Comparative Effect of Mo and Cr on Microstructure and Mechanical Properties in NbV-Microalloyed Bainitic Steels
Abstract
1. Introduction
2. Materials and Methods
- -
- Austenitising at 1250 °C for 180 s, followed by cooling to 1175 °C at a cooling rate of 1 °C·s−1;
- -
- First deformation at 1175 °C to 0.3 (low strain schedule) or 0.35 (high strain schedule) strain at 5 s−1 strain rate, followed by cooling to 1100 °C at a cooling rate of 2 °C·s−1;
- -
- Second deformation at 1100 °C to 0.35 (low strain schedule) or 0.50 (high strain schedule) strain at 5 s−1 strain rate, followed by cooling to 1000 °C at a cooling rate of 25 °C·s−1;
- -
- Third deformation at 1000 °C to 0.25 strain at 5 s−1 strain rate, followed by cooling to 900 °C at a cooling rate of 30 °C·s−1;
- -
- Fourth deformation at 900 °C to 0.25 strain at 5 s−1 strain rate, followed by holding at this temperature for 10 s and cooling to 500 °C at a cooling rate of 30 °C·s−1 to assure bainite transformation;
- -
- Holding at 500 °C for 900 s to simulate coiling, followed by air cooling to room temperature.
3. Results
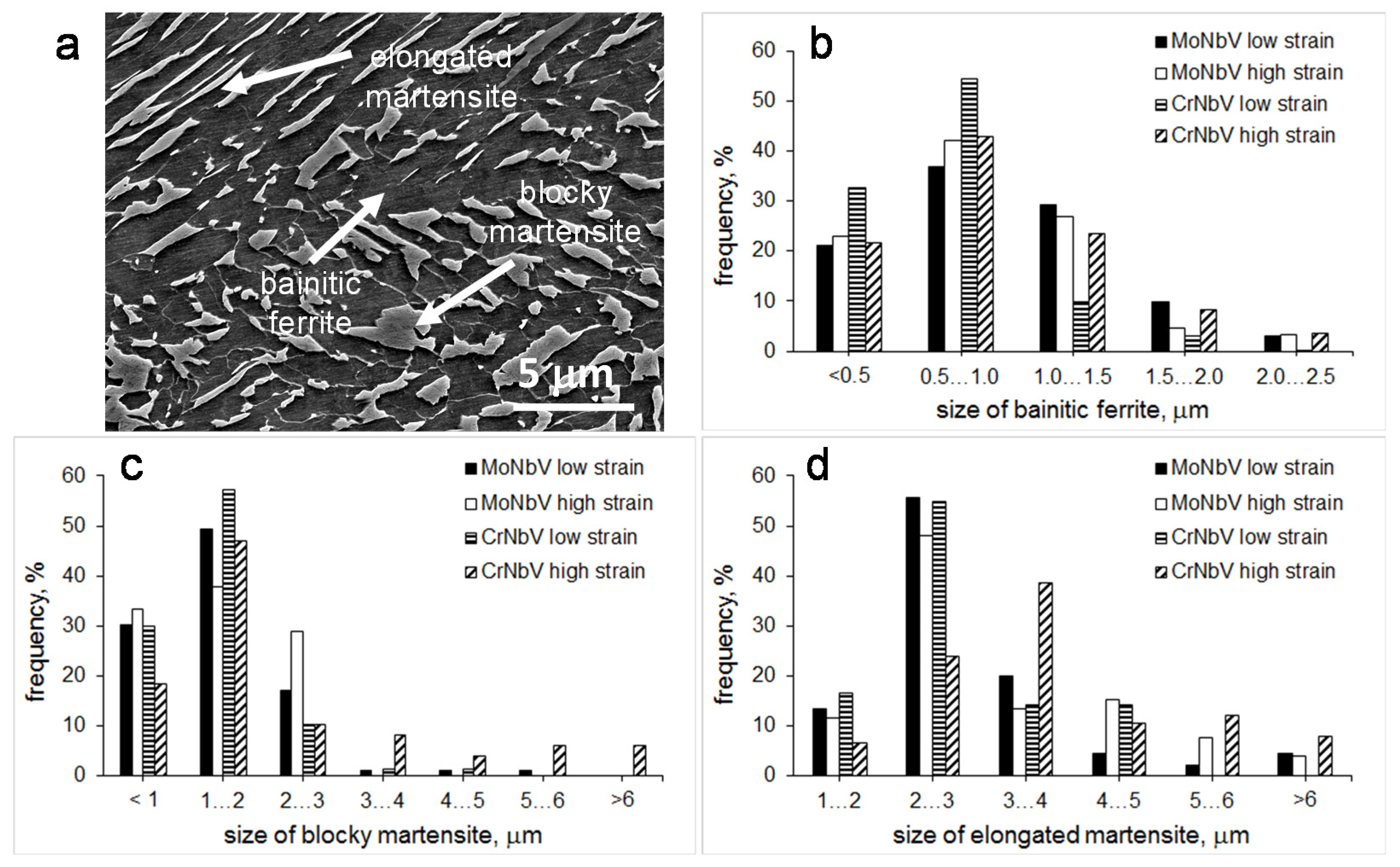
3.1. Grain Structure and Phase Balance
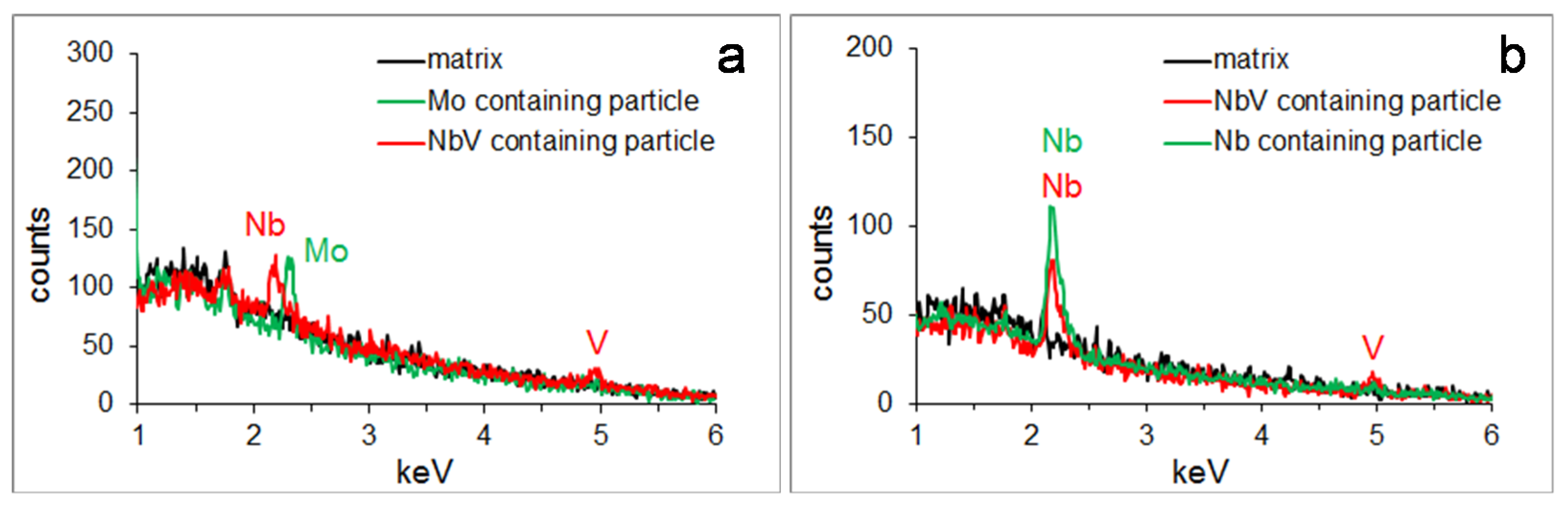
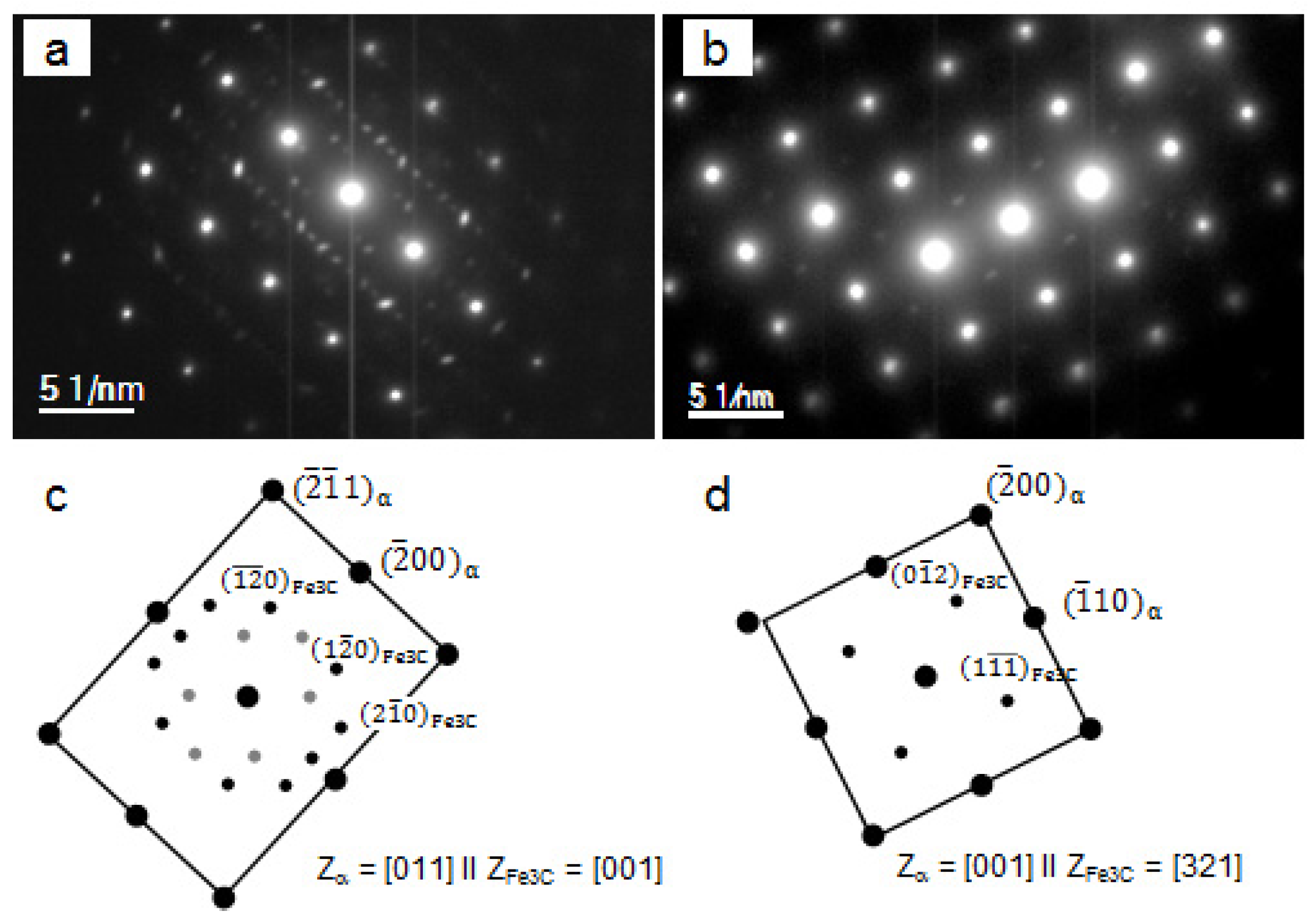
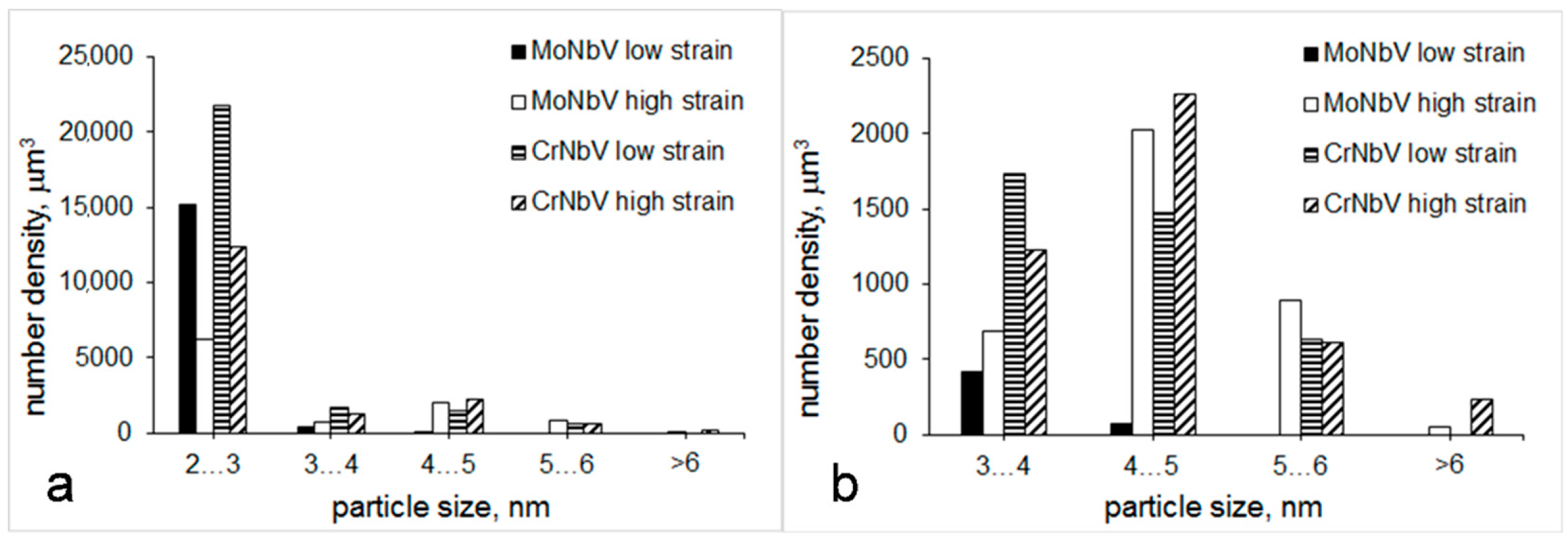
3.2. Particle Precipitation
3.3. Dislocation Structure
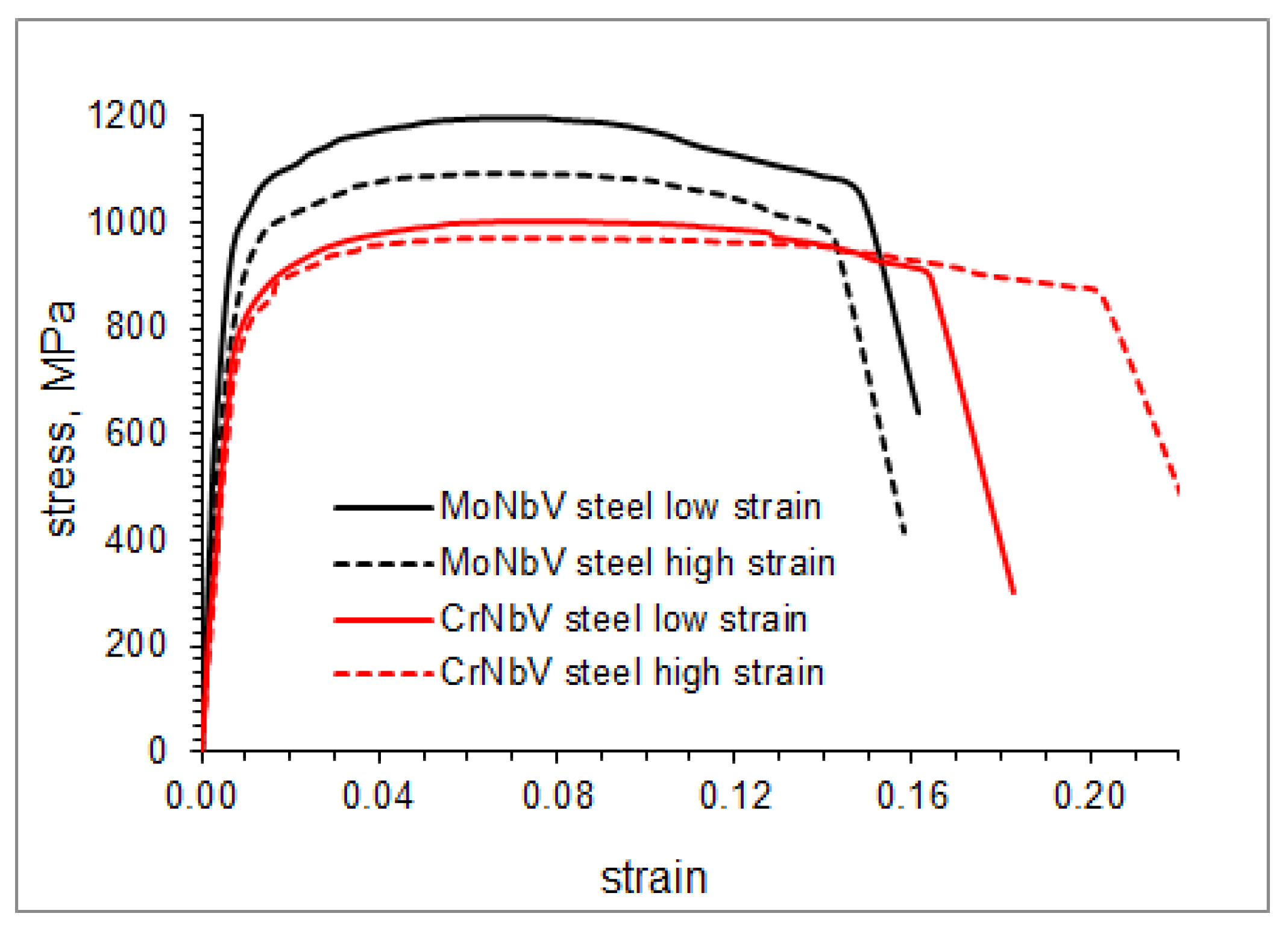
3.4. Mechanical Properties
4. Discussion
4.1. Strain Effect on Phase Transformation and Precipitation
4.2. Mo and Cr Effects on Phase Transformation
4.3. Mo and Cr Effects on Precipitation
4.4. Microstructure-Mechanical Properties Relationship: Role of Solute Atoms
- from grain boundaries using the Hall–Petch equation:where 0 = 15 MPa and k = 21.4 MPa·mm1/2 are accepted for pure iron [75], and d is the size of bainitic ferrite areas (the shortest distance between the martensitic grains);
- from precipitation of >20 nm particles using the Ashby-Orowan equation [76], which assumes the dislocation looping around relatively large particles:where f is the particle volume fraction and D is the particle diameter in μm;
- from precipitation of <20 nm particles using the order strengthening relationship [77], which assumes the dislocation cutting of relatively small, coherent particles:where M = 3 is the matrix orientation factor, b = 0.312–0.306 nm is Burgers vector accepted according to the measured unite cell size of bainitic ferrite (Table 1), γ is the matrix-particle interface energy assumed for the Fe-Fe3C interface to be γ = 0.5 J·m−2 [78], and f is the particle volume fraction;
- from dislocations using the long range work hardening theory [79]:where α = 0.5 is a constant, G = 85,000 MPa is the shear modulus, b = 0.312–0.306 nm is the Burgers vector and is the measured dislocation density;
- the solid solution strengthening contribution from Mn and Si was estimated using the matrix concentrations of these elements and the following relationship [76]:where CSi = 0.3 wt. % and CMn = 1.5 wt. % are Si and Mn concentrations in the bainitic ferrite matrix, respectively.Δσss(Si,Mn) = 83CSi + 32CMn,
5. Conclusions
