Failure Analysis of PHILOS Plate Construct Used for Pantalar Arthrodesis Paper I—Analysis of the Plate
Abstract
1. Introduction
1.1. Case Presentation (Clinical Summary)
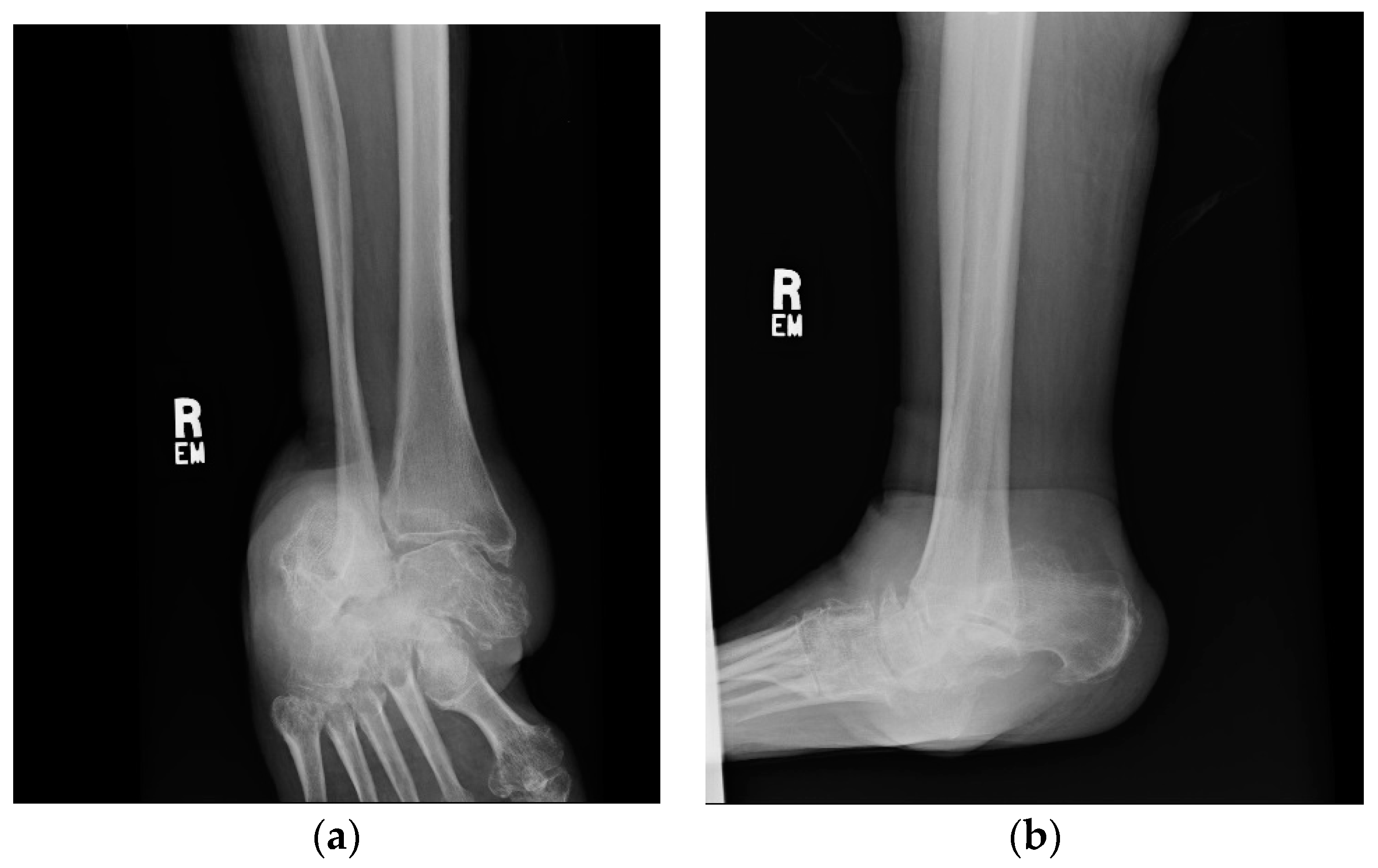
