Microstructural Characteristics and Mechanical Properties of 2205/AZ31B Laminates Fabricated by Explosive Welding
Abstract
:1. Introduction
2. Experimental Procedures
2.1. Materials
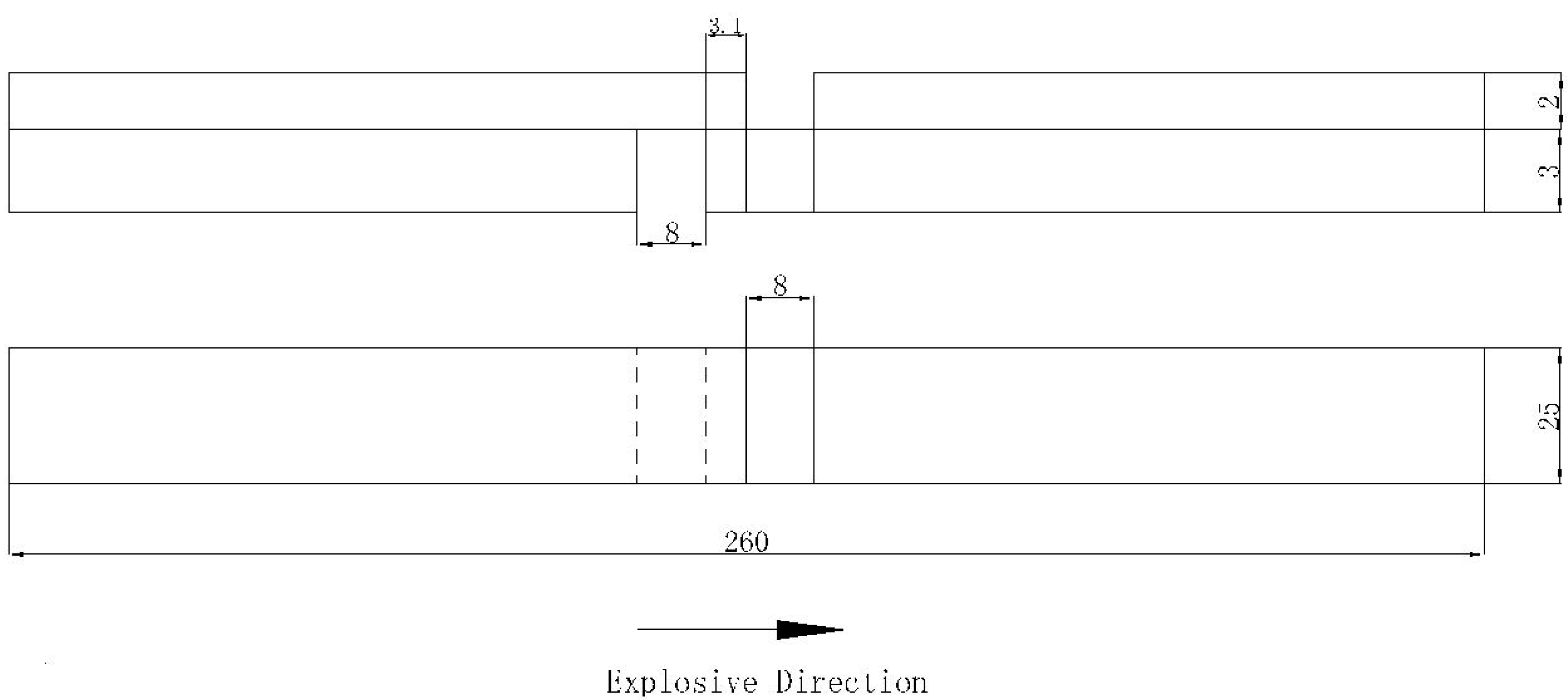
2.2. Explosive Cladding Process
2.3. Microstructural Work
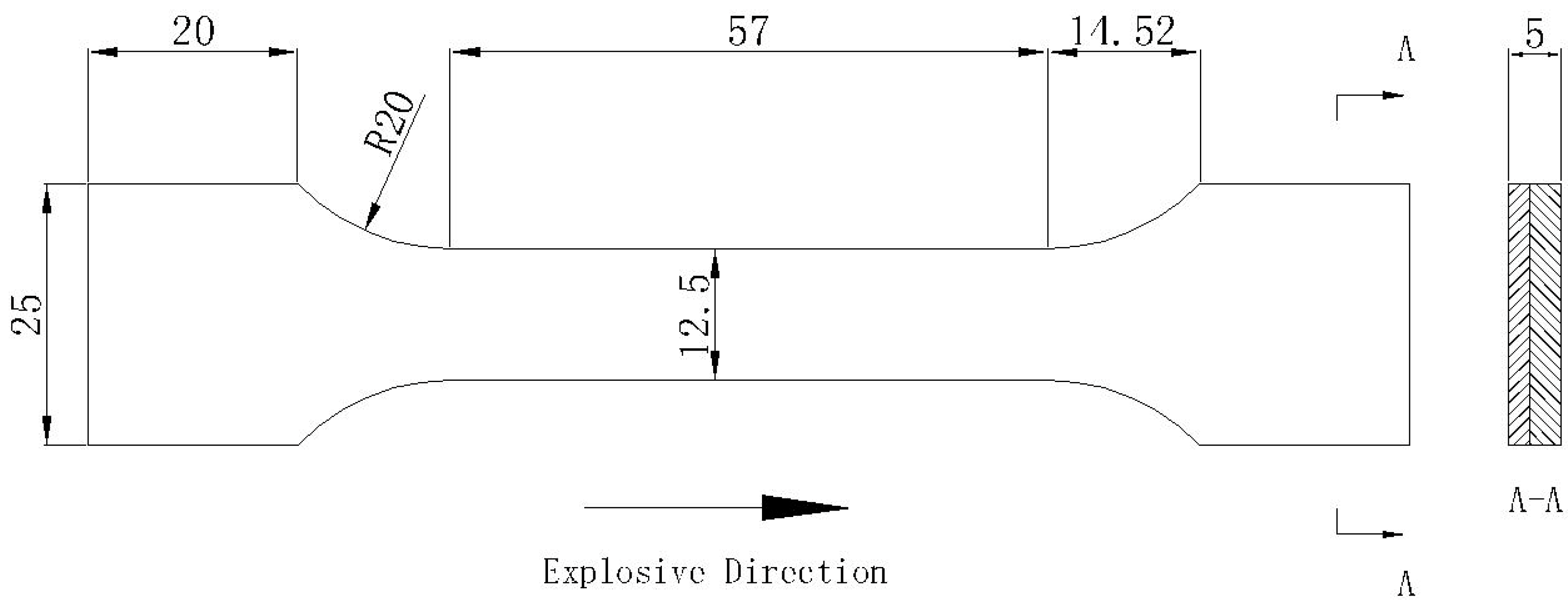
2.4. Mechanical Test
3. Results and Discussion
