Hydrogen Interaction with Deep Surface Modified Zr-1Nb Alloy by High Intensity Ti Ion Implantation
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Implantation Procedure
2.2. Hydrogenation
2.3. Characterization
3. Results and Discussion
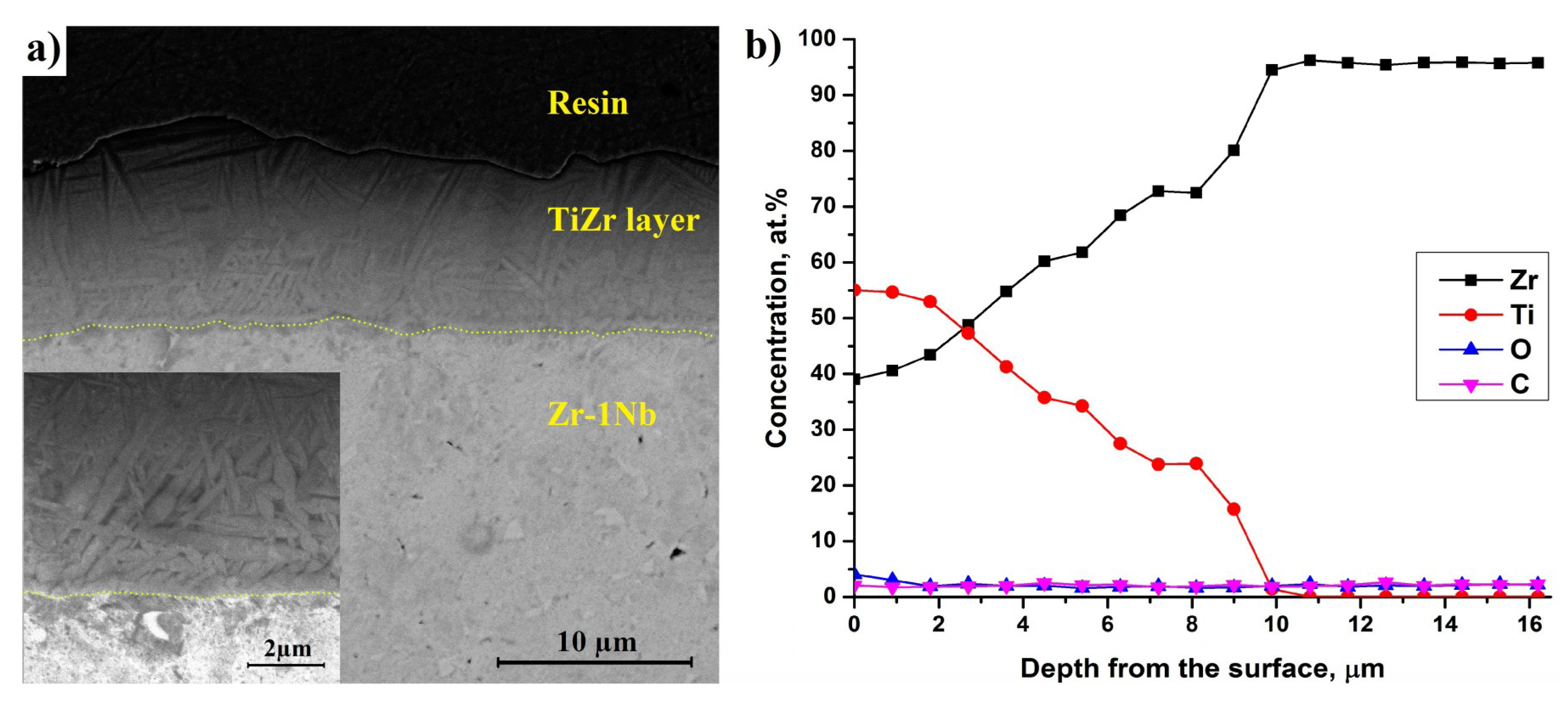
3.1. Structure and Composition of Ti-Implanted Zr-1Nb Alloy
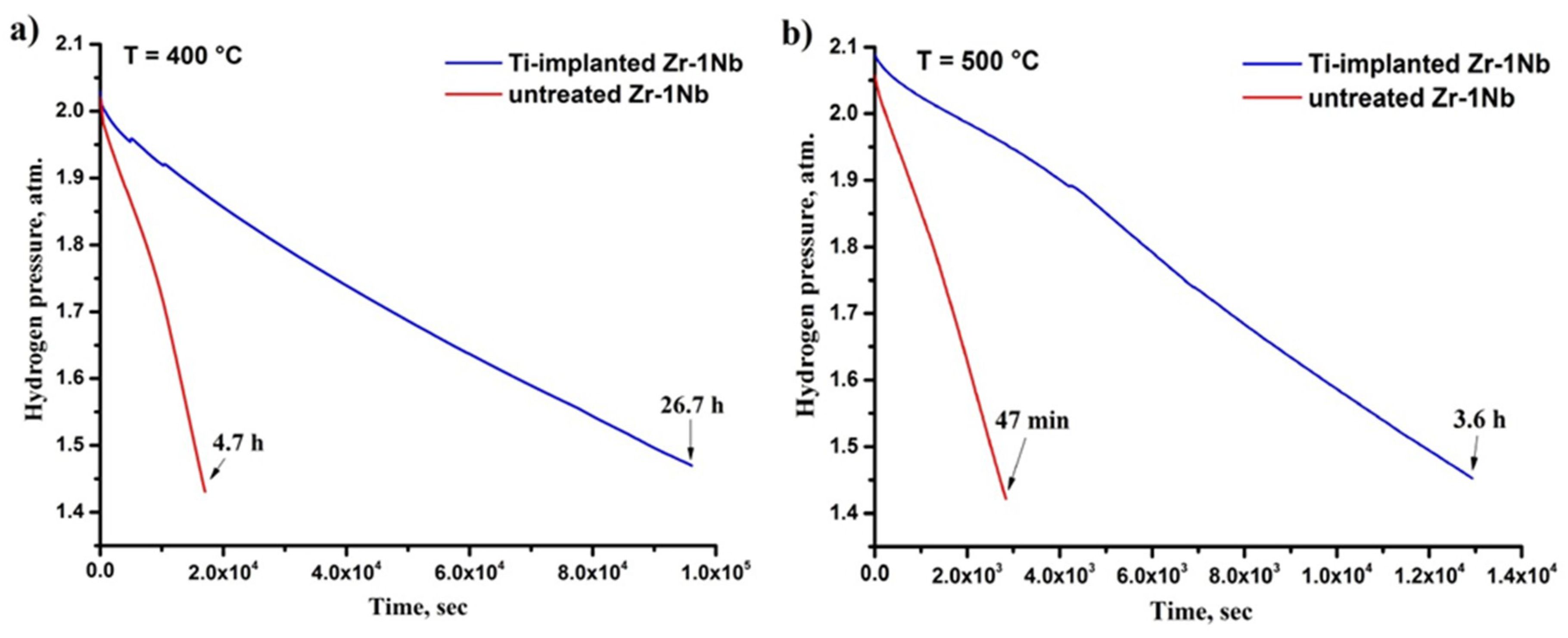
3.2. Hydrogen Sorption Behavior
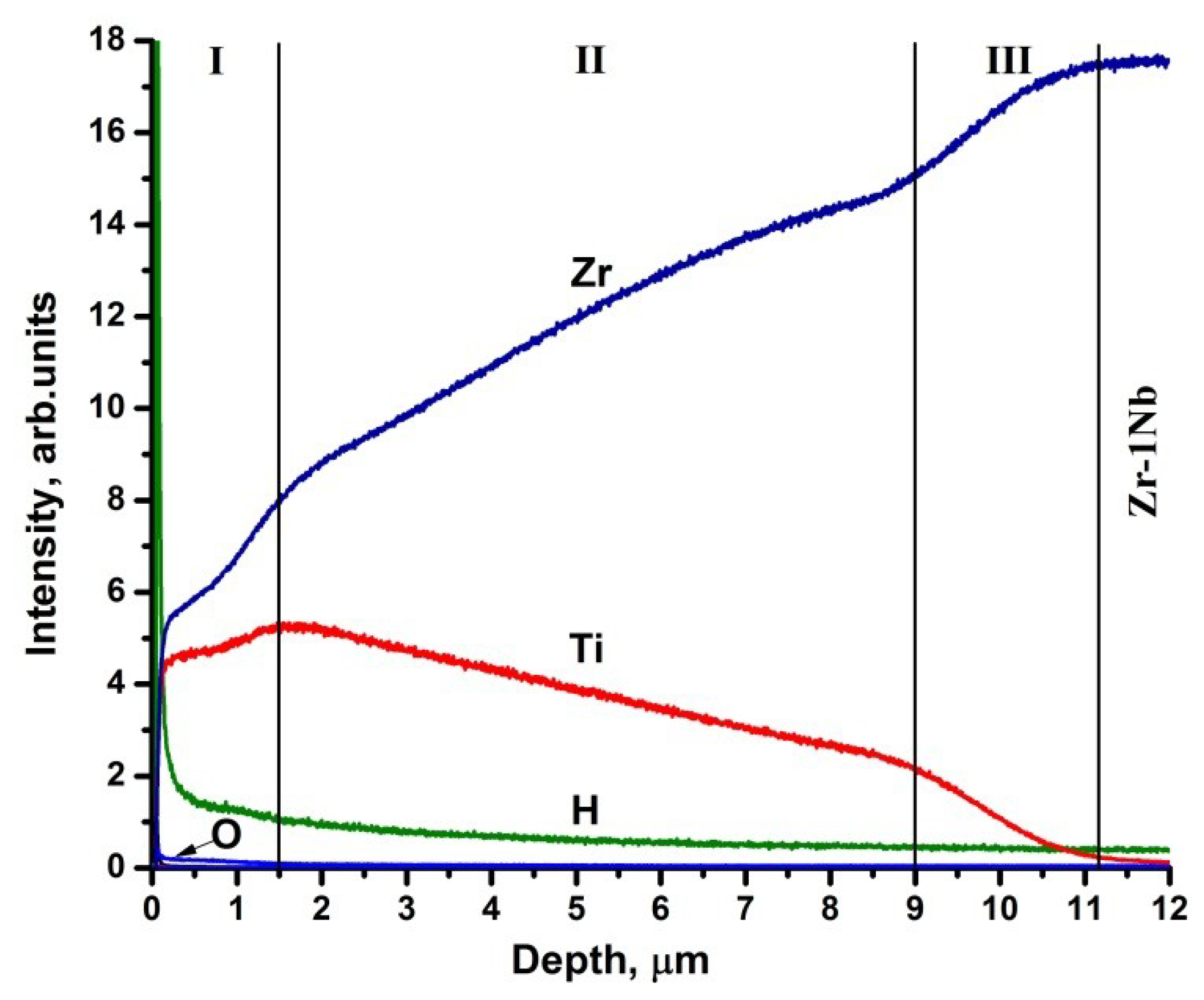
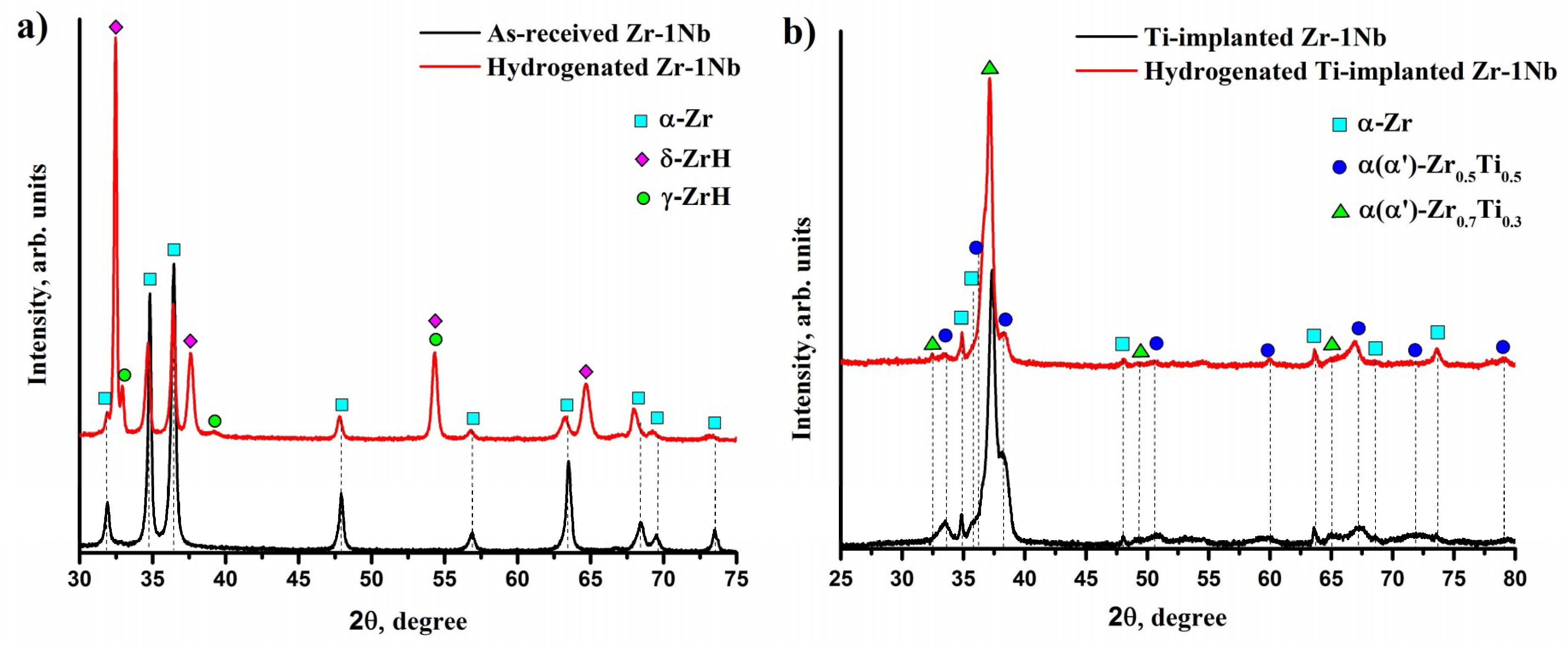
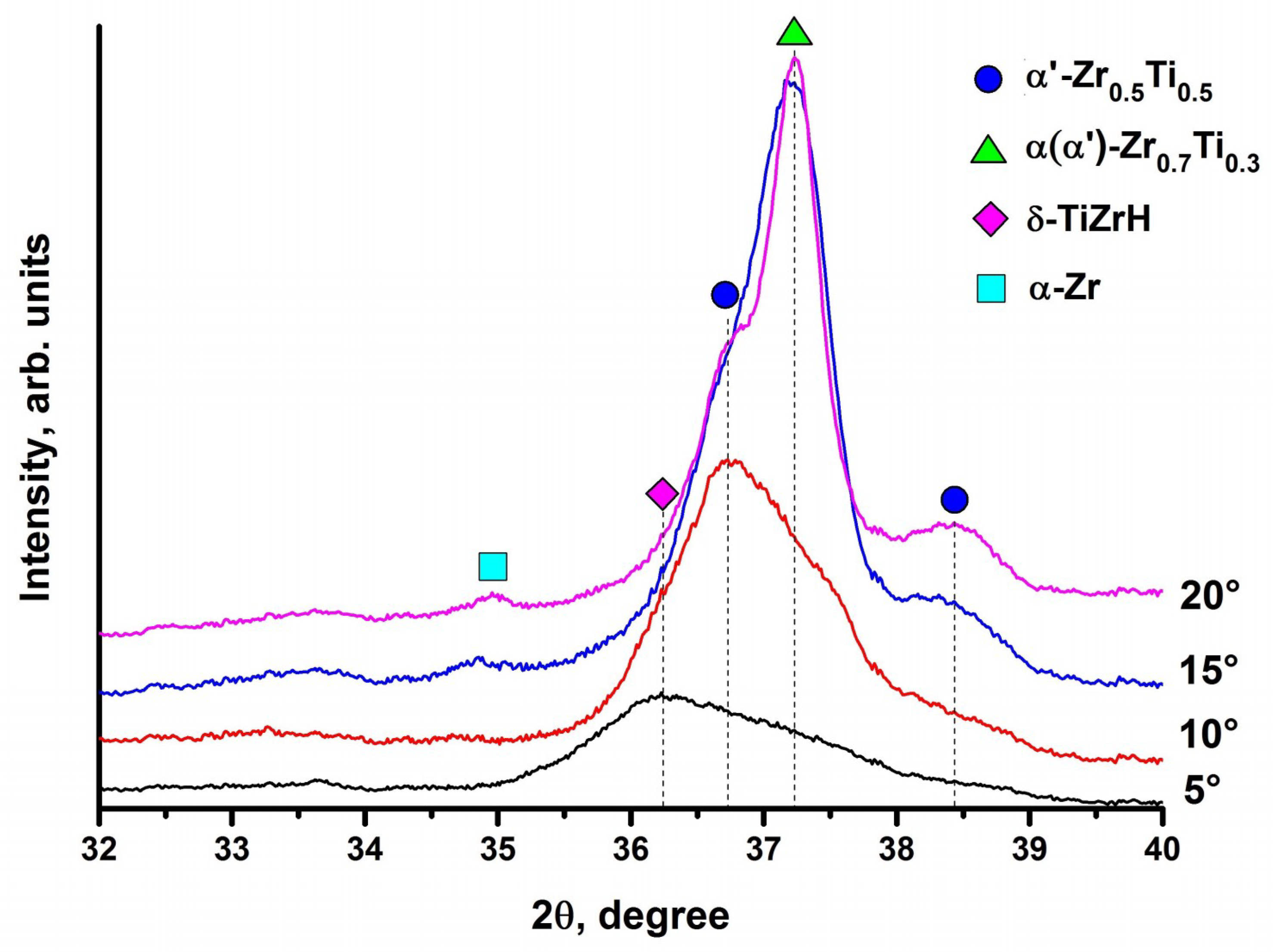
3.3. Structure and Elemental Distribution in Hydrogenated Samples
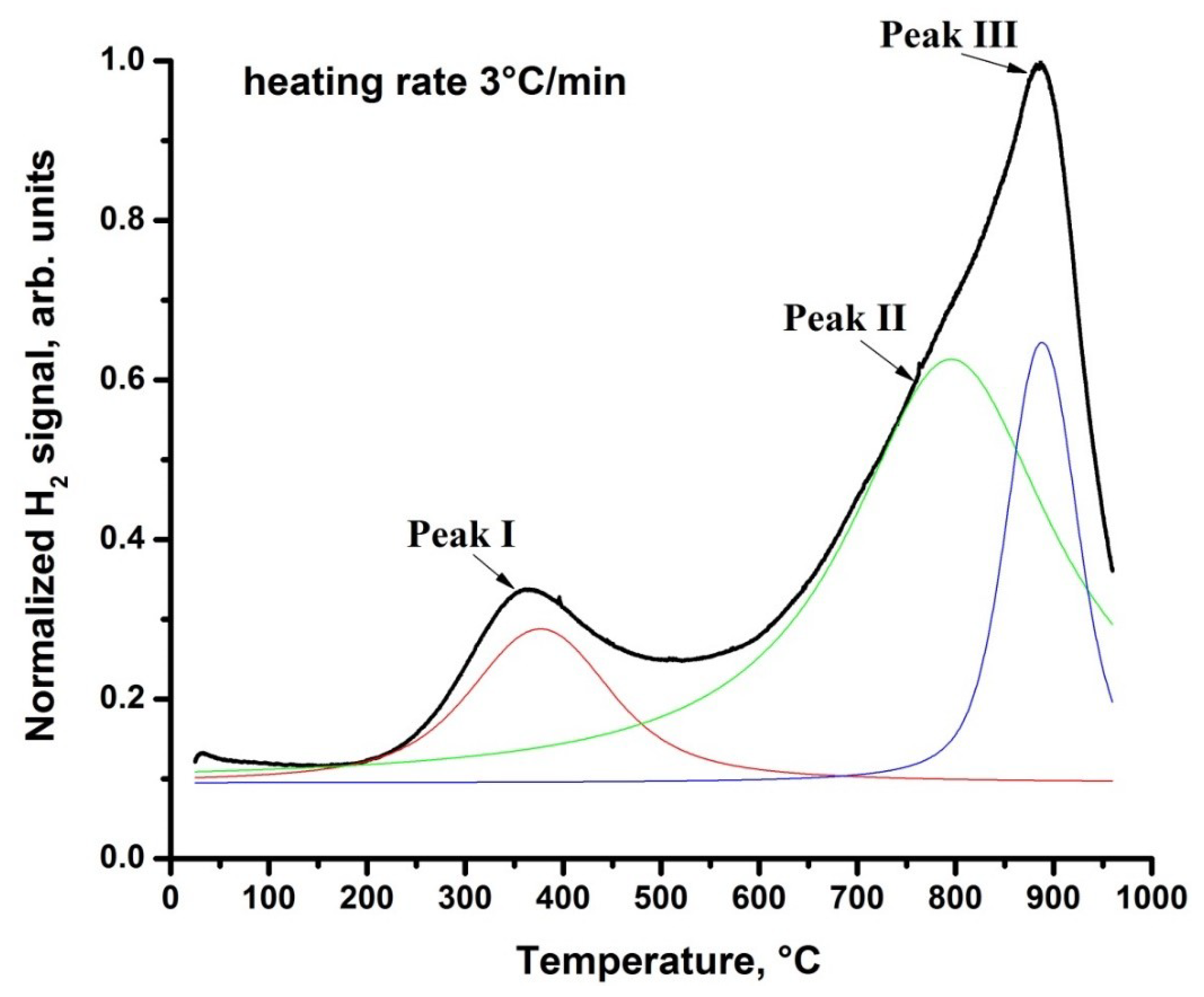
3.4. Thermal Desorption Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sarkar, A.; Chandanshive, S.A.; Thota, M.K.; Kapoor, R. High temperature deformation behavior of Zr-1Nb alloy. J. Alloys Compd. 2017, 703, 56–66. [Google Scholar] [CrossRef]
- Lee, C.M.; Mok, Y.-K.; Sohn, D.-S. High-temperature steam oxidation and oxide crack effects of Zr-1Nb-1Sn-0.1Fe fuel cladding. J. Nucl. Mater. 2017, 496, 343–352. [Google Scholar] [CrossRef]
- Ensor, B.; Lucente, A.M.; Frederick, M.J.; Sutliff, J.; Motta, A.T. The role of hydrogen in zirconium alloy corrosion. J. Nucl. Mater. 2017, 496, 301–312. [Google Scholar] [CrossRef]
- Kishore, R. Effect of hydrogen on the creep behavior of Zr-2.5%Nb alloy at 723 K. J. Nucl. Mater. 2009, 385, 591–594. [Google Scholar] [CrossRef]
- Wei, T.; Long, C.; Chen, H. Hydrogen uptake behaviors of Zr-1.0Cr-0.4Fe-(0.2Mo) and N18 alloys during corrosion in 500 °C steam. J. Alloys Compd. 2018, 731, 126–134. [Google Scholar] [CrossRef]
