High-Temperature Behavior of High-Pressure Diecast Alloys Based on the Al-Si-Cu System: The Role Played by Chemical Composition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Alloys and Specimen Manufacturing
2.2. Tensile Testing
2.3. Analyses on Tested Specimens
3. Results
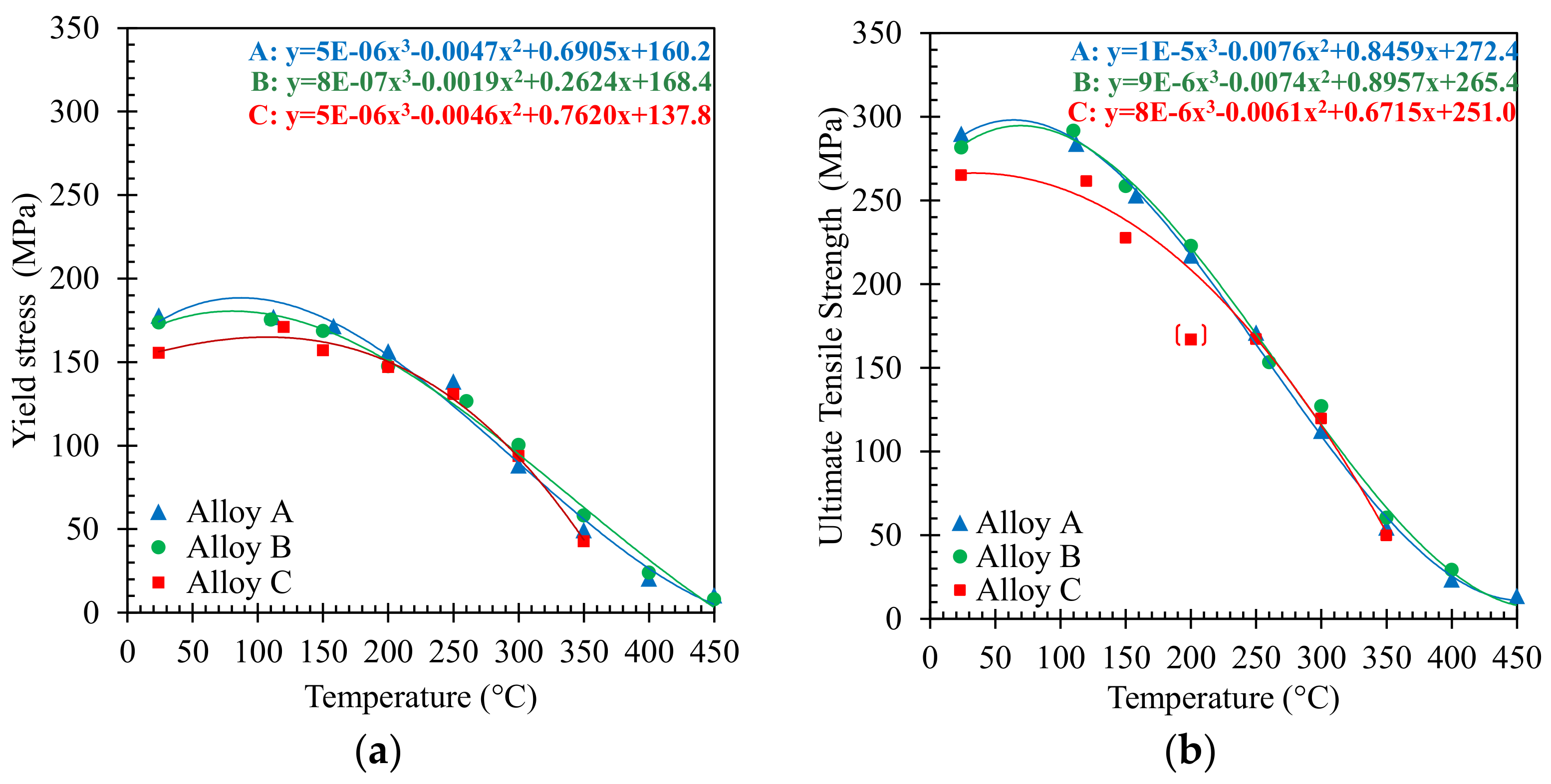
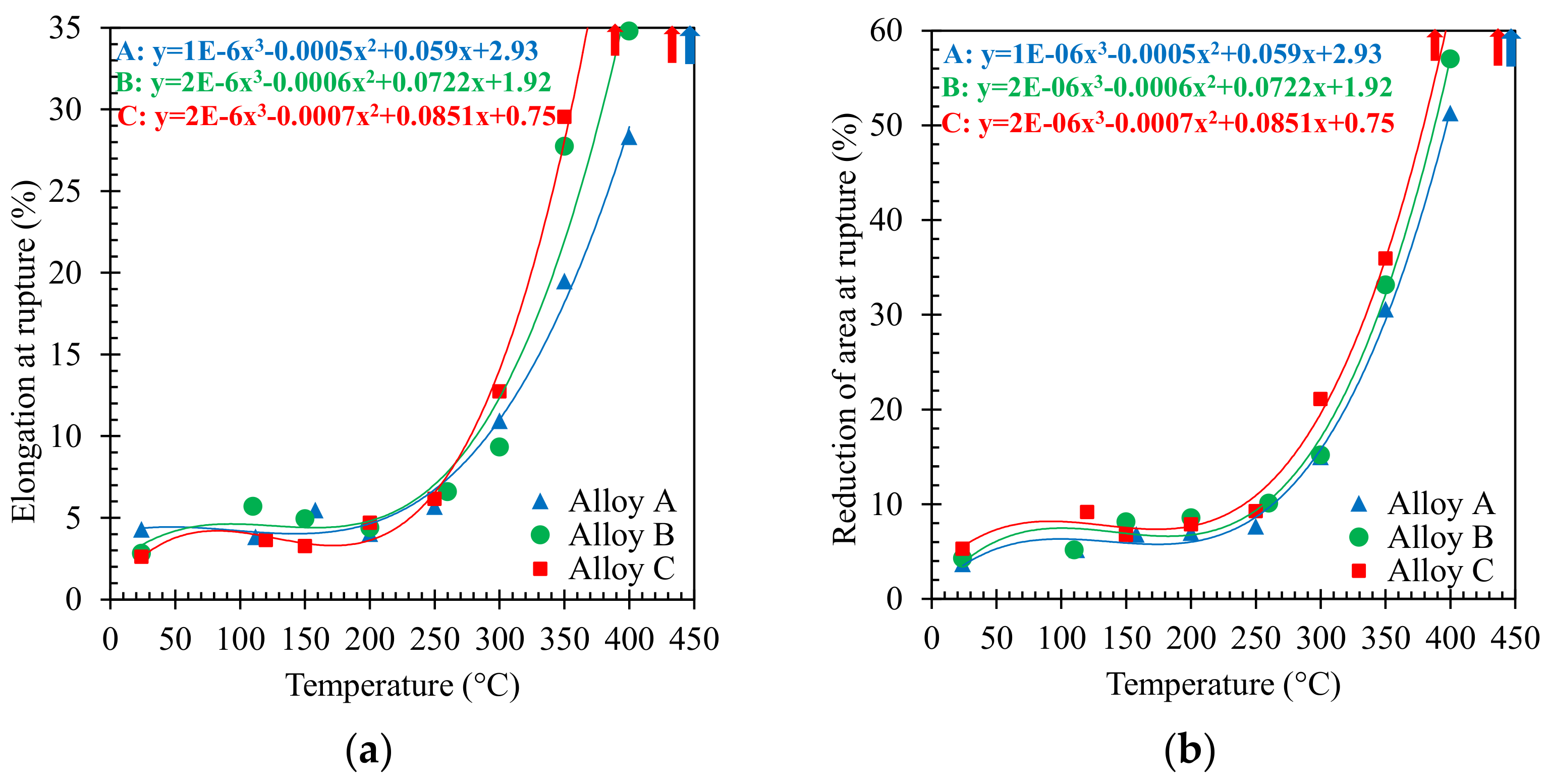
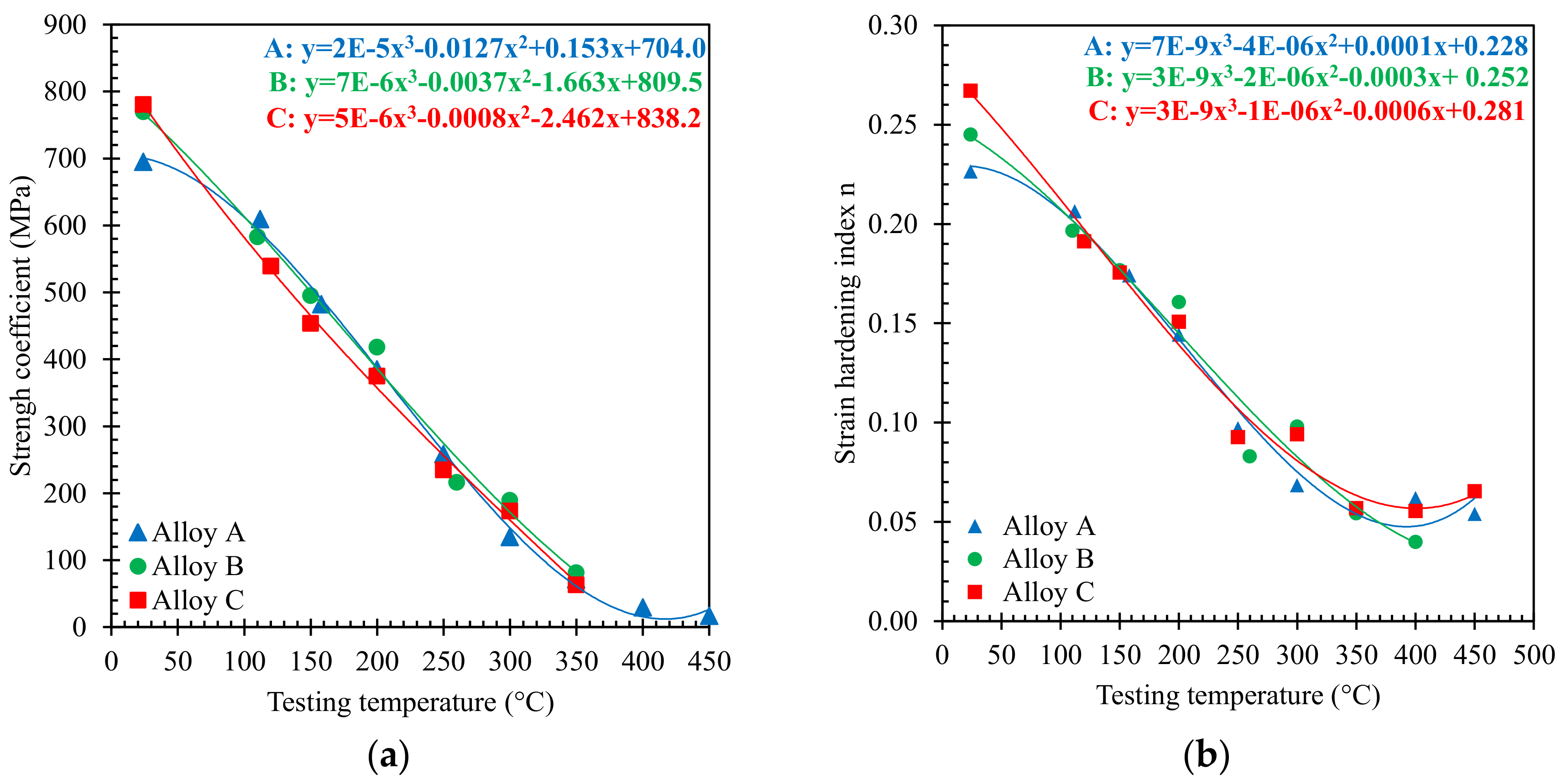
3.1. Tensile Behaviour
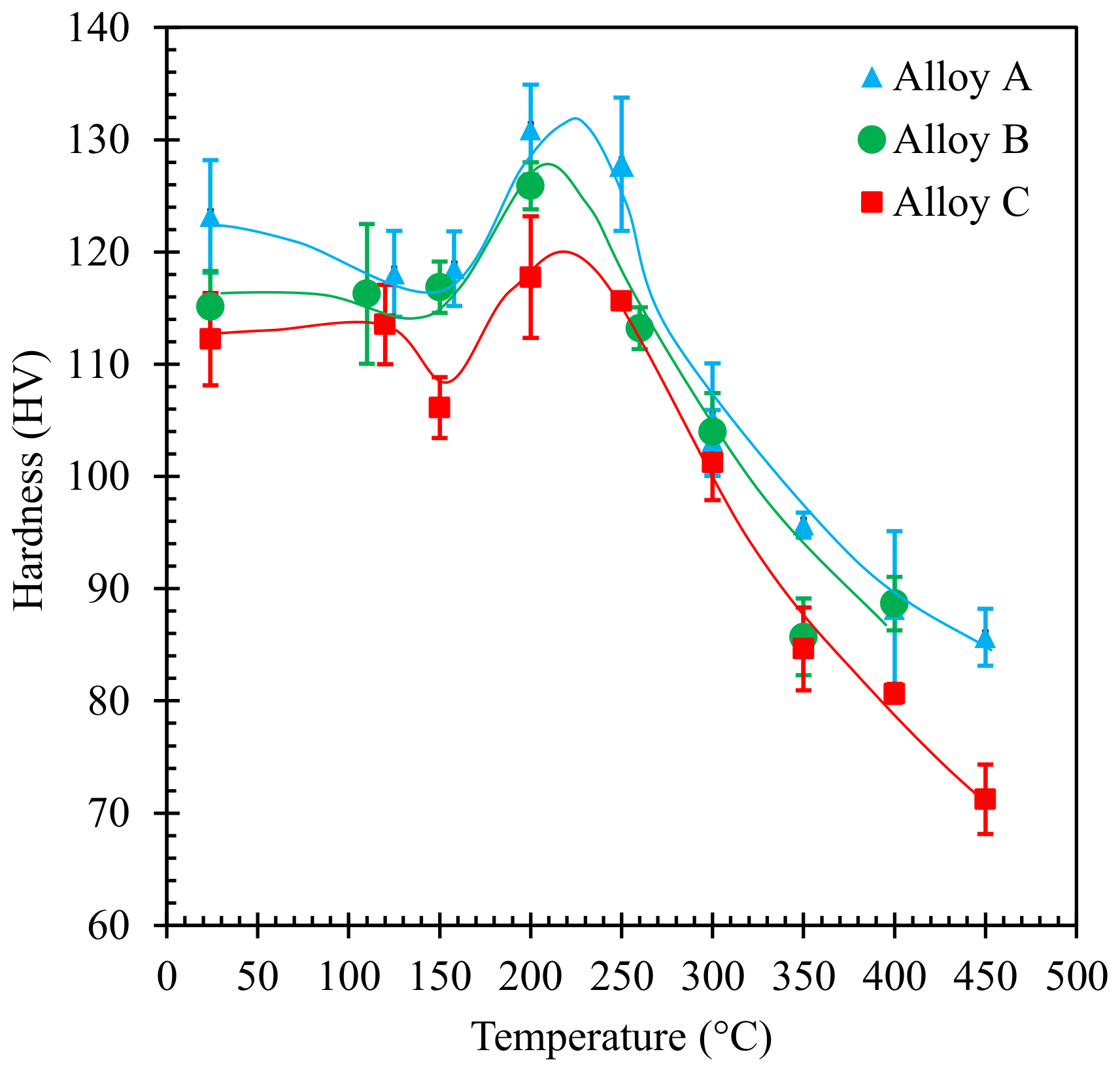
3.2. Hardness Testing
3.3. Microstructural Observations
