Microstructure, Microhardness, and Wear Properties of Cobalt Alloy Electrodes Coated with TiO2 Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
3. Results
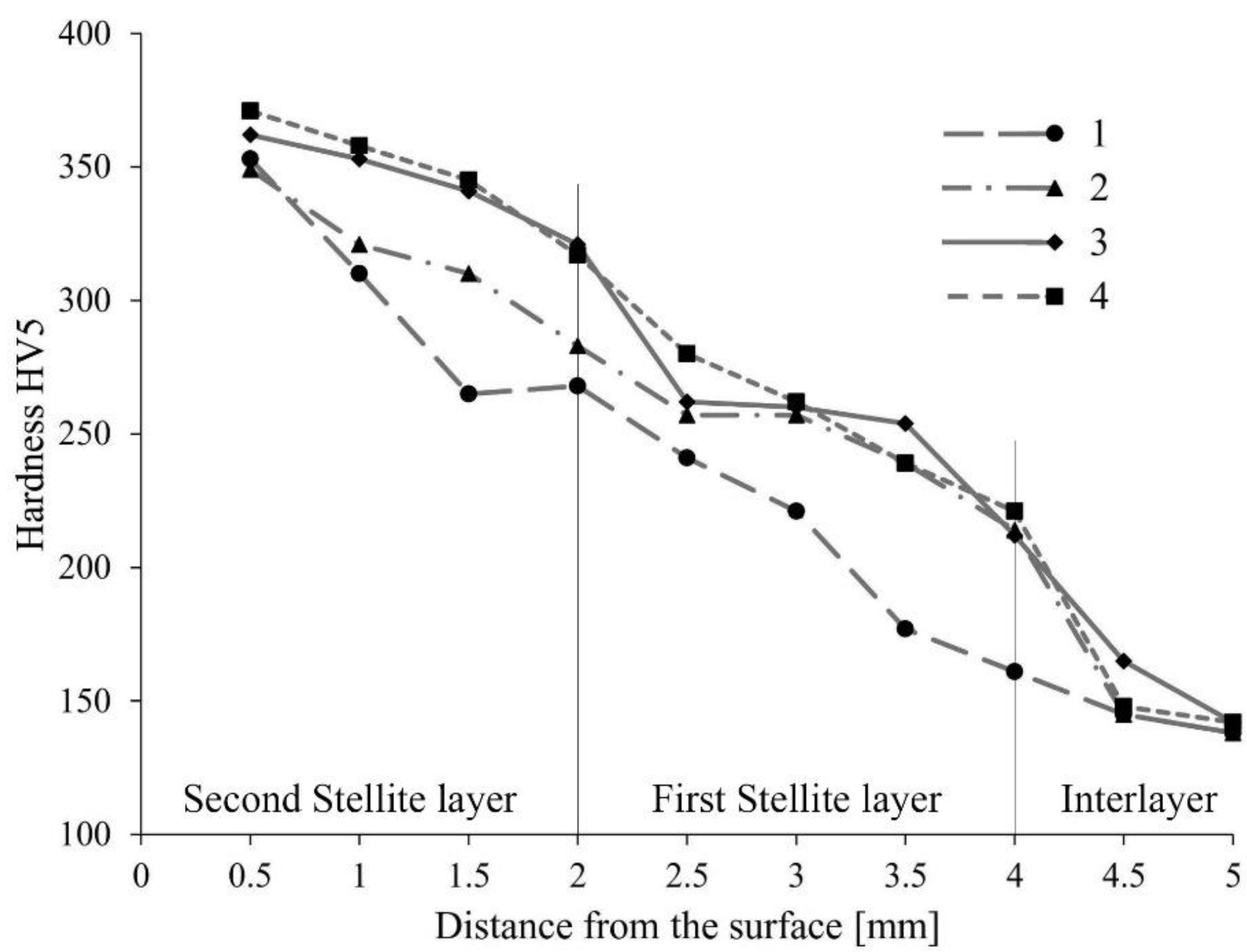
3.1. Hardness Profiles
3.2. Wear
3.3. Microstructure
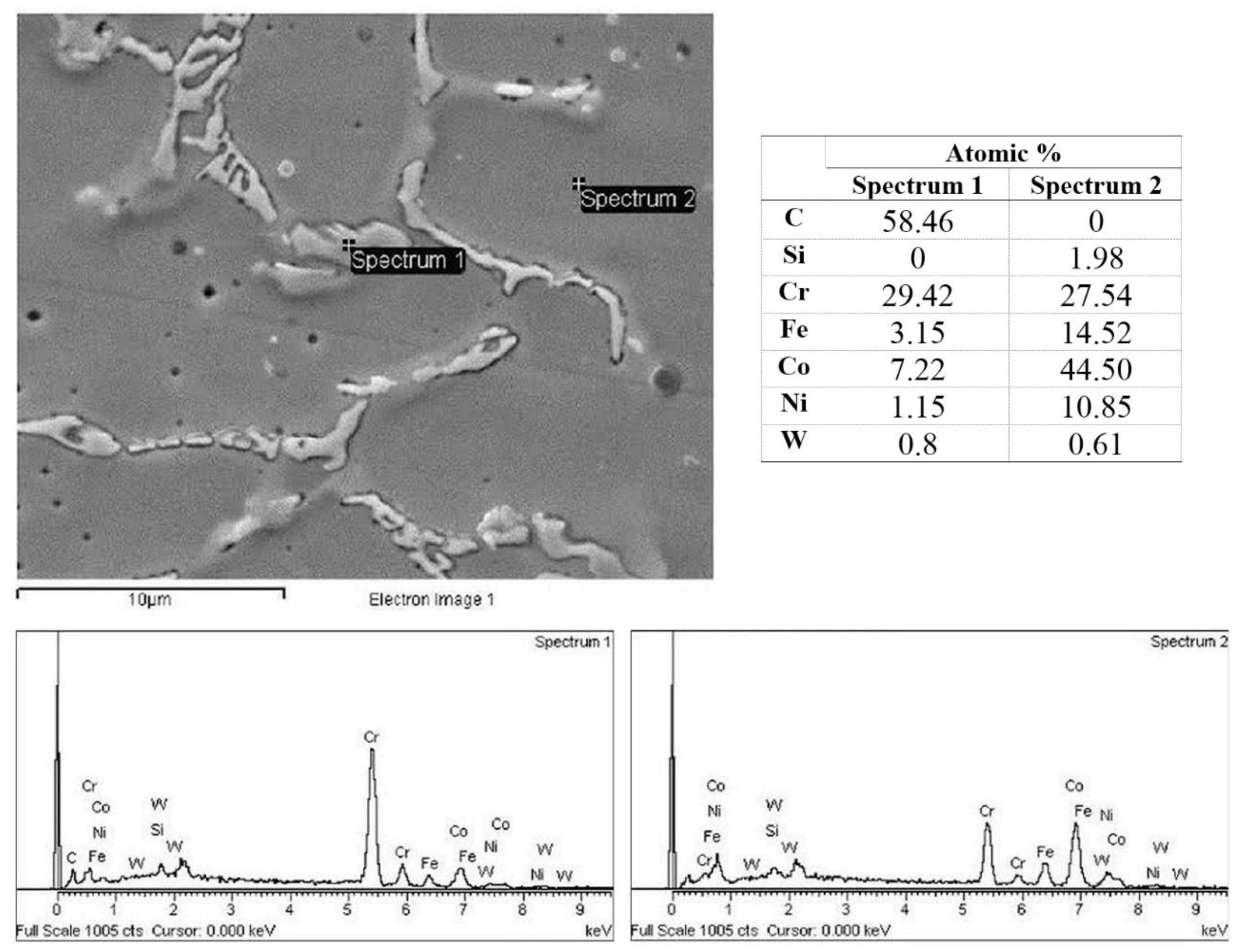
