The Influence of the Electrolyte Nature and PEO Process Parameters on Properties of Anodized Ti-15Mo Alloy Intended for Biomedical Applications
Abstract
1. Introduction
2. Methods and Materials
2.1. Sample and Electrolyte Preparation
2.2. PEO Process
2.3. Sample Characterisation
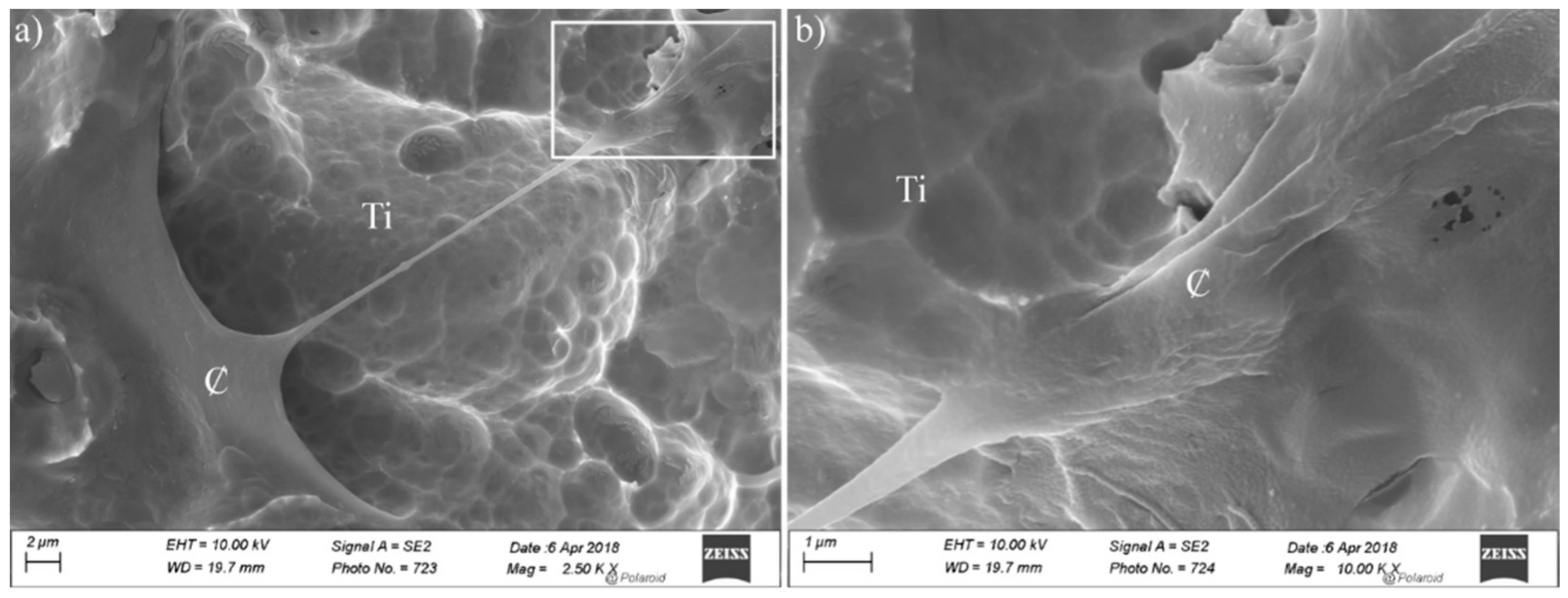
3. Results and Discussion
3.1. First Campaign: Selection of a Suitable Electrolyte for Ti-15Mo Anodization
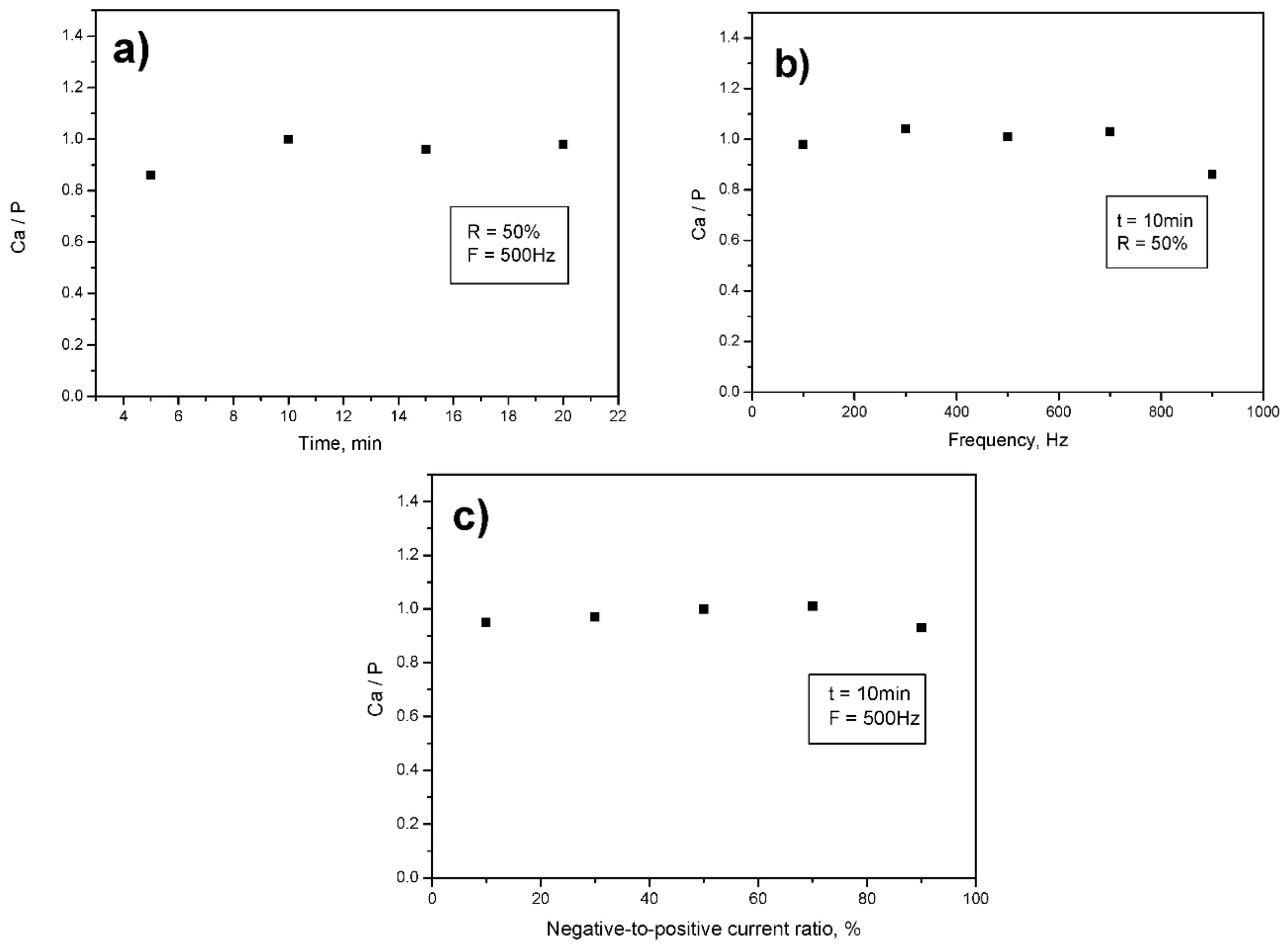
3.2. Second Campaign: Selection of the PEO Process Parameters
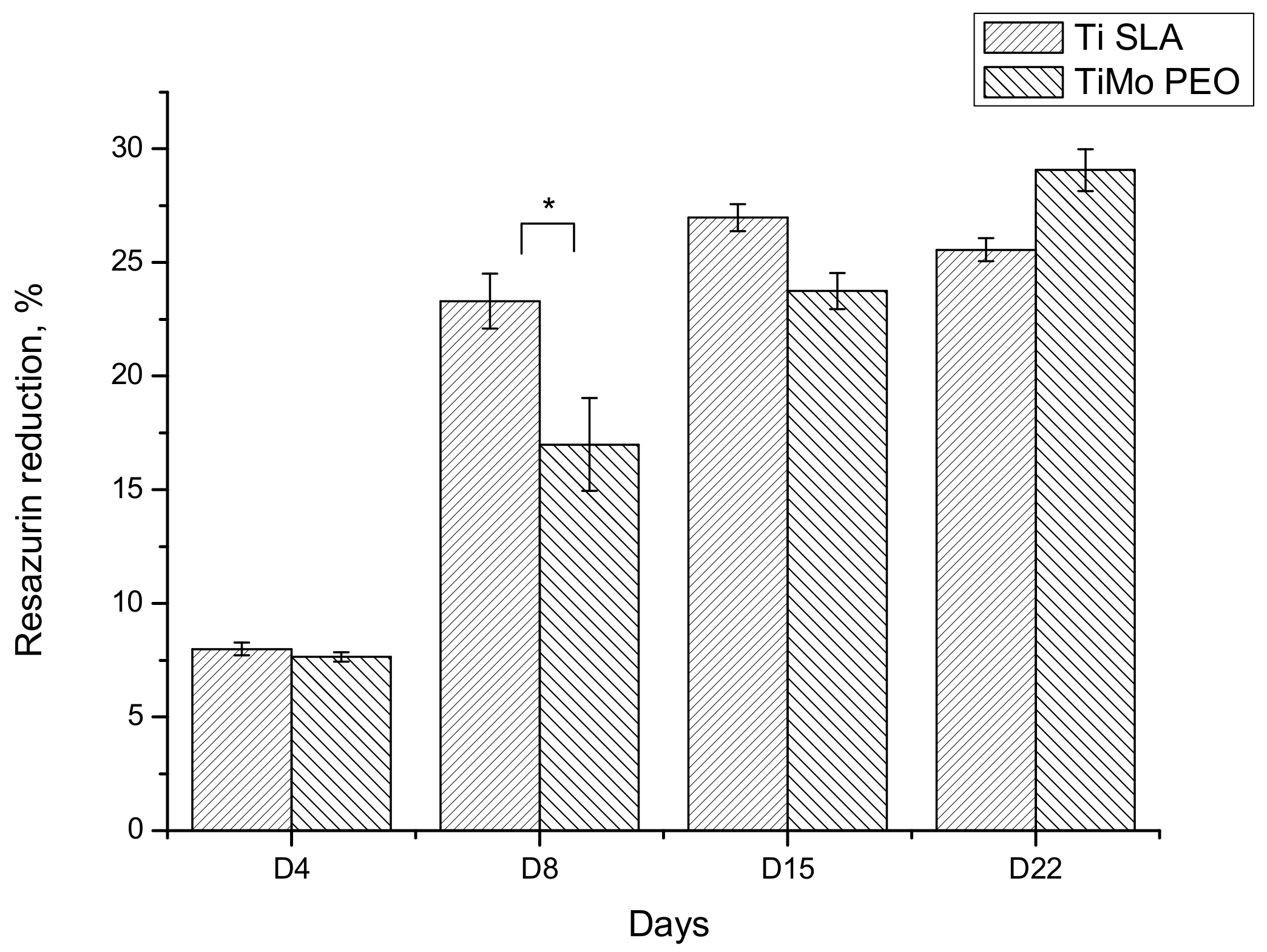
3.3. Biocompatibility Tests
4. Conclusions
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Özcan, M.; Hämmerle, C. Titanium as a reconstruction and implant material in dentistry: Advantages and pitfalls. Materials 2012, 5, 1528–1545. [Google Scholar] [CrossRef]
- Disegi, J.A. Titanium alloys for fracture fixation implants. Injury 2000, 31, 14–17. [Google Scholar] [CrossRef]
- Rack, H.J.; Qazi, J.I. Titanium alloys for biomedical applications. Mater. Sci. Eng. C 2006, 26, 1269–1277. [Google Scholar] [CrossRef]