- Couet, A.; Motta, A.T.; Comstock, R.J. Hydrogen pickup measurements in zirconium alloys: Relation to oxidation kinetics. J. Nucl. Mater. 2014, 451, 1–13. [Google Scholar] [CrossRef]
- Tagtstrom, P.; Limback, M.; Dahlback, M.; Andersson, T.; Pettersson, H. Effects of Hydrogen Pickup and Second- Phase Particle Dissolution on the In-Reactor Corrosion Performance of BWR Claddings. In Zirconium in the Nuclear Industry: 13th International Symposium, ASTM STP 1423; ASTM International: West Conshohocken, PA, USA, 2002; pp. 96–118. [Google Scholar]
- Krishna, K.M.; Srivastava, D.; Dey, G.K.; Hiwarkar, V.; Samajdar, I.; Banerjee, S. Role of grain/phase boundary nature on the formation of hydrides in Zr–2.5% Nb alloy. J. Nucl. Mater. 2011, 414, 270–275. [Google Scholar] [CrossRef]
- Bair, J.; Asle Zaeem, M.; Tonks, M. A review on hydride precipitation in zirconium alloys. J. Nucl. Mater. 2015, 466, 12–20. [Google Scholar] [CrossRef]
- McRae, G.A.; Coleman, C.E.; Leitch, B.W. The first step for delayed hydride cracking in zirconium alloys. J. Nucl. Mater. 2010, 396, 130–143. [Google Scholar] [CrossRef]
- Große, M.; Lehmann, E.; Steinbrück, M.; Kühne, G.; Stuckert, J. Influence of oxide layer morphology on hydrogen concentration in tin and niobium containing zirconium alloys after high temperature steam oxidation. J. Nucl. Mater. 2009, 385, 339–345. [Google Scholar] [CrossRef]
- Terrani, K.A. Accident tolerant fuel cladding development: Promise, status, and challenges. J. Nucl. Mater. 2018, 501, 13–30. [Google Scholar] [CrossRef]
- Cox, B. Some thoughts on the mechanisms of in-reactor corrosion of zirconium alloys. J. Nucl. Mater. 2005, 336, 331–368. [Google Scholar] [CrossRef]