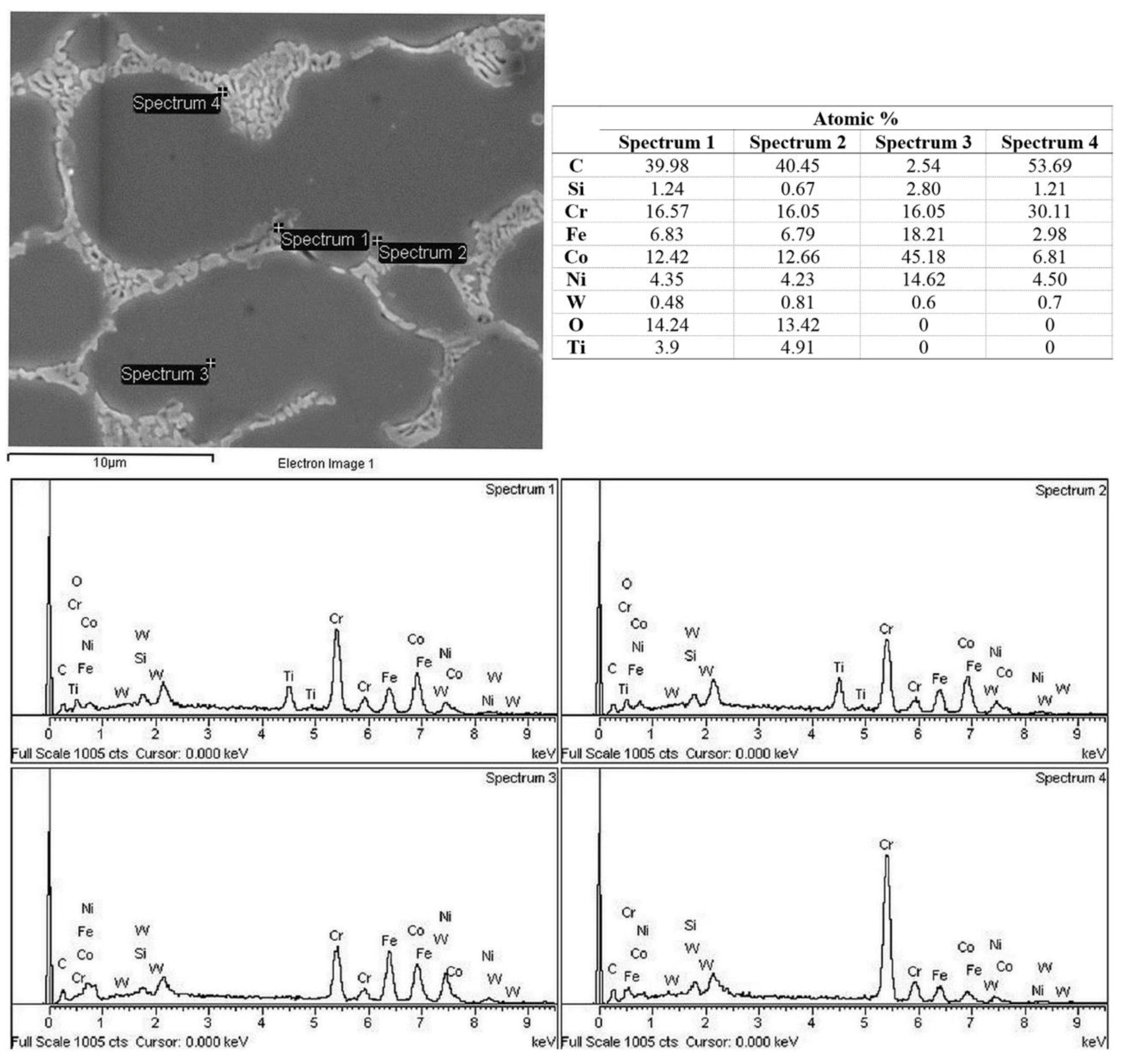
3.4. EDS Analysis
4. Discussion
5. Conclusions
- The hardness values of hard-faced layers obtained with TiO2 nanoparticle coating on the SMAW electrode are higher than those of the layers obtained with untreated electrodes.
- The drop of hardness towards the depth of the hard-faced layer is less pronounced in layers obtained with nanoparticle coated electrodes.
- The wear resistance of layers obtained with nanoparticle coatings is increased compared to specimens obtained with unmodified electrodes.