3.1. Macro Morphology of Composite Plate
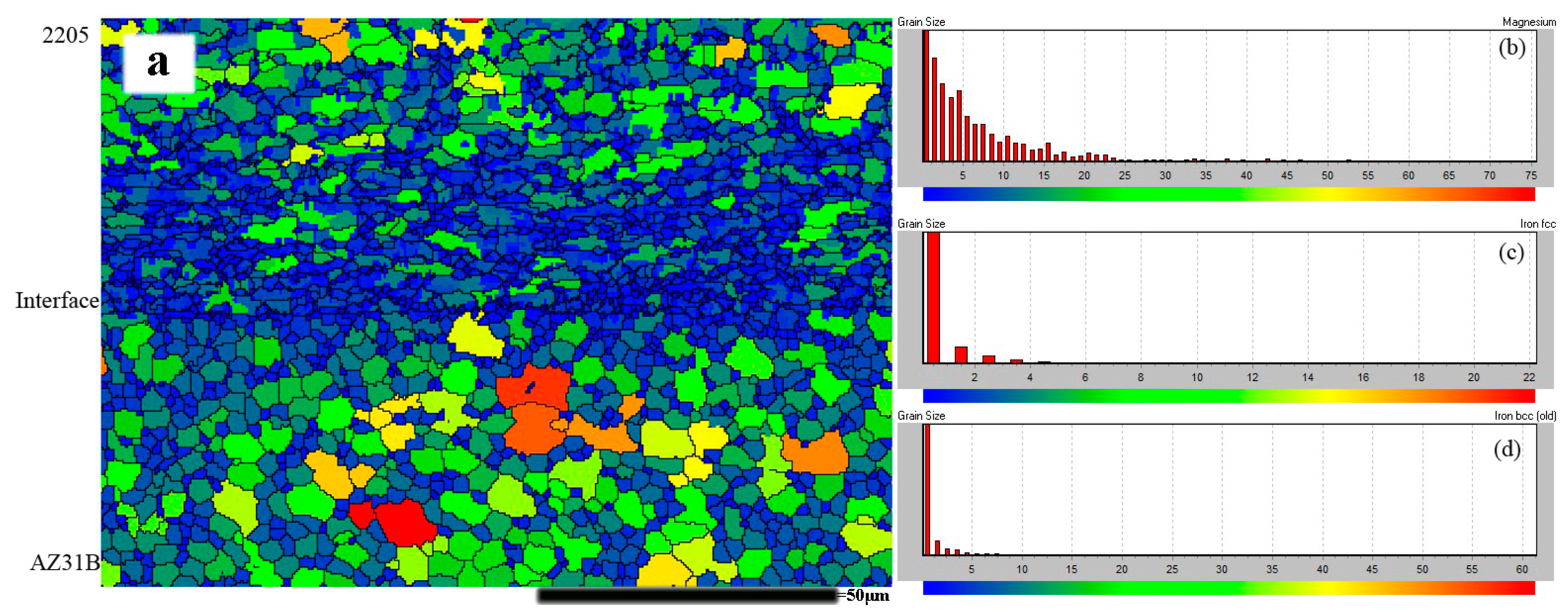
3.2. Morphology near the Interface
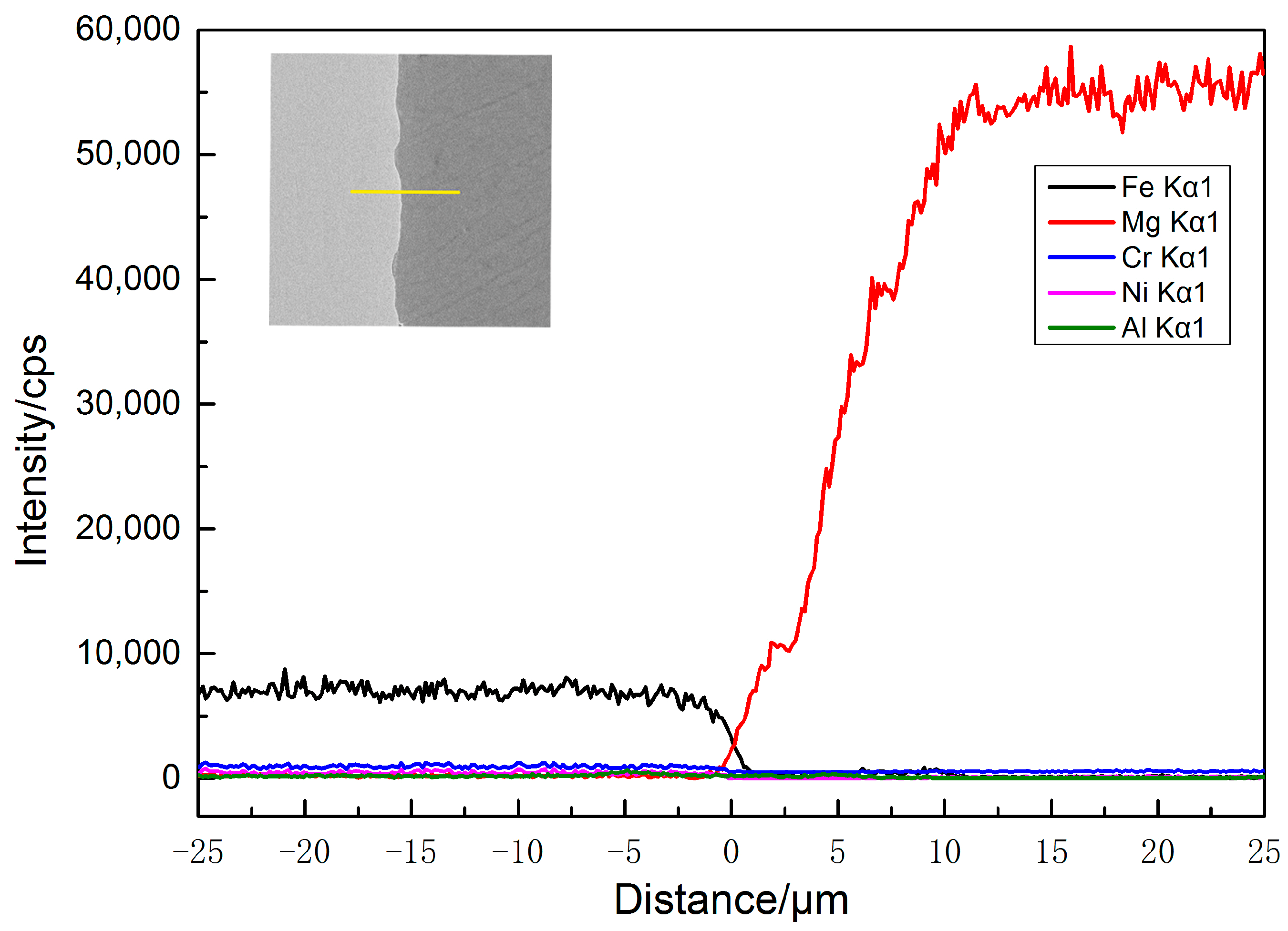