- Li, Y.; Yang, C.; Zhao, H.; Qu, S.; Li, X.; Li, Y. New developments of Ti-based alloys for biomedical applications. Materials 2014, 7, 1709–1800. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, N.T.C.; Guastaldi, A.C. Electrochemical behavior of Ti-Mo alloys applied as biomaterial. Corros. Sci. 2008, 50, 938–945. [Google Scholar] [CrossRef]
- Babilas, D.; Słuzalska, K.; Krza̧kała, A.; Maciej, A.; Socha, R.P.; Dercz, G.; Tylko, G.; Michalska, J.; Osyczka, A.M.; Simka, W. Plasma electrolytic oxidation of a Ti-15Mo alloy in silicate solutions. Mater. Lett. 2013, 100, 252–256. [Google Scholar] [CrossRef]
- Carayon, M.T.; Lacout, J.L. Study of the Ca/P atomic ratio of the amorphous phase in plasma-sprayed hydroxyapatite coatings. J. Solid State Chem. 2003, 172, 339–350. [Google Scholar] [CrossRef]
- Ribeiro, A.R.; Oliveira, F.; Boldrini, L.C.; Leite, P.E.; Falagan-Lotsch, P.; Linhares, A.B.R.; Zambuzzi, W.F.; Fragneaud, B.; Campos, A.P.C.; Gouvêa, C.P.; et al. Micro-arc oxidation as a tool to develop multifunctional calcium-rich surfaces for dental implant