- Duan, Z.; Yang, H.; Satoh, Y.; Murakami, K.; Kano, S.; Zhao, Z.; Shen, J.; Abe, H. Current status of materials development of nuclear fuel cladding tubes for light water reactors. Nucl. Eng. Des. 2017, 316, 131–150. [Google Scholar] [CrossRef]
- Kuprin, A.S.; Belous, V.A.; Voyevodin, V.N.; Vasilenko, R.L.; Ovcharenko, V.D.; Tolstolutskaya, G.D.; Kopanets, I.E.; Kolodiy, I.V. Irradiation resistance of vacuum arc chromium coatings for zirconium alloy fuel claddings. J. Nucl. Mater. 2018, 510, 163–167. [Google Scholar] [CrossRef]
- Bischoff, J.; Delafoy, C.; Vauglin, C.; Barberis, P.; Roubeyrie, C.; Perche, D.; Duthoo, D.; Schuster, F.; Brachet, J.-C.; Schweitzer, E.W.; et al. AREVA NP’s enhanced accident-tolerant fuel developments: Focus on Cr-coated M5 cladding. Nucl. Eng. Technol. 2018, 50, 223–228. [Google Scholar] [CrossRef]
- Alat, E.; Motta, A.T.; Comstock, R.J.; Partezana, J.M.; Wolfe, D.E. Multilayer (TiN, TiAlN) ceramic coatings for nuclear fuel cladding. J. Nucl. Mater. 2016, 478, 236–244. [Google Scholar] [CrossRef]
- Kim, I.; Khatkhatay, F.; Jiao, L.; Swadener, G.; Cole, J.I.; Gan, J.; Wang, H. TiN-based coatings on fuel cladding tubes for advanced nuclear reactors. J. Nucl. Mater. 2012, 429, 143–148. [Google Scholar] [CrossRef]
- Khatkhatay, F.; Jiao, L.; Jian, J.; Zhang, W.; Jiao, Z.; Gan, J.; Zhang, H.; Zhang, X.; Wang, H. Superior corrosion resistance properties of TiN-based coatings on Zircaloy tubes in supercritical water. J. Nucl. Mater. 2014, 451, 346–351. [Google Scholar] [CrossRef]
- Zou, Z.; Xue, W.; Jia, X.; Du, J.; Wang, R.; Weng, L. Effect of voltage on properties of microarc oxidation films prepared in phosphate electrolyte on Zr-1Nb alloy. Surf. Coat. Technol. 2013, 222, 62–67. [Google Scholar] [CrossRef]
