Alkali Treatment of Anodized Titanium Alloys Affects Cytocompatibility
Abstract
:1. Introduction
2. Experimental
2.1. Formation of Coatings
2.2. Bioactivity and Cytocompatibility
2.2.1. Simulated Body Fluid
2.2.2. Cytocompatibility
Cell Culture
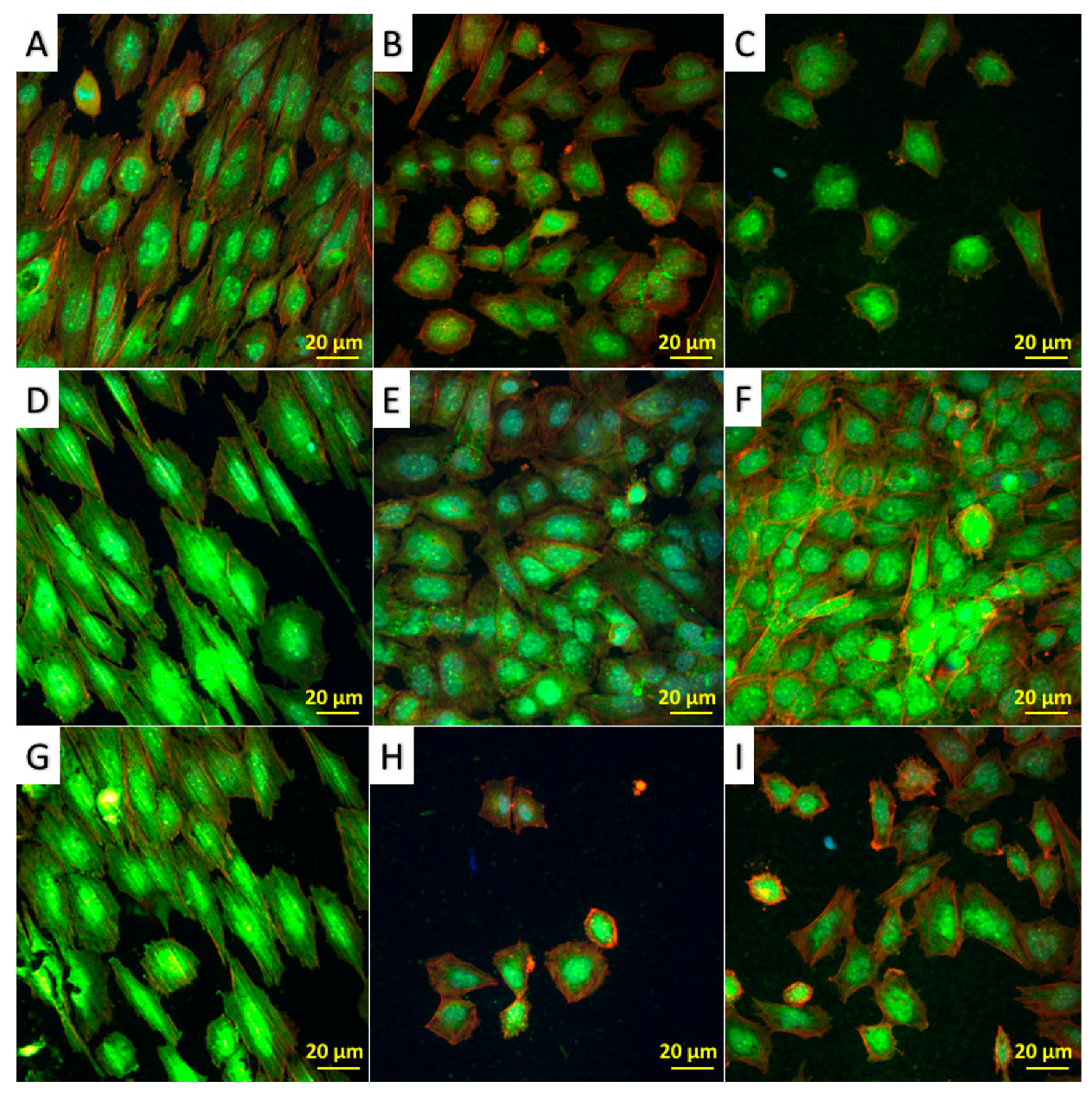
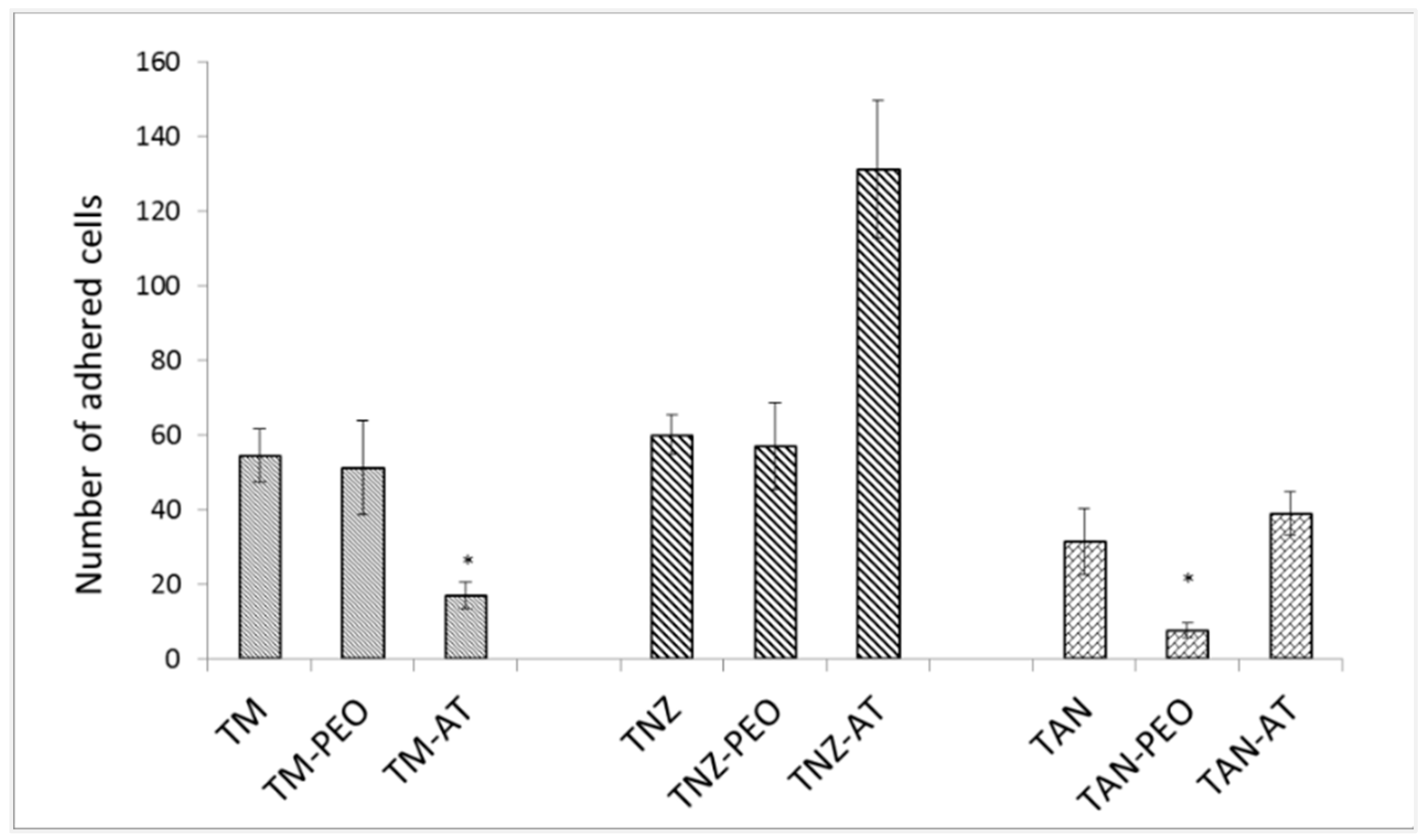
Initial Cell Adhesion and Cell Morphology
Statistical Analysis
Scanning Electron Microscopy at a Low Voltage (LV-SEM)
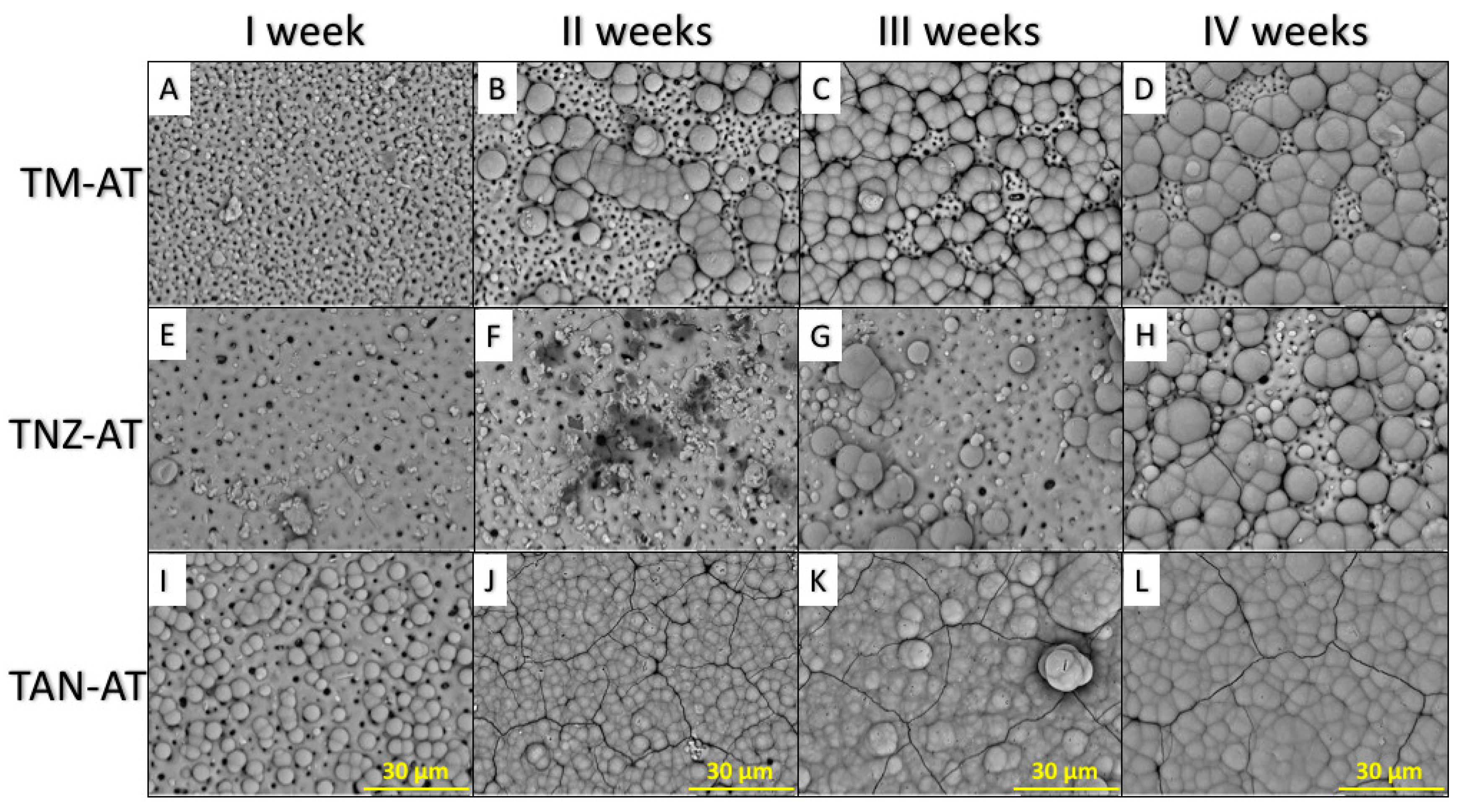
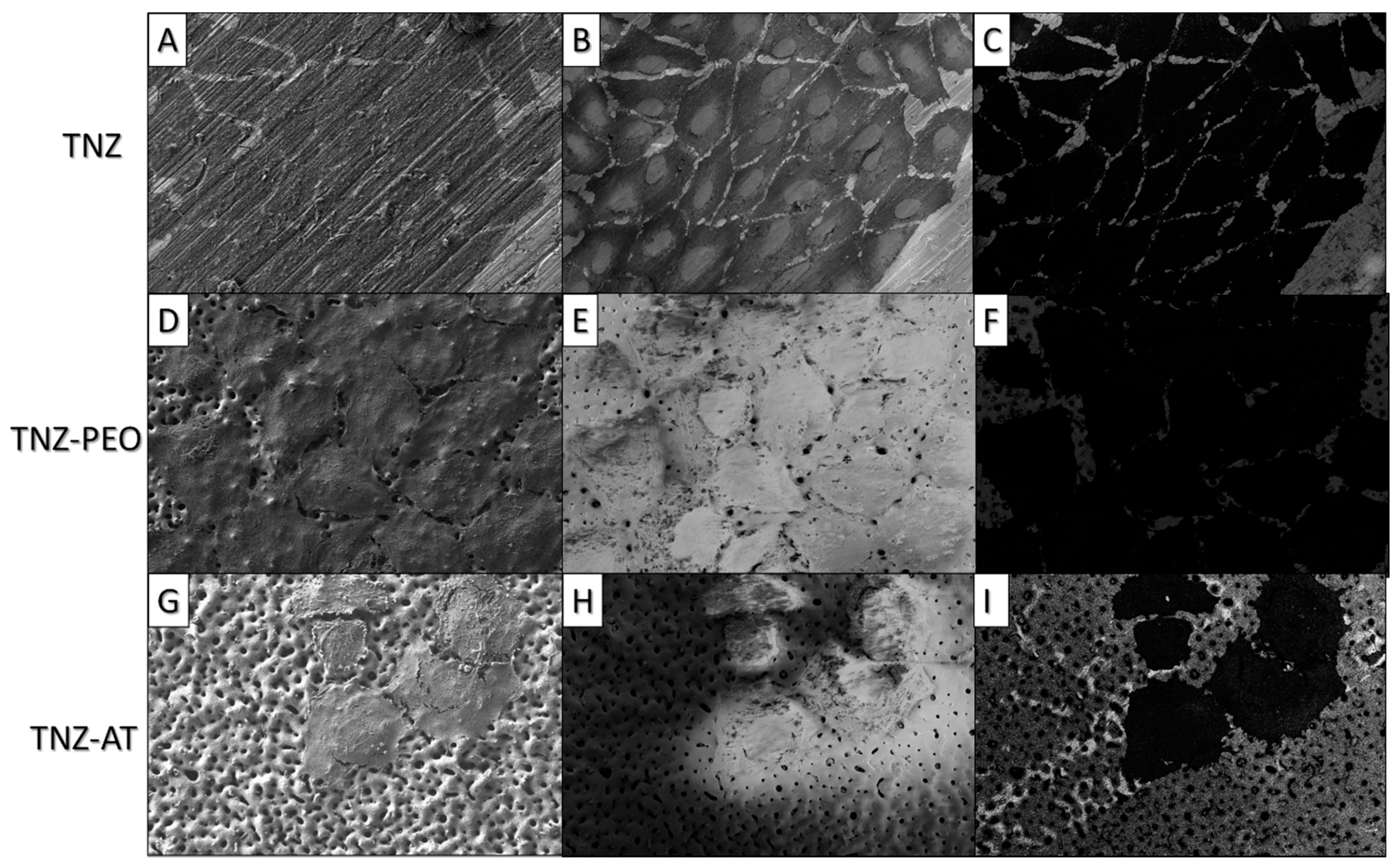
3. Results and Discussion
Bioactivity and Cytocompatibility of the Coatings
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Amini, A.R.; Laurecin, C.T.; Nukavarapu, S.P. Bone tissue engineering: Recent advances and challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.; Chen, M.T.; Liu, J.H. Structural evolution and adhesion of titanium oxide film containing phosphorus and calcium on titanium by anodic oxidation. J. Biomed. Mater. 2008, 85A, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Rokosz, K.; Hryniewicz, T.; Dudek, Ł.; Matýsek, D.; Valíček, J.; Harničárová, M. SEM and EDS analysis of surface layer formed on titanium after plasma electrolytic oxidation in phosphoric acid with the addition of copper nitrate (NanoOstrava 2015). J. Nanosci. Nanotechnol. 2016, 16, 7814–7817. [Google Scholar] [CrossRef]