4. Discussion
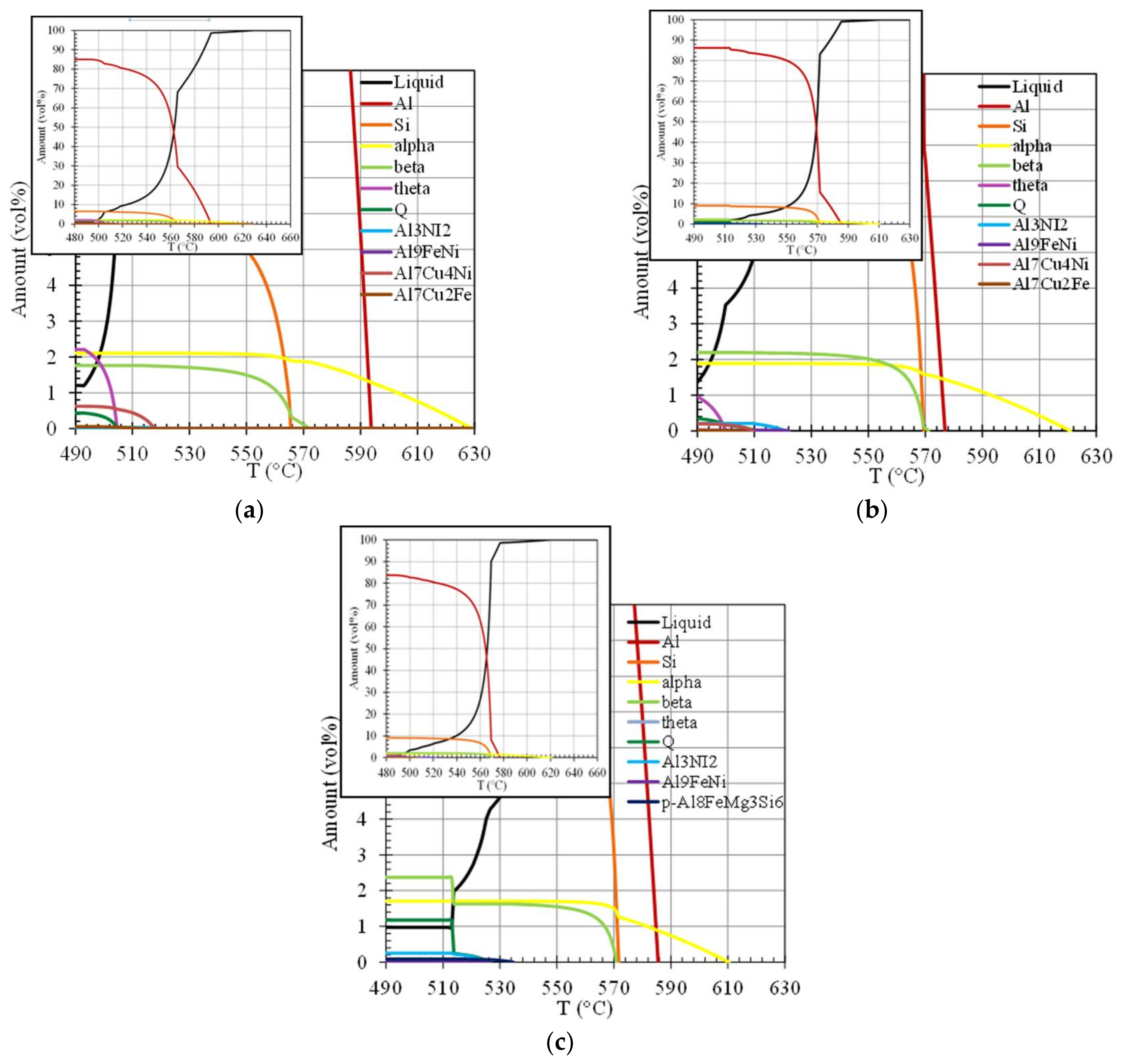
4.1. Thermodynamic Simulations and Microstructures/Properties at Room Temperature
4.2. High-Temperature Mechanical Behaviour and Microstructural Features
5. Conclusions
- The short-term tensile behavior of the three investigated Al-Si-Cu alloys showed a clear decrease of tensile properties above 200 °C, with a steep decrease occurring at testing temperatures between 250 and 350 °C, corresponding to a significant increase in ductility.