1.1.1. Initial Presentation
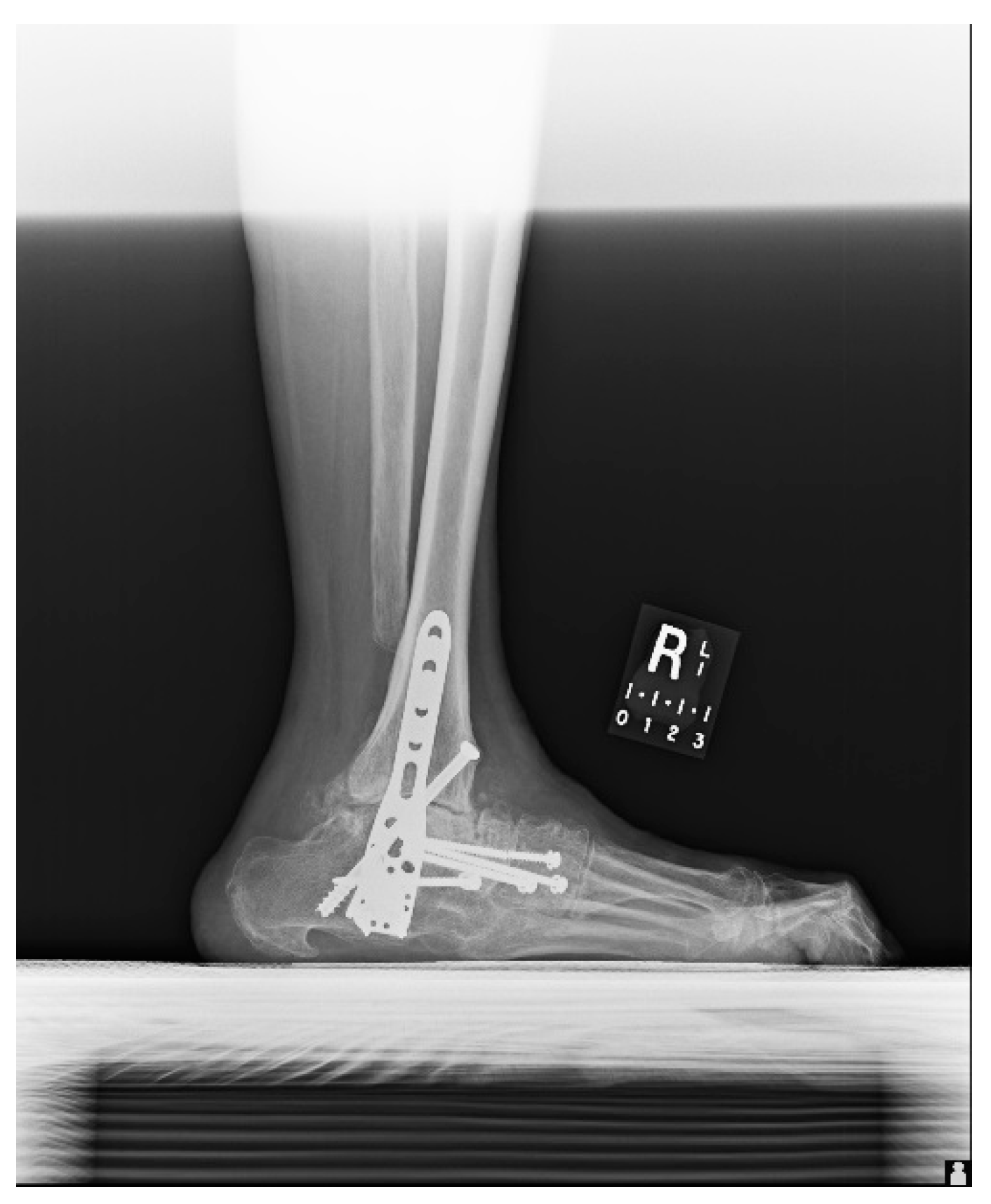
1.1.2. Operation
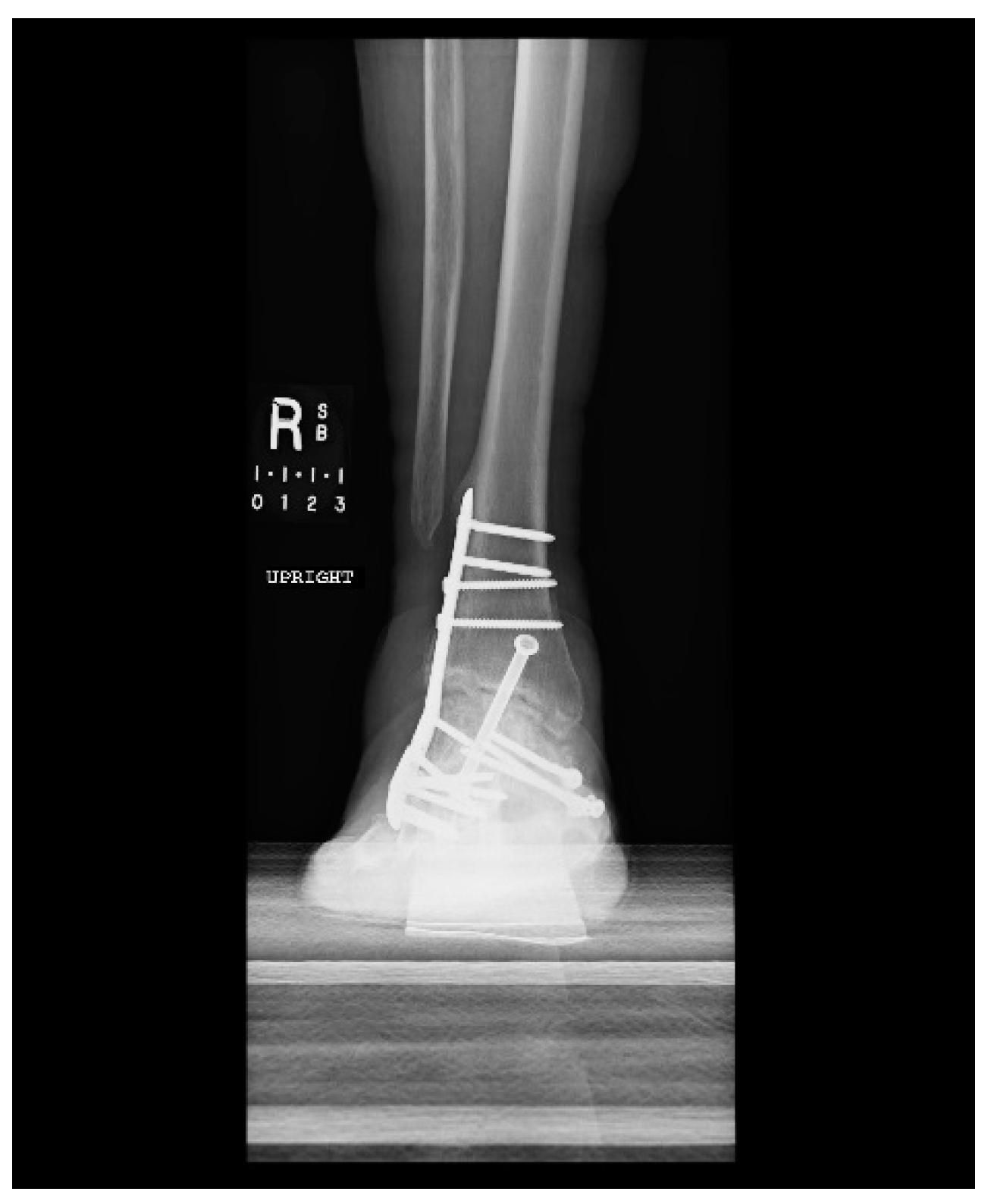
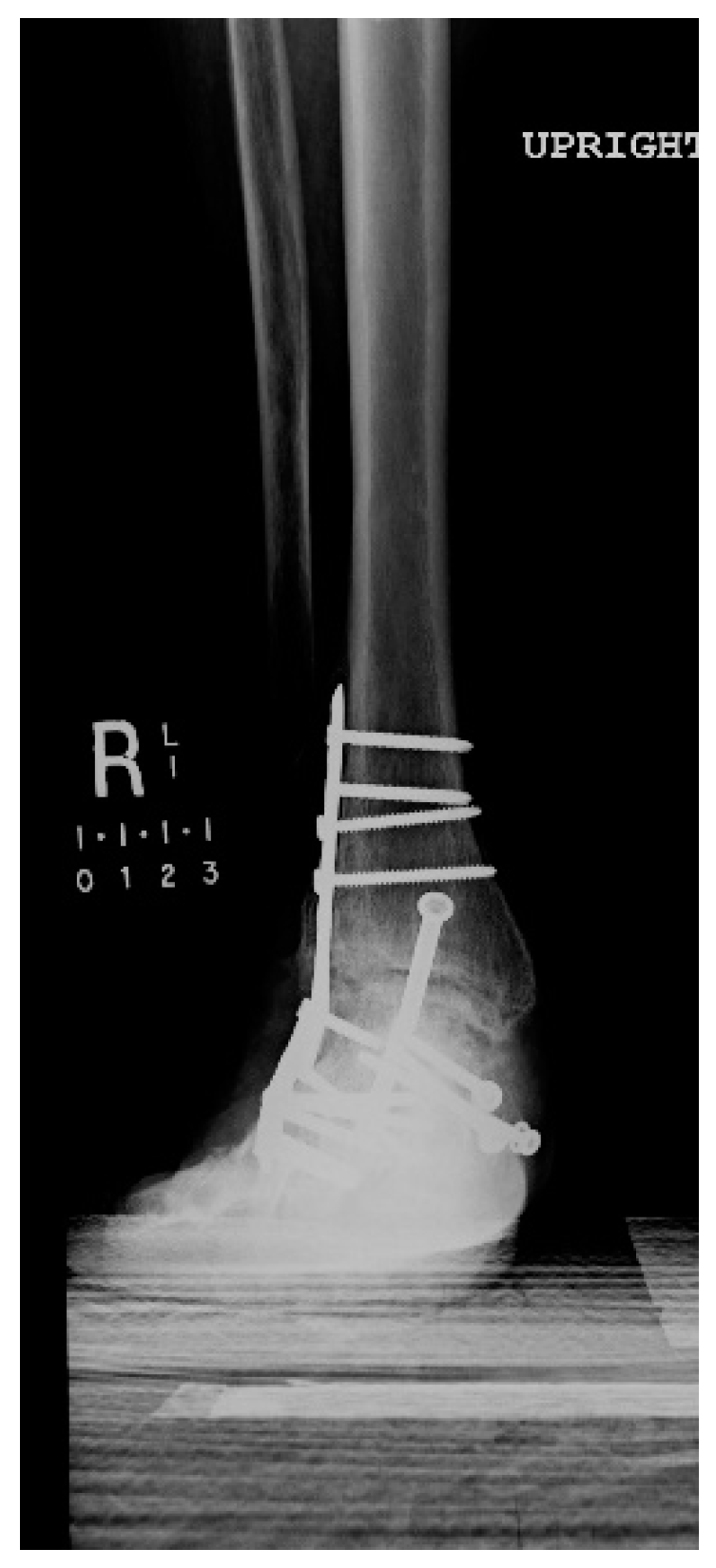
1.1.3. Post-Operative Follow-Up
