Environmentally Friendly and Controllable Pyrolysis Method to Synthesize Ni-Modified Graphene Nanosheets as Reinforcement of Lead-Free Solder
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Segmented Pyrolysis Ni Modification
2.3. Microstructural Characteristic
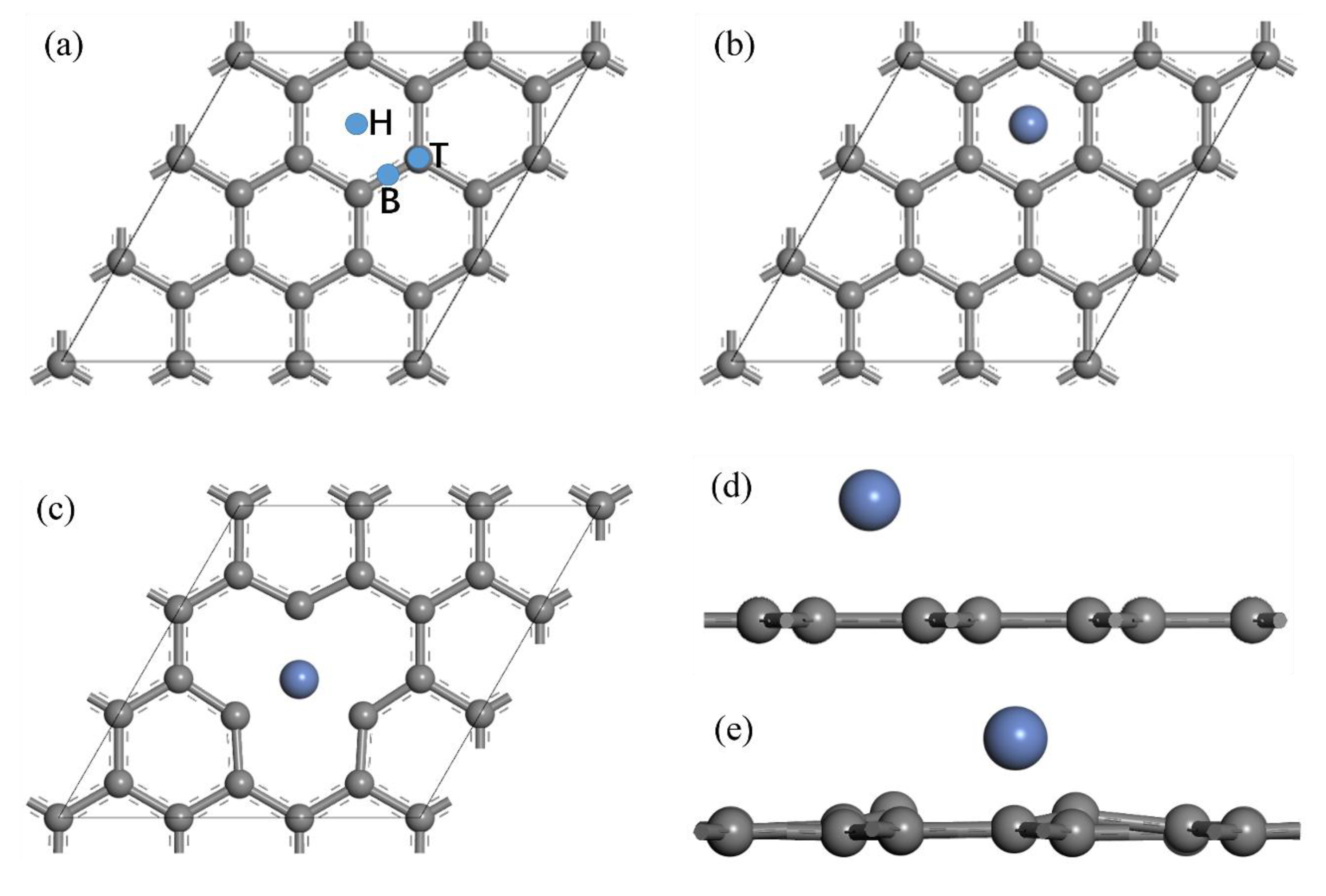
2.4. First Principle Calculation of the Adsorption Strength between Ni and GNSs
3. Results and Discussion
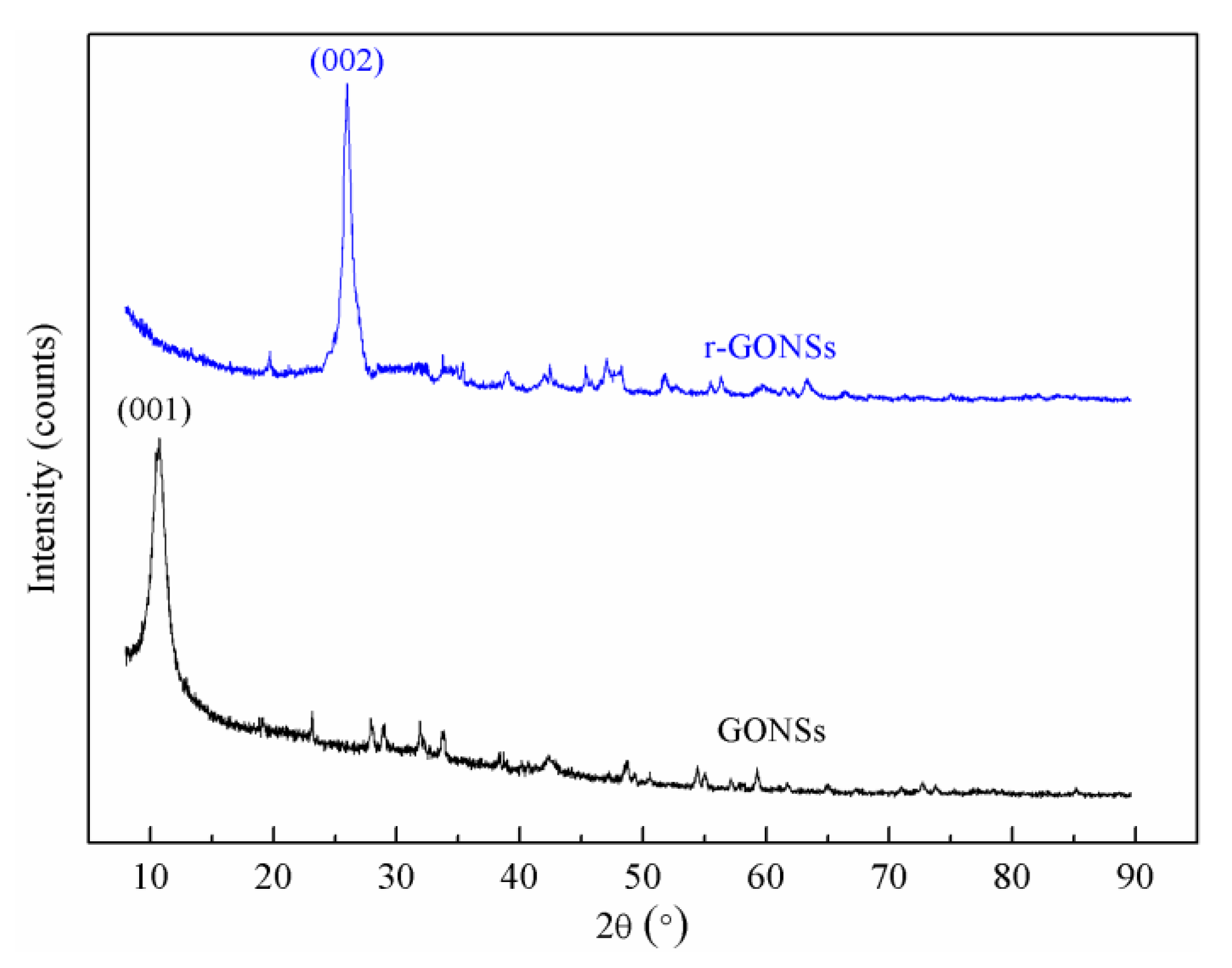
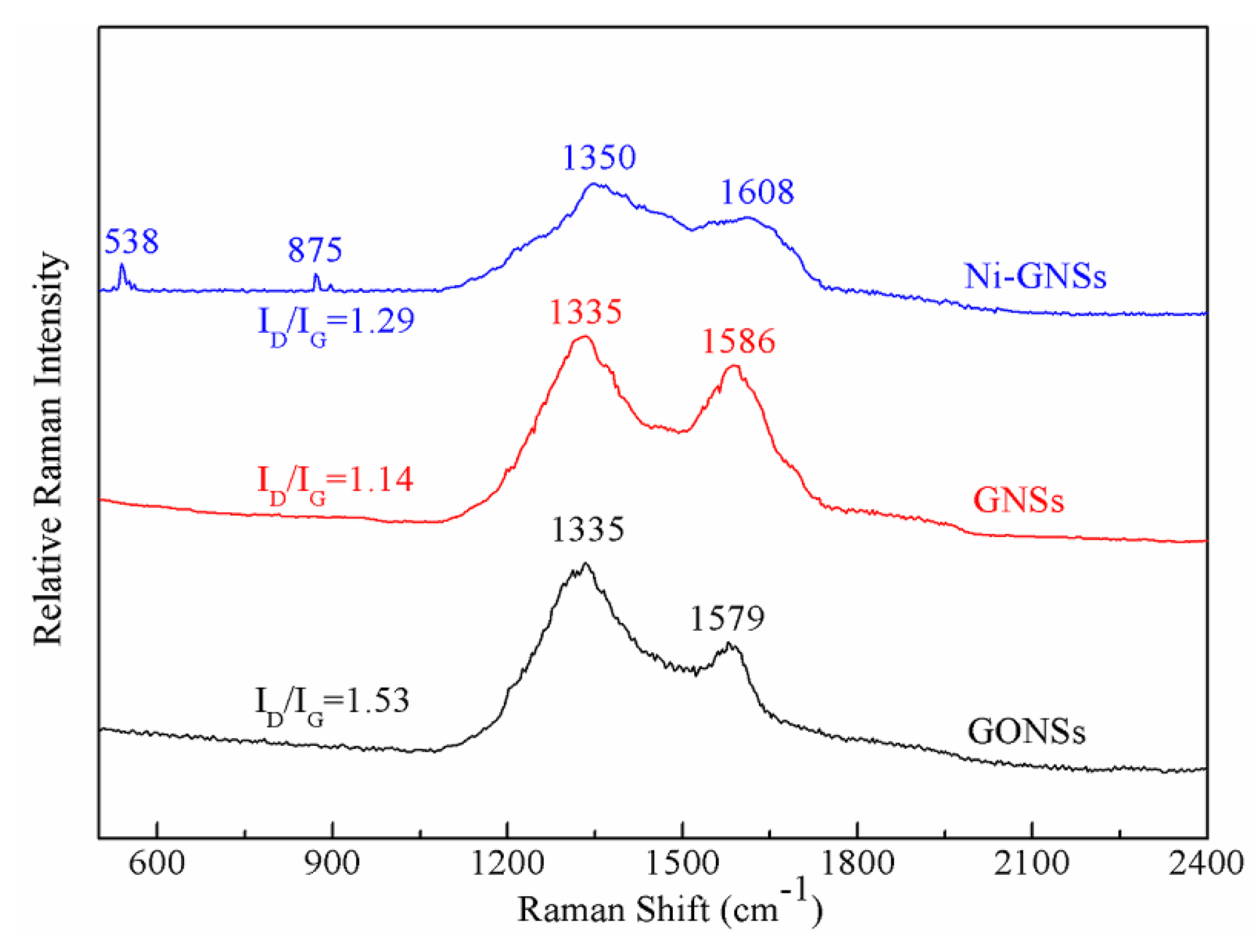
3.1. Pretreatment of GONSs
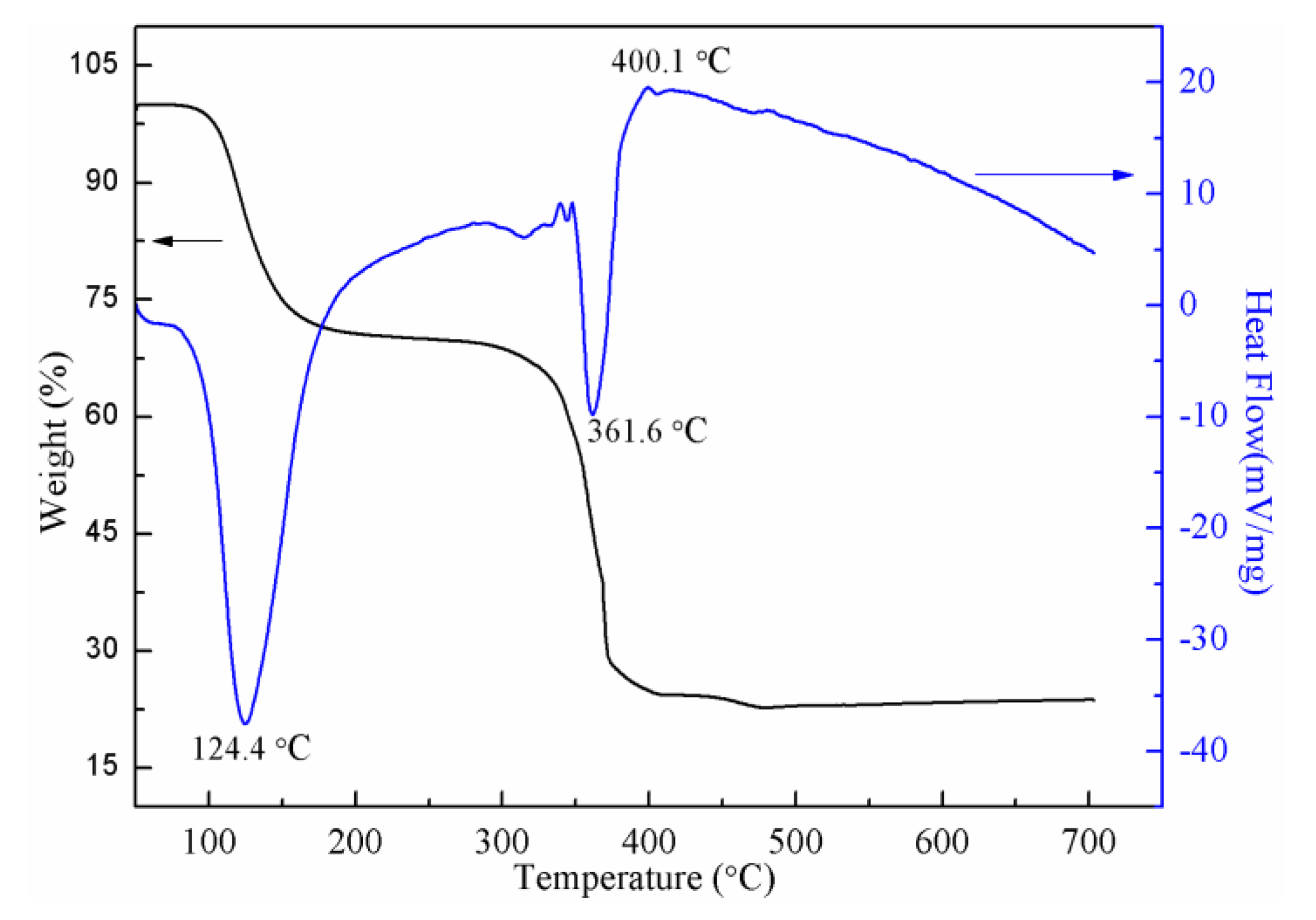
3.2. TG-DTA Analysis
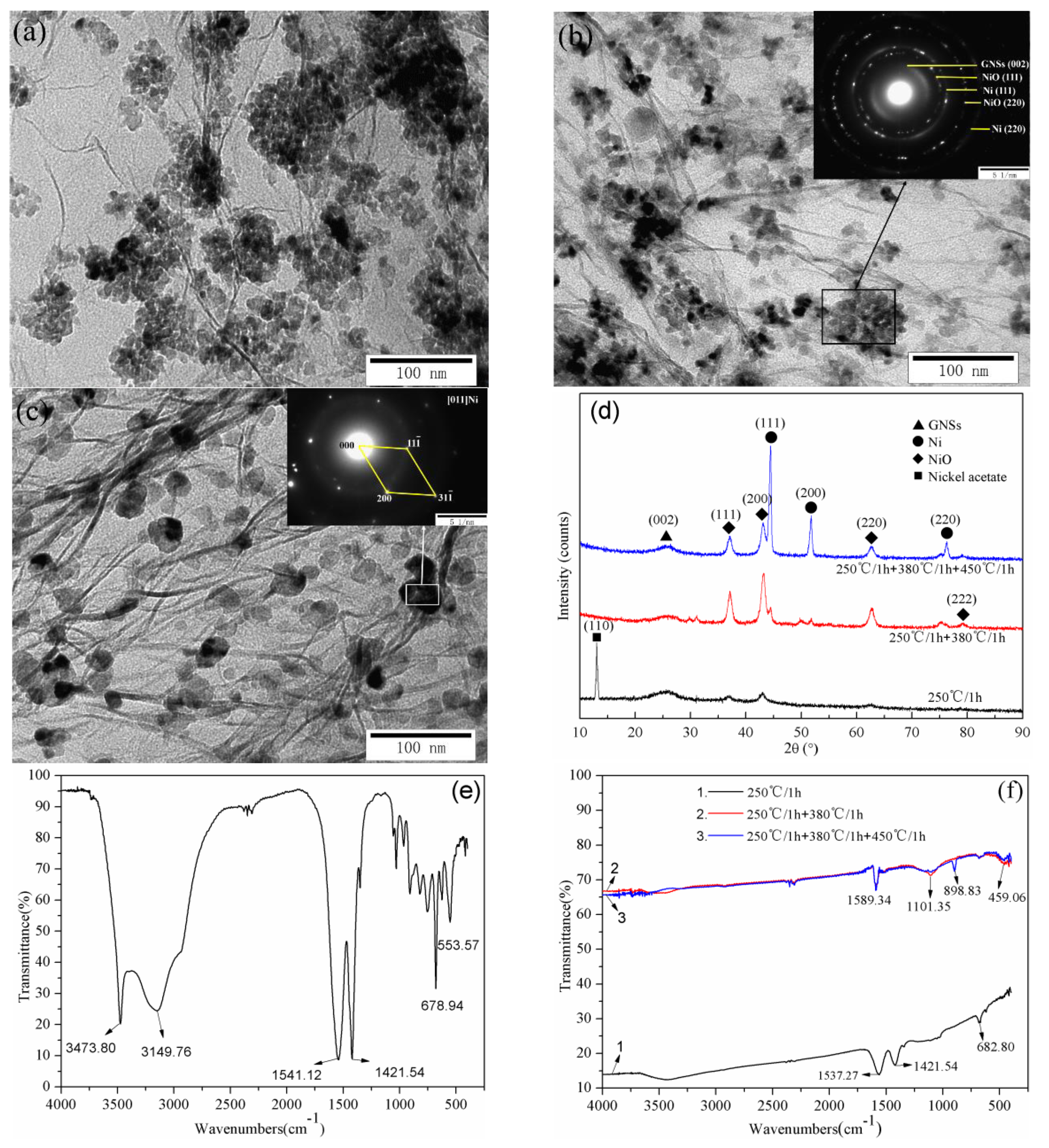
3.3. Segmented Pyrolysis Processes Analysis
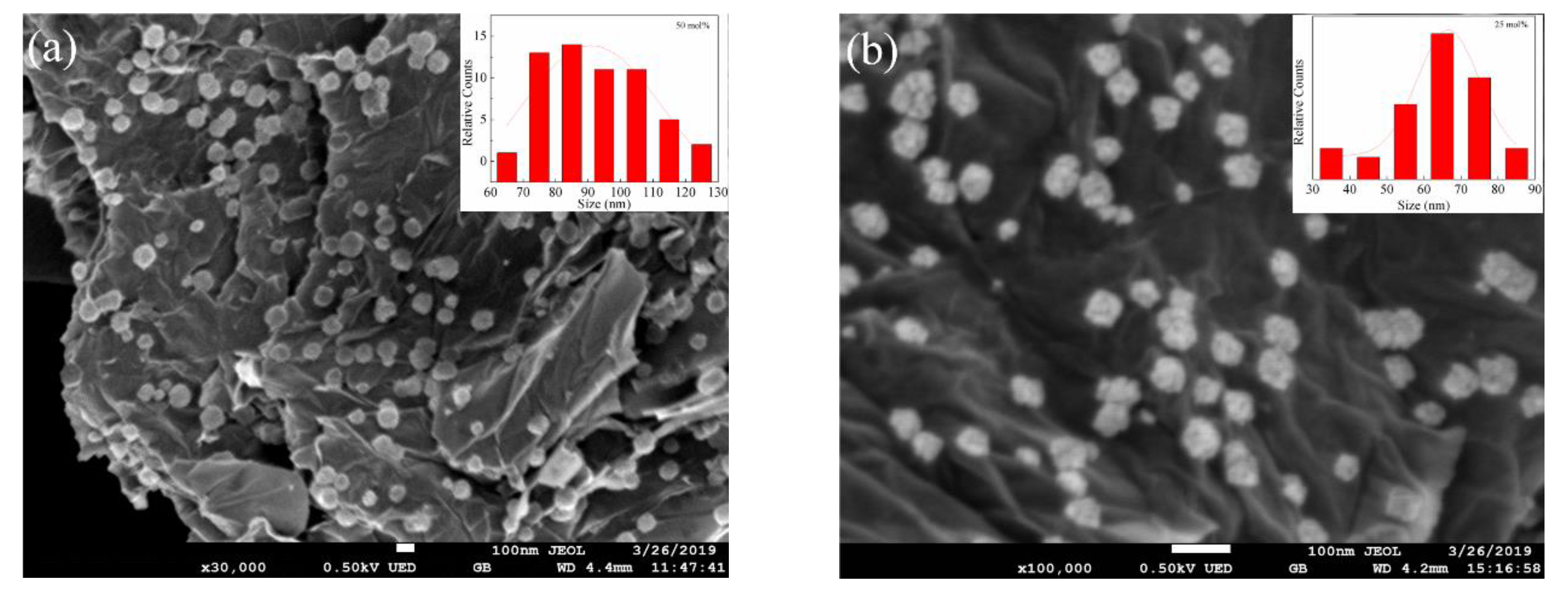
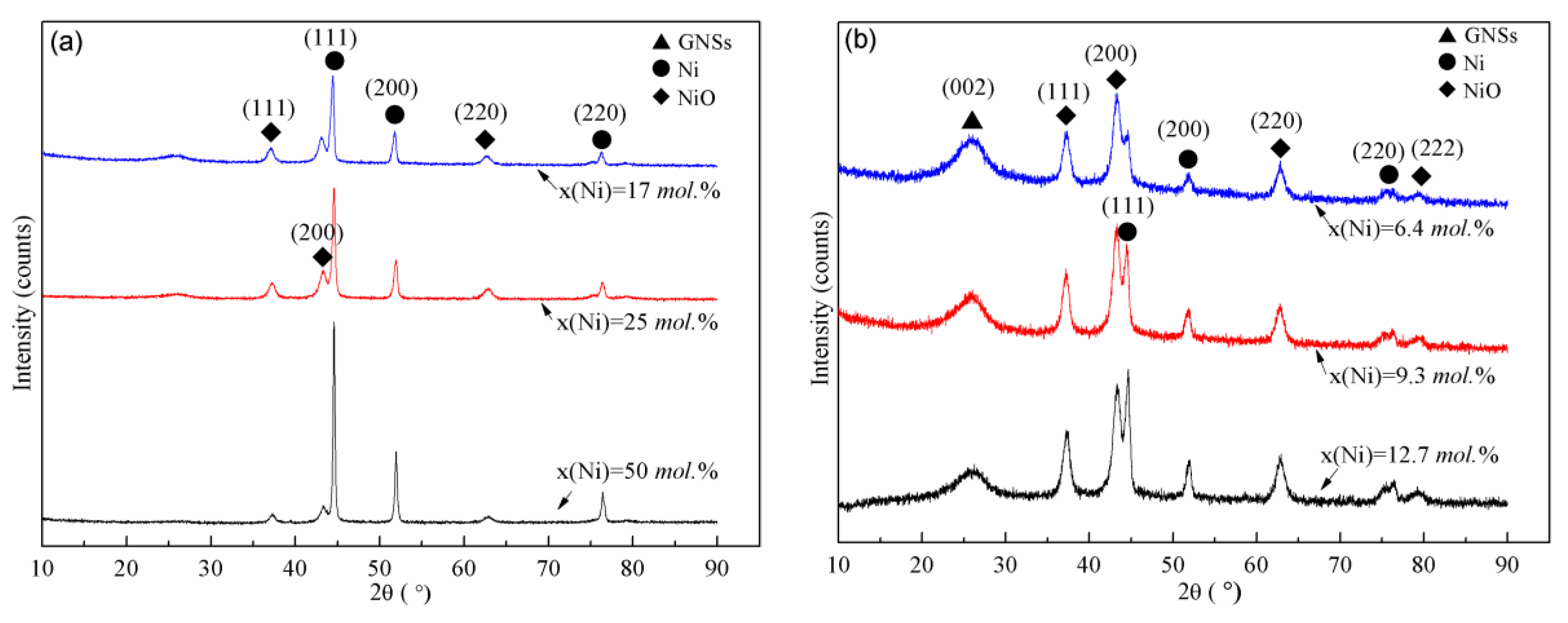
3.4. Effect of the Theoretical Ni Loading
3.5. Adsorption Strength of Nickel on Graphene Surface
3.6. First Principle Verification of the Bonding Strength between Ni and GNSs
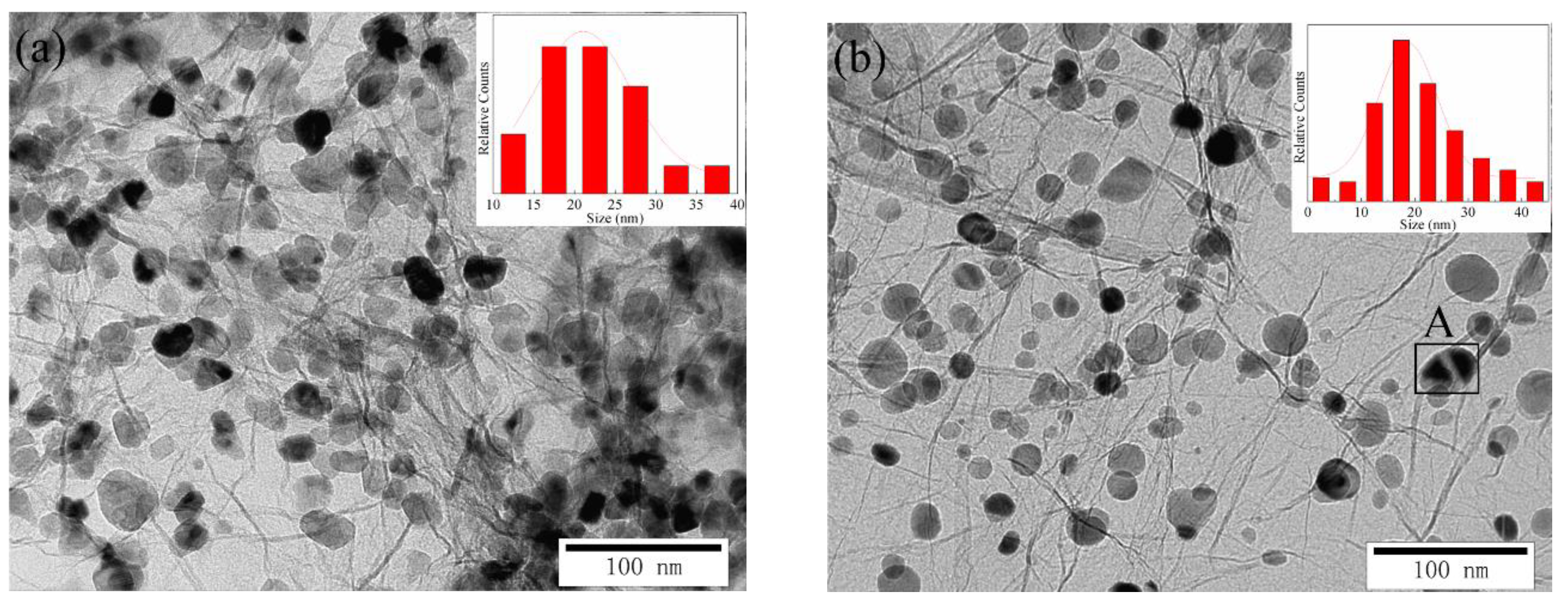
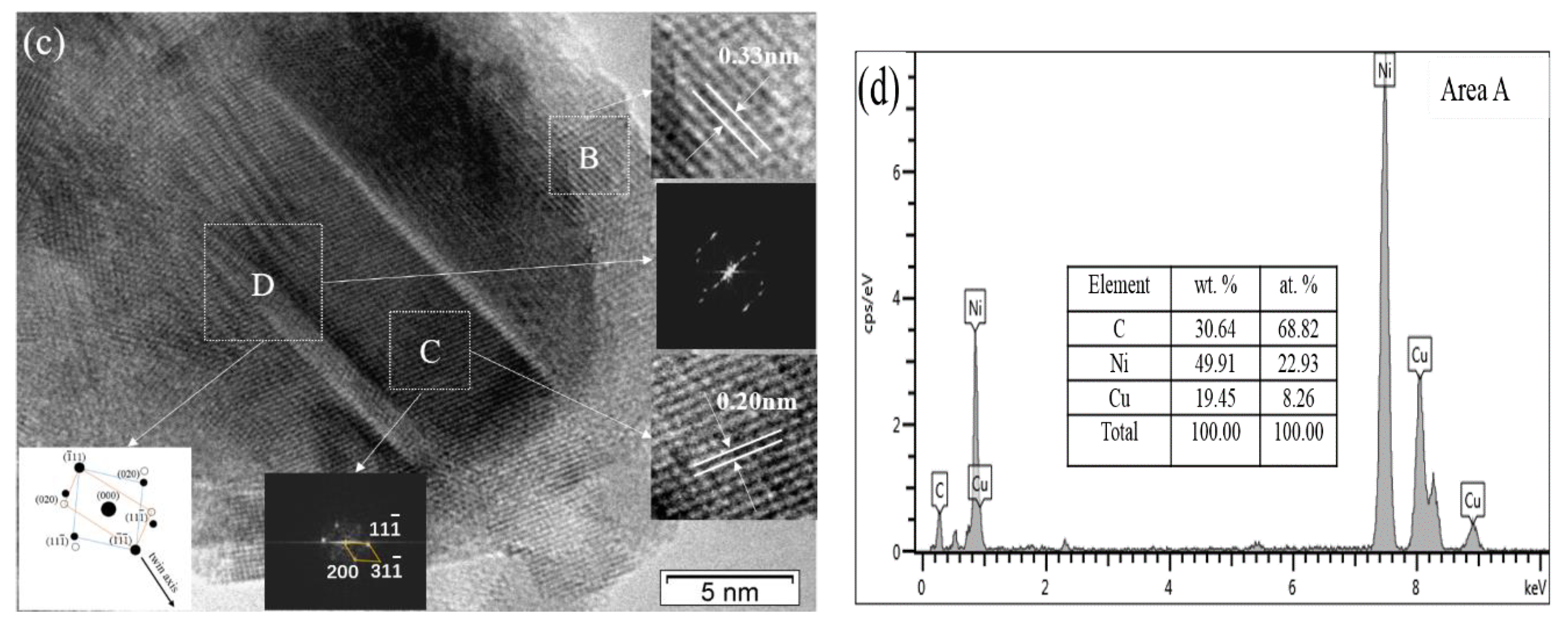
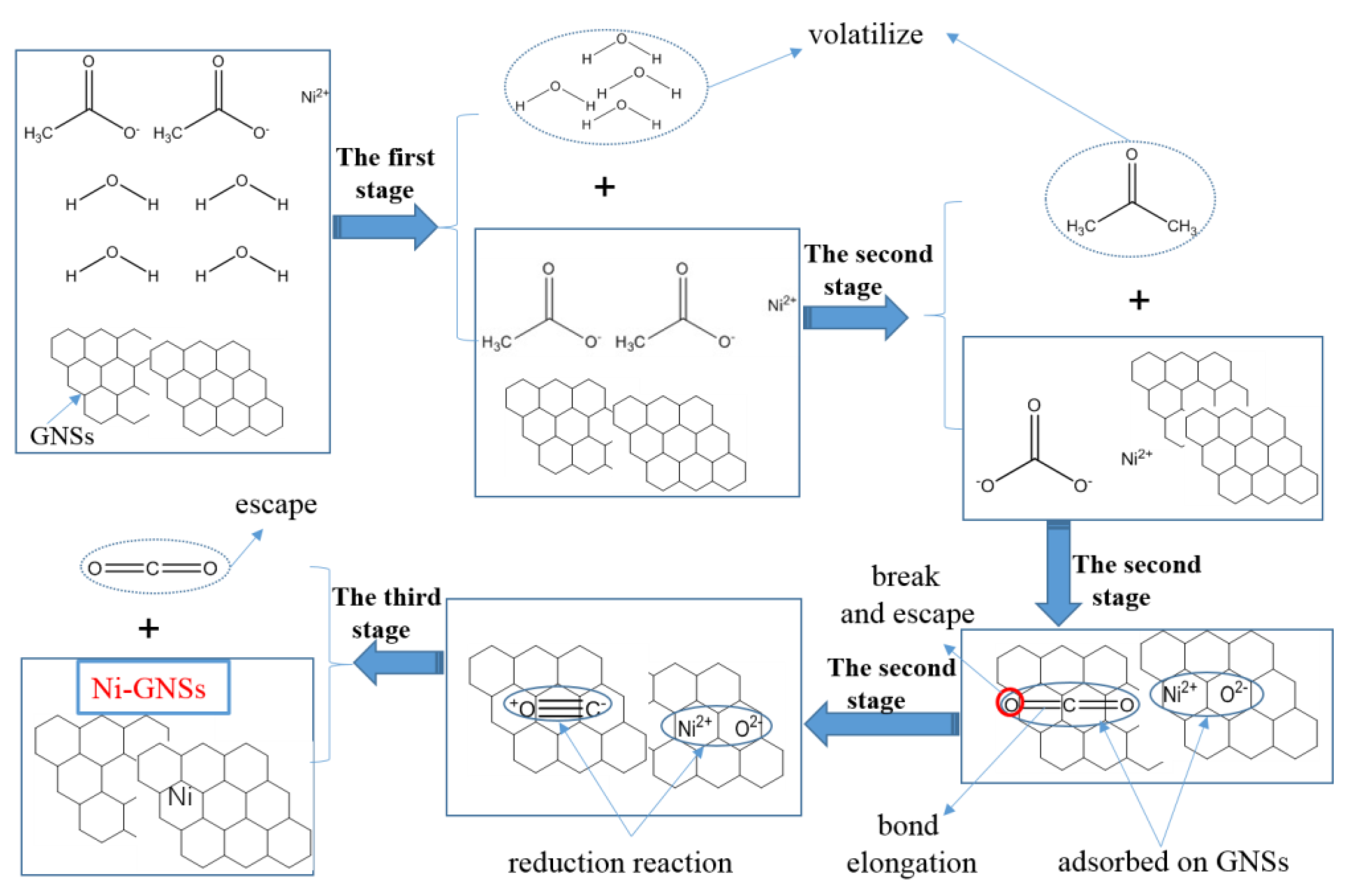
3.7. Formation Mechanism of Ni-Modified GNSs
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Geim, A.K. Graphene: Status and prospects. Science 2009, 324, 1530–1534. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, T.; Li, Y.; Zang, Z.; Peng, X.; Wei, H.; Zha, W.; Wang, F. Enhanced X-ray photon response in solution-synthesized CsPbBr3 nanoparticles wrapped by reduced graphene oxide. Sol. Energy Mater. Sol. Cells 2018, 187, 249–254. [Google Scholar] [CrossRef]
- Nag, A.; Mitra, A.; Mukhopadhyay, S.C. Graphene and its sensor-based applications: A review. Sens. Actuators A Phys. 2018, 270, 177–194. [Google Scholar] [CrossRef]
