | Y -> C | Multifunctional domain | |||
| 2, 69, 95, 214, 221, 233, 281 | S -> A5/9 §, T -> A, A -> T, I -> T8/9, T -> S, L -> P, S -> P | - | |||
| Tax_M | 167 | T -> I | CTL epitope & Leucine-rich domain | PL38 | 7 |
| 173 | L -> P | CTL epitope & Leucine-rich domain | |||
| 198 | A -> T | CTL epitope | |||
| 257 | Y -> C | Multifunctional domain | |||
| 69, 95, 198, 214, 221, 227, 233, 281 | T -> A, A -> T, A -> T, I -> V, T -> S, S ->T, L -> P, S -> P | - | |||
| Tax_N | 43 | R -> K | Zn finger domain | PL10 | 7 |
| 173 | L -> P | CTL epitope & Leucine-rich domain | |||
| 257 | Y -> C | Multifunctional domain | |||
| 2, 69, 95, 212, 221, 233, 281 | S -> A, T -> A, A -> T, V -> I, T -> S, L -> P, S -> P | - | |||
| Tax_O | 102, 214, 221 | V -> I7/9, I -> T6/9, T -> P 6/9 | - | PL17, PL18, PL19, PL24, PL26, PL29, PL31, PL37, PL47 | 4 |
| Variant Name | Position | Nucleotide | miRNAs | Respective Number of Cattle | Genotype |
|---|---|---|---|---|---|
| miRNA_A | 84, 171, 195, 210, 212 | A -> G, A -> G, G-> A, G-> A, T->A, | -Գ | PL8, PL14, PL15, PL25, PL28, PL36, PL38, PL39, PL46 | 7 |
| 310 | A -> del * | BLV-mir-B4-5p | |||
| 342 | G -> A | B4 pre-miRNA, putative A-box | |||
| 357 | A -> G | BLV-mir-B4-3p | |||
| 373, 392, 453 | G -> A, G -> A, C -> T | - | |||
| 462 | A -> G | BLV-mir-B5-5p | |||
| 497–498 | GA -> AG | BLV-mir-B5-3p | |||
| miRNA_B | 168 | T -> del14/15 | - | AL2, AL3, PL11, PL17, PL23, PL24, PL26, PL29, PL31, PL37, PL40, PL41, PL42, PL45, PL47 | 4 |
| miRNA_C | 106 | G -> A4/5 | - | PL5, PL12, PL33, PL43, LE48 | 4 |
| 154 | A -> G | BLV-mir-B2-3p | |||
| miRNA_D | 249 | C -> G | BLV-mir-B3-5p, putative A-box | PL21, PL30, PL35, PL44 | 4 |
| miRNA_E | 141 | G -> A | B2 pre-miRNA | PL9 | 4 |
| 168 | T -> del | - | |||
| miRNA_F | 141 | G -> A | B2 pre-miRNA | PL18 | 4 |
| 168 | T -> del | - | |||
| 171 | A -> G | putativeA-box | |||
| 91, 300 | A -> G, A -> G | - | |||
| miRNA_G | 64, 67, 84, 171, 195, 210, 212 | G -> A, A->T, A -> G, A -> G, G -> A, G -> A, T -> A | - | PL27 | 7 |
| 310 | A -> del | BLV-mir-B4-5p | |||
| 341 | C -> T | putative A-box | |||
| 342 | G -> A | B4 pre-miRNA, putative A-box | |||
| 357 | A -> G | BLV-mir-B4-3p | |||
| 373, 392, 453 | G -> A, G -> A, C -> T | - | |||
| 462 | A -> G | BLV-mir-B5-5p | |||
| 497–498 | GA -> AG | BLV-mir-B5-3p | |||
| miRNA_H | 97, 168 | A -> G, T -> del | - | PL16 | 4 |
| 420 | A -> G | putative B-box | |||
| miRNA_I | 168 | T -> del | - | PL19 | 4 |
| 420 | A -> G | putative B-box | |||
