Influence of Annealing on the Microstructures and Oxidation Behaviors of Al8(CoCrFeNi)92, Al15(CoCrFeNi)85, and Al30(CoCrFeNi)70 High-Entropy Alloys
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
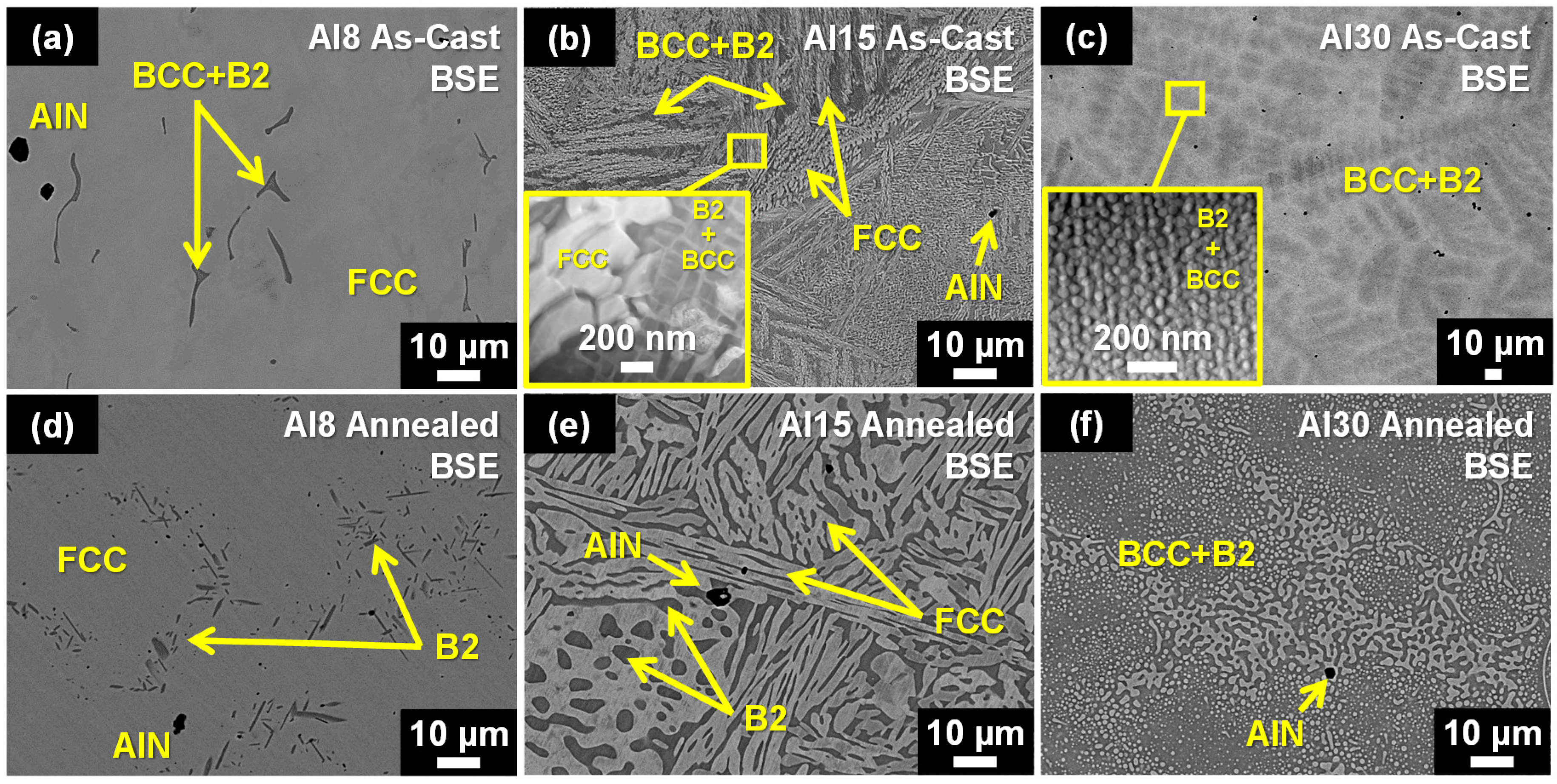
3.1. Microstructures of the As-Cast and Annealed HEAs
3.2. Oxidized Microstructures
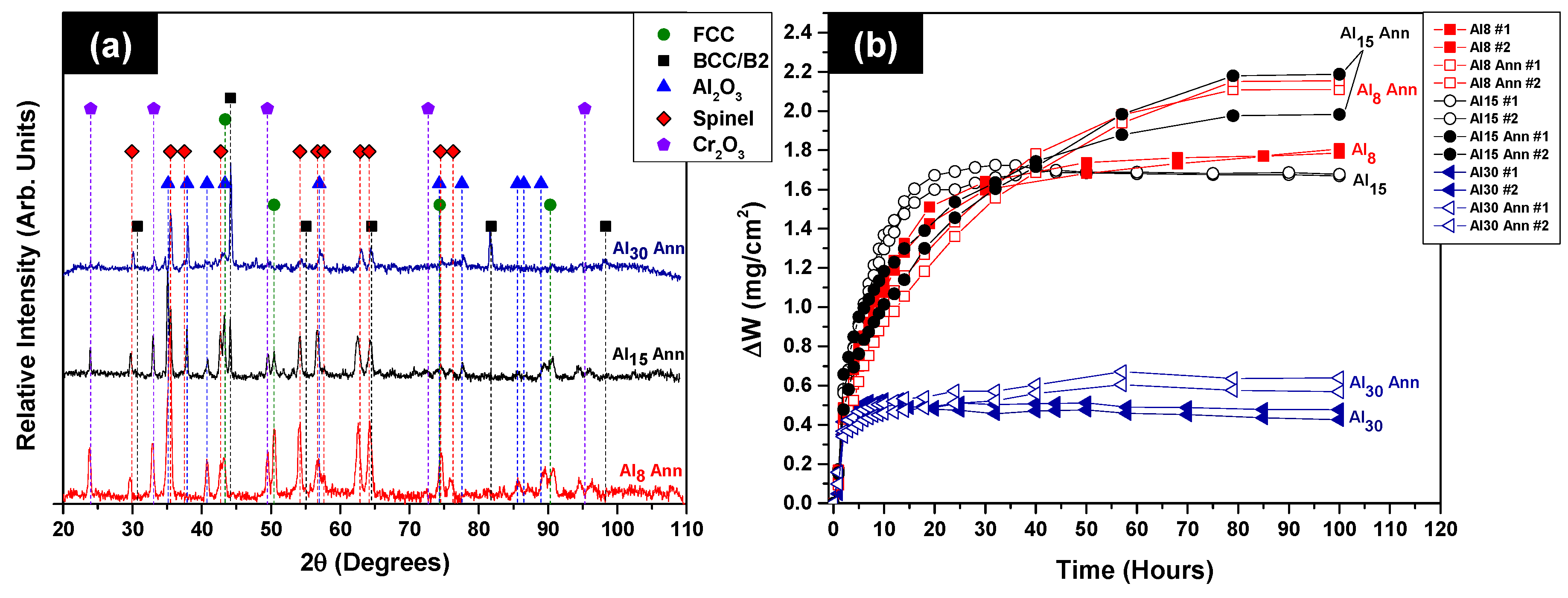
3.3. Oxidation Behavior
4. Discussion
5. Conclusions
- (1)
- The as-cast Al8 and Al15 HEAs consisted of a combination of FCC, BCC, and B2 solid solution phases. Annealing at 1050 °C for 120 h resulted in significant coarsening of the FCC and B2 phases and dissolution of the primary BCC phase. The as-cast Al30 HEA consisted of a uniform distribution of sub-micron sized BCC precipitates with spherical morphologies dispersed in a B2 matrix. Annealing of the Al30 HEA also caused substantial microstructural coarsening and the formation of additional nano-scale B2 precipitates inside the BCC phase. In all cases, annealing increased the phase fraction of the B2 phase at the expense of the FCC and/or BCC phases.
- (2)
- Discontinuous, isothermal oxidation tests at 1050 °C showed the as-cast HEAs to oxidize in the same way as model Group II and Group III Ni–Cr–Al alloys, with the initial formation of transient oxides and external Cr2O3, followed by the development of an internal Al2O3 scale. As expected, increased Al concentrations led to enhanced oxidation resistances. In contrast, the annealed HEAs tended to form external Al2O3 scales during oxidation with or without underlying spinel phases depending upon Al content. The change in oxidation mechanism was attributed to the increased phase fraction of Ni+Al-rich B2 phase. It is suggested that the enhanced distribution of B2 phase near the alloy free surfaces promoted the formation of Al2O3 and thus modifying the subsequent stages of oxidation.