- Foo, M.E.; Gopinath, S.C. Feasibility of graphene in biomedical applications. Biomed. Pharmacother. 2017, 94, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Zang, Z.; Zeng, X.; Wang, M.; Hu, W.; Liu, C.; Tang, X. Tunable photoluminescence of water-soluble AgInZnS–graphene oxide (GO) nanocomposites and their application in-vivo bioimaging. Sens. Actuators B Chem. 2017, 252, 1179–1186. [Google Scholar] [CrossRef]
- Chen, T.W.; Rajaji, U.; Chen, S.M.; Li, Y.L.; Ramalingam, R.J. Ultrasound-assisted synthesis of α-MnS (alabandite) nanoparticles decorated reduced graphene oxide hybrids: Enhanced electrocatalyst for electrochemical detection of Parkinson’s disease biomarker. Ultrason. Sonochem. 2019, 56, 378–385. [Google Scholar] [CrossRef]
- Aditya, T.; Nayak, A.K.; Pradhan, D.; Pal, A.; Pal, T. Fabrication of MoS2 decorated reduced graphene oxide sheets from solid Mo-precursor for electrocatalytic hydrogen evolution reaction. Electrochim. Acta 2019, 313, 341–351. [Google Scholar] [CrossRef]
- Ji, Z.; Wang, Y.; Shen, X.; Ma, H.; Yang, J.; Yuan, A.; Zhou, H. Facile synthesis and enhanced catalytic performance of reduced graphene oxide decorated with hexagonal structure Ni nanoparticles. J. Colloid Interface Sci. 2017, 487, 223–230. [Google Scholar] [CrossRef]
- Ren, S.; Rong, P.; Yu, Q. Preparations, properties and applications of graphene in functional devices: A concise review. Ceram. Int. 2018, 44, 11940–11955. [Google Scholar] [CrossRef]
- Kavinkumar, T.; Kavitha, P.; Naresh, N.; Manivannan, S.; Muneeswaran, M.; Neppolian, B. High performance flexible solid-state symmetric supercapacitorsbased on laser induced porous reduced graphene oxide-graphene oxide hybrid nanostructure devices. Appl. Surf. Sci. 2019, 480, 671–679. [Google Scholar] [CrossRef]