- The tensile behavior of alloys at high temperatures can be described by KT, nT parameters, for which simple correlations with temperature have been proposed.
- Material hardness and ductility indexes better evidence the differences in the mechanical behavior of the different alloys and can be related to the microstructural changes taking place at the test temperatures.
- Microstructural damage mechanisms are mainly correlated, at all of the investigated temperatures, to the amount and size of secondary phases and coarse intermetallic particles. The AlSi12Cu1(Fe) alloy, characterized by a lower amount of these particles, displayed the highest ductility indexes, particularly above 300 °C, when the highest ductility of its Alss grains combined to the lowest amount of coarse intermetallic phases characterizing this alloy.
- The hardness of specimens pulled at 150–250 °C suggests an age-hardening strengthening effect, even in the alloy stabilized at room temperature for a long time. The strengthening effect increased with the actual Cu content of the alloy, corresponding to a higher amount of fine particles formed within the Alss phase. Since their evolution is highly affected by the thermal cycle of the alloy, the actual peak temperature and hardness as well as other mechanical properties in the investigated temperature range are influenced by the thermal history of the material before the tensile test.
- The presence of coarse intermetallic phases in different alloys, related to their ductility, as well as the relevance of age-hardening effects by θ-Al2Cu and Q-Al5Cu2Mg8Si6 precipitation sequences, related to their strength at intermediate temperatures, can be derived from the results of thermodynamic simulations.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kaufman, J.G.; Rooy, E.L. Aluminium Alloy Castings: Properties, Processes and Applications; ASM International: Materials Park, OH, USA, 2004; pp. 61–68. ISBN 0-87170-803-5. [Google Scholar]
- Sjolander, E. Heat Treatment of Al-Si-Cu-Mg Casting Alloys. Ph.D. Thesis, Jönköping University, Jönköping, Sweden, 2011. [Google Scholar]
- Lumley, R.N.; ODonnell, R.G.; Gunasegaram, D.R.; Givord, M. Heat treatment of high-pressure die castings. Metall. Mater. Trans. 2007, 38, 2564–2574. [Google Scholar] [CrossRef]
- Hurtalova, L.; Tillova, E.; Chalupova, M. The structure analysis of secondary (recycled) AlSi9Cu3 cast alloy with and without heat treatment. Eng. Trans. 2013, 61, 197–218. [Google Scholar]
- Jeong, C.Y. Effect of alloying elements on high temperature mechanical properties for piston alloy. Mater. Trans. 2012, 53, 234–239. [Google Scholar] [CrossRef]
- Molina, R.; Amalberto, P.; Rosso, M. Mechanical characterization of aluminium alloys for high temperature applications Part1: Al-Si-Cu alloys. Metall. Sci. Technol. 2012, 29, 5–11. [Google Scholar]
- Ferguson, J.B.; Lopez, H.F.; Cho, K.; Kim, C.S. Temperature effects on the tensile properties of precipitation-hardened Al-Mg-Cu-Si alloys. Metals 2016, 6, 43. [Google Scholar] [CrossRef]
- Zamani, M.; Seifeddine, S.; Jarfors, A.E.W. High temperature tensile deformation behaviour and failure mechanisms of an Al–Si–Cu–Mg cast alloy—The microstructural scale effect. Mater. Des. 2015, 86, 361–370. [Google Scholar] [CrossRef]
- Yousefi, M.; Dehnavi, M.; Miresmaeili1, S.M. Microstructure and impression creep characteristics of Al-9Si-xCu aluminium alloys. Metall. Mater. Eng. 2015, 21, 115–125. [Google Scholar]
- EN 1706 Standard, Aluminium and Aluminium Alloys. Castings. Chemical Composition and Mechanical Properties; CEN–Comité Européen de Normalisation: Brussel, Belgium, 2007.
- ASTM B85/B85M—14 Standard Specification for Aluminum-Alloy Die Castings; ASTM International: West Conshohocken, PA, USA, 2014.