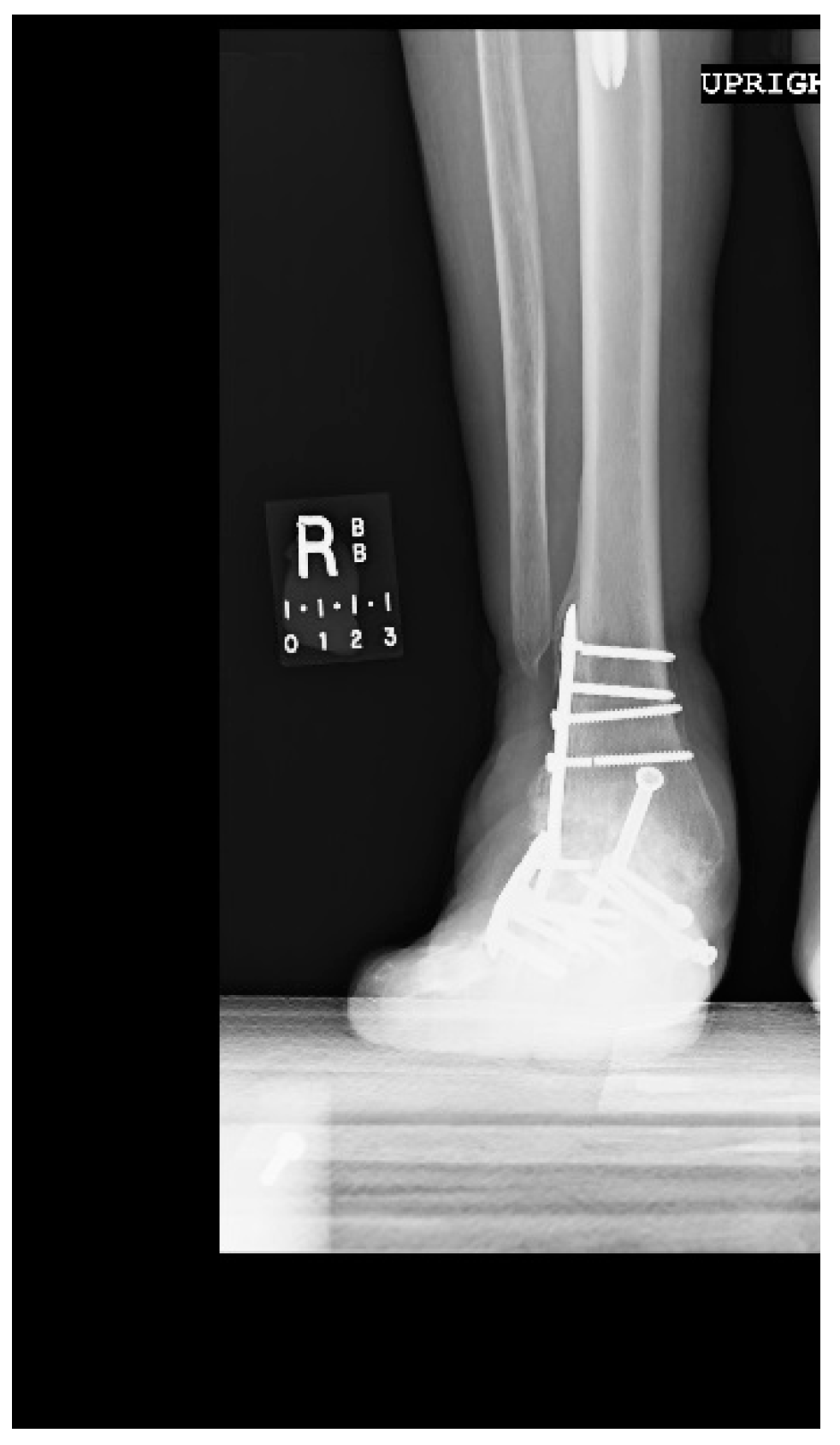
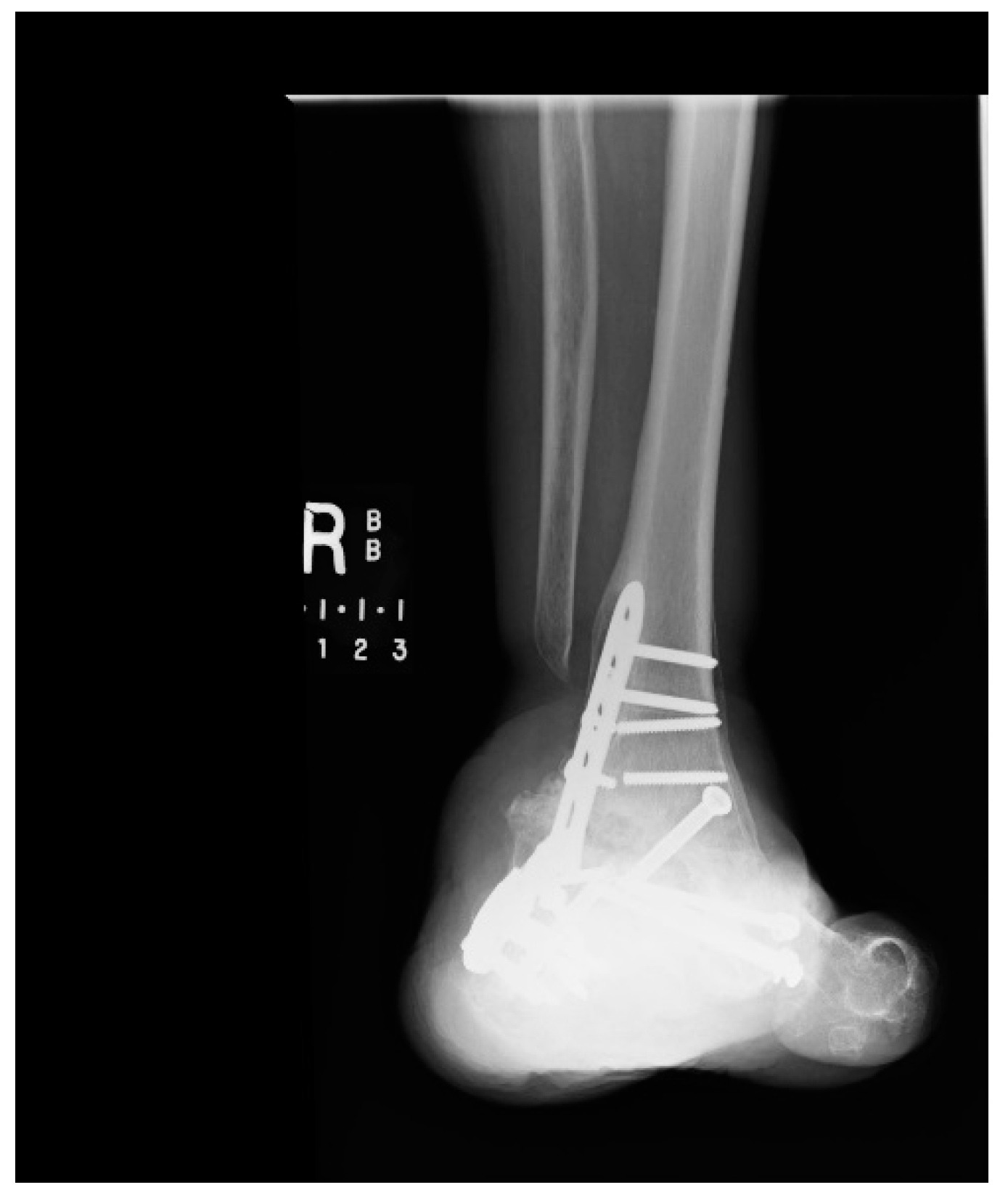
1.1.4. Post-Operative Imaging
2. Materials and Methods
3. Results and Discussion
3.1. Chemical Composition Characterization