- Yu, G.-H.; Han, Q.; Qu, L.-T. Graphene Fibers: Advancing Applications in Sensor, Energy Storage and Conversion. Chin. J. Polym. Sci. 2019, 37, 535–547. [Google Scholar] [CrossRef]
- Hu, X.; Chan, Y.; Zhang, K.; Yung, K. Effect of graphene doping on microstructural and mechanical properties of Sn–8Zn–3Bi solder joints together with electromigration analysis. J. Alloys Compd. 2013, 580, 162–171. [Google Scholar] [CrossRef]
- Ma, D.; Wu, P. Effects of coupled stressing and solid-state aging on the mechanical properties of graphene nanosheets reinforced Sn–58Bi–0.7Zn solder joint. Mater. Sci. Eng. A 2016, 651, 499–506. [Google Scholar] [CrossRef]
- Huang, Y.; Xiu, Z.; Wu, G.; Tian, Y.; He, P. Sn–3.0Ag–0.5Cu nanocomposite solders reinforced by graphene nanosheets. J. Mater. Sci. Mater. Electron. 2016, 27, 6809–6815. [Google Scholar] [CrossRef]
- Liu, X.; Han, Y.; Jing, H.; Wei, J.; Xu, L. Effect of graphene nanosheets reinforcement on the performance of Sn–Ag–Cu lead-free solder. Mater. Sci. Eng. A 2013, 562, 25–32. [Google Scholar] [CrossRef]
- Christian, M.S.; Otero-De-La-Roza, A.; Johnson, E.R. Adsorption of graphene to metal (111) surfaces using the exchange-hole dipole moment model. Carbon 2017, 124, 531–540. [Google Scholar] [CrossRef]
- Xie, K.F.; Jia, Q.Q.; Zhang, X.T.; Fu, L.; Zhao, G.H. Electronic and magnetic properties of Stone–Wales defected graphene decorated with the half-metallocene of M (M = Fe, Co, Ni): A first principle study. Nanomaterials 2018, 8, 552. [Google Scholar] [CrossRef]
- El-Daly, A.; El-Taher, A.; Dalloul, T. Improved creep resistance and thermal behavior of Ni-doped Sn–3.0Ag–0.5Cu lead-free solder. J. Alloys Compd. 2014, 587, 32–39. [Google Scholar] [CrossRef]
- Li, N.; Zhang, L.; Xu, M.; Xia, T.; Ruan, X.; Song, S.; Ma, H. Preparation and mechanical property of electrodeposited Al-graphene composite coating. Mater. Des. 2016, 111, 522–527. [Google Scholar] [CrossRef]