- Timelli, G.; Ferraro, S.; Groselle, F.; Bonollo, F.; Voltazza, F.; Capra, L. Caratterizzazione meccanica e microstrutturale di leghe di alluminio pressocolate. Metall. Ital. 2011, 1, 5–16. [Google Scholar]
- Timelli, G.; Bonollo, F. Quality mapping of aluminum alloy die castings. Metall. Sci. Technol. 2008, 26, 2–8. [Google Scholar]
- Ferrero, S.; Timelli, G. Influence of sludge particles on the tensile properties of die cast secondary aluminium alloys. Metall. Mater. Trans. B 2015, 46, 1022–1034. [Google Scholar] [CrossRef]
- Ozhoga-Maslovskaja, O.; Gariboldi, E.; Lemke, J.N. Conditions for blister formation during thermal cycles of Al-Si-Cu-Fe alloys for high pressure diecasting. Mater. Des. 2016, 92, 151–159. [Google Scholar] [CrossRef]
- ISO 6892-2:2011. Metallic Materials—Tensile Testing—Part 2: Method of Test at Elevated Temperature; ISO: Geneva, Switzerland, 2011.
- CEN T/R 16748:2014 Aluminium and Aluminium Alloys. Mechanical Potential of Al-Si Alloys for High Pressure, Low Pressure and Gravity Die Casting; CEN–Comité Européen de Normalisation: Brussel, Belgium, 2014.
- Ramberg, W.; Osgood, W.R. Description of Stress–Strain Currves by Three Parameters; Technical Note No. 902; National Advisory Committee for Aeronautics: Washington, DC, USA, 1943.
- Wiesner, D.J.; Watkins, T.R.; Ely, T.M.; Spooner, S.; Hubbard, C.R.; Williams, J.C. Residual stress measurements of cast aluminum engine blocks using diffraction. JCPDS-Int. Centre Diffr. Data Adv. X-ray Anal. 2005, 48, 136–142. [Google Scholar]
- Rossi, J.L.; Pilkington, R.; Trumper, R.L. Graphical Manipulation of EDS Data Obtained by SEM. In Proceedings of the 13 International Conference UMST X Ray Optics and Microanalysis 1992, Manchester, UK, 31 August–4 September 1992; pp. 275–278, ISBN 0-7503-0255-0. [Google Scholar]
- Hatch, J.E. Aluminium: Properties and Physical Metallurgy, 10th ed.; ASM International: Materials Park, OH, USA, 2005; p. 238. ISBN 978-0871701763. [Google Scholar]
- Rana, R.S.; Purohit, R.; Das, S. Reviews on the influences of alloying elements on the microstructure and mechanical properties of aluminum alloys and aluminum alloy composites. Int. J. Sci. Res. Publ. 2012, 2, 1–7. [Google Scholar]
- Belov, N.A.; Aksenov, A.A.; Eskin, D.G. Iron in Aluminium Alloys—Impurity and Alloying Element; Taylor and Francis: London, UK, 2002; pp. 78–80. ISBN 9780415273527. [Google Scholar]
- Kaufmann, H.; Fragner, W.; Uggowitzer, P.J. Influence of variations in alloy composition on castability and process stability. Part 1: Gravity and pressure casting. Int. J. Cast Met. Res. 2005, 18, 273–278. [Google Scholar] [CrossRef]
- Ferraro, S.; Fabrizi, A.; Timelli, G. Evolution of sludge particles in secondary die-cast aluminum alloys as function of Fe, Mn and Cr contents. Mater. Chem. Phys. 2015, 153, 168–179. [Google Scholar] [CrossRef]