- Additions of either 0.2 wt. % Mo or 0.2 wt. % Cr (above 0.3 wt. % Cr) resulted in formation of bainite microstructure with mixed bainite morphologies and similar parameters of bainitic ferrite matrix and martensite as the second phase. However, for higher strain processing (when the DRX may be completed faster) the size and fraction of martensite were lower in the MoNbV-steel. This could be related to a smaller prior austenite grain size in the MoNbV-steel, which would correspond to a stronger solute drag effect of Mo during DRX. The influence of prior austenite grain size on bainite phase transformation in the studied steels requires further investigation.
- The number density and area fraction of >20 nm NbV-containing particles were significantly lower in the MoNbV-steel. This supports a stronger effect of Mo than Cr on the increase of solubility of Nb and V in austenite, which results in their decreased precipitation. The number density and area fraction of <20 nm Fe3C particles were also lower in the MoNbV-steel. This supports a stronger effect of Mo than Cr on the retardation of Fe3C precipitation. Less pronounced precipitation in the MoNbV-steel, and related to this increased solid solute concentrations, corresponds to a wider expansion of the unit cell size of the bcc lattice of bainitic ferrite matrix.
- In addition to grain size strengthening, the solid solution/atom cluster strengthening effect was the second dominating in the MoNbV-steel; although in the CrNbV-steel the precipitation strengthening from >20 nm particles, <20 nm particles and dislocations equally contributed to the overall steel strength.
- Strain increase resulted in more pronounced strain induced precipitation of Mo, Nb and V in the MoNbV-steel and Nb and V in the CrNbV-steel. Solute depletion in microalloying elements could have contributed to decreased strength levels in both steels for the higher strain processing schedule.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Aaronson, H.I.; Reynolds, W.T., Jr.; Purdy, G.R. The incomplete transformation phenomenon in steel. Metall. Mater. Trans. A 2006, 37, 1731–1745. [Google Scholar] [CrossRef]
- Humphreys, E.S.; Fletcher, H.A.; Hutchins, J.D.; Garratt-Reed, A.J.; Reynolds, W.T., Jr.; Aaronson, H.I.; Purdy, G.R.; Smith, G.D.W. Molybdenum accumulation at ferrite: Austenite interfaces during isothermal transformation of an Fe-0.24 pct C-0.93 pct Mo alloy. Metall. Mater. Trans. A 2004, 35, 1223–1235. [Google Scholar] [CrossRef]
- Kong, J.; Xie, C. Effect of molybdenum on continuous cooling bainite transformation of low-carbon microalloyed steel. Mater. Des. 2006, 27, 1169–1173. [Google Scholar] [CrossRef]
- Hu, H.; Xu, G.; Zhou, M.; Yuan, Q. Effect of Mo content on microstructure and property of low-carbon bainitic steels. Metals 2016, 6, 173. [Google Scholar] [CrossRef]
- Sung, H.-K.; Lee, D.-H.; Shin, S.-Y.; Lee, S.; Yoo, J.-Y.; Hwang, B. Effect of finish cooling temperature on microstructure and mechanical properties of high-strength bainitic steels containing Cr, Mo and B. Mater. Sci. Eng. A 2015, 624, 14–22. [Google Scholar] [CrossRef]
- Uemori, R.; Chijiiwa, R.; Tamehiro, H.; Morikawa, H. AP-FIM study on the effect of Mo addition on microstructure in Ti-Nb steel. Appl. Surf. Sci. 1994, 76, 255–260. [Google Scholar] [CrossRef]