- (3)
- The parabolic oxidation behaviors of all of the HEAs in both the as-cast and annealed states indicate various levels of protection. The calculated parabolic oxide growth rate constants (kP) for all of the HEAs correspond to those expected for Group II and Group III Ni–Cr–Al alloys. Although annealing had a large impact on the post oxidation microstructures, it seemed to have lesser influence on the parabolic growth rate constants.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miracle, D.B.; Miller, J.D.; Senkov, O.N.; Woodward, C.; Uchic, M.D.; Tiley, J. Exploration and development of high entropy alloys for structural applications. Entropy 2014, 16, 494–525. [Google Scholar] [CrossRef]
- Yeh, J.-W. Alloy design strategies and future trends in high-entropy alloys. JOM 2013, 65, 1759–1771. [Google Scholar] [CrossRef]
- Yeh, J.W.; Chen, Y.L.; Lin, S.J.; Chen, S.K. High-entropy alloys—A new era of exploitation. Mater. Sci. Forum 2007, 560, 1–9. [Google Scholar] [CrossRef]
- Zhang, Y.; Zuo, T.T.; Tang, Z.; Gao, M.C.; Dahmen, K.A.; Liaw, P.K.; Lu, Z.P. Microstructures and properties of high-entropy alloys. Prog. Mater. Sci. 2014, 61, 1–93. [Google Scholar] [CrossRef]
- Tang, Z.; Huang, L.; He, W.; Liaw, P.K. Alloying and processing effects on the aqueous corrosion behavior of high-entropy alloys. Entropy 2014, 16, 895–911. [Google Scholar] [CrossRef]
- Butler, T.M.; Alfano, J.P.; Martens, R.L.; Weaver, M.L. High-temperature oxidation behavior of Al-Co-Cr-Ni-(Fe or Si) multicomponent high-entropy alloys. JOM 2015, 67, 246–259. [Google Scholar] [CrossRef]
- Butler, T.M.; Weaver, M.L. Oxidation behavior of arc melted AlCoCrFeNi multi-component high entropy alloys. J. Alloys Compd. 2016, 674, 229–244. [Google Scholar] [CrossRef]
- Chang, S.-Y.; Li, C.-E.; Huang, Y.-C.; Hsu, H.-F.; Yeh, J.-W.; Lin, S.-J. Structural and thermodynamic factors of suppressed interdiffusion kinetics in multi-component high-entropy materials. Sci. Rep. 2014, 4, 4162. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.Y.; Tsai, M.H.; Yeh, J.W. Sluggish diffusion in Co–Cr–Fe–Mn–Ni high-entropy alloys. Acta Mater. 2013, 61, 4887–4897. [Google Scholar] [CrossRef]
- Dąbrowa, J.; Kucza, W.; Cieślak, G.; Kulik, T.; Danielewski, M.; Yeh, J.-W. Interdiffusion in the fcc-structured Al-Co-Cr-Fe-Ni high entropy alloys: Experimental studies and numerical simulations. J. Alloys Compd. 2016, 674, 455–462. [Google Scholar] [CrossRef]
- Zhang, H.; He, Y.-Z.; Pan, Y.; Guo, S. Thermally stable laser cladded CoCrCuFeNi high-entropy alloy coating with low stacking fault energy. J. Alloys Compd. 2014, 600, 210–214. [Google Scholar] [CrossRef]
- Schuh, B.; Martin, F.M.; Volker, B.; George, E.P.; Clemens, H.; Pippan, R.; Hohenwarter, A. Mechanical properties, microstructure and thermal stability of a nanocrystalline CoCrFeMnNi high-entropy alloy after severe plastic deformation. Acta Mater. 2015, 96, 258–268. [Google Scholar] [CrossRef]
- Praveen, S.; Basu, J.; Kashyap, S.; Kottada, R.S. Exceptional resistance to grain growth in nanocrystalline CoCrFeNi high entropy alloy at high homologous temperatures. J. Alloys Compd. 2016, 662, 361–367. [Google Scholar] [CrossRef]
- Liu, Y.-X.; Cheng, C.-Q.; Shang, J.-L.; Wang, R.; Li, P.; Zhao, J. Oxidation behavior of high-entropy alloys AlxCoCrFeNi (x = 0.15, 0.4) in supercritical water and comparison with hr3c steel. Trans. Nonferr. Met. Soc. China 2015, 25, 1341–1351. [Google Scholar] [CrossRef]