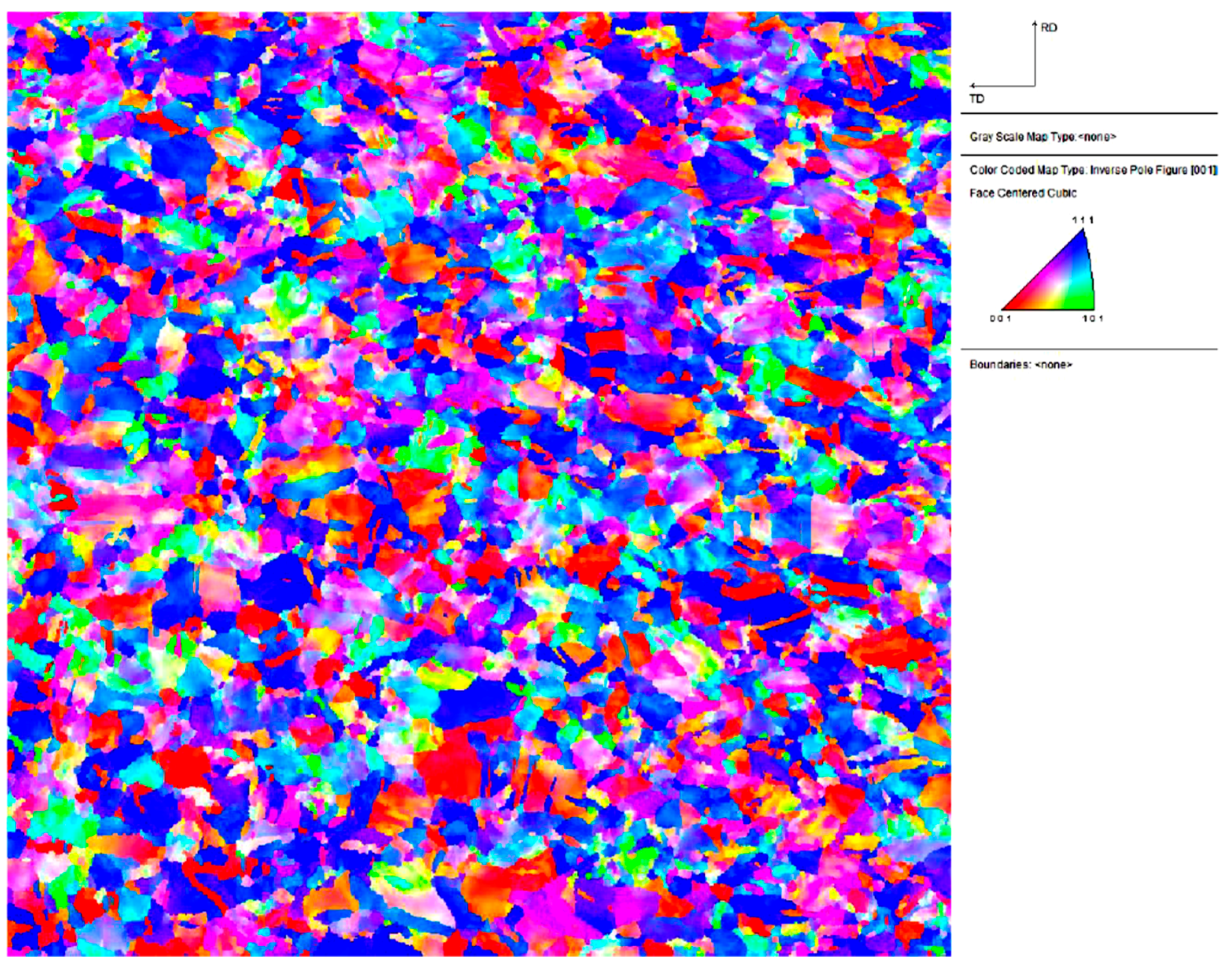
3.2. Microstructural Characterization
3.3. Material Property Analysis
3.4. Optical Microscopic Analysis
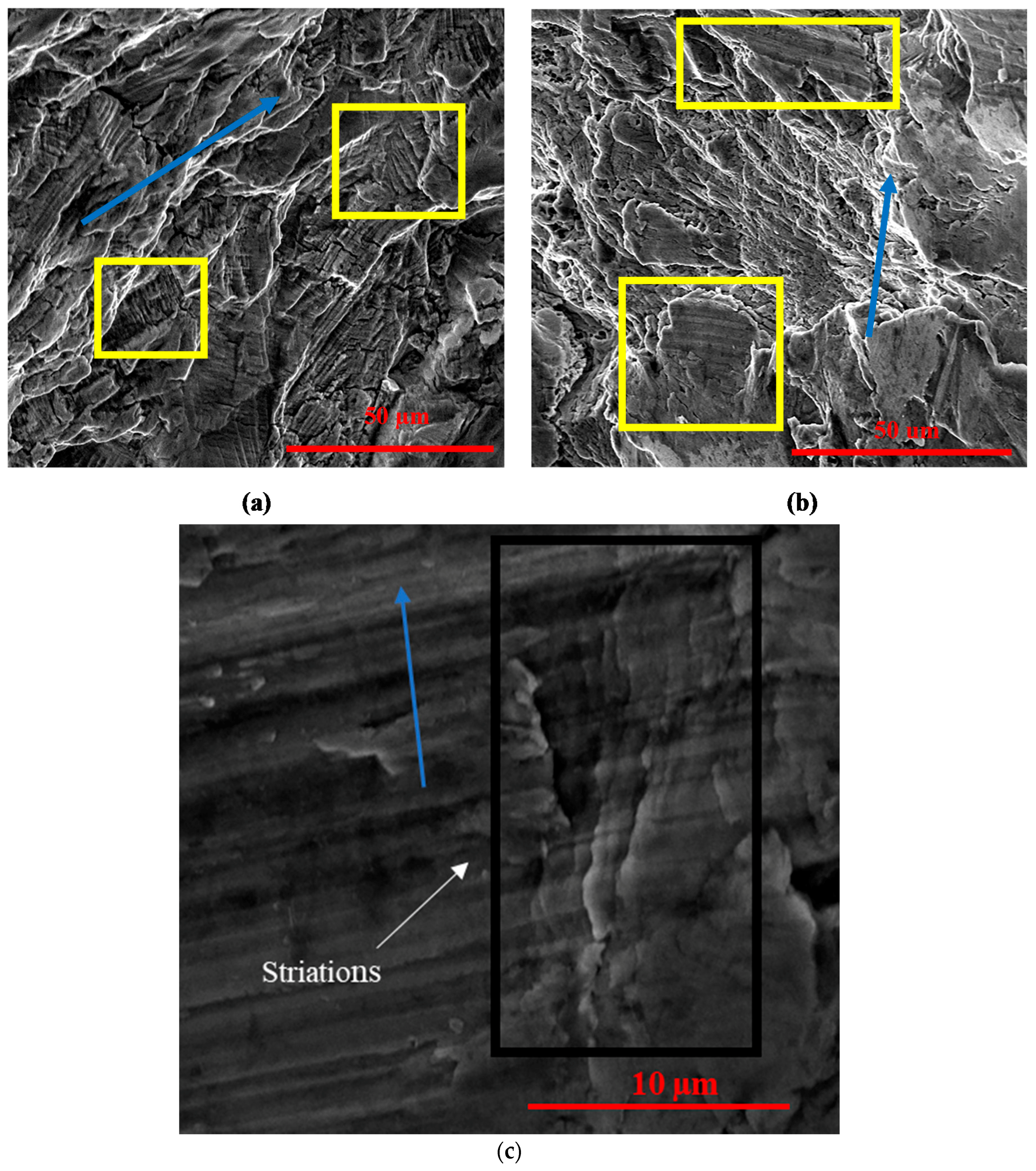
3.5. Fractography
4. Conclusions
- As we know that the plate was designed for the proximal humerus, it did not match all the requirements of pantalar arthrodesis. A slight mismatch would give rise to forces sufficient to cause overloading failure.