- Wan, R.; Sun, F.; Zhang, L.; Shan, A. Effect of Mo addition on strength of fire-resistant steel at elevated temperature. J. Mater. Eng. Perform. 2014, 23, 2780–2786. [Google Scholar] [CrossRef]
- Dimitriu, R.C.; Bhadeshia, H.K.D.H. Hot strength of creep resistant ferritic steels and relationship to creep rupture data. Mater. Sci. Technol. 2007, 23, 1127–1131. [Google Scholar] [CrossRef]
- He, R.; Jiang, L.; Dong, W. Development of high corrosion-resistant ferritic stainless steel and its application in the building cladding system. Baosteel Tech. Res. 2013, 7, 54–58. [Google Scholar]
- Akben, M.G.; Bacroix, B.; Jonas, J.J. Effect of vanadium and molybdenum addition on high temperature recovery, recrystallization and precipitation behaviour of niobium-based microalloyed steels. Acta Metall. 1983, 31, 161–174. [Google Scholar] [CrossRef]
- Andrade, H.L.; Akben, M.G.; Jonas, J.J. Effect of molybdenum, niobium, and vanadium on static recovery and recrystallization and on solute strengthening in microalloyed steels. Metall. Trans. A 1983, 14, 1967–1977. [Google Scholar] [CrossRef]
- Schambron, T.; Chen, L.; Gooch, T.; Dehghan-Manshadi, A.; Pereloma, E.V. Effect of Mo concentration on dynamic recrystallization behavior of low carbon microalloyed steels. Steel Res. Int. 2013, 84, 1191–1195. [Google Scholar] [CrossRef]
- Lu, J.; Omotoso, O.; Wiskel, J.B.; Ivey, D.G.; Henein, H. Strengthening mechanisms and their relative contributions to the yield strength of microalloyed steels. Metall. Mater. Trans. A 2012, 43, 3043–3061. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, C.; Yu, L.; Liu, Y.; Li, H. Phase transformation behavior and microstructural control of high-Cr martensitic/ferritic heat-resistant steels for power and nuclear plants: A review. J. Mater. Sci. Technol. 2015, 31, 235–242. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, C.; Li, R.; Gao, J.; Wang, M.; Zhang, F. Characterization of the microstructures and mechanical properties of 25CrMo48V martensitic steel tempered at different times. Mater. Sci. Eng. A 2013, 559, 130–134. [Google Scholar] [CrossRef]
- Chen, C.Y.; Chen, C.C.; Yang, J.R. Microstructure characterization of nanometer carbides heterogeneous precipitation in Ti–Nb and Ti–Nb–Mo steel. Mater. Charact. 2014, 88, 69–79. [Google Scholar] [CrossRef]
- Mukherjee, S.; Timokhina, I.; Zhu, C.; Ringer, S.P.; Hodgson, P.D. Clustering and precipitation processes in a ferritic titanium-molybdenum microalloyed steel. J. Alloy. Compd. 2017, 690, 621–632. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, H.; Guo, C.; Liu, W.; Yang, Z.; Sun, X.; Zhang, Z.; Jiang, F. Effect of molybdenum addition on the precipitation of carbides in the austenite matrix of titanium micro-alloyed steels. J. Mater. Sci. 2016, 51, 4996–5007. [Google Scholar] [CrossRef]
- Larzabal, G.; Isasti, N.; Rodriguez-Ibabe, J.M.; Uranga, P. Evaluating strengthening and impact toughness mechanisms for ferritic and bainitic microstructures in Nb, Nb-Mo and Ti-Mo microalloyed steels. Metals 2017, 7, 65. [Google Scholar] [CrossRef]