- The highest wear resistance was obtained with electrodes immersed for the longest period of time, 10 min. This effect can be observed for all tested abrasive grain sizes (P240–P500) and loadings (0.7–1.3 kg). The most pronounced increase in wear resistance was when coarser grains at higher loadings were applied.
- Several effects influence the increase in wear resistance: the formation of blocky titanium-oxides, the rise in carbide and oxide volume fraction, and the refinement of dendrites. The dendrite network becomes almost completely closed which can also be one of the effects beneficial for the increase in wear.
- The increased wear resistance of specimens obtained with modified electrodes influence the prolonged life of the component, which can provide significant savings in CRMs such as cobalt, tungsten, molybdenium, and silicon metal, present in the hard-faced alloy.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haynes, E. Metal Alloy. U.S. Patent No. 873,745, 17 December 1907. [Google Scholar]
- Drapier, J.M.; Davin, A.; Magnee, A.; Coutsouradis, D.; Habraken, L. Abrasion and corrosion resistant cobalt base alloys for hardfacing. Wear 1975, 33, 271–282. [Google Scholar] [CrossRef]
- Wood, P.D.; Evans, H.E.; Ponton, C.B. Investigation into the wear behaviour of Stellite 6 during rotation as an unlubricated bearing at 600 °C. Tribol. Int. 2011, 44, 1589–1597. [Google Scholar] [CrossRef]
- Gholipour, A.; Shamanian, M.; Ashrafizadeh, F. Microstructure and wear behaviour of Stellite 6 cladding on 174 PH stainless steel. J. Alloys Compd. 2011, 509, 4905–4909. [Google Scholar] [CrossRef]
- Da Silva, W.S.; Souza, R.M.; Mello, J.D.B.; Goldenstein, H. Room temperature mechanical properties and tribology of NICRALC and Stellite casting alloys. Wear 2011, 271, 1819–1827. [Google Scholar] [CrossRef]
- Berns, H. Microstructural properties of wear-resistant alloys. Wear 1995, 181, 271–279. [Google Scholar] [CrossRef]
- Yao, M.X.; Wu, J.B.C.; Xu, W.; Liu, R. Metallographic study and wear resistance of a high-C wrought Co-based alloy Stellite 706K. Mater. Sci. Eng. A Struct. 2005, 407, 291–298. [Google Scholar] [CrossRef]
- Davis, J.R. Nickel, Cobalt, Their Alloys; ASM International: Cleveland, OH, USA, 2001. [Google Scholar]
- Exocor Data Sheet. Available online: http://exocor.com (accessed on 10 August 2019).
- Kennametal Data Sheet. Available online: https://kennametal.com (accessed on 10 August 2019).
- Deloro Stellite 6 Data Sheet. Available online: http://www.deloro.com (accessed on 14 February 2017).
- Yao, M.X.; Wu, J.B.C.; Xie, Y. Wear, corrosion and cracking resistance of some W- or Mo-containing Stellite hardfacing alloys. Mater. Sci. Eng. A Struct. 2005, 407, 234–244. [Google Scholar] [CrossRef]
- Marques, F.P.; Bozzi, A.C.; Scandian, C.; Tschiptschin, A.P. Microabrasion of three experimental cobalt-chromium alloys: Wear rates and wear mechanisms. Wear 2017, 390, 176–183. [Google Scholar] [CrossRef]
- Luisa Grilli, M.; Bellezze, T.; Gamsjäger, E.; Rinaldi, A.; Novak, P.; Balos, S.; Piticescu, R.R.; Letizia Ruello, M. Solutions for critical raw materials under extreme conditions: A review. Materials 2017, 10, 285. [Google Scholar] [CrossRef] [PubMed]
- Report on Critical Raw Materials for EU, Report of the Ad-Hoc Working Group on Defining Critical Raw Materials for EU. May 2014. Available online: http://mima.geus.dk/report-on-critical-raw-materials en.pdf (accessed on 28 August 2019).