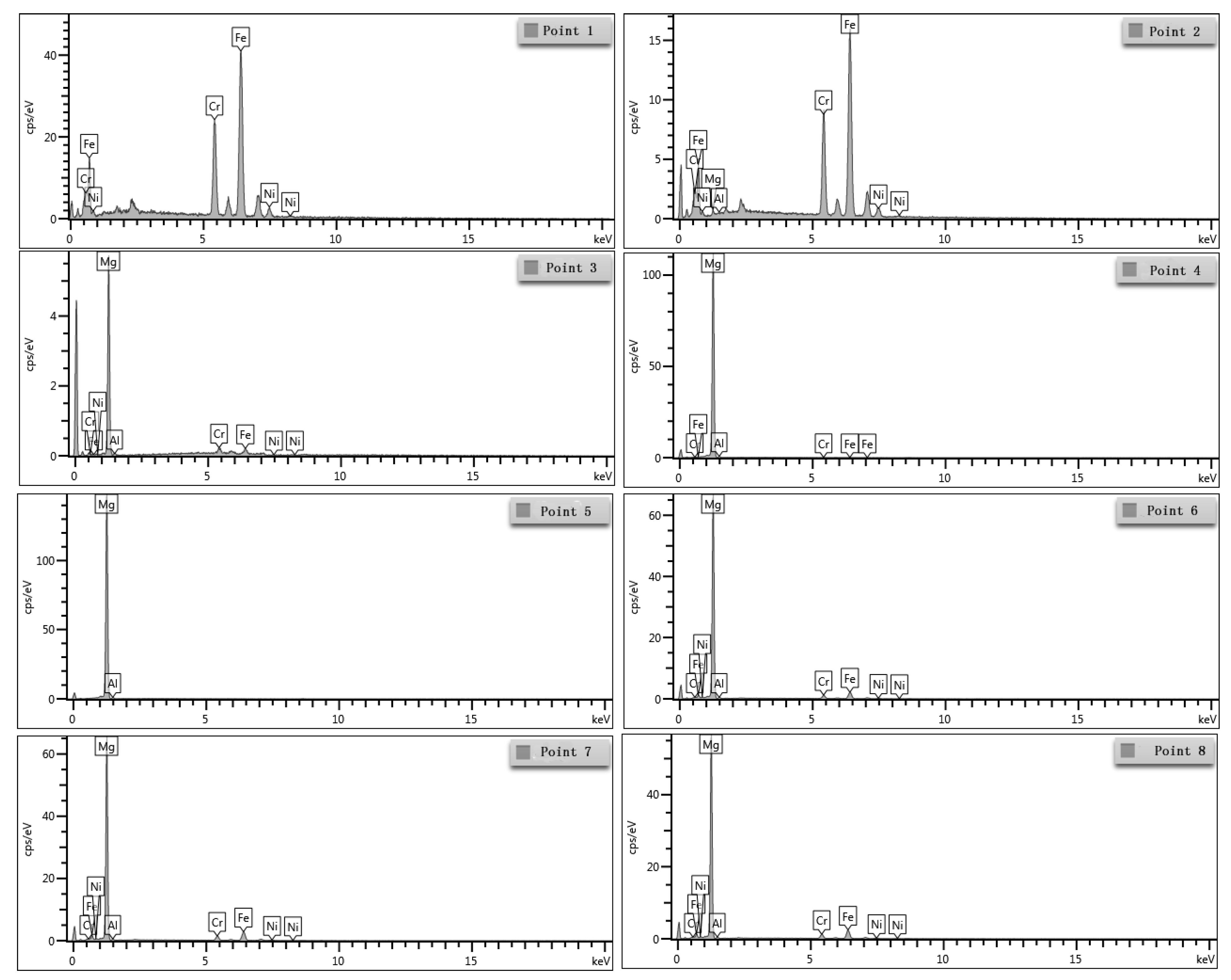
3.3. SEM Morphology and EDS Analyses near the Interface
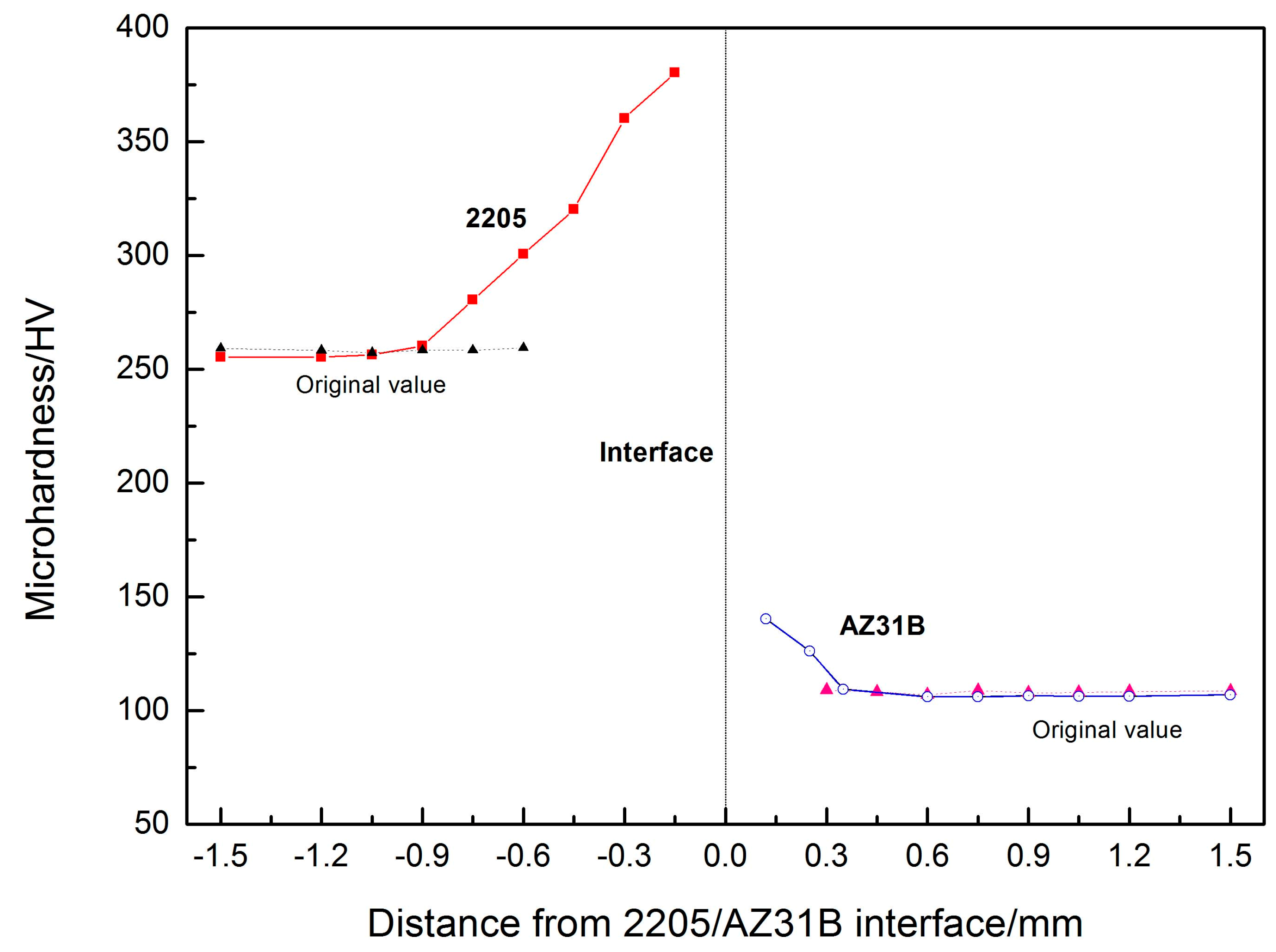
3.4. Microhardness Measurements
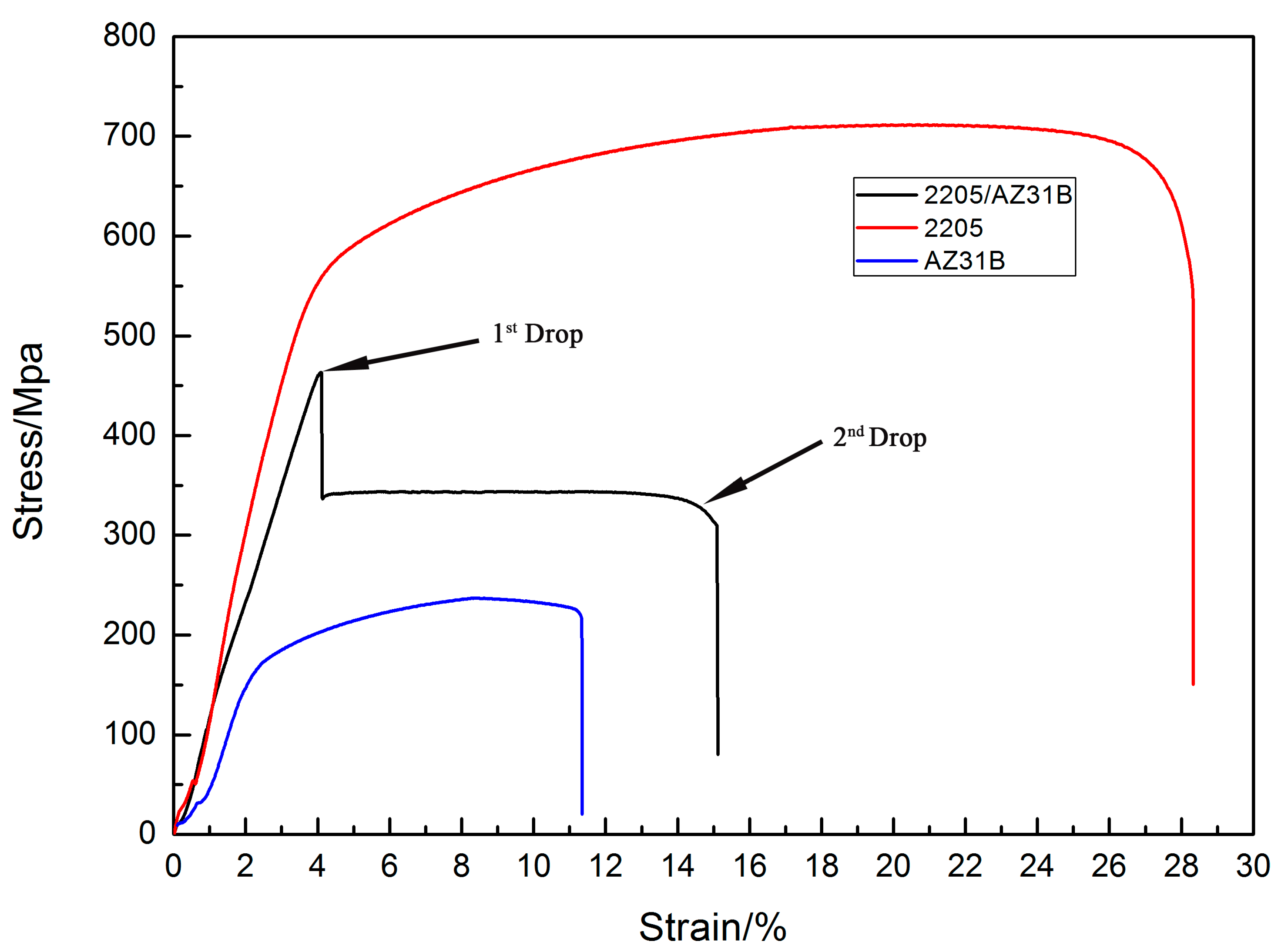
3.5. Tensile Strength Test Outcome
3.6. Tensile Shear Test
4. Conclusions
- (1)
- Explosive welding is a feasible technique to manufacture 2205/AZ31B composite plates.
