Solvent Extraction and Separation of Nd, Pr and Dy from Leach Liquor of Waste NdFeB Magnet Using the Nitrate Form of Mextral® 336At in the Presence of Aquo-Complexing Agent EDTA
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
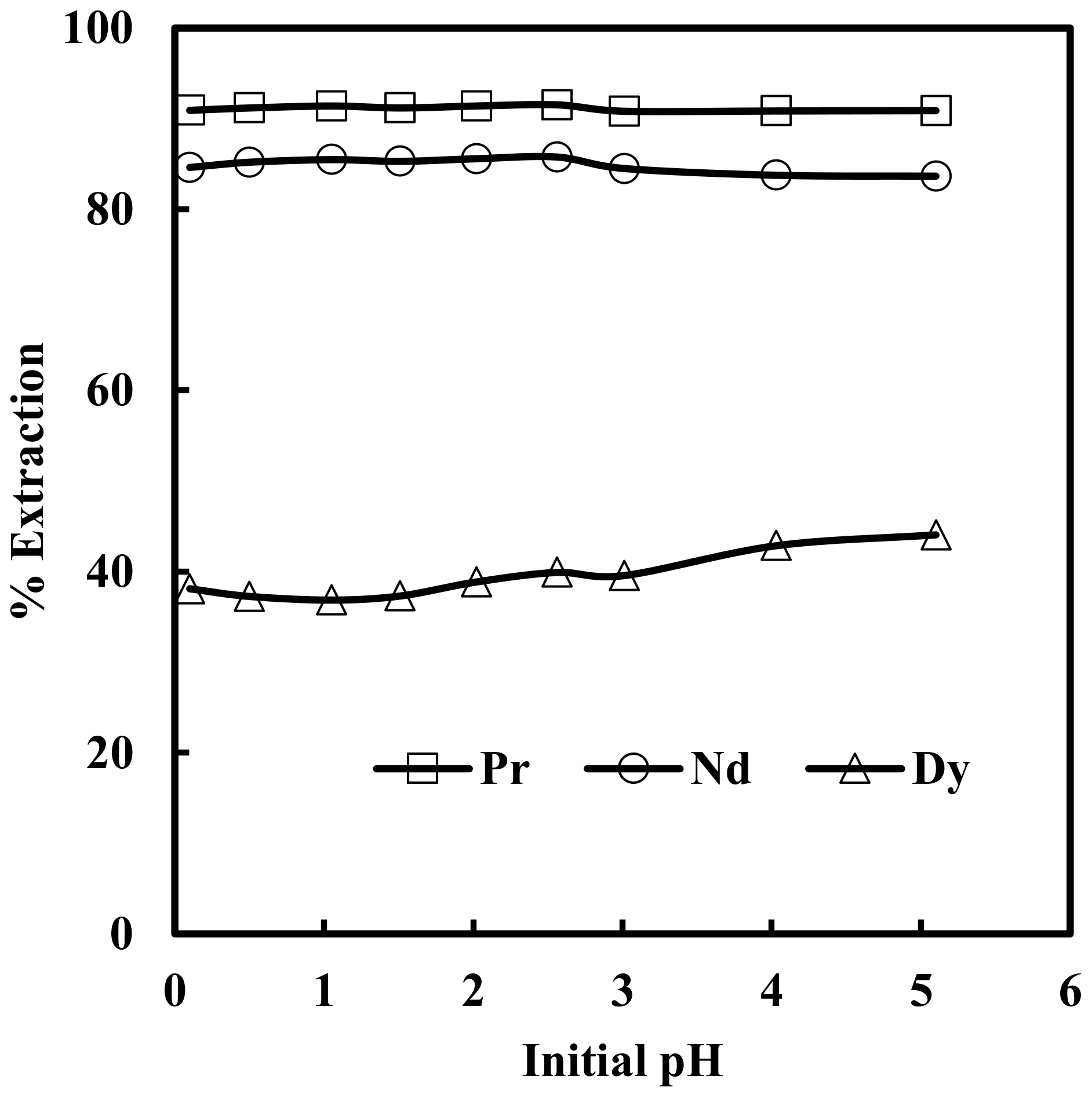
3.1. Effect of pH on Solvent Extraction of Nd, Pr and Dy
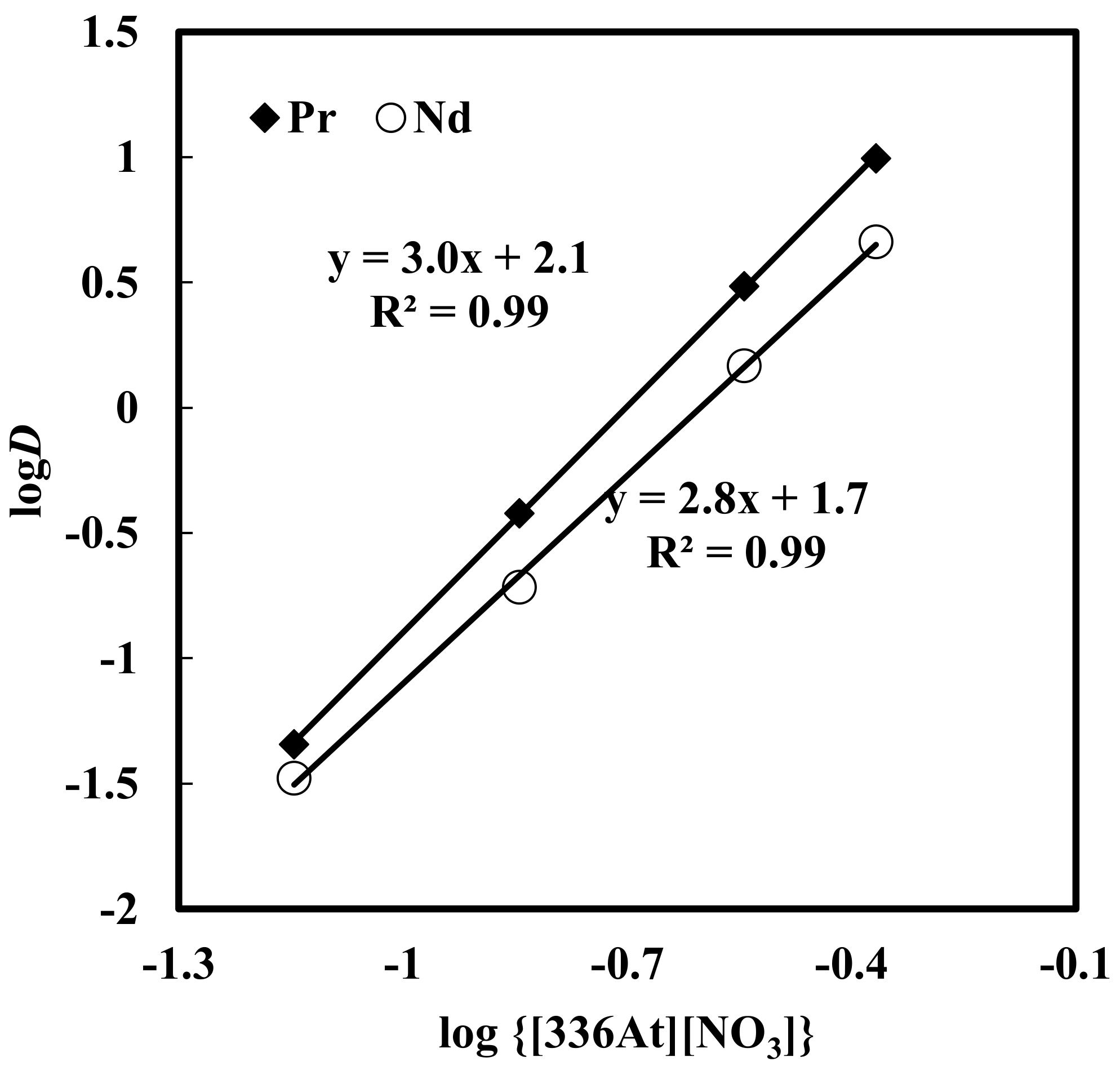
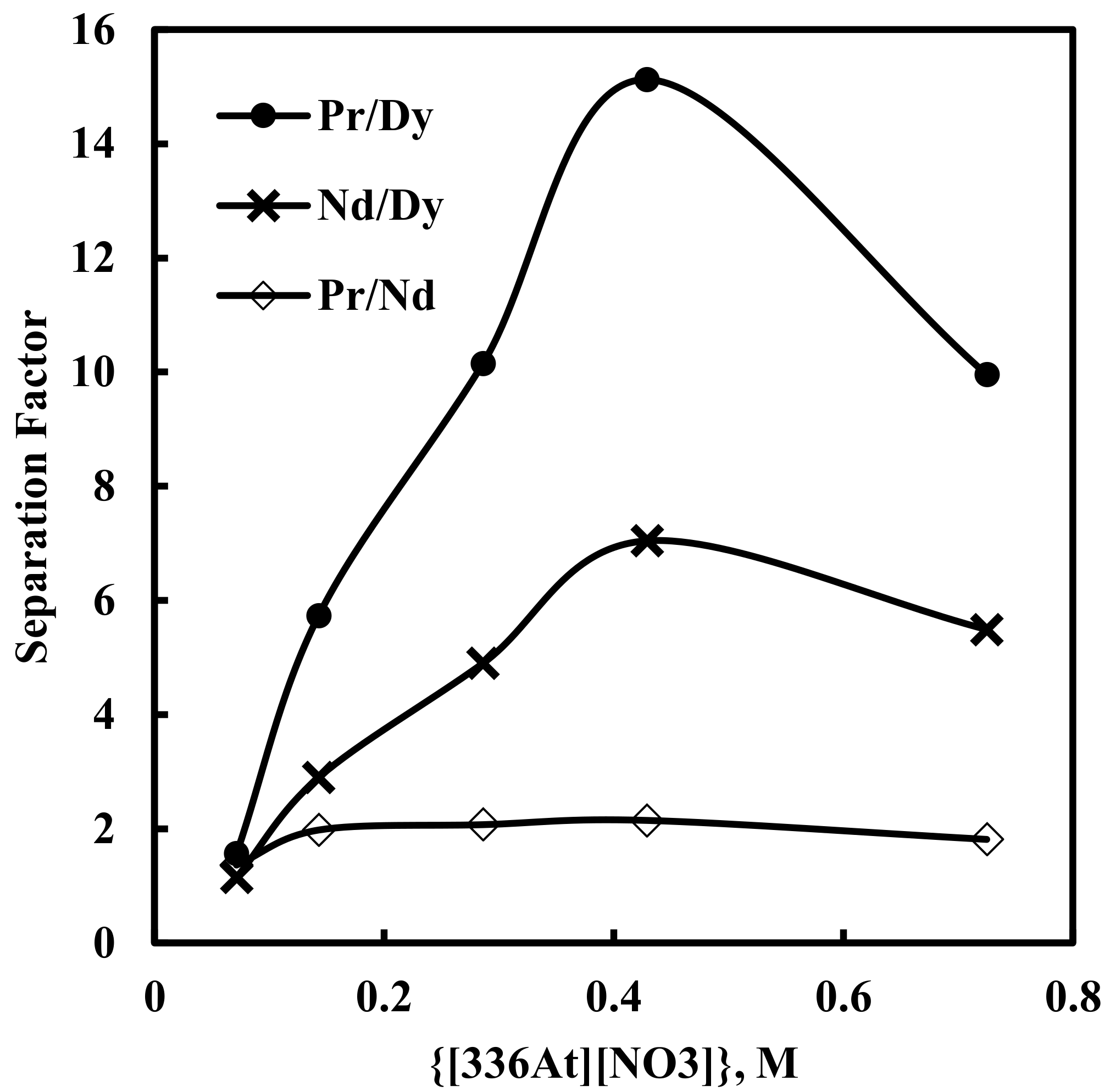
3.2. Effect of [336At][NO3] Concentration on Solvent Extraction of Nd, Pr and Dy
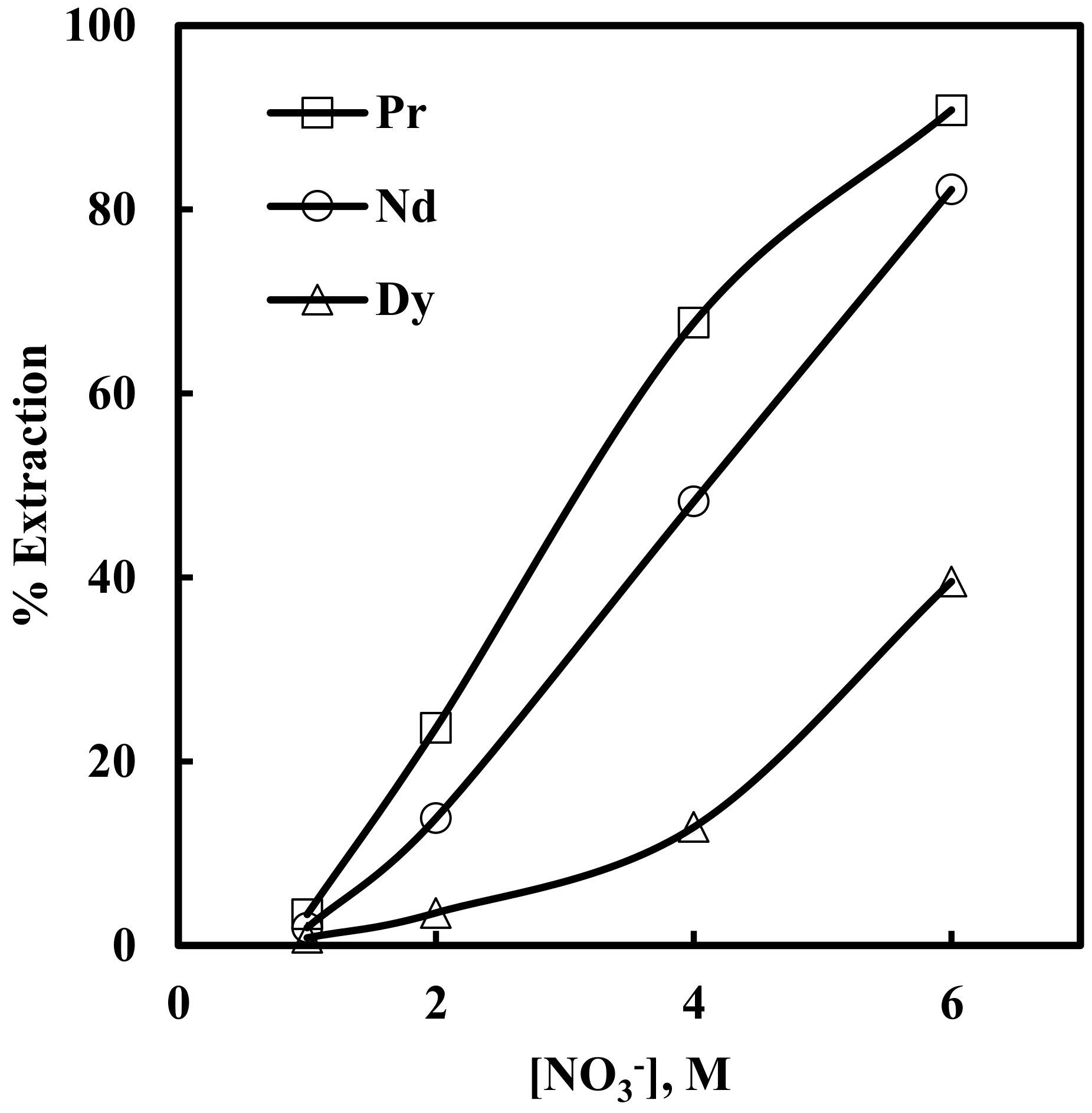
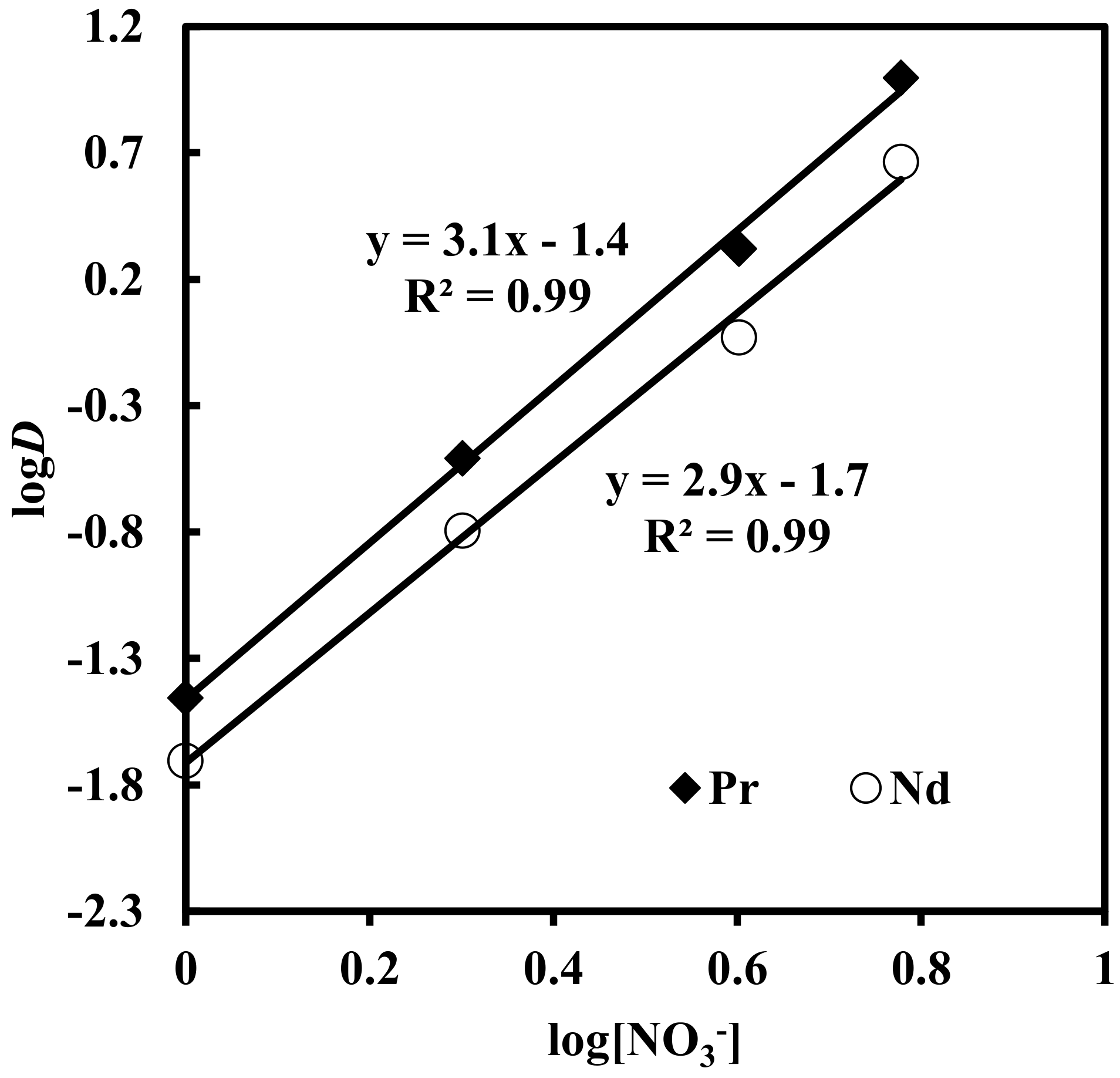
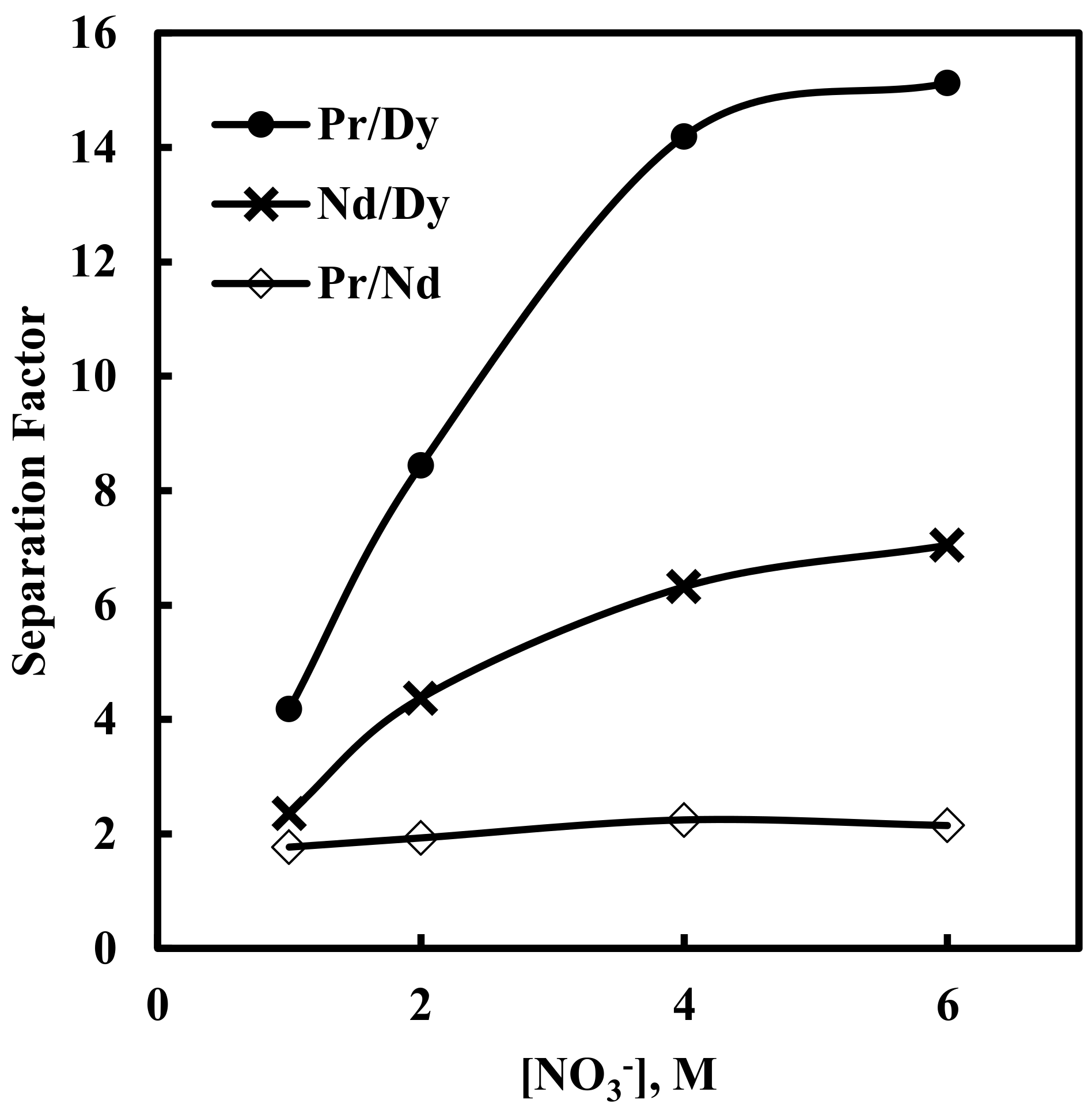
3.3. Effect of Nitrate Concentration on Solvent Extraction of Nd, Pr and Dy
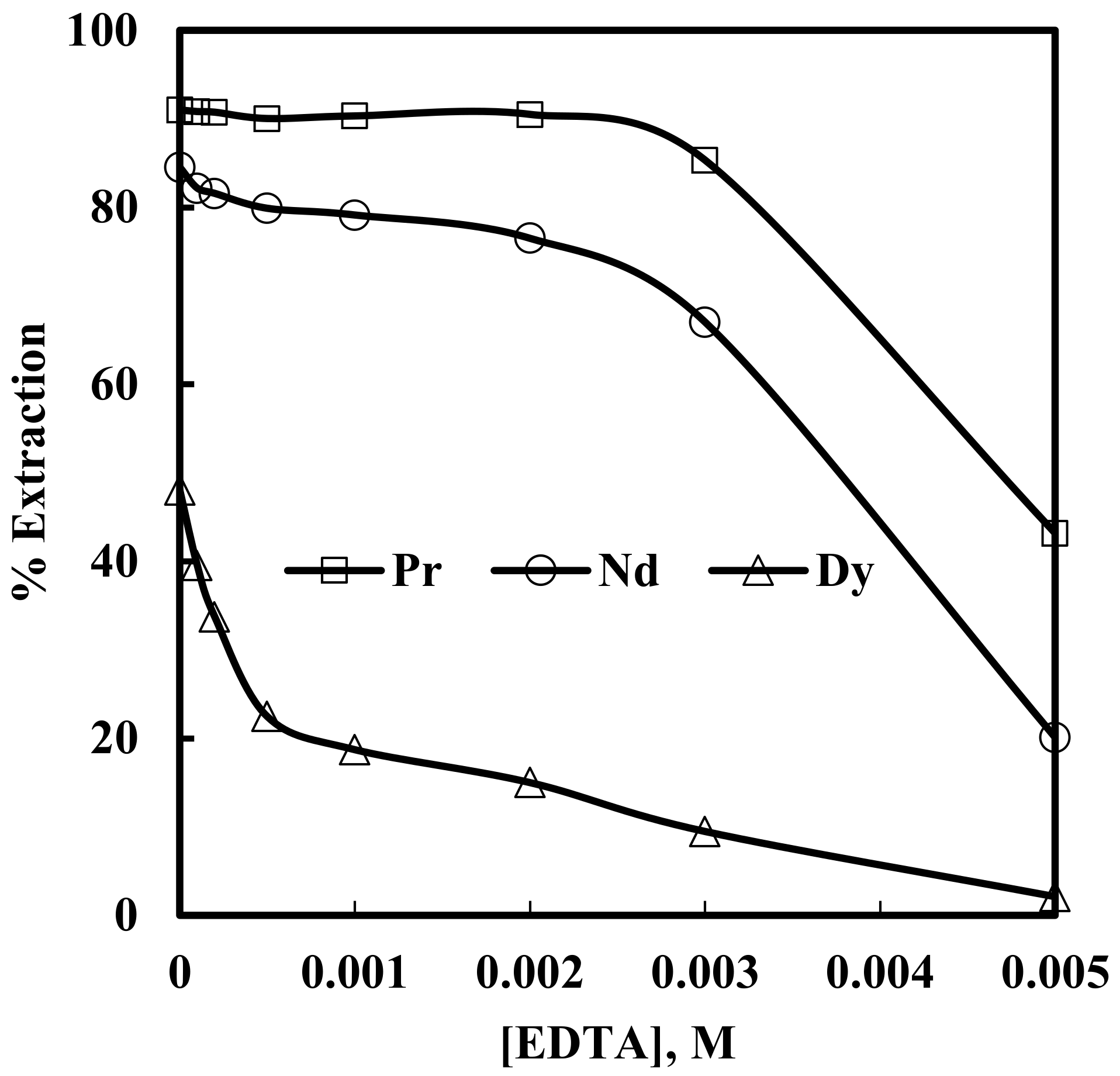
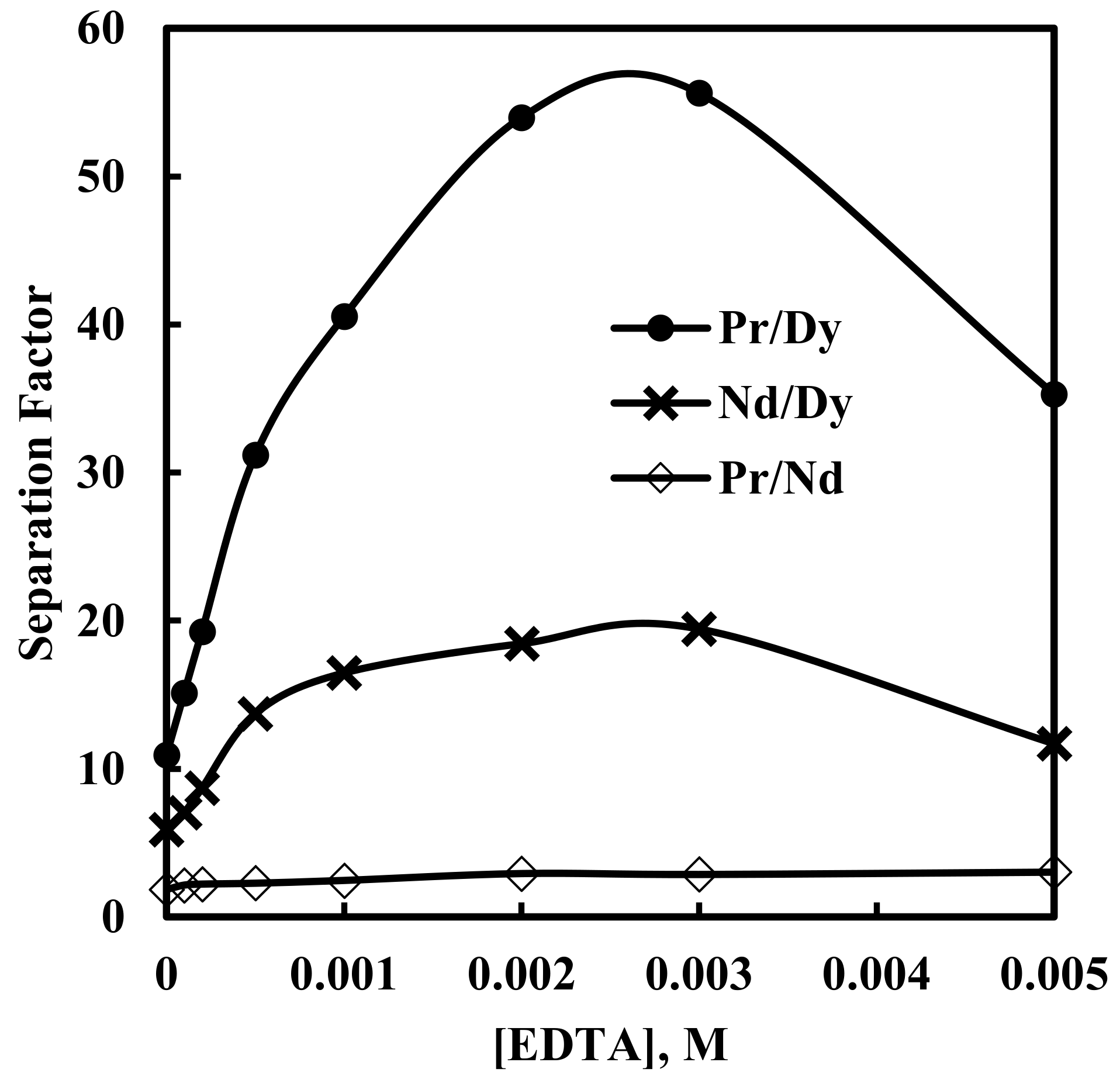
3.4. Effect of EDTA on Solvent Extraction of Nd, Pr and Dy
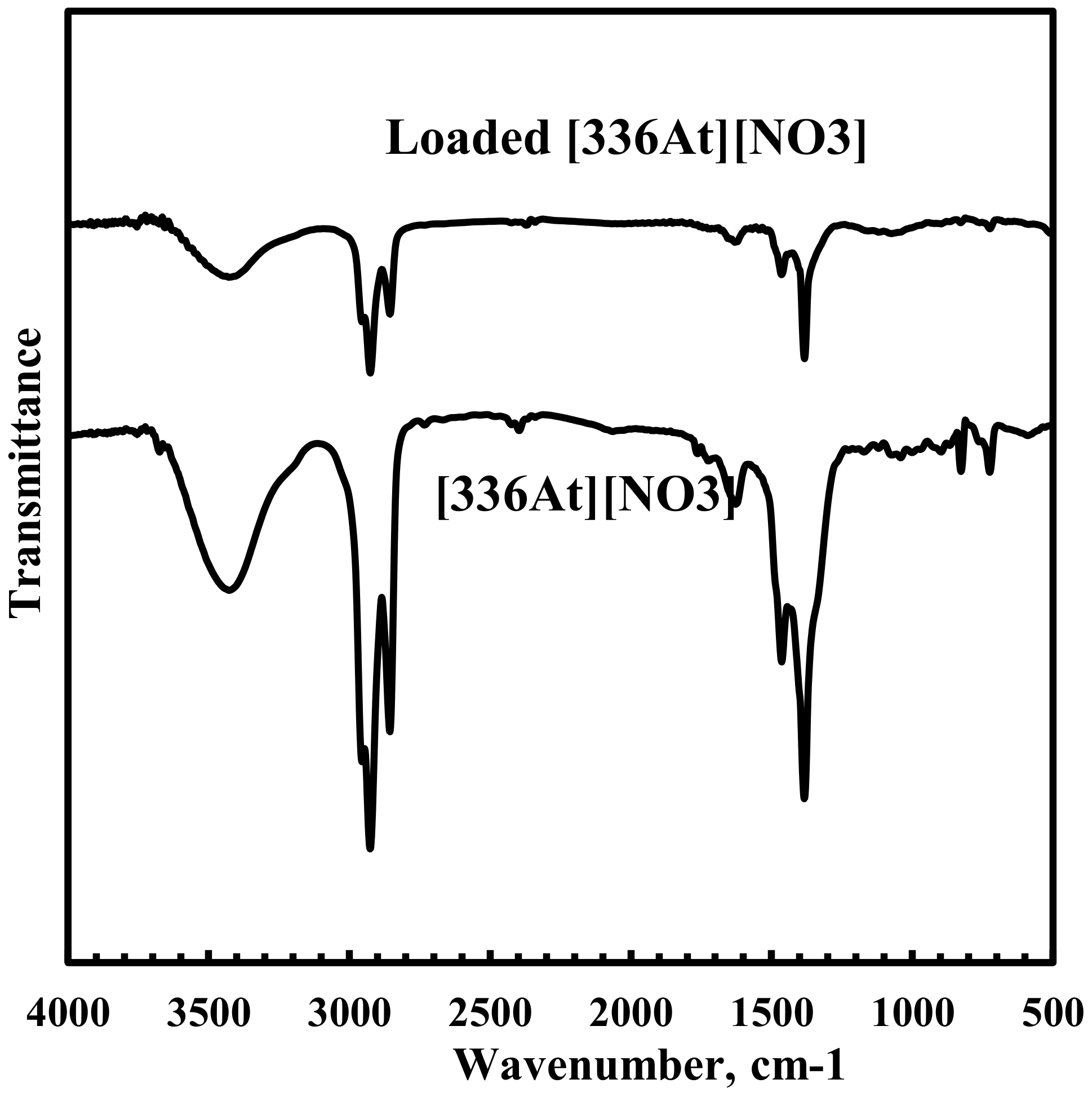
3.5. FTIR Spectra of [336At][NO3] and Nd Loaded [336At][NO3]
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tao, X.; Huiqing, P. Formation cause, composition analysis and comprehensive utilization of rare earth solid wastes. J. Rare Earth 2009, 27, 1096–1102. [Google Scholar]
- Pavel, C.C.; Lacal-Arántegui, R.; Marmier, A.; Schüler, D.; Tzimas, E.; Buchert, M.; Blagoeva, D. Substitution strategies for reducing the use of rare earths in wind turbines. Resour. Policy 2017, 52, 349–357. [Google Scholar] [CrossRef]
- Yang, Y.; Walton, A.; Sheridan, R.; Guth, K.; Gauß, R.; Gutfleisch, O.; Binnemans, K. REE recovery from end-of-life NdFeB permanent magnet scrap: A critical review. J. Sustain. Metall. 2017, 3, 122–149. [Google Scholar] [CrossRef]
- Sun, M.; Hu, X.; Peng, L.; Fu, P.; Ding, W.; Peng, Y. On the production of Mg-Nd master alloy from NdFeB magnet scraps. J. Mater. Proc. 2015, 218, 57–61. [Google Scholar] [CrossRef]