- Butler, T.M.; Weaver, M.L. Investigation of the microstructures and oxidation behavior of AlNiCoCrFe high-entropy alloys. In Proceedings of the Contributed Papers from Materials Science & Technology (MS&T) 2015, Columbus, OH, USA, 4–8 October 2015; TMS: Columbus, OH, USA, 2015; pp. 1257–1264. [Google Scholar]
- Jiang, J.; Luo, X. High temperature oxidation behaviour of AlCuTiFeNiCr high-entropy alloy. Adv. Mater. Res. 2013, 652–654, 1115–1118. [Google Scholar] [CrossRef]
- Laplanche, G.; Volkert, U.F.; Eggeler, G.; George, E.P. Oxidation behavior of the CrMnFeCoNi high-entropy alloy. Oxid. Met. 2016, 85, 629–645. [Google Scholar] [CrossRef]
- Chang, Y.-J.; Yeh, A.-C. The evolution of microstructures and high temperature properties of AlxCo1.5CrFeNi1.5Tiy high entropy alloys. J. Alloys Compd. 2015, 653, 379–385. [Google Scholar] [CrossRef]
- Tsao, T.-K.; Yeh, A.-C.; Kuo, C.-M.; Murakami, H. High temperature oxidation and corrosion properties of high entropy superalloys. Entropy 2016, 18, 62. [Google Scholar] [CrossRef]
- Chen, S.-T.; Tang, W.-Y.; Kuo, Y.-F.; Chen, S.-Y.; Tsau, C.-H.; Shun, T.-T.; Yeh, J.-W. Microstructure and properties of age-hardenable AlxCrFe1.5MnNi0.5 alloys. Mater. Sci. Eng. A 2010, 527, 5818–5825. [Google Scholar] [CrossRef]
- Yang, H.H.; Tsai, W.T.; Kuo, J.C. Effect of pre-oxidation on increasing resistance of Fe-Al-Ni-Cr-Co-Mn high entropy alloys to molten al attack. Corros. Eng. Sci. Technol. 2014, 49, 124–129. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Q.T.; Tang, Q.H.; Dai, P.Q. High temperature oxidation property of Al0.5FeCoCrNi(Si0.2, Ti0.5) high entropy alloys. Corros. Prot. 2013, 34, 561–565. [Google Scholar]
- Daoud, H.M.; Manzoni, A.M.; Volkl, R.; Wanderka, N.; Glatzel, U. Oxidation behavior of Al8Co17Cr17Cu8Fe17Ni33, Al23Co15Cr23Cu8Fe15Ni15, and Al17Co17Cr17Cu17Fe17Ni17 compositionally complex alloys (high-entropy alloys) at elevated temperatures in air. Adv. Eng. Mater. 2015, 17, 1134–1141. [Google Scholar] [CrossRef]
- Holcomb, G.R.; Tylczak, J.; Carney, C. Oxidation of CoCrFeMnNi high entropy alloys. JOM 2015, 67, 2326–2339. [Google Scholar] [CrossRef]
- Kai, W.; Li, C.C.; Cheng, F.P.; Chu, K.P.; Huang, R.T.; Tsay, L.W.; Kai, J.J. The oxidation behavior of an equimolar FeCoNiCrMn high-entropy alloy at 950 °C in various oxygen-containing atmospheres. Corros. Sci. 2016, 108, 209–214. [Google Scholar] [CrossRef]
- Senkov, O.N.; Senkova, S.V.; Dimiduk, D.M.; Woodward, C.; Miracle, D.B. Oxidation behavior of a refractory NbCrMo0.5Ta0.5TiZr alloy. J. Mater. Sci. 2012, 47, 6522–6534. [Google Scholar] [CrossRef]
- Gorr, B.; Azim, M.; Christ, H.J.; Mueller, T.; Schliephake, D.; Heilmaier, M. Phase equilibria, microstructure, and high temperature oxidation resistance of novel refractory high-entropy alloys. J. Alloys Compd. 2015, 624, 270–278. [Google Scholar] [CrossRef]
- Gorr, B.; Mueller, F.; Christ, H.J.; Mueller, T.; Chen, H.; Kauffmann, A.; Heilmaier, M. High temperature oxidation behavior of an equimolar refractory metal-based alloy 20Nb-20Mo-20Cr-20Ti-20Al with and without Si addition. J. Alloys Compd. 2016, 688, 468–477. [Google Scholar] [CrossRef]
- Liu, C.M.; Wang, H.M.; Zhang, S.Q.; Tang, H.B.; Zhang, A.L. Microstructure and oxidation behavior of new refractory high entropy alloys. J. Alloys Compd. 2014, 583, 162–169. [Google Scholar] [CrossRef]