- Van Nieuwenhove, R.; Andersson, V.; Balak, J.; Oberländer, B. In-Pile Testing of CrN, TiAlN, and AlCrN Coatings on Zircaloy Cladding in the Halden Reactor. In Zirconium in the Nuclear Industry: 18th International Symposium; ASTM International: West Conshohocken, PA, USA, 2018; pp. 965–982. [Google Scholar]
- Pushilina, N.S.; Kudiiarov, V.N.; Lider, A.M.; Teresov, A.D. Influence of surface structure on hydrogen interaction with Zr–1Nb alloy. J. Alloys Compd. 2015, 645, S476–S479. [Google Scholar] [CrossRef]
- Kashkarov, E.; Nikitenkov, N.; Sutygina, A.; Laptev, R.; Bordulev, Y.; Obrosov, A.; Liedke, M.O.; Wagner, A.; Zak, A.; Weiβ, S. Microstructure, defect structure and hydrogen trapping in zirconium alloy Zr-1Nb treated by plasma immersion Ti ion implantation and deposition. J. Alloys Compd. 2018, 732, 80–87. [Google Scholar] [CrossRef]
- Kashkarov, E.B.; Nikitenkov, N.N.; Sutygina, A.N.; Obrosov, A.; Manakhov, A.; Polčák, J.; Weiß, S. Hydrogen absorption by Ti-implanted Zr-1Nb alloy. Int. J. Hydrog. Energy 2018, 43, 2484–2491. [Google Scholar] [CrossRef]
- Peng, D.Q.; Bai, X.D.; Pan, F.; Sun, H.; Chen, B.S. Influence of titanium ions implantation on corrosion behavior of zirconium in 1 M H2SO4. Appl. Surf. Sci. 2006, 252, 2196–2203. [Google Scholar] [CrossRef]
- Obrosov, A.; Sutygina, A.; Manakhov, A.; Bolz, S.; Weiß, S.; Kashkarov, E. Oxidation Behavior of Zr–1Nb Corroded in Air at 400 °C after Plasma Immersion Titanium Implantation. Metals 2018, 8, 27. [Google Scholar] [CrossRef]
- Ryabchikov, A.I.; Kashkarov, E.B.; Pushilina, N.S.; Syrtanov, M.S.; Shevelev, A.E.; Korneva, O.S.; Sutygina, A.N.; Lider, A.M. High-intensity low energy titanium ion implantation into zirconium alloy. Appl. Surf. Sci. 2018, 439, 106–112. [Google Scholar] [CrossRef]