- Lee, W.-B.; Hong, S.-G.; Park, C.-G.; Park, S.-H. Carbide precipitation and high-temperature strength of hot-rolled high-strength, low-alloy steels containing Nb and Mo. Metall. Mater. Trans. A 2002, 33, 1689–1698. [Google Scholar] [CrossRef]
- Jang, J.H.; Heo, Y.-U.; Lee, C.-H.; Bhadeshia, H.K.D.H.; Suh, D.-W. Interphase precipitation in Ti–Nb and Ti–Nb–Mo bearing steel. Mater. Sci. Technol. 2013, 29, 309–313. [Google Scholar] [CrossRef]
- Funakawa, Y.; Shiozaki, T.; Tomita, K.; Yamamoto, T.; Maeda, E. Development of high strength hot-rolled sheet steel consisting of ferrite and nanometer-sized carbides. ISIJ Int. 2004, 44, 1945–1951. [Google Scholar] [CrossRef]
- Park, D.-B.; Huh, M.-Y.; Shim, J.-H.; Suh, J.-Y.; Lee, K.-H.; Jung, W.-S. Strengthening mechanism of hot rolled Ti and Nb microalloyed HSLA steels containing Mo and W with various coiling temperature. Mater. Sci. Eng. A 2013, 560, 528–534. [Google Scholar] [CrossRef]
- Bracke, L.; Xu, W. Effect of the Cr content and coiling temperature on the properties of hot rolled high strength lower bainitic steel. ISIJ Int. 2015, 55, 2206–2211. [Google Scholar] [CrossRef]
- Lee, H.-J.; Lee, H.-W. Effect of Cr content on microstructure and mechanical properties of low carbon steel welds. Int. J. Electrochem. Sci. 2015, 10, 8028–8040. [Google Scholar]
- Zhou, M.; Xu, G.; Tian, J.; Hu, H.; Yuan, Q. Bainitic transformation and properties of low carbon carbide-free bainitic steels with Cr addition. Metals 2017, 7, 263. [Google Scholar] [CrossRef]
- Mishra, S.K.; Das, S.; Ranganathan, S. Precipitation in high strength low alloy (HSLA) steel: A TEM study. Mater. Sci. Eng. A 2002, 323, 285–292. [Google Scholar]
- Janovec, J.; Svoboda, M.; Vyrostkova, A.; Kroupa, A. Time-temperature-precipitation diagrams of carbide evolution in low alloy steels. Mater. Sci. Eng. A 2005, 402, 288–293. [Google Scholar] [CrossRef]
- Timokhina, I.B.; Hodgson, P.D.; Ringer, S.P.; Zheng, R.K.; Pereloma, E.V. Precipitate characterisation of an advanced high-strength low-alloy (HSLA) steel using atom probe tomography. Scr. Mater. 2007, 56, 601–604. [Google Scholar] [CrossRef]
- Gorokhova, N.A.; Sarrak, V.I.; Suvorova, S.O. Solubility of titanium and niobium carbides in high-chromium ferrite. Met. Sci. Heat Treat. 1986, 28, 276–279. [Google Scholar] [CrossRef]
- Tsai, S.-P.; Su, T.-C.; Yang, J.-R.; Chen, C.-Y.; Wang, Y.-T.; Huang, C.-Y. Effect of Cr and Al additions on the development of interphase-precipitated carbides strengthened dual-phase Ti-bearing steels. Mater. Des. 2017, 119, 319–325. [Google Scholar] [CrossRef]
- Koyama, S.; Ishii, T.; Narita, K. Effects of Mn, Si, Cr and Ni on the solution and precipitation of Niobium carbide in iron austenite. J. Jpn. Inst. Met. 1971, 35, 1089–1094. [Google Scholar] [CrossRef]
- Ray, A. Niobium microalloying in rail steel. Mater. Sci. Technol. 2017, 33, 1584–1600. [Google Scholar] [CrossRef]
- Pereloma, E.V.; Bata, V.; Scott, R.I.; Smith, R.M. Effect of Cr on strain ageing behaviour of low carbon steel. Mater. Sci. Forum 2007, 539–543, 4214–4219. [Google Scholar] [CrossRef]
- Lu, Q.; Xu, W.; van der Zwaag, S. Designing new corrosion resistant ferritic heat resistant steel based on optimal solid solution strengthening and minimisation of undesirable microstructural components. Comput. Mater. Sci. 2014, 84, 198–205. [Google Scholar] [CrossRef]