- Giggins, C.S.; Pettit, F.S. Oxidation of Ni-Cr-Al alloys between 1000 °C and 1200 °C. J. Electrochem. Soc. 1971, 118, 1782–1790. [Google Scholar] [CrossRef]
- Tang, Q.H.; Huang, Y.; Huang, Y.Y.; Liao, X.Z.; Langdon, T.G.; Dai, P.Q. Hardening of an Al0.3CoCrFeNi high entropy alloy via high-pressure torsion and thermal annealing. Mater. Lett. 2015, 151, 126–129. [Google Scholar] [CrossRef]
- Wang, W.-R.; Wang, W.-L.; Yeh, J.-W. Phases, microstructure and mechanical properties of AlxCoCrFeNi high-entropy alloys at elevated temperatures. J. Alloys Compd. 2014, 589, 143–152. [Google Scholar] [CrossRef]
- Manzoni, A.; Daoud, H.; Völkl, R.; Glatzel, U.; Wanderka, N. Phase separation in equiatomic AlCoCrFeNi high-entropy alloy. Ultramicroscopy 2013, 132, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, F.; Chen, S.; Cao, W. Computational thermodynamics aided high-entropy alloy design. JOM 2012, 64, 839–845. [Google Scholar] [CrossRef]
- Wang, W.-R.; Wang, W.-L.; Wang, S.-C.; Tsai, Y.-C.; Lai, C.-H.; Yeh, J.-W. Effects of al addition on the microstructure and mechanical property of AlxCoCrFeNi high-entropy alloys. Intermetallics 2012, 26, 44–51. [Google Scholar] [CrossRef]
- Huang, J.C. Evaluation of tribological behavior of Al-Co-Cr-Fe-Ni high entropy alloys using molecular dynamics simulation. Scanning 2012, 34, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.M.; Fu, H.M.; Zhang, H.F.; Wang, A.M.; Li, H.; Hu, Z.Q. Microstructure and compressive properties of multiprincipal component AlCoCrFeNiCux alloys. J. Alloys Compd. 2011, 509, 3476–3480. [Google Scholar] [CrossRef]
- Kao, Y.-F.; Chen, T.-J.; Chen, S.-K.; Yeh, J.-W. Microstructure and mechanical property of as-cast, -homogenized, and -deformed AlxCoCrFeNi (0 ≤ x ≤ 2) high-entropy alloys. J. Alloys Compd. 2009, 488, 57–64. [Google Scholar] [CrossRef]
- Wang, Y.P.; Li, B.S.; Ren, M.X.; Yang, C.; Fu, H.Z. Microstructure and compressive properties of AlCrFeCoNi high entropy alloy. Mater. Sci. Eng. A 2008, 491, 154–158. [Google Scholar] [CrossRef]
- Tang, Z.; Senkov, O.N.; Parish, C.M.; Zhang, C.; Zhang, F.; Santodonato, L.J.; Wang, G.; Zhao, G.; Yang, F.; Liaw, P.K. Tensile ductility of an AlCoCrFeNi multi-phase high-entropy alloy through hot isostatic pressing (hip) and homogenization. Mater. Sci. Eng. A 2015, 647, 229–240. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Zhang, Y.; Wang, Y.L.; Chen, G.L. Solid solution alloys of AlCoCrFeNiTix with excellent room-temperature mechanical properties. Appl. Phys. Lett. 2007, 90, 181904. [Google Scholar] [CrossRef]
- Li, D.; Zhang, Y. The ultrahigh charpy impact toughness of forged AlxCoCrFeNi high entropy alloys at room and cryogenic temperatures. Intermetallics 2016, 70, 24–28. [Google Scholar] [CrossRef]
- Munitz, A.; Salhov, S.; Hayun, S.; Frage, N. Heat treatment impacts the micro-structure and mechanical properties of AlCoCrFeNi high entropy alloy. J. Alloys Compd. 2016, 683, 221–230. [Google Scholar] [CrossRef]
- Saunders, N.; Miodownik, A.P. Calphad (Calculation of Phase Diagrams): A Comprehensive Guide; Pergamon Press: Oxford, UK, 1998; p. 479. [Google Scholar]
- Andersson, J.O.; Helander, T.; Hoglund, L.; Shi, P.; Sundman, B. Thermo-calc and dictra, computational tools for materials science. Calphad 2002, 26, 273–312. [Google Scholar] [CrossRef]
- Thermocalc, version 2015a; ThermoCalc Software AB: Stckholm, Sweden, 2015.
