Influence of Scandium Addition on Stress Corrosion Cracking Susceptibility of Al-Zn-Mg Alloy in Different Corrosive Environments
Abstract
:1. Introduction
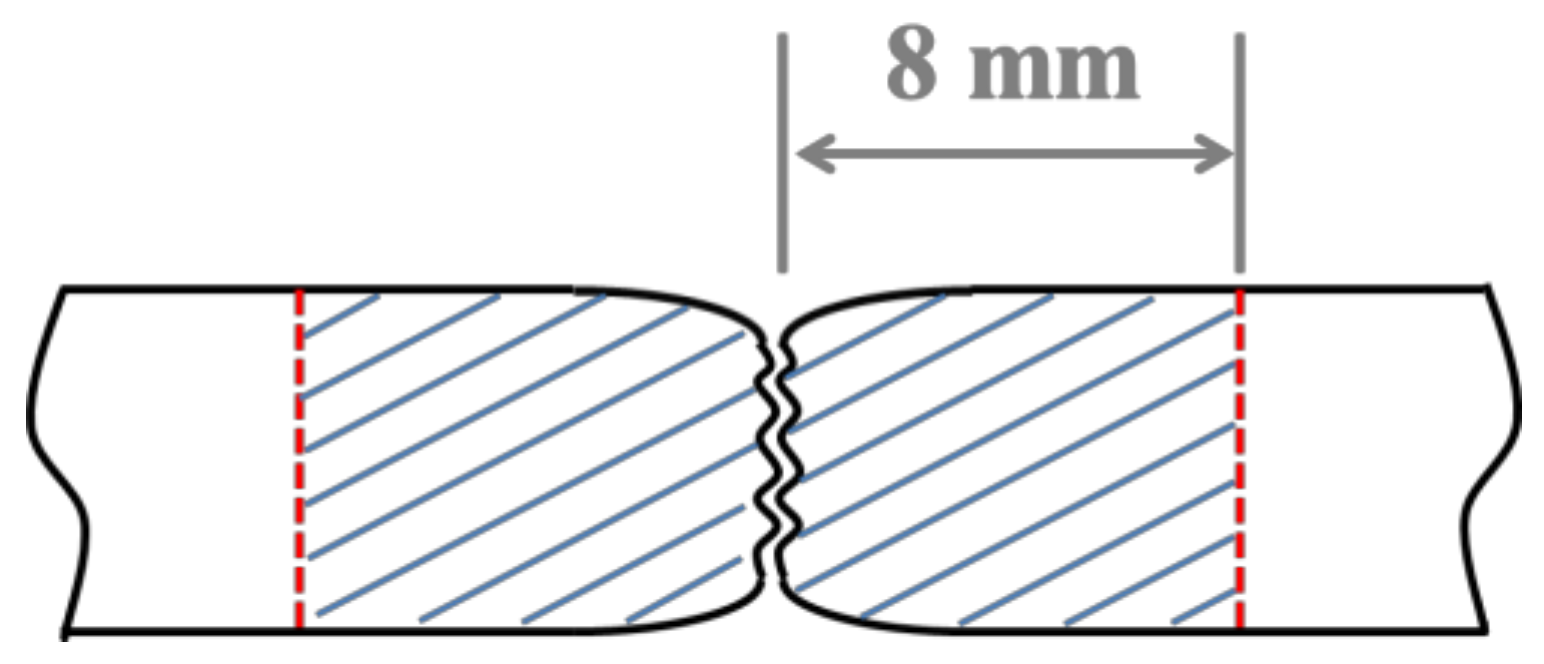
2. Materials and Methods
3. Results
3.1. The Microstructures of Al-Zn-Mg Alloys without and with Sc Addition
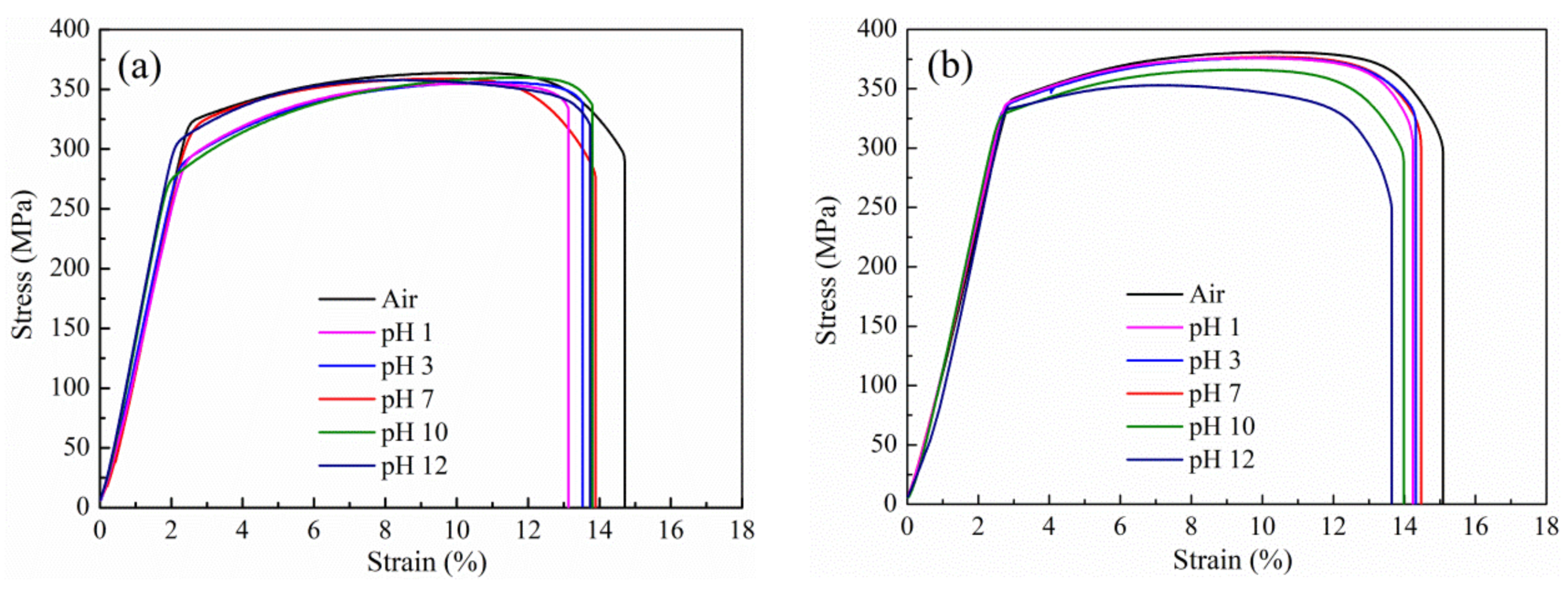