- Yamamoto, S.; Sakiyama, T.; Ouchi, C. Effect of alloying elements on recrystallization kinetics after hot deformation in austenitic stainless steels. Trans. ISIJ 1987, 27, 447–452. [Google Scholar] [CrossRef]
- De Abreu Martins, C.; Poliak, E.; Godefroid, L.B.; Fonstein, N. Determining the conditions for dynamic recrystallization in hot deformation of C–Mn–V steels and the effects of Cr and Mo additions. ISIJ Int. 2014, 54, 227–234. [Google Scholar] [CrossRef]
- Hutchinson, B.; Siwecki, T.; Komenda, J.; Hagstrӧm, J.; Lagneborg, R.; Hedin, J.-E.; Gladh, M. New vanadium-microalloyed bainitic 700 MPa strip steel product. Ironmak. Steelmak. 2014, 41, 1–6. [Google Scholar] [CrossRef]
- Kong, X.; Lan, L.; Hu, Z.; Li, B.; Sui, T. Optimization of mechanical properties of high strength bainitic steel using thermo-mechanical control and accelerated cooling process. J. Mater. Process. Technol. 2015, 217, 202–210. [Google Scholar] [CrossRef]
- Abbasi, E.; Rainforth, W.M. Effect of Nb-Mo additions on precipitation behaviour in V microalloyed TRIP-assisted steels. Mater. Sci. Technol. 2016, 32, 1721–1729. [Google Scholar] [CrossRef]
- Sourmail, T. Precipitation in creep resistant austenitic stainless steels. Mater. Sci. Technol. 2001, 17, 1–14. [Google Scholar] [CrossRef]
- Williams, D.; Carter, C.B. Transmission Electron Microscopy II—Diffraction; Plenum Press: New York, NY, USA, 1996; pp. 321–323. [Google Scholar]
- Pereloma, E.V.; Al-Harbi, F.; Gazder, A. The crystallography of carbide-free bainites in thermo-mechanically processed low Si transformation-induced plasticity steels. J. Alloy. Compd. 2014, 615, 96–110. [Google Scholar] [CrossRef]
- Pandit, A.; Murugaiyan, A.; Saha Podder, A.; Haldar, A.; Bhattacharjee, D.; Chandra, S.; Ray, R.K. Strain induced precipitation of complex carbonitrides in Nb–V and Ti–V microalloyed steels. Scr. Mater. 2005, 53, 1309–1314. [Google Scholar] [CrossRef]
- Cabibbo, M.; Fabrizi, A.; Merlin, M.; Garagnani, G.L. Effect of thermo-mechanical treatments on the microstructure of micro-alloyed low-carbon steels. J. Mater. Sci. 2008, 43, 6857–6865. [Google Scholar] [CrossRef]
- Kostryzhev, A.G.; Strangwood, M.; Davis, C.L. Mechanical property development during UOE forming of large diameter pipeline steels. Mater. Manuf. Process. 2010, 25, 41–47. [Google Scholar] [CrossRef]
- Miyata, K.; Omura, T.; Kushida, T.; Komizo, Y. Coarsening kinetics of multicomponent MC-type carbides in high-strength low-alloy steels. Metall. Mater. Trans. A 2003, 34, 1565–1573. [Google Scholar] [CrossRef]
- Jack, D.H.; Jack, K.H. Carbides and nitrides in steel. Mater. Sci. Eng. 1973, 11, 1–27. [Google Scholar] [CrossRef]
- Bagaryatskii, Y.A. Probable mechanism of martensite decomposition. Dokl. Acad. Nauk SSSR 1950, 73, 1161–1164. (In Russian) [Google Scholar]
- Andrews, K.W. Tabulation of interplanar spacings of cementite Fe3C. Acta Crystallogr. 1963, 16, 68. [Google Scholar] [CrossRef]
- Agrawal, B.K. Introduction to Engineering Materials; Tata McGraw-Hill: New Delhi, India, 1988. [Google Scholar]
- Irvine, J.; Baker, T.N. The influence of rolling variables on the strengthening mechanisms operating in Niobium steels. Mater. Sci. Eng. 1984, 64, 123–134. [Google Scholar] [CrossRef]