- Abrahami, S.T.; Xiao, Y.; Yang, Y. Rare-earth elements recovery from postconsumer hard-disc drives. Miner. Proces. Extr. Met. 2015, 124, 106–115. [Google Scholar] [CrossRef]
- Itoh, M.; Miura, K.; Machida, K.I. Novel rare earth recovery process on Nd-Fe-B magnet scrap by selective chlorination using NH4Cl. J. Alloy. Compd. 2009, 477, 484–487. [Google Scholar] [CrossRef]
- Banda, R.; Seok, H.; Seung, M. Hydrometallurgy Solvent extraction separation of La from chloride solution containing Pr and Nd with Cyanex 272. Hydrometallurgy 2012, 121–124, 74–80. [Google Scholar] [CrossRef]
- Singh, D.K.; Kotekar, M.K.; Singh, H. Development of a solvent extraction process for production of nuclear grade dysprosium oxide from a crude concentrate. Desalination 2008, 232, 49–58. [Google Scholar] [CrossRef]
- Parhi, P.K.; Sethy, T.R.; Rout, P.C.; Sarangi, K. Separation and recovery of neodymium and praseodymium from permanent magnet scrap through the hydrometallurgical route. Sep. Sci. Technol. 2016, 51, 2232–2241. [Google Scholar] [CrossRef]
- Yoon, H.S.; Kim, C.J.; Chung, K.W.; Kim, S.D.; Lee, J.Y.; Kumar, J.R. Solvent extraction, separation and recovery of dysprosium (Dy) and neodymium (Nd) from aqueous solutions: Waste recycling strategies for permanent magnet processing. Hydrometallurgy 2016, 165, 27–43. [Google Scholar] [CrossRef]
- Lee, M.S.; Lee, J.Y.; Kim, J.S.; Lee, G.S. Solvent extraction of neodymium ions from hydrochloric acid solution using PC88A and saponified PC88A. Sep. Puri. Tech. 2005, 46, 72–78. [Google Scholar] [CrossRef]
- Gupta, C.K.; Krishnamurthy, N. Extractive Metallurgy of Rare Earths; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Kumari, A.; Sinha, M.K.; Sahu, S.K.; Pandey, B.D. Solvent extraction and separation of trivalent lanthanides using cyphos IL 104, a novel phosphonium Ionic liquid as extractant. Solvent Extr. Ion Exc. 2016, 34, 469–484. [Google Scholar] [CrossRef]