- Hu, Q.-H.; Wang, X.-T.; Chen, H.; Wang, Z.-F. Synthesis of Ni/graphene sheets by an electroless Ni-plating method. Carbon 2012, 50, 3118. [Google Scholar] [CrossRef]
- Hui, X.; Qian, L.; Harris, G.; Wang, T.; Che, J. Fast fabrication of NiO@graphene composites for supercapacitor electrodes: Combination of reduction and deposition. Mater. Des. 2016, 109, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Jilani, A.; Othman, M.H.D.; Ansari, M.O.; Kumar, R.; Khan, I.U.; Abdel-Wahab, M.S.; Alshahrie, A.; Barakat, M.A.; Kurniawan, T.A. Structural, optical, and photocatalytic investigation of nickel oxide@graphene oxide nanocomposite thin films by RF magnetron sputtering. J. Mater. Sci. 2018, 53, 15034–15050. [Google Scholar] [CrossRef]
- Liu, C.-W.; Chen, W.-E.; Sun, Y.T.A.; Lin, C.-R. Fabrication and electrochemistry characteristics of nickel-doped diamond-like carbon film toward applications in non-enzymatic glucose detection. Appl. Surf. Sci. 2018, 436, 967–973. [Google Scholar] [CrossRef]
- Lee, S.; Jeong, Y.; Oh, H.-J.; Lee, J.-H.; Lee, O.Y.; Chi, C.-S. Synthesis of silver nanowire arrays by thermal decomposition of silver acetate. Met. Mater. Int. 2009, 15, 631–636. [Google Scholar] [CrossRef]
- Lin, Y.; Watson, K.A.; Fallbach, M.J.; Ghose, S.; Smith, J.G., Jr.; Delozier, D.M.; Cao, W.; Crooks, R.E.; Connell, J.W. Rapid, Solventless, Bulk Preparation of Metal Nanoparticle-Decorated Carbon Nanotubes. ACS Nano 2009, 3, 871–884. [Google Scholar] [CrossRef]
- Luo, X.; Chen, Y.; Yue, G.-H.; Peng, D.-L.; Luo, X. Preparation of hexagonal close-packed nickel nanoparticles via a thermal decomposition approach using nickel acetate tetrahydrate as a precursor. J. Alloys Compd. 2009, 476, 864–868. [Google Scholar] [CrossRef]