- The curvature in the plate is one of the important factors to be considered, due to which unequal load distribution could have taken place.
- Improper reduction may be due to the device. This improper reduction may have generated excessive loading on the LCP leading to a premature mechanical failure.
- The qualitative data of composition, non-metallic inclusions, hardness, and tensile strength of the plate show that the material conformity per ASTM standard may be an additional issue controlling the integrity of the device.
- SEM imaging supports a mechanism of corrosion-fatigue fracture from crack initiation sites in the distal areas of the plate due to the presence of inclusion bodies and pits. These crack initiation sites can then lead to crack propagation towards the proximal area (shown by the striations) and ultimately brittle fracture (supported by the cleavage sites and facets).
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Study | Device Details | Alloy | Failure Mode | Details of Failure |
|---|---|---|---|---|
| Kanchanomai et al. [10] | 14 hole, Locking compression plate (for femur) | 316L Stainless steel | Excessive walking before adequate healing of fracture |
|
| ||||
| ||||
| ||||
| ||||
| Thapa et al. [11] | 10 hole, Locking compression plate | Stainless steel | Corrosion-fatigue |
|
| ||||
| ||||
| ||||
| Karmacharya et al. [12] | 8 hole, Reconstructive locking plate | 316L Stainless steel | Corrosion |
|
| Majid et al. [13] | Lumbar plates (113 plates were studied) | 316L Stainless steel | Corrosion |
|
| ||||
| ||||
| Varadharajan et al. [14] | Hemi-toe implant | Cobalt chromium alloy | Failed clinically |
|
| ||||
| ||||
| Sudhakar et al. [15] | Nail for Shinbones | 316L Stainless steel | Ductile fracture and nonmetallic inclusions |
|
| Sivakumar et al. [16] | 6 hole, Tubular compression bone plate (for femur) | 316L Stainless steel | Improper fixation |
|
| ||||
| ||||
| ||||
| ||||
| ||||
| Marcomini et al. [17] | Femoral Locking compression plate | Stainless steel (not 316L due to higher content of Ni and P) | Non-conformity of the material |
|
| ||||
| ||||
| Cahoon et al. [18] | McLaughlin plate for hip (7 other implants discussed) | Cast Vitallium | Fracture in the area where maximum stresses were expected |
|
| ||||
| Cahoon et al. [18] | V-Moore plate and screws | 316L Stainless steel | Crevice corrosion |
|
| ||||
| Cahoon et al. [18] | Nail plate | 316L Stainless steel | Fracture due to bending (while trying to fit the patient) |
|
| ||||
| Azevedo et al. [19] | 13 hole reconstruction plate for osteosynthesis | Plate: CP Titanium Screws: Titanium-6Al-4V alloy | Corrosion-fatigue |
|
| ||||
| ||||
| ||||
| De Medeiros et al. [20] | Hemimandibles with assistive 4 hole plates | 5052-F Aluminum | Fracture due to ductile overload |
|
| ||||
| ||||
| Roffey et al. [21] | Femoral stem | M30NW High nitrogen Stainless steel | Failure at a point of maximum stress due to bending and torsional loading |
|
| ||||
| ||||
| ||||
| ||||
| ||||
| ||||
| Goswami et al. [22] | IM nail | Titanium | Axial, bending, and torsion forces |
|
| ||||
| ||||
| ||||
| Azevedo et al. [23] | Femoral compression plate | Stainless steel | Fretting-fatigue |
|
| ||||
| ||||
| ||||
| Azevedo et al. [26] | Femoral nail-plate | Stainless steel | Fatigue fracture |
|
| ||||
| Azevedo et al. [23] | Titanium oral maxilla-facial plate | CP Titanium | Corrosion-fatigue |
|
| ||||
| ||||
|
Appendix B
References
- Quill, G.E., Jr. Tibiotalocalcaneal arthrodesis. Tech. Orthop. 1996, 11, 269–273. [Google Scholar] [CrossRef]
- Coughlin, M.J.; Nery, C.; Baumfeld, D.; Jastifer, J. Tibiotalocalcaneal Arthrodesis with lateral compression Plate. Rev. Bras. Ortop. 2012, 47, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Ortho Info. Internal Fixation for Fractures. American Academy of Orthopaedic Surgeons. Available online: https://orthoinfo.aaos.org/en/treatment/internal-fixation-for-fractures/ (accessed on 13 August 2015).