- Liu, Y.; Jeon, H.S.; Lee, M.S. Solvent extraction of Pr and Nd from chloride solution by the mixtures of Cyanex 272 and amine extractants. Hydrometallurgy 2014, 150, 61–67. [Google Scholar] [CrossRef]
- Padhan, E.; Sarangi, K. Recovery of Nd and Pr from NdFeB magnet leachates with bi-functional ionic liquids based on Aliquat 336 and Cyanex 272. Hydrometallurgy 2017, 167, 134–140. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Matsumiya, M.; Kawakami, S. Extraction of rare earth ions from Nd–Fe–B magnet wastes with TBP in tri-capryl-methyl-ammonium nitrate. Solvent Extr. Res. Dev. 2014, 21, 137–145. [Google Scholar] [CrossRef]
- Riano, S.; Binnemans, K. Extraction and separation of neodymium and dysprosium from used NdFeB magnets: An application of ionic liquids in solvent extraction towards the recycling of magnets. Green Chem. 2015, 17, 2931–2942. [Google Scholar] [CrossRef]
- Rout, A.; Binnemans, K. Influence of the ionic liquid cation on the solvent extraction of trivalent rare-earth ions by mixtures of Cyanex 923 and ionic liquids. Dalton Trans. 2015, 44, 1379–1387. [Google Scholar] [CrossRef] [PubMed]
- Vander Hoogerstraete, T.; Onghena, B.; Binnemans, K. Homogeneous liquid-liquid extraction of rare earths with the betaine-betainium bis(trifluoromethylsulfonyl)imide ionic liquid system. Int. J. Mol. Sci. 2013, 14, 21353–21377. [Google Scholar] [CrossRef] [PubMed]
- Baba, Y.; Kubota, F.; Kamiya, N.; Goto, M. Selective recovery of dysprosium and neodymium ions by a supported liquid membrane based on ionic liquids. Solvent Extr. Res. Dev. 2011, 18, 193–198. [Google Scholar] [CrossRef]
- Kashi, E.; Habibpour, R.; Gorzin, H.; Maleki, A. Solvent extraction and separation of light rare earth elements (La, Pr and Nd) in the presence of lactic acid as a complexing agent by Cyanex 272 in kerosene and the effect of citric acid, acetic acid and Titriplex III as auxiliary agents. J. Rare Earth 2018, 36, 317–323. [Google Scholar] [CrossRef]