- Rokosz, K.; Hryniewicz, T.; Raaen, S.; Chapon, P. Investigation of porous coatings obtained on Ti-Nb-Zr-Sn alloy biomaterial by plasma electrolytic oxidation: Characterisation and modelling. Int. J. Adv. Manuf. Technol. 2016, 87, 3497–3512. [Google Scholar] [CrossRef]
- Rokosz, K.; Hryniewicz, T.; Dalibor, M.; Raaen, S.; Valicek, J.; Dudek, Ł.; Harnicarova, M. SEM, EDS and XPS analysis of the coatings obtained on titanium after plasma electrolytic oxidation in electrolytes containing copper nitrate. Materials 2016, 9, 318. [Google Scholar] [CrossRef] [PubMed]
- Rokosz, K.; Hryniewicz, T.; Raaen, S.; Chapon, P.; Dudek, Ł. GDOES, XPS and SEM with EDS analysis of porous coatings obtained on titanium after plasma electrolytic oxidation, surface and interface analysis. Adv. Mater. Sci. 2016, 49, 303–315. [Google Scholar]
- Rokosz, K.; Hryniewicz, T.; Malorny, W. Characterisation of porous coatings obtained on materials by plasma electrolytc oxidation. Mater. Sci. Forum 2016, 862, 86–95. [Google Scholar] [CrossRef]
- Lu, X.; Mohedano, M.; Blawert, C.; Matykina, E.; Arrabal, R.; Kainer, K.U.; Zheludkevich, M.L. Plasma electrolytic oxidation coatings with particle additions—A review. Surf. Coat. Technol. 2016, 307, 1165–1182. [Google Scholar] [CrossRef]
- Shin, K.R.; Kim, J.S.; Kim, G.W.; Yang, H.W.; Ko, J.G.; Shin, D.H. Effects of concentration of Ag nanoparticles on surface structure and in vitro biological responses of oxide layer on pure titanium via plasma electrolytic oxidation. Appl. Surf. Sci. 2015, 347, 574–582. [Google Scholar] [CrossRef]