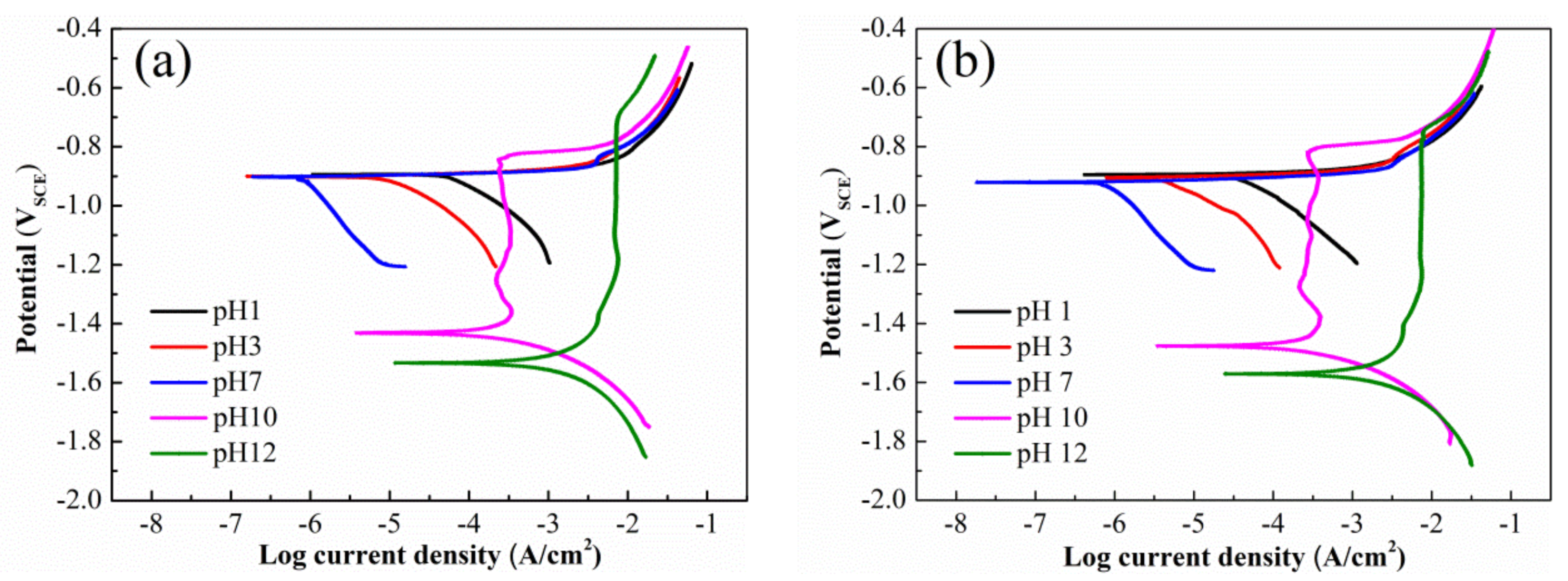
3.2. SCC Susceptibilities of Al-Zn-Mg Alloys without and with Sc Addition in Solution at Different pH
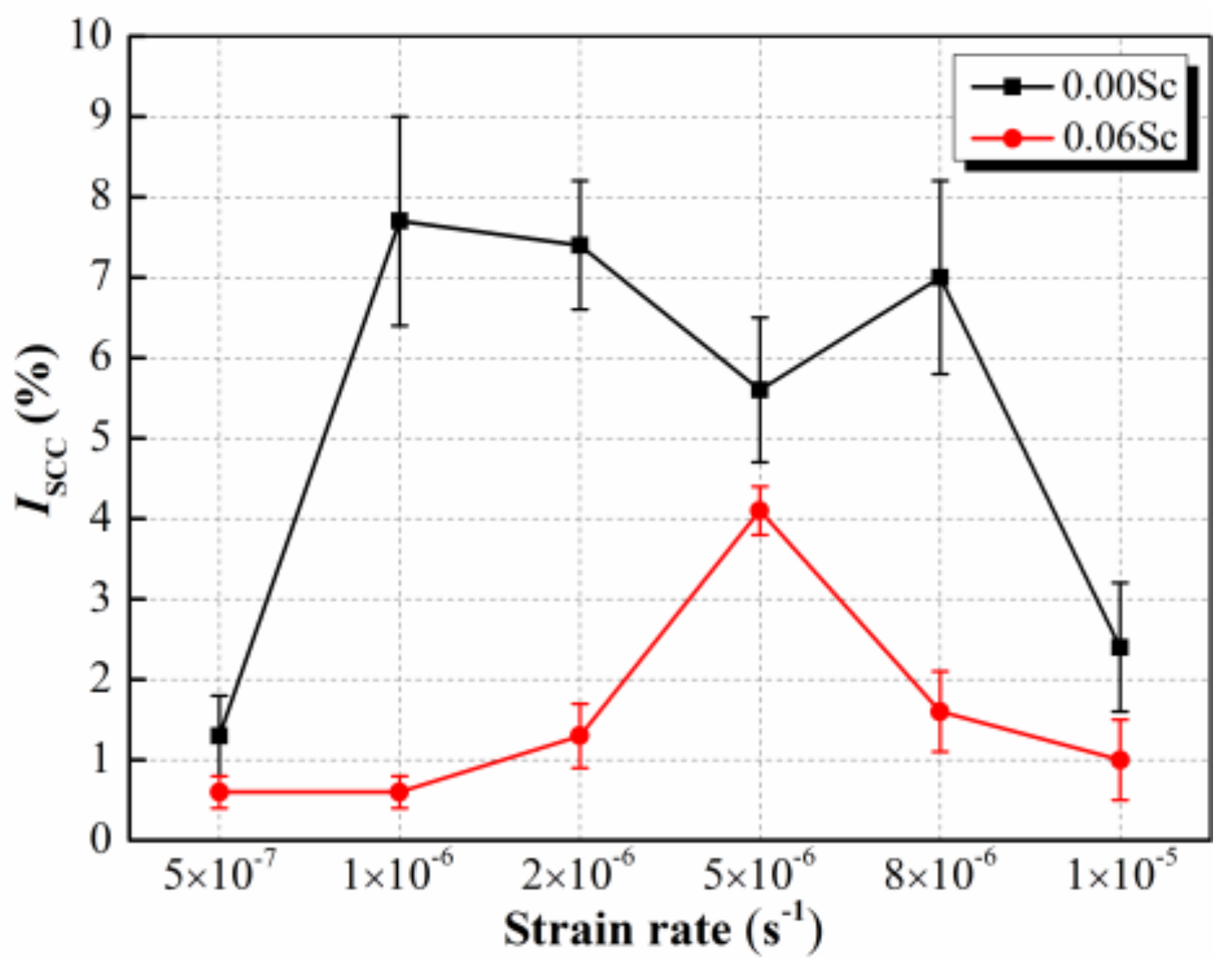
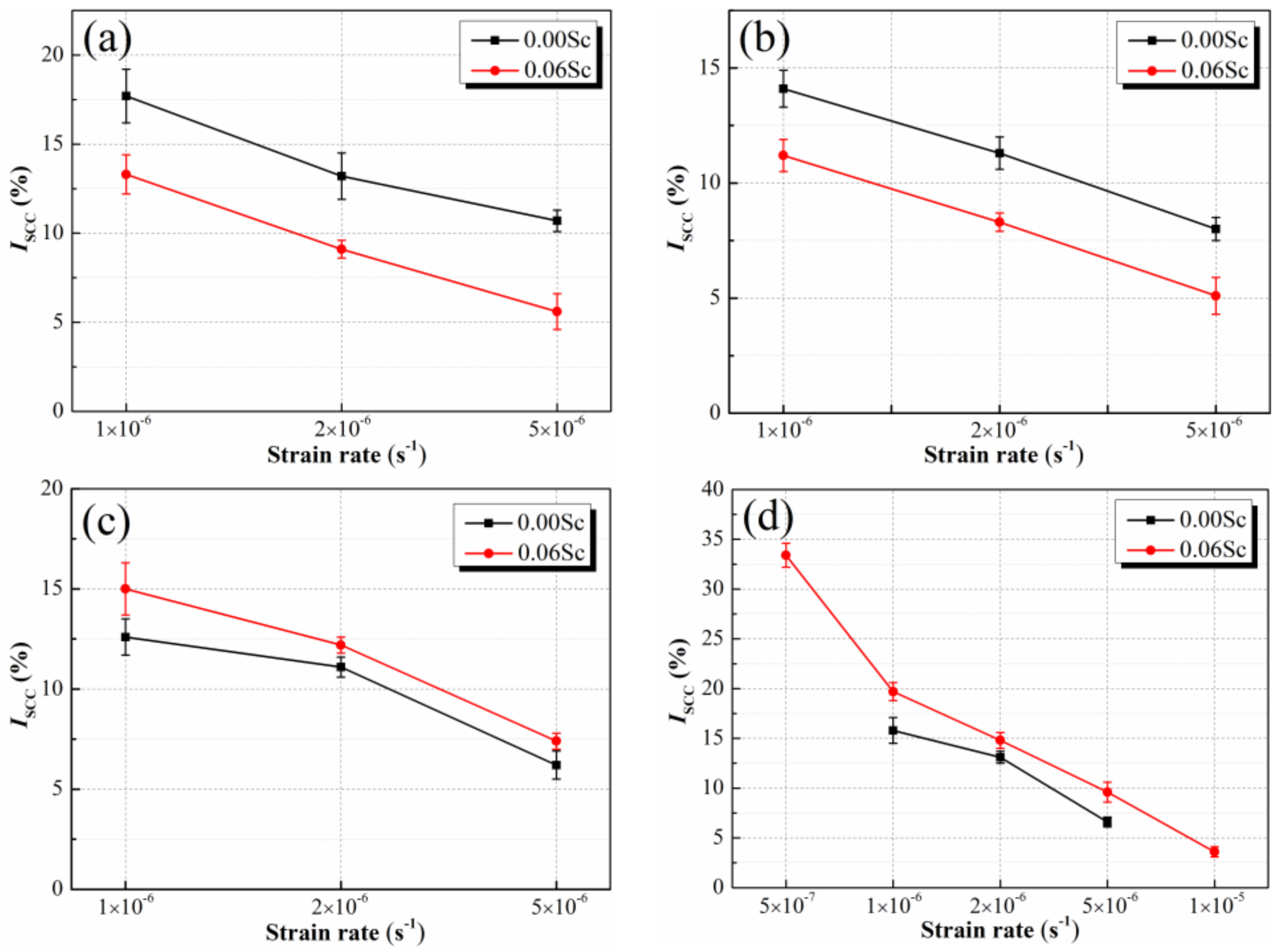
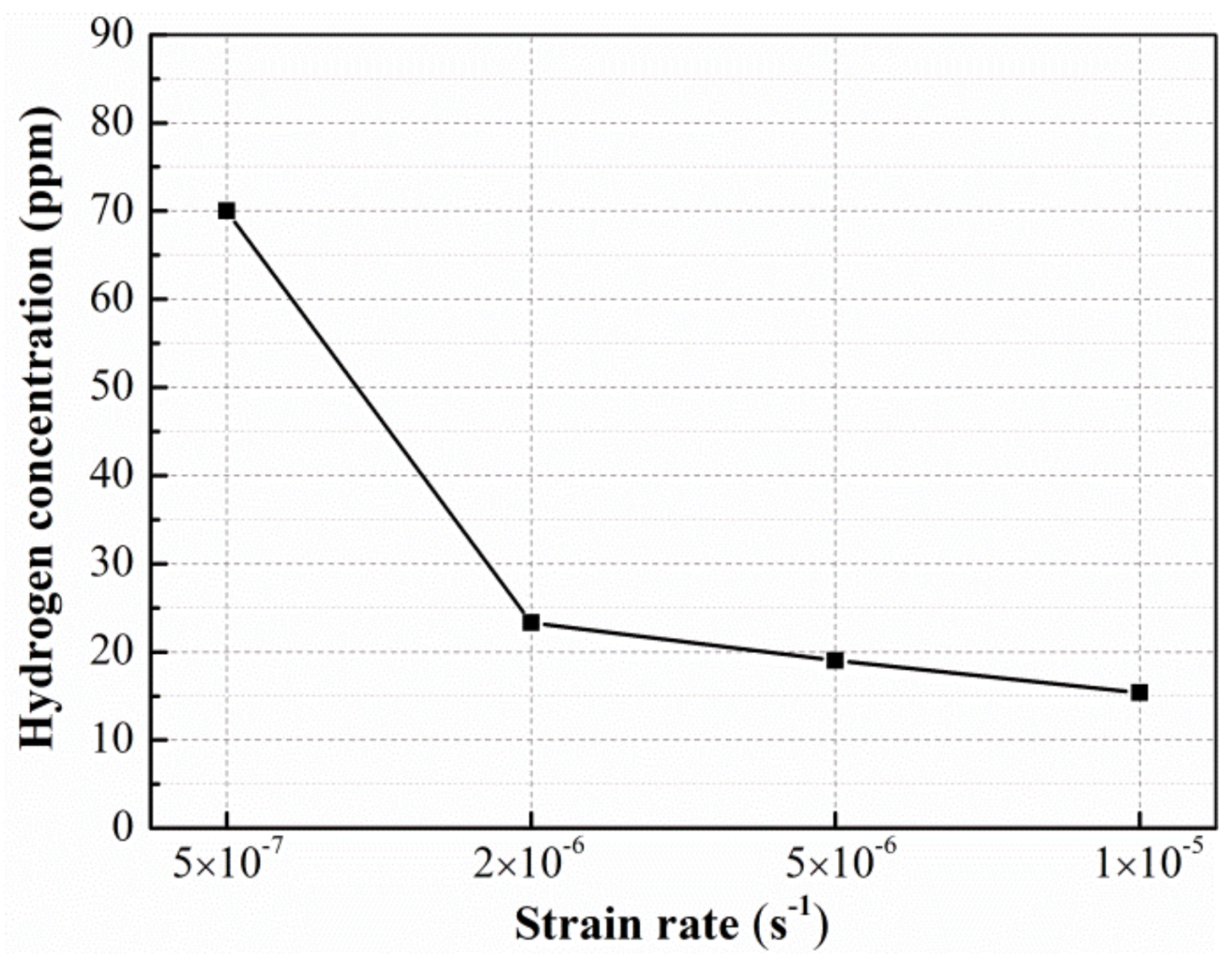
3.3. SCC Susceptibilities of Al-Zn-Mg Alloys without and with Sc Addition at Different Strain Rate
4. Discussion
4.1. Effect of Sc Addition on SCC Susceptibility of Al-Zn-Mg Alloy in Solution at Different pH
4.2. Effect of Sc Addition on SCC Susceptibility of Al-Zn-Mg Alloy at Different Strain Rate
5. Conclusions
- Sc addition reduces grain size and width of PFZ, and transforms GBPs from continuous distribution into interrupted distribution by inhibiting recrystallization.
- In neutral and acid 3.5% NaCl solution, Sc addition hinders SCC crack initiation and growth, thus reduces SCC susceptibility of Al-Zn-Mg alloy. However, in alkaline 3.5% NaCl solution, hydrogen atoms generated by reduction reaction can be absorbed into the matrix during SSRT process. Grain refinement significantly promotes the diffusion of hydrogen atoms into matrix, thus Sc addition increases SCC susceptibility of Al-Zn-Mg alloy.
- Sc addition can reduce the relatively susceptible strain rate range to SCC of Al-Zn-Mg alloy due to the improved resistance to localized corrosion of alloy surface in neutral 3.5% NaCl solution.