- Dorè, X.; Combeau, X.; Rappaz, M. Modelling of microsegregation in ternary alloys: Application to the solidification of Al-Mg-Si. Acta Mater. 2000, 48, 3951–3962. [Google Scholar] [CrossRef]
- Kaufmann, H.; Uggowitzer, P.J. Metallurgy and Processing of High Integrity Light Metal Pressure Castings; Schiele & Schon: Berlin, Germany, 2007; pp. 7–12, 187–191. ISBN 9783794907540. [Google Scholar]
- Zoqui, E.J. Alloys for semisolid processing. In Comprehensive Materials Processing, Volume 5—Casting, Semi-Solid Forming and Hot Metal Forming; Hashmi, S., Ed.; Elsevier: New York, NY, USA, 2014; pp. 163–188. ISBN 9780080965338. [Google Scholar]
- Shabestari, S.G. The effect of iron and manganese on the formation of intermetallic compounds in aluminum–silicon alloys. Mater. Sci. Eng. A 2004, 383, 289–298. [Google Scholar] [CrossRef]
- Cao, X. Effect of iron and manganese contents on convection-free precipitation and sedimentation of primaryα-Al(FeMn)Si phase in liquid Al-11.5Si-0.4Mg alloy. J. Mater. Sci. 2004, 39, 2303–2314. [Google Scholar] [CrossRef]
- Ferdian, M.D. Effect de la Vitesse de Refroidissment sur la Taille des Grains, la Modification Eutectique et la Precipitation D’intermetalliques Riches en fer dans des Alliages Al-Si Hypotectiques. Ph.D. Thesis, Universitè de Tolouse, Toulouse, France, 2014. [Google Scholar]
- Karl, M.V. A crystallographic identification of intermetallic phases in Al–Si alloys. Mater. Lett. 2005, 59, 2271–2276. [Google Scholar] [CrossRef]
- Taylor, J.A. Iron-containing intermetallic phases in Al-Si based casting alloys. Procedia Mater. Sci. 2012, 1, 19–33. [Google Scholar] [CrossRef]
- Ji, S.; Yang, W.; Gao, F.; Watson, D.; Fan, Z. Effect of iron on the microstructure and mechanical property of Al–Mg–Si–Mn and Al–Mg–Si diecast alloys. Mater. Sci. Eng. A 2013, 564, 30–139. [Google Scholar] [CrossRef]
- Timelli, G.; Fabrizi, A. The effects of microstructure heterogeneities and casting defects on the mechanical properties of high-pressure die-cast AlSi9Cu3(Fe) Alloys. Metall. Mater. Trans. A 2014, 45, 5486–5498. [Google Scholar] [CrossRef]
- Mohamed, A.M.A.; Samuel, A.M.; Samuel, F.H.; Doty, H.W. Influence of additives on the microstructure and tensile properties of near-eutectic Al–10.8% Si cast alloy. Mater. Des. 2009, 30, 3943–3957. [Google Scholar] [CrossRef]
- Czerwinski, F.; Shaha, S.K.; Kasprzak, W.; Friedman, J.; Chen, D.L. Aging characteristics of the Al-Si-Cu-Mg cast alloy modified with transition metals Zr, V and Ti. IOP Conf. Ser. Mater. Sci. Eng. 2016, 117, 012031. [Google Scholar] [CrossRef]
- Bassani, P.; Gariboldi, E.; Ripamonti, D. Thermal analysis of Al–Cu–Mg–Si alloy with Ag/Zr additions. J. Therm. Anal. Calorim. 2008, 91, 29–35. [Google Scholar] [CrossRef]