- Al-Nawas, B.; Brägger, U.; Meijer, H.J.A.; Naert, I.; Persson, R.; Perucchi, A.; Quirynen, M.; Raghoebar, G.M.; Reichert, T.E.; Romeo, E.; et al. A Double-Blind Randomized Controlled Trial (RCT) of Titanium-13Zirconium versus Titanium Grade IV Small-Diameter Bone Level Implants in Edentulous Mandibles—Results from a 1-Year Observation Period. Clin. Implant Dent. Relat. Res. 2012, 14, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.A.; Rocha-Filho, R.C.; Biaggio, S.R.; Bocchi, N. Corrosion resistance of the Ti–50Zr at.% alloy after anodization in different acidic electrolytes. Corros. Sci. 2010, 52, 4058–4063. [Google Scholar] [CrossRef]
- Li, Y.; Cui, Y.; Zhang, F.; Xu, H. Shape memory behavior in Ti-Zr alloys. Scr. Mater. 2011, 64, 584–587. [Google Scholar] [CrossRef]
- Frank, M.J.; Walter, M.S.; Lyngstadaas, S.P.; Wintermantel, E.; Haugen, H.J. Hydrogen content in titanium and a titanium–zirconium alloy after acid etching. Mater. Sci. Eng. C 2013, 33, 1282–1288. [Google Scholar] [CrossRef]
- Ryabchikov, A.I.; Ananin, P.S.; Dektyarev, S.V.; Sivin, D.O.; Shevelev, A.E. High intensity metal ion beam generation. Vacuum 2017, 143, 447–453. [Google Scholar] [CrossRef]
- Stout, V.L.; Gibbons, M.D. Gettering of gas by titanium. J. Appl. Phys. 1955, 26, 1488–1492. [Google Scholar] [CrossRef]
- Anders, A. (Ed.) Handbook of Plasma Immersion Ion Implantation and Deposition; Wiley: New York, NY, USA, 2000; Volume 17, p. 46. [Google Scholar]
- Yushkov, G.Y.; Anders, A.; Oks, E.M.; Brown, I.G. Ion velocities in vacuum arc plasmas. J. Appl. Phys. 2000, 88, 5618–5622. [Google Scholar] [CrossRef]