| 467 | A -> G | BLV-mir-B5-5p, putative B-box | |||
| miRNA_J | 107 | A -> G | - | PL20 | 4 |
| 239 | C -> T | BLV-mir-B3-5p | |||
| 249 | C -> G | BLV-mir-B3-5p, putative A-box | |||
| miRNA_K | 181 | G ->A | putative B-box | PL32 | 4 |
| 249 | C -> G | BLV-mir-B3-5p, putative A-box |
| Variant Name | Position | Nucleotide | Regulatory Element | Respective Number of Cattle | Genotype |
|---|---|---|---|---|---|
| LTR_A | −133 | G -> A | TRE2 | PL12, PL22, PL33, LE48 | 4 |
| +5 | C -> T | putative GR & PR site | |||
| +183 | C -> T | DAS | |||
| LTR_B | −133 | G -> A | TRE2 | PL43 | 4 |
| +5 | C -> T | putative GR & PR site | |||
| +150 | A -> C | Box A (DAS) | |||
| +183 | C -> T | DAS | |||
| +185 | C -> T | Box C (DAS) | |||
| LTR_C | +187 | C -> A | Box C (DAS) | AL1, AL2, PL23, PL26, PL31, PL37, PL45 | 4 |
| LTR_D | −4 | A -> G | CAP site | PL11, PL17, PL19, PL24, PL42 | 4 |
| +187 | C -> A | Box C (DAS) | |||
| LTR_E | −4 | A -> G | CAP site | PL9, PL18 | 4 |
| +5 | C -> T | putative GR & PR site | |||
| +187 | C -> A | Box C (DAS) | |||
| LTR_F | +161 | T -> C | DAS | PL13, PL20, PL30, PL32, PL44 | 4 |
| LTR_G | −10 | A -> G | CAP site | PL35 | 4 |
| +161 | T -> C | DAS | |||
| +190 | T -> C | Box C (DAS) | |||
| LTR_H | the consensus sequence-like | PL41 | 4 | ||
| LTR_I | −65 | T -> C | GRE | PL5 | 4 |
| LTR_J | −142 | G -> A | NF-kB-like protein | PL4 | 4 |
| −11 | T -> C | CAP site | |||
| LTR_K | −122 | T -> A | E box 2 | PL29, PL40, PL47 | 4 |
| LTR_L | −133 | G -> C | TRE2 | PL14, PL15, PL25, PL27, PL28, PL36, PL38, PL39, PL46 | 7 |
| +11 | C -> T | putative Sp1 & GR site | |||
| +150 | A -> G | Box A (DAS) | |||
| +160 | C -> T | DAS | |||
| LTR_M | +11 | C -> T | putative Sp1 & GR site | PL10 | 7 |
| +150 | A -> G | Box A (DAS) | |||
| Gene/Region | Variant | Mutation | Regulatory Domains/Elements |
|---|---|---|---|
| Tax | Tax H | F(178)Y I(246)T, F(249)L | CTL epitope & Leucine-rich domain Multifunctional domain |
| Tax I | - | - | |
| Tax N | R(43)K V(212)I | Zn finger domain adjacent to CTL epitope | |
| Tax E | - | - | |
| miRNA | miRNA_E | G(141)A | B2 pre-miRNA |
| miRNA_H | A(420)H | putative B-box | |
| miRNA_F | G(141)A | B2 pre-miRNA | |
| miRNA_I | A(420)H A(467)G | putative B-box BLV-mir-B5-5p & putative B-box | |
| miRNA_J | C(239)T | BLV-mir-B3-5p | |
| LTR | LTR_J | G(−142)A T(−11)C | NF-kB-like protein CAP site |
| LTR_I | T(−65)C | GRE | |
| LTR_M | - | - | |
| LTR_E | A(−4)G | CAP site | |
| LTR_D | A(−4)G | CAP site |