- Thermocalc Ni-Based Superalloys Database, version TCNI8; ThermoCalc Software AB: Stockholm, Sweden, 2015.
- Tomus, D.; Ng, H.P. In situ lift-out dedicated techniques using FIB–sem system for tem specimen preparation. Micron 2013, 44, 115–119. [Google Scholar] [CrossRef] [PubMed]
- German, R.M. Powder Metallurgy & Particular Materials Processing; Metal Powder Industries Federation: Princeton, NJ, USA, 2005; p. 528. [Google Scholar]
- Delaunay, D.; Huntz, A.M. Influence of structural parameters on oxidation of austenitic Fe-Ni-Cr-Al alloys. J. Mater. Sci. 1983, 18, 189–194. [Google Scholar] [CrossRef]
- Jang, C.; Kim, D.; Kim, D.; Sah, I.; Ryu, W.-S.; Yoo, Y.-S. Oxidation behaviors of wrought nickel-based superalloys in various high temperature environments. Trans. Nonferr. Met. Soc. China 2011, 21, 1524–1531. [Google Scholar] [CrossRef]
- Birks, N.; Meier, G.H.; Pettit, F.S. High-Temperature Oxidation of Metals, 2nd ed.; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]


| Alloy | Phase | Al | Ni | Co | Cr | Fe |
|---|---|---|---|---|---|---|
| Al8 | Target | 8.0 | 23.0 | 23.0 | 23.0 | 23.0 |
| As-cast Bulk | 8.2 ± 0.2 | 22.2 ± 0.5 | 22.8 ± 0.2 | 24.0 ± 0.3 | 22.8 ± 0.2 | |
| BCC (as-cast) ** | 17.1 ± 0.5 | 22.9 ± 0.8 | 16.8 ± 0.4 | 26.7 ± 1.1 | 16.5 ± 0.6 | |
| FCC (as-cast) ** | 10.4 ± 0.2 | 23.4 ± 0.1 | 20.8 ± 0.2 | 25.1 ± 0.1 | 20.3 ± 0.4 | |
| FCC (annealed) | 7.7 ± 0.2 | 21.9 ± 0.3 | 23.3 ± 0.1 | 24.2 ± 0.2 | 23.0 ± 0.3 | |
| B2 (as-cast) ** | 22.5 ± 0.1 | 28.6 ± 0.4 | 16.4 ± 0.1 | 18.6 ± 0.4 | 13.9 ± 0.4 | |
| B2 (annealed) | 25.2 ± 2.2 | 21.9 ± 0.3 | 16.9 ± 1.0 | 13.0 ± 2.3 | 14.3 ± 1.3 | |
| Al15 | Target | 15.0 | 21.25 | 21.25 | 21.25 | 21.25 |
| As-cast Bulk | 15.6 ± 0.1 | 20.5 ± 0.1 | 20.8 ± 0.1 | 21.9 ± 0.1 | 21.1 ± 0.1 | |
| BCC (as-cast) ** | 5.5 ± 1.3 | 21.3 ± 3.9 | 21.1 ± 0.3 | 30.8 ± 0.9 | 21.4 ± 2.0 | |
| FCC (as-cast) ** | 12.6 ± 0.7 | 21.4 ± 0.7 | 21.5 ± 0.7 | 21.8 ± 0.7 | 21.7 ± 0.1 | |
| FCC (annealed) | 6.9 ± 0.2 | 16.5 ± 0.2 | 22.9 ± 0.5 | 28.1 ± 0.4 | 25.6 ± 0.5 | |
| B2 (as-cast) ** | 19.7 ± 1.9 | 31.3 ± 3.6 | 21.5 ± 0.7 | 11.2 ± 3.6 | 16.5 ± 1.5 | |
| B2 (annealed) | 31.0 ± 0.6 | 29.4 ± 0.5 | 17.3 ± 0.4 | 10.0 ± 0.8 | 12.2 ± 0.6 | |
| Al30 | Target | 30 | 17.5 | 17.5 | 17.5 | 17.5 |
| As-cast Bulk | 29.2 ± 0.2 | 17.4 ± 0.1 | 17.6 ± 0.1 | 18.3 ± 0.1 | 17.6 ± 0.2 | |