- Ryabchikov, A.I.; Sivin, D.O.; Korneva, O.S.; Lopatin, I.V.; Ananin, P.S.; Prokopenko, N.A.; Akhmadeev, Y.K. High-current-density gas ion ribbon beam formation. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrom. Detect. Assoc. Equip. 2018, 906, 56–60. [Google Scholar] [CrossRef]
- Richmond, C.J. Ion Implantation; Ryssel, H., Ruge, I., Eds.; Wiley: Chichester, UK, 1986; 478p. [Google Scholar]
- Babikhina, M.N.; Kudiiarov, V.N.; Mostovshchikov, A.V.; Lider, A.M. Quantitative and Qualitative Analysis of Hydrogen Accumulation in Hydrogen-Storage Materials Using Hydrogen Extraction in an Inert Atmosphere. Metals 2018, 8, 672. [Google Scholar] [CrossRef]
- Dobromyslov, A.V.; Taluts, N.I. Structure investigation of quenched and tempered alloys of the Zr–Ti system. Phys. Met. Met. 1987, 63, 114–120. [Google Scholar]
- Zhou, Y.K.; Jing, R.; Ma, M.Z.; Liu, R.P. Tensile strength of Zr-Ti binary alloy. Chin. Phys. Lett. 2013, 30, 116201. [Google Scholar] [CrossRef]
- Liang, S.X.; Yin, L.X.; Zhou, Y.K.; Feng, X.J.; Ma, M.Z.; Liu, R.P.; Tan, C.L. Abnormal martensitic transformation of high Zr-containing Ti alloys. J. Alloys Compd. 2014, 615, 804–808. [Google Scholar] [CrossRef]
- Lin, C.; Yin, G.; Zhao, Y.; Ge, P.; Liu, Z. Analysis of the effect of alloy elements on martensitic transformation in titanium alloy with the use of valence electron structure parameters. Mater. Chem. Phys. 2011, 125, 411–417. [Google Scholar] [CrossRef]
- Ma, M.; Liang, L.; Wang, L.; Wang, Y.; Cheng, Y.; Tang, B.; Xiang, W.; Tan, X. Phase transformations of titanium hydride in thermal desorption process with different heating rates. Int. J. Hydrog. Energy 2015, 40, 8926–8934. [Google Scholar] [CrossRef]