applications. Mater. Sci. Eng. C 2015, 54, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Krzakała, A.; Słuzalska, K.; Dercz, G.; Maciej, A.; Kazek, A.; Szade, J.; Winiarski, A.; Dudek, M.; Michalska, J.; Tylko, G.; et al. Characterisation of bioactive films on Ti-6Al-4V alloy. Electrochim. Acta 2013, 104, 425–438. [Google Scholar] [CrossRef]
- Kazek-Kęsik, A.; Dercz, G.; Kalemba, I.; Suchanek, K.; Kukharenko, A.I.; Korotin, D.M.; Michalska, J.; Krzakala, A.; Piotrowski, J.; Kurmaev, E.Z.; et al. Surface characterisation of Ti-15Mo alloy modified by a PEO process in various suspensions. Mater. Sci. Eng. C 2014, 39, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Simka, W. Preliminary investigations on the anodic oxidation of Ti-13Nb-13Zr alloy in a solution containing calcium and phosphorus. Electrochim. Acta 2011, 56, 9831–9837. [Google Scholar] [CrossRef]
- Frauchiger, V.M.; Schlottig, F.; Gasser, B.; Textor, M. Anodic plasma-chemical treatment of CP titanium surfaces for biomedical applications. Biomaterials 2004, 25, 593–606. [Google Scholar] [CrossRef]