| BCC (as-cast) ** | 17.2 ± 0.5 | 13.2 ± 1.4 | 16.3 ± 1.9 | 29.3 ± 2.2 | 24.1 ± 1.7 | |
| BCC (annealed) | 11.7 ± 0.3 | 6.0 ± 0.7 | 13.3 ± 0.2 | 40.6 ± 0.1 | 28.2 ± 0.4 | |
| B2 (as-cast) ** | 32.2 ± 0.7 | 25.9 ± 0.2 | 24.1 ± 0.7 | 4.6 ± 0.4 | 13.1 ± 0.1 | |
| B2 (annealed) | 35.5 ± 1.3 | 20.4 ± 0.9 | 18.2 ± 0.8 | 12.0 ± 2.1 | 13.9 ± 0.8 |
| Alloy | Phase | Estimated (2D) Phase Fraction (%) As-Cast | Estimated (2D) Phase Fraction (%) Annealed | Phase Fraction (%) Thermo-Calc™ (at 1050 °C) |
|---|---|---|---|---|
| Al8 | FCC | 98 | 89 | 88 |
| BCC | 0.76 | --- | --- | |
| B2 | 1.24 | 11 | 12 | |
| Al15 | FCC | 54 | 59 | 53 |
| BCC | 26 | --- | --- | |
| B2 | 20 | 41 | 47 | |
| Al30 | BCC | 39 | 29 | 21 |
| B2 | 61 | 71 | 79 |
| Alloy | Initial Condition | kP (1) (g2/cm4·s) | Duration for kP (1) | kP (2) (g2/cm4·s) | Duration for kP (2) | Primary Oxides |
|---|---|---|---|---|---|---|
| Al8 | As-cast | ~2.5 × 10−11 | 1–30 h | ~2.5 × 10−12 | 30–100 h | Cr2O3/Al2O3 |
| Annealed | ~2.2 × 10−11 | 1–40 h | ~8.9 × 10−12 | 40–80 h | Al2O3/Spinel | |
| Al15 | As-cast | ~4.7 × 10−11 | 2–18 h | ~4.2 × 10−12 | 19–36 h | Cr2O3/Al2O3 |
| Annealed | ~2.4 × 10−11 | 2–20 h | ~7.8 × 10−12 | 20–80 h | Al2O3/Spinel | |
| Al30 | As-cast | ~1.9 × 10−12 | 2–10 h | --- | --- | Cr2O3/Al2O3 |
| Annealed | ~3.3 × 10−12 | 2–10 h | ~9.1 × 10−13 | 10–60 h | Al2O3 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butler, T.M.; Weaver, M.L. Influence of Annealing on the Microstructures and Oxidation Behaviors of Al8(CoCrFeNi)92, Al15(CoCrFeNi)85, and Al30(CoCrFeNi)70 High-Entropy Alloys. Metals 2016, 6, 222. https://doi.org/10.3390/met6090222
Butler TM, Weaver ML. Influence of Annealing on the Microstructures and Oxidation Behaviors of Al8(CoCrFeNi)92, Al15(CoCrFeNi)85, and Al30(CoCrFeNi)70 High-Entropy Alloys. Metals. 2016; 6(9):222. https://doi.org/10.3390/met6090222
Chicago/Turabian StyleButler, Todd M., and Mark L. Weaver. 2016. "Influence of Annealing on the Microstructures and Oxidation Behaviors of Al8(CoCrFeNi)92, Al15(CoCrFeNi)85, and Al30(CoCrFeNi)70 High-Entropy Alloys" Metals 6, no. 9: 222. https://doi.org/10.3390/met6090222
APA StyleButler, T. M., & Weaver, M. L. (2016). Influence of Annealing on the Microstructures and Oxidation Behaviors of Al8(CoCrFeNi)92, Al15(CoCrFeNi)85, and Al30(CoCrFeNi)70 High-Entropy Alloys. Metals, 6(9), 222. https://doi.org/10.3390/met6090222