- Szczesny, R.; Szlyk, E. Thermal decomposition of some silver(I) carboxylates under nitrogen atmosphere. J. Therm. Anal. Calorim. 2013, 111, 1325–1330. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, K.; Zhang, M. Fabrication and properties of Ni-modified graphene nanosheets reinforced Sn-Ag-Cu composite solder. J. Alloys Compd. 2019, 781, 761–772. [Google Scholar] [CrossRef]
- Bourlinos, A.B.; Gournis, D.; Petridis, D.; Szabo’, T.; Szeri, A.; De´ka´ny, I. Graphite oxide: Chemical reduction to graphite and surface modification with primary aliphatic amines and amino acids. Langmuir 2003, 19, 6050–6055. [Google Scholar] [CrossRef]
- Guo, H.-L.; Wang, X.-F.; Qian, Q.-Y.; Wang, F.-B.; Xia, X.-H. A Green Approach to the Synthesis of Graphene Nanosheets. ACS Nano 2009, 3, 2653–2659. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Qin, Z.; Li, T.; Dou, Z.; Zeng, F.; Cai, Y.; Zhu, M.; Zhou, Z. Poly(aniline-co-pyrrole) on the surface of reduced graphene oxide as high-performance electrode materials for supercapacitors. Electrochim. Acta 2015, 177, 335–342. [Google Scholar] [CrossRef]
- Jesus, J.C.D.; Gonzalez, I.; Quevedo, A.; Puerta, T. Thermal decomposition of nickel acetate tetrahydrate: An integrated study by TGA, QMS and XPS techniques. J. Mol. Catal. A Chem. 2005, 228, 283–291. [Google Scholar] [CrossRef]
- Galwey, A.K.; McKee, S.G.; Mitchell, T.R.; Brown, M.E.; Bean, A.F. A kinetic and mechanistic study of the thermal decomposition of nickel acetate. React. Solids 1988, 6, 173–186. [Google Scholar] [CrossRef]
- Wen, B.Y.; Sun, C.Z.; Bai, B.F. Molecular dynamics simulation of the separation of CH4/CO2 by nanoporous graphene. Acta Phys. Chim. Sin. 2015, 31, 261–267. [Google Scholar]