- Banakh, O.; Journot, T.; Gay, P.A.; Matthey, J.; Csefalvay, C.; Kalinichenko, O.; Sereda, O.; Moussa, M.; Durual, S.; Snizhko, L. Synthesis by anodic-spark deposition of Ca- and P-containing films on pure titanium and their biological response. Appl. Surf. Sci. 2016, 378, 207–215. [Google Scholar] [CrossRef]
- Moussa, M.; Banakh, O.; Wehrle-Haller, B.; Fontana, P.; Scherrer, S.; Cattani, M.; Wiskott, A.; Durual, S. TiNxOy coatings facilitate the initial adhesion of osteoblasts to create a suitable environment for their proliferation and the recruitment of endothelial cells. Biomed. Mater. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Durual, S.; Pernet, F.; Rieder, P.; Mekki, M.; Cattani-Lorente, M.; Wiskott, H.W.A. Titanium nitride oxide coating on rough titanium stimulates the proliferation of human primary osteoblasts. Clin. Oral Implants Res. 2011, 22, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Simka, W.; Krzakała, A.; Korotin, D.M.; Zhidkov, I.S.; Kurmaev, E.Z.; Cholakh, S.O.; Kuna, K.; Dercz, G.; Michalska, J.; Suchanek, K.; et al. Modification of a