- Khalid, E.A.; Edmonds, D.V. Mixed structures in continuously cooled low-carbon automotive steels. J. Phys. IV 1993, 3, 147–152. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G.; Capdevila, C.; Garcia de Andres, C. Estimation of dislocation density in bainitic microstructures using high-resolution dilatometry. Scr. Mater. 2009, 61, 855–858. [Google Scholar]
- Stevens, W.; Haynes, A.G. The temperature of formation of martensite and bainite in low-alloy steel. J. Iron Steel Inst. 1956, 183, 349–359. [Google Scholar]
- Kunitake, T.; Okada, Y. The estimation of bainite transformation temperatures in steels by the empirical formulas. J. Iron Steel Inst. Jpn. 1998, 84, 137–141. [Google Scholar] [CrossRef]
- Lee, Y.-K. Empirical formula of isothermal bainite start temperature of steel. J. Mater. Sci. Lett. 2002, 21, 1253–1255. [Google Scholar] [CrossRef]
- Lee, Y.-K.; Hong, J.-M.; Choi, C.-S.; Lee, J.-K. Continuous cooling transformation temperatures and microstructures of niobium bearing microalloyed steels. Mater. Sci. Forum 2005, 475–479, 65–68. [Google Scholar]
- Carpenter, K.R.; Killmore, C.R. The effect of Nb on the continuous cooling transformation curves of ultra-thin strip CASTRIP© Steels. Metals 2015, 5, 1857–1877. [Google Scholar] [CrossRef]
- Lambers, H.G.; Tschumak, S.; Maier, H.J.; Canading, D. Role of austenization and pre-deformation on the kinetics of the isothermal bainite transformation. Metall. Mater. Trans. A 2009, 40, 1355–1366. [Google Scholar] [CrossRef]
- Larn, R.H.; Yang, J.R. The effect of compressive deformation of austenite on the bainitic ferrite transformation in Fe-Mn-Si-C steels. Mater. Sci. Eng. A 2000, 278, 278–291. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Bainite in Steels; IOM Communications: London, UK, 2001; p. 221. [Google Scholar]
- Matsuzaki, A.; Bhadeshia, H.K.D.H. Effect of austenite grain size and bainite morphology on overall kinetics of bainite transformation in steels. Mater. Sci. Technol. 1999, 15, 518–522. [Google Scholar] [CrossRef]
- Kang, S.; Yoon, S.; Lee, S.-J. Prediction of bainite start temperature in alloy steels with different grain sizes. ISIJ Int. 2014, 54, 997–999. [Google Scholar] [CrossRef]
- Wada, H.; Pehlke, R.D. Nitrogen solubility and nitride formation in austenitic Fe-Ti alloys. Matall. Trans. B 1985, 16, 815–822. [Google Scholar] [CrossRef]
- Tagashira, K.; Mutsuji, T.; Endo, T. Effect of Mo-C dipole on the development of {111} recrystllisation texture in Mo added low carbon steels. Tetsu-to-Hagané 2000, 86, 466–471. [Google Scholar] [CrossRef]
- Bata, V. Minimising Ageing due to Carbon in Low Carbon Sheet Steel with the Aid of Mn, Cr, Mo or B Addition. Ph.D. Thesis, Monash University, Melbourne, Australia, June 2006. [Google Scholar]
- Johnson, R.A.; Diens, G.I.; Damask, A.C. Calculations of the energy and migration characteristics of carbon and nitrogen in α-iron and vanadium. Acta Metall. 1964, 12, 1215–1224. [Google Scholar] [CrossRef]
- Johnson, R.A. Clustering of carbon atoms in α-iron. Acta Metall. 1967, 15, 513–517. [Google Scholar] [CrossRef]