- Suwarno, S.; Yartys, V.A. Kinetics of hydrogen absorption and desorption in titanium. Bull. Chem. React. Eng. Catal. 2017, 12, 312–317. [Google Scholar] [CrossRef]
- Laptev, R.S.; Syrtanov, M.S.; Kudiiarov, V.N.; Shmakov, A.N.; Vinokurov, Z.S.; Mikhaylov, A.A.; Zolotarev, K.V. In Situ Investigation of Thermo-stimulated Decay of Hydrides of Titanium and Zirconium by Means of X-ray Diffraction of Synchrotron Radiation. In Physics Procedia; Elsevier: Amsterdam, The Netherlands, 2016; Volume 84, pp. 337–341. [Google Scholar]
- Jiménez, C.; Garcia-Moreno, F.; Pfretzschner, B.; Klaus, M.; Wollgarten, M.; Zizak, I.; Schumacher, G.; Tovar, M.; Banhart, J. Decomposition of TiH2studied in situ by synchrotron X-ray and neutron diffraction. Acta Mater. 2011, 59, 6318–6330. [Google Scholar] [CrossRef]
- Ma, M.; Liang, L.; Tang, B.; Xiang, W.; Wang, Y.; Cheng, Y.; Tan, X. Decomposition kinetics study of zirconium hydride by interrupted thermal desorption spectroscopy. J. Alloys Compd. 2015, 645, S217–S220. [Google Scholar] [CrossRef]
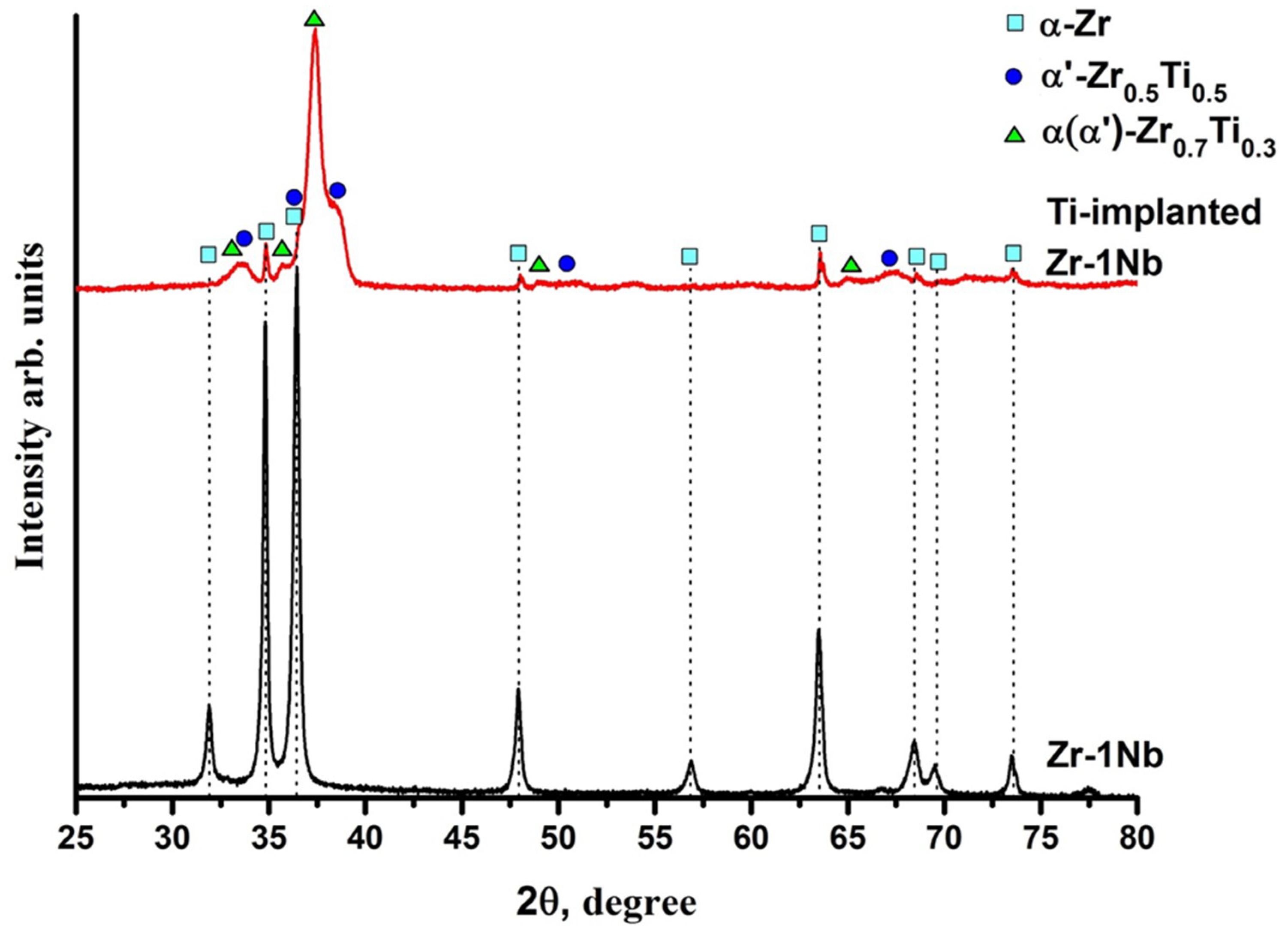

| Sample | Phase | Phase Content, % | Lattice Parameters, Å | c/a | |
|---|---|---|---|---|---|
| Zr-1Nb | α-Zr (hcp) | 100 | a = 3.2364 c = 5.1451 | 1.590 | 1.5 |
| Ti-implanted Zr-1Nb | α-Zr (hcp) | 4 | a = 3.2325 c = 5.1507 | 1.594 | 2.2 |
| Zr0.5Ti0.5(hcp) | 54 | a = 3.0886 c = 4.8438 | 1.568 | 4.3 | |
| Zr0.7Ti0.3(hcp) | 43 | a = 3.1613 c = 5.0229 | 1.589 | 4.8 |
| Temperature, °C | Hydrogen Absorption Rate Q, ×10−6 cm3Н2/(s∙cm2) | Reduction Ratio | |
|---|---|---|---|
| Zr-1Nb Alloy | Ti-implanted Zr-1Nb | ||