| Gene/Region | Variant | Mutation | Regulatory Domains/Elements |
|---|---|---|---|
| Tax | Tax O | - | - |
| Tax L | - | - | |
| Tax B | E(42)K, V(142)A | Zn finger domain, T-cell epitope | |
| Tax M | A(198)T | CD8+ T-cell epitope | |
| Tax G | - | - | |
| Tax C | E(51)K, I(131)V | Zn finger domain, T-cell epitope | |
| Tax J | Q(117)R | T-cell epitope | |
| miRNA | miRNA_K | C(249)G G(181)A | BLV-mir-B3-5p, putative B-box |
| miRNA_A | A(310)del *, G(342)A, A(357)G, A(462)G, GA(497–498)AG | BLV-mir-B4-5p, B4 pre-miRNA with putative A-box, BLV-mir-B4-3p, BLV-mir-B5-5p, BLV-mir-B5-3p | |
| miRNA_B | - | - | |
| miRNA_D | C(249)G | BLV-mir-B3-5p | |
| miRNA_C | A(154)G | BLV-mir-B2-3p | |
| LTR | LTR F | - | - |
| LTR L | G(−133)C, C(+160)T | CRE2, DAS | |
| LTR A | G(−133)A, C(+183)T | CRE2, DAS | |
| LTR K | T(−122)A | E box2 | |
| LTR H | - | - | |
| LTR C | - | - | |
| LTR B | G(−133)A, A(+150)C, C(+183)T, C(+185)T | CRE2, Box A (DAS), DAS, Box C (DAS) |
| Region | Predominant Residue (Position) | Chi-Square p-Value | |
|---|---|---|---|
| Low PL (Group I) | High PL (Group II) | ||
| Tax | I(214) | T(214) | 0.11416 |
| T(221) | S(221) | 0.34776 | |
| L(233) | P(233) | 0.35330 | |
| S(281) | P(281) | 0.35331 | |
| miRNA | del(168) | T(168) | 0.13392 |
| A(441) | G(441) | 0.11976 | |
| LTR | A(+187) | C(+187) | 0.00737 * |
| Cattle # | Lymphocyte × 103/µL | WBC × 103/µL | Clinical Stage | Breed | Herd # | Proviral Copy Number per 103 Cells |
|---|---|---|---|---|---|---|
| AL1 | 8.8 | 14 | AL | B-a-W | I | 0.01 |
| AL2 | 7.6 | 11 | AL | B-a-W | I | 224.8 |
| AL3 | 9.2 | 14 | AL | B-a-W | I | 0.7 |
| PL4 | 11.2 | 16 | PL | B-a-W | I | 1.1 |
| PL5 | 12.8 | 17 | PL | RedStep | II | 52.4 |
| PL6 | 12.2 | 17 | PL | B-a-W | I | 0.01 |
| PL7 | 11.9 | 17 | PL | Ayrshire | III | 0.1 |
| PL8 | 12.4 | 17.5 | PL | RedStep | II | 0.3 |
| PL9 | 14.4 | 17.5 | PL | B-a-W | IV | 0.4 |
| PL10 | 10.6 | 18 | PL | RedStep | II | 1.1 |
| PL11 | 11.7 | 18 | PL | B-a-W | IV | 1.3 |
| PL12 | 9.4 | 18 | PL | B-a-W | IV | 1.8 |
| PL13 | 10.3 | 18 | PL | Ayrshire | III | 3.9 |
| PL14 | 12.2 | 18 | PL | Ayrshire | III | 8.4 |
| PL15 | 12.6 | 18.3 | PL | Ayrshire | III | 16.0 |
| PL16 | 14.3 | 18.3 | PL | B-a-W | IV | 23.6 |
| PL17 | 11.5 | 18.3 | PL | B-a-W | IV | 50.7 |
| PL18 | 14.1 | 18.5 | PL | B-a-W | IV | 76.1 |
| PL19 | 12.0 | 18.5 | PL | B-a-W | IV | 90.8 |
| PL20 | 12.0 | 19 | PL | RedStep | II | 96.6 |
| PL21 | 14.3 | 19 | PL | Ayrshire | III | 112.6 |
| PL22 | 13.1 | 19 | PL | B-a-W | IV | 150.8 |
| PL23 | 13.3 | 19.5 | PL | B-a-W | IV | 159.1 |