- Sun, X.; Wang, Y.; Li, D. Selective separation of yttrium by CA-100 in the presence of a complexing agent. J. Alloy Compd. 2006, 408–412, 999–1002. [Google Scholar] [CrossRef]
- Matsuyama, H.; Azis, A.; Fujita, M.; Teramoto, M. Enhancement in extraction rates by addition of organic acids to aqueous phase in solvent extraction of rare earth metals in presence of diethylenetriamine pentacetic acid. J. Chem. Eng. Japan 1995, 29, 126–133. [Google Scholar] [CrossRef]
- Banda, R.; Jeon, H.S.; Lee, M.S. Extraction behavior of REEs (La, Pr, and Nd) in the presence of EDTA from chloride solutions. Korean J. Met. Mater. 2013, 51, 421–427. [Google Scholar] [CrossRef]
- Yin, S.; Wu, W.; Bian, X.; Luo, Y.; Zhang, F. Solvent Extraction of La(III) from Chloride Medium in the Presence of Two Water Soluble Complexing Agents with di-(2-ethylhexyl) Phosphoric Acid. Ind. Eng. Chem. Res. 2013, 52, 8558–8564. [Google Scholar] [CrossRef]
- Yin, S.; Wu, W.; Zhang, B.; Zhang, F.; Luo, Y.; Li, S.; Bian, X. Study on separation technology of Pr and Nd in D2EHPA-HCl-LA coordination extraction system. J. Rare Earth 2010, 28, 111–115. [Google Scholar] [CrossRef]
- Yin, S.; Li, S.; Wu, W.; Bian, X.; Peng, J.; Zhang, Li. Extraction and separation of Ce(III) and Pr(III) in the system containing two complexing agents with di-(2-ethylhexyl) phosphoric acid. RSC Adv. 2014, 4, 59997–60001. [Google Scholar] [CrossRef]
- Rout, A.; Binnemans, K. Separation of rare earths from transition metals by liquid-liquid extraction from a molten salt hydrate to an ionic liquid phase. Dalton Trans. 2014, 43, 3186–3195. [Google Scholar] [CrossRef] [PubMed]