| 400 | 4.4 | 0.7 | 6.3 |
| 500 | 28.7 | 6.1 | 4.7 |
| Sample | Phase | Content, vol.% | Lattice Parameter, Å | c/a |
|---|---|---|---|---|
| Zr-1Nb | α-Zr (hcp) | 42 | a = 3.2347; c = 5.1624 | 1.596 |
| δ-ZrH1.66 (fcc) | 53 | a = 4.7654 | - | |
| γ-ZrH (fct) | 5 | a = 4.5811; c = 4.9612 | 1.083 | |
| Ti-implanted Zr-1Nb | α-Zr (hcp) | 4 | a = 3.2567; c = 5.1508 | 1.582 |
| Zr0.5Ti0.5 (hcp) | 50 | a = 3.0901; c = 4.8931 | 1.583 | |
| Zr0.7Ti0.3 (hcp) | 46 | a = 3.1899; c = 4.9612 | 1.555 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kashkarov, E.B.; Ryabchikov, A.I.; Kurochkin, A.V.; Syrtanov, M.S.; Shevelev, A.E.; Obrosov, A.; Weiß, S. Hydrogen Interaction with Deep Surface Modified Zr-1Nb Alloy by High Intensity Ti Ion Implantation. Metals 2018, 8, 1081. https://doi.org/10.3390/met8121081
Kashkarov EB, Ryabchikov AI, Kurochkin AV, Syrtanov MS, Shevelev AE, Obrosov A, Weiß S. Hydrogen Interaction with Deep Surface Modified Zr-1Nb Alloy by High Intensity Ti Ion Implantation. Metals. 2018; 8(12):1081. https://doi.org/10.3390/met8121081
Chicago/Turabian StyleKashkarov, Egor B., Alexander I. Ryabchikov, Alexander V. Kurochkin, Maxim S. Syrtanov, Alexey E. Shevelev, Aleksei Obrosov, and Sabine Weiß. 2018. "Hydrogen Interaction with Deep Surface Modified Zr-1Nb Alloy by High Intensity Ti Ion Implantation" Metals 8, no. 12: 1081. https://doi.org/10.3390/met8121081
APA StyleKashkarov, E. B., Ryabchikov, A. I., Kurochkin, A. V., Syrtanov, M. S., Shevelev, A. E., Obrosov, A., & Weiß, S. (2018). Hydrogen Interaction with Deep Surface Modified Zr-1Nb Alloy by High Intensity Ti Ion Implantation. Metals, 8(12), 1081. https://doi.org/10.3390/met8121081