- Oliveira, N.T.C.; Guastaldi, A.C. Electrochemical stability and corrosion resistance of Ti-Mo alloys for biomedical applications. Acta Biomater. 2009, 5, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Y.; Xiong, J.; Hodgson, P.D.; Wen, C. Porous TiNbZr alloy scaffolds for biomedical applications. Acta Biomater. 2009, 5, 3616–3624. [Google Scholar] [CrossRef] [PubMed]
- Naritan, K.; Niinomi, M.; Nakai, M. Effects of micro- and nano-scale wave-like structures on fatigue strength of a beta-type titanium alloy developed as a biomaterial. J. Mech. Behav. Biomed. Mater. 2014, 29, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Morais, L.S.; Serra, G.G.; Palermo, E.F.A.; Andrade, L.R.; Müller, C.A.; Meyers, M.A.; Elias, C.N. Systemic levels of metallic ions released from orthodontic mini-implants. Am. J. Orthod. Dentofac. Orthop. 2009, 135, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Djouina, M.; Esquerre, N.; Desreumaux, P.; Vignal, C.; Body-Malapel, M. Toxicological consequences of experimental exposure to aluminum in human intestinal epithelial cells. Food Chem. Toxicol. 2016, 91, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Zaffea, D.; Bertoldi, C.; Consolo, U. Accumulation of aluminium in lamellar bone after implantation of titanium plates, Ti-6Al-4V screws, hydroxyapatite granules. Biomaterials 2004, 25, 3837–3844. [Google Scholar] [CrossRef] [PubMed]