- Liu, P.; Xing, W.; Cheng, X.; Li, D.; Li, Y.; Chen, X.-Q. Effects of dilute substitutional solutes on interstitial carbon in α-Fe: Interactions and associated carbon diffusion from first-principles calculations. Phys. Rev. B 2014, 90, 024103. [Google Scholar] [CrossRef]
- Golovin, I.S.; Blanter, M.S.; Magalas, L.B. Interactions of dissolved atoms and carbon diffusion in Fe-Cr and FeAl alloys. Defect Diffus. Forum 2001, 194–199, 73–78. [Google Scholar] [CrossRef]
- Pereloma, E.V.; Bata, V.; Scott, R.I.; Smith, R.M. Effect of Cr and Mo on strain ageing behaviour of low carbon steel. Mater. Sci. Eng. A 2010, 527, 2538–2546. [Google Scholar] [CrossRef]
- Sandomirskij, M.M.; Grigorkin, V.I.; Zemskij, S.V. Alloying element effect on carbon diffusion in ferrite of pearlitic steel at tempering. Izv. Vysshikh Uchebnykh Zavedenij. Chernaya Metall. 1985, 5, 116–119. [Google Scholar]
- Fleischer, R.L. Solution hardening. Acta Metall. 1961, 9, 996–1000. [Google Scholar] [CrossRef]
- Dingly, D.J.; McLean, D. Components of the flow stress of iron. Acta Metall. 1967, 15, 885–901. [Google Scholar] [CrossRef]
- Gladman, T. The Physical Metallurgy of Microalloyed Steels; The Institute of Materials, Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Seidman, D.N.; Marquis, E.A.; Dunand, D.C. Precipitation strengthening at ambient and elevated temperatures of heat-treatable Al(Sc) alloys. Acta Mater. 2002, 50, 4021–4035. [Google Scholar] [CrossRef]
- Zhang, X.; Hickel, T.; Rogal, J.; Fähler, S.; Drautz, R.; Neugebauer, J. Structural transformations among austenite, ferrite and cementite in Fe–C alloys: A unified theory based on ab initio simulations. Acta Mater. 2015, 99, 281–289. [Google Scholar] [CrossRef]
- Argon, A.S. Mechanical properties of single-phase crystalline media: Deformation at low temperature. In Physical Metallurgy; Cahn, R.W., Haasen, P., Eds.; North-Holland Publishing: Amsterdam, The Netherlands, 1996. [Google Scholar]
- Timokhina, I.B.; Enomoto, M.; Miller, M.K.; Pereloma, E.V. Microstructure-property relationship in the thermomechanically processed C-Mn-Si-Nb-Al-(Mo) transformation-induced plasticity steels before and after prestraining and bake hardening treatment. Metall. Mater. Trans. A 2012, 43, 2473–2483. [Google Scholar] [CrossRef]
- Xie, K.Y.; Zheng, T.; Cairney, J.M.; Kaul, H.; Williams, J.G.; Barbaro, F.J.; Killmore, C.R.; Ringer, S.P. Strengthening from Nb-rich clusters in a Nb-microalloyed steel. Scr. Mater. 2012, 66, 710–713. [Google Scholar] [CrossRef]
- Kostryzhev, A.G.; Al Shahrani, A.; Zhu, C.; Cairney, J.M.; Ringer, S.P.; Killmore, C.R.; Pereloma, E.V. Effect of niobium clustering and precipitation on strength of a NbTi-microalloyed ferritic steel. Mater. Sci. Eng. A 2014, 607, 226–235. [Google Scholar] [CrossRef]
- Zhang, Y.-J.; Miyamoto, G.; Shinbo, K.; Furuhara, T.; Ohmura, T.; Suzuki, T.; Tsuzaki, K. Effects of transformation temperature on VC interphase precipitation and resultant hardness in low-carbon steels. Acta Mater. 2015, 84, 375–384. [Google Scholar] [CrossRef]
- Hansen, N.; Barlow, C.Y. Microstructure evolution in whisker- and particle-containing materials. In Fundamentals of Metal-Matrix Composites; Surech, S., Mortensen, A., Needleman, A., Eds.; Elsevier: Boston, MA, USA, 1993; pp. 109–118. [Google Scholar]