Ti-Mo alloy surface via plasma electrolytic oxidation in a solution containing calcium and phosphorus. Electrochim. Acta 2013, 96, 180–190. [Google Scholar] [CrossRef]
- Saito, G.; Nakasugi, Y.; Akiyama, T. Excitation temperature of a solution plasma during nanoparticle synthesis. J. Appl. Phys. 2014, 116, 83301. [Google Scholar] [CrossRef]
- Saji, V.S.; Lee, C.W. Molybdenum, molybdenum oxides, and their electrochemistry. ChemSusChem 2012, 5, 1146–1161. [Google Scholar] [CrossRef] [PubMed]
- Habazaki, H.; Uozumi, M.; Konno, H.; Shimizu, K.; Nagata, S.; Asami, K.; Skeldon, P.; Thompson, G.E. Influence of molybdenum species on growth of anodic titania. Electrochim. Acta 2002, 47, 3837–3845. [Google Scholar] [CrossRef]
- Landmann, M.; Rauls, E.; Schmidt, W.G. The electronic structure and optical response of rutile, anatase and brookite TiO2. J. Phys. Condens. Matter 2012, 24, 195503. [Google Scholar] [CrossRef] [PubMed]


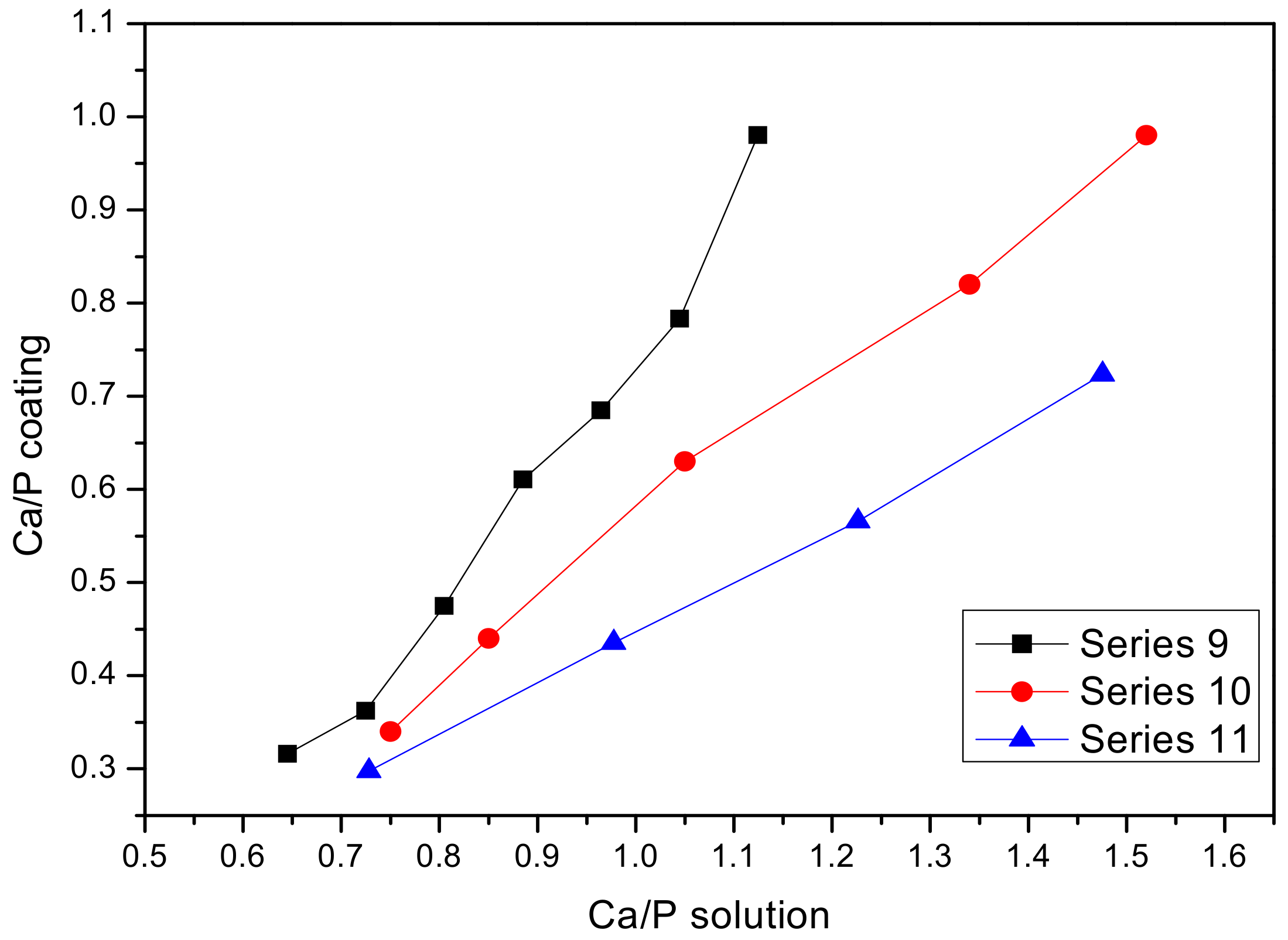


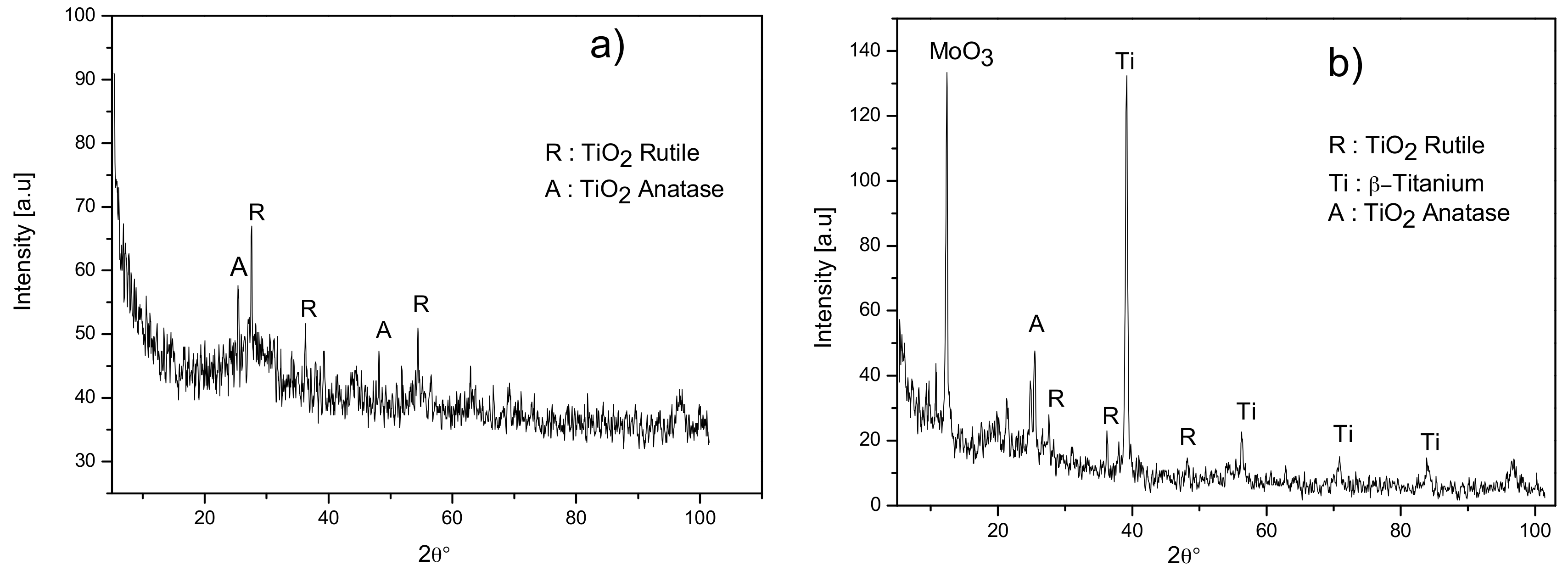


| Series | Compounds in the Solution | Ca/P Ratio | ||