- Czajka, M.; Sawicki, K.; Sikorska, K.; Popek, S.; Kruszewski, M.; Kapka-Skrzypczak, L. Toxicity of titanium dioxide nanoparticles in central nervous system. Toxicol. In Vitro 2015, 9, 1042–1052. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Liu, X.; Zheng, X.; Ding, C. In vivo evaluation of plasma-sprayed wollastonite coating. Biomaterials 2005, 26, 3455–3460. [Google Scholar] [CrossRef] [PubMed]
- Sadjadi, M.S.; Ebrahimi, H.R.; Meskinfam, M.; Zare, K. Silica enhanced formation of hydroxyapatite nanocrystals in simulated body fluid (SBF) at 37 °C. Mater. Chem. Phys. 2011, 130, 67–71. [Google Scholar] [CrossRef]
- Qian, S.; Liu, X. Cytocompatibility of Si-incorporated TiO2 nanopores films. Colloids Surf. B Biointerfaces 2015, 133, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Qiao, Y.; Liu, F.H.X.; Ding, C. Enhanced apatite-forming ability and cytocompatibility of porous and nanostructured TiO2/CaSiO3 coating on titanium, Colloids Surf. B Biointerfaces 2013, 101, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Cao, H.; Zhang, W.; Ding, X.; Yang, G.; Qiao, X.; Liu, X.; Jiang, X. Enhanced osseointegration of hierarchical micro/nanotopographic titanium fabricated by microarc oxidation and electrochemical treatment. ACS Appl. Mater. Interfaces 2016, 8, 3840–3852. [Google Scholar] [CrossRef] [PubMed]
- Zuldesmi, M.; Waki, A.; Kuroda, K.; Okido, M. Hydrothermal treatment of titanium alloys for the enhancement of osteoconductivity. Mater. Sci. Eng. C 2015, 49, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Müller, W.E.G.; Wang, X.; Lilja, L.; Shen, Z. Porous