| PL24 | 18.3 | 19.5 | PL | B-a-W | IV | 160.1 |
| PL25 | 9.0 | 19.5 | PL | Ayrshire | III | 226.7 |
| PL26 | 13.4 | 20 | PL | B-a-W | IV | 271.3 |
| PL27 | 11.4 | 20 | PL | Ayrshire | III | 300.0 |
| PL28 | 13.6 | 20 | PL | RedStep | II | 330.2 |
| PL29 | 14.3 | 21 | PL | Ayrshire | III | 41.6 |
| PL30 | 12.2 | 21 | PL | Ayrshire | III | 51.1 |
| PL31 | 14.9 | 21 | PL | B-a-W | I | 5.3 |
| PL32 | 18.0 | 22 | PL | RedStep | II | 266.2 |
| PL33 | 15.0 | 25 | PL | B-a-W | IV | 235.2 |
| PL34 | 16.2 | 24.6 | PL | B-a-W | IV | 314.6 |
| PL35 | 19.2 | 26 | PL | RedStep | II | 35.2 |
| PL36 | 16.5 | 28.5 | PL | Ayrshire | III | 44.9 |
| PL37 | 22.3 | 29 | PL | B-a-W | I | 312.3 |
| PL38 | 23.3 | 31 | PL | Ayrshire | III | 201.2 |
| PL39 | 27.2 | 32 | PL | RedStep | II | 228.4 |
| PL40 | 27.5 | 34 | PL | RedStep | II | 62.9 |
| PL41 | 27.2 | 34 | PL | B-a-W | I | 359.9 |
| PL42 | 25.6 | 35 | PL | B-a-W | IV | 109.4 |
| PL43 | 25.6 | 35 | PL | B-a-W | IV | 222.4 |
| PL44 | 21.6 | 36 | PL | Ayrshire | III | 182.8 |
| PL45 | 27.7 | 36 | PL | B-a-W | IV | 217.1 |
| PL46 | 30.0 | 38 | PL | Ayrshire | III | 143.6 |
| PL47 | 34.0 | 42 | PL | RedStep | II | 284.4 |
| LE48 | 44.8 | 56 | LE | B-a-W | IV | 175.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pluta, A.; Blazhko, N.V.; Ngirande, C.; Joris, T.; Willems, L.; Kuźmak, J. Analysis of Nucleotide Sequence of Tax, miRNA and LTR of Bovine Leukemia Virus in Cattle with Different Levels of Persistent Lymphocytosis in Russia. Pathogens 2021, 10, 246. https://doi.org/10.3390/pathogens10020246
Pluta A, Blazhko NV, Ngirande C, Joris T, Willems L, Kuźmak J. Analysis of Nucleotide Sequence of Tax, miRNA and LTR of Bovine Leukemia Virus in Cattle with Different Levels of Persistent Lymphocytosis in Russia. Pathogens. 2021; 10(2):246. https://doi.org/10.3390/pathogens10020246
Chicago/Turabian StylePluta, Aneta, Natalia V. Blazhko, Charity Ngirande, Thomas Joris, Luc Willems, and Jacek Kuźmak. 2021. "Analysis of Nucleotide Sequence of Tax, miRNA and LTR of Bovine Leukemia Virus in Cattle with Different Levels of Persistent Lymphocytosis in Russia" Pathogens 10, no. 2: 246. https://doi.org/10.3390/pathogens10020246
APA StylePluta, A., Blazhko, N. V., Ngirande, C., Joris, T., Willems, L., & Kuźmak, J. (2021). Analysis of Nucleotide Sequence of Tax, miRNA and LTR of Bovine Leukemia Virus in Cattle with Different Levels of Persistent Lymphocytosis in Russia. Pathogens, 10(2), 246. https://doi.org/10.3390/pathogens10020246