- In acidic 3.5% NaCl solution, reducing strain rate sharply aggravates the degree of localized corrosion of alloy surface, thus increases SCC susceptibilities of two aged alloys, and Sc addition reduces SCC susceptibility of Al-Zn-Mg alloy at different strain rate. However, in alkaline 3.5% NaCl solution, reducing strain rate increases the time available for substantial hydrogen entry and segregation, resulting in the increase of SCC susceptibilities of two aged alloys, and Sc addition increases SCC susceptibility of Al-Zn-Mg alloy at different strain rate due to grain refinement.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- He, J.L.; Zhang, D.T.; Zhang, W.W.; Qiu, C.; Zhang, W. Constitutive equation and hot compression deformation behavior of homogenized Al-7.5Zn-1.5Mg-0.2Cu-0.2Zr alloy. Materials 2017, 10, 1193. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.F.; Lei, Q.; Xiao, Z.; Wang, M.P. Precipitation behavior and quenching sensitivity of a spray deposited Al-Zn-Mg-Cu-Zr alloy. Materials 2017, 10, 1100. [Google Scholar] [CrossRef] [PubMed]
- Deschamps, A.; Ringeval, S.; Texier, G.; Delfaut-Durut, L. Quantitative characterization of the microstructure of an electron-beam welded medium strength Al-Zn-Mg alloy. Mater. Sci. Eng. A 2009, 517, 361–368. [Google Scholar] [CrossRef]
- Shi, C.J.; Lai, J.; Chen, X.G. Microstructural evolution and dynamic softening mechanisms of Al-Zn-Mg-Cu alloy during hot compressive deformation. Materials 2014, 7, 244–264. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; Wang, X.S.; Wang, Y.M.; Wang, Y.L.; Zhang, Z.H. Microarc oxidation coating combined with surface pore-sealing treatment enhances corrosion fatigue performance of 7075-T7351 Al alloy in different media. Materials 2017, 10, 609. [Google Scholar] [CrossRef] [PubMed]
- Harding, M.D.; Donaldson, I.W.; Junior, R.L.H.; Bishop, D.P. Effects of post-sinter processing on an Al-Zn-Mg-Cu powder metallurgy alloy. Metals 2017, 7, 370. [Google Scholar] [CrossRef]
- Qin, C.; Gou, G.Q.; Che, X.L.; Chen, H.; Chen, J.; Li, P.; Gao, W. Effect of composition on tensile properties and fracture toughness of Al-Zn-Mg alloy (A7N01S-T5) used in high speed trains. Mater. Des. 2016, 91, 278–285. [Google Scholar] [CrossRef]
- Liu, J.; Yao, P.; Zhao, N.; Shi, C.; Li, H.; Li, X.; Xi, D.; Yang, S. Effect of minor Sc and Zr on recrystallization behavior and mechanical properties of novel Al-Zn-Mg-Cu alloys. J. Alloy. Compd. 2016, 657, 717–725. [Google Scholar] [CrossRef]
- Chen, S.Y.; Chen, K.H.; Dong, P.X.; Ye, S.P.; Huang, L.P. Effect of recrystallization and heat treatment on strength and SCC of an Al-Zn-Mg-Cu alloy. J. Alloy. Compd. 2013, 581, 705–709. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Zhan, L.H. Effect of creep aging process on microstructures and properties of the retrogressed Al-Zn-Mg-Cu alloy. Metals 2016, 6, 189. [Google Scholar] [CrossRef]
- Kurt, H.I.; Oduncuoglu, M.; Kurt, M. A mathematical formulation to estimate the effect of grain refiners on the ultimate tensile strength of Al-Zn-Mg-Cu alloys. Metals 2015, 5, 836–849. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.L.; Ma, L.Y.; Yi, J.H. Effects of solution treatment on microstructure and high-cycle fatigue properties of 7075 aluminum alloy. Metals 2017, 7, 193. [Google Scholar] [CrossRef]
- Minoda, T.; Yoshida, H. Effect of grain boundary characteristics on intergranular corrosion resistance of 6061 aluminum alloy extrusion. Metall. Mater. Trans. A 2002, 33, 2891–2898. [Google Scholar] [CrossRef]
- Shi, Y.J.; Pan, Q.L.; Li, M.J.; Huang, X.; Li, B. Effect of Sc and Zr additions on corrosion behaviour of Al-Zn-Mg-Cu alloys. J. Alloy. Compd. 2014, 612, 42–50. [Google Scholar] [CrossRef]