titania surfaces on titanium with hierarchical macro- and mesoporosities for enhancing cell adhesion, proliferation and mineralization. Mater. Sci. Eng. C 2015, 47, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Ueno, T.; Tsukimura, N.; Yamada, M.; Ogawa, T. Enhanced bone-integration capability of alkali- and heat-treated nanopolymorphic titanium in micro-to-nanoscale hierarchy. Biomaterials 2011, 32, 7297–7308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, G.; Liu, Y.; Zhao, X.; Zou, D.; Zhu, C.; Jin, Y.; Huang, Q.; Sun, J.; Liu, X.; et al. The synergistic effect of hierarchical micro/nano-topography and bioactive ions for enhanced osseointegration. Biomaterials 2013, 34, 3184–3195. [Google Scholar] [CrossRef] [PubMed]
- Kazek-Kęsik, A.; Leśniak, K.; Zhidkov, I.S.; Korotin, D.M.; Kukharenko, A.I.; Cholakh, S.O.; Kalemba-Rec, I.; Suchanek, K.; Kurmaev, E.Z.; Simka, W. Influence of alkali treatment on anodized titanium alloys in wollastonite suspension. Metals 2017, 7, 322. [Google Scholar] [CrossRef]
- Kokubo, T. Apatite formation on surfaces of ceramics, metals and polymers in body environment. Acta Mater. 1998, 46, 2519–2527. [Google Scholar] [CrossRef]
- Kazek-Kęsik, A.; Krok-Borkowicz, M.; Dercz, G.; Donesz-Sikorska, A.; Pamuła, E.; Simka, W. Multilayer coatings formed on titanium alloy surfaces by plasma electrolytic oxidation-electrophoretic deposition methods. Electrochim. Acta 2016, 204, 294–306. [Google Scholar] [CrossRef]