- (2)
- The interface of 2205/AZ31B bimetallic composite has a less regular wavy morphology, with locally melted pockets. Adiabatic shear bands occurred only in the AZ31B side near explosive welding interface. The microstructure observed with EBSD showed a strong refinement near the interface zones.
- (3)
- Elemental mutual diffusion results in metallurgical bonding between 2205 duplex stainless steel and AZ31B magnesium alloy.
- (4)
- The microhardness value near the interface was higher than the original value, attributed to the violent plastic deformation and grain refinement at the collision interface.
- (5)
- The tensile shear strength of the bonding interface of 2205/AZ31B composite was 105.63MPa. Tensile strength of 2205/AZ31B composite material was higher than the base AZ31B.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Findik, F. Recent developments in explosive welding. Mater. Des. 2011, 32, 1081–1093. [Google Scholar] [CrossRef]
- Zhang, L.J.; Pei, Q.; Zhang, J.X.; Bi, Z.Y.; Li, P.C. Study on the microstructure and mechanical properties of explosive welded 2205/X65 bimetallic sheet. Mater. Des. 2014, 64, 462–476. [Google Scholar] [CrossRef]
- Mousavi, S.A.A.A.; Sartangi, P.F. Experimental investigation of explosive welding of cp-titanium/AISI 304 stainless steel. Mater. Des. 2009, 30, 459–468. [Google Scholar] [CrossRef]
- Fronczek, D.M.; Wojewoda-Budka, J.; Chulist, R.; Sypien, A.; Korneva, A.; Szulc, Z.; Schell, N.; Zieba, P. Structural properties of Ti/Al clads manufactured by explosive welding and annealing. Mater. Des. 2016, 91, 80–89. [Google Scholar] [CrossRef]
- Luo, A.A. Magnesium: Current and potential automotive applications. JOM 2002, 54, 42–48. [Google Scholar] [CrossRef]
- Li, D.; Chen, F.; Xie, Z.H.; Shan, S.; Zhong, C.J. Enhancing structure integrity and corrosion resistance of Mg alloy by a two-step deposition to avoid F etching to nano-SiO2, reinforcement. J. Alloys Compd. 2017, 705, 70–78. [Google Scholar] [CrossRef]
- Kaya, Y.; Kahraman, N. An investigation into the explosive welding/cladding of Grade A ship steel/AISI 316L austenitic stainless steel. Mater. Des. 2013, 52, 367–372. [Google Scholar] [CrossRef]
- Lee, K.S.; Kim, J.S.; Jo, Y.M.; Lee, S.E.; Heo, J.; Chang, Y.W.; Lee, Y.S. Interface-correlated deformation behavior of a stainless steel-Al–Mg 3-ply composite. Mater. Charact. 2013, 75, 138–149. [Google Scholar] [CrossRef]
- Guo, Q.; Guo, X.; Wang, F.; Wang, Y.; Du, K. Micro-Arc Oxidized Corrosion Resistant Interlayer on Magnesium Alloy with Electromagnetic Wave Absorption Coatings. ECS Electrochem. Lett. 2012, 1, C13–C16. [Google Scholar] [CrossRef]
- Mukai, T.; Mohri, T.; Mabuchi, M.; Nakamura, M.; Ishikawa, K.; Higashi, K. Experimental study of a structural magnesium alloy with high absorption energy under dynamic loading. Scr. Mater. 1998, 39, 1249–1253. [Google Scholar] [CrossRef]
- Darwin, J.D.; Lal, D.M.; Nagarajan, G. Optimization of cryogenic treatment to maximize the wear resistance of 18% Cr martensitic stainless steel by Taguchi method. J. Mater. Process. Technol. 2008, 195, 241–247. [Google Scholar] [CrossRef]
- Nayeb-Hashemi, A.A.; Clark, J.B.; Swartzendruber, L.J. The Fe–Mg (Iron-Magnesium) system. J. Phase Equilib. 1985, 6, 235–238. [Google Scholar] [CrossRef]
- Elthalabawy, W.; Khan, T. Liquid Phase Bonding of 316L Stainless Steel to AZ31 Magnesium Alloy. J. Mater. Sci. Technol. 2011, 27, 22–28. [Google Scholar] [CrossRef]