- Fang, H.C.; Chen, K.H.; Chen, X.; Chao, H.; Peng, G.S. Effect of Cr, Yb and Zr additions on localized corrosion of Al-Zn-Mg-Cu alloy. Corros. Sci. 2009, 51, 2872–2877. [Google Scholar] [CrossRef]
- Shi, Y.J.; Pan, Q.L.; Li, M.J.; Huang, X.; Li, B. Influence of alloyed Sc and Zr, and heat treatment on microstructures and stress corrosion cracking of Al-Zn-Mg-Cu alloys. Mater. Sci. Eng. A 2015, 621, 173–181. [Google Scholar] [CrossRef]
- Kannan, M.B.; Raja, V.S. Enhancing stress corrosion cracking resistance in Al-Zn-Mg-Cu-Zr alloy through inhibiting recrystallization. Eng. Fract. Mech. 2010, 77, 249–256. [Google Scholar] [CrossRef]
- Deng, Y.; Yin, Z.M.; Zhao, K.; Duan, J.Q.; Hu, J.; He, Z.B. Effects of Sc and Zr microalloying additions and aging time at 120 degrees C on the corrosion behaviour of an Al-Zn-Mg alloy. Corros. Sci. 2012, 65, 288–298. [Google Scholar] [CrossRef]
- Huang, X.; Pan, Q.L.; Li, B.; Liu, Z.M.; Huang, Z.Q.; Yin, Z.M. Microstructure, mechanical properties and stress corrosion cracking of Al-Zn-Mg-Zr alloy sheet with trace amount of Sc. J. Alloy. Compd. 2015, 650, 805–820. [Google Scholar] [CrossRef]
- Kannan, M.B.; Raja, V.S.; Mukhopadhyay, A.K.; Schmuki, P. Environmentally assisted cracking behavior of peak-aged 7010 aluminum alloy containing scandium. Metall. Mater. Trans. A 2005, 36, 3257–3262. [Google Scholar] [CrossRef]
- Deng, Y.; Yin, Z.M.; Zhao, K.; Duan, J.Q.; Hu, J.; He, Z.B. Effects of Sc and Zr microalloying additions on the microstructure and mechanical properties of new Al-Zn-Mg alloys. J. Alloy. Compd. 2012, 530, 71–80. [Google Scholar] [CrossRef]
- Li, G.; Zhao, N.Q.; Liu, T.; Li, J.J.; He, C.N.; Shi, C.S.; Liu, E.Z.; Sha, J.W. Effect of Sc/Zr ratio on the microstructure and mechanical properties of new type of Al-Zn-Mg-Sc-Zr alloys. Mater. Sci. Eng. A 2014, 617, 219–227. [Google Scholar] [CrossRef]
- Tsai, W.T.; Duh, J.B.; Yeh, J.J.; Lee, J.T.; Chang, Y.C. Effect of pH on stress corrosion cracking of 7050-T7451 aluminum alloy in 3.5 wt % NaCl solution. Corrosion 1990, 46, 444–449. [Google Scholar] [CrossRef]
- Rout, P.K.; Ghosh, M.M.; Ghosh, K.S. Effect of solution pH on electrochemical and stress corrosion cracking behaviour of a 7150 Al-Zn-Mg-Cu alloy. Mater. Sci. Eng. A 2014, 604, 156–165. [Google Scholar] [CrossRef]
- Deschamps, A.; Texier, G.; Ringeval, S.; Delfaut-Durut, L. Influence of cooling rate on the precipitation microstructure in a medium strength Al-Zn-Mg alloy. Mater. Sci. Eng. A 2009, 501, 133–139. [Google Scholar] [CrossRef]
- Wloka, J.; Virtanen, S. Influence of scandium on the pitting behaviour of Al-Zn-Mg-Cu alloys. Acta. Mater. 2007, 55, 6666–6672. [Google Scholar] [CrossRef]
- Ralston, K.D.; Birbilis, N.; Davies, C.H.J. Revealing the relationship between grain size and corrosion rate of metals. Scr. Mater. 2010, 63, 1201–1204. [Google Scholar] [CrossRef]
- Ganiev, I.N. Corrosion-electrochemical behavior of special-purity aluminum and its AK1 alloy alloyed with Scandium. Russ. J. Appl. Chem. 2004, 77, 925–929. [Google Scholar] [CrossRef]
- Santamaria, M.; Muratore, F.; Di Quarto, F. Growth and characterization of anodic films on Scandium. J. Electrochem. Soc. 2014, 161, C36–C41. [Google Scholar] [CrossRef]
- Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solutions, 2nd ed.; National Association of Corrosion Engineers: Houston, TX, USA, 1974; pp. 177–182. [Google Scholar]
- Sun, X.Y.; Zhang, B.; Lin, H.Q.; Zhou, Y.; Sun, L.; Wang, J.Q.; Han, E.H.; Ke, W. Correlations between stress corrosion cracking susceptibility and grain boundary microstructures for an Al-Zn-Mg alloy. Corros. Sci. 2013, 77, 103–112. [Google Scholar] [CrossRef]