- Wei, D.; Zhou, Y.; Jia, D.; Wang, Y. Chemical treatment of TiO2-based coatings formed by plasma electrolytic oxidation in electrolyte containing nano-HA, calcium salts and phosphates for biomedical applications. Appl. Surf. Sci. 2008, 254, 1775–1782. [Google Scholar] [CrossRef]
- Yang, Z.; Xia, L.; Li, W.; Han, J. Effect of Ca/P ratio on bioactivity of PEO coatings. Adv. Biomed. Eng. Technol. 2015, 2, 13–19. [Google Scholar] [CrossRef]
- Yan, Y.; Han, H.; Lu, C.G. The effect of chemical treatment on apatite-forming ability of the macroporous zirconia films formed by micro-arc oxidation. Appl. Surf. Sci. 2008, 254, 833–4839. [Google Scholar] [CrossRef]
- Pan, Y.K.; Chen, C.Z.; Wang, D.G.; Lin, Z.G. Preparation and bioactivity of micro-arc oxidized calcium phosphate coatings. Mater. Chem. Phys. 2013, 141, 842–849. [Google Scholar] [CrossRef]
- Robin, M.; Almeida, C.; Azaïs, T.; Haye, B.; Illoul, C.; Lesieur, J.; Giraud-Guille, M.N.; Nassif, N.; Hélary, C. Involvement of 3D osteoblast migration and bone apatite during in vitro early osteocytogenesis. Bone 2016, 88, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Addison, W.N.; Nelea, V.; Chicatun, F.; Chien, Y.C.; Tran-Khanh, N.; Buschmann, M.D.; Nazhat, S.N.; Kaartinen, M.T.; Vali, H.; Tecklenburg, M.M.; et al. Extracellular matrix mineralization in murine MC3T3-E1 osteoblast cultures: An ultrastructural, compositional and comparative analysis with mouse bone. Bone 2015, 71, 244–256. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Tolba, E.; Schröder, H.C.; Muñoz-Espí, R.; Diehl-Seifert, B.; Wang, X. Amorphous polyphosphate-hydroxyapatite: A morphogenetically active substrate for bone-related SaOS-2 cells in vitro. Acta Biomater. 2016, 31, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Olszta, M.J.; Cheng, X.; Jee, S.S.; Kumar, R.; Kim, Y.Y.; Kaufman, M.J.; Douglas, E.P.; Gower, L.B. Bone structure and formation: A new perspective. Mater. Sci. Eng. R 2007, 58, 77–116. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazek-Kęsik, A.; Leśniak, K.; Orzechowska, B.U.; Drab, M.; Wiśniewska, A.; Simka, W. Alkali Treatment of Anodized Titanium Alloys Affects Cytocompatibility. Metals 2018, 8, 29. https://doi.org/10.3390/met8010029
Kazek-Kęsik A, Leśniak K, Orzechowska BU, Drab M, Wiśniewska A, Simka W. Alkali Treatment of Anodized Titanium Alloys Affects Cytocompatibility. Metals. 2018; 8(1):29. https://doi.org/10.3390/met8010029
Chicago/Turabian StyleKazek-Kęsik, Alicja, Katarzyna Leśniak, Beata Urszula Orzechowska, Marek Drab, Agnieszka Wiśniewska, and Wojciech Simka. 2018. "Alkali Treatment of Anodized Titanium Alloys Affects Cytocompatibility" Metals 8, no. 1: 29. https://doi.org/10.3390/met8010029
APA StyleKazek-Kęsik, A., Leśniak, K., Orzechowska, B. U., Drab, M., Wiśniewska, A., & Simka, W. (2018). Alkali Treatment of Anodized Titanium Alloys Affects Cytocompatibility. Metals, 8(1), 29. https://doi.org/10.3390/met8010029