- Samuel, A.M.; Gauthier, J.; Samuel, F.H. Microstructural Aspects of the Dissolution and Melting of Al2Cu Phase in Al-Si Alloys during Solution Heat Treatment. Metall. Mater. Trans. A 1996, 27, 1785–1798. [Google Scholar] [CrossRef]






| Alloy | Si | Fe | Cu | Mn | Mg | Cr | Ni | Zn | Pb | Sn | Ti | Al | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | EN AC 46000-AlSi9Cu3Fe | actual | 8.227 | 0.799 | 2.825 | 0.261 | 0.252 | 0.083 | 0.081 | 0.895 | 0.083 | 0.026 | 0.041 | bal |
| limits | 8.0–11.0 | <1.3 | 2.0–4.0 | <0.55 | 0.05–0.55 | <0.15 | <0.55 | <1.20 | <0.35 | 0.15 | <0.25 | bal | ||
| B | EN AC 46100-AlSi11Cu2(Fe) | actual | 10.895 | 0.889 | 1.746 | 0.219 | 0.224 | 0.082 | 0.084 | 1.274 | 0.089 | 0.029 | 0.047 | bal |
| limits | 10.0–12.0 | <1.1 | 1.5–2.5 | <0.55 | <0.30 | <0.15 | <0.45 | <1.70 | <0.25 | 0.15 | <0.25 | bal | ||
| C | EN AC 47100-AlSi12Cu1(Fe) | actual | 10.510 | 0.721 | 0.941 | 0.232 | 0.242 | 0.045 | 0.080 | 0.354 | 0.055 | 0.025 | 0.038 | bal |
| limits | 10.5–13.5 | <1.3 | 0.7–1.2 | <0.55 | <0.35 | <0.10 | <0.30 | <0.55 | <0.20 | 0.10 | <0.20 | bal | ||
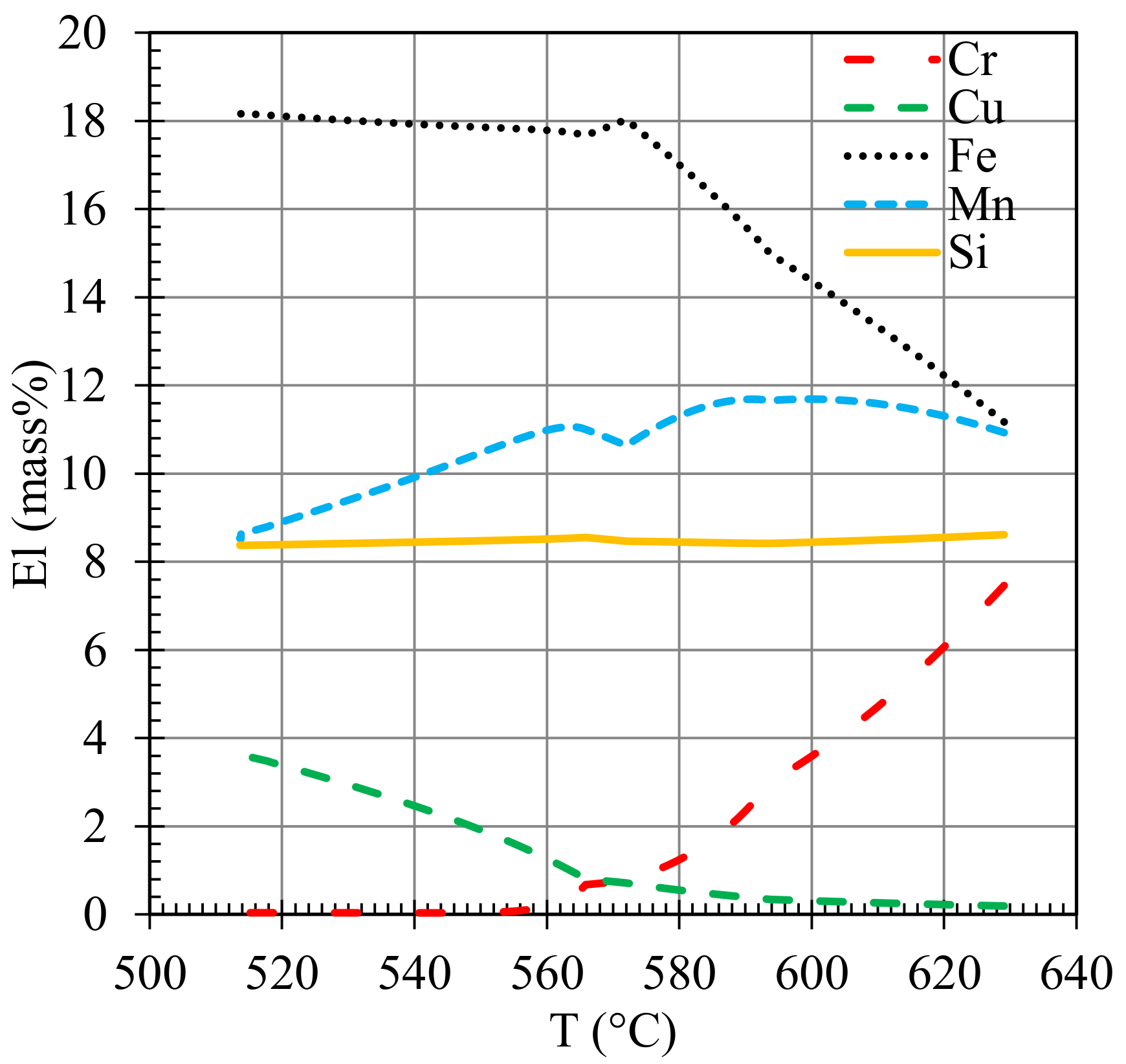
| Alloy | Figure | Point | Si | Fe | Cu | Mn | Mg | Cr | Ni | Al |
|---|---|---|---|---|---|---|---|---|---|---|
| A | 9c | A | 4.06 | 1.59 | 5.50 | - | - | - | Bal | |
| A | 9c | B | 8.57 | 16.07 | 4.94 | 4.85 | - | 1.20 | Bal | |
| A | 9c | C | 35.81 | 2.90 | 22.04 | - | 1.18 | 0.81 | Bal | |
| B | 9f | A | 8.54 | 5.39 | 13.27 | - | - | - | - | Bal |
| B | 9f | B | 8.82 | - | 24.95 | - | - | - | - | Bal |
| B | 9f | C | 4.35 | - | 25.4 | - | - | - | 2.18 | Bal |
| C | 9g | A | 11.59 | 12.14 | 0.61 | 4.85 | - | 1.17 | - | Bal |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gariboldi, E.; Lemke, J.N.; Rovatti, L.; Baer, O.; Timelli, G.; Bonollo, F. High-Temperature Behavior of High-Pressure Diecast Alloys Based on the Al-Si-Cu System: The Role Played by Chemical Composition. Metals 2018, 8, 348. https://doi.org/10.3390/met8050348
Gariboldi E, Lemke JN, Rovatti L, Baer O, Timelli G, Bonollo F. High-Temperature Behavior of High-Pressure Diecast Alloys Based on the Al-Si-Cu System: The Role Played by Chemical Composition. Metals. 2018; 8(5):348. https://doi.org/10.3390/met8050348
Chicago/Turabian StyleGariboldi, Elisabetta, Jannis Nicolas Lemke, Ludovica Rovatti, Oksana Baer, Giulio Timelli, and Franco Bonollo. 2018. "High-Temperature Behavior of High-Pressure Diecast Alloys Based on the Al-Si-Cu System: The Role Played by Chemical Composition" Metals 8, no. 5: 348. https://doi.org/10.3390/met8050348
APA StyleGariboldi, E., Lemke, J. N., Rovatti, L., Baer, O., Timelli, G., & Bonollo, F. (2018). High-Temperature Behavior of High-Pressure Diecast Alloys Based on the Al-Si-Cu System: The Role Played by Chemical Composition. Metals, 8(5), 348. https://doi.org/10.3390/met8050348