- Rout, A.; Binnemans, K. Solvent extraction of neodymium(III) by functionalized ionic liquid tri-octyl-methyl-ammonium dioctyl diglycolamate in fluorine-free ionic liquid diluent. Ind. Eng. Chem. Res. 2014, 53, 6500–6508. [Google Scholar] [CrossRef]
- Cerna, M.; Volaufova, E.; Rod, V. Extraction of light rare earth elements by amines at high inorganic nitrate concentration. Hydrometallurgy 1992, 28, 339–352. [Google Scholar] [CrossRef]
- Komasawa, I.; Hisada, K.; Miyamura, M. Extraction and separation of rare-earth elements by tri-octylmethylammonium nitrate. J. Chem. Eng. Japan 1990, 23, 308–315. [Google Scholar] [CrossRef]
- El-Kot, A.M. Solvent extraction of neodymium, europium and thulium by di-(2- ethylhexyl) phosphoric acid. J. Radioanal. Nucl. Chem. 1993, 170, 207–214. [Google Scholar] [CrossRef]
- Huang, Y.; Tanaka, M. Solvent extraction equilibrium of dysprosium(III) from nitric acid solutions with 2-ethylhexylphosphonic acid mono-2-ethylhexyl ester. Trans. Nonferrous Met. Soc. China 2010, 20, 707–711. [Google Scholar] [CrossRef]
- Thakur, N.V.; Jayawant, D.V.; Iyer, N.S.; Koppiker, K.S. Separation of neodymium from lighter rare earths using alkyl phosphonic acid, PC 88A. Hydrometallurgy 1993, 34, 99–108. [Google Scholar] [CrossRef]
- Preston, J.S. The recovery of rare earth oxides from a phosphoric acid byproduct. Part 4. The preparation of magnet-grade neodymium oxide from the light rare earth fraction. Hydrometallurgy 1996, 42, 15–167. [Google Scholar] [CrossRef]
- Panda, N.; Devi, N.; Mishra, S. Solvent extraction of neodymium(III) from acidic nitrate medium using Cyanex 921 in kerosene. J. Rare Earth 2012, 30, 794–797. [Google Scholar] [CrossRef]
- Vander Hoogerstraete, T.; Wellens, S.; Verachtert, K.; Binnemans, K. Removal of transition metals from rare earths by solvent extraction with an undiluted phosphonium ionic liquid: Separations relevant to rare-earth magnet recycling. Green Chem. 2013, 15, 919. [Google Scholar] [CrossRef]