|---|---|---|---|---|
| I | II | In the Solution, (Ca/P)sol | In the Layer, (Ca/P)coat | |
| 1 | Ca(CH3COO)2·H2O | Na4P2O7·10H2O | 1.53 | 0.64 |
| 2 | (NH4)H2PO4 | Ca(CH3COO)2·H2O | 1.53 | 1.27 |
| 3 | (NH4)H2PO4 | Ca(OH)2 | 1.99 | 0.84 |
| 4 | Na4P2O7·10H2O | CaC4H4O6 | 1.66 | 1.02 |
| 5 | (NH4)H2PO4 | CaC4H4O6 | 1.65 | 1.13 |
| 6 | Na3PO4·12H2O | Ca(CH3COO)2·H2O | 1.73 | 1.14 |
| 7 | Na3PO4·12H2O | CaC4H4O6 | 1.66 | 1.38 |
| 8 | Na3PO4·12H2O | Ca(OH)2 | 1.99 | 0.84 |
| Time, min | Frequency, Hz | R = In/Ip |
|---|---|---|
| 5 | 500 | 50 |
| 10 | 500 | 50 |
| 15 | 500 | 50 |
| 20 | 500 | 50 |
| 10 | 100 | 50 |
| 10 | 300 | 50 |
| 10 | 500 | 50 |
| 10 | 700 | 50 |
| 10 | 900 | 50 |
| 10 | 500 | 10 |
| 10 | 500 | 30 |
| 10 | 500 | 50 |
| 10 | 500 | 70 |
| 10 | 500 | 90 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banakh, O.; Snizhko, L.; Journot, T.; Gay, P.-A.; Csefalvay, C.; Kalinichenko, O.; Girin, O.; Marger, L.; Durual, S. The Influence of the Electrolyte Nature and PEO Process Parameters on Properties of Anodized Ti-15Mo Alloy Intended for Biomedical Applications. Metals 2018, 8, 370. https://doi.org/10.3390/met8050370
Banakh O, Snizhko L, Journot T, Gay P-A, Csefalvay C, Kalinichenko O, Girin O, Marger L, Durual S. The Influence of the Electrolyte Nature and PEO Process Parameters on Properties of Anodized Ti-15Mo Alloy Intended for Biomedical Applications. Metals. 2018; 8(5):370. https://doi.org/10.3390/met8050370
Chicago/Turabian StyleBanakh, Oksana, Lyubov Snizhko, Tony Journot, Pierre-Antoine Gay, Catherine Csefalvay, Oleg Kalinichenko, Oleg Girin, Laurine Marger, and Stéphane Durual. 2018. "The Influence of the Electrolyte Nature and PEO Process Parameters on Properties of Anodized Ti-15Mo Alloy Intended for Biomedical Applications" Metals 8, no. 5: 370. https://doi.org/10.3390/met8050370
APA StyleBanakh, O., Snizhko, L., Journot, T., Gay, P.-A., Csefalvay, C., Kalinichenko, O., Girin, O., Marger, L., & Durual, S. (2018). The Influence of the Electrolyte Nature and PEO Process Parameters on Properties of Anodized Ti-15Mo Alloy Intended for Biomedical Applications. Metals, 8(5), 370. https://doi.org/10.3390/met8050370