- Balos, S.; Sidjanin, L.; Dramicanin, M.; Labus Zlatanovic, D.; Pilic, B.; Jovicic, M. Modification of cellulose and rutile welding electrode coating by infiltrated TiO2 nanoparticles. Met. Mater. Int. 2016, 22, 509–518. [Google Scholar] [CrossRef]
- Balos, S.; Dramicanin, M.; Labus Zlatanovic, D.; Sidjanin, L. SMAW welding aided by TiO2 nano particles. In Proceedings of the 3rd IIW Sout-East European Welding Congress, Timisoara, Romania, 3–5 June 2015; pp. 1–6. [Google Scholar]
- Balos, S.; Dramicanin, M.; Labus Zlatanovic, S.; Sidjanin, L.; Pilic, B. Rutile Electrodes Enhanced with TiO2 nanoparticles. In Advanced Materials Research; Trans Tech Publications: Timisoara, Romania, 2016; pp. 69–74. [Google Scholar]
- Fattahi, M.; Nabhani, N.; Vaezi, M.R.; Rahimi, E. Improvement of impact toughness of AWS E6010 weld metal by adding TiO2 nanoparticles to the electrode coating. Mater. Sci. Eng. A Struct. 2011, 528, 8031–8039. [Google Scholar] [CrossRef]
- Byun, J.-S.; Shim, J.-H.; Suh, J.-Y.; Oh, Y.-J.; Cho, Y.W.; Shim, J.-D.; Lee, D.N. Inoculated acicular ferrite microstructure and mechanical properties. Mater. Sci. Eng. A Struct. 2011, 319, 326–331. [Google Scholar]
- Greenwood, N.; Earnshaw, A. Chemistry of the Elements, 2nd ed.; Butterworth-Heinemann: Woburn, UK, 1997; pp. 961–962. [Google Scholar]
- Hossein Nedjad, S.; Farzaneh, A. Formation of fine intragranular ferrite in cast plain carbon steel inoculated by titanium oxide nanopowder. Scr. Mater. 2007, 57, 937–940. [Google Scholar] [CrossRef]
- Kiviö, M.; Holappa, L.; Iung, T. Addition of dispersoid titanium oxide inclusions in steel and their influence on grain refinement. Metall. Mater. Trans. B 2010, 41, 1194–1204. [Google Scholar] [CrossRef]
- St-Laurent, S.; L’Esperance, G. Effects of chemistry, density and size distribution of inclusions on the nucleation of acicular ferrite of C-Mn steel shielded-metal-arc-welding weldments. Mater. Sci. Eng. A Struct. 1992, 149, 203–216. [Google Scholar] [CrossRef]
- Zhang, D.; Terasaki, H.; Komizo, Y. In situ observation of the formation of intragranular acicular ferrite at non-metallic inclusions in C–Mn steel. Acta Mater. 2010, 58, 1369–1378. [Google Scholar] [CrossRef]
- Byun, J.S.; Shim, J.H.; Cho, Y.W. Influence of Mn on microstructural evolution in Ti-killed C–Mn steel. Scr. Mater. 2003, 48, 449–454. [Google Scholar] [CrossRef]
- Davis, J.R. Alloying: Understanding the Basics; ASM International: Cleveland, OH, USA, 2001. [Google Scholar]
- Smallman, R.; Bishop, R.J. Modern Physical Metallurgy and Materials Engineering, 6th ed.; Butterworth Heinemann: Woburn, UK, 1999. [Google Scholar]
- Sun, N.; Apelian, D. Friction stir processing of aluminum cast alloys for high performance applications. JOM 2011, 63, 44–50. [Google Scholar] [CrossRef]



| Alloy | Co | Cr | W | C | Ni | Mo | Fe | Si | HRC | Properties | Applications |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Stellite 1 | Base | 31 | 12 | 2.45 | <3.0 | <1.0 | <3.0 | <2.0 | 51–56 | Erosion, abrasion resistance, crack sensitive | Valve seat inserts, bearings, cutter edges |