- Rout, A.; Kotlarska, J.; Dehaen, W.; Binnemans, K. Liquid-liquid extraction of neodymium(III) by dialkylphosphate ionic liquids from acidic medium: The importance of the ionic liquid cation. Phys. Chem. Chem. Phys. 2013, 15, 16533–16541. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M. Extractive separation of praseodymium and neodymium by di(2-ethylhexyl) phosphoric acid in the presence of water-soluble complexing agents using an electrostatic liquid-liquid contactor. Chem. Eng. Res. Des. 1997, 75, 447–452. [Google Scholar] [CrossRef]
- Lu, D.; Horng, J.S.; Hoh, Y.C. The separation of neodymium by quaternary amine from didymium nitrate solution. J. Alloy Compd. 1989, 149, 219–224. [Google Scholar] [CrossRef]
- Larson, K.; Binnemans, K. Separation of rare earths by solvent extraction with an undiluted nitrate ionic liquid. J. Sustain. Metall. 2017, 3, 73–78. [Google Scholar] [CrossRef]
- Riano, S.; Petranikova, M.; Onghena, B.; Hoogerstraete, T.V.; Banerjee, D.; Foreman, M.; Ekberg, C.; Binnemans, K. Separation of rare earths and other valuable metals from deep-eutectic solvents: A new alternative forthe recycling of used NdFeB magnets. Rsc. Adv. 2017, 7, 32100–32113. [Google Scholar] [CrossRef]
- Kumari, A.; Sinha, M.K.; Pramanik, S.; Sahu, S.K. Recovery of rare earths from spent NdFeB magnets of wind turbine: Leaching and kinetic aspects. Waste Manag. 2018, 75, 486–498. [Google Scholar] [CrossRef] [PubMed]
- Wheelwright, E.J.; Spedding, F.H. The Use of Chelating Agents in the Separation of the Rare Earth Elements by Ion-Exchange Methods. Ames Laboratory ISC Technical Reports 1955. Available online: https://lib.dr.iastate.edu/cgi/viewcontent.cgi?referer=https://www.google.com/&httpsredir=1&article=1098&context=ameslab_iscreports (accessed on 7 August 2018).