- Laxmi; Khan, S.; Kareem, A.; Zafar, F.; Nishat, N. Synthesis, vibrational spectrometry and thermal characterizations of coordination polymers derived from divalent metal ions and hydroxyl terminated polyurethane as ligand. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 188, 400–410. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.R.; Li, Q.R.; Xie, D.Y.; Xiao, H.Y.; Lu, H.X. Synthesis of NiO using pine as template and adsorption performance for Pb(II) from aqueous solution. Appl. Surf. Sci. 2013, 279, 129–136. [Google Scholar] [CrossRef]
- Kashif, M.; Sharmin, E.; Zafar, F.; Ahmad, S. Synthesis and Characterization of Ricinoleamide-Based Polyurethane. J. Am. Oil Chem. Soc. 2011, 88, 1989–1996. [Google Scholar] [CrossRef]
- Huo, F.; Zhang, K.; Zhang, M.; Wang, H. Preparation by Ball Milling–Thermal Decomposition Method and Characterization of Reduced Graphene Oxide Decorated with Ni Nanoparticles. JOM 2019. [Google Scholar] [CrossRef]
- Chaudhary, R.G.; Juneja, H.D.; Pagadala, R.; Gandhare, N.V.; Gharpure, M.P. Synthesis, characterisation and thermal degradation behaviour of some coordination polymers by using TG–DTG and DTA techniques. J. Saudi Chem. Soc. 2015, 19, 442–453. [Google Scholar] [CrossRef]
- Hussein, G.A.M.; Nohman, A.K.H.; Attyia, K.M.A. Characterization of the decomposition course of nickel acetate tetrahydrate in air. J. Therm. Anal. Calorim. 1994, 42, 1155–1165. [Google Scholar] [CrossRef]
- Huo, F.; Zhang, K.; Zhang, M.; Wang, H. The Interfacial Intermetallic and Shear Strength of Ni Nanoparticle-Decorated Reduced Graphene Oxide Reinforced Sn2.5Ag0.5Cu Lead-Free Composite Soldering Joints. Adv. Eng. Mater. 2018, 20, 1800147. [Google Scholar] [CrossRef]