- Sommer, C.; Gautier, E.; Müller, M.; Helfet, D.L.; Wagner, M. First clinical results of the Locking Compression Plate (LCP). Injury 2003, 34, 43–54. [Google Scholar] [CrossRef]
- Kubiak, E.N.; Fulkerson, E.; Strauss, E.; Egol, K.A. The evolution of locked plates. J. Bone Jt. Surg. 2006, 88 (Suppl. 4), 189–200. [Google Scholar]
- Ahmad, M.; Nanda, R.; Bajwa, A.S.; Candal-Couto, J.; Green, S.; Hui, A.C. Biomechanical testing of the locking compression plate: When does the distance between bone and implant significantly reduce construct stability? Injury 2007, 38, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Desai, K. A Biomechanical Comparison of Locking Compression Plate Constructs with Plugs/Screws in Osteoporotic Bone Model. Master’s Thesis, Wright State University, Fairborn, OH, USA, 2010. [Google Scholar]
- Ahmad, J.; Pour, A.E.; Raikin, S.M. The modified use of a proximal humeral locking plate for tibiotalocalcaneal arthrodesis. Foot Ankle Int. 2007, 28, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Twaij, H.; Damany, D. PHILOS humerus plate for a distal tibial fracture. J. Surg. Case Rep. 2013, 2013, rjs036. [Google Scholar] [CrossRef] [PubMed]
- Kanchanomai, C.; Phiphobmongkol, V.; Muanjan, P. Fatigue failure of an orthopedic implant—A locking compression plate. Eng. Fail. Anal. 2008, 15, 521–530. [Google Scholar] [CrossRef]
- Thapa, N.; Prayson, M.; Goswami, T. A failure study of a locking compression plate implant. Case Stud. Eng. Fail. Anal. 2015, 3, 68–72. [Google Scholar] [CrossRef]
- Karmacharya, S.; Goswami, T. Analysis of a clinically failed, mechanically intact, reconstructive compression plate. Case Studies in Engineering Failure Analysis. Trauma Cases Rev. 2015. [CrossRef]
- Majid, K.; Baker, K.C.; Baker, E.A.; Koueiter, D.M.; Herkowitz, H.N. High Rate of Corrosion of 316L Stainless Steel Plates Used in Posterior Thoracolumbar Fixation: A Retrieval Study. In Proceedings of the 55th Annual Meeting of the Orthopaedic Research Society, Las Vegas, NV, USA, 22–25 February 2011. [Google Scholar]
- Varadharajan, S.; Laughlin, R.; Goswami, T. Analysis of clinically failed, mechanically intact, hemi-toe implant. Case Studies in Engineering Failure Analysis. J. Biomed. Eng. Biosci. 2016, 3, 22–25. [Google Scholar]
- Sudhakar, K.V. Metallurgical investigation of a failure in 316L stainless steel orthopaedic implant. Eng. Fail. Anal. 2005, 12, 249–256. [Google Scholar] [CrossRef]
- Sivakumar, M.; Kamachi Mudali, U.; Rajeswari, S. Investigation of failures in stainless steel orthopaedic implant devices: Fatigue failure due to improper fixation of a compression bone plate. J. Mater. Sci. Lett. 1994, 13, 142–145. [Google Scholar] [CrossRef]
- Marcomini, J.B.; Baptista, C.A.R.P.; Pascon, J.P.; Teixeira, R.L.; Reis, F.P. Investigation of a fatigue failure in a stainless steel femoral plate. J. Mech. Behav. Biomed. Mater. 2014, 38, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Cahoon, J.R.; Paxton, H.W. Metallurgical analyses of failed orthopedic implants. J. Biomed. Mater. Res. 1968, 2, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, C.R.F. Failure analysis of a commercially pure titanium plate for osteosynthesis. Eng. Fail. Anal. 2003, 10, 153–164. [Google Scholar] [CrossRef]
- De Medeiros, R.C.; de Moura, A.L.; Rodrigues, D.C.; Mendes, M.B.M.; Sawazaki, R.; Moreira, R.W.F. Fractographic analysis of 2.0-mm plates with a screw locking system in simulated fractures of the mandibular body. J. Oral Maxillofac. Surg. 2014, 72, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Roffey, P. Case study: Failure of a high nitrogen stainless steel femoral stem. Eng. Fail. Anal. 2012, 20, 173–182. [Google Scholar] [CrossRef]
- Gundapaneni, D.; Slocum, M.; Paul, P.; Christof, S.; Goswami, T. Failure Investigation of a Tibiotalocalcaneal Arthrodesis System. Eng. Fail. Anal. 2016, 59, 588–604. [Google Scholar]
- SYNTHES. 3.5 mm LCP Proximal Humerus Plate—Stainless Steel and Titanium Technique Guide. 2002. Available online: http://synthes.vo.llnwd.net/o16/LLNWMB8/US%20Mobile/Synthes%20North%20America/Product%20Support%20Materials/Technique%20Guides/DSUSTRM10161133_ProxHumPl_STG_150dpi.pdf (accessed on 9 June 2015).
- SYNTHES. Small Fragment Locking Compression Plate (LCP) System. Stainless Steel and Titanium-Technique Guide. 2002. Available online: http://synthes.vo.llnwd.net/o16/Mobile/Synthes%20North%20America/Product%20Support%20Materials/Technique%20Guides/SUSA/SUTGSmFrgLCPJ3908I.pdf (accessed on 9 June 2015).
- Annual Book of ASTM Standards (Vol. 13.01), American Society for Testing and Materials: West Conshohocken, PA, USA, 2005.