| Rare Earths | Extractant | Diluent | Aqueous Medium | Remarks | Reference |
|---|---|---|---|---|---|
| La, Nd, Pr | Cyanex 272, D2EHPA, PC88A, Cyanex 301 | Escaid 110 | Chloride | Cyanex 272 showed the best extraction efficiency among all extractants. The separation of La, Nd and Pr improved by using saponified extractant. | [7] |
| Y, Dy, Tb, Gd | EHEHPA | Kerosene | Chloride | Dy2O3 were produced with more than 98% recovery. Two concentrates yttrium (>93%, 1st cycle) and terbium (>54%, 2nd cycle) were also produced. | [8] |
| Nd | PC88A and 40% saponified PC88A | Kerosene | Chloride | Distribution co-efficient was increased significantly by using saponified extractant. | [11] |
| La, Ce, Pr, Nd | Saponified PC-88A | Kerosene | Chloride | Nd2O3 of >97% purity were produced using extraction, scrubbing and stripping steps. | [12] |
| Pr and Nd | Mixture of Cyanex 272 and amine extractants (Alamine 336, TOA and TEHA) | Kerosene | Chloride | Equimolar Combination of Cyanex 272 and Alamine 336 showed the best extraction ability and the maximum synergistic effect for Pr and Nd separation. | [14] |
| Nd | [A336] [DGA] | [A336] [NO3] | Nitrate | Extraction of Nd was found better by using [A336][DGA] in [A336][NO3] in comparison to HDGA in [A336][NO3], at pH values > 2 | [29] |
| Nd, Eu, Tm | HDEHP | Benzene | Chloride, sulfate, nitrate | Antagonistic effects were observed the mixtures of HDEHP+TBP and HDEHP+TOPO | [32] |
| Dy | PC88A | Shellsol D70 | Nitrate | The experimental equilibrium data were correlated with chemically based model. | [33] |
| Pr, Nd, La, Ce, Sm | 20% saponified PC88A+iso-decanol | Kerosene | Chloride | Nd2O3 were produced with recovery of >85% having purity >96% using 6 extractions, 18 scrubbing and 6 stripping stages. The separation factor for Nd/Pr was obtained as 1.64. | [34] |
| Nd, Pr, Ce and La | [A336] [NO3] | Shellsol AB | Nitrate | Mini plant trial were carried out with 190 L of feed solution to obtain 95% Nd2O3 using 8 extractions, 6 scrubbing and 6 stripping stages. | [35] |
| Nd | Cyanex 921 | Kerosene | Nitrate | With the aqueous phase containing 0.001 mol/L HNO3 and 0.1 mol/L KNO3, 98% of Nd(III) was extracted using 0.5M extractant. | [36] |
| Pr, Nd, Dy | TBP | [A336][NO3] | Nitrate | By using 1.8M TBP, 95 g/L of Nd loaded in the ionic liquid phase at pH 2. Complete extraction of rare earths (Nd, Pr and Dy) in two contacts. | [16] |
| Nd, Dy, Co | trihexyl(tetradecyl)phosphonium nitrate | - | Nitrate | After separation of Cobalt, Na2EDTA along with NH4NO3 were used for selective stripping of Dy. By precipitation process, Nd2O3, Dy2O3 and CoO were obtained with purities of 99.6%, 99.8% and 99.8%, respectively. | [17] |
| Nd | Cyanex 923 | [C4mim][Tf2N], [N1444][Tf2N] [P66614][Tf2N], [C10mim][Tf2N], [N1888][Tf2N]. | Nitrate | Ionic liquids with hydrophilic cation, [C4mim][Tf2N] and [N1444][Tf2N] extract Nd(III) efficiently by ion-exchange mechanism.Hydrophobic cationextract Nd(III) less efficiently due toits low solubility in the aqueous phase. | [18] |
| Nd | [Hbet][Tf2N] | - | Chloride Nitrate, bis(trifluoromethylsulfonyl)imide | Extraction was possible from chloride, nitrate and bis(trifluoromethylsulfonyl)imide medium with similar distribution ratios. | [19] |
| Nd, Dy, Fe | DODGAA | [C8mim][Tf2N] | Nitrate | DODGAA showed higher selectivity for Dy and Nd compared with Fe through an SLM impregnated with the ionic liquid, [C8mim][Tf2N]. | [20] |
| Nd, Fe | Cyphos IL 101 | - | Chloride | More than 99% of iron selectively extracted in organic phase. Best separation factor of Nd/Fe was obtained as 8×105. | [37] |
| Nd | [C6mim][DEHP], [C6mpyr][DEHP], [N4444][DEHP] | [C6mim][NTf2], [C6mpyr][NTf2], [N1444][NTf2] | Nitrate | The extraction behaviour with [C6mim][DEHP] and [C6mpyr][DEHP] was found to be different in comparison with [N4444][DEHP], particularly for pH in the extraction process. | [38] |
| Rare Earths | Extractant | Diluent | Aqueous Medium | Water-soluble Complexing Agent | Remarks | Reference |
|---|---|---|---|---|---|---|
| La, Pr and Nd | Cyanex 272, PC88A | Escaid 110 | Chloride | EDTA | PC88A has higher extraction efficiency than Cyanex 272. Addition of EDTA in aqueous phase suppressed the extraction of Pr and Nd due to formation of anionic complexes. | [24] |