- Garcia-Garcia, F.J.; Skeldon, P.; Thompson, G.E.; Smithc, G.C. The effect of nickel on alloy microstructure and electrochemical behaviour of AA1050 aluminium alloy in acid and alkaline solutions. Electrochim. Acta 2012, 75, 229–238. [Google Scholar] [CrossRef]
- Boukerche, I.; Djerad, S.; Benmansour, L.; Tifouti, L.; Saleh, K. Degradability of aluminum in acidic and alkaline solutions. Corros. Sci. 2014, 78, 343–352. [Google Scholar] [CrossRef]
- Najjar, D.; Magnin, T.; Warner, T.J. Influence of critical surface defects and localized competition between anodic dissolution and hydrogen effects during stress corrosion cracking of a 7050 aluminium alloy. Mater. Sci. Eng. A 1997, 238, 293–302. [Google Scholar] [CrossRef]
- Argade, G.R.; Kumar, N.; Mishra, R.S. Stress corrosion cracking susceptibility of ultrafine grained Al-Mg-Sc alloy. Mater. Sci. Eng. A 2013, 565, 80–89. [Google Scholar] [CrossRef]
- Albrecht, J.; Bernstein, I.M.; Thompson, A.W. Evidence for dislocation transport of hydrogen in aluminum. Metall. Mater. Trans. A 1982, 13, 811–820. [Google Scholar] [CrossRef]
- Lynch, S. Mechanistic and fractographic aspects of stress corrosion cracking. Corros. Rev. 2012, 30, 63–104. [Google Scholar] [CrossRef]
- Young, G.A.; Scully, J.R. The diffusion and trapping of hydrogen in high purity aluminum. Acta Mater. 1998, 46, 6337–6349. [Google Scholar] [CrossRef]




| Alloys | Zn | Mg | Mn | Cr | Sc | Zr | Ti | Al |
|---|---|---|---|---|---|---|---|---|
| 0.00Sc | 4.19 | 1.36 | 0.32 | 0.21 | - | 0.10 | 0.06 | Bal. |
| 0.06Sc | 4.08 | 1.35 | 0.30 | 0.19 | 0.06 | 0.11 | 0.06 | Bal. |
| Corrosive Environments | UTS (MPa) | Ef (%) | ISCC (%) | |||
|---|---|---|---|---|---|---|
| 0.00Sc | 0.06Sc | 0.00Sc | 0.06Sc | 0.00Sc | 0.06Sc | |
| Air | 364 ± 4 | 381 ± 3 | 14.7 ± 0.3 | 15.1 ± 0.3 | - | - |
| pH 1 | 355 ± 5 | 376 ± 1 | 13.1 ± 0.2 | 14.2 ± 0.3 | 10.7 ± 0.6 | 5.6 ± 1.0 |
| pH 3 | 356 ± 2 | 376 ± 3 | 13.5 ± 0.2 | 14.3 ± 0.2 | 8.0 ± 0.5 | 5.1 ± 0.8 |
| pH 7 | 359 ± 5 | 377 ± 5 | 13.9 ± 0.2 | 14.5 ± 0.1 | 5.6 ± 0.9 | 4.1 ± 0.3 |
| pH 10 | 360 ± 6 | 366 ± 2 | 13.8 ± 0.2 | 14.0 ± 0.2 | 6.2 ± 0.7 | 7.4 ± 0.4 |
| pH 12 | 358 ± 3 | 353 ± 6 | 13.7 ± 0.1 | 13.6 ± 0.2 | 6.6 ± 0.4 | 9.6 ± 1.0 |
| Solution pH | Icorr (μA·cm−2) | Ecorr (VSCE) | ||
|---|---|---|---|---|
| 0.00Sc | 0.06Sc | 0.00Sc | 0.06Sc | |
| pH 1 | 66.073 ± 0.675 | 39.410 ± 9.393 | −0.894 ± 0.003 | −0.895 ± 0.003 |
| pH 3 | 11.078 ± 1.375 | 4.090 ± 0.096 | −0.901 ± 0.003 | −0.905 ± 0.004 |
| pH 7 | 0.901 ± 0.008 | 0.860 ± 0.004 | −0.902 ± 0.002 | −0.921 ± 0.005 |
| pH 10 | 253.219 ± 16.878 | 223.794 ± 11.977 | −1.431 ± 0.005 | −1.476 ± 0.006 |
| pH 12 | 1009.224 ± 40.130 | 1547.487 ± 185.988 | −1.533 ± 0.001 | −1.571 ± 0.004 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Jiang, H.; Yan, D.; Rong, L. Influence of Scandium Addition on Stress Corrosion Cracking Susceptibility of Al-Zn-Mg Alloy in Different Corrosive Environments. Metals 2018, 8, 225. https://doi.org/10.3390/met8040225
Li Z, Jiang H, Yan D, Rong L. Influence of Scandium Addition on Stress Corrosion Cracking Susceptibility of Al-Zn-Mg Alloy in Different Corrosive Environments. Metals. 2018; 8(4):225. https://doi.org/10.3390/met8040225
Chicago/Turabian StyleLi, Zhaoming, Haichang Jiang, Desheng Yan, and Lijian Rong. 2018. "Influence of Scandium Addition on Stress Corrosion Cracking Susceptibility of Al-Zn-Mg Alloy in Different Corrosive Environments" Metals 8, no. 4: 225. https://doi.org/10.3390/met8040225
APA StyleLi, Z., Jiang, H., Yan, D., & Rong, L. (2018). Influence of Scandium Addition on Stress Corrosion Cracking Susceptibility of Al-Zn-Mg Alloy in Different Corrosive Environments. Metals, 8(4), 225. https://doi.org/10.3390/met8040225