- Karthik, K.; Pandian, S.K.; Jaya, N.V. Effect of nickel doping on structural, optical and electrical properties of TiO2 nanoparticles by sol–gel method. Appl. Surf. Sci. 2010, 256, 6829–6833. [Google Scholar] [CrossRef]
- Kataura, H.; Kumazawa, Y.; Maniwa, Y.; Umezu, I.; Suzuki, S.; Ohtsuka, Y.; Achiba, Y. Optical Properties of Single-Wall Carbon Nanotubes. Synth. Met. 1999, 103, 2555–2558. [Google Scholar] [CrossRef]
- Juan, A.G.; Almudena, B.; Rocío, O.; Dolores, E.; César, J.S.; Julián, M.; Álvaro, C.; Francisco, J.R.S. A comparative study of particle size distribution of graphene nanosheets synthesized by an ultrasound-assisted method. Nanomaterials 2019, 9, 152–167. [Google Scholar]
- Eklund, P.; Holden, J.; Jishi, R. Vibrational modes of carbon nanotubes; Spectroscopy and theory. Carbon 1995, 33, 959–972. [Google Scholar] [CrossRef]
- Lee, J.; Novoselov, K.S.; Shin, H.S. Interaction between Metal and Graphene: Dependence on the Layer Number of Graphene. ACS Nano 2011, 5, 608–612. [Google Scholar] [CrossRef]
- Wu, J.X.; Xu, H.; Zhang, J. Raman spectroscopy of graphene. Acta Chim. Sin. 2014, 72, 301–318. [Google Scholar] [CrossRef]
- Maslar, J.E.; Hurst, W.S.; Bowers, W.J.; Hendricks, J.H.; Aquino, M.I. In Situ Raman Spectroscopic Investigation of Nickel Hydrothermal Corrosion. Corros. 2002, 58, 225–231. [Google Scholar] [CrossRef]
- Widjaja, H.; Altarawneh, M.; Jiang, Z.-T.; Yin, C.-Y.; Goh, B.-M.; Mondinos, N.; Dlugogorski, B.Z. Geometrical and orientational investigations on the electronic structure of graphene with adsorbed aluminium or silicon. Mater. Des. 2016, 89, 27–35. [Google Scholar] [CrossRef]
- Liu, X.; Wang, C.Z.; Yao, Y.X.; Lu, W.C.; Hupalo, M.; Tringides, M.C.; Ho, K.M. Bonding and charge transfer by metal adatom adsorption on graphene. Phys. Rev. B 2011, 83, 235411. [Google Scholar] [CrossRef]