| La, Ce, Pr, Nd | P2O4 | Kerosene | Chloride | Lactic acid, Citric acid | The separation factors for Ce/La, Pr/La and Nd/La were enhanced for the system containing both complexing agents, i.e., lactic acid and citric acid. | [25] |
| Pr and Nd | D2EHPA | Kerosene | Chloride | Lactic acid | Distribution ratio and separation factor increased by decreasing acidity and increasing lactic acid concentration. Best separation factor for Nd/Pr achieved was 1.57 at 0.6 M lactic acid concentration and pH 3.5. | [26] |
| Ce and Pr | P2O4 | Kerosene | Chloride | Lactic acid Citric acid | The distribution ratio increased with increase in ratio of citric acid: lactic acid. The maximum separation factor of 5.78 was achieved. | [27] |
| Pr and Nd | D2EHPA | ISOPAR M | Nitrate | EDTA, DTPA | The extraction degree of Nd and Pr in DTPA is higher than EDTA. Effects of mixtures of EDTA and DTPA on the extraction were also evaluated. Studies carried out in electrostatic liquid-liquid contactor. The best separation factor was achieved as 2. | [39] |
| Nd, Pr (didymium solution) | [A336] [NO3] | naphtha-100 | Nitrate | DTPA | DTPA forms 1:1 complex with rare earth ions, which helps in the separation of Nd-Pr. In 45 stage, 95% recovery of Nd was obtained with purity > 99%. | [40] |
| Light and Heavy Rare Earths | [A336][NO3] | - | Nitrate | EDTA | The addition of 0.2 M EDTA in 11 M nitrate aqueous solution enhanced the separation possibility between light and heavy rare earths as chelating agent forms stable complexes with Heavy rare earths. | [41] |
| Heavy Rare Earths | CA-100 | - | Chloride | EDTA | Selective separation of Yttrium from impurtites (Ho, Er, Tm, Yb, Lu due to formation of EDTA-Yttrium complex. | [22] |
| Nd, Dy, Fe, Co | [A336][SCN] | toluene | Chloride | EDTA | EDTA was used as stripping agent for the separation of Fe and Co from the deep eutectic solvent containing Nd, Dy, Fe, B and Co. | [42] |
| Properties | Mextral® 336At or [336At][Cl] | [336At][NO3] |
|---|---|---|
| IUPAC name | Trioctyl-methyl-ammonium chloride (C25H54NCl) | Trioctyl-methyl-ammonium nitrate (C25H54N2O3) |
| Appearance | Yellow cream | Yellow liquid |
| Molecular weight | 404.1 g/mol | 430.7 |
| Density | 0.88–0.89 g/mL | 0.90–0.95 g/mL |
| Structure | 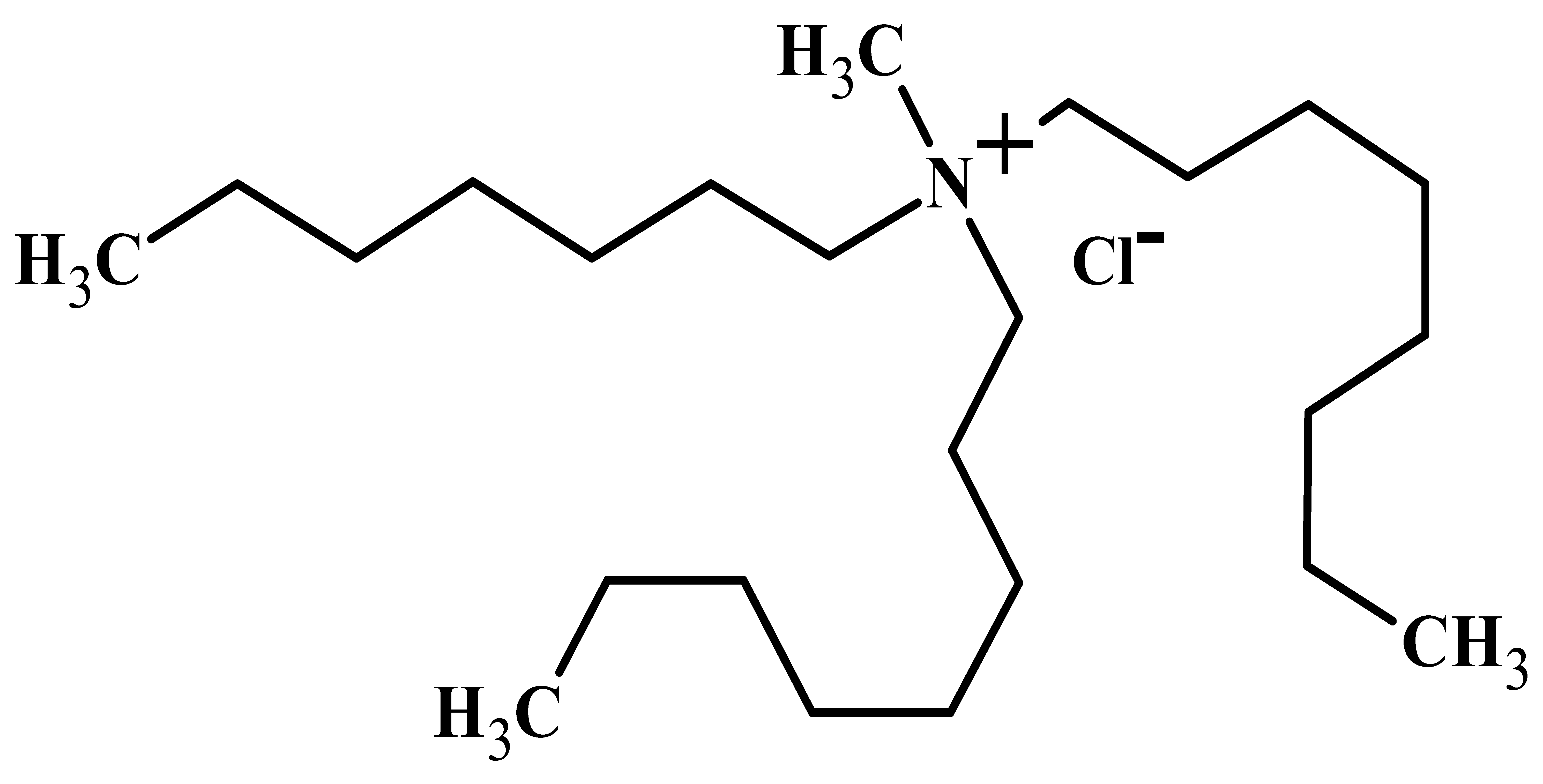 | 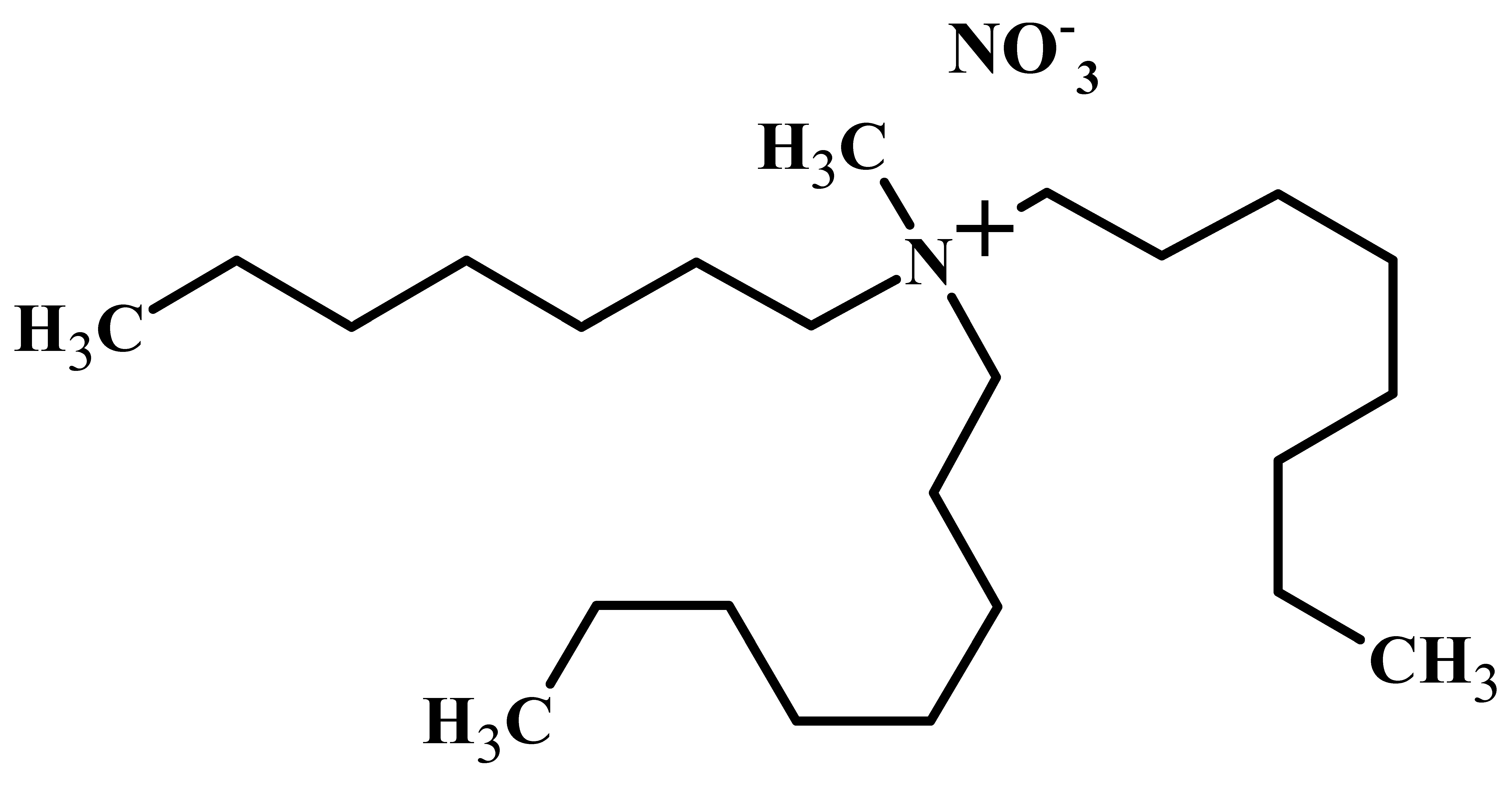 |
| In Aqueous Solution | % Extraction | Separation Factor (β) | ||||
|---|---|---|---|---|---|---|
| Pr | Nd | Dy | Pr/Dy | Nd/Dy | Pr/Nd | |
| Without adding EDTA | 91 | 84.5 | 48 | 10.9 | 5.9 | 1.8 |
| 0.003M EDTA | 85 | 67 | 9.5 | 55.6 | 19.4 | 2.9 |
| Separation Studies | Separation Factor for Pr and Nd | Reference |
|---|---|---|
| 0.43 M [A336][NO3] in kerosene from 6 M NaNO3 using 0.003 M EDTA as chelating agent | 2.9 | (present investigation) |
| 0.5 M [A336][NO3] in xylene from 4 M NaNO3 aqueous solution | 2.04 | [35] |
| 0.2 M EHEHPA in toluene from 0.4 M NaNO3 aqueous solution | 1.27 | |
| 0.2 M D2EHPA in toluene from 0.4 M NaNO3 aqueous solution | 1.25 | |
| 1 M D2EHPA in kerosene from aqueous solution of pH 3.5 using 0.6 M Lactic acid as chelating agent | 1.57 | [26] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumari, A.; Sahu, K.K.; Sahu, S.K. Solvent Extraction and Separation of Nd, Pr and Dy from Leach Liquor of Waste NdFeB Magnet Using the Nitrate Form of Mextral® 336At in the Presence of Aquo-Complexing Agent EDTA. Metals 2019, 9, 269. https://doi.org/10.3390/met9020269
Kumari A, Sahu KK, Sahu SK. Solvent Extraction and Separation of Nd, Pr and Dy from Leach Liquor of Waste NdFeB Magnet Using the Nitrate Form of Mextral® 336At in the Presence of Aquo-Complexing Agent EDTA. Metals. 2019; 9(2):269. https://doi.org/10.3390/met9020269
Chicago/Turabian StyleKumari, Aarti, Kamala Kanta Sahu, and Sushanta Kumar Sahu. 2019. "Solvent Extraction and Separation of Nd, Pr and Dy from Leach Liquor of Waste NdFeB Magnet Using the Nitrate Form of Mextral® 336At in the Presence of Aquo-Complexing Agent EDTA" Metals 9, no. 2: 269. https://doi.org/10.3390/met9020269
APA StyleKumari, A., Sahu, K. K., & Sahu, S. K. (2019). Solvent Extraction and Separation of Nd, Pr and Dy from Leach Liquor of Waste NdFeB Magnet Using the Nitrate Form of Mextral® 336At in the Presence of Aquo-Complexing Agent EDTA. Metals, 9(2), 269. https://doi.org/10.3390/met9020269