- Humphreys, F.J.; Hatherly, M. Recrystallisation and Related Annealing Phenomena; Elsevier: Oxford, UK, 2004. [Google Scholar]







| Parameters | MoNbV Steel | CrNbV Steel | |||
|---|---|---|---|---|---|
| Low Strain | High Strain | Low Strain | High Strain | ||
| size of bainitic ferrite areas #, μm | 0.95 ± 0.45 | 0.84 ± 0.42 | 0.72 ± 0.33 | 0.91 ± 0.45 | |
| martensite | fraction, % | 20 | 11 | 20 | 17 |
| average size of blocky grains, μm | 1.4 ± 0.7 | 1.4 ± 0.6 | 1.2 ± 0.7 | 2.3 ± 2.0 | |
| maximum size of blocky grains, μm | 5.0 | 2.8 | 4.2 | 10.4 | |
| average length of elongated grains, μm | 2.8 ± 1.2 | 3.0 ± 1.3 | 2.7 ± 0.9 | 3.7 ± 1.8 | |
| maximum length of elongated grains, μm | 8.3 | 8.0 | 4.8 | 10.4 | |
| >20 nm particles (SEM) | average size, nm | 42 ± 15 | 21 ± 4 | 27 ± 12 | 26 ± 13 |
| number density, μm−2 | 0.27 | 0.91 | 0.95 | 1.67 | |
| area fraction | 0.0003 | 0.0003 | 0.0006 | 0.0011 | |
| chemistry | 75% with MoNbV 25% with Mo | 36% with MoNbV 64% with Mo | 59% with NbV 41% with Nb | 58% with NbV 42% with Nb | |
| <20 nm particles (TEM) | average size, nm | 2.4 ± 0.5 | 3.2 ± 1.0 | 2.7 ± 0.7 | 2.8 ± 1.0 |
| number density, μm−3 | 15,667 | 9875 | 25,595 | 16,744 | |
| volume fraction | 0.0001 | 0.0002 | 0.0003 | 0.0003 | |
| chemistry | Cementite, Fe3C | ||||
| dislocation density, ×1015 m−2 | 0.93 ± 0.15 | 0.43 ± 0.10 | 0.85 ± 0.11 | 0.41 ± 0.10 | |
| matrix unite cell size, nm | 0.310 | 0.312 | 0.308 | 0.306 | |
| YS, MPa | 850 ± 30 | 775 ± 35 | 765 ± 30 | 700 ± 20 | |
| UTS, MPa | 1200 ± 45 | 1090 ± 40 | 1000 ± 25 | 975 ± 20 | |
| Elongation, % | 14 ± 2 | 13.5 ± 2 | 16 ± 3 | 19.5 ± 2 | |
| Steel | Strain | Calculated Contributions | Experimental YS | Δ * | |||||
|---|---|---|---|---|---|---|---|---|---|
| Grain Boundaries # | Particles | Dislocation Density | Solid Solute (Si, Mn) | Total | |||||
| >20 nm | >20 nm | ||||||||
| MoNbV | Low | 586 | 19 | 21 | 62 | 73 | 761 | 850 | +89 |
| High | 618 | 31 | 30 | 42 | 73 | 794 | 775 | −19 | |
| CrNbV | Low | 675 | 37 | 37 | 58 | 73 | 880 | 765 | −115 |
| High | 595 | 52 | 37 | 40 | 73 | 797 | 700 | −97 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostryzhev, A.; Singh, N.; Chen, L.; Killmore, C.; Pereloma, E. Comparative Effect of Mo and Cr on Microstructure and Mechanical Properties in NbV-Microalloyed Bainitic Steels. Metals 2018, 8, 134. https://doi.org/10.3390/met8020134
Kostryzhev A, Singh N, Chen L, Killmore C, Pereloma E. Comparative Effect of Mo and Cr on Microstructure and Mechanical Properties in NbV-Microalloyed Bainitic Steels. Metals. 2018; 8(2):134. https://doi.org/10.3390/met8020134
Chicago/Turabian StyleKostryzhev, Andrii, Navjeet Singh, Liang Chen, Chris Killmore, and Elena Pereloma. 2018. "Comparative Effect of Mo and Cr on Microstructure and Mechanical Properties in NbV-Microalloyed Bainitic Steels" Metals 8, no. 2: 134. https://doi.org/10.3390/met8020134
APA StyleKostryzhev, A., Singh, N., Chen, L., Killmore, C., & Pereloma, E. (2018). Comparative Effect of Mo and Cr on Microstructure and Mechanical Properties in NbV-Microalloyed Bainitic Steels. Metals, 8(2), 134. https://doi.org/10.3390/met8020134