- Bikulčius, G.; Miečinskas, P.; Ručinskienė, A.; Grigucevičienė, A.; Selskis, A.; Pakštas, V. Improvement of corrosion resistance of magnesium alloy by sputter coating with stainless steel. Trans. Inst. Met. Finish. 2012, 90, 125–128. [Google Scholar] [CrossRef]
- Yuan, X.J.; Sheng, G.M.; Luo, J.; Jia, L.I. Microstructural characteristics of joint region during diffusion-brazing of magnesium alloy and stainless steel using pure copper interlayer. Trans. Nonferr. Met. Soc. China 2013, 23, 599–604. [Google Scholar] [CrossRef]
- Miao, Y.; Han, D.; Yao, J.; Li, F. Effect of laser offsets on joint performance of laser penetration brazing for magnesium alloy and steel. Mater. Des. 2010, 31, 3121–3126. [Google Scholar] [CrossRef]
- Wei, Y.; Li, J.; Xiong, J.; Huang, F.; Zhang, F. Microstructures and mechanical properties of magnesium alloy and stainless steel weld-joint made by friction stir lap welding. Mater. Des. 2012, 33, 111–114. [Google Scholar] [CrossRef]
- Zhu, B.; Liang, W.; Li, X. Interfacial microstructure, bonding strength and fracture of magnesium–aluminum laminated composite plates fabricated by direct hot pressing. Mater. Sci. Eng. A 2011, 528, 6584–6588. [Google Scholar] [CrossRef]
- Yan, Y.B.; Zhang, Z.W.; Shen, W.; Wang, J.H.; Zhang, L.K.; Chin, B.A. Microstructure and properties of magnesium AZ31B–aluminum 7075 explosively welded composite plate. Mater. Sci. Eng. A 2010, 527, 2241–2245. [Google Scholar] [CrossRef]
- Acarer, M.; Gülenç, B.; Findik, F. The influence of some factors on steel/steel bonding quality on there characteristics of explosive welding joints. J. Mater. Sci. 2004, 39, 6457–6466. [Google Scholar] [CrossRef]
- Zheng, Y. Explosive Welding and Metallic Composite and Their Engineering Application; Central South University Press: Changsha, China, 2002. [Google Scholar]
- Mastanaiah, P.; Reddy, G.M.; Prasad, K.S.; Murthy, C.V.S. An investigation on microstructures and mechanical properties of explosive cladded C103 niobium alloy over C263 nimonic alloy. J. Mater. Process. Technol. 2014, 214, 2316–2324. [Google Scholar] [CrossRef]
- Lysak, V.I.; Kuzmin, S.V. Lower boundary in metal explosive welding. Evolution of ideas. J. Mater. Process. Technol. 2012, 212, 150–156. [Google Scholar] [CrossRef]
- Durgutlu, A.; Gülenç, B.; Findik, F. Examination of copper/stainless steel joints formed by explosive welding. Mater. Des. 2005, 26, 497–507. [Google Scholar] [CrossRef]
- Follansbee, P.S.; Murr, L.E.; Staudhammer, K.P.; Meyers, M.A. Metallurgical Applications of Shock Wave and High Strain Rate Phenomena; Marcel Dekker, Inc.: New York, NY, USA, 1986; p. 451. [Google Scholar]
- Yang, Y.; Xinming, Z.; Zhenghua, L.; Qingyun, L. Adiabatic shear band on the titanium side in the Ti/mild steel explosive cladding interface. Acta Mater. 1996, 44, 561–565. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, B.F.; Hu, B.; Hu, K.; Li, Z.G. The collective behavior and spacing of adiabatic shear bands in the explosive cladding plate interface. Mater. Sci. Eng. A 2005, 398, 291–296. [Google Scholar] [CrossRef]
- Zhang, N.; Wang, W.; Cao, X.; Wu, J. The effect of annealing on the interface microstructure and mechanical characteristics of AZ31B/AA6061 composite plates fabricated by explosive welding. Mater. Des. 2015, 65, 1100–1109. [Google Scholar] [CrossRef]
- Xia, H.B.; Wang, S.G.; Ben, H.F. Microstructure and mechanical properties of Ti/Al explosive cladding. Mater. Des. 2014, 56, 1014–1019. [Google Scholar] [CrossRef]
- Loureiro, A.; Mendes, R.; Ribeiro, J.B.; Leal, R.M.; Galvão, I. Effect of explosive mixture on quality of explosive welds of copper to aluminium. Mater. Des. 2016, 95, 256–267. [Google Scholar] [CrossRef]