- Azevedo, C.; Hippert, E. Failure analysis of surgical implants in Brazil. Eng. Fail. Anal. 2002, 9, 621–633. [Google Scholar] [CrossRef]
- Powell, G.W.; Mahmoud, S.E. ASM Handbook, 9th ed.; American Society for Metals: Metals Park, OH, USA, 1986; Volume 11. [Google Scholar]

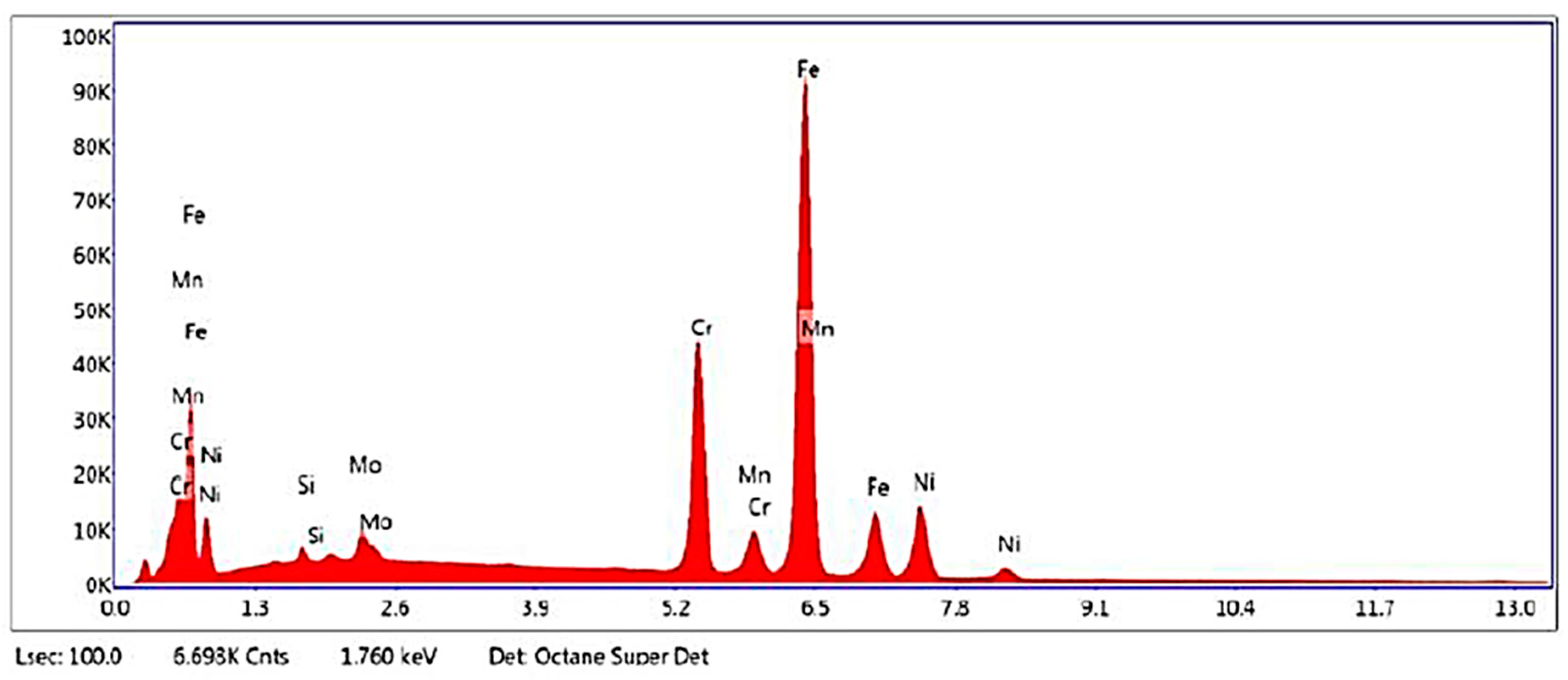






| Composition | ASTM Standard (Max) | Plate (Average) | Tolerance Limits |
|---|---|---|---|
| C | 0.03 | - | 0.005 |
| Mn | 2.0 | 1.81 | 0.04 |
| Si | 0.75 | 0.59 | 0.05 |
| P | 0.025 | - | 0.005 |
| S | 0.01 | - | 0.005 |
| Cr | 17–19 | 17.61 | 0.05 |
| Mo | 2.25–3.0 | 2.17 | 0.15 |
| Ni | 13–15 | 13.84 | 0.2 |
| N | 0.10 | - | 0.01 |
| Fe | Balance | 63.99 | - |
| The Test | Average | ASTM Standard | Proximal | Middle | Distal |
|---|---|---|---|---|---|
| Rockwell hardness | Average | 95 | 101.8 | 107.7 | 107.8 |
| 95% confidence interval | - | 101.1–102.5 | 106.1–109.3 | 106.6–109.5 | |
| Vickers hardness | Average | 213 | 266.5 | 330 | 334.5 |
| 95% confidence interval | - | 260–273 | 310–350 | 316–353 | |
| Tensile strength (psi) | Average (MPa) | 868 | 848 | 1057 | 1057 |
| 95% confidence interval | - | 837–859 | 1004–1110 | 1004–1110 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ina, J.; Vallentyne, M.; Hamandi, F.; Shugart, K.; Boin, M.; Laughlin, R.; Goswami, T. Failure Analysis of PHILOS Plate Construct Used for Pantalar Arthrodesis Paper I—Analysis of the Plate. Metals 2018, 8, 180. https://doi.org/10.3390/met8030180
Ina J, Vallentyne M, Hamandi F, Shugart K, Boin M, Laughlin R, Goswami T. Failure Analysis of PHILOS Plate Construct Used for Pantalar Arthrodesis Paper I—Analysis of the Plate. Metals. 2018; 8(3):180. https://doi.org/10.3390/met8030180
Chicago/Turabian StyleIna, Jason, Madhurima Vallentyne, Farah Hamandi, Kathleen Shugart, Michael Boin, Richard Laughlin, and Tarun Goswami. 2018. "Failure Analysis of PHILOS Plate Construct Used for Pantalar Arthrodesis Paper I—Analysis of the Plate" Metals 8, no. 3: 180. https://doi.org/10.3390/met8030180
APA StyleIna, J., Vallentyne, M., Hamandi, F., Shugart, K., Boin, M., Laughlin, R., & Goswami, T. (2018). Failure Analysis of PHILOS Plate Construct Used for Pantalar Arthrodesis Paper I—Analysis of the Plate. Metals, 8(3), 180. https://doi.org/10.3390/met8030180