| Stellite 6 | Base | 29 | 4 | 1.2 | <3.0 | <1.0 | <3.0 | <2.0 | 39–43 | General purpose wear, impact, cavitation resistance | Valve seats and gates, pump shafts and bearings, erosion shields, rolling couples |
| Stellite 12 | Base | 30 | 8 | 1.55 | <3.0 | <1.0 | <3.0 | <2.0 | 45–50 | High temperature and corrosion resistance, properties between Stellite 1 and 6 | Control plates, pump vanes, bearing bushes, narrow neck glass mold plungers, hard facing of engine valves, pinch rollers and rotor blade edges |
| Stellite 20 | Base | 32 | 16 | 2.45 | <3.0 | <1.0 | <3.0 | <2.0 | 53–57 | High abrasion, good corrosion resistance and low shock resistance. | Pump sleeves, rotary seal rings, wear pads, bearing sleeves, centerless grinder work rests |
| Stellite 21 | Base | 28 | - | 0.25 | 3 | 5.5 | <3.0 | <1.5 | 27–40 | Thermal, mechanical shock, cavitation resistance, not for abrasion | Building of forging and hot stamping dies |
| Stellite 25 | Base | 20 | 14 | 0.1 | 10 | <1.0 | <3.0 | <1.0 | 20–45 | Metal-on-metal wear, thermal fatigue, hot corrosion | Piercing points, forming tools, extrusion dies, furnace hardware |
| C | Si | Mn | S | Cr | P | Al | Cu | Mo | Ni | Fe |
|---|---|---|---|---|---|---|---|---|---|---|
| 0.09 | 0.14 | 0.02 | 0.041 | 0.001 | 0.011 | 0.006 | 0.39 | 0.011 | 0.07 | balance |
| C | Si | Mn | Cr | Fe | Mo | Nb | Co | Ni |
|---|---|---|---|---|---|---|---|---|
| 0.025 | 0.4 | 5.0 | 19.0 | 3.0 | 1.2 | 2.2 | 0.08 | balance |
| C | Si | Cr | W | Fe | Co |
|---|---|---|---|---|---|
| 1.1 | 1.0 | 28.0 | 4.5 | 3.0 | balance |
| Electrode Core Diameter × Length | Ø 3.2 × 450 mm |
|---|---|
| Current type | DC+ |
| Welding current | 85 A |
| Welding voltage | 22 V |
| Welding speed | 12 cm/min |
| Electrode inclination along welding direction | 60° |
| Specimen | Interdendritic Phase Volume Fraction [%] | ||
|---|---|---|---|
| Surface | 1.5 mm under the Surface | 2.5 mm under the Surface | |
| 1 | 12.0 | 11.5 | 7.9 |
| 2 | 12.3 | 12.0 | 10.3 |
| 3 | 12.5 | 12.1 | 10.1 |
| 4 | 14.0 | 12.4 | 11.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balos, S.; Janjatovic, P.; Dramicanin, M.; Labus Zlatanovic, D.; Pilic, B.; Hanus, P.; Jaworska, L. Microstructure, Microhardness, and Wear Properties of Cobalt Alloy Electrodes Coated with TiO2 Nanoparticles. Metals 2019, 9, 1186. https://doi.org/10.3390/met9111186
Balos S, Janjatovic P, Dramicanin M, Labus Zlatanovic D, Pilic B, Hanus P, Jaworska L. Microstructure, Microhardness, and Wear Properties of Cobalt Alloy Electrodes Coated with TiO2 Nanoparticles. Metals. 2019; 9(11):1186. https://doi.org/10.3390/met9111186
Chicago/Turabian StyleBalos, Sebastian, Petar Janjatovic, Miroslav Dramicanin, Danka Labus Zlatanovic, Branka Pilic, Pavel Hanus, and Lucyna Jaworska. 2019. "Microstructure, Microhardness, and Wear Properties of Cobalt Alloy Electrodes Coated with TiO2 Nanoparticles" Metals 9, no. 11: 1186. https://doi.org/10.3390/met9111186
APA StyleBalos, S., Janjatovic, P., Dramicanin, M., Labus Zlatanovic, D., Pilic, B., Hanus, P., & Jaworska, L. (2019). Microstructure, Microhardness, and Wear Properties of Cobalt Alloy Electrodes Coated with TiO2 Nanoparticles. Metals, 9(11), 1186. https://doi.org/10.3390/met9111186