- Grignon, F.; Benson, D.; Vecchio, K.S.; Meyers, M.A. Explosive Welding of Aluminum to Aluminum: Analysis, Computations and Experiments; American Institute of Physics: Melville, NY, USA, 2004; pp. 1333–1351. [Google Scholar]
- Song, J.; Kostka, A.; Veehmayer, M.; Raabe, D. Hierarchical microstructure of explosive joints: Example of titanium to steel cladding. Mater. Sci. Eng. A 2011, 528, 2641–2647. [Google Scholar] [CrossRef]
- Xu, W.; Chen, D.L.; Liu, L.; Mori, H.; Zhou, Y. Microstructure and mechanical properties of weld-bonded and resistance spot welded magnesium-to-steel dissimilar joints. Mater. Sci. Eng. A 2012, 537, 11–24. [Google Scholar] [CrossRef]
- Elthalabawy, W.M.; Khan, T.I. Microstructural development of diffusion-brazed austenitic stainless steel to magnesium alloy using a nickel interlayer. Mater. Charact. 2010, 61, 703–712. [Google Scholar] [CrossRef]
- Manikandan, P.; Lee, J.O.; Mizumachi, K.; Mori, A.; Raghukandan, K.; Hokamoto, K. Underwater explosive welding of thin tungsten foils and copper. J. Nucl. Mater. 2011, 418, 281–285. [Google Scholar] [CrossRef]
- Honarpisheh, M.; Asemabadi, M.; Sedighi, M. Investigation of annealing treatment on the interfacial properties of explosive-welded Al/Cu/Al multilayer. Mater. Des. 2012, 37, 122–127. [Google Scholar] [CrossRef]
- Kim, I.K.; Hong, S.I. Roll-Bonded Tri-Layered Mg/Al/Stainless Steel Clad Composites and their Deformation and Fracture Behavior. Metall. Mater. Trans. A 2013, 44, 3890–3900. [Google Scholar] [CrossRef]
- Mousavi, S.A.A.A.; Al-Hassani, S.T.S. Finite element simulation of explosively-driven plate impact with application to explosive welding. Mater. Des. 2008, 29, 1–19. [Google Scholar] [CrossRef]









| Element | C | Si | Cr | Mn | Ni | Mo | P | S | Fe |
|---|---|---|---|---|---|---|---|---|---|
| Wt. % | 0.021 | 0.56 | 22.59 | 1.13 | 5.29 | 3.45 | 0.018 | 0.001 | Balanced |
| Element | Mn | Zn | Si | Fe | Al | Mg |
|---|---|---|---|---|---|---|
| Wt. % | 0.63 | 1.10 | 0.10 | 0.05 | 3.02 | Balanced |
| Point | Fe | Cr | Ni | Mg | Al |
|---|---|---|---|---|---|
| 1 | 69.80 | 23.80 | 6.40 | - | - |
| 2 | 70.13 | 23.74 | 5.23 | 0.84 | 0.06 |
| 3 | 10.72 | 6.61 | 0.61 | 81.54 | 0.52 |
| 4 | 0.18 | 0.04 | 0.13 | 98.09 | 1.56 |
| 5 | - | - | - | 97.58 | 2.42 |
| 6 | 15.68 | 5.00 | 1.06 | 76.94 | 1.32 |
| 7 | 12.88 | 4.41 | 1.29 | 80.21 | 1.21 |
| 8 | 15.05 | 5.29 | 1.16 | 77.30 | 1.20 |
| Material | Yield Strength (Mpa) | Ultimate Tensile Strength (Mpa) | Elongation (%) |
|---|---|---|---|
| 2205 base metal | 560.6 | 716.6 | 28.3 |
| AZ31B base metal | 160.75 | 237.9 | 11.6 |
| 2205/AZ31B composite | - | 486.6 | 15.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Wu, Z. Microstructural Characteristics and Mechanical Properties of 2205/AZ31B Laminates Fabricated by Explosive Welding. Metals 2017, 7, 125. https://doi.org/10.3390/met7040125
Li Y, Wu Z. Microstructural Characteristics and Mechanical Properties of 2205/AZ31B Laminates Fabricated by Explosive Welding. Metals. 2017; 7(4):125. https://doi.org/10.3390/met7040125
Chicago/Turabian StyleLi, Yan, and Zhisheng Wu. 2017. "Microstructural Characteristics and Mechanical Properties of 2205/AZ31B Laminates Fabricated by Explosive Welding" Metals 7, no. 4: 125. https://doi.org/10.3390/met7040125
APA StyleLi, Y., & Wu, Z. (2017). Microstructural Characteristics and Mechanical Properties of 2205/AZ31B Laminates Fabricated by Explosive Welding. Metals, 7(4), 125. https://doi.org/10.3390/met7040125