- Hupalo, M.; Liu, X.; Wang, C.-Z.; Lu, W.-C.; Yao, Y.-X.; Ho, K.-M.; Tringides, M.C. Metal Nanostructure Formation on Graphene: Weak versus Strong Bonding. Adv. Mater. 2011, 23, 2082–2087. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Tian, Y.; Chen, G.; Zhao, J. Theoretical insights into the energetics and electronic properties of MPt12 (M = Fe, Co, Ni, Cu, and Pd) nanoparticles supported by N-doped defective graphene. Appl. Surf. Sci. 2017, 397, 199–205. [Google Scholar] [CrossRef]
- Ben Mansour, N.; Najeh, I.; Mansouri, S.; El Mir, L. Effect of pyrolysis temperature on the properties of carbon/nickel nanocomposites prepared by sol–gel method. Appl. Surf. Sci. 2015, 337, 158–165. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Zhang, K.; Yin, C.; Zhang, M. Environmentally Friendly and Controllable Pyrolysis Method to Synthesize Ni-Modified Graphene Nanosheets as Reinforcement of Lead-Free Solder. Metals 2019, 9, 1123. https://doi.org/10.3390/met9101123
Wang H, Zhang K, Yin C, Zhang M. Environmentally Friendly and Controllable Pyrolysis Method to Synthesize Ni-Modified Graphene Nanosheets as Reinforcement of Lead-Free Solder. Metals. 2019; 9(10):1123. https://doi.org/10.3390/met9101123
Chicago/Turabian StyleWang, Huigai, Keke Zhang, Chenxiang Yin, and Meng Zhang. 2019. "Environmentally Friendly and Controllable Pyrolysis Method to Synthesize Ni-Modified Graphene Nanosheets as Reinforcement of Lead-Free Solder" Metals 9, no. 10: 1123. https://doi.org/10.3390/met9101123
APA StyleWang, H., Zhang, K., Yin, C., & Zhang, M. (2019). Environmentally Friendly and Controllable Pyrolysis Method to Synthesize Ni-Modified Graphene Nanosheets as Reinforcement of Lead-Free Solder. Metals, 9(10), 1123. https://doi.org/10.3390/met9101123




